Abstract
The stomach is a digestive organ that has important roles in human physiology and pathophysiology. The developmental origin of the stomach is the embryonic foregut, which also gives rise a number of other structures. There are several signaling pathways and transcription factors that are known to regulate stomach development at different stages, including foregut patterning, stomach specification, and gastric regionalization. These developmental events have important implications in later homeostasis and disease in the adult stomach. Here we will review the literature that has shaped our current understanding of the molecular mechanisms that coordinate gastric organogenesis. Further we will discuss how developmental paradigms have guided recent efforts to differentiate stomach tissue from pluripotent stem cells.
Keywords: endoderm, foregut, stomach, corpus, fundus, antrum
1. INTRODUCTION
The stomach is an evolutionarily diverse structure that has numerous functions including digestion of food, immune defense, and hormonal regulation of metabolic homeostasis. Depending on their unique dietary needs and habits, vertebrates have adapted variations in their structural and histological organization of the stomach [1]. In humans, the gastric mucosa entirely comprises a glandular, columnar epithelium, and it is regionalized into two distinct compartments containing specialized cell types that work in a complementary fashion. The more proximal region of the stomach contains fundic glands that contain acid-secreting parietal cells, enzyme-producing chief cells, and protective mucus-forming cells, whereas the distal region of the stomach, the antrum, consists largely of mucous cells and endocrine cells including G-cells that secrete gastrin. In addition to these, however, mice have a large forestomach in the anterior, which consists of a stratified squamous epithelium similar to that of the esophagus.
Regardless of its ultimate composition, the mechanisms that direct early embryonic development of the stomach appear to be conserved across vertebrate species. While the species-specific differences in architecture can sometimes hamper our ability to directly translate findings from animals to humans, we have learned a great deal about stomach development from model organisms including mouse, chick, frog, and fish. Additionally human pluripotent stem cell (hPSC)-derived endoderm has recently proven to be a valuable in vitro model system for interrogating the dynamic processes of foregut and stomach development. In this review, we will summarize the early developmental mechanisms of stomach development, focusing on the signaling pathways and transcriptional regulators that control endoderm patterning, gastric specification, stomach regionalization, and morphogenesis. While the later control of gastric stem cell behavior and homeostasis likely has parallels to early developmental processes, this literature has been reviewed recently [1–3] and will not be discussed here.
2. SPECIFICATION OF GASTRIC LINEAGE
The embryonic stomach derives from posterior foregut, and it develops amid a number of neighboring tissues including the esophagus, intestine, liver, gallbladder and pancreas. As with development of all endoderm organs, a complex array of epithelial-mesenchymal interactions is responsible for promoting gastric fate. In both rodent and chick embryos, presumptive gastric mesoderm is required to specify a gastric fate in the nearby endoderm, and it can re-specify endoderm taken from non-gastric regions of the gut [4,5]; however, the responding endoderm is only competent to respond to gastric-inducing signals during a short temporal window in early development [6], suggesting that gastric specification is a temporally and spatially dynamic process. The work of several groups has led to the elucidation of signaling pathway and transcription factor networks that govern some of the early stages stomach development.
2.1 Patterning posterior foregut endoderm
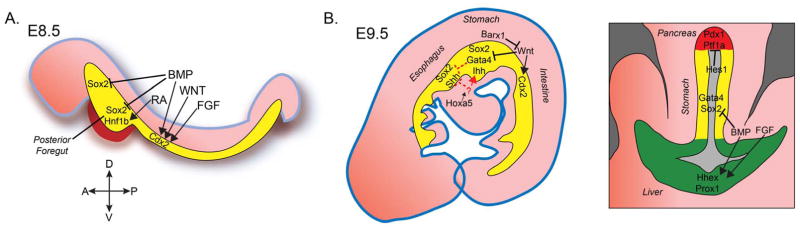
Following gastrulation a series of instructive signals patterns nascent endoderm resulting in a three-dimensional gut tube that is roughly divided into anterior and posterior domains (Reviewed in [7]). The posterior region, or hindgut, expresses the homeodomain transcription factor Cdx2 [8,9], and it gives rise to the small and large intestines. The anterior endoderm is the foregut, which expresses the HMG box transcription factor Sox2 [9]. The signals responsible for this early anterior-posterior patterning have been elucidated in numerous model organisms, and they include WNT, FGF, and BMP [10–13]. Interestingly, all of these pathways have been shown to have a posteriorizing effect on early endoderm. In the nascent foregut, expression of Sox2 is negatively regulated by BMP signaling [14–16]. Moreover, repression of BMP signaling occurs throughout early stages of esophagus and stomach development [17], suggesting that these organ fates require a low-BMP state. Although WNT, FGF, and BMP have direct effects on endoderm [18–20], they only have patterning activity in the presence of mesoderm, suggesting that multiple signals might act synergistically to coordinate endoderm patterning. Indeed, activity of all three pathways is required to differentiate hPSCs into Cdx2-positive hindgut tissue [16,21].
Shortly after its formation, the Sox2-positive foregut tube is segregated into a number of primitive organ domains. The anterior foregut, in the pharyngeal region of the embryo, forms the oropharynx, thyroid, thymus and parathyroid glands. More posterior portions of the foregut differentiate into the epithelial lining of the esophagus and glandular stomach, as well as budding organs including lungs, liver, pancreas, and extrahepatic biliary system. The ability to give rise to such a diversity of organ lineages implies that the foregut itself might be patterned into sub-domains that have distinct developmental potentials. However this stage of development is brief and difficult to access experimentally, so there are limited data that address this concept of “foregut patterning.” Evidence to support this model comes via the regionalized expression of several transcription factors that are expressed in the posterior foregut prior to organ specification, including Hnf1β, Hnf6, and Prox1 [9,22–24]. In particular, Hnf1β appears to be a master regulator of posterior foregut development as its deletion leads to aberrant specification of the pancreas, liver, and glandular stomach [24,25].
On key regulator of posterior foregut development is retinoic acid (RA), which has numerous roles in pattern formation in embryonic development (reviewed in [26]). Raldh2, the primary synthetic enzyme in the pathway, is expressed in posterior mesodermal tissues surrounding the distal foregut and hindgut [27], implicating RA as an important foregut patterning molecule. Early RA activity is required for specification of several posterior foregut derivatives including the pancreas [28,29] and lung [15], and it represses the development of anterior foregut lineages such as the thyroid [30]. The glandular hindstomach fails to form in mice lacking Raldh2 [31], providing further evidence for a role for RA in posterior foregut development. Consistent with this, application of exogenous RA is critical for the in vitro differentiation of human gastric tissues from hPSCs [16]. In these models RA promotes expression of HNF1β, suggesting that a RA-Hnf1β regulatory network might control early foregut patterning to set up competence for specification into posterior foregut lineages.
Aside from the few studies described above, there is a paucity of information known regarding the molecular pathways that govern formation of the posterior foregut. Reduction in Activin receptor signaling leads to similar gastric phenotypes as observed in Raldh2 mutants [32], but the TGFβ pathway has not been more thoroughly investigated for its role in stomach development. With there being high interest in generating fully functional foregut organ tissues from hPSCs, it is essential to better delineate the mechanisms that segregate early foregut endoderm into its derivatives.
2.2 Defining the anterior gastric boundary
After initial patterning, the posterior foregut is further divided into distinct organ lineages. Again, the molecular mechanisms that precisely drive these events are poorly understood. Instead, much of our knowledge is derived from studies focusing on the establishment of organ-organ boundaries, i.e. binary fate decisions. The presumptive gastric epithelium forms adjacent to the esophagus, small intestine, pancreas, and liver; thus, stomach specification in the posterior foregut involves processes that exclude or repress each of these other organ fates.
The process of establishing the gastro-esophageal boundary is likely to be different in mice than it is in humans because of the divergent anatomy between mammalian species. Because the mouse has a forestomach that resembles the esophagus, the gastric columnar epithelium only begins at the convergence of the forestomach and hindstomach. In contrast, the squamo-columnar junction in humans occurs at the organ boundary between the stomach and esophagus. At the molecular level, it seems that gene expression boundaries follow the squamo-columnar junction pattern rather in mice rather than the gastro-esophageal junction. Moreover, the embryologic precursors of the esophagus and forestomach display similar developmental potential. For example, ectopic activation of β-catenin is sufficient to convert presumptive esophageal progenitors into the Nkx2-1-positive respiratory lineage, and it has a similar effect on anterior but not posterior stomach epithelium [33,34]. Therefore, it seems most likely that the forestomach-hindstomach boundary in rodent embryos is analogous to the gastro-esophageal boundary in human development.
Hedgehog pathway ligands are generally expressed by the endodermal lining of the developing gut tube and they primarily signal to the adjacent mesodermal tissue. Most of the early endoderm in mice expresses Sonic hedgehog (Shh), but the presumptive glandular hindstomach epithelium specifically expresses Indian hedgehog (Ihh; [35]). Thus, there is a Shh-Ihh expression boundary at the junction of the forestomach and hindstomach. There are two lines of evidence that the gastric mesenchyme plays an essential role in establishing this junction. First, deletion of the mesenchymal factor Hoxa5 disrupts the normal Shh-Ihh expression pattern leading to defective hindstomach differentiation [36]. Second, deletion of Shh, which is believed to only signal directly to the mesenchyme [37], also disrupts the forestomach-hindstomach boundary. In both Hoxa5 and Shh mutants [38], the hindstomach also adopts some intestinal features. These data suggest a model whereby Hoxa5, acting through an unknown paracrine factor, regulates epithelial Hh ligand expression, which in turn regulate mesenchymal growth factors that further reinforce the forestomach-hindstomach junction. Thus, the extrinsic signaling mechanisms that direct epithelial cell fate decisions at the anterior gastric boundary remain largely unknown.
A gastric-specific transcriptional “master regulator” has not yet been identified during embryonic stages. However there are several broadly expressed factors that contribute to establishing the esophageal-gastric boundary. The zinc finger transcription factor Gata4 is robustly expressed in the hindstomach and completely excluded from the forestomach. Gata4-null cells in chimeric embryos do not contribute to the glandular epithelium of the stomach, and deletion studies demonstrated that Gata4 interactions with its transcriptional co-activators FOG1/2 are required for normal gastric development [39,40]. Further, deletion of Gata4 from the already-specified distal stomach causes cell-autonomous migration of cells from the hindstomach into the forestomach [41]. A related factor, Gata6, has a similar localization pattern [42] but has not been investigated for a role in stomach development. Thus, Gata4 remains one of the very few genes known to be required for stomach specification. The early foregut gene Sox2 remains expressed in both the esophagus and stomach throughout much of development. While it is required for proper differentiation of the squamous epithelia of the foregut [43], glandular stomach differentiation appears unperturbed in Sox2 hypomorphs. Though it would be an important finding that Sox2 is dispensable for gastric development, complete loss-of function analyses have not yet been performed.
2.3 Defining the posterior gastric boundary
The gastro-intestinal junction is established as early as E8.5 in the mouse embryo, as it is identified as the Sox2-Cdx2 expression boundary. Although a small number of cells co-express these factors in the early endoderm [9], the border is sharpened and refined over developmental time [44]. These transcription factors are important regulators of foregut and hindgut fates, respectively. Ectopic activation of Sox2 in the hindgut suppresses the intestinal phenotype and induces features of the glandular stomach, but it does not repress Cdx2 itself [45]. Similarly, forced expression of Cdx2 in previously specified developing gastric epithelium is sufficient to induce intestinal metaplasia [46] but not down-regulation of Sox2 [47]. These findings that Sox2 and Cdx2 are not mutually repressive are interesting, given that they have dominant effects on gastric and intestinal differentiation. Moreover, it is also fascinating that Cdx2 loss-of-function has different effects depending on the timing of deletion. Early loss of Cdx2 (via Foxa3-Cre) leads to impaired intestinal differentiation, but the intestine adopts an esophageal, rather than gastric, phenotype [48]. In this model, the gastric boundary is not impacted. Conversely, later deletion of Cdx2 with Villin-CreER produces pyloric-like gastric glands in the intestine [49]. Taken together, these studies have revealed that Sox2 and Cdx2 have key functions in establishing the gastro-intestinal boundary and maintaining organ fate. However, the findings also suggest that there must be unidentified factors that contribute to determining gastric and intestinal fates.
Early formation of the gastro-intestinal boundary is orchestrated by a mesenchymal-epithelial network that was elucidated in a key series of papers on the transcription factor Barx1 [50,51]. This factor is specifically expressed in the presumptive stomach mesenchyme, and it promotes expression of secreted WNT signaling antagonists (sFRPs) in this region. This Barx1-mediated WNT inhibition is required to suppress intestinal transformation in the presumptive gastric epithelium. While WNT/β-catenin activation is able promote hindgut gene expression in several model systems including frog, mouse, and human endoderm [10,19,21], ectopic induction of the Barx1-sFRP cassette in hindgut mesenchyme does not impact differentiation of the intestinal epithelium [52]. This discrepancy could be explained by either timing of WNT signaling repression or the existence of other signaling pathways that regulate this fate decision. One example is the BMP signaling pathway, which in human endoderm models in vitro is necessary for intestinal specification, and it must be inactive during early foregut development [16]. Along similar lines, BMP is sufficient to repress Sox2 expression in the foregut [14], and BMP activity is accordingly low in the embryonic stomach and esophagus. Taken together, these studies indicate that multiple signaling pathways, including at least WNT and BMP, act in combination to establish organ boundaries at the foregut-hindgut junction.
2.4 Gene expression boundaries between stomach and accessory organs
In addition to the competing lineages along the longitudinal axis of the alimentary canal, early gastric progenitors must also be specified apart from the budding organs of the posterior foregut – the pancreas and liver. Again, there are transcription factor boundaries that distinguish between these early organs domains. While the pancreatic factor Pdx1 is also expressed in the distal stomach and proximal intestine epithelia, the bHLH factor Ptf1a is highly specific to the pancreas at E9.5 [53,54]. As an example of the early plasticity of posterior foregut endoderm, ectopic Ptf1a expression is sufficient to induce pancreatic fate in gastric progenitors [55,56]. Deletion of Hes1, another bHLH factor, also leads to ectopic pancreatic differentiation along the greater curvature of the stomach [57], suggesting that Hes1 represses pancreatic fate in the presumptive stomach. Although Hes1 often functions downstream of Notch signaling, gastric specification defects are not observed in other Notch pathway mutants [58]. Therefore Hes1 might act independent of Notch, but neither its function nor that of the Notch pathway has been further explored in the developing stomach.
As described previously, the stomach and liver develop adjacent to each other and express a number of common markers, such as Hnf1β. Despite their proximity in the foregut, little is known regarding the mechanisms that segregate early gastric and hepatic lineages, except that BMP and FGF activation promote liver specification [59,60]. Thus absence of these factors is thus likely permissive for stomach development, again emphasizes the inhibitory role of BMP in stomach specification. One interesting observation is that while the lateral (presumptive gastric) foregut endoderm at E9.5 expresses Gata4, the budding hepatic diverticulum is Gata4-negative [61]. These data again imply a potentially important role for Gata4 in foregut patterning and lineage segregation, but early endoderm-specific knockouts have not yet been described.
The key signaling pathways and transcription factors required for posterior foregut patterning and gastric specification are summarized in Figure 1.
Figure 1. Schematic overview of developmental mechanisms leading to stomach specification.
A. Patterning of posterior foregut endoderm. During endoderm patterning, which occurs around E8.5 in the mouse, WNT, Bmp, and Fgf signals promote hindgut (Cdx2) gene expression. BMP represses the foregut marker Sox2. RA activity is required for specification of the posterior region of the foregut, where one of its target genes is Hnf1β. B. Sagittal and transverse representations of lineage restriction and gastric specification in the mouse.
3. PATTERNING THE EMBRYONIC STOMACH
Once specified, gastric progenitors are further divided along the rostro-caudal axis. The proximal region forms the epithelium of the corpus and fundus (and additionally the forestomach in mice), while the distal portion develops into the antral stomach. During embryonic stages, one of the few known molecular differences between these gastric domains is Pdx1, which is specifically localized to the presumptive antral region at E10.5 [62]. Surprisingly, the role for Pdx1 in stomach patterning has not been investigated, outside of its known requirement for the differentiation of antral G-cells at later stages [63]. Pdx1 mutant embryos do not exhibit gross or obvious defects in stomach morphology [64], but it would be interesting to see whether this transcription factor impacts gastric differentiation more generally. The transcription factor Nkx6.3 is also expressed in an antrum-specific fashion, but seems to only be required for G-cell specification within the antrum [65]. In order to more fully understand the mechanisms of stomach pattern formation, it is imperative to identify additional genes that exhibit region-specific expression profiles.
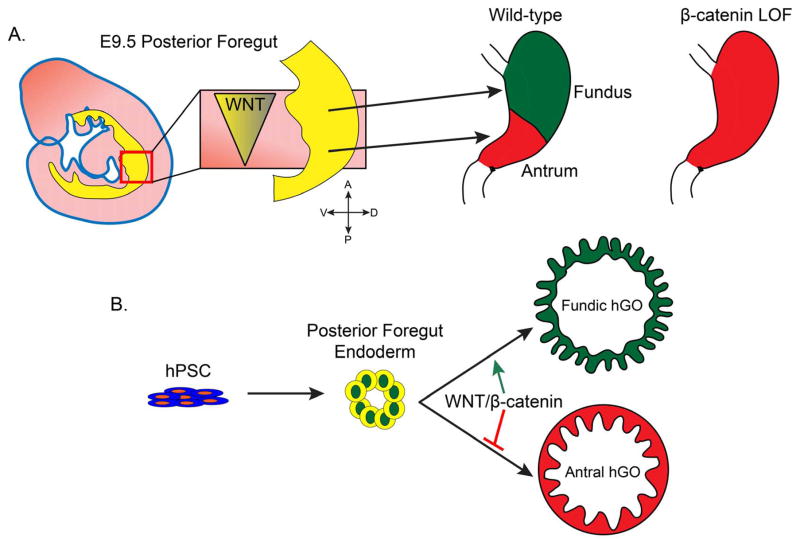
Predictably, mesenchymal-epithelial interactions appear to play an important role in gastric patterning. Bapx1 is a transcription factor expressed in mesenchyme surrounding the distal stomach and proximal intestine [66]. When it is deleted, the antral segment of the stomach is reduced and parietal cells are found near the gastro-intestinal junction. The signaling mechanism underlying this phenomenon has not been identified in rodents. However in the avian stomach, Bapx1 represses the expression of Wnt5a and Bmp4 [67], suggesting that WNT and/or BMP signaling may play a role in stomach patterning. It has been observed that canonical WNT activity is restricted to the rostral embryonic stomach and excluded from the presumptive antrum [50,68,69]. Following up this observation, it was shown that deletion of the canonical WNT effector Ctnnb1 in the epithelium led to ectopic Pdx1 expression and subsequent antralization of the proximal stomach. In these mice, loss of Ctnnb1 resulted in disruption of fundic-type cytodifferentiation in a cell autonomous manner. These data support a model in which WNT/β-catenin signaling can toggle between cell fates in the developing glandular stomach. High WNT activity is required for differentiation of the corpus/fundus region, while suppression of the pathway permits specification of the antral domain (summarized in Figure 2). Deletion in these animals largely occurred prior to gastric patterning, so it is yet unclear whether WNT/β-catenin manipulation can impact gastric fate after initial patterning has already occurred. This principle was further applied to modulate cell fate during the in vitro differentiation of PSCs into human gastric organoids (hGOs). In the absence of WNT signaling hGOs contain Pdx1-positive antral epithelium, but early WNT stimulation induces fundic-type hGOs that contain parietal and chief cells [70].
Figure 2. Summary of WNT-mediated gastric patterning mechanisms in vivo and in vitro.
A. WNT/β-catenin signaling promotes fundus specification from the pool of gastric progenitor cells during E9–10. B. WNT activation promotes generation of fundic human gastric organoids (hGOs), while repressing antral specification, from hPSC-derived posterior foregut spheroids.
The newer data that support a role for WNT/β-catenin signaling in formation of the corpus/fundus appear incompatible with previous models in which WNT signaling must be repressed during stomach development. There are several potential explanations that could resolve this discrepancy. Time is an important variable in embryonic development, and it is thus possible that early WNT promotes intestinal specification while a later WNT signal regulates gastric patterning. Further, the epithelial response to WNT could be modulated by activity of other pathways such as BMP, as described above. Regardless, more work is necessary to address these questions and identify how gastric fate is determined.
4. MORPHOGENESIS OF THE GASTRIC EPITHELIUM
The early foregut epithelium is a simple cuboidal lining that lacks architectural complexity. Following gastric specification and patterning, the mucosa transitions through a pseudostratified stage prior to adopting the tall columnar morphology that is found in the adult organ, similar to events observed in the intestine [71]. Accompanying these cell shape changes, the epithelium undergoes folding and glandular morphogenesis that progressively develops through postnatal stages, resulting in the highly organized, complex structure found in the mature stomach. The precise mechanisms that coordinate these morphogenetic events are not fully understood, but several embryonic signaling molecules are known to play a role.
Similar to its role in regulating cell fate decisions, the gastric mesenchyme is essential for proper growth and morphogenesis of the stomach. Epithelial-derived Hh ligands sustain mesenchymal cell survival and proliferation, and simultaneous deletion of Shh and Ihh thus lead to severely stunted stomach and intestinal growth [72]. Despite their aberrant morphogenesis, proper gastric specification and patterning are maintained in these mutants. Downstream of the epithelial Hh signal, FoxF and FoxL transcription factors mediate a complex signaling network in the associated mesenchyme [73,74]. An important mesenchyme-derived growth factor is Fgf10, which signals to the epithelium through the Fgfr2 receptor throughout the later stages of development. Loss of either molecule results in reduced proliferation and defective glandular morphogenesis in the gastric epithelium [75,76]. Conversely, overexpression of Fgf10 leads to glandular hyperplasia and impaired cytodifferentiation [77]. Gland formation in the avian proventriculus is also regulated by BMP, WNT5a, Notch, and Shh [78–81]. In mouse, it was shown that Wnt5a activates the WNT/PCP pathway, which is essential for maintaining epithelial cell polarity, morphogenesis, and growth and elongation of the anterior portion of the stomach [69]. Further, canonical WNT signaling contributes to stomach growth as epithelial deletion of Ctnnb1 leads to a dramatic reduction in stomach size [70], albeit downstream of yet unidentified WNT ligands. Finally, EGF is another important epithelial mitogen required for normal proliferation and growth of the rodent mucosa [82,83], and ectopic EGF is required for in vitro differentiation of embryonic gastric structures [16].
5. SUMMARY
Overall, a number of developmental signaling pathways are used reiteratively during stomach development to regulate endoderm patterning, gastric specification, stomach regionalization, and morphogenesis. Included in this group are WNT, Bmp, Fgf, and RA, and together they precisely coordinate expression of transcription factors that determine cell identity. Our current understanding of the transcriptional regulators of stomach development is lacking, and it is imperative for future work to further elucidate the signaling paradigms underlying this process. Nevertheless, use of known embryologic mechanisms, as well as newly discovered principles, has recently led development of protocols for the de novo differentiation of gastric tissue from PSCs [16,70,84]. These systems, in turn, should prove to be effective models for studying gastric development and disease. For example, there are currently no available data on the epigenetic control of stomach development, and in vitro tissues are well suited for epigenetic studies.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Kim T-H, Shivdasani RA. Stomach development, stem cells and disease. Development. 2016;143:554–65. doi: 10.1242/dev.124891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mills JC, Shivdasani RA. Gastric epithelial stem cells. Gastroenterology. 2011;140:412–24. doi: 10.1053/j.gastro.2010.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Han M-E, Oh S-O. Gastric stem cells and gastric cancer stem cells. Anat Cell Biol. 2013;46:8–18. doi: 10.5115/acb.2013.46.1.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fukamachi H, Mizuno T, Takayama S. Epithelial-mesenchymal interactions in differentiation of stomach epithelium in fetal mice. Anat Embryol. 1979;157:151–60. doi: 10.1007/BF00305155. [DOI] [PubMed] [Google Scholar]
- 5.Koike T, Yasugi S. In vitro analysis of mesenchymal influences on the differentiation of stomach epithelial cells of the chicken embryo. Differentiation. 1999;65:13–25. doi: 10.1046/j.1432-0436.1999.6510013.x. [DOI] [PubMed] [Google Scholar]
- 6.Duluc I, Freund JN, Leberquier C, Kedinger M. Fetal endoderm primarily holds the temporal and positional information required for mammalian intestinal development. J Cell Biol. 1994;126:211–21. doi: 10.1083/jcb.126.1.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zorn AM, Wells JM. Vertebrate Endoderm Development and Organ Formation. Annu Rev Cell Dev Biol. 2009;25:221–51. doi: 10.1146/annurev.cellbio.042308.113344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Silberg DG, Swain GP, Suh ER, Traber PG. Cdx1 and cdx2 expression during intestinal development. Gastroenterology. 2000;119:961–71. doi: 10.1053/gast.2000.18142. [DOI] [PubMed] [Google Scholar]
- 9.Sherwood RI, Chen T-YA, Melton DA. Transcriptional dynamics of endodermal organ formation. 2009;238:29–42. doi: 10.1002/dvdy.21810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McLin VA, Rankin SA, Zorn AM. Repression of WNT/beta-catenin signaling in the anterior endoderm is essential for liver and pancreas development. Development. 2007;134:2207–17. doi: 10.1242/dev.001230. [DOI] [PubMed] [Google Scholar]
- 11.Wells JM, Melton DA. Early mouse endoderm is patterned by soluble factors from adjacent germ layers. Development. 2000;127:1563–72. doi: 10.1242/dev.127.8.1563. [DOI] [PubMed] [Google Scholar]
- 12.Tiso N, Filippi A, Pauls S, Bortolussi M, Argenton F. BMP signalling regulates anteroposterior endoderm patterning in zebrafish. Mechanisms of Development. 2002;118:29–37. doi: 10.1016/S0925-4773(02)00252-6. [DOI] [PubMed] [Google Scholar]
- 13.Kumar M, Jordan N, Melton D, Grapin-Botton A. Signals from lateral plate mesoderm instruct endoderm toward a pancreatic fate. Developmental Biology. 2003;259:109–22. doi: 10.1016/S0012-1606(03)00183-0. [DOI] [PubMed] [Google Scholar]
- 14.Domyan ET, Ferretti E, Throckmorton K, Mishina Y, Nicolis SK, Sun X. Signaling through BMP receptors promotes respiratory identity in the foregut via repression of Sox2. Development. 2011;138:971–81. doi: 10.1242/dev.053694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rankin SA, Han L, McCracken KW, Kenny AP, Anglin CT, Grigg EA, et al. A Retinoic Acid-Hedgehog Cascade Coordinates Mesoderm-Inducing Signals and Endoderm Competence during Lung Specification. Cell Reports. 2016;16:66–78. doi: 10.1016/j.celrep.2016.05.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.McCracken KW, Catá EM, Crawford CM, Sinagoga KL, Schumacher M, Rockich BE, et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature. 2014;516:400–4. doi: 10.1038/nature13863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rodriguez P, Da Silva S, Oxburgh L, Wang F, Hogan BLM, Que J. BMP signaling in the development of the mouse esophagus and forestomach. Development. 2010;137:4171–6. doi: 10.1242/dev.056077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dessimoz J, Opoka R, Kordich JJ, Grapin-Botton A, Wells JM. FGF signaling is necessary for establishing gut tube domains along the anterior–posterior axis in vivo. Mechanisms of Development. 2006;123:42–55. doi: 10.1016/j.mod.2005.10.001. [DOI] [PubMed] [Google Scholar]
- 19.Sherwood RI, Maehr R, Mazzoni EO, Melton DA. WNT signaling specifies and patterns intestinal endoderm. Mechanisms of Development. 2011;128:387–400. doi: 10.1016/j.mod.2011.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rankin SA, Kormish J, Kofron M, Jegga A, Zorn AM. A gene regulatory network controlling hhex transcription in the anterior endoderm of the organizer. Developmental Biology. 2011;351:297–310. doi: 10.1016/j.ydbio.2010.11.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Spence JR, Mayhew CN, Rankin SA, Kuhar MF, Vallance JE, Tolle K, et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature. 2011;470:105–9. doi: 10.1038/nature09691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jacquemin P, Yoshitomi H, Kashima Y, Rousseau GG, Lemaigre FP, Zaret KS. An endothelial-mesenchymal relay pathway regulates early phases of pancreas development. Developmental Biology. 2006;290:189–99. doi: 10.1016/j.ydbio.2005.11.023. [DOI] [PubMed] [Google Scholar]
- 23.Burke Z, Oliver G. Prox1 is an early specific marker for the developing liver and pancreas in the mammalian foregut endoderm. Mechanisms of Development. 2002;118:147–55. doi: 10.1016/S0925-4773(02)00240-X. [DOI] [PubMed] [Google Scholar]
- 24.Haumaitre C, Barbacci E, Jenny M, Ott MO, Gradwohl G, Cereghini S. Lack of TCF2/vHNF1 in mice leads to pancreas agenesis. Proceedings of the National Academy of Sciences. 2005;102:1490–5. doi: 10.1073/pnas.0405776102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lokmane L, Haumaitre C, Garcia-Villalba P, Anselme I, Schneider-Maunoury S, Cereghini S. Crucial role of vHNF1 in vertebrate hepatic specification. Development. 2008;135:2777–86. doi: 10.1242/dev.023010. [DOI] [PubMed] [Google Scholar]
- 26.Duester G. Retinoic Acid Synthesis and Signaling during Early Organogenesis. Cell. 2008;134:921–31. doi: 10.1016/j.cell.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Niederreither K. The regional pattern of retinoic acid synthesis by RALDH2 is essential for the development of posterior pharyngeal arches and the enteric nervous system. Development. 2003;130:2525–34. doi: 10.1242/dev.00463. [DOI] [PubMed] [Google Scholar]
- 28.Molotkov A, Molotkova N, Duester G. Retinoic acid generated by Raldh2 in mesoderm is required for mouse dorsal endodermal pancreas development. Dev Dyn. 2005;232:950–7. doi: 10.1002/dvdy.20256. [DOI] [PubMed] [Google Scholar]
- 29.Martín M, Gallego-Llamas J, Ribes V, Kedinger M, Niederreither K, Chambon P, et al. Dorsal pancreas agenesis in retinoic acid-deficient Raldh2 mutant mice. Developmental Biology. 2005;284:399–411. doi: 10.1016/j.ydbio.2005.05.035. [DOI] [PubMed] [Google Scholar]
- 30.Wang JH, Deimling SJ, D’Alessandro NE, Zhao L, Possmayer F, Drysdale TA. Retinoic acid is a key regulatory switch determining the difference between lung and thyroid fates in Xenopus laevis. BMC Developmental Biology. 2011;11:75. doi: 10.1186/1471-213X-11-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang Z, Dollé P, Cardoso WV, Niederreither K. Retinoic acid regulates morphogenesis and patterning of posterior foregut derivatives. Developmental Biology. 2006;297:433–45. doi: 10.1016/j.ydbio.2006.05.019. [DOI] [PubMed] [Google Scholar]
- 32.Kim SK, Hebrok M, Li E, Oh SP, Schrewe H, Harmon EB, et al. Activin receptor patterning of foregut organogenesis. Genes & Development. 2000;14:1866–71. doi: 10.1101/gad.14.15.1866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Goss AM, Tian Y, Tsukiyama T, Cohen ED, Zhou D, Lu MM, et al. WNT2/2b and beta-catenin signaling are necessary and sufficient to specify lung progenitors in the foregut. Developmental Cell. 2009;17:290–8. doi: 10.1016/j.devcel.2009.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Harris-Johnson KS, Domyan ET, Vezina CM, Sun X. beta-Catenin promotes respiratory progenitor identity in mouse foregut. Proc Natl Acad Sci USa. 2009;106:16287–92. doi: 10.1073/pnas.0902274106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bitgood MJ, McMahon AP. Hedgehog and Bmp genes are co-expressed at many diverse sites of cell-cell interaction in the mouse embryo. Developmental Biology. 1995;172:126–38. doi: 10.1006/dbio.1995.0010. [DOI] [PubMed] [Google Scholar]
- 36.Aubin J, Déry U, Lemieux M, Chailler P, Jeannotte L. Stomach regional specification requires Hoxa5-driven mesenchymal-epithelial signaling. Development. 2002;129:4075–87. doi: 10.1242/dev.129.17.4075. [DOI] [PubMed] [Google Scholar]
- 37.Kolterud Å, Grosse AS, Zacharias WJ, Walton KD, Kretovich KE, Madison BB, et al. Paracrine Hedgehog Signaling in Stomach and Intestine: New Roles for Hedgehog in Gastrointestinal Patterning. Gastroenterology. 2009;137:618–28. doi: 10.1053/j.gastro.2009.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ramalho-Santos M, Melton DA, McMahon AP. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development. 2000;127:2763–72. doi: 10.1242/dev.127.12.2763. [DOI] [PubMed] [Google Scholar]
- 39.Jacobsen CM, Narita N, Bielinska M, Syder AJ, Gordon JI, Wilson DB. Genetic mosaic analysis reveals that GATA-4 is required for proper differentiation of mouse gastric epithelium. Developmental Biology. 2002;241:34–46. doi: 10.1006/dbio.2001.0424. [DOI] [PubMed] [Google Scholar]
- 40.Jacobsen CM, Mannisto S, Porter-Tinge S, Genova E, Parviainen H, Heikinheimo M, et al. GATA-4:FOG interactions regulate gastric epithelial development in the mouse. Dev Dyn. 2005;234:355–62. doi: 10.1002/dvdy.20552. [DOI] [PubMed] [Google Scholar]
- 41.Xuan S, Borok MJ, Decker KJ, Battle MA, Duncan SA, Hale MA, et al. Pancreas-specific deletion of mouse Gata4 and Gata6 causes pancreatic agenesis. J Clin Invest. 2012;122:3516–28. doi: 10.1172/JCI63352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Freyer L, Schröter C, Saiz N, Schrode N, Nowotschin S, Martinez-Arias A, et al. A loss-of-function and H2B-Venus transcriptional reporter allele for Gata6 in mice. BMC Developmental Biology. 2015;15:38. doi: 10.1186/s12861-015-0086-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Que J, Okubo T, Goldenring JR, Nam KT, Kurotani R, Morrisey EE, et al. Multiple dose-dependent roles for Sox2 in the patterning and differentiation of anterior foregut endoderm. Development. 2007;134:2521–31. doi: 10.1242/dev.003855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li X, Udager AM, Hu C, Qiao XT, Richards N, Gumucio DL. Dynamic patterning at the pylorus: formation of an epithelial intestine-stomach boundary in late fetal life. Dev Dyn. 2009;238:3205–17. doi: 10.1002/dvdy.22134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Raghoebir L, Bakker ERM, Mills JC, Swagemakers S, Kempen MB-V, Munck AB-D, et al. SOX2 redirects the developmental fate of the intestinal epithelium toward a premature gastric phenotype. J Mol Cell Biol. 2012;4:377–85. doi: 10.1093/jmcb/mjs030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Silberg DG, Sullivan J, Kang E, Swain GP, Moffett J, Sund NJ, et al. Cdx2 ectopic expression induces gastric intestinal metaplasia in transgenic mice. Gastroenterology. 2002;122:689–96. doi: 10.1053/gast.2002.31902. [DOI] [PubMed] [Google Scholar]
- 47.Mutoh H, Sashikawa M, Sugano K. Sox2 expression is maintained while gastric phenotype is completely lost in Cdx2-induced intestinal metaplastic mucosa. Differentiation. 2011;81:92–8. doi: 10.1016/j.diff.2010.10.002. [DOI] [PubMed] [Google Scholar]
- 48.Gao N, White P, Kaestner KH. Establishment of intestinal identity and epithelial-mesenchymal signaling by Cdx2. Developmental Cell. 2009;16:588–99. doi: 10.1016/j.devcel.2009.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Grainger S, Savory JGA, Lohnes D. Cdx2 regulates patterning of the intestinal epithelium. Developmental Biology. 2010;339:155–65. doi: 10.1016/j.ydbio.2009.12.025. [DOI] [PubMed] [Google Scholar]
- 50.Kim B-M, Buchner G, Miletich I, Sharpe PT, Shivdasani RA. The stomach mesenchymal transcription factor Barx1 specifies gastric epithelial identity through inhibition of transient WNT signaling. Developmental Cell. 2005;8:611–22. doi: 10.1016/j.devcel.2005.01.015. [DOI] [PubMed] [Google Scholar]
- 51.Kim B-M, Woo J, Kanellopoulou C, Shivdasani RA. Regulation of mouse stomach development and Barx1 expression by specific microRNAs. Development. 2011;138:1081–6. doi: 10.1242/dev.056317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jayewickreme CD, Shivdasani RA. Control of stomach smooth muscle development and intestinal rotation by transcription factor BARX1. Developmental Biology. 2015;405:21–32. doi: 10.1016/j.ydbio.2015.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Krapp A, Knofler M, Ledermann B, Burki K, Berney C, Zoerkler N, et al. The bHLH protein PTF1-p48 is essential for the formation of the exocrine and the correct spatial organization of the endocrine pancreas. Genes & Development. 1998;12:3752–63. doi: 10.1101/gad.12.23.3752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Burlison JS, Long Q, Fujitani Y, Wright CVE, Magnuson MA. Pdx-1 and Ptf1a concurrently determine fate specification of pancreatic multipotent progenitor cells. Developmental Biology. 2008;316:74–86. doi: 10.1016/j.ydbio.2008.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jarikji ZH, Vanamala S, Beck CW, Wright CVE, Leach SD, Horb ME. Differential ability of Ptf1a and Ptf1a-VP16 to convert stomach, duodenum and liver to pancreas. Developmental Biology. 2007;304:786–99. doi: 10.1016/j.ydbio.2007.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Willet SG, Hale MA, Grapin-Botton A, Magnuson MA, MacDonald RJ, Wright CVE. Dominant and context-specific control of endodermal organ allocation by Ptf1a. Development. 2014;141:4385–94. doi: 10.1242/dev.114165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Fukuda A, Kawaguchi Y, Furuyama K, Kodama S, Horiguchi M, Kuhara T, et al. Ectopic pancreas formation in Hes1-knockout mice reveals plasticity of endodermal progenitors of the gut, bile duct, and pancreas. J Clin Invest. 2006;116:1484–93. doi: 10.1172/JCI27704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Apelqvist A, Li H, Sommer L, Beatus P, Anderson DJ, Honjo T, et al. Notch signalling controls pancreatic cell differentiation. Nature. 1999;400:877–81. doi: 10.1038/23716. [DOI] [PubMed] [Google Scholar]
- 59.Rossi JM, Dunn NR, Hogan BL, Zaret KS. Distinct mesodermal signals, including BMPs from the septum transversum mesenchyme, are required in combination for hepatogenesis from the endoderm. Genes & Development. 2001;15:1998–2009. doi: 10.1101/gad.904601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Deutsch G, Jung J, Zheng M, Lóra J, Zaret KS. A bipotential precursor population for pancreas and liver within the embryonic endoderm. Development. 2001;128:871–81. doi: 10.1242/dev.128.6.871. [DOI] [PubMed] [Google Scholar]
- 61.Watt AJ, Zhao R, Li J, Duncan SA. Development of the mammalian liver and ventral pancreas is dependent on GATA4. BMC Developmental Biology. 2007;7:37–11. doi: 10.1186/1471-213X-7-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Fukuda A, Kawaguchi Y, Furuyama K, Kodama S, Kuhara T, Horiguchi M, et al. Loss of the Major Duodenal Papilla Results in Brown Pigment Biliary Stone Formation in Pdx1 Null Mice. Gastroenterology. 2006;130:855–67. doi: 10.1053/j.gastro.2005.11.031. [DOI] [PubMed] [Google Scholar]
- 63.Larsson L-I, Madsen OD, Serup P, Jonsson J, Edlund H. Pancreatic-duodenal homeobox 1-role in gastric endocrine patterning. Mechanisms of Development. 1996;60:175–84. doi: 10.1016/S0925-4773(96)00609-0. [DOI] [PubMed] [Google Scholar]
- 64.Fujitani Y, Fujitani S, Boyer DF, Gannon M, Kawaguchi Y, Ray M, et al. Targeted deletion of a cis-regulatory region reveals differential gene dosage requirements for Pdx1 in foregut organ differentiation and pancreas formation. Genes & Development. 2006;20:253–66. doi: 10.1101/gad.1360106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Choi MY, Romer AI, Wang Y, Wu MP, Ito S, Leiter AB, et al. Requirement of the tissue-restricted homeodomain transcription factor Nkx6. 3 in differentiation of gastrin-producing G cells in the stomach antrum. Molecular and Cellular Biology. 2008;28:3208–18. doi: 10.1128/MCB.01737-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Verzi MP, Stanfel MN, Moses KA, Kim B-M, Zhang Y, Schwartz RJ, et al. Role of the homeodomain transcription factor Bapx1 in mouse distal stomach development. Gastroenterology. 2009;136:1701–10. doi: 10.1053/j.gastro.2009.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nielsen C, Murtaugh LC, Chyung JC, Lassar A, Roberts DJ. Gizzard Formation and the Role of Bapx1. Developmental Biology. 2001;231:164–74. doi: 10.1006/dbio.2000.0151. [DOI] [PubMed] [Google Scholar]
- 68.Cervantes S, Yamaguchi TP, Hebrok M. WNT5a is essential for intestinal elongation in mice. Developmental Biology. 2009;326:285–94. doi: 10.1016/j.ydbio.2008.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Matsuyama M, Aizawa S, Shimono A. Sfrp Controls Apicobasal Polarity and Oriented Cell Division in Developing Gut Epithelium. PLoS Genet. 2009;5:e1000427–15. doi: 10.1371/journal.pgen.1000427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.McCracken KW, Aihara E, Martin B, Crawford CM, Broda T, Treguier J, et al. WNT/β-catenin promotes gastric fundus specification in mice and humans. Nature. 2017;541:182–7. doi: 10.1038/nature21021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Grosse AS, Pressprich MF, Curley LB, Hamilton KL, Margolis B, Hildebrand JD, et al. Cell dynamics in fetal intestinal epithelium: implications for intestinal growth and morphogenesis. Development. 2011;138:4423–32. doi: 10.1242/dev.065789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mao J, Kim B-M, Rajurkar M, Shivdasani RA, McMahon AP. Hedgehog signaling controls mesenchymal growth in the developing mammalian digestive tract. Development. 2010;137:1721–9. doi: 10.1242/dev.044586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ormestad M. Foxf1 and Foxf2 control murine gut development by limiting mesenchymal WNT signaling and promoting extracellular matrix production. Development. 2006;133:833–43. doi: 10.1242/dev.02252. [DOI] [PubMed] [Google Scholar]
- 74.Madison BB, McKenna LB, Dolson D, Epstein DJ, Kaestner KH. FoxF1 and FoxL1 Link Hedgehog Signaling and the Control of Epithelial Proliferation in the Developing Stomach and Intestine. Journal of Biological Chemistry. 2009;284:5936–44. doi: 10.1074/jbc.M808103200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Spencer Dene B, Sala FG, Bellusci S, Gschmeissner S, Stamp G, Dickson C. Stomach development is dependent on fibroblast growth factor 10/fibroblast growth factor receptor 2b-mediated signaling. Gastroenterology. 2006;130:1233–44. doi: 10.1053/j.gastro.2006.02.018. [DOI] [PubMed] [Google Scholar]
- 76.Shin M, Noji S, Neubüser A, Yasugi S. FGF10 is required for cell proliferation and gland formation in the stomach epithelium of the chicken embryo. Developmental Biology. 2006;294:11–23. doi: 10.1016/j.ydbio.2005.12.019. [DOI] [PubMed] [Google Scholar]
- 77.Nyeng P, Norgaard GA, Kobberup S, Jensen J. FGF10 signaling controls stomach morphogenesis. Developmental Biology. 2007;303:295–310. doi: 10.1016/j.ydbio.2006.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Listyorini D, Yasugi S. Expression and function of WNT5a in the development of the glandular stomach in the chicken embryo. Develop Growth Differ. 2006;48:243–52. doi: 10.1111/j.1440-169X.2006.00861.x. [DOI] [PubMed] [Google Scholar]
- 79.Matsuda Y, Wakamatsu Y, Kohyama J, Okano H, Fukuda K, Yasugi S. Notch signaling functions as a binary switch for the determination of glandular and luminal fates of endodermal epithelium during chicken stomach development. Development. 2005;132:2783–93. doi: 10.1242/dev.01853. [DOI] [PubMed] [Google Scholar]
- 80.Narita T, Saitoh K, Kameda T, Kuroiwa A, Mizutani M, Koike C, et al. BMPs are necessary for stomach gland formation in the chicken embryo: a study using virally induced BMP-2 and Noggin expression. Development. 2000;127:981–8. doi: 10.1242/dev.127.5.981. [DOI] [PubMed] [Google Scholar]
- 81.Fukuda K, Kameda T, Saitoh K, Iba H, Yasugi S. Down-regulation of endodermal Shh is required for gland formation in chicken stomach. Mechanisms of Development. 2003;120:801–9. doi: 10.1016/s0925-4773(03)00069-8. [DOI] [PubMed] [Google Scholar]
- 82.Dembinski AB, Johnson LR. Effect of epidermal growth factor on the development of rat gastric mucosa. Endocrinology. 1985;116:90–4. doi: 10.1210/endo-116-1-90. [DOI] [PubMed] [Google Scholar]
- 83.Johnson LR, Guthrie PD. Stimulation of rat oxyntic gland mucosal growth by epidermal growth factor. Am J Physiol. 1980;238:G45–9. doi: 10.1152/ajpgi.1980.238.1.G45. [DOI] [PubMed] [Google Scholar]
- 84.Noguchi T-AK, Ninomiya N, Sekine M, Komazaki S, Wang P-C, Asashima M, et al. Generation of stomach tissue from mouse embryonic stem cells. Nature Cell Biology. 2015;17:984–93. doi: 10.1038/ncb3200. [DOI] [PubMed] [Google Scholar]