Abstract
Polycomb repressive complex 2 (PRC2), a H3K27me3 methyltransferase complex, promotes the development of many organs by silencing ectopic transcription program. However, currently little is known about the role of PRC2 in blood and vascular development. In this study, we interrogated the function of embryonic ectoderm development (EED), a core PRC2 component, in both endothelial and hematopoietic tissues by inactivating a floxed murine EED allele with Tie2Cre, which catalyzes recombination in endothelial and hematopoietic lineages. Murine EEDfl/fl;Tie2Cre (EEDCKO) embryos died at embryonic day (E) 13.5. We did not observe structural abnormalities of blood vessels or cardiac valves, suggesting that EED is dispensable in endothelial cells for initial steps of vascular development. EEDCKO embryos were pale and had abnormal livers. Flow cytometry of fetal liver cells showed that EED depletion significantly impeded erythroid maturation. There was a corresponding increase in myeloid progenitors and granulocytes and macrophages, suggesting an attenuated differentiation path in myeloid lineages. Moreover, EED depletion impaired the generation of hematopoietic stem cells. Collectively, our study demonstrates that within Tie2Cre-recombined embryonic cells, EED is required for proper erythropoiesis and for formation of hematopoietic progenitor and stem cells, but is dispensable for endothelial lineage commitment and early vascular patterning.
Polycomb repressive complex 2 (PRC2) represses gene transcription by trimethylating histone H3 on lysine 27 (H3K27me3). The core PRC2 complex is comprised of embryonic ectoderm development (EED), Suppressor of Zeste12 homolog (SUZ12), retinoblastoma-associated protein 48 (RbAp48), and a catalytic subunit, either Enhancer of Zeste Homolog 1 or 2 (EZH1 or EZH2).1, 2 EZH1 and EZH2 are mutually exclusive in the same PRC2 complex and are partially redundant at certain developmental and disease stages.3, 4 EED is a core component of PRC2 complex that interacts with EZH1 or EZH2 through a WD40 domain. In addition, EED recognizes H3K27me3 through an aromatic cage structure to recruit PRC2 to established H3K27me3 and thereby enforce maintenance of this repressive epigenetic mark.5, 6
PRC2 has essential roles of regulating cell fate commitment, organogenesis, homeostasis and disease-related tissue remodeling.1 In early developing mouse embryos, homozygous Eed null mutation abolished global H3K27 methylation and caused defects in primitive streak formation and fetal lethality at E9.5.7, 8 EZH2 also controls the fate of multiple types of tissue progenitors.9 Depletion of EZH2 in epidermal precursor cells promoted premature epidermal differentiation and barrier acquisition.10, 11 EZH2 also had developmental-stage-specific roles in regulating cardiomyocyte differentiation and proliferation.12, 13 In adult vasculature, EZH2 was required for VEGF-induced silencing of vasohibin1 (VASH1), which promoted angiogenesis in ovarian cancer.14
Both endothelial cell (EC) and hematopoietic cells originate from a common progenitor, the hemangioblast. Hemogenic ECs within the yolk sac give rise to the first population of hematopoietic stem cells (HSCs), which migrate to aorta-gonad mesonephros (AGM) and then to the fetal liver, which then becomes the main site of fetal hematopoiesis. Fetal liver HSCs establish the common myeloid progenitor (CMP) and the common lymphoid progenitor (CLP), multipotent progenitors that differentiate into the major blood lineages. CMPs further differentiate into megakaryocyte–erythrocyte progenitors (MEPs), precursors of erythrocytes and megakaryocytes, and granulocyte–macrophage progenitors (GMPs), precursors of granulocytes and monocyte/macrophages.15, 16, 17
Functional links between PRC2, hematopoiesis, and hematopoietic malignancies have been recently unveiled. Human genetic studies revealed that mutations affecting EZH2, EED, SUZ12, and JARID2, encoding PRC2 subunits and associated proteins, cause hematopoietic diseases including myelodysplastic syndrome, T-cell acute lymphoblastic leukemia, and B cell lymphoma.18, 19, 20, 21 However, the functions of PRC2 in hematopoietic development are currently less clear. Conditional deletion of Ezh2 in hematopoietic and vascular lineages of mouse embryos by Tie2Cre was lethal at mid-gestation due to insufficient expansion of hematopoietic stem/progenitor cells (HSCs).22 In contrast, conditional deletion of EED in HSCs by VavCre did not significantly influence fetal HSC hemostasis but became significant in the adulthood.23 Thus, the role of PRC2 and its component subunits in blood and vessel development require elucidation.
In this study, we used Tie2Cre to inactivate EED in developing blood and vascular lineages. Because there is no functional redundant ortholog of EED, this should abolish PRC2 function in these lineages and circumvent functional redundancy between EZH1 and EZH2. We found that EED is dispensable for EC specification and initial development into vessels and cardiac valve precursors, but is essential for normal population of the fetal liver by HSCs and for erythropoiesis. Combined with previous studies on PRC2, our results illustrated a tissue-context-dependent and developmental-stage-specific role of PRC2 in controlling hematopoiesis.
Results
EED is required in the Tie2Cre lineage for normal embryonic development
EEDflox mice were described previously and develop normally.23 Although heterozygosity for a null Eed allele was previously reported to cause myelo- and lympohoproliferative defects,24 Tie2Cre;EEDfl/wt mice survived normally to adulthood and were fertile (Table 1). However, no live Tie2Cre;EEDfl/fl (EED conditional knockout, abbreviated as EEDCKO) mice were observed at birth (Table 1), indicating that EED is required in the Tie2Cre lineage for normal embryonic development.
Table 1. Survival rate of Tie2Cre;EEDfl/fl knockout mouse.
| Age | EEDfl/wt | EEDfl/fl | TieCre+EEDfl/wt | TieCre+EEDfl/fl |
|---|---|---|---|---|
| P0 | 16 (33%) | 16 (33%) | 16 (33%) | 0 (0%) |
| E13.5 | 16 (24%) | 20 (29%) | 18 (26%) | 14 (4 viable=6%) |
| E11.5 | 30 (30%) | 27 (27%) | 20 (20%) | 23 (23%) |
Table indicates number and (%) of embryos found at each stage.
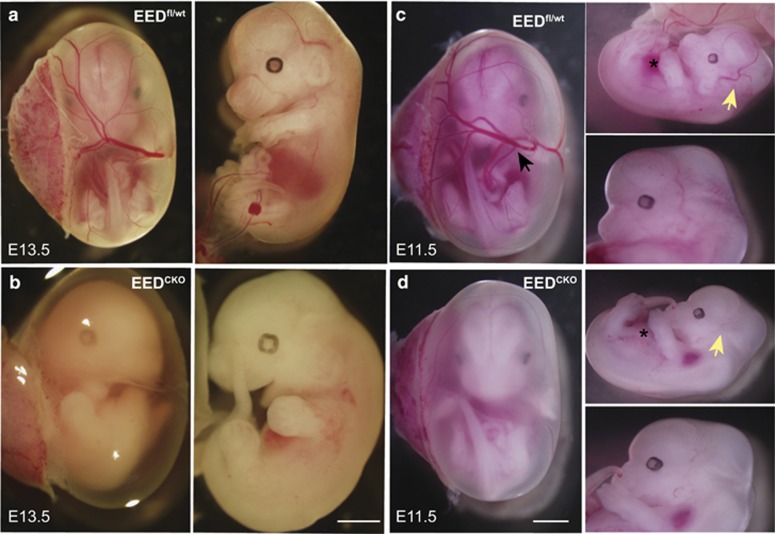
To determine the time of embryonic lethality, we analyzed litters at E11.5 and E13.5. At E11.5, EEDCKO were present at the expected Mendelian frequency, whereas 70% of mutant embryos were no longer viable at E13.5 (Table 1, Figures 1a and b). However, E11.5 EEDCKO embryos already showed developmental abnormalities, as they lacked large blood-perfused vessels including vitelline veins in the yolk sac (Figures 1c and d, black arrow) and primary head veins of the embryo (Figures 1c and d, yellow arrow). The region occupied by the liver was also notably pale (Figures 1c and d, asterisk).
Figure 1.
EEDCKO mouse is embryonic lethal at mid-gestation. (a and b) Images of wild-type and EEDCKO embryos at E13.5. Most EEDCKO embryos had died by this stage. (c and d) Images of wild-type and EEDCKO embryos at E11.5. EEDCKO embryos show significant paleness and absence of blood-perfused vasculature. Asterisks overlie the fetal liver. Black arrow points to a vitelline vessel. Yellow arrow indicates primary head vein. Bar, 0.2 cm (b) or 0.3 cm (d)
EED deletion did not significantly alter the vascular patterning
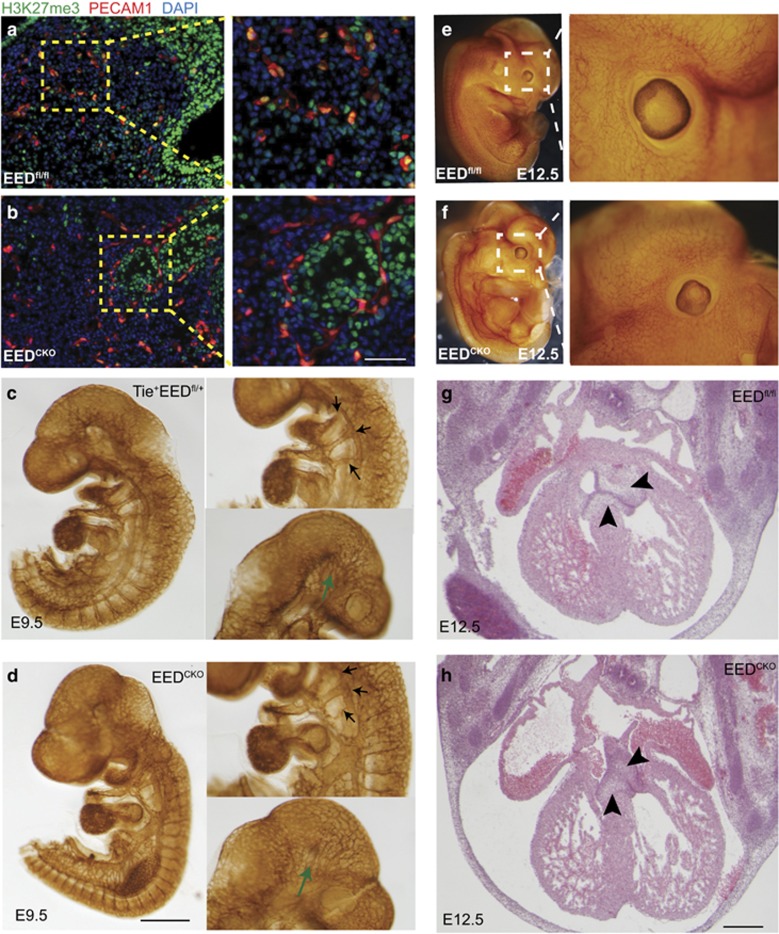
Given that EED is an essential component of PRC2, which deposits H3K27me3, we performed immunostaining to measure bulk H3K27me3 levels. Immunofluorescent staining demonstrated substantially weaker H3K27me3 signal in endothelial cells (Figures 2a and b), consistent with efficient EED deletion in these cells by Tie2Cre. This also suggested the possibility that the observed absence of blood-filled vessels in EEDCKO embryos was due to abnormal vascular development (Figures 2a and b).
Figure 2.
Normal blood vessel and cardiac valve development in EEDCKO. (a and b) Immunofluorescent images of skin tissue from E12.5 stage embryos. Overall, H3K27me3 signal was reduced in endothelial cells. Bar, 0.1 cm. (c and d) PECAM1 whole-mount staining of E9.5 embryos. Black arrows point at branchial arch arteries. Green arrows point at cerebral vascular plexus. Bar, 0.1 cm. (e and f) PECAM1 whole-mount staining of E12.5 embryos. (g and h) H&E staining of E12.5 embryonic heart section. Arrowheads point at atrioventricular endocardial cushion. Bar, 50 μm
To test this possibility, we examined blood vessels of wild-type and EEDCKO embryos at E9.5 and E12.5 by PECAM1 whole-mount staining, a marker of vascular endothelial cell. Unexpectedly, at E9.5, there was no global angiodysplasia observed in EED mutants. The large vessels such as the pharyngeal arch arteries, midline dorsa aorta, cardinal vein, intersomitic vessels, and large cerebral vessels were well developed in EEDCKO (Figures 2c and d). The small vessels formed complete vascular plexuses with similar density and pattern to control littermates. We also checked the vasculature of E12.5 embryos and found no significant difference in vascular density or pattern in EEDCKO compared with control (Figures 2e and f).
Endothelial cells of the heart contribute to heart valve mesenchyme formation through endothelial-to-mesenchymal transition, and Tie2Cre-driven mutations in EC genes have caused lethal heart valve defects.25, 26 To test whether the deletion of EED results in valve hypoplasia, we examined heart valves in E12.5 EEDCKO and control littermates. Cardiac valve size and morphology were comparable between mutant and wild-type embryos (Figures 2g and h). Taken together, these data suggest that EED depletion did not significantly influence vessel or valve formation, and therefore are unlikely to account for the observed embryonic lethality.
Deletion of EED attenuated the myeloid lineages commitment
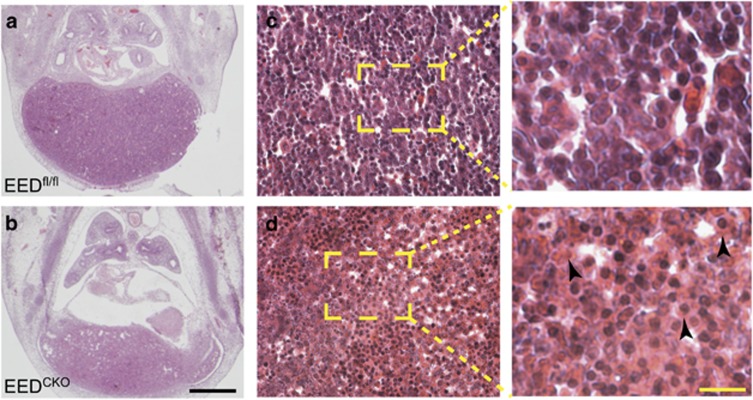
Tie2Cre drives recombination in almost all hematopoietic cells, in addition to endothelial cells.27 As the deletion of PRC2 has been reported to induce defective hematopoiesis,23, 24 we hypothesized that death of EEDCKO embryos is due to the abnormal hematopoiesis. Between E11 and E15.5, the fetal liver is the primary site of hematopoiesis.15, 28 Given the abnormal appearance of the fetal liver on gross embryo inspection (Figures 1c and d), we examined fetal liver morphology on H&E-stained histological sections and found that EEDCKO liver size was significantly reduced (Figures 3a and b). Hepatic structure was poorly organized, and the cytoplasm of blood cells within the liver were swollen and more eosinophilic than controls (Figure 3b). All together, these data suggested disrupted fetal hematopoiesis.
Figure 3.
Hepatic abnormalities of EEDCKO embryos. (a and b) H&E staining of E12.5 histological section through the liver. Arrowheads indicate swollen cells with eosinophilic cytoplasm. Black bar, 50 μm. Yellow bar, 10 μm
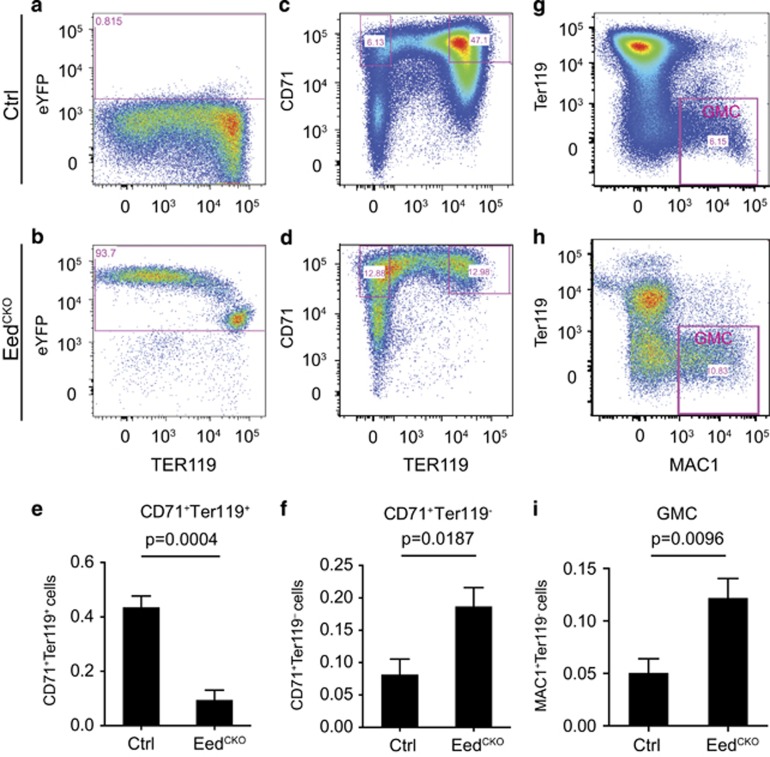
To check the status of hematopoiesis, especially erythropoiesis, whose deficiency could have contributed to the pale appearance of EEDCKO embryos, we isolated hematopoietic cells from fetal liver and examined their lineage composition by flow cytometry. Using a Cre-activated Rosa26-floxed-stop-YFP (Rosa26fsYFP) reporter, we determined that over 90% of hematopoietic cells were YFP+ (Figures 4a and b), confirming efficient Tie2Cre-mediated recombination within the hematopoietic lineage.27 We then evaluated erythropoiesis using flow cytometry to measure lineage differentiation markers. CD71+Ter119+ cells, representing more mature erythroblasts, were dramatically depleted compared with control (9.4% versus 43%, P=0.0004; Figures 4c–e). In contrast, less mature CD71+Ter119− erythroblasts were significantly increased (18.6% versus 8.1%, P=0.0001, Figures 4c, d and f), which suggested that EED is required for erythroblast maturation.
Figure 4.
EEDCKO caused defective erythrocyte maturation. Flow cytometry analysis of hematopoietic cells from hepatic liver. (a and b) Detection of hematopoietic cells that expressed YFP from Tie2Cre-recombined Rosa26fsYFP. (c and d) Flow cytometry analysis of erythroblasts (CD71+Ter119+). (e) Quantitative analysis of C71+Ter119+ erythroblasts. Unpaired t-test, n=4–7. (f and g) Flow cytometry analysis of less mature erythroblasts (CD71+Ter119−). (h) Quantitative analysis of CD71+Ter119– erythroblasts. Unpaired t-test, n=4–7. (i and j) Flow cytometry analysis of the granulocyte lineage (MAC1+Ter119–). (k) Quantitative analysis of granulocyte lineage. Unpaired t-test, n=4–7
Erythroblasts arise from a common myeloid progenitor (CMP), which also differentiates into the granulocyte–macrophage progenitor (GMP), which subsequently differentiates into granulocytes and macrophages (GMC).17, 29 The granulocytes and macrophages express the cell surface marker Mac1 and lack Ter119.17 By flow cytometry, MAC1+Ter119– cells increased almost 2.4-fold (P=0.0001) in EEDCKO (Figures 4g–i). This result suggests that EED depletion biases CMP differentiation towards GMC at the expense of the myeloid lineage.
EED depletion exhausted the HSC pool
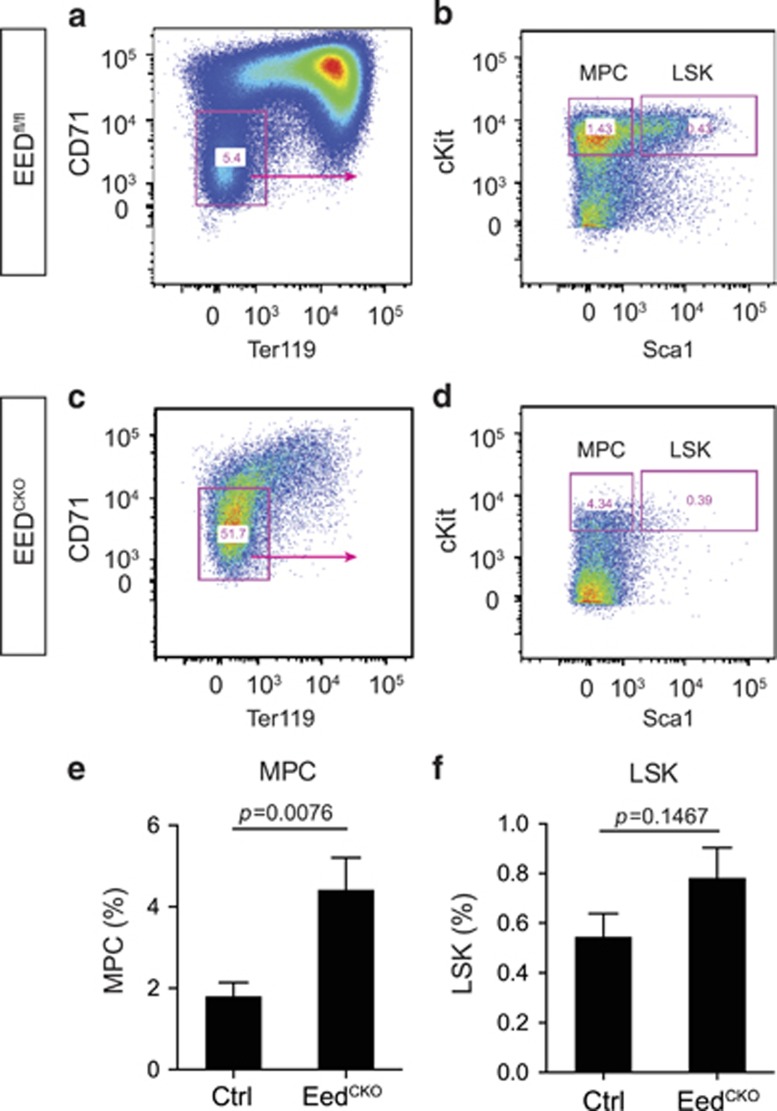
Erythroblasts and granulocytes are derived from myeloid progenitors. To investigate whether EED is required for normal formation of myeloid progenitor cells, we measured multipotent hematopoietic and myeloid progenitors by flow cytometry. Myeloid progenitor cell (MPC) lacks canonical erythrocyte maker of CD71 and Ter119. Thus, we first isolated the CD71–Ter119– blood cells and further stained them with typical progenitor markers of Sca1 and cKit to gate the MPC (Figures 5a–d). The percentage of CD71–Ter119–cKit+Sca1– myeloid progenitors was increased by 2.5-fold in EEDCKO compared with control littermates (P=0.0076; Figure 5e), indicating EED depletion hindered MPC differentiation or promotes its proliferation or self-renewal. The total number of CD71–Ter119– Sca1+cKit+ (LSK) cells was not altered in EEDCKO although they were significantly depleted in C71−Ter119− population (P=0.1467; Figure 5f).
Figure 5.
Eed deletion resulted in defective development of myeloid progenitor cells. (a–d) Flow cytometry analysis of myeloid progenitor cells (MPCs) and LSK cells from control and EEDCKO fetal liver. (e) Quantitative analysis of MPCs. Unpaired t-test, n=3–6. (f) Quantitative analysis of LSK cells. Unpaired t-test, n=3–6
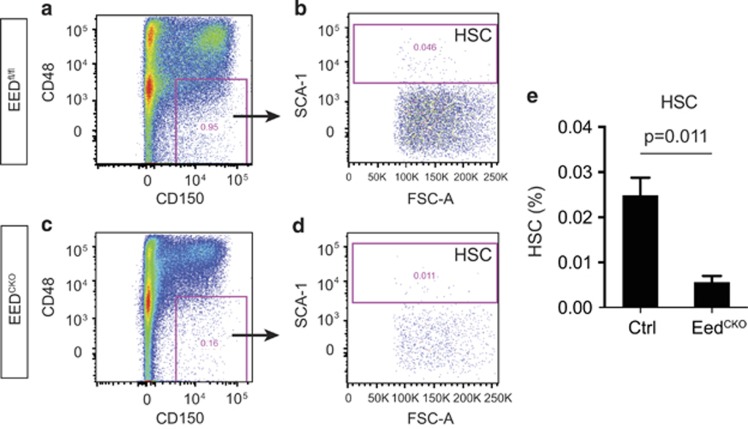
The depletion of cKit+ cells in CD71−Ter119− population indicates the loss of HSC in EEDCKO mouse. HSCs are hematopoiesis-committed multipotent cells that self-renew and give rise to all hematopoietic lineages. To test whether PRC2 regulates HSC generation or homeostasis, we stained for cell surface markers C48 and C150. Within Lin−CD48–CD150+ cells, HSC cells are the subset that expresses the pan-stem cell marker Sca123 (Figures 6a–d). Consistent with a previous study showing the impaired expansion of HSC cells following EZH2 depletion, the HSC population in EEDKO was significantly reduced (0.025 versus 0.0056%, P=0.011; Figure 6e). Taken together, these data indicate that EED deletion impaired the generation or maintenance of HSCs.
Figure 6.
Eed deletion resulted in defective HSC development. (a–d) FACS strategy defining HSC cells in wild-type and EEDCKO mouse. (e) Quantitative analysis of HSC cells. Unpaired t-test, n=3–5
Discussion
The PRC2 complex is a master regulator of differentiation programs and organogenesis. In this study, we depleted EED, a core component of PRC2. Unlike EZH1 or EZH2, there is no known functional redundancy for EED within PRC2, and therefore EED inactivation should delineate required PRC2 functions. Inactivation of EEDflox using Tie2Cre illustrated that EED depletion did not strongly affect vascular development but significantly disrupted erythroid maturation as well as HSC formation, homeostasis, and differentiation, which together led to embryonic lethality. These findings reinforced current knowledge of the essential role of PRC2 in regulating cell differentiation and homeostasis, while unveiling new roles for PRC2 in regulating hematopoiesis.
PRC2 and hematopoiesis
PRC2 has been recently shown to regulate the development and malignancy of multiple hematopoietic lineages,20, 22, 23, 30, 31 but its roles in these processes are still imprecisely defined. Ectopic expression of EZH2 in bone marrow HSCs promoted HSC proliferation and myeloproliferative disease.32 Conversely, inactivating mutations of PRC2 core members SUZ12 and EED, induced by N-ethyl-N-nitrosourea (ENU), disrupted HSC regeneration.30 Depletion of EZH2 by Tie2Cre caused defective HSC development in the fetal liver, but did not compromise adult HSC capacity to reconstitute the bone marrow.22 However, a recent study reported that deletion of EED by VavCre, which is active in hematopoietic cells, was compatible with fetal survival, leading to the opposite conclusion that PRC2 is dispensable for fetal HSC differentiation and maintenance.23 Results from these EZH2 and EED knockout studies have been difficult to compare due to the different Cre drivers, functional redundancy of EZH1/2, and non-canonical roles of EZH2 that are not dependent on EED.33 Furthermore, a recent study reported that EED functions as a scaffold protein that interacts with catalytic components of the PRC1 complex,34 adding further complexity to comparisons between EZH2 and EED knockout studies.
Our study reconciles the results of the Tie2Cre;Ezh2fl/fl and VavCre;EEDfl/fl studies to a large extent by deleting EEDfl/fl with Tie2Cre. We found that EED depletion by Tie2Cre disrupted HSC homeostasis and severely impaired erythropoiesis. We reason the different results from two experiments might be due to the earlier onset of Tie2Cre deletion in hemangioblasts. EED-deficient hemangioblasts might be defective in maitenance or further differentiation, whereas this defect may not be exposed when EED ablation occurs in HSCs or their descendants. Although lethal effects of EED depletion on hematopoiesis may require its inactivation in angioblasts, the functional defect may not occur in angioblasts, as we have previously observed that altering PRC2 in progenitors may not have functional consequences until a later developmental time point,12 perhaps reflecting a 'memory' effect of the chromatin landscape.
Tie2Cre also efficiently targets endothelial cells, which contribute to form a stem cell niche to nurse multiple types of hemotapoietic progenitors including HSC by paracrine mechanisms.15, 35 Hence, even though we did not observe vascular abnormalities in the EEDCKO mutants, it is possible that Tie2Cre-mediated EED deletion in EC could influence this paracrine mechanism and thereby cell non-autonomously influence HSC maintenance and differentiation. To test this hypothesis, a more EC-restricted Cre such as CDH5CreERT23 will need to be used.36
PRC2 and vascular development
Recently Tie2Cre;EZH2fl/fl mice were reported to have defective vascular integrity and severe hemorrhage evident at E12.5 and due to the activation of Mmp9.37 In our study, we did not find significant hemorrhage up to E12.5, a day before death. Thus, it is less likely that embryonic lethality of our Tie2Cre;EEDfl/fl mutants was due to vascular leakage. The divergent results might result from technical differences in mouse strain background or kinetics of Cre inactivation. An alternative intriguing possibility is that the more severe vascular phenotype of EZH2fl/fl mutants reflects a non-canonical, EED-independent role of EZH2 in regulating endothelial cell genes such as Mmp9. Another potential contributor to the divergent results is the recently reported role of EED in PRC1 activity.34
In adult mice, siRNA-mediated Ezh2 knockdown was used to show that EZH2 promotes tumor angiogenesis by repressing anti-angiogenic factor Vash1.14 We did not find an essential role for EED in developmental angiogenesis. These studies may point out differences in the dependence of developmental versus tumor angiogenesis on PRC2 or they may further highlight non-canonical roles of EZH2. Alternatively, technical differences in the method of gene inactivation may have led to divergent conclusions. A genetic deletion of Eed or Ezh2 by inducible CDH5CreETR2 in the adult mouse would help to solidify our current understanding of the role of PRC2 in angiogenesis and even possibly generate new insights into the functions of EED and EZH2 within PRC2 and in non-canonical roles.33
PRC2 and hematopoietic malignancies
Mutations of genes encoding PRC2 subunits have been linked to multiple types of human hematopoietic malignancies. Somatic mutation of EED or chromosome deletion of EZH2 led to myelodysplastic syndrome and related neoplasm, which is consistent to our and others studies that depletion of EZH2 and EED in hematopoietic cells impaired erythroblast generation, erythrocyte maturation, and caused cytopenia.31, 32, 38 Moreover, structural rearrange of EZH2, SUZ12, and EED both in mouse and human results in the T-cell acute lymphoblast leukemia.20 The malignant transformation in hematopoietic cells lacking PRC2 suggests that compensating for PRC2 deficiency may be a productive therapeutic strategy for some hematopoietic disorders. Consistent with this idea, overexpression of EZH2 in mice augmented HSC regeneration and prevented the HSC exhaustion during the transplantation assays.32 However, elevation of EZH2 and other PRC2 components is associated with aggressive forms in solid cancers and related to cancer progression.39, 40 As a result, PRC2 inhibition has emerged as a potential strategy cancer treatment strategy, and is currently being tested in clinical trials. Given the deleterious effects of PRC2 inhibition on HSCs and hematopoiesis illuminated in this study and others, the side effects and therapeutic index of these agents needs to be carefully evaluated.
Materials and Methods
Mice
All animal procedures were approved by Institutional Animal Care and Use Committee (IACUC) of Shanghai Jiao Tong University and Boston Children's Hospital. The EEDflox, Tie2Cre, and Rosa26fsYFP alleles were described previously.23, 41, 42 All mouse strains were maintained in a C57BL/6 and 129 mixed background. EEDfl/fl female mice were mated with Tie2Cre;EEDfl/wt male mice to generate TieCre;EEDfl/fl embyros, to avoid germline deletion that can result from Tie2Cre transmitted through the female germline. Gestational age of embryos was determined by checking vaginal plugs, with noon of the day of the plug defined as embryonic day (E) E0.5.
Immunofluorescent staining
Immunofluorescent staining of embryo sections was performed as described previously.43, 44 Cryopreserved embryos were sectioned, fixed in 4% PFA for 15 min, and blocked and permeabilized with Blocking buffer (1% BSA in PBS containing 5% goat serum) containing 0.1% Triton X-100. Following 1 h of blocking at room temperature, the sections were incubated with primary antibodies (H3K27me3, 17-622, Millipore; PECAM1, clone MEC13.3, BD Bioscience, San Diego, USA) overnight at 4 ˚C. The samples were washed three times with PBS containing 0.05% Tween-20, incubated with Alexa-conjugated secondary antibodies (1:200, Fisher Scientific, Shanghai, China) for 1 h and then counterstained with DAPI (1 μg/ml, Roche) for 5 min before mounting. The images were captured on a Olympus FluoView FV1000 confocal microscope (China).
Whole-mount staining
Whole-mount immunohistochemical staining of E9.5 and E12.5 mouse embryos using PECAM1 antibody was performed as reported previously.43, 44 Briefly, embryos were dissected from the uterus, fixed in 4% paraformaldehyde overnight, and dehydrated in 100% methanol until use. The embryo was quenched with 5% hydrogen peroxide in methanol to remove the endogenous peroxidase and then rehydrated in 75, 50, and 25% methanol before use. The blocking process was performed with incubation of blocking buffer (2% nonfat milk, 5% goat serum and 0.2% Triton X-100) for 2 h at room temperature. After blocking, PECAM1 antibody (MEC13.3, 1:200; BD Pharmingen) in blocking buffer was applied to embryos with gentle shaking overnight. The embryos were washed with PBS five times, incubated overnight with biotinylated goal anti-rat antibody (1:50, BD Pharmingen) in PBS containing 0.2% Triton X-100, and then incubated with HRP-conjugated streptavidin for 2 h before color development in DAB substrates (Vector Labs, Burlingame, CA, USA). The images were obtained using an SMZ800 stereo microscope.
Flow cytometry
Fetal livers were isolated from E12.5 mouse embryos, triturated, and passed through 70 μm nylon mesh to obtain a single-cell suspension. The RBCs were lysed in 1 × erythroid lysis buffer. The isolated cells were first incubated on ice with Fc-Block. For selection of Lin− cells, the cells were incubated with biotin-conjugated antibodies against lineage-specific markers (Ter119[TER119] CD71; B220[RA3-6B2] Gr1[RB6-8C5]), and biotin was subsequently bound by PerCP-Cy5.5 conjugated streptavidin. The cells were stained as indicated for surface markers, including FITC-CD34, phycoerythrin(PE)-Cy7-conjugated Sca1(D7), allophycocyanin (APC)-Cy7-conjugated cKit (2B8), and PE-conjugated CD48 (HM48-1). These antibodies were purchased from BD Biosciences (San Jose, CA, USA), Thermo Fisher (Shanghai, China) and BioLegend (San Diego, CA, USA). The dead cells were excluded by staining with 7-AAD (1 μg/ml, Thermo Fisher). Antibody-labeled cells were run on an LSR II/LSR Fortessa for analysis or on a FACSAria II for cell sorting. Flowjo software (Ashland, OR, USA) was used to analyze the cytometry data.
Statistics
The results are shown as mean±S.E.M. Student's t-test was used to determine whether groups were significantly different.
Acknowledgments
WTP was supported by an AHA Established Investigator Award and NIH U01HL098166. BZ was funded by National Science Foundation of China (91539109 and 31671503), A Thousand Young Talent Award (16Z127060017), NIH T32 HL7572-28, and an AHA Scientist Development Grant 14SDG20380866.
Author contributions
BZ conceived of the study. WY, FZ, BZ and WTP designed the experiments. WY, SW and YF performed the bench work. WY, FZ, JC and BZ performed the data analysis. WY, WTP and BZ drafted the manuscript. All the authors read and revised the paper.
Footnotes
Edited by H-U Simon
The authors declare no conflict of interest.
References
- Margueron R, Reinberg D. The Polycomb complex PRC2 and its mark in life. Nature 2011; 469: 343–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitcomb SJ, Basu A, Allis CD, Bernstein E. Polycomb Group proteins: an evolutionary perspective. Trends Genet 2007; 23: 494–502. [DOI] [PubMed] [Google Scholar]
- Margueron R, Li G, Sarma K, Li G, Sarma K, Blais A et al. Ezh1 and Ezh2 maintain repressive chromatin through different mechanisms. Mol Cell 2008; 32: 503–518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen X, Liu Y, Hsu YJ, Liu Y, Hsu YJ, Fujiwara Y et al. EZH1 mediates methylation on histone H3 lysine 27 and complements EZH2 in maintaining stem cell identity and executing pluripotency. Mol Cell 2008; 32: 491–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao R, Wang L, Wang H, Wang L, Wang H, Xia L et al. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 2002; 298: 1039–1043. [DOI] [PubMed] [Google Scholar]
- Kuzmichev A, Nishioka K, Erdjument-Bromage H, Tempst P, Reinberg D. Histone methyltransferase activity associated with a human multiprotein complex containing the Enhancer of Zeste protein. Genes Dev 2002; 16: 2893–2905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faust C, Schumacher A, Holdener B, Magnuson T. The eed mutation disrupts anterior mesoderm production in mice. Development 1995; 121: 273–285. [DOI] [PubMed] [Google Scholar]
- Faust C, Lawson KA, Schork NJ, Thiel B, Magnuson T. The Polycomb-group gene eed is required for normal morphogenetic movements during gastrulation in the mouse embryo. Development 1998; 125: 4495–4506. [DOI] [PubMed] [Google Scholar]
- Tee WW, Reinberg D. Chromatin features and the epigenetic regulation of pluripotency states in ESCs. Development 2014; 141: 2376–2390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezhkova E, Pasolli HA, Parker JS, Pasolli HA, Parker JS, Stokes N et al. Ezh2 orchestrates gene expression for the stepwise differentiation of tissue-specific stem cells. Cell 2009; 136: 1122–1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ezhkova E, Lien WH, Stokes N, Pasolli HA, Silva JM, Fuchs E. EZH1 and EZH2 cogovern histone H3K27 trimethylation and are essential for hair follicle homeostasis and wound repair. Genes Dev 2011; 25: 485–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He A, Ma Q, Cao J, Ma Q, Cao J, von Gise A et al. Polycomb repressive complex 2 regulates normal development of the mouse heart. Circ Res 2012; 110: 406–415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delgado-Olguin P, Huang Y, Li X, Christodoulou D, Seidman CE, Seidman JG et al. Epigenetic repression of cardiac progenitor gene expression by Ezh2 is required for postnatal cardiac homeostasis. Nat Genet 2012; 44: 343–347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu C, Han HD, Mangala LS, Ali-Fehmi R, Newton CS, Ozbun L et al. Regulation of tumor angiogenesis by EZH2. Cancer Cell 2010; 18: 185–197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orkin SH, Zon LI. Hematopoiesis: an evolving paradigm for stem cell biology. Cell 2008; 132: 631–644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akashi K, Traver D, Kondo M, Weissman IL. Lymphoid development from hematopoietic stem cells. Int J Hematol 1999; 69: 217–226. [PubMed] [Google Scholar]
- Iwasaki H, Akashi K. Myeloid lineage commitment from the hematopoietic stem cell. Immunity 2007; 26: 726–740. [DOI] [PubMed] [Google Scholar]
- Ueda T, Sanada M, Matsui H, Yamasaki N, Honda ZI, Shih LY et al. EED mutants impair polycomb repressive complex 2 in myelodysplastic syndrome and related neoplasms.[letter]. Leukemia 2012; 26: 2557–2560. [DOI] [PubMed] [Google Scholar]
- Stepanik VA, Harte PJ. A mutation in the E(Z) methyltransferase that increases trimethylation of histone H3 lysine 27 and causes inappropriate silencing of active Polycomb target genes. Dev Biol 2012; 364: 249–258. [DOI] [PubMed] [Google Scholar]
- Simon C, Chagraoui J, Krosl J, Gendron P, Wilhelm B, Lemieux S et al. A key role for EZH2 and associated genes in mouse and human adult T-cell acute leukemia. Genes Dev 2012; 26: 651–656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sauvageau M, Sauvageau G. Polycomb group proteins: multi-faceted regulators of somatic stem cells and cancer. Cell Stem Cell 2010; 7: 299–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mochizuki-Kashio M, Mishima Y, Miyagi S, Negishi M, Saraya A, Konuma T et al. Dependency on the polycomb gene Ezh2 distinguishes fetal from adult hematopoietic stem cells. Blood 2011; 118: 6553–6561. [DOI] [PubMed] [Google Scholar]
- Xie H, Xu J, Hsu JH, Nguyen M, Fujiwara Y, Peng C et al. Polycomb repressive complex 2 regulates normal hematopoietic stem cell function in a developmental-stage-specific?thyc?> manner. Cell Stem Cell 2014; 14: 68–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lessard J, Schumacher A, Thorsteinsdottir U, van Lohuizen M, Magnuson T, Sauvageau G. Functional antagonism of the Polycomb-Group genes eed and Bmi1 in hemopoietic cell proliferation. Genes Dev 1999; 13: 2691–2703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivera-Feliciano J, Lee KH, Kong SW, Rajagopal S, Ma Q, Springer Z et al. Development of heart valves requires Gata4 expression in endothelial-derived cells. Development 2006; 133: 3607–3618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, von Gise A, Liu Q, Hu T, Tian X, He L et al. Yap1 is required for endothelial to mesenchymal transition of the atrioventricular cushion. J Biol Chem 2014; 289: 18681–18692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liakhovitskaia A, Gribi R, Stamateris E, Villain G, Jaffredo T, Wilkie R et al. Restoration of Runx1 expression in the Tie2 cell compartment rescues definitive hematopoietic stem cells and extends life of Runx1 knockout animals until birth. Stem Cells 2009; 27: 1616–1624. [DOI] [PubMed] [Google Scholar]
- Palis J, Robertson S, Kennedy M, Wall C, Keller G. Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development 1999; 126: 5073–5084. [DOI] [PubMed] [Google Scholar]
- Arinobu Y, Iwasaki H, Gurish MF, Mizuno S, Shigematsu H, Ozawa H et al. Developmental checkpoints of the basophil/mast cell lineages in adult murine hematopoiesis. Proc Natl Acad Sci USA 2005; 102: 18105–18110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamminga LM, Bystrykh LV, de Boer A et al, Houwer S, Douma J, Weersing E The Polycomb group gene Ezh2 prevents hematopoietic stem cell exhaustion. Blood 2006; 107: 2170–2179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makishima H, Jankowska AM, Tiu RV, Szpurka H, Sugimoto Y, Hu Z et al. Novel homo- and hemizygous mutations in EZH2 in myeloid malignancies.[letter]. Leukemia 2010; 24: 1799–1804. [DOI] [PubMed] [Google Scholar]
- Herrera-Merchan A, Arranz L, Ligos JM, de Molina A, Dominguez O, Gonzalez S. Ectopic expression of the histone methyltransferase Ezh2 in haematopoietic stem cells causes myeloproliferative disease. Nat Commun 2012; 3: 623. [DOI] [PubMed] [Google Scholar]
- Xu K, Wu ZJ, Groner AC, He HH, Cai C, Lis RT et al. EZH2 oncogenic activity in castration-resistant prostate cancer cells is Polycomb-independent. Science 2012; 338: 1465–1469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Q, Wang X, Zhao M, Yang R, Malik R, Qiao Y et al. The central role of EED in the orchestration of polycomb group complexes. Nat Commun 2014; 5: 3127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rafii S, Butler JM, Ding BS. Angiocrine functions of organ-specific endothelial cells. Nature 2016; 529: 316–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pitulescu ME, Schmidt I, Benedito R, Adams RH. Inducible gene targeting in the neonatal vasculature and analysis of retinal angiogenesis in mice. Nat Protoc 2010; 5: 1518–1534. [DOI] [PubMed] [Google Scholar]
- Delgado-Olguín P, Dang LT, He D, Thomas S, Chi L, Sukonnik T et al. Ezh2-mediated repression of a transcriptional pathway upstream of Mmp9 maintains integrity of the developing vasculature. Development 2014; 141: 4610–4617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morin RD, Johnson NA, Severson TM, Mungall AJ, An J, Goya R et al. Somatic mutations altering EZH2 (Tyr641) in follicular and diffuse large B-cell lymphomas of germinal-center origin. Nat Genet 2010; 42: 181–185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee ST, Li Z, Wu Z, Aau M, Guan P, Karuturi RK et al. Context-specific regulation of NF-κB target gene expression by EZH2 in breast cancers. Mol Cell 2011; 43: 798–810. [DOI] [PubMed] [Google Scholar]
- Varambally S, Dhanasekaran SM, Zhou M, Barrette TR, Kumar-Sinha C, Sanda MG et al. The polycomb group protein EZH2 is involved in progression of prostate cancer. Nature 2002; 419: 624–629. [DOI] [PubMed] [Google Scholar]
- Srinivas S, Watanabe T, Lin CS, William CM, Tanabe Y, Jessell TM et al. Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC. Dev Biol 2001; 1: 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kisanuki YY, Hammer RE, Miyazaki J, Williams SC, Richardson JA, Yanagisawa M. Tie2-Cre transgenic mice: a new model for endothelial cell-lineage analysis in vivo. Dev Biol 2001; 230: 230–242. [DOI] [PubMed] [Google Scholar]
- Zhang B, Xiao W, Qiu H, Zhang F, Moniz HA, Jaworski A et al. Heparan sulfate deficiency disrupts developmental angiogenesis and causes congenital diaphragmatic hernia. J Clin Invest 2014; 124: 209–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang B, Dietrich UM, Geng JG, Bicknell R, Esko JD, Wang L. Repulsive axon guidance molecule Slit3 is a novel angiogenic factor. Blood 2009; 114: 4300–4309. [DOI] [PMC free article] [PubMed] [Google Scholar]