Abstract
Recent genome-wide association studies implicate microglia in Alzheimer’s disease (AD) pathogenesis; however, their biological significance remains poorly understood. Synapse loss is a significant correlate of cognitive decline that serves as a critical hallmark of AD and other neurodegenerative diseases; however, mechanisms underlying synaptic vulnerability remain elusive. Emerging research on microglia function in the healthy brain is providing new insight into fundamental roles of microglia and immune molecules in brain wiring. Among their many roles, microglia prune developing synapses and regulate synaptic plasticity and function. Here, we review and discuss how this emerging work may provide new insight into how disruptions in microglia–synapse interactions could contribute to synapse loss and dysfunction, and consequently cognitive impairment, in AD.
Introduction
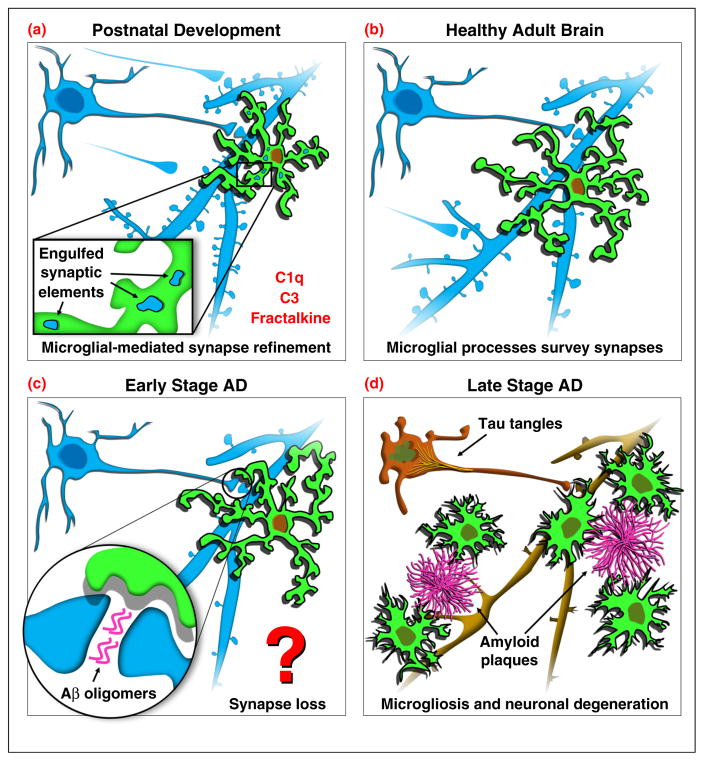
Alzheimer’s disease (AD) is a progressive neurological disorder in which the earliest clinical symptoms involve impaired declarative memory and general cognitive impairment, such as compromised judgment, decision-making and orientation. The two pathological hallmarks of AD are extracellular neuritic plaques, in which the amyloid β-protein (Aβ) is the principal component, and intraneuronal tangles, which are non-membrane bound masses of paired helical filaments composed primarily of hyperphosphorylated tau. A frequent observation in brains of both AD patients and animal models of AD is the surrounding of neuritic amyloid plaques by highly reactive, phagocytic microglia. Thus, there has been extensive research to understand the role of microglia in the AD brain and their interaction with amyloid plaques and tangles, and their contributions to neuroinflammation and AD pathology (see comprehensive review in [1]). Recent genome-wide association studies (GWAS) and other integrated network studies have identified immune-related pathways as risk factors for late-onset AD, implicating microglia, the resident immune cells of the central nervous system (CNS), as central players in AD pathogenesis. Among these are Trem2, CR1, ApoJ/Clusterin, CD33, and immune-specific and microglia-specific network modules that include TYR-OBP [2–9]; however, the biological significance of these findings remains elusive. Recent research has focused on understanding how microglia and these immune-related molecules contribute to amyloid plaque deposition, maintenance and clearance, but little is known on what microglia do in earlier, pre-plaque stages of AD when synapses are already vulnerable. Emerging research implicates microglia and immune-related mechanisms in synapse elimination during development, raising the intriguing hypothesis that these developmental pruning mechanisms may be aberrantly reactivated in the aged brain to contribute to synapse loss and cognitive impairment in AD and other neurodegenerative diseases (NDDs) (Figure 1).
Figure 1.
Potential roles of microglia in the healthy and diseased brain. (a) During early postnatal development, microglia (illustrated in green) help refine excessive synaptic connections (illustrated in blue). Insert highlights microglial engulfment of synaptic elements. Microglia-related proteins including complement proteins and fractalkine have been suggested to mediate this process. (b) In the healthy adult brain, microglial processes are dynamic and continuously survey surrounding synapses. (c) Synapse loss is an early hallmark of AD pathology, thought to be initiated by Aβ oligomers. What microglia do in this early stage of AD remains poorly understood. (d) Late stage AD is characterized by the presence of extracellular plaque deposition, intraneuronal tangle formation and neurodegeneration, often accompanied by significant levels of microgliosis and neuroinflammation. Recent research has given insight into how microglia may impact plaque and plaque-related neuropathology in late stage AD; however, what microglia do in early AD, in particular, how their dynamic interactions with synapses are impacted, is unknown. An intriguing hypothesis is whether microglia and immune-related molecules that mediate synaptic engulfment in the developing brain (a) may be aberrantly reactivated in the AD brain (c) to mediate synapse loss and degeneration.
Microglia and their potential roles in Aβ plaque maintenance
One of the cardinal features of AD brains is the presence of reactive microglia surrounding senile Aβ plaques along with prominent activation of inflammatory processes and immune responses. Recent in vivo imaging and microglial ablation studies have provided insight into how microglia may impact plaque deposition, maintanence and clearance. Time lapse imaging in cerebral cortex of plaque-burdened AD mouse models revealed microglia to be recruited rapidly (within 48 hours) to newly formed plaques and that microglial processes remain very dynamic [10,11]. Microglia that surround plaques appear to constitute a barrier to not only restrict plaque growth, but also may prevent diffusion of synaptotoxic Aβ oligomers from plaques [10,11,12•]. Furthermore, microglia that surround plaques may ultimately become dysfunctional, displaying decreased process number and directed process motility and impaired phagocytic activity in a manner that temporally and spatially correlates with plaque deposition [13,14•]. However, microglial ablation studies suggest that microglia may have a minimal role in maintaining plaques: ablating microglia for up to four weeks in two mouse models of AD crossed with CD11b-HSVTK mice, which allows deletion of microglia following treatment with ganciclovir, had little to no impact on either plaque number or size or on amyloid-associated neuritic dystrophy, regardless of whether microglia were ablated before or after plaques deposit in these mice [15]. A recent study using PLX5622, an inhibitor against colony-stimulating factor 1 receptor (CSF1R) signaling upon which microglia are dependent on survival, induced a partial, chronic reduction of microglia in an AD mouse model, but failed to detect any alteration of plaque load [16•]. However, the PLX5622-treated AD mice performed better in learning and memory tests than control-treated AD mice, despite having similar levels of plaque load. Altogether, these studies suggest that microglia may have roles in the AD brain beyond plaque maintenance, with potential behavioral consequences impacting learning and memory.
Dynamic microglia–synapse interactions in the healthy brain
Microglia derive from myeloid progenitor cells in the yolk sac and colonize the brain early during embryonic development [17–19]. Once in the CNS, they adopt tissue-specific signatures and functions through local microenvironment signals [20–24]. Until a decade ago, microglia were studied mostly in the context of CNS injury and disease [25]. However, groundbreaking in vivo imaging studies revealed that microglia in the healthy brain have highly dynamic processes and continually survey their local microenvironment [26,27]. Moreover, microglial processes constantly contact dendritic spines, axons and synapses, raising questions about whether and how microglia regulate synaptic structure and function [28–33,34••,35•].
Emerging research implicates microglia and immune-related proteins in the elimination and refinement of synaptic connections in the healthy developing brain [29–32,36,37••]. Microglial processes make direct and transient connections with neuronal synapses where, intriguingly, the frequency of these connections is related to basal neuronal activity and/or experience [28,29]. Direct evidence of microglia sculpting the brain came from electron microscopy and high resolution in vivo engulfment assays where presynaptic and postsynaptic elements were found inside microglial lysosomes [29,31]. Interestingly, neuronal activity regulated the microglial engulfment of synaptic input, where microglia preferentially phagocytosed less active inputs [31]. Furthermore, disruption of immune-related molecules or receptors expressed on microglia, such as complement proteins or complement and fractalkine receptors, resulted in synaptic and wiring abnormalities in both prenatal and postnatal brain development [30–33,35•], implicating microglia in sculpting synaptic connectivity. Interestingly, ablating microglia in the adult brain induced impairment in learning tasks that correlated with reduction in learning-induced synapse formation in the motor cortex [34••], suggesting that microglia regulate synaptic formation and/or elimination depending on context (i.e. different brain regions, developmental time points and behavior).
AD: a disorder of degenerating selected synapses
Region-specific degeneration and loss of synapses that precede neuronal death is an early hallmark that differentiates AD from normal aging [38]. Furthermore, synapse loss in the hippocampus and association cortices serves as a much stronger correlate of cognitive impairment in AD than do counts of amyloid plaques or neurofibrillary tangles [39]. Research in the last two decades has provided extensive evidence that soluble oligomers of Aβ, and not plaques per se, act as prime synaptotoxic agents (see comprehensive review in [40]). In multiple mouse models of AD, synapse numbers are significantly lower than in non-transgenic controls already at a very young age, long before these mice develop amyloid plaques [41–45]. In another striking example, mice expressing a familial APP mutation where the synaptotoxic Aβ oligomers are present and stabilized in the absence of fibrillar amyloid plaques display profound synaptic and neuronal loss in their cortex [46].
Despite the physical degeneration of synapses being an early, critical event in AD, whether microglia play a role in these early, pre-plaque stages of pathogenesis and how they may impact synaptic function remains little explored. As mentioned above, much of the research on the role of microglia has focused on their relationship with plaques. Although these are important studies that help us understand whether microglia may impact plaques and plaque-related neurodegeneration, there already exists a significant level of widespread neuroinflammation and associated gliosis at these late stages, clouding investigation of potential neuroimmune perturbations that may directly contribute to the initiation of synaptic and cognitive impairment. Therefore, it will be pertinent to shift the focus of investigation away from late stages of AD toward earlier stages when synapses are already vulnerable to explore potential dysfunction of microglia or immune-related pathways and how these may relate to synaptotoxic Aβ oligomers.
Possible mechanisms of microglia–synapse dysfunction in the AD brain
Recent GWAS and other integrated network studies in AD have revealed microglia-specific and immune-related genes [2–9], suggesting microglia could play a major role in AD pathogenesis. However, the biological significance remains elusive, and whether microglia become dysfunctional in the AD brain regions vulnerable to synapse loss prior to plaque accumulation is unknown.
Among the GWAS-identified variants are CLU, also known as complement lysis inhibitor or APOJ, and CR1, which encodes for the receptor for complement component C3b [3]. In the AD brain, complement proteins, in particular, C1q, C3 and C4, are often found upregulated and localized to neuritic plaques. Moreover, Aβ has been shown to bind and regulate the expression and localization of complement [1,47]. Genetic deletion of C1qa in an AD mouse model resulted in less plaque-related neuronal damage and gliosis, whereas genetic deletion of C3 resulted in more plaques and increased plaque-related neurodegeneration [48–50]. These studies suggest that complement proteins may have multiple roles in plaque-related pathology; however, it is not yet known whether complement is dysregulated early in the AD brain or whether this immune pathway contributes to synaptic or cognitive impairment.
In the healthy developing brain, classical complement cascade proteins, C1q and C3, are widely expressed, localize to subsets of immature synapses, and mediate synapse elimination, a developmental process critical for precise synaptic connectivity [31,36,37••,51]. Importantly, disruption of this pruning pathway results in sustained defects on synaptic connectivity and brain wiring. In the peripheral immune system, classical complement proteins are ‘eat me’ signals that promote the rapid clearance of invading pathogens or cellular debris by circulating macrophages that express complement receptors (C1qR and CR3) [52]. In the developing visual system, microglia phagocytose synapses that are undergoing pruning in a manner that is dependent on complement (C1q, C3 and CR3) and neuronal activity [31,37••]. Several complement components, including C3, are normally downregulated in the healthy adult brain [31,51] and complement-mediated synaptic pruning is regulated by several signals, including cytokine TGFβ [37••]. Interestingly, C1q is highly upregulated and deposits on synapses with normal aging in human and mouse brains, particularly in the hippocampus, one of the brain regions most vulnerable to synapse loss in AD [53••]. C3 has also been shown to contribute to synapse loss and dysfunction in the mouse hippocampus during normal aging [54••]. Altogether, these findings raise the intriguing question of whether C1q and/or C3 increases the susceptibility of the hippocampus to further insults such as accumulation of synaptotoxic Aβ oligomers. An important question for future investigation is whether the normal developmental pruning pathway becomes aberrantly upregulated to mediate synapse loss by microglia in early stages of AD and other NDDs [51]. Indeed, emerging data suggest complement may be a key mechanism underlying synaptic vulnerability in early stages of AD [55].
Another immune-related pathway that warrants further studies on synaptic health in the AD brain is TREM2, which is expressed on myeloid cells and whose missense mutations lead to a significant risk of developing AD [6,7]. Recent exciting studies have extensively focused on whether TREM2 may affect Aβ plaque pathogenesis; TREM2 appears to affect the number of plaque-associated microglia but its overall impact on plaque burden seems to depend on age and on which mouse model was utilized [56,57•,58•,59••]. TREM2 has been shown to impact inflammatory signals as well as phagocytosis in the peripheral immune system [60]; however, the normal function of TREM2 in the CNS remains poorly understood. Interestingly, loss-of-function mutations in TREM2 or DAP12, an adaptor protein for TREM2 signaling, underlie the Nasu–Hakola disease, in which patients display progressive presenile dementia [61,62], and mice expressing mutations in DAP12 display impaired synaptic maturation [63]. These findings suggest that TREM2 may play an important role for maturation and maintenance of synaptic function and connectivity. Interestingly, TREM2 has recently been suggested to interact with certain lipids to promote microglial survival [59••]. This is particularly intriguing given that certain lipid structures on neuronal membranes have been implicated to bind Aβ oligomers to potentially mediate downstream synaptic dysfunction [64]. Collectively, these findings raise the possibility that TREM2 could play a role in early synaptic dysfunction in AD and the need to better understand the biology of TREM2 in the context of both the healthy and diseased CNS.
Role of microglia in region-specific network dysfunction
Region-specific loss of synaptic integrity and aberrant neuronal networks have been linked to many developmental and neurodegenerative diseases [65–68]. In AD, elaborate in vivo tools including structural and functional magnetic resonance imaging, positron emission tomography (PET) and microdialysis in humans and animal models have revealed an intricate relationship among cognitive status, neural or astroglial activity, Aβ dynamics, and CNS structural changes [69–74]. Interestingly, areas of the human brain that develop the most Aβ deposits may also have among the highest basal rates of metabolic and neural activity [71,73,75]. This is particularly intriguing as neuronal activity has been shown to regulate Aβ production [76,77] and also to modulate microglia–synapse interactions [28,29] and microglial elimination of synapses in the developing brain [31]. We propose a model in which aberrant neuronal activity through genetic or other causes may trigger local activation of microglia and other immune-related molecules and thus contribute to region-specific vulnerability in AD and potentially other NDDs.
Conclusion
Emerging evidence implicates microglia as key participants in the wiring of the developing brain. Given the intricate relationship between synaptic/neuronal activity, cognition and microglial function in development, it is a salient time for AD research to focus on understanding whether, and how, microglia contribute to synaptic loss and impairment with attendant behavioral outcomes. If so, microglia and relevant immune-related pathways may act as early therapeutic targets in AD and potentially other NDDs involving synaptic dysfunction and memory decline.
Acknowledgments
This work was funded by Edward R. and Anne G. Lefler Fellowship (SH), National Institutes of Health AG000222 (SH), Lundbeck Foundation (LDO), Coins for Alzheimer’s Research Trust (BS), and Anonymous (BS).
Footnotes
Conflict of interest statement
B.S. serves on the Scientific Advisory Board of and is a minor shareholder of Annexon LLC. All other authors declare no competing financial interests.
References and recommended reading
Papers of particular interest, published within the period of review, have been highlighted as:
• of special interest
•• of outstanding interest
- 1.Wyss-Coray T, Rogers J. Inflammation in Alzheimer disease — a brief review of the basic science and clinical literature. Cold Spring Harb Perspect Med. 2012;2:a006346. doi: 10.1101/cshperspect.a006346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bertram L, Lange C, Mullin K, Parkinson M, Hsiao M, Hogan MF, Schjeide BMM, Hooli B, DiVito J, Ionita I, et al. Genome-wide association analysis reveals putative Alzheimer’s disease susceptibility loci in addition to APOE. Am J Hum Genet. 2008;83:623–632. doi: 10.1016/j.ajhg.2008.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lambert J-C, Heath S, Even G, Campion D, Sleegers K, Hiltunen M, Combarros O, Zelenika D, Bullido MJ, Tavernier B, et al. Genome-wide association study identifies variants at CLU and CR1 associated with Alzheimer’s disease. Nat Genet. 2009;41:1094–1099. doi: 10.1038/ng.439. [DOI] [PubMed] [Google Scholar]
- 4.Hollingworth P, Harold D, Sims R, Gerrish A. Common variants at ABCA7, MS4A6A/MS4A4E, EPHA1, CD33 and CD2AP are associated with Alzheimer’s disease. Nature. 2011 doi: 10.1038/ng.803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Naj AC, Jun G, Beecham GW, Wang L-S, Vardarajan BN, Buros J, Gallins PJ, Buxbaum JD, Jarvik GP, Crane PK, et al. Common variants at MS4A4/MS4A6E, CD2AP, CD33 and EPHA1 are associated with late-onset Alzheimer’s disease. Nat Genet. 2011;43:436–441. doi: 10.1038/ng.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Guerreiro R, Wojtas A, Bras J, Carrasquillo M, Rogaeva E, Majounie E, Cruchaga C, Sassi C, Kauwe JSK, Younkin S, et al. TREM2 variants in Alzheimer’s disease. N Engl J Med. 2013;368:117–127. doi: 10.1056/NEJMoa1211851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jonsson T, Stefansson H, Steinberg S, Jonsdottir I, Jonsson PV, Snaedal J, Bjornsson S, Huttenlocher J, Levey AI, Lah JJ, et al. Variant of TREM2 associated with the risk of Alzheimer’s disease. N Engl J Med. 2013;368:107–116. doi: 10.1056/NEJMoa1211103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang B, Gaiteri C, Bodea L-G, Wang Z, McElwee J, Podtelezhnikov AA, Zhang C, Xie T, Tran L, Dobrin R, et al. Integrated systems approach identifies genetic nodes and networks in late-onset Alzheimer’s disease. Cell. 2013;153:707–720. doi: 10.1016/j.cell.2013.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lambert J-C, Ibrahim-Verbaas CA, Harold D, Naj AC, Sims R, Bellenguez C, Jun G, DeStefano AL, Bis JC, Beecham GW, et al. Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat Genet. 2013;45:1452–1458. doi: 10.1038/ng.2802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meyer-Luehmann M, Spires-Jones TL, Prada C, Garcia-Alloza M, De Calignon A, Rozkalne A, Koenigsknecht-Talboo J, Holtzman DM, Bacskai BJ, Hyman BT. Rapid appearance and local toxicity of amyloid-β plaques in a mouse model of Alzheimer’s disease. Nature. 2008;451:720–724. doi: 10.1038/nature06616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bolmont T, Haiss F, Eicke D, Radde R, Mathis CA, Klunk WE, Kohsaka S, Jucker M, Calhoun ME. Dynamics of the microglial/amyloid interaction indicate a role in plaque maintenance. J Neurosci. 2008;28:4283–4292. doi: 10.1523/JNEUROSCI.4814-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12•.Condello C, Yuan P, Schain A, Grutzendler J. Microglia constitute a barrier that prevents neurotoxic protofibrillar Aβ42 hotspots around plaques. Nat Commun. 2015;6:6176. doi: 10.1038/ncomms7176. This study shows that areas of plaques that are covered by microglial processes are more compact and have a lower affinity for neurotoxic Aβ. Their data indicate that microglia exert a functional role in preventing neuritic dystrophy. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Koenigsknecht-Talboo J, Meyer-Luehmann M, Parsadanian M, Garcia-Alloza M, Finn MB, Hyman BT, Bacskai BJ, Holtzman DM. Rapid microglial response around amyloid pathology after systemic anti-Abeta antibody administration in PDAPP mice. J Neurosci. 2008;28:14156–14164. doi: 10.1523/JNEUROSCI.4147-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14•.Krabbe G, Halle A, Matyash V, Rinnenthal JL, Eom GD, Bernhardt U, Miller KR, Prokop S, Kettenmann H, Heppner FL. Functional impairment of microglia coincides with beta-amyloid deposition in mice with Alzheimer-like pathology. PLoS ONE. 2013;8:e60921. doi: 10.1371/journal.pone.0060921. Using both in vivo and ex vivo two-photon imaging, the authors demonstrate that microglia become dysfunctional in mice with AD-like pathology, in particular in association with plaque deposition. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Grathwohl SA, Kälin RE, Bolmont T, Prokop S, Winkelmann G, Kaeser SA, Odenthal J, Radde R, Eldh T, Gandy S, et al. Formation and maintenance of Alzheimer’s disease beta-amyloid plaques in the absence of microglia. Nat Neurosci. 2009;12:1361–1363. doi: 10.1038/nn.2432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16•.Dagher NN, Najafi AR, Kayala KMN, Elmore MRP, White TE, Medeiros R, West BL, Green KN. Colony-stimulating factor 1 receptor inhibition prevents microglial plaque association and improves cognition in 3xTg-AD mice. J Neuroinflamm. 2015;12:139. doi: 10.1186/s12974-015-0366-9. Sustained microglia elimination or dissociation with plaques, by inhibition of CSF1R, averts decline in learning and memory in a mouse model of AD. The Green lab was the first to show that inhibition of CSF1R alone is sufficient to eliminate microglia (see Elmore et al. Neuron 2014) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ginhoux F, Greter M, Leboeuf M, Nandi S, See P, Gokhan S, Mehler MF, Conway SJ, Ng LG, Stanley ER, et al. Fate mapping analysis reveals that adult microglia derive from primitive macrophages. Science. 2010;330:841–845. doi: 10.1126/science.1194637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Perdiguero EG, Klapproth K, Schulz C, Busch K, Azzoni E, Crozet L, Garner H, Trouillet C, de Bruijn MF, Geissmann F, et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature. 2014 doi: 10.1038/nature13989. http://dx.doi.org/10.1038/nature13989. [DOI] [PMC free article] [PubMed]
- 19.Ginhoux F, Prinz M. Origin of microglia: current concepts and past controversies. Cold Spring Harb Perspect Med. 2015 doi: 10.1101/cshperspect.a020537. http://dx.doi.org/10.1101/cshperspect.a020537. [DOI] [PMC free article] [PubMed]
- 20.Gautier EL, Shay T, Miller J, Greter M, Jakubzick C, Ivanov S, Helft J, Chow A, Elpek KG, Gordonov S, et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat Immunol. 2012;13:1118–1128. doi: 10.1038/ni.2419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hickman SE, Kingery ND, Ohsumi TK, Borowsky ML, Wang L-C, Means TK, Khoury ElJ. The microglial sensome revealed by direct RNA sequencing. Nat Neurosci. 2013;16:1896–1905. doi: 10.1038/nn.3554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Butovsky O, Jedrychowski MP, Moore CS, Cialic R, Lanser AJ, Gabriely G, Koeglsperger T, Dake B, Wu PM, Doykan CE, et al. Identification of a unique TGF-β-dependent molecular and functional signature in microglia. Nat Neurosci. 2013;17:131–143. doi: 10.1038/nn.3599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gosselin D, Link VM, Romanoski CE, Fonseca GJ, Eichenfield DZ, Spann NJ, Stender JD, Chun HB, Garner H, Geissmann F, et al. Environment drives selectionand function of enhancers controlling tissue-specific macrophage identities. Cell. 2014;159:1327–1340. doi: 10.1016/j.cell.2014.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lavin Y, Winter D, Blecher-Gonen R, David E, Keren-Shaul H, Merad M, Jung S, Amit I. Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell. 2014;159:1312–1326. doi: 10.1016/j.cell.2014.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hanisch U-K, Kettenmann H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci. 2007;10:1387–1394. doi: 10.1038/nn1997. [DOI] [PubMed] [Google Scholar]
- 26.Davalos D, Grutzendler J, Yang G, Kim JV, Zuo Y, Jung S, Littman DR, Dustin ML, Gan W-B. ATP mediates rapid microglial response to local brain injury in vivo. Nat Neurosci. 2005;8:752–758. doi: 10.1038/nn1472. [DOI] [PubMed] [Google Scholar]
- 27.Nimmerjahn A, Kirchhoff F, Helmchen F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science. 2005;308:1314–1318. doi: 10.1126/science.1110647. [DOI] [PubMed] [Google Scholar]
- 28.Wake H, Moorhouse AJ, Jinno S, Kohsaka S, Nabekura J. Resting microglia directly monitor the functional state of synapses in vivo and determine the fate of ischemic terminals. J Neurosci. 2009;29:3974–3980. doi: 10.1523/JNEUROSCI.4363-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tremblay M-È, Lowery RL, Majewska AK. Microglial interactions with synapses are modulated by visual experience. PLoS Biol. 2010;8:e1000527. doi: 10.1371/journal.pbio.1000527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Paolicelli RC, Bolasco G, Pagani F, Maggi L, Scianni M, Panzanelli P, Giustetto M, Ferreira TA, Guiducci E, Dumas L, et al. Synaptic pruning by microglia is necessary for normal brain development. Science. 2011;333:1456–1458. doi: 10.1126/science.1202529. [DOI] [PubMed] [Google Scholar]
- 31.Schafer DP, Lehrman EK, Kautzman AG, Koyama R, Mardinly AR, Yamasaki R, Ransohoff RM, Greenberg ME, Barres BA, Stevens B. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron. 2012;74:691–705. doi: 10.1016/j.neuron.2012.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hoshiko M, Arnoux I, Avignone E, Yamamoto N, Audinat E. Deficiency of the microglial receptor CX3CR1 impairs postnatal functional development of thalamocortical synapses in the barrel cortex. J Neurosci. 2012;32:15106–15111. doi: 10.1523/JNEUROSCI.1167-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ueno M, Fujita Y, Tanaka T, Nakamura Y, Kikuta J, Ishii M, Yamashita T. Layer V cortical neurons require microglial support for survival during postnatal development. Nat Neurosci. 2013;16:543–551. doi: 10.1038/nn.3358. [DOI] [PubMed] [Google Scholar]
- 34••.Parkhurst CN, Yang G, Ninan I, Savas JN, Yates JR, Lafaille JJ, Hempstead BL, Littman DR, Gan W-B. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell. 2013;155:1596–1609. doi: 10.1016/j.cell.2013.11.030. By selective depletion of microglial cells or microglial BDNF, the authors demonstrated that microglia promote learning-dependent synapse formation, potentially through BDNF signaling. This study is the first to selectively manipulate microglial cells in vivo without affecting peripheral immune cells. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35•.Squarzoni P, Oller G, Hoeffel G, Pont-Lezica L, Rostaing P, Low D, Bessis A, Ginhoux F, Garel S. Microglia modulate wiring of the embryonic forebrain. Cell Rep. 2014;8:1271–1279. doi: 10.1016/j.celrep.2014.07.042. The authors manipulated multiple microglia-specific pathways using different transgenic mouse lines to show that embryonic microglia may regulate the wiring of forebrain circuits. The data indicates that both dopaminergic axon outgrowth and neocortical interneuron positioning are modulated by microglia. [DOI] [PubMed] [Google Scholar]
- 36.Stevens B, Allen NJ, Vazquez LE, Howell GR, Christopherson KS, Nouri N, Micheva KD, Mehalow AK, Huberman AD, Stafford B, et al. The classical complement cascade mediates CNS synapse elimination. Cell. 2007;131:1164–1178. doi: 10.1016/j.cell.2007.10.036. [DOI] [PubMed] [Google Scholar]
- 37••.Bialas AR, Stevens B. TGF-β signaling regulates neuronal C1q expression and developmental synaptic refinement. Nat Neurosci. 2013;16:1773–1782. doi: 10.1038/nn.3560. Complement proteins C1q and C3 mediate synaptic refinement in the developing visual system. Here, the authors identify astrocytic TGF-β as a key regulator of neuronal C1q expression and downstream microglia-mediated synaptic pruning. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 38.West MJ, Coleman PD, Flood DG, Troncoso JC. Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer’s disease. Lancet. 1994;344:769–772. doi: 10.1016/s0140-6736(94)92338-8. [DOI] [PubMed] [Google Scholar]
- 39.Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R, Hansen LA, Katzman R. Physical basis of cognitive alterations in Alzheimer’s disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol. 1991;30:572–580. doi: 10.1002/ana.410300410. [DOI] [PubMed] [Google Scholar]
- 40.Mucke L, Selkoe DJ. Neurotoxicity of amyloid β-protein: synaptic and network dysfunction. Cold Spring Harb Perspect Med. 2012;2:a006346. doi: 10.1101/cshperspect.a006338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hsia AY, Masliah E, McConlogue L, Yu GQ, Tatsuno G, Hu K, Kholodenko D, Malenka RC, Nicoll RA, Mucke L. Plaque-independent disruption of neural circuits in Alzheimer’s disease mouse models. Proc Natl Acad Sci U S A. 1999;96:3228–3233. doi: 10.1073/pnas.96.6.3228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mucke L, Masliah E, Yu G-Q, Mallory M, Rockenstein E, Tatsuno G, Hu K, Kholodenko D, Johnson-Wood K, Mcconlogue L. High-level neuronal expression of Abeta 1-42 in wild-type human amyloid protein precursor transgenic mice: synaptotoxicity without plaque formation. J Neurosci. 2000;20:4050. doi: 10.1523/JNEUROSCI.20-11-04050.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Palop J, Jones B, Kekonius L, Chin J, Yu G, Raber J, Masliah E, Mucke L. Neuronal depletion of calcium-dependent proteins in the dentate gyrus is tightly linked to Alzheimer’s disease-related cognitive deficits. Proc Natl Acad Sci U S A. 2003;100:9572. doi: 10.1073/pnas.1133381100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shankar GM, Leissring MA, Adame A, Sun X, Spooner E, Masliah E, Selkoe DJ, Lemere CA, Walsh DM. Biochemical and immunohistochemical analysis of an Alzheimer’s disease mouse model reveals the presence of multiple cerebral Aβ assembly forms throughout life. Neurobiol Dis. 2009;36:293–302. doi: 10.1016/j.nbd.2009.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Harris JA, Devidze N, Halabisky B, Lo I, Thwin MT, Yu G-Q, Bredesen DE, Masliah E, Mucke L. Many neuronal and behavioral impairments in transgenic mouse models of Alzheimer’s disease are independent of caspase cleavage of the amyloid precursor protein. J Neurosci. 2010;30:372–381. doi: 10.1523/JNEUROSCI.5341-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tomiyama T, Matsuyama S, Iso H, Umeda T, Takuma H, Ohnishi K, Ishibashi K, Teraoka R, Sakama N, Yamashita T. A mouse model of amyloid {beta} oligomers: their contribution to synaptic alteration, abnormal tau phosphorylation, glial activation, and neuronal loss in vivo. J Neurosci. 2010;30:4845. doi: 10.1523/JNEUROSCI.5825-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Benoit ME, Hernandez MX, Dinh ML, Benavente F, Vasquez O, Tenner AJ. C1q-induced LRP1B and GPR6 proteins expressed early in Alzheimer disease mouse models, are essential for the C1q-mediated protection against amyloid-β neurotoxicity. J Biol Chem. 2013;288:654–665. doi: 10.1074/jbc.M112.400168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wyss-Coray T. Prominent neurodegeneration and increased plaque formation in complement-inhibited Alzheimer’s mice. Proc Natl Acad Sci U S A. 2002;99:10837–10842. doi: 10.1073/pnas.162350199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fonseca MI, Zhou J, Botto M, Tenner AJ. Absence of C1q leads to less neuropathology in transgenic mouse models of Alzheimer’s disease. J Neurosci. 2004;24:6457–6465. doi: 10.1523/JNEUROSCI.0901-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Maier M, Peng Y, Jiang L, Seabrook TJ, Carroll MC, Lemere CA. Complement C3 deficiency leads to accelerated amyloid beta plaque deposition and neurodegeneration and modulation of the microglia/macrophage phenotype in amyloid precursor protein transgenic mice. J Neurosci. 2008;28:6333–6341. doi: 10.1523/JNEUROSCI.0829-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Stephan AH, Barres BA, Stevens B. The complement system: an unexpected role in synaptic pruning during development and disease. Annu Rev Neurosci. 2012;35:369–389. doi: 10.1146/annurev-neuro-061010-113810. [DOI] [PubMed] [Google Scholar]
- 52.Gasque P. Complement a unique innate immune sensor for danger signals. Mol Immunol. 2004;41:1089–1098. doi: 10.1016/j.molimm.2004.06.011. [DOI] [PubMed] [Google Scholar]
- 53••.Stephan AH, Madison DV, Mateos JM, Fraser DA, Lovelett EA, Coutellier L, Kim L, Tsai HH, Huang EJ, Rowitch DH, et al. A dramatic increase of C1q protein in the CNS during normal aging. J Neurosci. 2013;33:13460–13474. doi: 10.1523/JNEUROSCI.1333-13.2013. This paper demonstrates for the first time a significant increase of C1q with aging, particularly in the hippocampus where it deposits near synapses. Aged C1q-deficient mice showed enhanced synaptic plasticity and performed better in memory tests compared to wild-type controls. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54••.Shi Q, Colodner KJ, Matousek SB, Merry K, Hong S, Kenison JE, Frost JL, Le KX, Li S, Dodart J-C, et al. Complement C3-deficient mice fail to display age-related hippocampal decline. J Neurosci. 2015;35:13029–13042. doi: 10.1523/JNEUROSCI.1698-15.2015. Wild-type mice show age-related synaptic and neuronal loss in the hippocampus along with cognitive decline; however, aged C3-deficient mice fail to show such age-related effects. This paper suggests that C3 may play a central role in normal age-related synaptic decline. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hong S, Nfonoyim B, Merry K, Ramakrishnan S, Shi Q, Barres BA, Lemere C, Selkoe D, Stevens B. Classical complement cascade mediates early synapse loss in ad mouse models. Nanosymposium Session Presented at: SFN 2014 44th Annual Meeting of the Society for Neuroscience; Washington, DC. 2014. [Google Scholar]
- 56.Ulrich JD, Finn MB, Wang Y, Shen A, Mahan TE, Jiang H, Stewart FR, Piccio L, Colonna M, Holtzman DM. Altered microglial response to Aβ plaques in APPPS1-21 mice heterozygous for TREM2. Mol Neurodegener. 2014;9:20. doi: 10.1186/1750-1326-9-20. The authors demonstrate for the first time that loss of a single Trem2 allele failed to affect Aβ plaque burden in an AD mouse model. However, there was a substantial decrease in plaque-associated microglia, suggesting that TREM2 may affect microglia response to Aβ pathology. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57•.Kleinberger G, Yamanishi Y, Suarez-Calvet M, Czirr E, Lohmann E, Cuyvers E, Struyfs H, Pettkus N, Wenninger-Weinzierl A, Mazaheri F, et al. TREM2 mutations implicated in neurodegeneration impair cell surface transport and phagocytosis. Sci Transl Med. 2014;6:243ra86. doi: 10.1126/scitranslmed.3009093. This paper demonstrates for the first time that mutations associated with neurodegenerative diseases may prevent maturation, transport, and shedding of TREM2 at cell surface, ultimately leading to reduced microglial phagocytic activity. Interestingly, patients with sporadic frontotemporal dementia and AD showed reduced levels of soluble TREM2 in their CSF. [DOI] [PubMed] [Google Scholar]
- 58•.Jay TR, Miller CM, Cheng PJ, Graham LC, Bemiller S, Broihier ML, Xu G, Margevicius D, Karlo JC, Sousa GL, et al. TREM2 deficiency eliminates TREM2+ inflammatory macrophages and ameliorates pathology in Alzheimer’s disease mouse models. J Exp Med. 2015;212:287–295. doi: 10.1084/jem.20142322. The authors demonstrate that cells surround Aβ plaques in AD mouse models and human AD tissue express TREM2 and that these cells are more likely immune cells infiltrating from the periphery than resident parenchymal microglia. Depletion of Trem2 is associated with reduced infiltration of immune cells in the diseased CNS, along with reduced inflammation and amyloid and tau pathologies. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59••.Wang Y, Cella M, Mallinson K, Ulrich JD, Young KL, Robinette ML, Gilfillan S, Krishnan GM, Sudhakar S, Zinselmeyer BH, et al. TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell. 2015 doi: 10.1016/j.cell.2015.01.049. http://dx.doi.org/10.1016/j.cell.2015.01.049 This paper suggests a novel function of TREM2 by demonstrating that TREM2 acts as a sensor of a broad array of anionic and zwitterionic lipids known to associate with Aβ on damaged lipid membranes. Importantly, this lipid sensing seems to be impaired in the R47H Trem2 mutation that increases risk for AD. Similar to Ulrich et al., the authors report that Trem2 deficiency leads to a dysfunctional microglial response around plaques. [DOI] [PMC free article] [PubMed]
- 60.Colonna M. TREMs in the immune system and beyond. Nat Rev Immunol. 2003;3:445–453. doi: 10.1038/nri1106. [DOI] [PubMed] [Google Scholar]
- 61.Paloneva J, Kestilä M, Wu J, Salminen A, Böhling T, Ruotsalainen V, Hakola P, Bakker AB, Phillips JH, Pekkarinen P, et al. Loss-of-function mutations in TYROBP (DAP12) result in a presenile dementia with bone cysts. Nat Genet. 2000;25:357–361. doi: 10.1038/77153. [DOI] [PubMed] [Google Scholar]
- 62.Paloneva J, Manninen T, Christman G, Hovanes K, Mandelin J, Adolfsson R, Bianchin M, Bird T, Miranda R, Salmaggi A, et al. Mutations in Two genes encoding different subunits of a receptor signaling complex result in an identical disease phenotype. Am J Hum Genet. 2002;71:656–662. doi: 10.1086/342259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Roumier A, Béchade C, Poncer J-C, Smalla K-H, Tomasello E, Vivier E, Gundelfinger ED, Triller A, Bessis A. Impaired synaptic function in the microglial KARAP/DAP12-deficient mouse. J Neurosci. 2004;24:11421–11428. doi: 10.1523/JNEUROSCI.2251-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hong S, Ostaszewski BL, Yang T, O’Malley TT, Jin M, Yanagisawa K, Li S, Bartels T, Selkoe DJ. Soluble Aβ oligomers are rapidly sequestered from brain ISF in vivo and bind GM1 ganglioside on cellular membranes. Neuron. 2014 doi: 10.1016/j.neuron.2014.02.027. http://dx.doi.org/10.1016/j.neuron.2014.02.027. [DOI] [PMC free article] [PubMed]
- 65.Ehrnhoefer DE, Wong BKY, Hayden MR. Convergent pathogenic pathways in Alzheimer’s and Huntington’s diseases: shared targets for drug development. Nat Rev Drug Discov. 2011;10:853–867. doi: 10.1038/nrd3556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Faludi G, Mirnics K. Synaptic changes in the brain of subjects with schizophrenia. Int J Dev Neurosci. 2011;29:305–309. doi: 10.1016/j.ijdevneu.2011.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Palop JJ, Chin J, Mucke L. A network dysfunction perspective on neurodegenerative diseases. Nature. 2006;443:768–773. doi: 10.1038/nature05289. [DOI] [PubMed] [Google Scholar]
- 68.Seeley WW, Crawford RK, Zhou J, Miller BL, Greicius MD. Neurodegenerative diseases target large-scale human brain networks. Neuron. 2009;62:42–52. doi: 10.1016/j.neuron.2009.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Palop JJ, Chin J, Roberson ED, Wang J, Thwin MT, Bien-Ly N, Yoo J, Ho KO, Yu G-Q, Kreitzer A, et al. Aberrant excitatory neuronal activity and compensatory remodeling of inhibitory hippocampal circuits in mouse models of Alzheimer’s disease. Neuron. 2007;55:697–711. doi: 10.1016/j.neuron.2007.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Edison P, Archer HA, Gerhard A, Hinz R, Pavese N, Turkheimer FE, Hammers A, Tai YF, Fox N, Kennedy A, et al. Microglia, amyloid, and cognition in Alzheimer’s disease: an [11C](R)PK11195-PET and [11C]PIB-PET study. Neurobiol Dis. 2008;32:412–419. doi: 10.1016/j.nbd.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 71.Sperling RA, Laviolette PS, O’Keefe K, O’Brien J, Rentz DM, Pihlajamaki M, Marshall G, Hyman BT, Selkoe DJ, Hedden T, et al. Amyloid deposition is associated with impaired default network function in older persons without dementia. Neuron. 2009;63:178–188. doi: 10.1016/j.neuron.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Palop JJ, Mucke L. Amyloid-β-induced neuronal dysfunction in Alzheimer’s disease: from synapses toward neural networks. Nat Neurosci. 2010;13:812–818. doi: 10.1038/nn.2583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Putcha D, Brickhouse M, O’Keefe K, Sullivan C, Rentz D, Marshall G, Dickerson B, Sperling R. Hippocampal hyperactivation associated with cortical thinning in Alzheimer’s disease signature regions in non-demented elderly adults. J Neurosci. 2011;31:17680–17688. doi: 10.1523/JNEUROSCI.4740-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Bero AW, Yan P, Roh JH, Cirrito JR, Stewart FR, Raichle ME, Lee J-M, Holtzman DM. Neuronal activity regulates the regional vulnerability to amyloid-[beta] deposition. Nat Neurosci. 2011 doi: 10.1038/nn.2801. http://dx.doi.org/10.1038/nn.2801. [DOI] [PMC free article] [PubMed]
- 75.Buckner RL, Snyder AZ, Shannon BJ, LaRossa G, Sachs R, Fotenos AF, Sheline YI, Klunk WE, Mathis CA, Morris JC, Mintun MA. Molecular, structural, and functional characterization of Alzheimer’s disease: evidence for a relationship between default activity, amyloid, and memory. J Neurosci. 2005;25:7709–7717. doi: 10.1523/JNEUROSCI.2177-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kamenetz F, Tomita T, Hsieh H, Seabrook G, Borchelt D, Iwatsubo T, Sisodia S, Malinow R. APP processing and synaptic function. Neuron. 2003;37:925–937. doi: 10.1016/s0896-6273(03)00124-7. [DOI] [PubMed] [Google Scholar]
- 77.Cirrito JR, Yamada KA, Finn MB, Sloviter RS, Bales KR, May PC, Schoepp DD, Paul SM, Mennerick S, Holtzman DM. Synaptic activity regulates interstitial fluid amyloid-beta levels in vivo. Neuron. 2005;48:913–922. doi: 10.1016/j.neuron.2005.10.028. [DOI] [PubMed] [Google Scholar]