Abstract
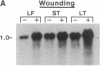
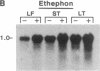
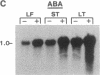
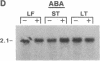
Hevein is a chitin-binding protein that is present in laticifers of the rubber tree (Hevea brasiliensis). A cDNA clone (HEV1) encoding hevein was isolated by using the polymerase chain reaction with mixed oligonucleotides corresponding to two regions of hevein as primers and a Hevea latex cDNA library as a template. HEV1 is 1018 base pairs long and includes an open reading frame of 204 amino acids. The deduced amino acid sequence contains a putative signal sequence of 17 amino acid residues followed by a 187-amino acid polypeptide. This polypeptide has two striking features. The amino-terminal region (43 amino acids) is identical to hevein and shows homology to several chitin-binding proteins and to the amino termini of wound-inducible proteins in potato and poplar. The carboxyl-terminal portion of the polypeptide (144 amino acids) is 74-79% homologous to the carboxyl-terminal region of wound-inducible genes of potato. Wounding, as well as application of the plant hormones abscisic acid and ethylene, resulted in accumulation of hevein transcripts in leaves, stems, and latex but not in roots.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bohlmann H., Clausen S., Behnke S., Giese H., Hiller C., Reimann-Philipp U., Schrader G., Barkholt V., Apel K. Leaf-specific thionins of barley-a novel class of cell wall proteins toxic to plant-pathogenic fungi and possibly involved in the defence mechanism of plants. EMBO J. 1988 Jun;7(6):1559–1565. doi: 10.1002/j.1460-2075.1988.tb02980.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boutry M., Chua N. H. A nuclear gene encoding the beta subunit of the mitochondrial ATP synthase in Nicotiana plumbaginifolia. EMBO J. 1985 Sep;4(9):2159–2165. doi: 10.1002/j.1460-2075.1985.tb03910.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broekaert W. F., VAN Parijs J., Leyns F., Joos H., Peumans W. J. A chitin-binding lectin from stinging nettle rhizomes with antifungal properties. Science. 1989 Sep 8;245(4922):1100–1102. doi: 10.1126/science.245.4922.1100. [DOI] [PubMed] [Google Scholar]
- Broglie K. E., Gaynor J. J., Broglie R. M. Ethylene-regulated gene expression: molecular cloning of the genes encoding an endochitinase from Phaseolus vulgaris. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6820–6824. doi: 10.1073/pnas.83.18.6820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dale R. M., Arrow A. A rapid single-stranded cloning, sequencing, insertion, and deletion strategy. Methods Enzymol. 1987;155:204–214. doi: 10.1016/0076-6879(87)55017-0. [DOI] [PubMed] [Google Scholar]
- Ecker J. R., Davis R. W. Plant defense genes are regulated by ethylene. Proc Natl Acad Sci U S A. 1987 Aug;84(15):5202–5206. doi: 10.1073/pnas.84.15.5202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. "A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity". Addendum. Anal Biochem. 1984 Feb;137(1):266–267. doi: 10.1016/0003-2697(84)90381-6. [DOI] [PubMed] [Google Scholar]
- Gubler U., Hoffman B. J. A simple and very efficient method for generating cDNA libraries. Gene. 1983 Nov;25(2-3):263–269. doi: 10.1016/0378-1119(83)90230-5. [DOI] [PubMed] [Google Scholar]
- Hedrick S. A., Bell J. N., Boller T., Lamb C. J. Chitinase cDNA cloning and mRNA induction by fungal elicitor, wounding, and infection. Plant Physiol. 1988 Jan;86(1):182–186. doi: 10.1104/pp.86.1.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henstrand J. M., Handa A. K. Effect of Ethylene Action Inhibitors upon Wound-Induced Gene Expression in Tomato Pericarp. Plant Physiol. 1989 Sep;91(1):157–162. doi: 10.1104/pp.91.1.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirk M. M., Kirk D. L. Translational regulation of protein synthesis, in response to light, at a critical stage of Volvox development. Cell. 1985 Jun;41(2):419–428. doi: 10.1016/s0092-8674(85)80015-5. [DOI] [PubMed] [Google Scholar]
- Kush A., Goyvaerts E., Chye M. L., Chua N. H. Laticifer-specific gene expression in Hevea brasiliensis (rubber tree). Proc Natl Acad Sci U S A. 1990 Mar;87(5):1787–1790. doi: 10.1073/pnas.87.5.1787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lerner D. R., Raikhel N. V. Cloning and characterization of root-specific barley lectin. Plant Physiol. 1989 Sep;91(1):124–129. doi: 10.1104/pp.91.1.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizusawa S., Nishimura S., Seela F. Improvement of the dideoxy chain termination method of DNA sequencing by use of deoxy-7-deazaguanosine triphosphate in place of dGTP. Nucleic Acids Res. 1986 Feb 11;14(3):1319–1324. doi: 10.1093/nar/14.3.1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons T. J., Bradshaw H. D., Jr, Gordon M. P. Systemic accumulation of specific mRNAs in response to wounding in poplar trees. Proc Natl Acad Sci U S A. 1989 Oct;86(20):7895–7899. doi: 10.1073/pnas.86.20.7895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pēna-Cortés H., Sánchez-Serrano J. J., Mertens R., Willmitzer L., Prat S. Abscisic acid is involved in the wound-induced expression of the proteinase inhibitor II gene in potato and tomato. Proc Natl Acad Sci U S A. 1989 Dec;86(24):9851–9855. doi: 10.1073/pnas.86.24.9851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raikhel N. V., Wilkins T. A. Isolation and characterization of a cDNA clone encoding wheat germ agglutinin. Proc Natl Acad Sci U S A. 1987 Oct;84(19):6745–6749. doi: 10.1073/pnas.84.19.6745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinshi H., Mohnen D., Meins F. Regulation of a plant pathogenesis-related enzyme: Inhibition of chitinase and chitinase mRNA accumulation in cultured tobacco tissues by auxin and cytokinin. Proc Natl Acad Sci U S A. 1987 Jan;84(1):89–93. doi: 10.1073/pnas.84.1.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silflow C. D., Hammett J. R., Key J. L. Sequence complexity of polyadenylated ribonucleic acid from soybean suspension culture cells. Biochemistry. 1979 Jun 26;18(13):2725–2731. doi: 10.1021/bi00580a006. [DOI] [PubMed] [Google Scholar]
- Stanford A., Bevan M., Northcote D. Differential expression within a family of novel wound-induced genes in potato. Mol Gen Genet. 1989 Jan;215(2):200–208. doi: 10.1007/BF00339718. [DOI] [PubMed] [Google Scholar]
- Vieira J., Messing J. Production of single-stranded plasmid DNA. Methods Enzymol. 1987;153:3–11. doi: 10.1016/0076-6879(87)53044-0. [DOI] [PubMed] [Google Scholar]
- Wilkins T. A., Raikhel N. V. Expression of rice lectin is governed by two temporally and spatially regulated mRNAs in developing embryos. Plant Cell. 1989 May;1(5):541–549. doi: 10.1105/tpc.1.5.541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright C. S., Gavilanes F., Peterson D. L. Primary structure of wheat germ agglutinin isolectin 2. Peptide order deduced from X-ray structure. Biochemistry. 1984 Jan 17;23(2):280–287. doi: 10.1021/bi00297a017. [DOI] [PubMed] [Google Scholar]
- von Heijne G. Signal sequences. The limits of variation. J Mol Biol. 1985 Jul 5;184(1):99–105. doi: 10.1016/0022-2836(85)90046-4. [DOI] [PubMed] [Google Scholar]