Abstract
Using population-based surveillance data, we analyzed antiviral treatment among hospitalized patients with laboratory-confirmed influenza. Treatment increased after the influenza A(H1N1) 2009 pandemic from 72% in 2010–2011 to 89% in 2014–2015 (P < .001). Overall, treatment was higher in adults (86%) than in children (72%); only 56% of cases received antivirals on the day of admission.
Keywords: influenza, antiviral, oseltamivir
Early antiviral treatment can reduce influenza-associated complications including lower respiratory tract infections [1, 2] and death in hospitalized patients [3, 4]. Antivirals have been recommended for all hospitalized patients with suspected or confirmed influenza since before the influenza A(H1N1)2009 pandemic [5]. During the 2014–2015 influenza season, because of detection of drifted influenza A(H3N2) viruses, the Centers for Disease Control and Prevention (CDC) issued an advisory to reemphasize the importance of early antiviral treatment. While the frequency of antiviral treatment in hospitalized patients with laboratory-confirmed influenza increased to approximately 80% in children and adults during the 2009 pandemic compared with a range of 37%–57% in prior seasons, a significant decline was observed in 2010–2011, most notably to 52% among children aged <2 years [6, 7]. To compare patterns of antiviral treatment in recent seasons and evaluate antiviral use in patients at high risk of developing influenza-associated complications, we analyzed population-based surveillance data of hospitalized patients with laboratory-confirmed influenza during the 2010–2011 through 2014–2015 influenza seasons.
METHODS
Data were collected prospectively from 1 October to 31 April each season from all patients hospitalized with laboratory-confirmed influenza as part of the Influenza Hospitalization Surveillance Network (FluSurv-NET), a national population-based influenza-associated hospitalization surveillance system. Cases were defined as residents of a catchment area admitted to a hospital ≤14 days after a positive influenza test [8]. The catchment area included 267 acute care hospitals and laboratories in California, Colorado, Connecticut, Georgia, Iowa, Maryland, Michigan, Minnesota, New Mexico, New York, Ohio, Oklahoma, Oregon, Rhode Island, Tennessee, and Utah, and represented approximately 9% of the US population, with distribution of age, sex, race/ethnicity, and health indicators similar to that of the overall population [8].
Laboratory-confirmed influenza cases were identified from laboratory logs, infection preventionist databases, hospital discharge summaries, or reportable conditions databases at surveillance sites [8]. Surveillance officers reviewed medical records to obtain information about treatment, vaccination, and medical conditions that confer a higher risk of developing influenza-associated complications, including organ disease, chronic metabolic disease, blood disorders, immunosuppression, morbid obesity, and pregnancy (Supplementary Table 1) [9].
Hospital laboratory testing for influenza was performed at clinician discretion by rapid influenza diagnostic test (RIDT), viral culture, fluorescent antibody staining, and/or reverse transcription polymerase chain reaction (RT-PCR). Because RT-PCR testing of specimens with negative RIDT results may have been performed subsequently at public health laboratories as part of surveillance alone and not for clinical care, not all patients with negative RIDT and positive PCR results may have been known to the clinician in real time. These patients were excluded because we were unable to identify timing of the PCR results. Patients with missing data on the use or timing of initiation of antiviral treatment were also excluded.
We compared characteristics of treated and untreated patients and described the timing of antiviral treatment in relation to illness onset and hospital admission. The earliest treatment date was used for patients with multiple treatment dates. We defined prompt antiviral treatment as initiation within 2 days of admission (ie, on day of admission [day 0] or day 1). Admission within 2 days of respiratory illness onset was similarly defined as admission on day 0 or day 1 of illness.
The χ2 test and Cochran-Armitage test for trend were used to assess the association between antiviral treatment and clinical characteristics, and treatment over time, respectively, using SAS software (version 9.3).
RESULTS
During the 2010–2011 through 2014–2015 seasons, 48 456 laboratory-confirmed influenza cases were identified in FluSurv-NET (Supplementary Figure 1). We included patients who received antiviral treatment prior to hospitalization (n = 1238), of whom 775 (63%) received antivirals within 1 day prior to admission. To exclude potential nosocomial infections, we excluded patients with positive influenza test >3 days after admission (n = 1358; Supplementary Figure 1). An additional 3145 patients with negative RIDT and positive PCR results and 245 with missing antiviral data were excluded. Of the remaining 43 708 with data available on antiviral use, 36 780 (84%) received antivirals over the 5 combined seasons. Overall, 32 124 of 37 239 (86%) adults (≥18 years) received antivirals compared with 4656 of 6469 (72%) children (<18 years) (P < .001). Almost all treated patients (36 685 [>99%]) received oseltamivir.
Antiviral treatment significantly increased during the study period: 72% in 2010–2011, 75% in 2011–2012, 83% in 2012–2013, 87% in 2013–2014, and 89% in 2014–2015 (P for trend <.001) (Figure 1). The percentage treated increased across every season for almost all ages. The only significant declines were among 2- to 4-year olds, by 19% from 2010–2011 to 2011–2012 (P = .002), and 8% from 2013–2014 to 2014–2015 (P = .03). Children aged <1 year had the greatest overall treatment increase across seasons, from 51% to 82%. Treatment among adults aged 18–64 years remained at or above 77% across all seasons and by 2014–2015 approached 90%.
Figure 1.
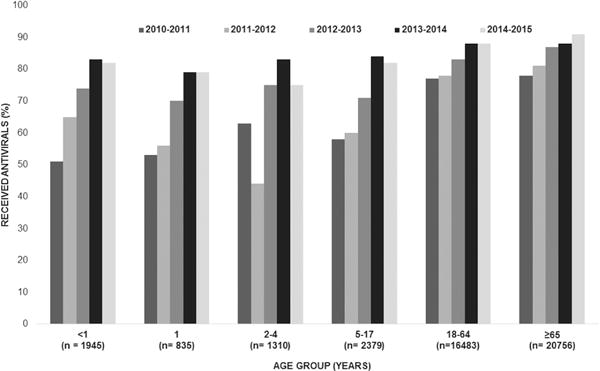
Antiviral treatment in patients hospitalized with laboratory-confirmed influenza, by age group and influenza season, 2010–2011 to 2014–2015 (n = 43 708). Test for trend across the 5 seasons was significant in all age groups (P < .001).
Among 35 393 patients with high-risk conditions, 30 407 (86%) received antivirals compared with 6373 of 8315 (77%) without (P < .001). Across all seasons, treatment increased over time from 76% to 90% in patients with high-risk conditions (P for trend <.001). Among 3051 children aged <18 years with high-risk conditions, 2346 (77%) were treated compared with 28 061 of 32 342 (87%) adults with high-risk conditions (P < .001) (Supplementary Table 1). Among treated patients, 83% had a high-risk condition compared with 72% of those not treated (P < .001) (Supplementary Table 1).
Of 40 868 patients with illness dates available (Supplementary Figure 1), 12 629 (31%) were admitted within 2 days of illness onset, 19 608 (48%) on days 2–4, and 8631 (21%) after day 4. Among patients admitted within 2 days of illness onset, and on days 2–4 of illness, 11 032 (87%) and 16 845 (86%) received antivirals, respectively. Of patients admitted after day 4 from illness, 6559 (76%) of patients were treated.
Among treated patients, a total of 34 112 had treatment dates and 19,193 (56%) were treated on the day of hospital admission. (Supplementary Figure 1). A total of 10 937 patients were admitted within 2 days of illness onset; 208 (2%) were treated prior to admission, 5977 (55%) on the day of admission, and 3456 (32%) on the day after admission (Supplementary Figure 2). Among 16 700 patients admitted on days 2–4 of illness, 9900 (59%) and 4710 (28%) were treated on the day of admission and the day after admission, respectively.
DISCUSSION
Antiviral treatment of hospitalized patients of all ages with laboratory-confirmed influenza significantly increased from 2010–2011 to 2014–2015 among participating FluSurv-NET hospitals. While use of antiviral treatment started at nearly 80% in 2010–2011, it increased over time to nearly 90% among adults, and improved from 51% to 82% among children <1 year old. Among infants, treatment increased the most across the 5 seasons and remained constant over the last 2 seasons.
Antiviral treatment in infants expanded during the 2009 pandemic, when oseltamivir was used for treatment in children aged <1 year under an Emergency Use Authorization (EUA) [9]. Given the high risk of complications in this group, oseltamivir continued to be recommended by the CDC and the American Academy of Pediatrics for children aged <1 year after the EUA expiration in June 2010. The US Food and Drug Administration approved oseltamivir for use in children <1 year in December 2012.
Despite the recommendation for early antiviral treatment for hospitalized patients with suspected or confirmed influenza, 87% of patients admitted within 2 days of illness onset were treated in that same early time window, but only 55% received treatment on the day of admission. Early antiviral treatment among hospitalized patients is associated with reduced mortality in adults and shortened length of stay in children and older adults [3, 4, 10]. The earlier treatment is initiated in the patient’s illness, the better the clinical outcomes [4, 11]. More than two-thirds of patients were admitted to the hospital ≥2 days after illness onset. Although clinical benefit is greatest when antiviral treatment is initiated <2 days from illness onset, some studies suggest that treatment may provide benefit in hospitalized patients even when started 4–5 days after onset [3, 4].
Our analysis had potential limitations. Influenza testing was performed based on clinician discretion. Clinicians may have preferentially tested patients for influenza who were admitted shortly after illness onset. In a study of adults aged ≥50 years hospitalized with acute respiratory illness during the 2006–2012 influenza seasons, only 26% had provider-initiated influenza testing and 26% of patients with laboratory-confirmed influenza received treatment [12]. Our findings may overestimate antiviral treatment of patients hospitalized with influenza as we cannot address the extent to which treatment occurred in patients who were not tested.
Our findings demonstrate that antiviral treatment of hospitalized children and adults with laboratory-confirmed influenza significantly increased from 2010–2011 to 2014–2015. Although these gains are encouraging, 11% of hospitalized patients with laboratory-confirmed influenza remained untreated in 2014–2015. This may be partly explained by delays to hospital admission relative to symptom onset, as only one-third of patients were admitted within 2 days after symptoms began. Improving initiation of antiviral therapy among high-risk outpatients may provide additional benefits for patients seeking care early in illness. Availability and use of sensitive, point-of-care influenza testing in the ambulatory setting, including clinics and emergency departments, would increase timely provider diagnosis of influenza. Additional studies are needed to understand barriers to influenza antiviral treatment and increase optimal use of antivirals in hospitalized patients with influenza, especially among children.
Supplementary Material
Acknowledgments
We thank the following individuals for their expertise and assistance with statistics, study coordination and management, and/or surveillance efforts: Sue Reynolds, Jessie Chung, Michelle De Leon, Jessica Cohen, and Charisse Nitura in the CDC Influenza Division; Kyle Openo, and Monica M. Farley, in the Georgia Emerging Infections Program; Elizabeth M. Mermel, in the Rhode Island Department of Health; Kimberly Yousey-Hindes, Tamara Rissman, and Amber Maslar in the Yale Emerging Infections Program; Nancy Spina, from New York State Department of Health, Albany; Deborah Aragon in the Colorado Department of Health; Karen Leib, Gail Hughett, Katie Dyer, and Danielle Ndi from the Tennessee Emerging Infections Program; Craig Morin, Melissa McMahon, Sara Vetter, Anna Strain, and Dave Boxrud from the Minnesota Department of Health; Erin Parker, Lauren Pasutti, and Ashley Coates, from the California Emerging Infections Program.
Financial support
FluSurv-NET is a collaboration of state health departments, academic institutions, and local partners and is supported by the Centers for Disease Control and Prevention (cooperative agreements CDC-RFA-CK12-1202 and 5U38HM000414).
Footnotes
Supplementary Data
Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the author to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the author, so questions or comments should be addressed to the author.
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the CDC.
Potential conflicts of interest
All authors: No reported conflicts. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Dobson J, Whitley RJ, Pocock S, Monto AS. Oseltamivir treatment for influenza in adults: a meta-analysis of randomised controlled trials. Lancet. 2015;385:1729–37. doi: 10.1016/S0140-6736(14)62449-1. [DOI] [PubMed] [Google Scholar]
- 2.Lipsitch M, Hernán MA. Oseltamivir effect on antibiotic-treated lower respiratory tract complications in virologically positive randomized trial participants. Clin Infect Dis. 2013;57:1368–9. doi: 10.1093/cid/cit481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Louie JK, Yang S, Acosta M, et al. Treatment with neuraminidase inhibitors for critically ill patients with influenza A (H1N1)pdm09. Clin Infect Dis. 2012;55:1198–204. doi: 10.1093/cid/cis636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Muthuri SG, Venkatesan S, Myles PR, et al. PRIDE Consortium Investigators Effectiveness of neuraminidase inhibitors in reducing mortality in patients admitted to hospital with influenza A H1N1pdm09 virus infection: a meta-analysis of individual participant data. Lancet Respir Med. 2014;2:395–404. doi: 10.1016/S2213-2600(14)70041-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Harper SA, Bradley JS, Englund JA, et al. Expert Panel of the Infectious Diseases Society of America Seasonal influenza in adults and children—diagnosis, treatment, chemoprophylaxis, and institutional outbreak management: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis. 2009;48:1003–32. doi: 10.1086/604670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Doshi S, Kamimoto L, Finelli L, et al. Description of antiviral treatment among adults hospitalized with influenza before and during the 2009 pandemic: United States, 2005–2009. J Infect Dis. 2011;204:1848–56. doi: 10.1093/infdis/jir648. [DOI] [PubMed] [Google Scholar]
- 7.Garg S, Chaves SS, Pérez A, et al. Reduced influenza antiviral treatment among children and adults hospitalized with laboratory-confirmed influenza infection in the year after the 2009 pandemic. Clin Infect Dis. 2012;55:e18–21. doi: 10.1093/cid/cis442. [DOI] [PubMed] [Google Scholar]
- 8.Chaves SS, Lynfield R, Lindegren ML, Bresee J, Finelli L. The US Influenza Hospitalization Surveillance Network. Emerg Infect Dis. 2015;21:1543–50. doi: 10.3201/eid2109.141912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fiore AE, Fry A, Shay D, Gubareva L, Bresee JS, Uyeki TM, Centers for Disease Control and Prevention (CDC) Antiviral agents for the treatment and chemoprophylaxis of influenza—recommendations of the Advisory Committee on Immunization Practices (ACIP) MMWR Recomm Rep. 2011;60:1–24. [PubMed] [Google Scholar]
- 10.Chaves SS, Pérez A, Miller L, et al. Impact of prompt influenza antiviral treatment on extended care needs after influenza hospitalization among community-dwelling older adults. Clin Infect Dis. 2015;61:1807–14. doi: 10.1093/cid/civ733. [DOI] [PubMed] [Google Scholar]
- 11.Aoki FY, Macleod MD, Paggiaro P, et al. IMPACT Study Group Early administration of oral oseltamivir increases the benefits of influenza treatment. J Antimicrob Chemother. 2003;51:123–9. doi: 10.1093/jac/dkg007. [DOI] [PubMed] [Google Scholar]
- 12.Lindegren ML, Griffin MR, Williams JV, et al. Antiviral treatment among older adults hospitalized with influenza, 2006–2012. PLoS One. 10:e0121952. doi: 10.1371/journal.pone.0121952. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


