Abstract
Background
Green tea contains numerous polyphenols, which have health-promoting effects. The purpose of this study was to evaluate the effect of tannase-converted green tea extract (TGE) formulation on the physical stability and activities of skin-related enzymes.
Methods
Physical stability was evaluated by measuring the pH, precipitation, and colors at 25 ± 2 °C/ambient humidity and at 40 ± 2 °C/70% ± 5% relative humidity for 4 months. Activities of collagenase, elastase, and tyrosinase as skin-related enzymes were assessed on TGE formulation.
Results
The concentrations of epigallocatechin-3-gallate and epicatechin-3-gallate in green tea extract were greatly decreased to the extent of negligible level when treated with tannase. The formulation containing 5% tannase-converted green tea extract showed relatively stable pH, precipitation, and color features for 16 weeks. When TGE was added to the formulation, there was a significant increase in the inhibition of elastase and tyrosinase activities (p < 0.05) compared with the formulation containing 5% normal green tea extract.
Conclusion
The TGE could be used in cosmetics as skin antiwrinkling or depigmenting agent.
Keywords: elastase, green tea, stability, tannase, tyrosinase
1. Introduction
Facial appearance is one of the important factors influencing social relations in many situations. People tend to judge their personality based on facial appearance.1 Cosmeceuticals, which are applied to skin, are widely used to improve such an appearance. Ingredients in cosmeceuticals should penetrate the skin and become systemically available to be effective.2 Therefore, the research and development for cosmeceuticals should not only include sources, structures, skin-interactive mechanisms of active ingredients, but also their safety and efficacy on the targeted components of skin.3
Plant extracts have been widely used as ingredients of topical agents for wound healing and antiaging. Some of the commonly used plants for this purpose are ginkgo, ginseng, grape seeds, papaya, citrus fruits (e.g., lemon), lavender, rosemary, soy, aloe vera, and green tea, etc.4 Plant extracts usually contain polyphenols such as flavonoids, which react with reactive oxygen species (ROS) to neutralize free radicals.5 At present, there is a strong tendency in the cosmetic industry to develop multifunctional cosmetics with high antioxidant activity. According to the oxidative stress theory, the major causes of skin aging are an excessive production of ROS6 and a reduction of antioxidant activity with age.
Catechins, the polyphenols in green tea, have beneficial effects on human health.7 In recent years, they have received a great deal of attention due to their potent antioxidant activities,8 and it was reported that green tea extract can be used to help treat hypercholesterolemia, and protect individuals against disorders caused by the aging process.9 Moreover, several studies have reported that the catechins in green tea inhibit the activities of collagenase10 and tyrosinase activity, subsequently improving skin health.11
Tannase, also known as tannin acyl hydrolase (EC 3.1.1.20), is a kind of inducible enzyme produced by fungi, yeast, and bacteria. Tannase has mostly been characterized by its hydrolyzing activity on the ester bond (galloyl ester of an alcohol moiety) and the depside bond [galloyl ester of gallic acid (GA)] of substrates such as tannic acid, (–)-epigallocatechin-3-gallate (EGCG), (–)-epicatechin-3-gallate (ECG), and chlorogenic acid. Lu and Chen12 reported that the tannase-derived bioconversion on catechins compositions in green tea enhanced the scavenging ability on radicals, such as superoxide anions, and hydrogen peroxide. It has also been reported that the antioxidant activities and chelation of metal ions are improved by tannase treatment.12 However, only limited studies are available on the physical stability and physiological effects of a tannase-converted green tea extract (TGE) formulation.
In this study, we investigated the stability of a TGE formulation and its effect on collagenase, elastase, and tyrosinase activities as major enzyme markers for skin health.
2. Methods
2.1. Preparation of TGE
Green tea leaves were obtained from the Hadong area of Korea. The green tea leaves were ground and pulverized in a mortar, and then mixed with distilled water (DW) for 30 minutes in a 10-L reactor (200 g/4 L). The mixture was incubated at 80 °C for 20 minutes, and centrifuged at 3000 g for 15 minutes at 5 °C. The clear supernatant [normal green tea extract (NGE)] was used for experiments. The green tea extract (4 L) thus prepared was combined with 1 g of tannase (Visionbiochem, Gyeonggi-do, Korea) and then incubated in a water bath at 35 °C for 20 minutes followed by centrifuging at 3000 g for 15 minutes at 5 °C. The supernatant was used as the TGE for further analyses. The compositions of the base formulation (vehicle), the formulation containing 5% NGE (FNGE), and the formulation containing 5% tannase-converted green tea extract (FTGE) are shown in Table 1.
Table 1.
Compositions of cosmetic formulations.
| Compositions (%) | Vehicle | FNGE | FTGE |
|---|---|---|---|
| Deionized water | 61.5 | 56.5 | 56.5 |
| Carbomer (1% solution) | 30 | 30 | 30 |
| Hyaluronic acid | 5 | 5 | 5 |
| Beta glucan | 3 | 3 | 3 |
| Triethanolamine | 0.3 | 0.3 | 0.3 |
| Methylparaben | 0.1 | 0.1 | 0.1 |
| DS-49 | 0.1 | 0.1 | 0.1 |
| NGE | 0 | 5 | 0 |
| TGE | 0 | 0 | 5 |
| Total | 100 | 100 | 100 |
DS-49, disodium-2,2′-dihydroxy-4,4′-dimethoxy benzophenone sulfonic acid; FNGE, formulation containing 5% normal green tea extract; FTGE, formulation containing 5% tannase-converted green tea extract; NGE, normal green tea extract; TGE, tannase-converted green tea extract; vehicle, base formulation.
2.2. Physical stability
The physical stability assay used in this study was based on the method reported by Zhang et al.13 The samples in impermeable polypropylene containers were stored at 25 ± 2 °C with ambient humidity (AH), and at 40 ± 2 °C with 70% ± 5% relative humidity (RH) for 4 months. The pH, precipitation, and color features of samples were evaluated after storage for 1, 2, 3, 4, 8, 12, and 16 weeks at room temperature. Approximately 1 g of the sample was diluted with distilled water up to a volume of 10 mL. This sample mixture was then homogenized. The pH of the samples was measured using a pH meter (Systronics, Inc., Ahmadabad, India). Precipitation was conducted by measuring the supernatant after centrifuging at 3000 g for 30 minutes. The color of samples was measured using a colorimeter (Model CR300; Minolta Camera Co. Ltd. Inc., Osaka, Japan). The colorimeter was calibrated using a Minolta standard-white reflector plate in advance. The data were expressed as L (degree of lightness), a (degree of redness), and b (degree of yellowness) values based on the Hunter color system. The total color difference (ΔE) is calculated as follows:
where L, a, and b are colors of the samples; L′, a′, and b′ are colors of the base at time zero.
2.3. Analysis of total polyphenol and flavonoid contents
Total polyphenol contents were examined using the Folin–Ciocalteu method.14 The reaction mixture includes 0.79 mL of DW, 0.01 mL of sample, and 0.05 mL of Folin–Ciocalteu reagent. After exactly 1 minute, 0.15 mL of 20% sodium carbonate was added to the mixture, followed by standing at room temperature in darkness for 120 minutes. The absorbance was measured at 750 nm, and the total polyphenol content was calculated with GA as the standard. The total flavonoid content was determined by the method of Li et al14 with minor modifications. AlCl3 in 2% ethanol (0.5 mL) was added to 0.5 mL of the sample, and allowed to stand for 1 hour at room temperature. The absorbance was measured at 420 nm. Total flavonoids were calculated with catechins as the standard.
2.4. Inhibition of collagenase and elastase activities
The assay of collagenase inhibitory activity was performed according to the method reported by Van Wart and Steinbrink15 with minor modifications. The 50 mM tricine buffer (pH 7.5) containing 10 mM calcium chloride and 400 mM sodium chloride was prepared. A 50 mL volume of 1.0 mM N-[3-(2-furyl)acryloyl]-Leu–Gly–Pro–Ala solution and collagenase were added in the presence or absence of samples for the reaction. The reaction was stopped by adding citric acid (6%). The reaction mixture was separated by adding ethyl acetate. The absorbance of supernatant was measured at 345 nm. The percentage of inhibition was calculated as follows:
where A is a mixture with collagenase but without sample and B is a mixture with both sample and collagenase.
The inhibitory activity of elastase was evaluated spectrophotometrically by the method reported by Kraunsoe et al.16 N-succinyl–Ala–Ala–Ala–p-nitroanilide (Suc–Ala–Ala–Ala–pNA; Bachem Feinchemikalien AG, Bubendorf, Switzerland) was used as the substrate, and the release of p-nitroaniline was monitored for 20 minutes at 25 °C. A portion (1 μg) of porcine pancreatic elastase (PPE) type IV (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in 1 mL of 0.2 M Tris–HCl buffer (pH 8.0). The reaction mixture contained 0.2 M Tris–HCl buffer (pH 8.0), 1 ppm PPE, 0.8 mM Suc–Ala–Ala–Ala–pNA, the sample, and the aforementioned substrate. Absorbance at 214 nm was measured. The percentage of elastase inhibition was calculated by the same method that was used to measure inhibition of collagenase.
2.5. Inhibition of tyrosinase activity
The inhibitory activity of tyrosinase was measured according to the method reported by Mason and Peterson17 adapted to a 96-well plate. A premixture solution containing 70 μL of 0.1 M phosphate buffer (pH 6.8), 30 μL of mushroom tyrosinase (167 U/mL), and 20 μL of sample was incubated for 5 minutes at 30 °C. Approximately 100 μL of 3,4-dihydroxyphenyl-l-alanine (l-DOPA) was then added to initiate the enzymatic reaction. The absorbance at 492 nm was measured to observe DOPAchrome formation for 20 minutes. The inhibition ratio was calculated as follows:
where A is a mixture with tyrosinase but without sample; B is a mixture without sample and tyrosinase; C is a mixture with sample and tyrosinase; and D is a mixture with sample but without tyrosinase.
2.6. Statistical analysis
All expressed values are the means of three experiments. All statistical analyses were performed using the Statistical Package for Social Sciences version 12.0 (SPSS Inc., Chicago, IL, USA). The changes in physical stability according to time interval were analyzed by repeated measures analysis of variance (ANOVA). The differences among formulations (vehicle, FNGE, and FTGE) were analyzed by one-way ANOVA and Turkey multiple test. The results are reported as means ± standard deviation. All tests were two-sided at the 5% significance level.
3. Results
3.1. Green tea biotransformation by tannase
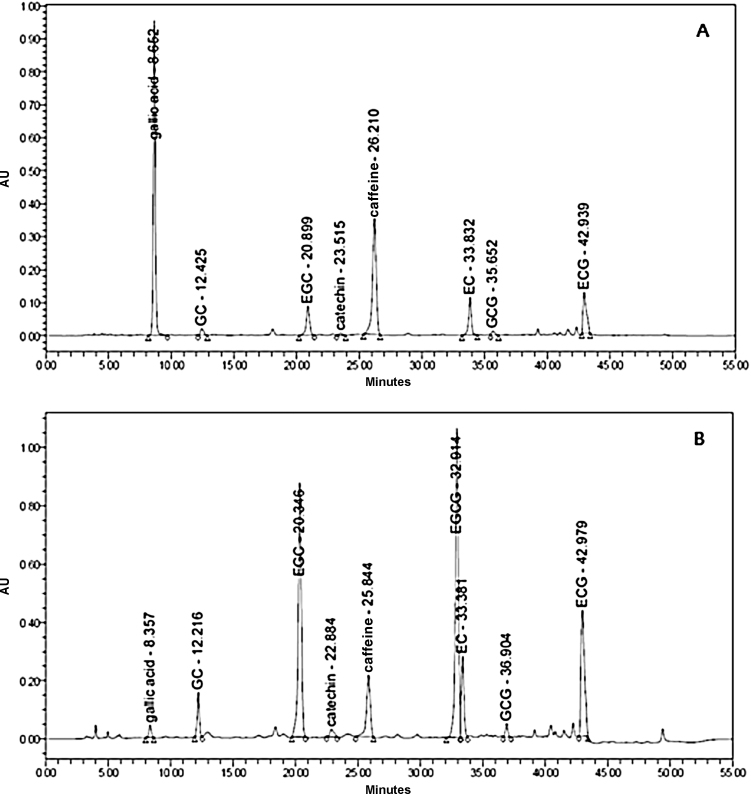
The bioconversion of green tea was achieved by tannase treatment. The composition of catechins in tannase-treated green tea extract was analyzed by high-performance liquid chromatography (HPLC; Fig. 1). We observed that the hydrolysis of green tea catechins usually increased with the reaction time (Table 2). The relative levels of each component in the green tea extracts were altered by tannase treatment. Tannase treatment induced the decrease of EGCG and ECG levels, and increase in the levels of (–)-epigallocatechin (EGC), (–)-epicatechin (EC), and GA. However, the relative amounts of caffeine were not changed after tannase treatment.
Fig. 1.
High-performance liquid chromatography chromatogram for catechins in green tea extract (A) with or (B) without tannase treatment under optimum extraction conditions. GC, (–)-gallocatechin; EGC, (–)-epigallocatechin; EGCG, (–)-epigallocatechin gallate; EC, (–)-epicatechin; GCG, (–)-gallocatechin gallate; ECG; (–)-epicatechin gallate.
Table 2.
Catechin composition of green tea extract before and after tannase treatment.
| Tea extract | Gallic acid | GC | EGC | Catechin | Caffeine | EGCG | EC | GCG | ECG |
|---|---|---|---|---|---|---|---|---|---|
| NGE | 254.6 ± 2.2 | 203.2 ± 1.7 | 1557.9 ± 14.9 | 99.2 ± 0.2 | 55.6 ± 0.7 | 3990.9 ± 35.4 | 640.6 ± 22.3 | 196.0 ± 2.7 | 1473.7 ± 11.8 |
| TGE | 3056.0 ± 17.8 | 354.3 ± 4.1 | 5284.4 ± 63.6 | 94.0 ± 7.9 | 51.5 ± 0.2 | 3.3 ± 0.1 | 1445.0 ± 5.7 | 26.0 ± 5.8 | 240.2 ± 11.4 |
EC, epicatechin; ECG (–)-epicatechin-3-gallate; EGCG, (–)-epigallocatechin-3-gallate; EGC, (–)-epigallocatechin; GC, gallocatechin; GCG, (–)-gallocatechin gallate; NGE, normal green tea extract (green tea extract before tannase treatment); TGE, tannase-converted green tea extract (green tea extract after tannase treatment).
3.2. Physical stability
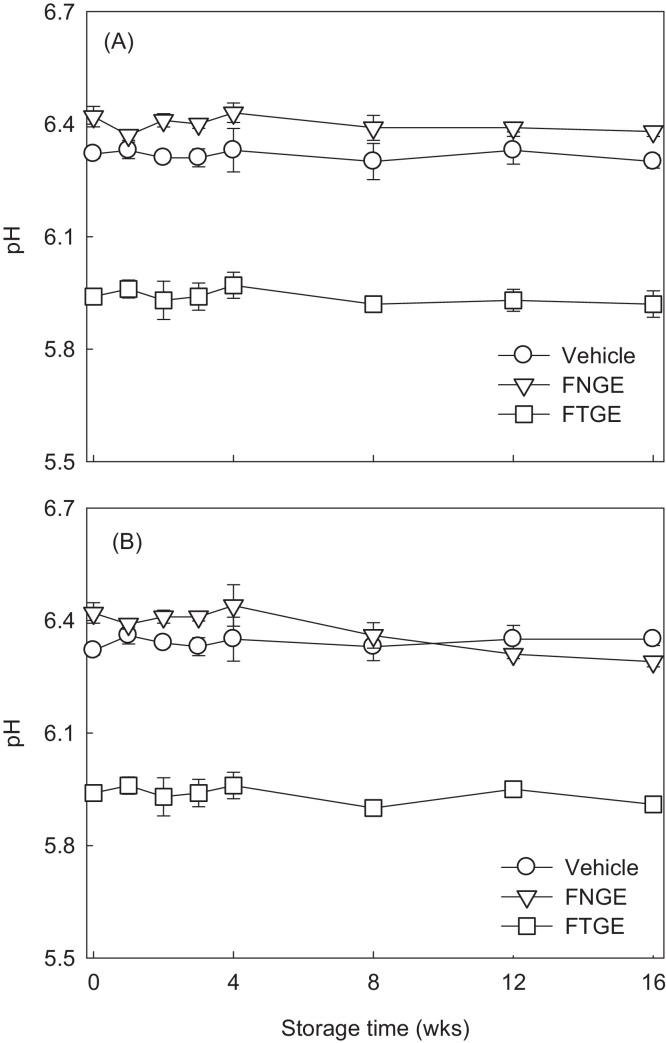
Fig. 2 shows the pH variation of the cosmetic formulations stored at different temperatures and humidities for 4 months. The pH values of each sample did not exhibit a significant change over time at 25 ± 2 °C/AH; they showed minimal changes within the ranges of 6.32–6.30, 6.42–6.38, and 5.94–5.92 for vehicle, FNGE, and FTGE, respectively (Fig. 2A). The change in pH values at 40 ± 2 °C with 70% ± 5% RH (Fig. 2B) was also similar to those at 25 ± 2 °C with AH condition. This result showed that the pH of green tea extract formulation is relatively stable at different temperatures, humidities, and storage durations. Tannase treatment leads to a significant decrease in pH value compared with the control; the pH of FTGE was approximately 5.9, whereas FNGE has pH values of 6.3–6.4 in both Figs. 2A and 2B. Our data showed that green tea extract formulations, especially FTGE, maintained constant weak acidic pH during the storage period (Fig. 2).
Fig. 2.
pH changes of formulation containing 5% tannase-converted green tea extract stored at (A) 25 ± 2 °C/ambient humidity and at (B) 40 ± 2 °C/70% ± 5% relative humidity for 4 months. Values are means ± standard deviation of three determinations. FNGE, formulation containing 5% normal green tea extract; FTGE, formulation containing 5% tannase-converted green tea extract.
After centrifugation, phase separation was not observed in any of the three formulations stored for different periods at 25 °C. The FNGE kept at 40 °C for 8 weeks showed a little phase separation, and the centrifugal precipitations in FNGE were present in very small quantities. However, no centrifugal precipitation was found at any storage time in the FTGE kept at 40 °C (data not shown).
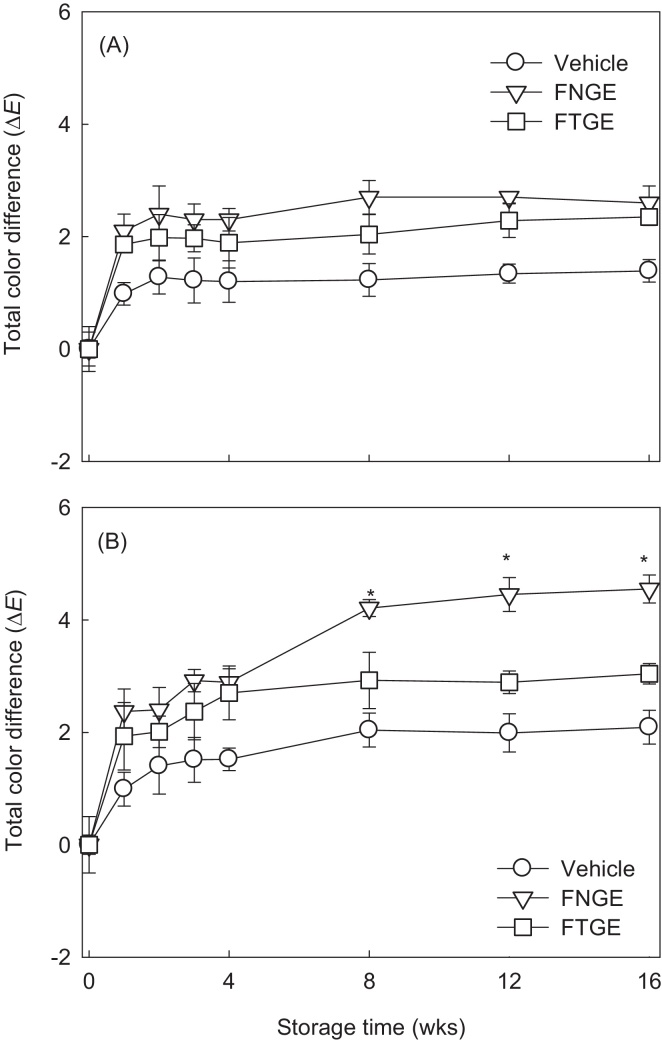
The color changes of the cosmetic formulations were also analyzed in the stability study (Fig. 3). In particular, the total color difference (ΔE) of the FNGE was higher than that of the FTGE. The total color differences of the FTGE were slightly increased with increasing storage times, whereas no significant differences were observed between baseline and changed values. However, FNGE started to show significant (p < 0.05) darkness after 56 days of storage at 40 ± 2 °C with 70% ± 5% RH. The color changes shown by FNGE were significantly different between baseline values and values at 8, 12, and 16 weeks (Fig. 3B). Therefore, our results indicate good color stability of FTGE.
Fig. 3.
Color changes of formulation containing 5% tannase-converted green tea extract stored at (A) 25 ± 2 °C/ambient humidity and at (B) 40 ± 2 °C/70% ± 5% relative humidity for 4 months. Values are means ± standard deviation of three determinations. Asterisk indicates a significant difference (p < 0.05) between baseline and each week by a paired t test. L (degree of lightness), a (degree of redness), and b (degree of yellowness) are colors of samples; L′, a′, and b′ are colors of base at zero time. FNGE, formulation containing 5% normal green tea extract; FTGE, formulation containing 5% tannase-converted green tea extract.
3.3. Total polyphenol and flavonoid contents
We examined the effect of tannase treatment on the levels of total polyphenol and flavonoids in the green tea extract formulation. As shown in Fig. 4A, TGE allowed a significantly (p < 0.05) increased level of total polyphenol contents compared with NGE and vehicle; FTGE had 0.92 mg/mL of total polyphenolic content, while vehicle and FNGE had 0.60 and 0.83 mg/mL, respectively. In addition, the flavonoid content of FTGE (0.57 mg/mL) was higher than the levels in other formulations (vehicle = 0.09 and FNGE = 0.45 mg/mL; p < 0.05; Fig. 4B). This result suggests that polyphenol and flavonoid contents in green tea were increased by tannase-derived bioconversion.
Fig. 4.
Total (A) polyphenol and (B) flavonoid contents in formulation containing 5% tannase-converted green tea extract. Bars are means ± standard deviation of three determinations. Asterisks indicate significant differences (p < 0.05) among samples by Turkey multiple range test. FNGE, formulation containing 5% normal green tea extract; FTGE, formulation containing 5% tannase-converted green tea extract.
In our HPLC analysis, tannase treatment induced changes in the compositions of catechin (Fig. 1); the FNGE contained 2.4 mg/g GA, 5.9 mg/g EGC, and 3.6 mg/g EC compared with the FTGE levels of 17.2, 9.4, and 4.9 mg/g, respectively, and the FTGE contained less EGCG (1.0 mg/g) and ECG (1.1 mg/g) than the contents of FNGE (EGCG = 33.3 mg/g and ECG = 11.9 mg/g; data not shown).
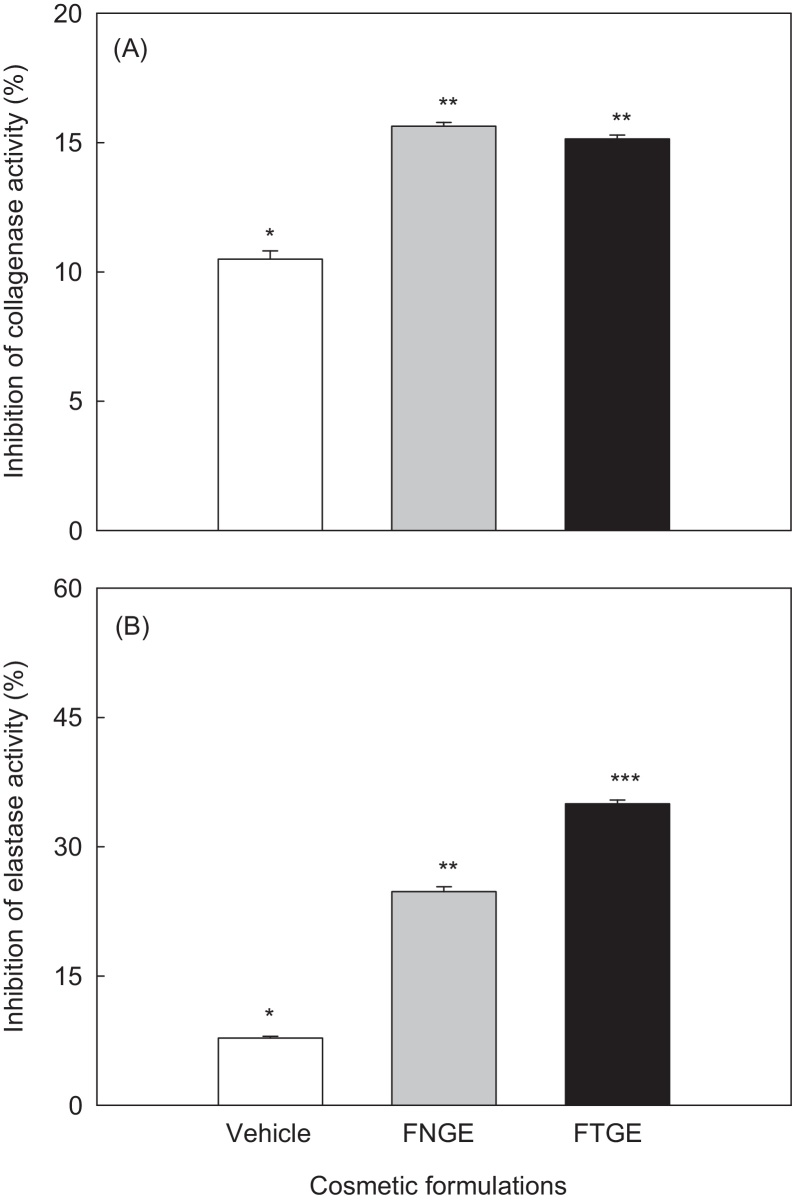
3.4. Inhibition of collagenase, elastase and tyrosinase activities
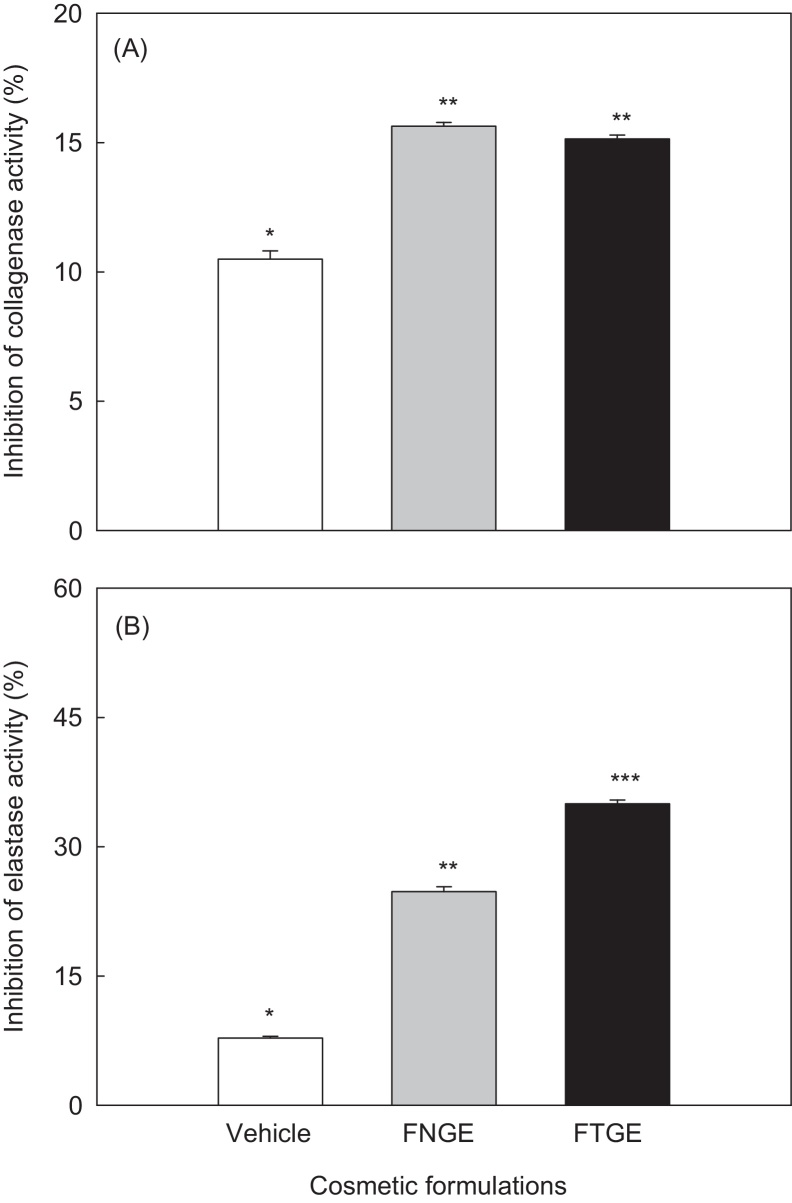
Collagenase and elastase are known to be major enzymes responsible for dehydration and wrinkle formation on the skin surface. We examined the inhibitory effects of green tea extract on these two enzymes in the presence or absence of tannase. The inhibitory activities of collagenase on the cosmetic formulations are shown in Fig. 5A. The inhibition of collagenase activity was not significantly different between FNGE (15.63%) and FTGE (15.14%). The elastase inhibitory activity of the cosmetic formulations is shown in Fig. 5B. The inhibition of elastase activity for FNGE and FTGE was 24.79% and 34.98%, respectively. This result showed that FTGE has an inhibitory effect on elastase rather than on collagenase, which could contribute to antiwrinkle effects. Our results indicate that FTGE possesses equivalent or higher inhibitory effect on collagenase and elastase compared with FNGE. The inhibition of elastase by FTGE was significantly (p < 0.05) higher than that by FNGE.
Fig. 5.
(A) Collagenase and (B) elastase inhibitory activities of formulation containing 5% tannase-converted green tea extract. Collagenase inhibition activity (%) = [(A–B)/A] × 100. (A) With collagenase but without sample; (B) with sample and collagenase. The percentage of elastase inhibition was calculated using the same method used for collagenase inhibition. Bars are means ± standard deviation of three determinations. Asterisks indicate significant differences (p < 0.05) among samples by Turkey multiple range test. FNGE, formulation containing 5% normal green tea extract; FTGE, formulation containing 5% tannase-converted green tea extract.
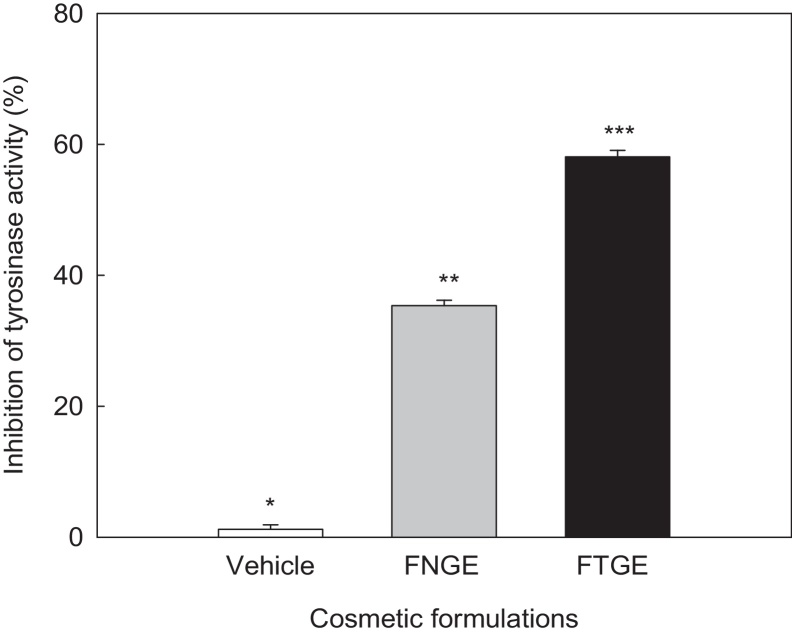
The tyrosinase inhibitory activities of the cosmetic formulations were measured as shown in Fig. 6. FTGE (58.06%) showed a higher inhibitory activity (58.06%) than that of FNGE (35.38%). This result showed that tannase treatment enhanced the inhibitory effect of tyrosinase activity.
Fig. 6.
Tyrosinase inhibitory activities of formulation containing 5% tannase-converted green tea extract. Tyrosinase inhibition activity (%) = [(A − B) − (C − D)]/(A − B) × 100. (A) With tyrosinase but without sample; (B) without sample and tyrosinase; (C) with sample and tyrosinase; and (D) with sample but without tyrosinase. Bars are means ± standard deviation of three determinations. Asterisks indicate significant differences (p < 0.05) among samples by Turkey multiple range test. FNGE, formulation containing 5% normal green tea extract; FTGE, formulation containing 5% tannase-converted green tea extract.
4. Discussion
Gallated catechins, such as EGCG and ECG, were thought to be hydrolyzed to degallated catechins (EGC or EC), and such conversion was maximized in 2 h. This change in the compositions of green tea extract is due to the action of tannase, which cleaves the ester bonds between EGCG (or ECG) and GAs. Aspergillus oryzae is a known source of tannase hydrolyzing the ester bonds of natural substrates.18 More such sources are expected to be discovered in the future.
Stability test is one of the crucial areas in the cosmetic testing program, because of the instability of the product involved in the safety, efficacy, and quality of cosmetic formulations. For physical stability, changes in pH, level of precipitation, and color features for storage period are tested. These pH conditions of green tea extract well correspond to weak acidic environments on the skin surface, which provides resistance to foreign pathogens. The efficacy of cosmetic product depends on the delivery of the functional molecule into the skin, which is affected by the type of molecules.19 These agreements of pH between green tea extract and skin are thought to be desirable for delivery of active compounds into the skin. Therefore, our results indicate that TGE did not change the pH during storage, providing more acidic pH conditions for skin application. Centrifugation is one of the alternative methods to rapidly assess the stability of different formulations. The hydrolytic activity of tannase could be expected to decrease the binding ability of EGCG to protein, due to the cleavage of ester bonds. However, our tannase treatments did not cause aggregation of macromolecules or the formation of precipitates during storage periods.
Many cosmetic products currently use plant extracts as a source of flavonoid. The increase in the use of flavonoids in cosmetic is due to their beneficial biochemical effect such as antioxidant activity.20 Catechins are the major polyphenols in green-tea extracts. A recent study reported on the various health beneficial effects of catechin including its sedative effect on skin and inhibitory effects on photoaging and cancer.21 Green tea usually contains four major types of catechins, namely, EC, (–)-ECG, (–)-EGC, and (–)-EGCG. The EGCG constitutes 65% of the total catechins.22 Compared with other hydrophobic substances it is difficult for catechins to permeate the dermal surfaces due to their hydrophilic nature and their chemical interactions with the skin lipid bilayer.23 Several studies on dermal penetration of catechins showed that degallated catechins such as EC and EGC more easily permeated the skin than EGCG and ECG.24 EC and EGC is thought to penetrate the skin more than EGCG and ECG due to their increased hydrophobicity. Therefore, FTGE might be able to permeate the skin more easily than FNGE, which provides a higher availability in cosmetics.
Collagen and elastin are major components of the connective tissue of the skin. Collagen accounts for 70–80% of the skin weight, providing structural stability. However, collagen is known to be rapidly dismantled by collagenase.25 Plant phenolic compounds have been known to inhibit collagenase activity,26 accompanied by free-radical scavenging ability to control aging. Catechins including EGCG have been known to inhibit the activity of collagenase and elastase.27 Persimmon (Diospyros kaki) leaf-derived polyphenols showed anticollagenase and antielastase activity.28 Rosemary (Rosmarinus officinalis) extract has also been known to have strong antielastase activity.29 The principle compound responsible for this inhibitory activity is considered to be the flavonoids present in plant extracts. Madhan et al.10 showed that the conformational change in collagenase induced by the green tea polyphenols is responsible for the inhibition of Clostridium histolyticum collagenase I. The hydroxyl part of polyphenols can form hydrogen bond with the functional groups of collagenase. The benzene group of the polyphenols can also be involved in hydrophobic interactions with collagenase.
Elastin, an insoluble fibrous protein, occupies only 2–4% of the dermis matrix, but plays an important role in presenting the elasticity of the skin. It also comprises a network with collagenous fibers under the epidermis.30 It is usually degraded by elastase, which can hydrolyze peripheral and structural proteins in the connective tissue.31 Because decomposition of elastin results from activation of elastase caused by ultraviolet light or ROS, inhibition of elastase activity could also be a therapeutic target to protect elastin-induced skin aging.32 ROS is known to induce expression of proteinases, which cause remodeling of skin extracellular matrix. Activation of such proteases (e.g., matrix metalloproteinases and serine proteases) may be involved in the lack of skin elasticity.32, 33 The inhibitory effect of FTGE on elastase could be correlated to our data, which showed more total polyphenol and flavonoid contents in FTGE (Fig. 4). FTGE can strongly interact with elastase than FNGE due to hydrophobic interactions, providing the conformational change of elastase, resulting in strong inhibition of elastase activity. Therefore, our results indicate that FTGE has been demonstrated to be a beneficial ingredient to treat the effects of skin aging.
Melanin is responsible for the formation of spots and freckles on skin. Tyrosinase is a major enzyme of the melanin synthetic pathway in melanocytes. Therefore, inhibition of melanocyte metabolic enzyme such as tyrosinase could be an important strategy for blocking melanogenesis.34 In reality, tyrosinase inhibitors have been used as important ingredients of cosmetics for skin whitening.11 Search for the tyrosinase inhibitor usually using an in vitro mushroom tyrosinase inhibition assay.35 Kojic acid, arbutin, ascorbic acid derivatives, retinoic acid, azelaic acid, and many other natural inhibitors are now commercialized or used for localized hyperpigmentation, which is found in dermatological disorders. Recent studies reported on arbutin, a fungal metabolite, and hydroquinone, which are used as tyrosinase inhibitors in cosmetics.36, 37 However, some of these inhibitors involve several limitations in activity, toxicity, and penetration. Therefore, safe and effective tyrosinase inhibitors are needed to be searched and developed. Edible plant-derived bioactive compounds usually receive a lot of attention as tyrosinase inhibitor because they are relatively safe compared with wild plants. Potent tyrosinase inhibitors, such as cuminaldehyde, oxyresveratrol, kaempferol, quercetin, and GA derivatives, have been isolated from various plants.38
The majority of natural tyrosinase inhibitors consist of a phenolic structure or are a metal chelater.39 In general, the level of the antioxidant or tyrosinase inhibitory activities is proportion to the level of the phenolic content. Thus, phenolics contained in our samples may play a major role in inhibition of tyrosinase activity in FTGE. Polyphenol may be also used as depigmentation agents because of their structural similarity to tyrosine, a substrate of tyrosinase.38 In addition, antioxidants inhibit pigmentation by various mechanisms including scavenging of ROS and reactive nitrogen species,40 and reduction of o-quinones or other intermediates in melanin biosynthesis, thus delaying oxidative polymerization.41 α-Tocopherol, an antioxidant, is usually known to inhibit melanin formation by suppressing oxidative polymerization of phenylalanines.42 Therefore, polyphenolic compounds are partially responsible for the efficacy of substances used as whitening agents in skin-care products.
According to Kubo et al. 43 all catechins could inhibit tyrosinase by their ability to chelate copper. Many catechins have been isolated from green tea 44 as strong tyrosinase inhibitors. This study indicates that optimal tyrosinase inhibition requires the structure of flavan-3-ol skeleton with a galloyl moiety at the 3 position. Moreover, addition of a hydroxyl group enhanced the inhibitory effect on tyrosinase, whereas addition of a methyl group reduced the inhibitory activity. Meanwhile, an unsaturated alkyl side chain produced a stronger inhibition compared with a saturated side chain. Oxyresveratrol presents strong tyrosinase inhibitory activity mainly because of the hydroxy groups in the ring.44 No et al. 44 reported that EC, EGC, and catechin, which are isolated from green tea extract, showed <10%, 40%, and <10% of inhibitory effects, respectively, against mushroom tyrosinase at a 40 μM concentration. Our data showed that the inhibitory effect of FTGE on tyrosinase activity was higher than that of FNGE. Thus, TGE could be more useful in the development of cosmetic formulations for whitening based on the tyrosinase inhibition.
5. Conclusion
In this study, we found that FTGE retained its physical stability in environmental changes such as temperature and pH, and maintained higher level of total polyphenols and flavonoids than FNGE. Moreover, our result indicates that tannase-treated formulation may be more effective for inhibiting elastase and tyrosinase activities. Thus, this study suggests that TGE may be supplemented as a pharmacologic agent in skin-care products such as antiaging and whitening creams.
Conflicts of interest
The authors declare no conflicts of interest.
Acknowledgment
This study was supported by the fund from Kaya University.
References
- 1.Makrantonaki E., Zouboulis C.C. Skin alterations and diseases in advanced age. Drug Discov Today Dis Mech. 2008;5:e153–e162. [Google Scholar]
- 2.Morganti P. Reflections on cosmetics, cosmeceuticals, and nutraceuticals. Clin Dermatol. 2008;26:318–320. doi: 10.1016/j.clindermatol.2008.01.001. [DOI] [PubMed] [Google Scholar]
- 3.Gao X.H., Zhang L., Wei H., Chen H.D. Efficacy and safety of innovative cosmeceuticals. Clin Dermatol. 2008;26:367–374. doi: 10.1016/j.clindermatol.2008.01.013. [DOI] [PubMed] [Google Scholar]
- 4.Rababah T.M., Hettiarachchy N.S., Horax R. Total phenolics and antioxidant activities of fenugreek, green tea, black tea, grape seed, ginger, rosemary, gotu kola, and ginkgo extracts, vitamin E, and tert-butylhydroquinone. J Agric Food Chem. 2004;52:5183–5186. doi: 10.1021/jf049645z. [DOI] [PubMed] [Google Scholar]
- 5.Rice-Evans C.A., Miller N.J. Antioxidant activities of flavonoids as bioactive components of food. Biochem Soc Trans. 1996;24:790–794. doi: 10.1042/bst0240790. [DOI] [PubMed] [Google Scholar]
- 6.Ma W., Wlaschek M., Tantcheva-Poór I., Schneider L.A., Naderi L., Razi-Wolf Z. Chronological ageing and photoageing of the fibroblasts and the dermal connective tissue. Clin Exp Dermatol. 2001;26:592–599. doi: 10.1046/j.1365-2230.2001.00905.x. [DOI] [PubMed] [Google Scholar]
- 7.Hsu S. Green tea and the skin. J Am Acad Dermatol. 2005;52:1049–1059. doi: 10.1016/j.jaad.2004.12.044. [DOI] [PubMed] [Google Scholar]
- 8.Katiyar S., Elmets C.A., Katiyar S.K. Green tea and skin cancer: photoimmunology, angiogenesis and DNA repair. J Nutr Biochem. 2007;18:287–296. doi: 10.1016/j.jnutbio.2006.08.004. [DOI] [PubMed] [Google Scholar]
- 9.Borek C. Antioxidant health effects of aged garlic extract. J Nutr. 2001;131 doi: 10.1093/jn/131.3.1010S. 1010S–15S. [DOI] [PubMed] [Google Scholar]
- 10.Madhan B., Krishnamoorthy G., Rao J.R., Nair B.U. Role of green tea polyphenols in the inhibition of collagenolytic activity by collagenase. Int J Biol Macromol. 2007;41:16–22. doi: 10.1016/j.ijbiomac.2006.11.013. [DOI] [PubMed] [Google Scholar]
- 11.An B.J., Kwak J.H., Son J.H., Park J.M., Lee J.Y., Park T.S. Physiological activity of irradiated green tea polyphenol on the human skin. Am J Chin Med. 2005;33:535–546. doi: 10.1142/S0192415X05003144. [DOI] [PubMed] [Google Scholar]
- 12.Lu M.-J., Chen C. Enzymatic tannase treatment of green tea increases in vitro inhibitory activity against N-nitrosation of dimethylamine. Process Biochem. 2007;42:1285–1290. [Google Scholar]
- 13.Zhang C., Lu Y., Tao L., Tao X., Su X., Wei D. Tyrosinase inhibitory effects and inhibition mechanisms of nobiletin and hesperidin from citrus peel crude extracts. J Enzyme Inhib Med Chem. 2007;22:91–98. doi: 10.1080/14756360600988989. [DOI] [PubMed] [Google Scholar]
- 14.Li C., Du H., Wang L., Shu Q., Zheng Y., Xu Y. Flavonoid composition and antioxidant activity of tree peony (Paeonia section moutan) yellow flowers. J Agric Food Chem. 2009;57:8496–8503. doi: 10.1021/jf902103b. [DOI] [PubMed] [Google Scholar]
- 15.Van Wart H.E., Steinbrink D.R. A continuous spectrophotometric assay for Clostridium histolyticum collagenase. Anal Biochem. 1981;113:356–365. doi: 10.1016/0003-2697(81)90089-0. [DOI] [PubMed] [Google Scholar]
- 16.Kraunsoe J.A., Claridge T.D., Lowe G. Inhibition of human leukocyte and porcine pancreatic elastase by homologues of bovine pancreatic trypsin inhibitor. Biochemistry. 1996;35:9090–9096. doi: 10.1021/bi953013b. [DOI] [PubMed] [Google Scholar]
- 17.Mason H.S., Peterson E.W. Melanoproteins. I. Reactions between enzyme-generated quinones and amino acids. Biochim Biophys Acta. 1965;111:134–146. doi: 10.1016/0304-4165(65)90479-4. [DOI] [PubMed] [Google Scholar]
- 18.García-Conesa M.-T., Østergaard P., Kauppinen S., Williamson G. Hydrolysis of diethyl diferulates by a tannase from Aspergillus oryzae. Carbohydr Polym. 2001;44:319–324. [Google Scholar]
- 19.Wu X., Landfester K., Musyanovych A., Guy R.H. Disposition of charged nanoparticles after their topical application to the skin. Skin Pharmacol Physiol. 2010;23:117–123. doi: 10.1159/000270381. [DOI] [PubMed] [Google Scholar]
- 20.Arct J., Oborska A., Mojski M., Binkowska A., Swidzikowska B. Common cosmetic hydrophilic ingredients as penetration modifiers of flavonoids. Int J Cosmet Sci. 2002;24:357–366. doi: 10.1046/j.1467-2494.2002.00166.x. [DOI] [PubMed] [Google Scholar]
- 21.Yusuf N., Irby C., Katiyar S.K., Elmets C.A. Photoprotective effects of green tea polyphenols. Photodermatol Photoimmunol Photomed. 2007;23:48–56. doi: 10.1111/j.1600-0781.2007.00262.x. [DOI] [PubMed] [Google Scholar]
- 22.Perva-Uzunalić A., Škerget M., Knez Ž, Weinreich B., Otto F., Grüner S. Extraction of active ingredients from green tea (Camellia sinensis): extraction efficiency of major catechins and caffeine. Food Chem. 2006;96:597–605. [Google Scholar]
- 23.Sun Y., Hung W.-C., Chen F.-Y., Lee C.-C., Huang H.W. Interaction of tea catechin (–)-epigallocatechin gallate with lipid bilayers. Biophys J. 2009;96:1026–1035. doi: 10.1016/j.bpj.2008.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.dal Belo S.E., Gaspar L.R., Maia Campos P.M., Marty J.P. Skin penetration of epigallocatechin-3-gallate and quercetin from green tea and Ginkgo biloba extracts vehiculated in cosmetic formulations. Skin Pharmacol Physiol. 2009;22:299–304. doi: 10.1159/000241299. [DOI] [PubMed] [Google Scholar]
- 25.Demina N.S., Lysenko S.V. Collagenolytic enzymes synthesized by microorganisms. Mikrobiologiia. 1996;65:293–304. [Article in Russian] [PubMed] [Google Scholar]
- 26.Miao Y.P., Wen R., Aoshima H., Zhou P.G. Synthesis and antioxidative activity of 2-substituted phenyl-5-(3′-indolyl)-oxazole derivatives. Yao Xue Xue Bao. 2004;39:37–40. [PubMed] [Google Scholar]
- 27.Kim Y.H., Chung C.B., Kim J.G., Ko K.I., Park S.H., Kim J.H. Anti-wrinkle activity of ziyuglycoside I isolated from a Sanguisorba officinalis root extract and its application as a cosmeceutical ingredient. Biosci Biotechnol Biochem. 2008;72:303–311. doi: 10.1271/bbb.70268. [DOI] [PubMed] [Google Scholar]
- 28.An B.J., Kwak J.H., Park J.M., Lee J.Y., Park T.S., Lee J.T. Inhibition of enzyme activities and the antiwrinkle effect of polyphenol isolated from the persimmon leaf (Diospyros kaki folium) on human skin. Dermatol Surg. 2005;31:848–854. doi: 10.1111/j.1524-4725.2005.31730. discussion 854. [DOI] [PubMed] [Google Scholar]
- 29.Baylac S., Racine P. Inhibition of human leukocyte elastase by natural fragrant extracts of aromatic plants. Int J Aromather. 2004;14:179–182. [Google Scholar]
- 30.Thring T.S., Hili P., Naughton D.P. Anti-collagenase, anti-elastase and anti-oxidant activities of extracts from 21 plants. BMC Complement Altern Med. 2009;9:27. doi: 10.1186/1472-6882-9-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhi-ying Y., Guo-xiong G., Wei-qin Z., Zhe-fu L. Elastolytic activity from Flavobacterium odoratum. Microbial screening and cultivation, enzyme production and purification. Process Biochem. 1994;29:427–436. [Google Scholar]
- 32.Meyer W., Neurand K., Radke B. Elastic fibre arrangement in the skin of the pig. Arch Dermatol Res. 1981;270:391–401. doi: 10.1007/BF00403783. [DOI] [PubMed] [Google Scholar]
- 33.Wiedow O., Schröder J.M., Gregory H., Young J.A., Christophers E. Elafin: an elastase-specific inhibitor of human skin. Purification, characterization, and complete amino acid sequence. J Biol Chem. 1990;265:14791–14795. [PubMed] [Google Scholar]
- 34.Park S.H., Kim D.S., Kim W.G., Ryoo I.J., Lee D.H., Huh C.H. Terrein: a new melanogenesis inhibitor and its mechanism. Cell Mol Life Sci. 2004;61:2878–2885. doi: 10.1007/s00018-004-4341-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Madhosingh C., Sundberg L. Purification and properties of tyrosinase inhibitor from mushroom. FEBS Lett. 1974;49:156–158. doi: 10.1016/0014-5793(74)80500-4. [DOI] [PubMed] [Google Scholar]
- 36.Rendon M.I., Gaviria J.I. Review of skin-lightening agents. Dermatol Surg. 2005;31:886–889. doi: 10.1111/j.1524-4725.2005.31736. discussion 889. [DOI] [PubMed] [Google Scholar]
- 37.Maeda K., Fukuda M. In vitro effectiveness of several whitening cosmetic components in human melanocytes. J Soc Cosmet Chem. 1991;42:361–368. [Google Scholar]
- 38.Parvez S., Kang M., Chung H.S., Bae H. Naturally occurring tyrosinase inhibitors: mechanism and applications in skin health, cosmetics and agriculture industries. Phytother Res. 2007;21:805–816. doi: 10.1002/ptr.2184. [DOI] [PubMed] [Google Scholar]
- 39.Mayer A.M. Polyphenol oxidases in plants—recent progress. Phytochemistry. 1986;26:11–20. [Google Scholar]
- 40.Seo S.Y., Sharma V.K., Sharma N. Mushroom tyrosinase: recent prospects. J Agric Food Chem. 2003;51:2837–2853. doi: 10.1021/jf020826f. [DOI] [PubMed] [Google Scholar]
- 41.Karg E., Odh G., Wittbjer A., Rosengren E., Rorsman H. Hydrogen peroxide as an inducer of elevated tyrosinase level in melanoma cells. J Invest Dermatol. 1993;100 209S–13S. [PubMed] [Google Scholar]
- 42.Nilsson S.E., Sundelin S.P., Wihlmark U., Brunk U.T. Aging of cultured retinal pigment epithelial cells: oxidative reactions, lipofuscin formation and blue light damage. Doc Ophthalmol. 2003;106:13–16. doi: 10.1023/a:1022419606629. [DOI] [PubMed] [Google Scholar]
- 43.Kubo I., Kinst-Hori I., Kubo Y., Yamagiwa Y., Kamikawa T., Haraguchi H. Molecular design of antibrowning agents. J Agric Food Chem. 2000;48:1393–1399. doi: 10.1021/jf990926u. [DOI] [PubMed] [Google Scholar]
- 44.No J.K., Soung D.Y., Kim Y.J., Shim K.H., Jun Y.S., Rhee S.H. Inhibition of tyrosinase by green tea components. Life Sci. 1999;65:PL241–PL246. doi: 10.1016/s0024-3205(99)00492-0. [DOI] [PubMed] [Google Scholar]