Abstract
Background
Asymmetric and symmetric dimethylarginines (ADMA and SDMA) are putative uremic toxins that may exert toxicity by a number of mechanisms including impaired nitric oxide synthesis and generation of reactive oxygen species. The study goal was to determine the association between these metabolites and cardiovascular outcomes in hemodialysis patients.
Study Design
Post hoc analysis of the Hemodialysis (HEMO) Study.
Setting & Participants
1,276 prevalent hemodialysis patients with available samples 3–6 months after randomization.
Predictor
ADMA and SDMA measured in stored specimens.
Outcomes
Cardiac death, sudden cardiac death, first cardiovascular event, and any-cause death. Association with predictors analyzed using Cox regression adjusted for potential confounders (including demographics, clinical characteristics, comorbidities, albumin, and residual kidney function).
Results
Mean age of patients was 57 ±14 (SD) years, 63% were Black and 57% were female. Mean ADMA (0.9 ± 0.2 µM) and SDMA (4.3 ± 1.4 µM) were moderately correlated (r=0.418). Higher dialysis dose or longer session length were not associated with lower predialysis concentrations of ADMA or SDMA. In fully adjusted models, each doubling of ADMA was associated with higher risk (HR per 2-fold higher concentration; 95% CI) of cardiac death (1.83; 1.29–2.58), sudden cardiac death (1.79; 1.19–2.69), first cardiovascular event (1.50; 1.20–1.87), and any-cause death (1.44; 1.13–1.83). Compared to the lowest ADMA quintile (≤0.745 µM), the highest ADMA quintile (≥1.07 µM) was associated with higher risk (HR; 95% CI) of cardiac death (2.10; 1.44–3.05), sudden cardiac death (2.06; 1.46–2.90), first cardiovascular event (1.75; 1.35–2.27), and any-cause death (1.56; 1.21–2.00). SDMA was associated with higher risk of cardiac death (1.40; 1.03–1.92) but this was no longer statistically significant after adjusting for ADMA (1.20; 0.86–1.68).
Limitations
Single time-point measurement of ADMA and SDMA.
Conclusions
ADMA and, to a lesser extent SDMA are associated with cardiovascular outcomes in hemodialysis patients.
Keywords: Cardiovascular Mortality, Dialysis Outcomes, Asymmetric Dimethylarginine (ADMA), Symmetric Dimethylarginine (SDMA), hemodialysis, end-stage renal disease (ESRD), uremic toxins, cardiovascular morbidity, cardiac death, sudden cardiac death
Patients undergoing dialysis continue to have excessive morbidity and mortality despite many advances in care.(1) Much of this excessive risk is due to cardiovascular disease, however, the underlying mechanisms for the accelerated cardiovascular disease phenotype in dialysis patients remain undefined.(2, 3) Some of this risk may be due to solutes that accumulate in the body in kidney failure.(4) The identity of these uremic toxins remains incompletely known.
Asymmetric dimethylarginine (ADMA) and symmetric dimethylarginine (SDMA) are produced endogenously by the metabolism of arginine-containing proteins.(5) Both metabolites accumulate in patients with kidney failure.(6) A body of evidence supports toxicity of ADMA and, to a lesser extent, SDMA.(5, 7, 8) Whereas ADMA inhibits nitric oxide synthesis, SDMA may have vasculotoxic and proatherogenic effects. Both ADMA and SDMA have been associated with cardiovascular mortality in large general population studies(9–12), and previous studies also suggest associations with cardiovascular outcomes in dialysis patients.(13, 14)
We measured ADMA and SDMA in specimens of the Hemodialysis (HEMO) Study, a US multicenter trial of hemodialysis dose and flux. Our aim was to examine the longitudinal association between these solutes and cardiovascular morbidity and mortality in patients receiving dialysis. The design of the HEMO Study16, 17 --- including its large sample size, national multicenter design, inclusion of patients without significant residual kidney function, and physician-adjudicated outcomes --- provided us with a unique opportunity to examine the associations between the dimethylarginines and outcomes in hemodialysis patients.
METHODS
Study Design
The HEMO Study was a clinical trial that randomized 1846 prevalent hemodialysis patients to standard or high dialyzer urea clearance (assessed by Kt/Vurea, an index of urea clearance by dialysis) and to low-flux or high-flux dialysis membranes (assessed by β2-microglobulin clearance).(15, 16) The patients were enrolled May 1995–February 2001 from 15 clinical centers in the United States comprising 72 dialysis units and followed up for outcomes until death, kidney transplantation or end of study in December 2001. Major exclusion criteria included residual urea clearance >1.5 mL/min/35 L urea volume of distribution, unstable angina, active systemic infection, New York Heart Association class IV congestive heart failure and severe hypoalbuminemia (<2.6 g/dL). Our study sample included all HEMO Study participants who had available stored sera collected 3–6 months post-randomization (N=1276), a time-point allowing adequate separation between the trial intervention arms. The participating institutions’ institutional review boards reviewed and approved the HEMO Study and all participants provided informed consent. The Johns Hopkins Medicine Institutional Review Board reviewed and approved this study (IRB00081893).
Data Collection
Laboratory Measurements
We measured ADMA/SDMA by liquid chromatography–tandem mass spectrometry (LC-MS/MS) using ADMA-d7 (Cambridge isotope laboratories, Andover, MA) and SDMA-d6 (Toronto Research Chemicals, Toronto, ON) as internal standards. Plasma was deproteinized by mixture with an internal standard solution and methanol (2:1:20 vol:vol:vol). Five µL of each sample supernatant was injected in a Shimadzu Prominence LC-20A system (Kyoto, Japan) and analytes were separated on a silica column (150 × 2.1 mm, 3 Om Luna silica; Phenomenex, Torrance, CA) at room temperature. The mobile phase was 90% methanol containing 10 mM ammonium formate and 0.2% formic acid (volume/volume) at a flow rate of 0.2 mL/min. Mass spectrometry was performed on an API 4000 triple quadrupole mass spectrometer (AB Sciex, Canada) with electrospray ionization in the positive mode. Ion transitions used for quantitation were m/z 203→70 for both ADMA and SDMA with corresponding transitions for the internal standards. For ADMA and SDMA, recoveries averaged 102±8% and 115±6%, respectively. The intra-day coefficients of variation for ADMA were 0.3% at 0.461 µM, 2.1% at 1.71 µM and 1.0% at 3 µM and for SDMA were 1.4% at 0.737 µM, 3.7% at 2.1 µM and 0.3% at 3.7 µM; inter-day data were similar. When measured values fell below 80% of the lowest standard, a value halfway between zero and the low end of the standard curve was imputed. For other laboratory tests including urea, albumin, and β2-microglobulin, we used data collected as part of the HEMO Study.
Outcomes
The primary outcomes for our analyses were cardiac death, sudden cardiac death and first cardiovascular event (composite of first cardiovascular hospitalization or death from any cause). Secondary outcome was all-cause mortality. Cardiac death was defined as deaths due to coronary events, congestive heart failure, arrhythmias, and other heart diseases and conditions. Sudden cardiac death was defined as a witnessed death with preceding duration of symptoms less than 24 hours or unwitnessed unexpected death with symptom duration less than the interval since the last dialysis session.(17) Cardiovascular hospitalizations were defined as hospitalizations for ischemic heart disease, heart failure, arrhythmias, other cardiac conditions, hypertension, and peripheral vascular disease. Causes for death and hospitalizations in the HEMO Study were determined locally and then adjudicated by an outcomes committee that was unaware of treatment-group assignments.(18)
Other Covariates
Demographics and clinical information was available for all participants at baseline. We used the Index of Coexisting Disease (ICED) score, assessed by chart abstraction by trained nurses at baseline and then annually, for comorbidity assessment. The final ICED score ranges from 0 to 3 with higher numbers indicating greater comorbidity. We assessed dietary information that was collected at baseline and then annually using 2-day assisted recall. We assessed residual kidney function at baseline from a timed urine collection with measurement of urinary urea clearance. Other baseline data included self-reported appetite(19, 20) and the mental health index subscale of the 36-Item Short-Form Health Survey (SF-36) questionnaire, which correlates with depressive symptoms in dialysis patients.(21) We used data for systolic blood pressure, weight and volume removed on dialysis collected as per the dialysis unit routine and recorded on the monthly HEMO kinetic modelling day, the same date as the blood sample collection. We calculated relative volume removed as predialysis weight minus post dialysis weight divided by predialysis weight and body mass index (BMI) as target weight (in kilograms) divided by height (in meters) squared. We used data for Kt/Vurea and normalized protein catabolic rate (an index of protein intake) provided in the HEMO database.
Statistical Analysis
We analyzed the baseline characteristics of the participants overall and compared difference in included and excluded participants using chi-squared test for categorical variables and linear regression for continuous variables. Covariates with missing values included race (0.1%), cause of end-stage renal disease (2.3%), systolic blood pressure (0.1%), albumin (0.5%), and residual kidney function (0.1%). To avoid listwise deletion,(22) we imputed missing data with 10 data replicates using multiple imputation by chained equations method implemented by “proc mi” procedure in SAS and used “proc mianalyze” to combine the results. We censored participants at kidney transplantation or end of the study for mortality analyses and also for transfer to non-participating clinical centers for hospitalization analyses, as the hospitalization information was not collected after transfer. For survival analyses, we set the time origin as the date of dialysis initiation with at-risk time starting at the date of sample collection (left censoring; accounts for dialysis vintage prior to enrollment). We used Cox proportional hazards models to analyze the association between dimethylarginines and outcomes modeling the dimethylarginines as a natural log and as quintiles. We checked proportionality assumptions by Schoenfeld residual plots. We adjusted the Cox models for the following prespecified factors: age, sex, race, ICED score, cause of end-stage renal disease, BMI (categorized as <18, 18–25, and >25 kg/m2), systolic blood pressure (categorized as <130, 130–160 and >160 mm Hg), relative volume removed, serum albumin, and residual kidney function (urinary standard Kt/Vurea calculated from urinary urea clearance). We prespecified the following subgroup analyses: age (above or below median), sex, race (Blacks versus non-Blacks), diabetes, cardiac disease, gastrointestinal disease, BMI (<18, 18–25, or >25), albumin (above or below median), residual kidney function (any versus none), and trial interventions. We generated plots for the adjusted hazard of outcomes with the dimethylarginines modelled as a restricted cubic spline with 5 knots(23) to allow visual assessment of the association, calculating p-linearity by Wald test. Additional analyses included the following: a) association with first infection-related hospitalization or any-cause death; b) predictors of serum ADMA and SDMA using multivariable linear regression; c) association with baseline medications, depressive symptoms (mental health index), appetite, and day of the week. We considered two sided p<0.05 as statistically significant. We conducted all analyses using SAS 9.4 (SAS Institute Inc., Cary, NC) and STATA 13.0 (StataCorp LP).
RESULTS
Participant Characteristics
Baseline characteristics of the participants are presented in Table 1. Mean age of the participants was 57 years, 63% were Black and 57% were female. Mean and median concentrations of ADMA were 0.9 ±0.2 (standard deviation) µM and 0.9 (interquartile range [IQR], 0.8–1.0) µM, respectively, and of SDMA were 4.3 ±1.4 and 4.1 (IQR, 3.3–5.0) µM, respectively. Both ADMA and SDMA were moderately correlated (Pearson r = 0.418). The patients included in this study were generally similar to participants of the HEMO Study (Table S1, available as online supplementary material).
Table 1.
Baseline Characteristics of 1276 Patients from HEMO Study in total and by ADMA quintiles
| Characteristic | All | Q1: <0.745 µM |
Q2: 0.745– 0.841 µM |
Q3: 0.842– 0.938 µM |
Q4: 0.939– 1.06 µM |
Q5: ≥ 1.07 µM |
P for trend |
|---|---|---|---|---|---|---|---|
| ADMA | |||||||
| Mean, µM | 0.9 ± 0.2 | 0.7 ± 0.1 | 0.8 ± 0.0 | 0.9 ± 0.0 | 1.0 ± 0.0 | 1.3 ± 0.2 | ― |
| Median, µM | 0.9 [0.8–1.0] | 0.7 [0.6–0.7] | 0.8 [0.8–0.8] | 0.9 [0.9–0.9] | 1.0 [1.0–1.0] | 1.2 [1.1–1.3] | |
| SDMA | |||||||
| Mean, µM | 4.3 ± 1.4 | 3.6 ± 0.9 | 4.0 ± 1.2 | 4.2 ± 1.2 | 4.5 ± 1.3 | 5.1 ± 1.7 | <0.001 |
| Median, µM | 4.1 [3.3–5.0] | 3.6 [3.0–4.3] | 3.8 [3.2–4.5] | 4.1 [3.3–4.9] | 4.3 [3.5–5.2] | 4.9 [4.0–6.1] | |
| Demographics | |||||||
| Age, y | 57.5 ± 14.0 | 59.1 ± 13.1 | 57.7 ± 14.7 | 58.1 ± 14.0 | 57.2 ± 14.2 | 55.5 ± 13.8 | 0.004 |
| Female sex | 724 (56.7) | 124 (48.6) | 142 (55.9) | 146 (57.3) | 153 (60.2) | 159 (61.6) | 0.003 |
| Black race | 801 (62.8) | 188 (73.7) | 170 (66.9) | 153 (60.0) | 147 (57.9) | 143 (55.4) | <0.001 |
| Clinical Characteristics | |||||||
| Diabetes | 577 (45.2) | 105 (41.2) | 116 (45.7) | 115 (45.1) | 123 (48.4) | 118 (45.7) | 0.3 |
| Cardiac Disease | 1005 (78.8) | 191 (74.9) | 197 (77.6) | 201 (78.8) | 211 (83.1) | 205 (79.4) | 0.1 |
| ICED Score | 2.0 ± 0.8 | 1.8 ± 0.8 | 1.9 ± 0.8 | 2.0 ± 0.8 | 2.0 ± 0.8 | 2.1 ± 0.9 | <0.001 |
| Gastrointestinal Disease | 478 (37.5) | 82 (32.2) | 88 (34.6) | 97 (38.0) | 98 (38.6) | 113 (43.8) | 0.004 |
| Residual Kidney Urea Clearance, ml/min/35L TBW | 0.3 ± 0.5 | 0.3 ± 0.7 | 0.3 ± 0.4 | 0.2 ± 0.5 | 0.2 ± 0.4 | 0.2 ± 0.4 | <0.001 |
| BMI, kg/m2 | 25.8 ± 5.4 | 25.8 ± 4.8 | 26.0 ± 5.4 | 25.7 ± 5.4 | 26.0 ± 5.5 | 25.3 ± 5.9 | 0.3 |
| Body surface area, m2 | 1.8 ± 0.2 | 1.8 ± 0.2 | 1.8 ± 0.2 | 1.7 ± 0.2 | 1.8 ± 0.2 | 1.7 ± 0.2 | 0.002 |
| Dialysis Characteristics | |||||||
| Dialysis vintage, y | 3.5 ± 4.1 | 2.6 ± 3.2 | 3.2 ± 3.6 | 3.7 ± 4.3 | 3.4 ± 4.0 | 4.4 ± 5.1 | <0.001 |
| Predialysis SBP, mm Hg | 152.5 ± 25.8 | 151.3 ± 25.0 | 152.3 ± 25.3 | 152.3 ± 26.5 | 153.2 ± 26.2 | 153.4 ± 26.1 | 0.3 |
| Postdialysis Weight, kg | 70.0 ± 15.3 | 71.6 ± 13.6 | 70.7 ± 14.5 | 69.6 ± 15.0 | 70.0 ± 16.3 | 68.2 ± 16.7 | 0.01 |
| Relative Volume Removed, % | 4.1 ± 1.7 | 4.2 ± 1.7 | 4.1 ± 1.7 | 4.0 ± 1.7 | 4.0 ± 1.7 | 4.2 ± 1.7 | 0.9 |
| Dose Intervention | 635 (49.8) | 128 (50.2) | 117 (46.1) | 129 (50.6) | 130 (51.2) | 131 (50.8) | 0.6 |
| Flux Intervention | 636 (49.8) | 137 (53.7) | 116 (45.7) | 133 (52.2) | 125 (49.2) | 125 (48.4) | 0.5 |
| Session length, min | 206.8 ± 28.2 | 207.7 ± 27.3 | 205.5 ± 27.4 | 206.9 ± 29.1 | 205.9 ± 29.0 | 208.1 ± 28.4 | 0.7 |
| Blood Flow Rate, ml/min | 343.1 ± 60.7 | 351.2 ± 60.0 | 344.0 ± 58.7 | 344.9 ± 60.7 | 343.1 ± 58.7 | 332.4 ± 64.0 | <0.001 |
| Dialysate Flow Rate, ml/min | 673.1 ± 129.8 | 685.5 ± 127.6 | 661.4 ± 133.7 | 680.4 ± 128.3 | 673.6 ± 130.3 | 664.7 ± 128.3 | 0.2 |
| Predialysis Laboratory Tests | |||||||
| Serum urea nitrogen, mg/dL | 59.7 ± 18.7 | 61.7 ± 20.3 | 60.9 ± 19.2 | 59.0 ± 17.8 | 58.2 ± 18.1 | 58.6 ± 18.1 | 0.03 |
| Single-pool Kt/Vurea | 1.5 ± 0.3 | 1.5 ± 0.3 | 1.5 ± 0.3 | 1.5 ± 0.3 | 1.5 ± 0.3 | 1.5 ± 0.3 | 0.3 |
| Serum albumin, g/dL | 3.6 ± 0.4 | 3.6 ± 0.4 | 3.6 ± 0.4 | 3.6 ± 0.4 | 3.6 ± 0.4 | 3.6 ± 0.4 | 0.09 |
| Serum β2-microglobulin, mg/L | 36.7 ± 14.2 | 34.0 ± 13.8 | 35.4 ± 13.0 | 36.1 ± 14.9 | 37.7 ± 14.2 | 40.1 ± 14.4 | <0.001 |
| Equilibrated nPCR, g/kg/d | 1.0 ± 0.3 | 1.0 ± 0.3 | 1.0 ± 0.3 | 1.0 ± 0.2 | 1.0 ± 0.3 | 1.0 ± 0.2 | 0.1 |
Note: Values for categorical variables are given as count (percentage); values for continuous variables, as mean ± standard deviation or median [interquartile range]. Conversion factor for serum urea nitrogen in mg/dL to mmol/L, ×0.357.
Abbreviations: Asymmetric dimethylarginine (ADMA); symmetric dimethylarginine (SDMA); BMI, body mass index; HEMO, Hemodialysis; ICED, Index of Coexistent Disease; nPCR, normalized protein catabolic rate; Q, quintile; SBP, systolic blood pressure; TBW, total body water
Outcomes During Follow-up
There were 220 cardiac deaths during 3,282 person-years of follow-up (median, 2.3 years) with a crude cardiac death rate of 67 per 1000 person-years. The adjudicated causes of cardiac death included ischemic heart disease (62.3%), congestive heart failure (11.4%), arrhythmias and other conduction disorders (15.0%), and other heart diseases (11.4%). During follow-up, there were 126 sudden cardiac deaths (crude mortality rate, 38 per 1000 person-years), 644 cardiovascular events or any-cause deaths (crude event rate, 274 per 1000 person-years) and 565 any-cause deaths (crude mortality rate, 172 per 1000 person-years).
Association Between ADMA and Outcomes
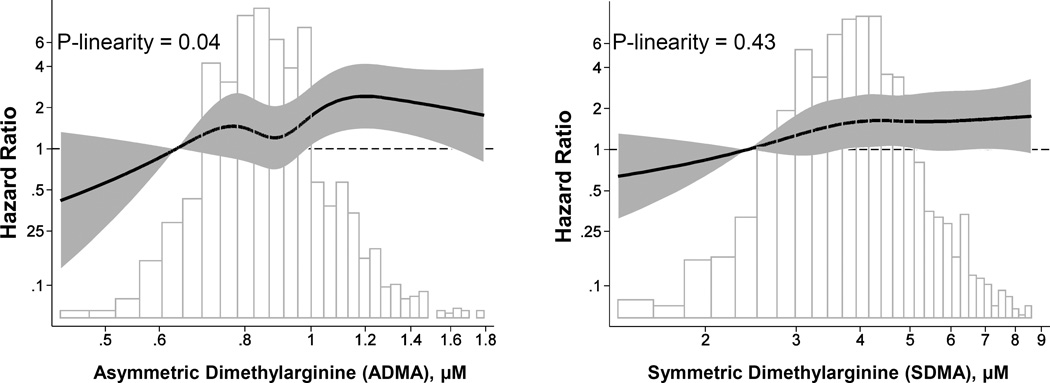
In fully-adjusted models (Table 2), each doubling of ADMA was associated with an 83% higher risk of cardiac death (hazard ratio [HR] per 2-fold higher concentration, 1.83; 95% confidence interval [CI], 1.29–2.58; p<0.001). With ADMA modeled as quintiles (Table S2), there was a 2.1-fold higher risk of cardiac death for patients in the highest quintile (≥1.07 µM) compared with the lowest quintile (<0.745 µM) and 76% higher risk when comparing those in the highest quintile (≥1.07 µM) to those with lower (<1.07 µM) ADMA concentrations (Table S3). Higher ADMA concentrations were also associated (Table 2) with a 79% higher risk of sudden cardiac death (p=0.006), 50% higher risk of first cardiovascular event (p<0.001) and 44% higher risk of any-cause death (p=0.003). Figure 1 and Figure S1 present the fully adjusted association between ADMA and outcomes. The association between ADMA and outcomes was generally linear for cardiac death (p=0.04) and sudden cardiac death (p=0.1) with suggestion of non-linearity for all-cause mortality (p=0.001) and first cardiovascular event (p=0.03). Table S4 presents the subgroup analyses. The results should be interpreted with caution due to multiple comparisons, and a p-value of 0.05/11=0.004 is suggested as a significant interaction between the groups. No significant differences were noted in subgroups except for a trend towards higher risk of sudden cardiac death with higher ADMA concentrations among patients without diabetes (uncorrected p for interaction = 0.03) and among patients randomized to high-flux dialysis (uncorrected p for interaction = 0.04).
Table 2.
Association of ADMA and SDMA with Outcomes in the HEMO Study
| Model 1a | Model 2b | Model 3c | Model 4: Final Modeld | Model 5: Additional Analysese |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| HR* (95% CI) | P | HR* (95% CI) | P | HR* (95% CI) | P | HR* (95% CI) | P | HR* (95% CI) | P | |
| Cardiac Death | ||||||||||
| (N=1276; Events=220; IR=67) | ||||||||||
| ADMA | 2.16 (1.51–3.09) | <0.001 | 2.16 (1.61–2.89) | <0.001 | 1.85 (1.32–2.60) | <0.001 | 1.83 (1.29–2.58) | <0.001 | 1.78 (1.19–2.67) | 0.005 |
| SDMA | 0.96 (0.74–1.25) | 0.7 | 1.30 (0.98–1.72) | 0.07 | 1.42 (1.05–1.93) | 0.02 | 1.40 (1.03–1.92) | 0.03 | 1.20 (0.86–1.68) | 0.3 |
| Sudden Cardiac Death | ||||||||||
| (N=1276; Events=126; IR=38) | ||||||||||
| ADMA | 2.16 (1.38–3.36) | <0.001 | 2.15 (1.49–3.11) | <0.001 | 1.80 (1.20–2.71) | 0.005 | 1.79 (1.19–2.69) | 0.006 | 1.69 (0.99–2.88) | 0.06 |
| SDMA | 0.92 (0.61–1.40) | 0.7 | 1.23 (0.83–1.82) | 0.3 | 1.42 (0.97–2.08) | 0.07 | 1.40 (0.96–2.06) | 0.08 | 1.22 (0.77–1.93) | 0.4 |
| First CV Event or Any-Cause Death | ||||||||||
| (N=1186; Events=644; IR=274) | ||||||||||
| ADMA | 1.59 (1.30–1.94) | <0.001 | 1.60 (1.29–1.99) | <0.001 | 1.50 (1.20–1.86) | <0.001 | 1.50 (1.20–1.87) | <0.001 | 1.73 (1.35–2.22) | <0.001 |
| SDMA | 0.73 (0.62–0.87) | <0.001 | 0.95 (0.80–1.12) | 0.5 | 0.98 (0.83–1.16) | 0.8 | 0.98 (0.83–1.16) | 0.8 | 0.85 (0.70–1.02) | 0.09 |
| Any-Cause Death | ||||||||||
| (N=1276; Events=565; IR=172) | ||||||||||
| ADMA | 1.61 (1.27–2.04) | <0.001 | 1.63 (1.31–2.03) | <0.001 | 1.44 (1.14–1.82) | 0.002 | 1.44 (1.13–1.83) | 0.003 | 1.42 (1.06–1.89) | 0.02 |
| SDMA | 0.85 (0.69–1.04) | 0.1 | 1.13 (0.94–1.36) | 0.2 | 1.21 (1.01–1.46) | 0.04 | 1.21 (1.00–1.47) | 0.05 | 1.12 (0.89–1.41) | 0.3 |
| First Infection-Related Hospitalization or Any-Cause Death | ||||||||||
| (N=1183; Events=663; IR=293) | ||||||||||
| ADMA | 1.56 (1.32–1.84) | <0.001 | 1.55 (1.30–1.84) | <0.001 | 1.42 (1.19–1.70) | <0.001 | 1.41 (1.18–1.69) | <0.001 | 1.55 (1.23–1.97) | <0.001 |
| SDMA | 0.85 (0.71–1.00) | 0.05 | 0.98 (0.83–1.14) | 0.8 | 1.04 (0.88–1.23) | 0.6 | 1.04 (0.88–1.23) | 0.7 | 0.90 (0.75–1.08) | 0.3 |
Abbreviation: CV, cardiovascular; IR, incidence rate (per 1000 person-years); HEMO, Hemodialysis; HR, hazard ratio; CI, confidence interval; ADMA, asymmetric dimethylarginine; SDMA, symmetric dimethylarginine
Model 1 was unadjusted.
Model 2 adjusted for age, sex, and race.
Model 3 adjusted for variables in model 2 + Index of Coexisting Disease severity score, cause of end-stage renal disease, body mass index (<18, 18–25, or >25 kg/m2), systolic blood pressure (<130, 130–160, or >160 mm Hg), albumin, and relative volume removed on dialysis.
Model 4 adjusted for variables in Model 3 + residual kidney function (urinary standardized Kt/Vurea calculated from urinary urea clearance).
Model 5 adjusted for variables in Model 4 + SDMA in models for ADMA and ADMA in models for SDMA.
HR represents increase in risk per 2-fold increase in ADMA or SDMA concentrations. Modeled as natural log transformed variable/natural log of 2.
Figure 1. Association of ADMA and SDMA with Cardiac Death in the HEMO Study.
Relative hazard predicted using Cox proportional hazards regression adjusted for age, sex, race, Index of Coexisting Disease (ICED) severity score, cause of end-stage renal disease, body mass index (categorized as <18, 18–25 and >25 kg/m2), systolic blood pressure (categorized as <130, 130–160 and >160 mm Hg), albumin, relative volume removed on dialysis, and residual kidney function (urinary standardized Kt/Vurea calculated from urinary urea clearance). ADMA and SDMA are modeled as a restricted cubic spline with 5 knots (5th, 27.5th, 50th, 72.5th and 95th percentiles); 10th percentile is used as the reference (HR = 1). The lines represent the adjusted HR and the shaded area is the 95% CI of the HR. Vertical bars are the frequency histogram, showing the distribution of the solutes. Extreme observations, defined as values >99th percentile, are excluded; ADMA >1.8 µM (n=12) and SDMA >8.6 µM (n=12).
Panel A (left): Asymmetric Dimethylarginine (ADMA) and Cardiac Death
Panel B (right): Symmetric Dimethylarginine (SDMA) and Cardiac Death.
Association Between SDMA and Outcomes
In fully-adjusted models (Table 2 and Table S2), each 2-fold higher SDMA was associated with a 40% higher risk of cardiac death (p=0.03), 40% higher risk of sudden cardiac death (p=0.08) and 21% higher risk of any-cause death (p=0.05). There were no statistically significant interactions in subgroup analyses (Table S5).
Other Analyses
Additionally, ADMA was associated with the risk of first infection-related hospitalization or any-cause death (HR, 1.41; 95% CI, 1.18–1.69; p<0.001; Table 2 and Table S2). Addition of SDMA to the fully adjusted model of the association of ADMA with outcomes (Model 5) did not change any of the observed associations. However, after adjustment for ADMA in models for SDMA, the association of SDMA with outcomes were attenuated and were no longer statistically significant (Table 2).
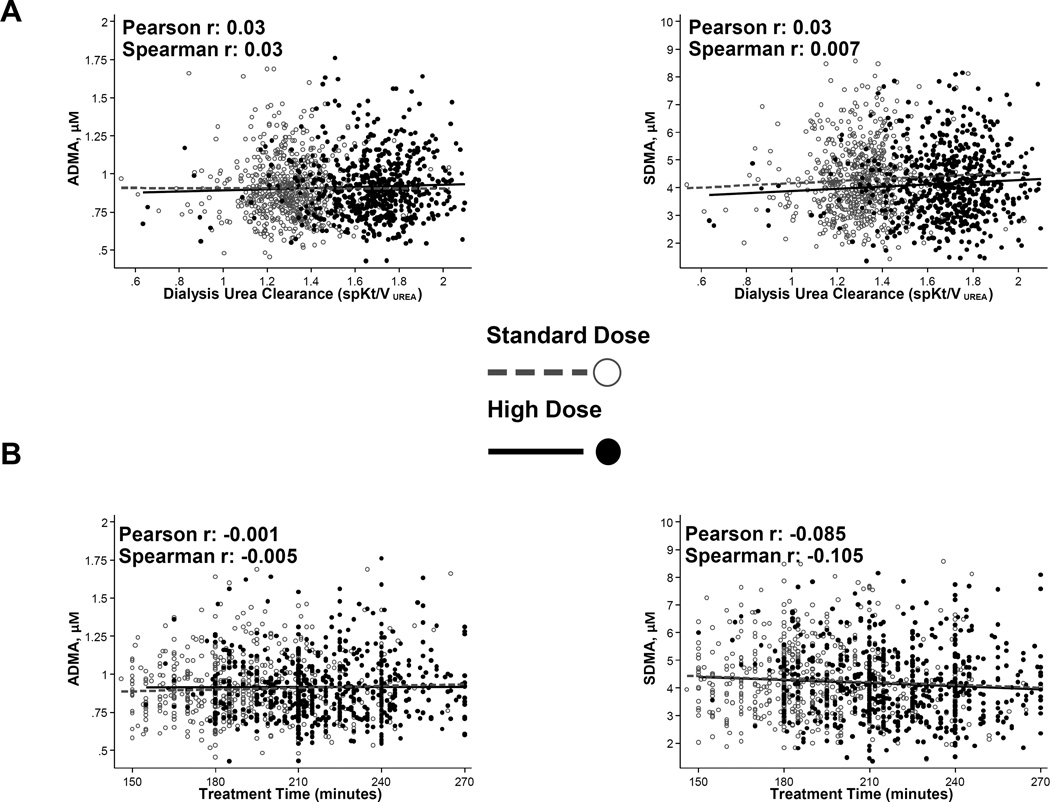
Table 3 presents the results of univariate and multivariable cross-sectional associations of the solutes. These predictors explained little variability in ADMA and SDMA concentration; the best-fit model only explained 6.3% variability in ADMA and 21.4% variability in SDMA. Notably, there was no association of ADMA or SDMA with treatment time or Kt/Vurea (Figure 2). Medications prescribed at baseline were not associated with ADMA concentrations (Table S6). Calcium channel blocker use was associated with higher SDMA concentrations, whereas aspirin, warfarin and nitrate use were associated with lower SDMA concentrations. Depressive symptoms, assessed by mental health index, were not associated with ADMA (Pearson r = −0.031; p=0.3) or SDMA (Pearson r = 0.003; p=0.9). The association between poor appetite and ADMA concentrations was inconsistent (Table S7). There was no association between appetite and SDMA. Average ADMA concentrations were lowest at the beginning of the week (Monday or Tuesday; p for trend=0.001; Table S8). There was no association between SDMA and day of sample collection.
Table 3.
Predictors of ADMA and SDMA in 1276 Patients of HEMO Study
| ADMA* | SDMA** | |||||||
|---|---|---|---|---|---|---|---|---|
| Univariate | Multivariable*** | Univariate | Multivariable | |||||
| Characteristics and Modeling | β (SE) | p | β (SE) | p | β (SE) | p | β (SE) | p |
| Demographics | ||||||||
| Age, per 10-y older | −0.011 (0.005) | 0.02 | — | — | −0.279 (0.026) | <0.001 | −0.178 (0.028) | <0.001 |
| Sex, female vs male | 0.031 (0.013) | 0.02 | 0.045 (0.013) | <0.001 | −0.216 (0.078) | 0.006 | — | — |
| Race, black vs nonblack | −0.048 (0.013) | <0.001 | −0.051 (0.014) | <0.001 | 0.008 (0.080) | 0.9 | — | — |
| Clinical Characteristics | ||||||||
| Diabetes, Yes vs No | 0.015 (0.013) | 0.2 | — | — | −0.756 (0.075) | <0.001 | −0.372 (0.078) | <0.001 |
| Cardiac Disease, Yes vs No | 0.018 (0.016) | 0.3 | — | — | −0.179 (0.094) | 0.06 | — | — |
| ICED Score, <3 vs 3 | 0.057 (0.014) | <0.001 | 0.047 (0.014) | <0.001 | −0.129 (0.082) | 0.1 | — | — |
| Gastrointestinal Disease, Yes vs No | 0.044 (0.013) | 0.001 | — | — | −0.168 (0.080) | 0.04 | — | — |
| Residual Kidney Urea Clearance, per 0.5–mL/min/35 L TBW greater |
−0.021 (0.007) | 0.001 | — | — | −0.182 (0.039) | <0.001 | — | — |
| BMI,≥25 vs <25 kg/m2 | −0.011 (0.013) | 0.4 | — | — | −0.567 (0.076) | <0.001 | — | — |
| Body Surface Area,per 0.5-m2 greater | −0.035 (0.016) | 0.03 | — | — | −0.477 (0.097) | <0.001 | — | — |
| Dialysis Characteristics | ||||||||
| Dialysis vintage, per 1-y greater | 0.009 (0.002) | <0.001 | 0.008 (0.002) | <0.001 | 0.074 (0.009) | <0.001 | 0.034 (0.009) | <0.001 |
| Predialysis SBP, per 10–mm Hg greater | 0.003 (0.003) | 0.2 | — | — | 0.017 (0.015) | 0.3 | — | — |
| Postdialysis Weight, per 10-kg greater | −0.005 (0.004) | 0.2 | — | — | −0.191 (0.025) | <0.001 | −0.141(0.024) | <0.001 |
| Relative Volume Removed, per 1% greater | −0.002 (0.004) | 0.7 | — | — | 0.035 (0.023) | 0.1 | −0.045 (0.023) | 0.05 |
| Dose Intervention, High Dose vs Standard Dose | 0.005 (0.013) | 0.7 | — | — | −0.172 (0.077) | 0.03 | −0.161(0.072) | 0.03 |
| Flux Intervention, High Flux vs Low Flux | −0.022 (0.013) | 0.09 | — | — | −0.341 (0.077) | <0.001 | −0.187 (0.075) | 0.01 |
| Session length, per 30-min greater | 0.005 (0.007) | 0.4 | — | — | −0.126 (0.041) | 0.002 | — | — |
| Blood Flow Rate, per 50-ml/min greater | −0.016 (0.005) | 0.003 | — | — | −0.101 (0.032) | 0.001 | — | — |
| Dialysate Flow Rate, per 100-ml/min greater | −0.008 (0.005) | 0.1 | — | — | −0.049 (0.030) | 0.1 | — | — |
| Predialysis Laboratory Tests | ||||||||
| Serum Urea Nitrogen, per 10-mg/dL greater | −0.007 (0.003) | 0.03 | — | — | 0.088 (0.020) | <0.001 | 0.077 (0.020) | <0.001 |
| Single-pool Kt/Vurea, per 0.2-unit greater | 0.003 (0.005) | 0.5 | — | — | −0.013 (0.028) | 0.6 | — | — |
| Serum albumin, per 0.5-g/dL greater | −0.014 (0.008) | 0.1 | — | — | 0.283 (0.049) | <0.001 | 0.115 (0.048) | 0.02 |
| Serum β2-microglobulin, per 10-mg/L greater | 0.022 (0.005) | <0.001 | 0.019 (0.005) | <0.001 | 0.262 (0.027) | <0.001 | 0.139 (0.028) | <0.001 |
| Equilibrated nPCR, per 0.2-g/kg/d greater | −0.008 (0.005) | 0.1 | — | — | 0.129 (0.030) | <0.001 | — | — |
Abbreviations: ADMA, asymmetric dimethylarginine; BMI, body mass index; SDMA, symmetric dimethylarginine; SBP, systolic blood pressure SE, Standard Error; HEMO, Hemodialysis; ICED, Index of Coexistent Disease; nPCR, normalized protein catabolic rate; RMSE, root mean squared error; TBW, total body water
Note: β coefficients are from linear regression of ADMA or SDMA (natural scale) on predictors in separate models (univariate associations) and in a forward selection model with model entry specified as p≤0.05 (multivariable model). Conversion factor for serum urea nitrogen in mg/dL to mmol/L, ×0.357.
ADMA mean, 0.9 ±0.2 (standard deviation) µM; median, 0.9 [interquartile range, 0.8–1.0] µM
SDMA mean, 4.3 ± 1.4 (standard deviation) µM; median, 4.1 [interquartile range, 3.3–5.0] µM
model characteristics: RMSE = 0.051; R^2=6.3%
model characteristics: RMSE = 1.522; R^2=21.4%
Figure 2. Association of ADMA and SDMA with single-pool Kt/Vurea and Dialysis Treatment Time in the HEMO Study.
Scatterplot of ADMA and SDMA concentrations and single-pool Kt/Vurea (Panel A) and dialysis treatment time (Panel B). Patients randomized to standard dose intervention are represented by open circles and those randomized to high dose intervention are represented by solid circles. Lines represent linear regression of the solute on single-pool Kt/Vurea or treatment time with broken line representing standard dose group and solid line representing high dose group. Pearson and Spearman correlation coefficients are also displayed.
DISCUSSION
We measured ADMA and SDMA in samples from a large, national, multicenter study of prevalent hemodialysis patients in the United States and report an association between these solutes and the risk of death and cardiovascular events. Predialysis ADMA and SDMA concentrations were 2-fold and 8-fold higher, respectively, compared with concentrations reported in patients with normal kidney function. Dialysis dose (Kt/Vurea) or treatment time were not associated with ADMA or SDMA concentrations. Each 2-fold higher ADMA was associated with an 83% higher risk of cardiac death, 79% higher risk of sudden cardiac death, 50% higher risk of first cardiovascular event and 44% higher risk of any-cause death. Patients in the highest quintile of ADMA (≥1.07 µM) had a 2-fold higher risk of cardiac death compared to those in the lower quintile. There were relatively weaker associations of SDMA with outcomes; 2-fold higher SDMA was associated with a 40% higher risk of cardiac death and trends towards higher risk of sudden cardiac death and any-cause death. The SDMA associations were no longer statistically significant after adjustment for ADMA.
Patients undergoing dialysis continue to have high rates of morbidity and mortality despite improvements in general medical care and dialyzer urea removal (Kt/Vurea).(1) The age, sex and race adjusted median 3-year survival for hemodialysis patients is 55%, and 5-year survival is only 40%.(1) Cardiovascular disease accounts for about half of all deaths in dialysis patients, but the mechanisms of accelerated cardiovascular disease remain elusive; retention of uremic toxins is a likely culprit.(4) Increasing dialyzer urea clearance (Kt/Vurea) without targeting specific uremic toxins does not improve survival, as demonstrated by the results of the HEMO Study.(15) The HEMO Study was designed with the hypothesis that increasing Kt/Vurea will increase removal of the unknown uremic toxins and improve survival. The high-dose group received 30% higher dialyzer urea clearance (average single-pool Kt/Vurea, 1.7) than the standard-dose group (average single-pool Kt/Vurea, 1.4). However, the higher dose did not reduce the risk of death in study participants.(15) Results of other trials attempting reduction of unknown uremic toxins by improving dialyzer urea clearance have also generally been negative.(24–27) It is likely that some of the residual illness in contemporary dialysis patients is due to uremic toxins that are not effectively lowered by conventional dialysis.(28) We previously reported that compared to the standard-dose group, the high-dose group in the HEMO Study had significantly lower predialysis urea nitrogen concentrations (relative difference, −9%; p<0.001), however, the predialysis ADMA concentrations were not lower (relative difference, 0.5%; p=0.7) and SDMA concentrations were only slightly lower (relative difference, −4%; p=0.02).(29) The present study suggests that the elevated concentrations of ADMA and, to a lesser extent, SDMA are cardiovascular risk factors in hemodialysis patients, and therefore, potential therapeutic targets for a randomized controlled trial.
Metabolism of arginine-containing proteins by the enzyme PRMT (protein arginine N-methyl transferase) endogenously produce ADMA.(30, 31) Of the generated ADMA, only 20% is excreted unchanged in the urine, whereas 80% is removed through non-renal clearance by the enzyme DDAH (dimethylarginine dimethylaminohydrolase).(6, 7); DDAH is expressed in many tissues, including the kidney. In kidney failure ADMA accumulates and contributes to cardiovascular toxicity by inhibiting nitric oxide synthase (NOS) and reducing nitric oxide (NO) production.(8, 32, 33) A central role is played by NO in vascular biology and health; it leads to vasodilation and reduces platelet aggregation, vascular smooth muscle proliferation, oxidation of low-density lipoprotein cholesterol and free oxygen species generation.(34, 35)
Higher ADMA concentrations are associated with mortality in a number of general population studies.(9, 10, 12) Prior studies have also reported association of ADMA with outcomes in the dialysis patients. In the Cardiovascular Risk Extended Evaluation in Dialysis (CREED) cohort from Europe (N=225), ADMA was associated with the risk of death and cardiovascular events.(14) In the Cognition and Dialysis Study from the United States (N=259), ADMA was associated with death in Whites but not in Blacks (p for interaction=0.03).(13)
Our study extends these findings in a large, national, multicenter study with carefully assessed comorbidities and physician-adjudicated outcomes. Our findings of 83% higher risk of cardiac death, 79% higher risk of sudden cardiac death, 50% higher risk of first cardiovascular event and 44% higher risk of any-cause death with each 2-fold higher ADMA, extends previously noted associations with all-cause mortality in dialysis patients. Similar to the Cognition and Dialysis Study cohort, we noted that the ADMA concentrations were lower in Blacks compared with Whites, however, we did not find that the risk of outcomes differed by race.
Our findings support the evidence for toxicity of ADMA(5, 36) and highlight the need for a randomized controlled trial with ADMA as a therapeutic target. Elevated ADMA concentration may be reduced by down regulating synthesis (PRMT) and/or enhancing endogenous removal (DDAH).(5) PRMT may be downregulated (reducing ADMA production) by reducing cholesterol concentration.(7) DDAH may be modified by reducing inhibitory influences (proton pump inhibitors,(37) smoking(5)), statins (38) or via upregulation by PPAR-γ (peroxisome proliferator-activated receptor γ) ligands (such as pioglitazone), (39) omega-3 fatty acids(40), and erythropoietin.(41) Interestingly, angiotensin-converting enzyme inhibitors may increase ADMA concentrations in hemodialysis patients via bradykinin signaling and may need discontinuation in select patients.(42) Upregulating NOS, increasing NO availability, and augmenting NO signaling are also potential therapeutic strategies.(35) Such therapies may be selectively targeted to patients with elevated ADMA concentrations, personalizing management of uremia.
SDMA is generated from arginine-containing proteins by the action of PRMT.(5) It is almost completely excreted in the urine and its concentrations increase to a greater degree than those of ADMA as kidney function declines.(43) Once considered to be an “inert” isomer of ADMA, SDMA reduces NO availability by reducing availability of L-arginine to NOS(44) and scavenging NO.(45) Additionally, SDMA may have direct inflammatory and atherogenic effects.(46, 47) It has been associated with death and cardiovascular events in general population studies.(10, 11) In the Ludwigshafen Risk and Cardiovascular Health study (N=3229), SDMA was associated with all-cause and cardiovascular mortality in patients undergoing cardiac catheterization.(10) In dialysis patients, however, no association between SDMA concentrations and mortality were noted in a cohort of hemodialysis patients in Italy (N=288).(48)
We report that SDMA is associated with the risk of cardiac death in dialysis patients. However, the association of SDMA with outcomes was attenuated after adjustment for ADMA, suggesting that perhaps some of the risk associated with SDMA study could be explained by elevated ADMA concentrations. Both SDMA and ADMA are metabolized by the mitochondrial enzyme AGXT2 (alanine-glyoxylate aminotransferase 2).(8) Elevated concentration of both ADMA and SDMA may be due to mitochondrial dysfunction in dialysis patients with reduction in AGXT2 activity. Another possibility of the relatively weaker association of SDMA with outcomes is the presence of a threshold effect where the high SDMA concentrations present in dialysis patients are uniformly toxic. SDMA is highly correlated with kidney function(43), and confounding by residual kidney function can also be potential concern. However, the HEMO Study is uniquely suitable to address this limitation as it excluded patients with residual urea clearance >1.5 mL/min. Our findings of the risk of cardiac mortality with SDMA were similar in patients before and after adjustment for residual kidney function and in patients with or without residual kidney function (p for interaction=0.8). In one animal study, chronic SDMA infusion did not have any effect on blood pressure, fibrosis or NO production, which suggests that perhaps SDMA may not be associated with toxicity.(49)
There are some limitations to our study. Both ADMA and SDMA were measured at single time point and the concentration may change over time. Freeze-thaw cycles may also effect metabolite concentrations, however, both ADMA and SDMA are robust to multiple freeze/thaw cycles.(50, 51) We did not measure L-arginine, which is the substrate for NO synthase. However, clinical trials have failed to show benefits of large L-arginine supplementation, suggesting that the NO pathway may not be modifiable by increasing L-arginine supply.(52–55) These limitations are balanced by major strengths of our study which include its large, national, prospective design, exclusion of patients with significant residual kidney function, careful collection of samples, long duration of follow-up and carefully adjudicated cardiovascular outcomes.
In conclusion, the dimethylarginines ADMA and SDMA are associated with cardiovascular morbidity and mortality in hemodialysis patients. Because the concentrations of ADMA and SDMA are not effectively reduced by conventional hemodialysis,(56) preventing their toxicity will require reducing their production, enhancing non-renal clearance or modifying downstream pathways. Our findings present opportunities for developing targeted therapeutics and testing if personalized uremia management with individualized therapies targeted to specific uremic toxins will be effective in reducing the extraordinarily high risk of death in dialysis patients. These strategies must be tested in a carefully designed randomized clinical trial.
Supplementary Material
Acknowledgments
We thank the HEMO Study participants, investigators, and staff for their contributions to the HEMO Study.
Support: CHOICE was supported by R01-HS-008365 (Agency for Healthcare Quality and Research AHRQ) from 7/1994 to 6/1999, by RO1-HL-62985 (National Heart, Lung, and Blood Institute (NHLBI) from 9/00 to 6/06, R01-DK-059616 (National Institute of Diabetes & Digestive & Kidney Diseases NIDDK) from 9/2000 to 6/2005 and by R01-DK-080123 from 8/2008 to 1/2016 (National Institute of Diabetes & Digestive & Kidney Diseases NIDDK). Drs Shafi (K23-DK-083514 and R03-DK-104012) and Powe (in part by R01-DK-080123 and K24 DK002643) were supported by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Financial Disclosure: The authors declare that they have no other relevant financial interests.
Contributions: Research idea and study design: NRP, THH, TWM, MLM, TS; data acquisition: XH, THH, TWM; data analysis/interpretation: all Authors; statistical analysis: TS, SH; supervision: NRP, THH, TWM. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved. TS takes responsibility that this study has been reported honestly, accurately, and transparently; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned have been explained.
Supplementary Material
Table S1: Comparison of baseline characteristics of included vs excluded participants.
Table S2: Association of ADMA and SDMA quintiles and outcomes.
Table S3: Association of ADMA categorized and outcomes.
Table S4: Subgroup analyses for association between ADMA and outcomes.
Table S5: Subgroup analyses for association between SDMA and outcomes.
Table S6: Association of baseline medication use with ADMA and SDMA concentrations.
Table S7: Association of baseline appetite with ADMA and SDMA concentrations.
Table S8: Day of the week and solute concentration.
Figure S1: Association of ADMA and SDMA with outcomes.
Note: The supplementary material accompanying this article (doi:_______) is available at www.ajkd.org
Supplementary Material Descriptive Text for Online Delivery
Supplementary Table S1 (PDF). Comparison of baseline characteristics of included vs excluded participants.
Supplementary Table S2 (PDF). Association of ADMA and SDMA quintiles and outcomes.
Supplementary Table S3 (PDF). Association of ADMA categorized and outcomes.
Supplementary Table S4 (PDF). Subgroup analyses for association between ADMA and outcomes.
Supplementary Table S5 (PDF). Subgroup analyses for association between SDMA and outcomes.
Supplementary Table S6 (PDF). Association of baseline medication use with ADMA and SDMA concentrations.
Supplementary Table S7 (PDF). Association of baseline appetite with ADMA and SDMA concentrations.
Supplementary Table S8 (PDF). Day of the week and solute concentration.
Supplementary Figure S1 (PDF). Association of ADMA and SDMA with outcomes.
REFERENCES
- 1.United States Renal Data System. 2015 USRDS annual data report: Epidemiology of kidney disease in the United States. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; 2015. [Google Scholar]
- 2.Foley RN, Parfrey PS, Sarnak MJ. Epidemiology of cardiovascular disease in chronic renal disease. J Am Soc Nephrol. 1998;9:S16–S23. [PubMed] [Google Scholar]
- 3.Himmelfarb J, Ikizler TA. Hemodialysis. N Engl J Med. 2010;363:1833–1845. doi: 10.1056/NEJMra0902710. [DOI] [PubMed] [Google Scholar]
- 4.Meyer TW, Hostetter TH. Approaches to uremia. J Am Soc Nephrol. 2014;25:2151–2158. doi: 10.1681/ASN.2013121264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schepers E, Speer T, Bode-Boger SM, Fliser D, Kielstein JT. Dimethylarginines ADMA and SDMA: the real water-soluble small toxins? Semin Nephrol. 2014;34:97–105. doi: 10.1016/j.semnephrol.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 6.Vallance P, Leone A, Calver A, Collier J, Moncada S. Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet. 1992;339:572–575. doi: 10.1016/0140-6736(92)90865-z. [DOI] [PubMed] [Google Scholar]
- 7.Kielstein JT, Frolich JC, Haller H, Fliser D. ADMA (asymmetric dimethylarginine): an atherosclerotic disease mediating agent in patients with renal disease? Nephrol Dial Transplant. 2001;16:1742–1745. doi: 10.1093/ndt/16.9.1742. [DOI] [PubMed] [Google Scholar]
- 8.Caplin B, Leiper J. Endogenous nitric oxide synthase inhibitors in the biology of disease: markers, mediators, and regulators? Arterioscler Thromb Vasc Biol. 2012;32:1343–1353. doi: 10.1161/ATVBAHA.112.247726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boger RH, Sullivan LM, Schwedhelm E, et al. Plasma asymmetric dimethylarginine and incidence of cardiovascular disease and death in the community. Circulation. 2009;119:1592–1600. doi: 10.1161/CIRCULATIONAHA.108.838268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meinitzer A, Kielstein JT, Pilz S, et al. Symmetrical and asymmetrical dimethylarginine as predictors for mortality in patients referred for coronary angiography: the Ludwigshafen Risk and Cardiovascular Health study. Clinical chemistry. 2011;57:112–121. doi: 10.1373/clinchem.2010.150854. [DOI] [PubMed] [Google Scholar]
- 11.Schulze F, Carter AM, Schwedhelm E, et al. Symmetric dimethylarginine predicts all-cause mortality following ischemic stroke. Atherosclerosis. 2010;208:518–523. doi: 10.1016/j.atherosclerosis.2009.06.039. [DOI] [PubMed] [Google Scholar]
- 12.Willeit P, Freitag DF, Laukkanen JA, et al. Asymmetric dimethylarginine and cardiovascular risk: systematic review and meta-analysis of 22 prospective studies. J Am Heart Assoc. 2015;4:e001833. doi: 10.1161/JAHA.115.001833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Drew DA, Tighiouart H, Scott T, et al. Asymmetric dimethylarginine, race, and mortality in hemodialysis patients. Clin J Am Soc Nephrol. 2014;9:1426–1433. doi: 10.2215/CJN.00770114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zoccali C, Bode-Boger S, Mallamaci F, et al. Plasma concentration of asymmetrical dimethylarginine and mortality in patients with end-stage renal disease: a prospective study. Lancet. 2001;358:2113–2117. doi: 10.1016/s0140-6736(01)07217-8. [DOI] [PubMed] [Google Scholar]
- 15.Eknoyan G, Beck GJ, Cheung AK, et al. Effect of dialysis dose and membrane flux in maintenance hemodialysis. N Engl J Med. 2002;347:2010–2019. doi: 10.1056/NEJMoa021583. [DOI] [PubMed] [Google Scholar]
- 16.Greene T, Beck GJ, Gassman JJ, et al. Design and statistical issues of the hemodialysis (HEMO) study. Control Clin Trials. 2000;21:502–525. doi: 10.1016/s0197-2456(00)00062-3. [DOI] [PubMed] [Google Scholar]
- 17.Shastri S, Tangri N, Tighiouart H, et al. Predictors of sudden cardiac death: a competing risk approach in the hemodialysis study. Clin J Am Soc Nephrol. 2012;7:123–130. doi: 10.2215/CJN.06320611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rocco MV, Yan G, Gassman J, et al. Comparison of causes of death using HEMO Study and HCFA end-stage renal disease death notification classification systems. The National Institutes of Health-funded Hemodialysis. Health Care Financing Administration. Am J Kidney Dis. 2002;39:146–153. doi: 10.1053/ajkd.2002.29905. [DOI] [PubMed] [Google Scholar]
- 19.Burrowes JD, Larive B, Chertow GM, et al. Self-reported appetite, hospitalization and death in haemodialysis patients: findings from the Hemodialysis (HEMO) Study. Nephrol Dial Transplant. 2005;20:2765–2774. doi: 10.1093/ndt/gfi132. [DOI] [PubMed] [Google Scholar]
- 20.Thong MS, van Dijk S, Noordzij M, et al. Symptom clusters in incident dialysis patients: associations with clinical variables and quality of life. Nephrol Dial Transplant. 2009;24:225–230. doi: 10.1093/ndt/gfn449. [DOI] [PubMed] [Google Scholar]
- 21.Kang EW, Pike F, Ramer S, et al. The association of mental health over time with cardiac outcomes in HEMO study patients. Clin J Am Soc Nephrol. 2012;7:957–964. doi: 10.2215/CJN.06730711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Donders AR, van der Heijden GJ, Stijnen T, Moons KG. Review: a gentle introduction to imputation of missing values. J Clin Epidemiol. 2006;59:1087–1091. doi: 10.1016/j.jclinepi.2006.01.014. [DOI] [PubMed] [Google Scholar]
- 23.Harrell FE., Jr . Regression Modeling Strategies: With Applications to Linear Models, Logistic Regression, and Survival Analysis. New York: Springer; 2001. [Google Scholar]
- 24.Locatelli F, Martin-Malo A, Hannedouche T, et al. Effect of membrane permeability on survival of hemodialysis patients. J Am Soc Nephrol. 2009;20:645–654. doi: 10.1681/ASN.2008060590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Grooteman MP, van den Dorpel MA, Bots ML, et al. Effect of online hemodiafiltration on all-cause mortality and cardiovascular outcomes. J Am Soc Nephrol. 2012;23:1087–1096. doi: 10.1681/ASN.2011121140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ok E, Asci G, Toz H, et al. Mortality and cardiovascular events in online haemodiafiltration (OL-HDF) compared with high-flux dialysis: results from the Turkish OL-HDF Study. Nephrol Dial Transplant. 2013;28:192–202. doi: 10.1093/ndt/gfs407. [DOI] [PubMed] [Google Scholar]
- 27.Maduell F, Moreso F, Pons M, et al. High-efficiency postdilution online hemodiafiltration reduces all-cause mortality in hemodialysis patients. J Am Soc Nephrol. 2013;24:487–497. doi: 10.1681/ASN.2012080875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Depner TA. Uremic toxicity: urea and beyond. Semin Dial. 2001;14:246–251. doi: 10.1046/j.1525-139x.2001.00072.x. [DOI] [PubMed] [Google Scholar]
- 29.Meyer TW, Sirich TL, Fong K, et al. The Effect of Increasing Kt/Vurea on Levels of Non-Urea Solutes in the HEMO Study. Journal of the American Society of Nephrology. 2016 Mar 29; pii: ASN.2015091035. [Epub ahead of print] [Google Scholar]
- 30.MacAllister RJ, Fickling SA, Whitley GS, Vallance P. Metabolism of methylarginines by human vasculature; implications for the regulation of nitric oxide synthesis. British journal of pharmacology. 1994;112:43–48. doi: 10.1111/j.1476-5381.1994.tb13026.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tang J, Kao PN, Herschman HR. Protein-arginine methyltransferase I, the predominant protein-arginine methyltransferase in cells, interacts with and is regulated by interleukin enhancer-binding factor 3. J Biol Chem. 2000;275:19866–19876. doi: 10.1074/jbc.M000023200. [DOI] [PubMed] [Google Scholar]
- 32.Faraci FM, Brian JE, Jr, Heistad DD. Response of cerebral blood vessels to an endogenous inhibitor of nitric oxide synthase. Am J Physiol. 1995;269:H1522–H1527. doi: 10.1152/ajpheart.1995.269.5.H1522. [DOI] [PubMed] [Google Scholar]
- 33.Segarra G, Medina P, Ballester RM, et al. Effects of some guanidino compounds on human cerebral arteries. Stroke. 1999;30:2206–2210. doi: 10.1161/01.str.30.10.2206. discussion 2210–2211. [DOI] [PubMed] [Google Scholar]
- 34.Moncada S, Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med. 1993;329:2002–2012. doi: 10.1056/NEJM199312303292706. [DOI] [PubMed] [Google Scholar]
- 35.Steinhorn BS, Loscalzo J, Michel T. Nitroglycerin and Nitric Oxide--A Rondo of Themes in Cardiovascular Therapeutics. N Engl J Med. 2015;373:277–280. doi: 10.1056/NEJMsr1503311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kielstein JT, Zoccali C. Asymmetric dimethylarginine: a cardiovascular risk factor and a uremic toxin coming of age? Am J Kidney Dis. 2005;46:186–202. doi: 10.1053/j.ajkd.2005.05.009. [DOI] [PubMed] [Google Scholar]
- 37.Ghebremariam YT, LePendu P, Lee JC, et al. Unexpected effect of proton pump inhibitors: elevation of the cardiovascular risk factor asymmetric dimethylarginine. Circulation. 2013;128:845–853. doi: 10.1161/CIRCULATIONAHA.113.003602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Serban C, Sahebkar A, Ursoniu S, et al. A systematic review and meta-analysis of the effect of statins on plasma asymmetric dimethylarginine concentrations. Sci Rep. 2015;5:9902. doi: 10.1038/srep09902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Shokouh P, Joharimoghadam A, Roohafza H, et al. Effects of Pioglitazone on Asymmetric Dimethylarginine and Components of the Metabolic Syndrome in Nondiabetic Patients (EPICAMP Study): A Double-Blind, Randomized Clinical Trial. PPAR Res. 2013;2013:358074. doi: 10.1155/2013/358074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Borgeraas H, Strand E, Ringdal Pedersen E, et al. Omega-3 Status and the Relationship between Plasma Asymmetric Dimethylarginine and Risk of Myocardial Infarction in Patients with Suspected Coronary Artery Disease. Cardiol Res Pract. 2012;2012:201742. doi: 10.1155/2012/201742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Scalera F, Kielstein JT, Martens-Lobenhoffer J, Postel SC, Tager M, Bode-Boger SM. Erythropoietin increases asymmetric dimethylarginine in endothelial cells: role of dimethylarginine dimethylaminohydrolase. J Am Soc Nephrol. 2005;16:892–898. doi: 10.1681/ASN.2004090735. [DOI] [PubMed] [Google Scholar]
- 42.Gamboa JL, Pretorius M, Sprinkel KC, Brown NJ, Ikizler TA. Angiotensin converting enzyme inhibition increases ADMA concentration in patients on maintenance hemodialysis--a randomized cross-over study. BMC Nephrol. 2015;16:167. doi: 10.1186/s12882-015-0162-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kielstein JT, Salpeter SR, Bode-Boeger SM, Cooke JP, Fliser D. Symmetric dimethylarginine (SDMA) as endogenous marker of renal function--a meta-analysis. Nephrol Dial Transplant. 2006;21:2446–2451. doi: 10.1093/ndt/gfl292. [DOI] [PubMed] [Google Scholar]
- 44.Closs EI, Basha FZ, Habermeier A, Forstermann U. Interference of L-arginine analogues with L-arginine transport mediated by the y+ carrier hCAT-2B. Nitric Oxide. 1997;1:65–73. doi: 10.1006/niox.1996.0106. [DOI] [PubMed] [Google Scholar]
- 45.Bode-Boger SM, Scalera F, Kielstein JT, et al. Symmetrical dimethylarginine: a new combined parameter for renal function and extent of coronary artery disease. J Am Soc Nephrol. 2006;17:1128–1134. doi: 10.1681/ASN.2005101119. [DOI] [PubMed] [Google Scholar]
- 46.Schepers E, Glorieux G, Dhondt A, Leybaert L, Vanholder R. Role of symmetric dimethylarginine in vascular damage by increasing ROS via store-operated calcium influx in monocytes. Nephrol Dial Transplant. 2009;24:1429–1435. doi: 10.1093/ndt/gfn670. [DOI] [PubMed] [Google Scholar]
- 47.Schepers E, Glorieux G, Dou L, et al. Guanidino compounds as cause of cardiovascular damage in chronic kidney disease: an in vitro evaluation. Blood Purif. 2010;30:277–287. doi: 10.1159/000320765. [DOI] [PubMed] [Google Scholar]
- 48.Aucella F, Maas R, Vigilante M, et al. Methylarginines and mortality in patients with end stage renal disease: a prospective cohort study. Atherosclerosis. 2009;207:541–545. doi: 10.1016/j.atherosclerosis.2009.05.011. [DOI] [PubMed] [Google Scholar]
- 49.Veldink H, Faulhaber-Walter R, Park JK, et al. Effects of chronic SDMA infusion on glomerular filtration rate, blood pressure, myocardial function and renal histology in C57BL6/J mice. Nephrol Dial Transplant. 2013;28:1434–1439. doi: 10.1093/ndt/gfs554. [DOI] [PubMed] [Google Scholar]
- 50.El-Khoury JM, Bunch DR, Reineks E, Jackson R, Steinle R, Wang S. A simple and fast liquid chromatography-tandem mass spectrometry method for measurement of underivatized L-arginine, symmetric dimethylarginine, and asymmetric dimethylarginine and establishment of the reference ranges. Anal Bioanal Chem. 2012;402:771–779. doi: 10.1007/s00216-011-5462-9. [DOI] [PubMed] [Google Scholar]
- 51.Valtonen P, Karppi J, Nyyssonen K, Valkonen VP, Halonen T, Punnonen K. Comparison of HPLC method and commercial ELISA assay for asymmetric dimethylarginine (ADMA) determination in human serum. J Chromatogr B Analyt Technol Biomed Life Sci. 2005;828:97–102. doi: 10.1016/j.jchromb.2005.09.023. [DOI] [PubMed] [Google Scholar]
- 52.Bennett-Richards KJ, Kattenhorn M, Donald AE, et al. Oral L-arginine does not improve endothelial dysfunction in children with chronic renal failure. Kidney Int. 2002;62:1372–1378. doi: 10.1111/j.1523-1755.2002.kid555.x. [DOI] [PubMed] [Google Scholar]
- 53.Schulman SP, Becker LC, Kass DA, et al. L-arginine therapy in acute myocardial infarction: the Vascular Interaction With Age in Myocardial Infarction (VINTAGE MI) randomized clinical trial. Jama. 2006;295:58–64. doi: 10.1001/jama.295.1.58. [DOI] [PubMed] [Google Scholar]
- 54.Vega-Lopez S, Matthan NR, Ausman LM, et al. Altering dietary lysine:arginine ratio has little effect on cardiovascular risk factors and vascular reactivity in moderately hypercholesterolemic adults. Atherosclerosis. 2010;210:555–562. doi: 10.1016/j.atherosclerosis.2009.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wilson AM, Harada R, Nair N, Balasubramanian N, Cooke JP. L-arginine supplementation in peripheral arterial disease: no benefit and possible harm. Circulation. 2007;116:188–195. doi: 10.1161/CIRCULATIONAHA.106.683656. [DOI] [PubMed] [Google Scholar]
- 56.Kielstein JT, Boger RH, Bode-Boger SM, et al. Low dialysance of asymmetric dimethylarginine (ADMA)--in vivo and in vitro evidence of significant protein binding. Clin Nephrol. 2004;62:295–300. doi: 10.5414/cnp62295. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.