Abstract
Background
Vertebral compression fractures (VCF) are common in COPD patients, with osteoporosis being the main cause. The clinical impact of VCF derives mostly from both pain and chest deformity, which may lead to ventilatory and physical activity limitations. Surprisingly, the consequences of VCF on the quality outcomes of hospital care are poorly known.
Objective
To assess these indicators in patients hospitalized due to a COPD exacerbation (ECOPD) who also have VCF.
Methods
Clinical characteristics and quality care indicators were assessed in two one-year periods, one retrospective (exploratory) and one prospective (validation), in all consecutive patients hospitalized for ECOPD. Diagnosis of VCF was based on the reduction of >20% height of the vertebral body evaluated in standard lateral chest X-ray (three independent observers).
Results
From the 248 patients admitted during the exploratory phase, a third had at least one VCF. Underdiagnosis rate was 97.6%, and patients with VCF had more admissions (normalized for survival), longer hospital stays, and higher mortality than patients without (4 [25th–75th percentiles, 2–8] vs 3 [1–6] admissions, P<0.01; 12 [6–30] vs 9 [6–18] days, P<0.05; and 50 vs 32.1% deaths, P<0.01, respectively). The risk of dying in the two following years was also higher in VCF patients (odds ratio: 2.11 [1.2–3.6], P<0.01). The validation cohort consisted of 250 patients who showed very similar results. The logistic regression analysis indicated that both VCF and age were factors independently associated with mortality.
Conclusion
Although VCF is frequently underdiagnosed in patients hospitalized for ECOPD, it is strongly associated with a worse prognosis and quality care outcomes.
Keywords: vertebral fracture, COPD, prognosis, hospitalizations
Introduction
COPD is a complex and heterogeneous disorder where the lung involvement is often associated with aging, comorbidities, and systemic manifestations.1,2 The latter includes the involvement of the musculoskeletal system and, more specifically, muscle dysfunction and bone disorders. However, relatively little attention has been paid to the latter despite its high prevalence. In fact, it is estimated that between 20% and 35% of patients with COPD have associated osteoporosis,3,4 and similar percentages have vertebral fractures.4–10 Steroid therapy, smoking, sedentary lifestyle, vitamin D deficiency, and systemic inflammation have been suggested as the main factors causing such bone disorders.3,7,11 As for the consequences of vertebral fractures, the most important are the subsequent pain and decreased mobility and deformity of the rib cage. Both can contribute to the development of ventilatory restriction.6,12,13
Moreover, COPD exacerbations (ECOPD) are acute episodes that appear during the course of the disease and adversely modify its evolution.1,2 As a result, different guidelines related to the management of COPD have included the presence of frequent exacerbations among the elements to properly classify and treat this disorder.1,2 Moderate and severe ECOPD also constitute a major health problem because they usually involve hospital admissions, with significant consumption of resources.14 The relationship between vertebral fractures and exacerbations is not well known, but it seems logical to assume that mechanical limitations secondary to the former would lead to a worse ventilatory response during the latter. Conversely, frequent exacerbations appear to be associated with a loss of bone mineral density, probably due to the concomitant presence of some of the abovementioned factors.15
The level of underdiagnosis of osteoporosis, and vertebral fractures in particular, is very high.16 Up to 30% of patients admitted for ECOPD had vertebral compression fractures (VCF) in some of the previous studies, although such a diagnosis remains unnoticed in around 80% of the cases.4,17 Other studies have shown that of ECOPD patients who are diagnosed with VCF, only half of them receive a specific treatment and/or special considerations regarding their respiratory disease.5 All of this is probably the consequence of the relative lack of data on the specific implications that the VCF presence has on clinical outcomes of ECOPD. In two recent retrospective studies, Kim et al17 and Glaser and El-Haddad18 observed that COPD patients with VCF showed higher mortality and increased risk of readmission, respectively, than those without. In a similar line of argument, we hypothesized that VCF will have an important impact not only on mortality but also on other relevant outcomes related to both the prognosis of the disease and the quality of the health care. Accordingly, the present study was designed to investigate potential associations between this bone abnormality and the most relevant clinical outcomes of patients admitted to the hospital for ECOPD.
Methods
Design, population and variables
The study was divided into two phases. The first one was retrospective, assessing data from all patients admitted to our hospital for ECOPD during one year. The analysis included sociodemographic, clinical, nutritional, respiratory function, radiological, and treatment data, including cumulative dose of corticosteroids during the two previous years. In addition, the most prominent clinical outcomes were analyzed: admissions (normalized by survival, ie, admissions per days of survival), as well as length of hospital stays, mortality, and risk of death in the year of recruitment plus the two following years. The second phase was prospective (validation), collecting the same data in an additional year. Exclusion criteria were diagnosis of pneumonia or the concomitant presence of other relevant diseases with potential respiratory impact (lung cancer, left heart failure, neuromuscular disorder, etc.). The study was designed according to local and European legislation, with particular emphasis on regulations regarding data privacy (Spanish Organic Law 15/1999 on Protection of Personal Data [LOPD]) and was approved by the Ethics Committee at our institution (Comité Étic d’Investigació Clínica, CEIC). Written informed consent was obtained from all individuals or their closest relatives (if patients not alive in the retrospective cohort).
Radiological analysis
The chest X-ray obtained during hospitalization by the care team were reanalyzed in the lateral projection by three independent observers with extensive experience in chest radiology. The initial diagnosis of VCF was established in the presence of at least a 20% reduction in the height of the vertebral body in its anterior, middle, or posterior cephalo-caudal lines (Figure 1). Definitive diagnosis involved the coincidence of at least two of the three observers.
Figure 1.
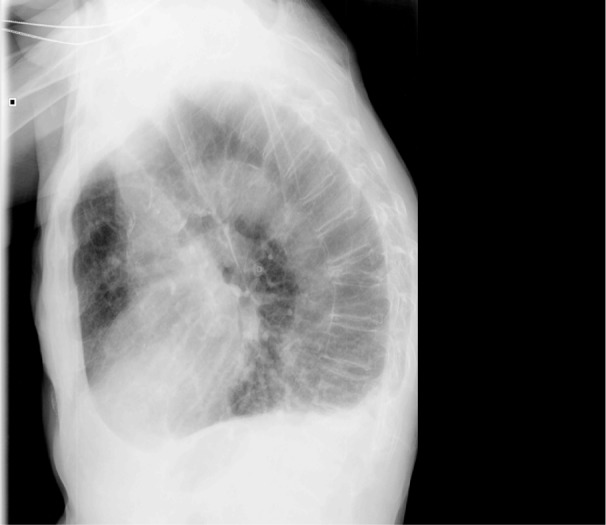
Lateral projection of a chest radiograph.
Notes: Wedge fractures can be observed in different vertebral bodies. That of T9 is severe (loss of >40% of its anterior component) and that of L1 can be considered as moderate (25%–40% loss) according to Genant’s classification. No vertebra presents retropulsion of the posterior wall or associated listhesis.
Cumulative doses of corticosteroids
To calculate the cumulative steroid doses received by patients during the two years preceding hospital admission institutional records (Catalan Health System, CatSalut) of drug deliveries were analyzed. Such calculations included outpatient (visits to the emergency room, specialists or family doctors) and hospital prescriptions as well as drug withdrawals from the pharmacy offices, and both intravenous (methyl-prednisolone and hydrocortisone) and/or oral (prednisone) administrations. Methyl-prednisolone and hydrocortisone were converted to prednisone doses using the following formula: cumulative dose of corticosteroids = (mg of methyl-prednisolone* 0.8) + (mg hydrocortisone/4) + (mg prednisone).19
Statistical analysis
The normal distribution of different variables was tested using the Kolmogorov–Smirnov test. While those with normal distribution are expressed as mean ± standard deviation, those with nonnormal distribution are expressed as median (25%–75% percentiles). The κ index was used for the assessment of interobserver agreement in the diagnosis of VCF. The Student’s t-test or the Mann–Whitney U test were used to compare groups with and without VCF, when appropriate. Calculation of odds ratio (OR) was used for the analysis of mortality, which was represented by Kaplan–Meier curves. Finally, logistic regression analyses were performed to evaluate the independence of those variables associated with mortality. All statistical analyses were performed using SPSS 16.0 (SPSS Inc., Chicago, IL, USA), and a P-value ≤0.05 was considered significant.
Results
A total of 248 patients were admitted for ECOPD during the retrospective exploratory phase. A third of them had at least one VCF, mainly located in the dorsal spine and more precisely in the anterior portion of the vertebral body. The agreement between the three observers for chest radiograph was excellent (κ index of 0.689, 0.763, and 0.723, P<0.001 in all cases). Unfortunately, only 2.4% of ECOPD patients with VCF were diagnosed with this fracture and/or osteoporosis during the hospitalization. Sociodemographic, anthropometric, and functional characteristics of patients, with and without VCF, are summarized in Table 1. No significant differences were observed between them, except for the body mass index (BMI) and the cumulative dose of steroids that were lower and higher, respectively, in the former group. The treatment of ECOPD as well as radiological and analytical determinations were those commonly used following current COPD guidelines.1,2 That is to say, patients were treated with bronchodilators, antibiotics, and a short run of systemic corticosteroids. Bone density and metabolism as well as vitamin D or parathyroid hormone (PTH) levels were not assessed by the clinical team taking care of the patients at that time.
Table 1.
Patients’ characteristics of the retrospective (exploratory) phase of the study
| Clinical variable | VCF | Non-VCF | Significance |
|---|---|---|---|
| Age (years) | 74.5 (67–80) | 73 (63–80) | ns |
| Sex (M/F, %) | 87.2/12.8 | 77.8/22.2 | ns |
| FEV1 (% pred.) | 33.5 (23–45) | 35 (27–51) | ns |
| FVC (% pred.) | 53.8±21.6 | 55.9±14.2 | ns |
| TLC (% pred.) | 97.2±22.1 | 98.5±23.7 | ns |
| % RV/TLC | 62.7±10.6 | 61.5±11.4 | ns |
| DLCO (% pred.) | 43.3±17.2 | 46.8±20.8 | ns |
| PaO2 (mmHg) | 66.3±14.1 | 66.1±10.3 | ns |
| PaCO2 (mmHg) | 47 (42–51) | 45 (41–49) | ns |
| BMI (m/kg2) | 25.9±6.1 | 27.9±7.4 | P<0.05 |
| NIMV (%) | 39.5 | 34.8 | ns |
| P. aeruginosa (%) | 41.9 | 31.1 | ns |
| Cumulated dose of Corticosteroids (mg of prednisone) | 1,440 (757–3,242) | 1,092 (546–2,189) | P<0.05 |
| Smoking (pack/yr) | 58 (43–75) | 61 (44–77) | ns |
Notes: Variables are expressed as mean and standard deviation for those with normal distribution and as median and (percentiles 25%–75%) for those others with nonnormal distribution. The presence of P. aeruginosa in respiratory secretions is routinely tested in patients admitted to Hospital del Mar (Barcelona) for exacerbation of COPD.
Abbreviations: FEV1, forced expiratory volume in the first second; FVC, forced vital capacity; TLC, total lung capacity; RV, residual volume; DLCO, transfer coefficient for CO; PaO2 and PaCO2, partial pressures of oxygen and carbon dioxide in arterial blood, respectively; P. aeruginosa, Pseudomonas aeruginosa; BMI, body mass index; NIMV, non-invasive mechanical ventilation; ns, not significant; VCF, vertebral compression fractures.
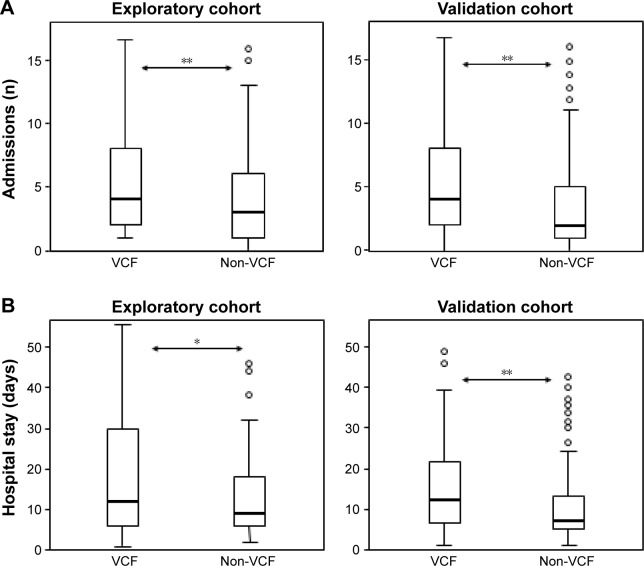
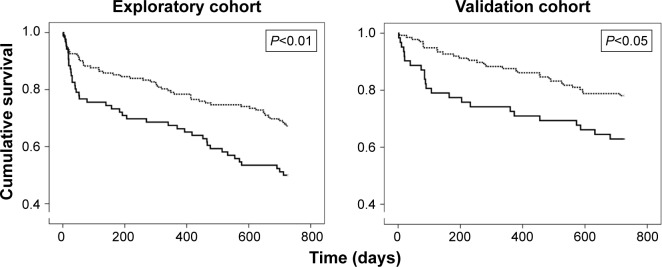
Patients with VCF had a higher number of admissions per year and significantly longer hospital stay than the group without such a bone alteration (4 [2–8] vs 3 [1–6] admissions, respectively, P<0.01; and 12 [6–30] vs 9 [6–18] days/yr, respectively, P<0.05) (Figure 2A and B). The analysis of readmissions within 30 days following discharge could not be done due to their extremely low frequency. The overall mortality in the period analyzed was 38.2%, but VCF patients showed a higher mortality rate and an increased risk of dying in the following two years (50% vs 32.1%, P<0.01, OR: 2.11 [1.2–3.6], P<0.01) (Figure 3). Other variables also associated with mortality were age (P=0.001), BMI (P<0.001), accumulated length of hospitalizations per year (P<0.01), total number of admissions (P=0.01), the presence of multiresistant Pseudomonas in respiratory secretions (OR: 2.37 [1.31–4.05], P<0.01), and the cumulated dose of corticosteroids (P<0.001). The subsequent logistic regression analysis showed that age (P<0.05), BMI (P<0.001), and the total dose of steroids (P<0.05) were independent predictors of mortality after adjusting for the other factors, and VCF was at the limit of statistical significance.
Figure 2.
Comparison between patients with and without at least one VCF.
Notes: (A) Hospital admissions and (B) Mean length of hospital stays. *P<0.05; **P<0.01.
Abbreviation: VCF, vertebral compression fractures.
Figure 3.
Mortality in the two cohorts during the two years follow-up of the ECOPD that led to the inclusion in the study.
Notes: Continuous bold line, patients with VCF; dashed line, patients without VCF.
Abbreviations: VCF, Vertebral compression fractures; ECOPD, COPD exacerbation.
As for the subsequent validation cohort consisting of 250 ECOPD patients, 32.8% had at least one VCF, although such a diagnosis and/or that of osteoporosis were only made in 10.7% of all of them. Again, no significant differences were found regarding sociodemographic, anthropometric, and functional characteristics between patients with and without VCF (Table 2). In this case, the only exception was the cumulative dose of corticosteroids, which was significantly higher in patients with the spine fracture. The overall mortality was 31.5% in this cohort, also confirming that ECOPD patients with VCF have a higher number of hospital admissions and longer hospital stays, as well as an increased mortality and risk of death in the following two years than patients without (3.5 [2–7] vs 2 [1–5] admissions, respectively, P<0.01; 11.5 [6–21] vs 7 [5–13] days/yr, respectively, P<0.01; 37.7% vs 23.4%, P<0.05; OR: 1.98 [1.03–3.81], P<0.05) (Figures 2A, B and 3). Age was another variable also associated with mortality (P<0.001). The subsequent logistic regression analysis showed that both VCF and age were independent predictors of mortality (P<0.05 and <0.01, respectively).
Table 2.
Patients’ characteristics of the prospective (validation) phase of the study
| Clinical variable | VCF | Non-VCF | Significance |
|---|---|---|---|
| Age (years) | 73 (65.2–79) | 71 (62.2–79) | ns |
| Sex (M/F, %) | 75.9/24.1 | 78.3/21.7 | ns |
| FEV1 (% pred.) | 35 (24–45) | 40 (28–51) | ns |
| FVC (% pred.) | 55.6±16.7 | 55.6±15.6 | ns |
| TLC (% pred.) | 100.2±19.4 | 99.9±25.4 | ns |
| % RV/TLC | 62.2±9.9 | 60.6±10.8 | ns |
| DLCO (% pred.) | 43.2±16.8 | 48.6±19 | ns |
| PaO2 (mmHg) | 67±10.6 | 65.4±10.9 | ns |
| PaCO2 (mmHg) | 45 (40.5–49) | 44 (41–49) | ns |
| BMI (m/kg2) | 25.7±6 | 27±5.9 | ns |
| NIMV (%) | 36.1 | 37.6 | ns |
| P. aeruginosa (%) | 32.9 | 23 | ns |
| Cumulated dose of Corticosteroids (mg of prednisone) | 1,294 (556–2,231) | 919 (472–1,508) | P<0.05 |
| Smoking (pack/yr) | 60 (45–80) | 60 (40–80) | ns |
Note: P. aeruginosa, presence of this microorganism in respiratory secretions.
Abbreviations: FEV1, forced expiratory volume in the first second; FVC, forced vital capacity; TLC, total lung capacity; RV, residual volume; DLCO, transfer coefficient for CO; PaO2 and PaCO2, partial pressures of oxygen and carbon dioxide in arterial blood, respectively; P. aeruginosa, Pseudomonas aeruginosa; BMI, body mass index; NIMV, non-invasive mechanical ventilation; ns, not significant; VCF, vertebral compression fractures.
A post hoc logistic regression analysis including both cohorts in order to obtain greater statistical power and a more representative sample of the population confirmed that both VCF and age were independent predictors of mortality (OR: 2.05 [1.34–3.13], P<0.05; and OR: 1.05 [1.03–1.08], P<0.001).
Discussion
The present study demonstrates a clear association between the presence of VCF and different clinical outcomes of quality care in ECOPD patients. Indeed, those subjects with VCF had more hospital admissions, longer hospital stays, and higher mortality than similar individuals without this bone fracture. Despite our striking findings, the rate of VCF underdiagnosis was very high and consequently, did not involve specific measures in the clinical management. This indicates a lack of awareness among health professionals about the presence of significant bone problems in COPD patients.
Certainly, the design of the study cannot allow us to demonstrate a cause–effect relationship between VCF and bad prognosis, but it does indicate that both elements are closely related. In fact, this association might have bidirectional components. On the one hand, the presence of VCF may induce pain and/or changes in thoracic architecture, resulting in ventilatory problems and limiting general physical activity, which in turn would facilitate a worse outcome. On the other hand, the presence of VCF may simply indicate that this is a patient with a history of repeated exacerbations, frequent steroid treatments, and low physical activity. In either case, it can be concluded that VCF acts as a biomarker of poor prognosis in hospitalized ECOPD patients. However, and surprisingly, in most of the previous studies, spinal fractures have not been considered as an important element associated with morbidity and mortality in ECOPD patients, at least as an individual factor differentiated from other comorbidities.20,21 In other words, only very few previous studies have specifically examined the impact of the presence of VCF in the prognosis of COPD patients. In one of them, Kim et al17 analyzed a retrospective cohort of 254 COPD patients with a recent history of hospitalization showing that those with VCF had a higher mortality in a follow-up of two years. In contrast, they were unable to demonstrate an impact of VCF in other clinical or health care outcomes. Glaser and El-Haddad18 in turn found that VCF was also associated with an increased risk of 30-day readmission in ECOPD patients. In the present study, we demonstrate the association between the presence of this fracture not only with mortality but also in the total number of admissions and length of the corresponding stays in the two following years using an exploratory cohort, validating our data in a new and prospective cohort.
The prevalence of VCF in the present series is in the upper limit of the range previously reported by other authors,4–10,17 which also showed a high level of underdiagnosis.4,22 This lack of diagnosis is somewhat surprising since it is well known that COPD patients have up to four times greater risk of osteoporosis than control subjects with similar characteristics.4 Osteoporosis appears to be the main cause of VCF, which are also very frequent in COPD patients,4 especially in those with advanced disease.7 Vertebral wedge fracture is the most common form of VCF, but it can also appear as a central or generalized collapse of the vertebral body. Moreover, the subjects with one VCF have a five times greater chance of developing new spine fractures and up to three times higher probability of having a fracture in other parts of the body.23
As previously mentioned, the consequences of VCF are essentially chest pain and deformation. The former results in a reduction of thoracic mobility and general physical activity,12,13,24 leading to ventilatory problems among other consequences. Moreover, rib cage deformation itself can negatively impact respiratory function, since each VCF can decrease forced vital capacity by around 9%12 and can also affect the mechanical properties of respiratory muscles. In fact, VCF are associated with an increased risk of death even in the general population.25 Despite all of this, they are asymptomatic in two-thirds of the cases, which undoubtedly contributes to the high level of underdiagnosis.22 A point that merits special attention is that since most VCF (80%–90%) occur in the dorsal (D7–D8) and the thoracolumbar (D12-L1) spinal regions,26 they should be easily diagnosed in conventional chest radiography. This indicates that underdiagnosis may have elements of malpractice, especially in light of the results of the present study, since VCF emerges as a marker of many worse clinical outcomes. In the second part of the present study, we found, however, a relative improvement in the level of underdiagnosis, probably because bone abnormalities associated with different chronic conditions have received a little more attention recently in the medical literature.
The causes of osteoporosis and VCF in COPD patients are numerous, although as in our case the use of systemic steroids appears to play an important role.7,27 Moreover, some series pointed out that up to 60% of COPD patients with prolonged treatment on oral steroids have VCF.7 The total cumulative dose of these drugs is also important. It has been proven that a total dose of more than 1,000 mg of oral prednisone, even if given intermittently (as is the case in many frequent exacerbators), also increases the risk of osteoporosis and VCF.28 Regarding inhaled steroids there is no absolute consensus,7,29,30 but it does seem that standard doses used in COPD patients cause no significant adverse effects on their bones.28,30 In the present study, the cumulative dose of corticosteroids during the preceding years was significantly higher in ECOPD patients with VCF compared with their controls and widely exceeded the aforementioned limit, suggesting a major role for this factor.
However, VCF is also present in a large number of patients who have not received high doses of steroids.7 Moreover, bone abnormalities are also present in a significant number of COPD patients who have never even received inhaled steroids.31 Therefore, other factors could be implicated. Some of them are the same that have been involved in other systemic manifestations of the disease. This is the case of smoking, which exerts a direct negative effect on bone mass and increases the risk of VCF.32,33 If alcohol abuse associates with smoking, the risk increases even more.32 The reduction in physical activity is an additional factor that can be implicated in VCF in COPD patients. Low physical activity induces inhibition of trophic signals to bone, resulting in bone mass loss and increased risk of fractures.34 Systemic inflammation is another factor that could be involved in the pathophysiology of fractures in COPD patients.31 Nutritional disturbances, which are frequently observed in COPD patients, lead to the loss of muscle mass and have also been associated with osteoporosis and fractures. The mechanism for this appears to be an increase in protein destruction, and mostly of collagen.35 Interestingly, low bone density and the risk of fractures are also associated with the extent of emphysema,36 and it is worth noting that patients with an emphysematous phenotype characteristically show low muscle mass, which is mainly due to an increase in protein breakdown.37 Certainly, many of the previously mentioned deleterious factors involved in the loss of bone mass would be even more pronounced during ECOPD. Therefore, it is not surprising that exacerbations also impact negatively on body weight, muscle mass, and bone mass in COPD patients.38 Hormonal factors may also be involved in their bone loss. Hypogonadism, which seems to be common in men with the disease, has also been involved in their bone disorders.39 In women after menopause, in turn, hormonal deficiency also leads to a significant loss of bone mass, acting synergistically with the lung disease.40 Furthermore, growth hormone dysfunction, which also seems to be relatively frequent in COPD, can lead to loss of bone mineralization.41 Vitamin D deficiency, also common in these patients and proportional to the severity of the lung disease, appears to play an important role in the occurrence of bone changes.42–44 Its deficit has been attributed to both nutritional abnormalities and low exposure to ultraviolet rays (ie, reduction in outdoor activities). Finally, it is worth noting that there is a frequent association of COPD with aging and many different chronic comorbidities, which are also potential contributors to bone abnormalities. Interestingly, recent studies of some members of our group using text mining suggest that bone abnormalities share molecular mechanisms with other COPD comorbidities such as cardiovascular disorders.45 This has also been proved experimentally.46
Whatever the causes of bone loss, it is clear that it is very intense in COPD patients43 and is a direct result of an imbalance between formation and destruction of bone.43,46
The management of COPD patients with suspected bone disorders should include routine screening with conventional radiology, as well as periodic determination of markers of bone metabolism (calcium, phosphorus, vitamin D, and eventually, PTH) and the assessment of bone density and risk of fractures.31 Patients should avoid tobacco and alcohol and ensure a balanced nutrition and an appropriate physical activity level, preferably outdoors. Moreover, the use of systemic steroids should be optimized, avoiding inappropriate high doses or long regimes.47 As to the pharmacological prevention of VCF (ie, drug therapy of osteoporosis), it should be started early, especially in those patients who already have a fracture.23 There are several alternatives but few have been specifically tested in COPD patients, so the consensus is to follow general guidelines.31,48 In cases with proven hypogonadism, hormone replacement therapy may be useful. Supplementation with calcium and vitamin D may also be desirable in subjects exposed to prolonged steroid therapy, but these should be combined with other drugs.31 Finally, bisphosphonates appear to be effective in preventing fractures, especially if used early in high-risk subjects.49 It is interesting to consider that surgical treatments such as kyphoplasty can improve lung function in COPD patients with VCF,50,51 although its impact on prognosis and quality care outcomes still remains unexplored.
As for the potential limitations of the present study, we should recognize that diagnosis using simple chest X-ray may have underestimated small fractures. However, the use of three independent expert observers has probably minimized this possibility. Moreover, the objectives of the study were to assess the impact of VCF on clinical outcomes and the level of underdiagnosis when using the usual clinical approach employed with ECOPD patients. For the same reason, the present study did not include a specific questionnaire on VCF symptoms, a study of bone metabolism, or the assessment of bone density. In addition, our series had a net predominance of male patients, which is still the scenario in our environment. This is not the case in other countries where COPD is more evenly distributed among both sexes. However, a higher presence of female postmenopausal patients would probably have resulted in an even greater frequency of bone disturbances. Finally, our study design did not enable us to define the incidence or prevalence of VCF but an approximation to its frequency in hospitalized COPD patients and the association with their clinical outcomes.
The design of the present study is one of its main strengths since results obtained in the exploratory retrospective cohort were prospectively validated in a second one.
Conclusion
VCF is common in ECOPD patients, being associated with more hospitalizations, larger hospital stays, and higher mortality. Unfortunately, these bone abnormalities often remain underdiagnosed, and therefore do not lead to a specific clinical management and treatment of the patients.
Acknowledgments
We thank Pilar Torre and Susana Font for their invaluable help in collecting and processing data, Sergi Mojal for his statistical advice, and Jonathan McFarland for his editing aid.
Footnotes
Disclosure
This study has been partially funded by SEPAR (264/2012), CIBERES, FIS (12/02534), Plan Nacional I+D+i (SAF-2014 – 54371), Generalitat de Catalunya (2009-SGR-393), and FUCAP (2012). The authors report no other conflicts of interest in this work.
References
- 1.Vestbo J, Hurd SS, Agusti AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary, disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187:347365. doi: 10.1164/rccm.201204-0596PP. [DOI] [PubMed] [Google Scholar]
- 2.Miravitlles M, Soler-Cataluna JJ, Calle M, et al. Spanish Guideline for COPD (GesEPOC). Update 2014. Arch Bronconeumol. 2014;50(Suppl 1):1–16. doi: 10.1016/S0300-2896(14)70070-5. [DOI] [PubMed] [Google Scholar]
- 3.Lehouck A, Boonen S, Decramer M, Janssens W. COPD, bone metabolism, and osteoporosis. Chest. 2011;139:648–657. doi: 10.1378/chest.10-1427. [DOI] [PubMed] [Google Scholar]
- 4.Papaioannou A, Parkinson W, Ferko N, et al. Prevalence of vertebral fractures among patients with chronic obstructive pulmonary disease in Canada. Osteoporos Int. 2003;14:913–917. doi: 10.1007/s00198-003-1449-5. [DOI] [PubMed] [Google Scholar]
- 5.Majumdar SR, Villa-Roel C, Lyons KJ, Rowe BH. Prevalence and predictors of vertebral fracture in patients with chronic obstructive pulmonary disease. Respir Med. 2010;104:260–266. doi: 10.1016/j.rmed.2009.09.013. [DOI] [PubMed] [Google Scholar]
- 6.Carter JD, Patel S, Sultan FL, et al. The recognition and treatment of vertebral fractures in males with chronic obstructive pulmonary disease. Respir Med. 2008;102:1165–1172. doi: 10.1016/j.rmed.2008.03.003. [DOI] [PubMed] [Google Scholar]
- 7.McEvoy CE, Ensrud KE, Bender E, et al. Association between corticosteroid use and vertebral fractures in older men with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1998;157:704–709. doi: 10.1164/ajrccm.157.3.9703080. [DOI] [PubMed] [Google Scholar]
- 8.Jorgensen NR, Schwarz P, Holme I, Henriksen BM, Petersen LJ, Backer V. The prevalence of osteoporosis in patients with chronic obstructive pulmonary disease: a cross sectional study. Respir Med. 2007;101:177–185. doi: 10.1016/j.rmed.2006.03.029. [DOI] [PubMed] [Google Scholar]
- 9.Nuti R, Siviero P, Maggi S, et al. Vertebral fractures in patients with chronic obstructive pulmonary disease: the EOLO study. Osteoporos Int. 2009;20:989–998. doi: 10.1007/s00198-008-0770-4. [DOI] [PubMed] [Google Scholar]
- 10.Liao KM, Liang FW, Li CY. Risks of all-cause and site-specific fractures among hospitalized patients with COPD. Medicine (Baltimore) 2016;95:e5070. doi: 10.1097/MD.0000000000005070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brightling CE. Biomarkers that predict and guide therapy for exacerbations of chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2013;10(Suppl):S214–S219. doi: 10.1513/AnnalsATS.201302-023AW. [DOI] [PubMed] [Google Scholar]
- 12.Leech JA, Dulberg C, Kellie S, Pattee L, Gay J. Relationship of lung function to severity of osteoporosis in women. Am Rev Respir Dis. 1990;141:68–71. doi: 10.1164/ajrccm/141.1.68. [DOI] [PubMed] [Google Scholar]
- 13.Culham EG, Jimenez HA, King CE. Thoracic kyphosis, rib mobility, and lung volumes in normal women and women with osteoporosis. Spine. 1994;19:1250–1255. doi: 10.1097/00007632-199405310-00010. [DOI] [PubMed] [Google Scholar]
- 14.Blasi F, Cesana G, Conti S, et al. The clinical and economic impact of exacerbations of chronic obstructive pulmonary disease: a cohort of hospitalized patients. PLoS One. 2014;9:e101228. doi: 10.1371/journal.pone.0101228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kiyokawa H, Muro S, Oguma T, et al. lmpact of COPD exacerbations on osteoporosis assessed by chest CT scan. COPD. 2012;9:235–242. doi: 10.3109/15412555.2011.650243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Graat-Vérboom L, Spruit MA, van den Bome BE, et al. CIRO Network Correlates of osteoporosis in chronic obstructive pulmonary disease: An underestimated systemic component. Respir Med. 2009;103:1143–1151. doi: 10.1016/j.rmed.2009.02.014. [DOI] [PubMed] [Google Scholar]
- 17.Kim GW, Joo HJ, Park TS, et al. Vertebral compression fractures may increase mortality in male patients with chronic obstructive pulmonary disease. Int J Tuberc Lung Dis. 2015;19:603–609. doi: 10.5588/ijtld.14.0754. [DOI] [PubMed] [Google Scholar]
- 18.Glaser JB, El-Haddad H. Exploring novel medicare readmission risk variables in chronic obstructive pulmonary disease patients at high risk of readmission within 30 days of hospital discharge. Ann Am Thorac Soc. 2015;12:1288–1293. doi: 10.1513/AnnalsATS.201504-228OC. [DOI] [PubMed] [Google Scholar]
- 19.Hope NH, Ray SM, Franks AS, Heidel E. Impact of an educational intervention on steroid prescribing and dosing effect on patient outcomes in COPD exacerbations. Pharm Pract. 2010;8:162–166. doi: 10.4321/s1886-36552010000300002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.de Granda-Orive JI, Solano-Reina S. Chronic obstructive pulmonar disease mortality. SEPAR COPD-Smoking Year. Arch Bronconeumol. 2016;52:407–408. doi: 10.1016/j.arbres.2016.01.015. [DOI] [PubMed] [Google Scholar]
- 21.Escarrabill J, Torrente E, Esquinas C, et al. En nombre del equipo del Plan Director de las Enfermedades del Aparato Respiratorio (PDMAR) y del grupo colaborador del proyecto MAG-1 Clinical audit of patients hospitalized due to COPD exacerbation. MAG-1 Study. Arch Bronconeumol. 2015;51:483–489. doi: 10.1016/j.arbres.2014.06.023. [DOI] [PubMed] [Google Scholar]
- 22.Cooper C, Melton LJ. Vertebral fractures. BMJ. 1992;304:793–794. doi: 10.1136/bmj.304.6830.793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Klotzbuecher CM, Ross PD, Landsman PB, Abbott TA, 3rd, Berger M. Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res. 2000;15:721–739. doi: 10.1359/jbmr.2000.15.4.721. [DOI] [PubMed] [Google Scholar]
- 24.Nevitt MC, Ettinger B, Black DM, et al. The association of radio-graphically detected vertebral fractures with back pain and function: a prospective study. Ann Intern Med. 1998;128:793–800. doi: 10.7326/0003-4819-128-10-199805150-00001. [DOI] [PubMed] [Google Scholar]
- 25.Ensrud KE, Thompson DE, Cautey JA, et al. Prevalent vertebral deformities predict mortality and hospitalization in older women with low bone mass. Fracture Intervention Trial Research Group. J Am Geriatr Soc. 2000;48:241–249. doi: 10.1111/j.1532-5415.2000.tb02641.x. [DOI] [PubMed] [Google Scholar]
- 26.Kim N, Rowe BH, Raymond G, et al. Underreporting of vertebral fractures on routine chest radiography. Am J Roentgenol. 2004;182:297–300. doi: 10.2214/ajr.182.2.1820297. [DOI] [PubMed] [Google Scholar]
- 27.Dam TT, Harrison S, Fink HA, Ramsdell J, Barrett-Connor E, Osteoporotic Fractures in Men (MrOS) Research Group Bone mineral density and fractures in older men with chronic obstructive pulmonary disease or asthma. Osteoporos Int. 2010;21:1341–1349. doi: 10.1007/s00198-009-1076-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Leib ES, Saag KG, Adachi JD, et al. FRAX(®) Position Development Conference Members. Official Positions for FRAX clinical regarding glucocorticoids: the impact of the use of glucocorticoids on the estimate by FRAX of the 10 year risk of fracture from Joint Official Positions Development Conference of the International Society for Clinical Densitometry and International Osteoporosis Foundation on FRAX. J Clin Densitom. 2011;14:212–219. doi: 10.1016/j.jocd.2011.05.014. [DOI] [PubMed] [Google Scholar]
- 29.Hubbard R, Tattersfield A, Smith C, West J, Smeeth L, Fletcher A. Use of inhaled corticosteroids and the risk of fracture. Chest. 2006;130:1082–1088. doi: 10.1378/chest.130.4.1082. [DOI] [PubMed] [Google Scholar]
- 30.Pauwels RA, Liifdahl CG, Laitinen LA, et al. Long-term treatment with inhaled budesonide in persons with mild chronic obstructive pulmonary disease who continue smoking. European Respiratory Society Study on Chronic Obstructive Pulmonary Disease. N Engl J Med. 1999;340:1948–1953. doi: 10.1056/NEJM199906243402503. [DOI] [PubMed] [Google Scholar]
- 31.Biskobing DM. COPD and osteoporosis. Chest. 2002;121:609–620. doi: 10.1378/chest.121.2.609. [DOI] [PubMed] [Google Scholar]
- 32.Seeman E, Melton LJ, 3rd, O’Fallon WM, Riggs BL. Risk factors for spinal osteoporosis in men. Am J Med. 1983;75:977–983. doi: 10.1016/0002-9343(83)90878-1. [DOI] [PubMed] [Google Scholar]
- 33.Jaramillo JD, Wilson C, Stinson DS, et al. COPD Gene Investigators Reduced bone density and vertebral fractures in smokers. Men and COPD patients at increased risk. Ann Am Thorac Soc. 2015;12:648–656. doi: 10.1513/AnnalsATS.201412-591OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu WT, Kuo HP, Liao TH, et al. Low bone mineral density in COPD patients with osteoporosis is related to low daily physical activity and high COPD assessment test scores. Int J Chron Obstruct Pulmon Dis. 2015;10:1737–1744. doi: 10.2147/COPD.S87110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bolton CE, Lonescu AA, Shiels KM, et al. Associated loss of fat-free mass and bone mineral density in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2004;170:1286–1293. doi: 10.1164/rccm.200406-754OC. [DOI] [PubMed] [Google Scholar]
- 36.Pompe E, de Jong PA, van Rikxoort EM, et al. Smokers with emphysema and small airway disease on computed tomography have lower bone density. Int J Chron Obstruct Pulmon Dis. 2016;11:1207–1216. doi: 10.2147/COPD.S103680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gea J, Agustí A, Roca J. Pathophysiology of muscle dysfunction in COPD. J Appl Physiol (1985) 2013;114:1222–1234. doi: 10.1152/japplphysiol.00981.2012. [DOI] [PubMed] [Google Scholar]
- 38.Hopkinson NS, Tennant RC, Dayer MJ, et al. A prospective study of decline in fat free mass and skeletal muscle strength in chronic obstructive pulmonary disease. Respir Res. 2007;8:25. doi: 10.1186/1465-9921-8-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Balasubramanian V, Naing S. Hypogonadism in chronic obstructive pulmonary disease: incidence and effects. Curr Opin Pulm Med. 2012;18:112–117. doi: 10.1097/MCP.0b013e32834feb37. [DOI] [PubMed] [Google Scholar]
- 40.Dennison EM, Compston JE, Flahive J, et al. GLOW Investigators Effect of comorbidities on fracture risk: findings from the Global Longitudinal Study of Osteoporosis in Women (GLOW) Bone. 2012;50:1288–1293. doi: 10.1016/j.bone.2012.02.639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Creutzberg EC, Casaburi R. Endocrinological disturbances in chronic obstructive pulmonary disease. Eur Respir J. 2003;46(Suppl):76s–80s. doi: 10.1183/09031936.03.00004610. [DOI] [PubMed] [Google Scholar]
- 42.Janssens W, Bouillon R, Claes B, et al. Vitamin D deficiency is highly prevalent in COPD and correlates with variants in the vitamin D-binding gene. Thorax. 2010;65:21520. doi: 10.1136/thx.2009.120659. [DOI] [PubMed] [Google Scholar]
- 43.Duckers JM, Evans BA, Fraser WD, Stone MD, Bolton CE, Shale DJ. Low bone mineral density in men with chronic obstructive pulmonary disease. Respir Res. 2011;12:101. doi: 10.1186/1465-9921-12-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.García de Tena J, El Hachem Debek A, Hernández Gutiérrez C, Izquierdo Alonso JL. The role of vitamin D in chronic obstructive pulmonary disease, asthma and other respiratory diseases. Arch Bronconeumol. 2014;50:179–184. doi: 10.1016/j.arbres.2013.11.023. [DOI] [PubMed] [Google Scholar]
- 45.Grosdidier S, Ferrer A, Faner R, et al. Network medicine analysis of COPD multimorbidities. Respir Res. 2014;15:111. doi: 10.1186/s12931-014-0111-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Graat-Verboom L, van den Borne BE, Smeenk FW, Spruit MA, Wouters EF. Osteoporosis in COPD outpatients based on bone mineral density and vertebral fractures. J Bone Miner Res. 2011;26:561–568. doi: 10.1002/jbmr.257. [DOI] [PubMed] [Google Scholar]
- 47.Marcos PJ, Huerta A, Márquez-Martín E. 2014 GesEPOC and systemic steroids in chronic obstructive pulmonary disease (COPD) exacerbation: an update. Arch Bronconeumol. 2015;51:360. doi: 10.1016/j.arbres.2014.05.007. [DOI] [PubMed] [Google Scholar]
- 48.Inoue D, Watanabe R, Okazaki R. COPD and osteoporosis: links, risks, and treatment challenges. Int J COPD. 2016;11:637–648. doi: 10.2147/COPD.S79638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cohen S, Levy RM, Keller M, et al. Risedronate therapy prevents corticosteroid-induced bone loss: a twelve-month, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum. 1999;42:2309–2318. doi: 10.1002/1529-0131(199911)42:11<2309::AID-ANR8>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 50.Masala S, Magrini A, Taglieri A, et al. Chronic obstructive pulmonary disease (COPD) patients with osteoporotic vertebral compression fractures (OVCFs): improvement of pulmonary function after percutaneous vertebroplasty (VTP) Eur Radiol. 2014;24:1577–1585. doi: 10.1007/s00330-014-3165-2. [DOI] [PubMed] [Google Scholar]
- 51.Sheng S, Zhenzhong S, Weimin J, Yimeng W, Qudong Y, Jinhui S. Improvement in pulmonary function of chronic obstructive pulmonary disease (COPD) patients with osteoporotic vertebral compression fractures (OVCFs) after kyphoplasty under local anesthesia. Int Surg. 2015;100:503–509. doi: 10.9738/INTSURG-D-14-00173.1. [DOI] [PMC free article] [PubMed] [Google Scholar]