Abstract
Objective
Maternal weight is thought to impact labor. With rising rates of obesity and inductions, we sought to evaluate labor times among induced women by body mass index (BMI) category.
Methods
Retrospective cohort study of term inductions from 2005 to 2010. BMI categories were: normal weight (NW), overweight (OW), and obese (Ob) (18.5–24.9, 25–29.9, ≥30kg/m2). Kruskal-Wallis tests compared median latent labor (LL) length and active labor (AL) length. Chi-square determined associations. Multivariable logistic regression controlled for confounders. Analyses were stratified by parity.
Results
A total of 448 inductions were analyzed. For nulliparas, there was no difference in LL by BMI category (p = 0.22). However, OW nulliparas had a longer AL compared to NW and Ob nulliparas (3.2, 1.7, 2.0 h, p = 0.005). For multiparas, NW had the shortest LL (5.5 h, p = 0.025) with no difference in AL among BMI categories (p = 0.42). The overall cesarean rate was 23% with no difference by BMI category (p = 0.95). However, Ob women had a greater percentage of first stage cesareans (41%) and NW had a greater percentage of second stage cesareans (55%), p = 0.06.
Conclusion
The association between BMI and labor length among inductions differs by phase of labor and parity. BMI also influences the stage of labor in which a cesarean occurs.
Keywords: Body mass index, induction of labor, labor curves, labor length, obesity
Introduction
More than one-half of pregnant women are overweight (OW) or obese (Ob) [1,2]. This rate continues to rise. Simultaneously, the rate of labor induction is also increasing with more than 20% of delivering women undergoing induction [3]. Evidence supports increasing obstetric complications in Ob women, and this risk leads to more inductions. Previous work has demonstrated that obesity is a risk factor for induction of labor, prolonged labor and cesarean delivery [2,4–10]. Given the continued rise in obesity, studies specifically addressing whether an increased body mass index (BMI) affects various aspects of labor, specifically in women being induced, are necessary in order to optimally care for the OW or Ob parturient.
Recent studies suggest that labor is less likely to progress according to standard labor curves in Ob women, especially in active labor (AL) [11–14]. Ob women undergoing induction have also been shown to have greater median dose and duration of pre-delivery oxytocin [12]. Additionally, Ob nulliparous women have a significantly increased rate of cesarean delivery [6]. Several studies, however, have demonstrated no association between maternal BMI and length of the second stage in nulliparous women [11,15,16], suggesting that it is not the second stage of labor, but rather the first stage, that may be most affected by increasing maternal weight in nulliparas.
While prior studies evaluated the effect of maternal weight on labor curves, few have specifically evaluated the length of time in each phase of labor. They also have not focused on women undergoing an induction. Additionally, many studies did not include all BMI categories, specifically OW women. Given the increasing number of OW and Ob pregnant women, the number undergoing an induction of labor, and the concurrent call to prevent the first cesarean delivery [17], our objective was to compare the length of latent and active phases of labor among normal weight (NW), OW, and Ob women undergoing a term induction of labor. Our hypothesis is that OW and Ob women will have a longer latent phase and active phase of labor compared to NW women.
Methods
This was a planned secondary analysis of a large retrospective cohort study of women with two consecutive deliveries at the Hospital of the University of Pennsylvania during the years 2005–2010. The primary study compared women that underwent a term (≥37 weeks) induction of labor to those that went into spontaneous term labor and evaluated their rates of spontaneous preterm birth in a subsequent pregnancy [18]. The starting year was chosen as 2005 as this was the start of electronic medical records at our institution. Approval from the University of Pennsylvania Institutional Review Board was obtained prior to initiation of the study.
Our analysis evaluated only those women undergoing a term induction in the first of the two pregnancies from the parent cohort (n = 622). Both nulliparous and multiparous women were included. Women were included if their first BMI was recorded at ≤20 weeks gestation to limit overestimation of pre-pregnancy BMI due to gestational weight gain. The measured and documented weight at the first prenatal visit and reported height were used to calculate BMI given that self reported pre-pregnancy weight was frequently uncertain and not universally available. Women with multiple gestations and women with a prior cesarean delivery were excluded. Underweight women were also excluded, as our goal was to evaluate labor times in OW and Ob women compared to NW women.
Women undergoing a term induction were identified using a combination of International Classification of Disease, 9th edition (ICD-9) codes (73.01, 73.1, 73.4) and detailed chart review. The detailed chart review was performed to confirm that induction of labor met our strict definitions as described below. Data collection was through chart abstraction from maternal and neonatal electronic medical records. Variables collected included maternal demographics, obstetrical and medical history, and labor and delivery information. All data abstraction was performed in parallel by two of the investigators (L.D.L., A.H.).
We defined induction of labor as (1) use of any cervical ripening agent (prostaglandin or cervical foley); (2) artificial rupture of membranes or oxytocin use in the setting of contractions with cervical dilation <4 cm and (3) cervical dilation of <4 cm in the absence of contractions. Latent labor (LL) was defined as the interval between the start of induction and AL. AL was defined as cervical dilation ≥5cm. The generally accepted definition of failed induction of labor at our institution is the inability to achieve AL after 24 h of induction or 12 h of ruptured membranes in the absence of a non-reassuring fetal heart rate. The first stage of labor was defined as the start of induction to complete dilation. The second stage of labor was defined as time from complete dilation to delivery. Nulliparity was defined as no prior delivery ≥20 weeks gestation. BMI was calculated based on measured weight and patient reported height at the first prenatal visit and defined according to the Institute of Medicine BMI categories: NW: 18.5–24.9 kg/m2, OW: 25.0–29.9kg/m2 and Ob: ≥30.0kg/m2 [19].
Our objective was to compare the length of latent and active phases of labor among NW, OW and Ob women undergoing a term induction of labor. Our primary outcomes were length of time in the latent phase and the active phase of labor. Our secondary outcomes were cesarean delivery rate and indication for cesarean delivery.
Median labor times were analyzed by the Kruskal–Wallis test. Associations between categorical variables were calculated using Chi-square tests. Additionally, associations between BMI categories and mode of delivery were determined using logistic regression and adjusted for confounders including maternal age and race. Given the known difference in labor curves between nulliparous and multiparous women [20–22], all results were stratified by parity. LL and AL times were analyzed separately given the importance of separating different phases of labor for the purposes of understanding potential applications of labor management. Additionally, a subgroup analysis amongst women who achieved vaginal delivery versus those who had cesarean delivery was performed as time to delivery would be decreased in those who had cesarean deliveries early in the labor process and affect the overall results. Data analysis was performed using STATA 12.0 for Windows (STATA Corporation, College Station, TX). Statistical significance was determined by a p value of less than 0.05.
Results
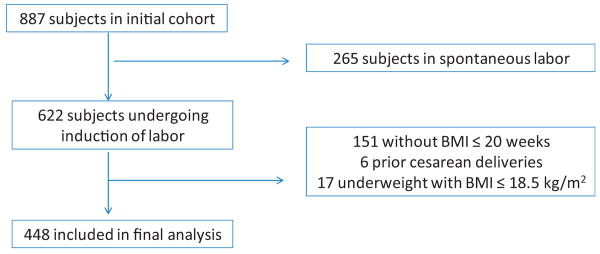
There were 887 patients included in the parent cohort [18]. A total of 448 term women undergoing induction of labor with a BMI recorded at ≤20 weeks gestation were evaluated in this study (Figure 1).
Figure 1.
Flowchart of included subjects.
Baseline demographic characteristics are listed in Table 1. Of the 448 women who met inclusion criteria, 30% (n = 134) were OW and 30% were Ob (n = 133) at the initiation of prenatal care. There was no difference in indication for induction between the BMI categories (p = 0.74) or cervical dilation in centimeters at start of induction (p = 0.76).
Table 1.
Demographics by BMI category.
| NW 181 (40) | OW 134 (30) | Ob 133 (30) | p value | |
|---|---|---|---|---|
| Gestational age at delivery (weeks) | 39.2 [38.7–41] | 39.7 [38.7–40.6] | 39.9 [38.9–41] | 0.60 |
| Race | ||||
| African American | 99 (55) | 99 (74) | 110 (83) | <0.001 |
| Caucasian | 49 (27) | 24 (18) | 16 (12) | |
| Asian | 18 (10) | 1 (1) | 0 | |
| Other | 6 (3) | 5 (4) | 2 (2) | |
| Unknown | 9 (5) | 5 (4) | 5 (4) | |
| Age (years) | 26 [20–31] | 25 [20–31] | 24 [21–30] | 0.69 |
| Parity | ||||
| Nulliparous | 148 (82) | 95 (71) | 70 (53) | <0.001 |
| Multiparous | 33 (18) | 39 (29) | 63 (47) | |
| Indication for induction* | ||||
| PROM | 26 (14) | 22 (16) | 16 (12) | 0.74 |
| Post dates | 39 (22) | 20 (15) | 25 (19) | |
| Maternal | 37 (20) | 37 (28) | 35 (26) | |
| Fetal | 60 (33) | 39 (29) | 42 (32) | |
| Elective | 19 (11) | 16 (12) | 15 (11) | |
| Cervical dilation at start of induction (cm) | ||||
| 0 | 39 (22) | 37 (28) | 27 (20) | 0.76 |
| 1 | 70 (39) | 40 (30) | 47 (35) | |
| 2 | 39 (22) | 27 (20) | 32 (24) | |
| 3 | 19 (16) | 27 (20) | 23 (17) | |
| 4 | 4 (2) | 3 (2) | 4 (3) | |
| Neonatal weight (g) | 3270 [2945–3535] | 3293 [3010–3535] | 3400 [2950–3730] | 0.13 |
This table describes the demographic characteristics of the study population by BMI category. n(%), mean [IQR].
PROM: premature rupture of membranes, Post dates: ≥41 weeks, Maternal: medical comorbidity, Fetal: amniotic fluid disorders, IUGR, anomalies, fetal decelerations.
Among all NW, OW and Ob women in our cohort, there was no significant difference in median LL time (10.3, 10.3, 10.5 h, respectively, p = 0.9), even when restricting the analysis to women who eventually had a vaginally delivery (9.42, 9.38, 9.17 h, p = 0.99). There was, however, a significant difference in overall median AL time by BMI category among NW, OW and Ob women (1.7, 2.8, 1.6 h, p = 0.004). This remained when including only those women who had a vaginal delivery (1.65, 2.8, 1.5 h, p = 0.002). We next stratified the analysis by parity.
The LL and AL times of the nulliparous study population for each BMI category are described in Table 2. Similar to the overall results, there was no significant difference in median LL time by BMI category in the nulliparous strata. Of the nulliparous women who achieved AL, median AL time was significantly different by BMI category.
Table 2.
Median labor times (h) among nulliparous women by BMI category.
| NW 40 (181) | OW 30 (134) | Ob 30 (133) | p value | |
|---|---|---|---|---|
| Median LL time, n = 313 | 11.17 [7.73–15.44] | 11.83 [6.75–18.27] | 12.69 [8.83–17.50] | 0.22 |
| Median LL time in those with vaginal delivery, n = 290 | 10.50 [7.42–14.42] | 9.75 [5.83–14.75] | 12.98 [7.38–18.24] | 0.37 |
| Median AL time, n =246 | 1.7 [0.50–3.20] | 3.20 [1.50–5.30] | 2.00 [0.80–3.80] | 0.005 |
| Median AL time in those with vaginal delivery, n = 180 | 1.3 [0–2.80] | 3.20 [1.50–5.30] | 2.00 [0.80–3.80] | 0.0021 |
This table describes median labor times among nulliparous women by BMI category, phase of labor, and those with a vaginal delivery. %(n), median [inter-quartile range].
Table 3 demonstrates LL and AL times for the multiparous women in each BMI category. There was a significant difference in LL time with OW multiparas having the longest LL. This difference in LL, however, did not remain when restricted to multiparous women that had vaginal deliveries. For multiparous women who achieved AL, there was no significant difference in median AL times overall or when restricted to those who had a vaginal delivery.
Table 3.
Median labor times (h) among multiparous women by BMI category.
| NW 40 (181) | OW 30 (134) | Ob 30 (133) | p value | |
|---|---|---|---|---|
| Median LL time, n = 135 | 5.50 [3.75–9.33] | 8.75 [6.00–12.25] | 8.17 [5.42–11.63] | 0.025 |
| Median LL time in those with vaginal delivery, n = 110 | 6.25 [4.12–9.33] | 7.78 [6.33–12.75] | 7.94 [5.43–11.63] | 0.11 |
| Median AL time, n = 126 | 1.30 [0.30–2.50] | 1.70 [0.60–3.80] | 1.50 [0.30–2.50] | 0.42 |
| Median AL time in those with vaginal delivery, n = 110 | 1.0 [0.10–2.40] | 1.70 [0.60–3.80] | 1.50 [0.30–2.50] | 0.43 |
This table describes median labor times among multiparous women by BMI category, phase of labor, and those with a vaginal delivery. %(n); media [inter-quartile range].
The cesarean delivery rate for the entire cohort was 23% with no difference by BMI category (Table 4). Additionally, when looking at all indications for cesarean delivery, there was no statistically significant association between BMI category and indication (p = 0.17). However, when comparing arrest in the first stage of labor (defined as a failed induction or arrest of dilation) and arrest in the second stage, there was a trend towards a significant difference (p = 0.059). Ob women had a greater percentage of first stage cesareans (41%), whereas NW women had a greater percentage of second stage cesareans (55%).
Table 4.
Cesarean delivery among BMI categories.
| NW | OW | Ob | p value | |
|---|---|---|---|---|
| Cesarean rate (overall) | 41 (23) | 32 (24) | 32 (24) | 0.95 |
| Nulliparous | 35 (24) | 28 (29) | 26 (37) | 0.12 |
| Multiparous | 6 (18) | 4 (10) | 6 (10) | 0.43 |
| Indication for cesarean | ||||
| Failed induction | 3 (8) | 10 (31) | 7 (22) | 0.17 |
| Arrest of dilation | 7 (18) | 3(9) | 9 (28) | |
| Arrest of descent | 10 (25) | 5 (16) | 4 (13) | |
| Failed operative | 2 (1) | 1 (1) | 0 (0) | |
| Non-reassuring tracing | 15 (38) | 13 (41) | 12 (38) | |
| Other | 3 (2) | 0 (0) | 0 (0) | |
| Cesarean for arrest disorders | ||||
| Arrest in first stage* | 10 (26) | 13 (33) | 16 (41) | 0.059 |
| Arrest in second stage† | 12 (55) | 6 (27) | 4 (18) | |
This table describes the Cesarean delivery rate and indications for Cesarean delivery by BMI category. %(n).
Arrest in 1st stage: failed IOL or arrest of dilation in AL
Arrest in 2nd stage: arrest of descent or failed operative.
While not statistically significant, there is a suggestion that Ob women have a different cesarean delivery rate than NW woman. Therefore, comparisons were made for both Ob and OW women separately compared to NW women. Among nulliparous women, the risk of cesarean delivery was significantly higher for Ob women compared to NW women (OR 1.91 [1.03–3.53], p = 0.040). This remained after adjusting for maternal age and race (OR 2.05 [1.07–3.94, p = 0.031). There was no statistical difference in the risk of cesarean delivery when comparing nulliparous OW women to NW women (OR 1.35 [0.75–2.41], p = 0.31). Among multiparous women, there was no statistical difference in the risk of cesarean delivery when comparing OW or Ob women to NW women (OR 0.51 [0.13–2.00], p = 0.34; OR 0.47 [0.14–1.61], p = 0.23 respectively).
Discussion
Our findings demonstrate that the association between obesity and labor length in women undergoing induction of labor differs based on phase of labor and parity. We also found no difference in the overall cesarean rate among BMI categories; however, Ob women were more likely to have a cesarean delivery for arrest in the first stage of labor, whereas NW women were more likely to have a cesarean delivery for arrest in the second stage.
Our findings support prior studies by Nuthalapaty et al. [12] and Vaharatian et al. [11], who found that maternal weight was inversely proportional to the cervical dilation rate in women undergoing induction of labor. Similar to our findings, Nuthalapaty et al. did not observe a difference in labor length for multiparous women after 4 cm cervical dilation. Vaharatian also found significantly longer median duration of labor from 4 to 10 cm in OW and Ob women compared with NW women [11]. Interestingly, in our cohort of women undergoing induction and categorized by strict BMI definitions, OW women were found to have the longest labor lengths. Selection and provider bias may have influenced this result, as physicians may have been more likely to perform a controlled cesarean delivery on an Ob nulliparous patient with slightly protracted labor while allowing an OW nulliparous woman more time for cervical dilation despite a similar labor course due to differences in BMI.
The progression of labor in our cohort, regardless of BMI, was faster than in prior studies evaluating induction of labor [11,14,23]. This is possibly due to our strict definition of AL that required a cervical dilation of at least 5 cm. The abovementioned studies [11,14,23] predominantly focused on labor starting at 4 cm. Prior studies show slower cervical dilation between 4 and 6 cm in women in spontaneous labor [21,22,24], even when restricting to women with BMIs > 30 [14].
Contrary to previously reported literature, we did not find a statistically significant association between cesarean delivery rates and BMI categories. However, our data suggested a trend toward an increased rate of cesarean delivery in OW and Ob women, and when compared to NW women, Ob nulliparous women had a higher risk of cesarean. Our data, however, did support prior studies by Fyfe et al. [4] and Robinson et al. [15] suggesting that Ob women are at increased risk for arrest disorders in the first stage of labor and not in the second stage.
Our study has several strengths. It is a large cohort study using data from one large urban hospital which minimizes practice variation during labor and delivery. We had a significant prevalence of OW and Ob women to study this question. Given the concern for inaccuracies with ICD-9 coding and the inability to appropriately classify BMI with coding, detailed chart abstraction and data collection were performed solely by two investigators (L.D.L., A.H.) with the use of strict definitions for induction and BMI classification to limit misclassification bias. Additionally, various induction methods and indications for induction were included, making this study generalizable.
Our study also has some limitations. First, this was a planned secondary analysis of a primary study and therefore the initial study population was not chosen to specifically answer our study question. As this is a retrospective study, the time spent in LL or AL is based on the timing of exams and it is impossible to know exactly when a woman first reached 5 cm dilation. However, this is a limitation of any labor study where data is based on clinical timing of exams. Our difference in methods may explain the different labor progression pattern we found compared to studies using interval-censored regression methods [11,23]. We also did not exclude those with neonatal macrosomia, diabetes or other factors associated with obesity that increase the risk of dysfunctional labor in our analysis [3,10], but we believe this makes our study more generalizable. Additionally, there was no standardized management for induction of labor, LL and AL, as this was provider dependent. This introduces a confounding factor of provider bias; however, limiting the data to one institution may have minimized this practice variation. Furthermore, our sample size may limit our ability to detect smaller differences in LL and AL duration in various BMI categories. It is also possible there are differences in labor duration by induction indication or method, but our sample size limited our ability to perform these sub-analyses.
It is encouraging to find that LL times did not significantly vary by BMI category nor did cesarean delivery rate. Additional research is needed to further understand labor curves in OW and Ob women to optimize labor and delivery management. If in fact AL progresses more slowly with increasing maternal weight both in induction and spontaneous labor, as indicated in our study and prior studies, then creation of separate definitions of protraction and arrest disorders for OW and Ob women may be warranted. This will have to be balanced with the overall success rate of vaginal deliveries and neonatal outcomes that result from allowing longer time for labor progression. However, this may in turn decrease potentially unnecessary cesarean deliveries in this population, an important public health concern considering the rising cesarean delivery rate [17] and the known increase in long-term maternal morbidity with subsequent pregnancies [25], as well as the multitude of surgical complications for which Ob women are at increased risk [2,26] after cesarean delivery.
Footnotes
Declaration of interest: Poster (#295) entitled “Labor length and cesarean delivery rate among OW and Ob women undergoing induction” was presented at the 33rd Annual Meeting of the Society for Maternal-Fetal Medicine in San Francisco, California on 14 February 2013. The authors report no other declarations of interest.
References
- 1.Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. JAMA. 2012;307:491–7. doi: 10.1001/jama.2012.39. [DOI] [PubMed] [Google Scholar]
- 2.Obesity in pregnancy. Committee Opinion No: 549, American College of Obstetricians and Gynecologists. Obstet Gynecol. 2013;121:213–7. doi: 10.1097/01.aog.0000425667.10377.60. [DOI] [PubMed] [Google Scholar]
- 3.Induction of Labor. ACOG Practice Bulletin No: 107. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2009;114:386–97. doi: 10.1097/AOG.0b013e3181b48ef5. [DOI] [PubMed] [Google Scholar]
- 4.Fyfe EM, Anderson NH, North RA, et al. Risk of first-stage and second-stage cesarean delivery by maternal body mass index among nulliparous women in labor at term. Obstet Gynecol. 2011;117:1315–22. doi: 10.1097/AOG.0b013e318217922a. [DOI] [PubMed] [Google Scholar]
- 5.Perlow JH, Morgan MA. Massive maternal obesity and perioperative cesarean morbidity. Am J Obstet Gynecol. 1994;170:560–5. doi: 10.1016/s0002-9378(94)70227-6. [DOI] [PubMed] [Google Scholar]
- 6.Weiss JL, Malone FD, Emig D, et al. Obesity, obstetric complications and cesarean delivery rate – a population-based screening study. FASTER Research Consortium. Am J Obstet Gynecol. 2004;190:1091–7. doi: 10.1016/j.ajog.2003.09.058. [DOI] [PubMed] [Google Scholar]
- 7.Crane SS, Wojtowoycz MA, Dye TD, et al. Association between pre-pregnancy obesity and the risk of cesarean delivery. Obstet Gynecol. 1997;89:213–16. doi: 10.1016/S0029-7844(96)00449-8. [DOI] [PubMed] [Google Scholar]
- 8.Kaiser PS, Kirby RS. Obesity as risk factor for cesarean in a low-risk population. Obstet Gynecol. 2001;97:39–43. doi: 10.1016/s0029-7844(00)01078-4. [DOI] [PubMed] [Google Scholar]
- 9.Brost BC, Goldenberg RL, Mercer BM, et al. The preterm prediction study: association of cesearean delivery with increases in maternal weight and body mass index. Am J Obstet Gynecol. 1997;177:333–41. doi: 10.1016/s0002-9378(97)70195-9. [DOI] [PubMed] [Google Scholar]
- 10.Ehrenberg HM, Durnwald CP, Catalano P, Mercer BM. The influence of obesity and diabetes on risk of cesarean delivery. Am J Obstet Gynecol. 2004;191:969–74. doi: 10.1016/j.ajog.2004.06.057. [DOI] [PubMed] [Google Scholar]
- 11.Vaharatian A, Zhang J, Troendle JF, et al. Maternal prepregnancy overweight and obesity and the pattern of labor progression in term nulliparous women. Obstet Gynecol. 2004;104:943–51. doi: 10.1097/01.AOG.0000142713.53197.91. [DOI] [PubMed] [Google Scholar]
- 12.Nuthalapaty FS, Rouse DJ, Owen J. The association of maternal weight with cesarean risk, labor duration, and cervical dilation rate during labor induction. Obstet Gynecol. 2003;103:452–6. doi: 10.1097/01.AOG.0000102706.84063.C7. [DOI] [PubMed] [Google Scholar]
- 13.Verdiales M, Pacheco C, Cohen WR. The effect of maternal obesity on course of labor. J Perinatal Med. 2009;37:651–5. doi: 10.1515/JPM.2009.110. [DOI] [PubMed] [Google Scholar]
- 14.Norman SM, Tuuli MG, Odibo AO, et al. The effects of obesity on the first stage of labor. Obstet Gynecol. 2012;120:130–5. doi: 10.1097/AOG.0b013e318259589c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Robinson BK, Mapp DC, Bloom SL, et al. Increasing maternal body mass index and characteristics of the second stage of labor. Obstet Gynecol. 2011;118:1309–13. doi: 10.1097/AOG.0b013e318236fbd1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Buhimschi CS, Buhimschi IA, Malinow AM, Weiner CP. Intrauterine pressure during the second stage of labor in obese women. Obstet Gynecol. 2004;103:225–30. doi: 10.1097/01.AOG.0000102706.84063.C7. [DOI] [PubMed] [Google Scholar]
- 17.Spong CY, Berghella V, Wenstrom KD, et al. Preventing the first cesarean delivery. Obstet Gynecol. 2012;120:1181–93. doi: 10.1097/aog.0b013e3182704880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Levine LD, Bogner HR, Hirshberg A, et al. Term induction of labor and subsequent preterm birth. Am J Obstet Gynecol. 2013 Oct 30; doi: 10.1016/j.ajog.2013.10.877. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Institute of Medicine and National Research Council Weight gain during pregnancy: reexamining the guidelines. The National Academies Press; 2009. [PubMed] [Google Scholar]
- 20.Friedman EA. Primigravid labor; a graphicostatistical analysis. Obstet Gynecol. 1955;6:567–89. doi: 10.1016/s0029-7844(02)02398-0. [DOI] [PubMed] [Google Scholar]
- 21.Zhang J, Landy HL, Branch DW, et al. Contemporary patterns of spontaneous labor with normal neonatal outcomes. Obstet Gynecol. 2010;116:1281–7. doi: 10.1097/AOG.0b013e3181fdef6e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang J, Troendle J, Mikolajczk R, et al. The natural history of the normal first stage of labor. Obstet Gynecol. 2010;115:705–10. doi: 10.1097/AOG.0b013e3181d55925. [DOI] [PubMed] [Google Scholar]
- 23.Harper LM, Caughey AB, Odibo AO, et al. Normal progress of induced labor. Obstet Gynecol. 2012;119:1113–18. doi: 10.1097/AOG.0b013e318253d7aa. [DOI] [PubMed] [Google Scholar]
- 24.Peisner DB, Rosen MG. Transition from latent to active labor. Obstet Gynecol. 1986;68:448–51. [PubMed] [Google Scholar]
- 25.Miller ES, Hahn K, Grobman WA. Consequences of a primary elective cesarean delivery across the reproductive life. Obstet Gynecol. 2013;121:789–97. doi: 10.1097/AOG.0b013e3182878b43. [DOI] [PubMed] [Google Scholar]
- 26.Cedergran M. Maternal morbid obesity and the risk of adverse pregnancy outcomes. Obstet Gynecol. 2004;103:219–24. doi: 10.1097/01.AOG.0000107291.46159.00. [DOI] [PubMed] [Google Scholar]