ABSTRACT
The multidrug resistance profiles of Klebsiella pneumoniae carbapenemase (KPC) producers have led to increased clinical polymyxin use. Combination therapy with polymyxins may improve treatment outcomes, but it is uncertain which combinations are most effective. Clinical successes with intravenous minocycline-based combination treatments have been reported for infections caused by carbapenemase-producing bacteria. The objective of this study was to evaluate the in vitro activity of polymyxin B and minocycline combination therapy against six KPC-2-producing K. pneumoniae isolates (minocycline MIC range, 2 to 32 mg/liter). Polymyxin B monotherapy (0.5, 1, 2, 4, and 16 mg/liter) resulted in a rapid reduction of up to 6 log in bactericidal activity followed by regrowth by 24 h. Minocycline monotherapy (1, 2, 4, 8, and 16 mg/liter) showed no reduction of activity of >1.34 log against all isolates, although concentrations of 8 and 16 mg/liter prolonged the time to regrowth. When the therapies were used in combination, rapid bactericidal activity was followed by slower regrowth, with synergy (60 of 120 combinations at 24 h, 19 of 120 combinations at 48 h) and additivity (43 of 120 combinations at 24 h, 44 of 120 combinations at 48 h) against all isolates. The extent of killing was greatest against the more susceptible polymyxin B isolates (MICs of ≤0.5 mg/liter) regardless of the minocycline MIC. The pharmacodynamic activity of combined polymyxin B-minocycline therapy against KPC-producing K. pneumoniae is dependent on polymyxin B susceptibility. Further in vitro and animal studies must be performed to fully evaluate the efficacy of this drug combination.
KEYWORDS: polymyxin, minocycline, Klebsiella pneumoniae, KPC, MDR, antimicrobial combinations, multidrug resistance
INTRODUCTION
Klebsiella pneumoniae is a ubiquitous colonizer of the gastrointestinal tract and a common cause of nosocomial pneumonia and of bloodstream and urinary tract infections (1, 2). Drug-resistant K. pneumoniae infections are often treated with carbapenems, but the increasing dissemination of carbapenem-resistant strains has led to higher mortality rates and longer hospital stays for affected individuals (2–4). Since K. pneumoniae carbapenemase (KPC) was first detected in 2001 in North Carolina, KPC-producing Enterobacteriaceae have become endemic in the United States, Greece, Israel, Italy, China, Brazil, Colombia, and Argentina (5–7). Monotherapy against KPC-producing bacteria has higher treatment failure rates than combination therapy (8), with the development of resistance likely to be partially responsible. Cases of colistin (polymyxin E)-resistant carbapenemase-producing Enterobacteriaceae strains arising from suboptimal exposure to colistin have been reported in some countries (9, 10). Further, the recent emergence of the first plasmid-mediated polymyxin resistance mechanism in Enterobacteriaceae is indicative of the need for combination therapy against these bacteria (9, 11–13).
Although minocycline is not commonly used for the treatment of KPC-producing K. pneumoniae infections, there are reports of clinical success with intravenous minocycline-based combinations against carbapenemase-producing K. pneumoniae and Acinetobacter baumannii infections. Further research into the efficacy of minocycline and polymyxin combination regimens is therefore warranted (14–16). Minocycline inhibits bacterial protein synthesis and has favorable tissue penetrability due to its lipophilicity (16). Minocycline combined with colistin is synergistic against multidrug-resistant (MDR) A. baumannii (17–19) based on the hypothesis that polymyxin disrupts outer membrane integrity to raise intracellular concentrations of minocycline in Gram-negative bacteria (20, 21). The objective of this study was to evaluate the rate and extent of killing activity of polymyxin B and minocycline alone and in combination against KPC-2-producing K. pneumoniae isolates using static in vitro time-kill studies.
RESULTS
Characterization of clinical isolates revealed KPC-2 production in all isolates. No additional β-lactamases were detected. The mgrB gene was functional in all polymyxin B-sensitive isolates and was truncated in the polymyxin B-resistant BRKP57 isolate. None of the isolates contained the tet(B) resistance determinant.
Monotherapy.
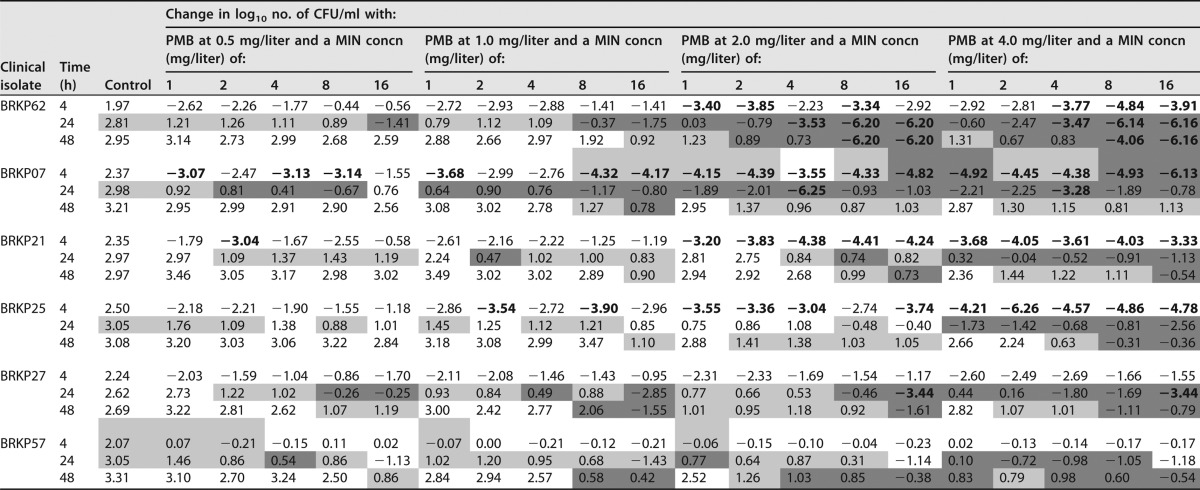
Log changes in CFU per milliliter at 4, 24, and 48 h compared to the starting inoculum at 0 h for both polymyxin B monotherapy and minocycline monotherapy are shown in Table 1. Polymyxin B monotherapy displayed concentration-dependent rapid bactericidal activity for all concentrations that peaked by 4 h against the polymyxin B-sensitive BRKP62, BRKP07, BRKP21, BRKP25, and BRKP27 isolates (Table 1; see also Fig. S1 in the supplemental material). However, bactericidal activity was not sustained beyond 8 h, and regrowth similar to the growth control results occurred beyond 24 h at polymyxin B concentrations of ≤4 mg/liter. The highest polymyxin B concentration evaluated (16 mg/liter) demonstrated a lower rate of regrowth similar to the growth control results, with a slight delay against BRKP62, BRKP21, and BRKP25 (Fig. S1a, c, and d). Against the polymyxin B-resistant BRKP57 isolate, polymyxin B at 16 mg/liter showed minimal activity, resulting in a <1 log10 reduction followed by regrowth.
TABLE 1.
The change in log10 CFU per milliliter at 4, 24, and 48 h during time-kill experiments with monotherapy with polymyxin B or minocycline against an inoculum of ∼106 CFU/ml of KPC-producing K. pneumoniae clinical isolatesa
| Clinical isolate | Time (h) | Change in log10 no. of CFU/ml with: |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | PMB (mg/liter) |
MIN (mg/liter) |
||||||||||
| 0.5 | 1 | 2 | 4 | 16 | 1 | 2 | 4 | 8 | 16 | |||
| BRKP62 | 4 | 1.97 | −3.32 | −4.58 | −4.84 | −6.11 | −6.25 | 2.19 | 1.41 | −0.12 | −0.15 | −0.30 |
| 24 | 2.81 | 2.90 | 2.61 | 2.74 | 2.51 | −3.04 | 3.27 | 3.03 | 2.48 | 2.38 | 1.06 | |
| 48 | 2.95 | 3.09 | 2.90 | 3.10 | 3.07 | 2.06 | 3.20 | 3.37 | 3.18 | 3.11 | 3.14 | |
| BRKP07 | 4 | 2.12 | −3.49 | −2.77 | −2.70 | −2.60 | −6.01 | 1.71 | −0.18 | −0.79 | −0.66 | −0.55 |
| 24 | 2.98 | 3.00 | 3.08 | 3.06 | 2.97 | 2.20 | 2.88 | 2.90 | 2.78 | 2.70 | 1.38 | |
| 48 | 3.21 | 3.16 | 3.21 | 3.32 | 3.21 | 3.19 | 2.77 | 2.91 | 2.84 | 2.79 | 2.91 | |
| BRKP21 | 4 | 2.35 | −3.48 | −3.44 | −3.66 | −6.21 | −6.00 | 2.29 | 0.70 | 0.30 | −0.06 | −0.87 |
| 24 | 2.97 | 3.03 | 2.71 | 2.83 | 2.89 | −1.20 | 3.00 | 3.04 | 2.91 | 2.91 | 2.69 | |
| 48 | 2.97 | 3.24 | 2.88 | 2.89 | 3.05 | 3.12 | 2.95 | 3.04 | 2.86 | 3.12 | 3.19 | |
| BRKP25 | 4 | 2.42 | −4.00 | −4.26 | −4.18 | −4.62 | −5.87 | 2.55 | 2.34 | 0.80 | −0.01 | −0.20 |
| 24 | 3.05 | 3.15 | 3.07 | 1.27 | 2.91 | −4.09 | 3.07 | 2.24 | 2.14 | 2.26 | 0.10 | |
| 48 | 3.08 | 3.28 | 3.48 | 3.32 | 3.41 | 2.09 | 3.21 | 2.73 | 2.56 | 2.58 | 2.73 | |
| BRKP27 | 4 | 1.93 | −3.07 | −1.99 | −2.29 | −2.56 | −4.49 | 1.17 | 1.27 | −0.78 | −0.78 | −1.34 |
| 24 | 2.62 | 2.83 | 2.76 | 2.00 | 2.47 | 2.89 | 3.07 | 2.91 | 2.77 | 2.99 | 2.75 | |
| 48 | 2.69 | 2.65 | 2.89 | 2.62 | 2.29 | 2.61 | 2.99 | 3.00 | 2.99 | 3.05 | 2.55 | |
| BRKP57 | 4 | 2.07 | 1.44 | 1.52 | 1.13 | 0.59 | −0.48 | 1.74 | 0.83 | 0.50 | 0.00 | −0.35 |
| 24 | 3.05 | 2.98 | 3.09 | 3.16 | 3.09 | 3.15 | 3.02 | 2.54 | 2.64 | 2.11 | −0.34 | |
| 48 | 3.31 | 3.16 | 3.20 | 3.40 | 3.37 | 3.19 | 3.15 | 2.73 | 3.06 | 2.97 | 2.59 | |
Bactericidal activity (≥3 log10 CFU/ml reduction compared to the initial inoculum) is shown in bold. PMB, polymyxin B; MIN, minocycline.
Minocycline monotherapy did not result in bactericidal activity against any of the isolates at any of the minocycline concentrations (Table 1). All monotherapy concentrations eventually resulted in growth similar to the growth control results by 48 h, although the rate of regrowth decreased with increasing minocycline concentrations (Fig. S2). The results seen with minocycline concentrations of 1, 2, and 4 mg/liter were comparable to the growth control results regardless of the minocycline susceptibility of the isolate. Minocycline at 8 mg/liter resulted in a <1 log10 reduction at 4 h against all isolates (Table 1), with regrowth observed by 24 h in the minocycline-susceptible (BRKP07, BRKP62, and BRKP57) isolates and by 8 h in the minocycline-resistant (BRKP27, BRKP21, and BRKP25) isolates. Minocycline at 16 mg/liter performed slightly better, resulting in a maximal reduction of 1.34 log10 at 4 h followed by regrowth in all isolates by 28 h.
Combination therapy.
Log changes in CFU per milliliter at 4, 24, and 48 h compared to the starting inoculum that resulted from the use of minocycline and polymyxin B in combination are shown in Table 2. Time-kill profiles for polymyxin B in combination with minocycline against four isolates with various levels of polymyxin B susceptibility (for BRKP62, polymyxin B MIC = <0.5 mg/liter; for BRKP25, polymyxin B MIC = 0.5 mg/liter; for BRKP27, polymyxin B MIC = 1 mg/liter; for BRKP57, polymyxin B MIC = 64 mg/liter) are shown in Fig. 1.
TABLE 2.
The change in log10 CFU per milliliter at 4, 24, and 48 h during time-kill experiments with polymyxin B in combination with minocycline against an inoculum of ∼106 CFU/ml of KPC-producing K. pneumoniae clinical isolatesa
Bactericidal activity (≥3 log10 CFU/ml reduction compared to the initial inoculum) is indicated in bold. Additivity is defined as a reduction of between 1 and 2 log10 CFU/ml and synergy is defined as a reduction of ≥2 log10 CFU/ml caused by the combination compared to the results seen with the most active single agent in the combination. Additivity is highlighted with light gray shading and synergy with dark gray shading. PMB, polymyxin B; MIN, minocycline.
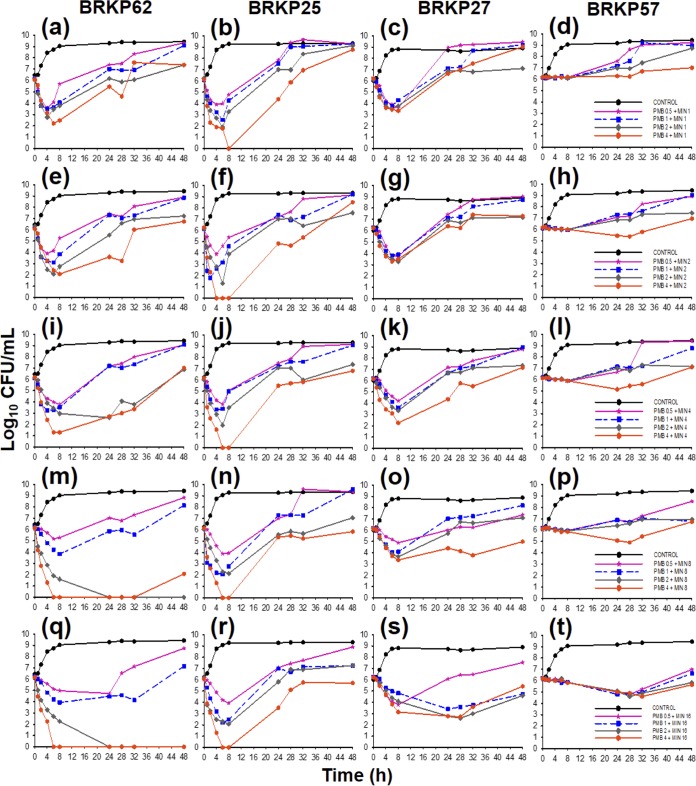
FIG 1.
Time-kill curves of polymyxin B (PMB) at 0.5, 1, 2, and 4 mg/liter in combination with minocycline (MIN) at 1, 2, 4, 8, and 16 mg/liter against an inoculum of ∼106 CFU/ml of the following KPC-producing K. pneumoniae clinical isolates: (i) BRKP62 (polymyxin B MIC, <0.5 mg/liter; minocycline MIC, 4 mg/liter), (ii) BRKP25 (polymyxin B MIC, 0.5 mg/liter; minocycline MIC, 32 mg/liter), (iii) BRKP27 (polymyxin B MIC, 1 mg/liter; minocycline MIC, 8 mg/liter), and (iv) BRKP57 (polymyxin B MIC, 64 mg/liter; minocycline MIC, 4 mg/liter).
Against BRKP62, an isolate susceptible to both polymyxin B and minocycline (polymyxin B MIC, <0.5 mg/liter; minocycline MIC, 4 mg/liter), polymyxin at 0.5 and 1 mg/liter in combination with all minocycline concentrations resulted in a >2 log reduction by 8 h followed by regrowth similar to the growth control results at 48 h. Polymyxin B at ≥2 mg/liter in combination with minocycline at 1, 2, and 4 mg/liter resulted in bactericidal activity by 4 h followed by regrowth at up to 107 CFU/ml at 48 h (Fig. 1a, e, and i and Table 2). Polymyxin B at 2 and 4 mg/liter combined with minocycline at 8 and 16 mg/liter resulted in undetectable levels of isolate BRKP62 by 6 h that were sustained up to 48 h (Fig. 1m and q and Table 2).
Against the polymyxin B-sensitive and minocycline-resistant BRKP25 isolate (polymyxin B MIC, 0.5 mg/liter; minocycline MIC, 32 mg/liter), polymyxin B concentrations of ≥2 mg/liter in combination with all minocycline concentrations were bactericidal by 4 h followed by regrowth beyond 24 h (Fig. 1b, f, j, n, and r). Surprisingly, the same activity was seen against BRKP07 and BRKP21, which also have a polymyxin B MIC of 0.5 mg/liter but different minocycline MICs of 2 and 16 mg/liter, respectively (Table 2; see also Fig. S3). Increasing minocycline concentrations against these isolates failed to improve the extent of killing regardless of the minocycline MIC.
Against BRKP27 (polymyxin B MIC, 1.0 mg/liter; minocycline MIC, 8 mg/liter), polymyxin B and minocycline together at all concentrations resulted in a >2 log reduction by 8 h followed by regrowth by 48 h (Fig. 1c, g, k, o, and s).
The combination demonstrated minimal activity against the highly polymyxin B-resistant and minocycline-sensitive BRKP57 isolate (polymyxin B MIC, 64 mg/liter; minocycline MIC, 4 mg/liter) (Fig. 1d, h, l, p, and t). Polymyxin B at all concentrations combined with minocycline at 16 mg/liter resulted in a >1 log reduction at 24 h followed by regrowth by 48 h. Across all isolates, the pharmacodynamic activity decreased with increasing polymyxin B MIC regardless of the level of minocycline susceptibility (Fig. 1).
Combination therapy did not result in synergy before 4 h except for BRKP07, as the addition of minocycline did not increase the extent of early bactericidal activity seen with polymyxin B alone (Table 2). At 24 h, 60 of 120 (50%) combinations were considered synergistic and 43 of 120 (36%) combinations were considered additive. At 48 h, 19 of 120 (16%) combinations were synergistic and 44 of 120 (37%) were additive. A striking similarity between all isolates was that combinations with polymyxin B at 4 mg/liter were synergistic.
DISCUSSION
Here we evaluated polymyxin B and minocycline as monotherapy and in combination against six clinical K. pneumoniae isolates with various levels of susceptibilities to each antibiotic. Polymyxin B monotherapy resulted in rapid bactericidal activity followed by regrowth by 24 h. While minocycline monotherapy at high concentrations increased the time to regrowth of all isolates, no bactericidal activity was observed. In combination with polymyxin B, the rapid bactericidal killing was followed by slower regrowth, as evidenced by synergy up to 48 h against all isolates. The extent of killing was greatest against the more susceptible polymyxin B isolates, as early bactericidal activity was seen when polymyxin B MICs were ≤0.5 mg/liter regardless of the minocycline MIC. Bactericidal activity was sustained at 48 h only against the most polymyxin B-susceptible isolate (MIC, <0.5). Killing activity was attenuated with increasing polymyxin B MIC. In addition, bactericidal activity was observed at polymyxin B concentrations of ≥2 mg/liter, and synergy was observed against all isolates at the high concentration of 4 mg/liter. These findings further confirm that the pharmacodynamic activity of this combination is dependent on the level of polymyxin B susceptibility.
Previous studies have evaluated polymyxin (polymyxin B or colistin) in combination with minocycline against A. baumannii in both in vitro and in vivo models of infection with favorable results (17–19). Bowers et al. (20) determined that subinhibitory concentrations of polymyxin B (0.5 mg/liter) could increase intracellular minocycline concentrations (2 and 8 mg/liter) and enhance minocycline's activity in static time-kill experiments against three clinical isolates and one laboratory isolate (20) with polymyxin B MICs of ≥2 mg/liter and minocycline MICs ranging between 0.25 and 16 mg/liter. Zhang et al. (21) determined that the combination significantly reduced the MICs (polymyxin B, 0.25 to 4 mg/liter; minocycline, 0.25 to 8 mg/liter) against 25 resistant A. baumannii strains compared to the MIC for either polymyxin B (1 to 8 mg/liter) or minocycline (4 to 16 mg/liter) used as monotherapy (21). They also determined the synergy of the combination using the fractional inhibitory concentration index (FICI). Yang et al. (22) tested the colistin and minocycline combination against four minocycline-resistant and colistin-sensitive isolates (minocycline MICs of 16 and 32 mg/liter; colistin MIC, 0.5 mg/liter) and revealed similar FICI results using a checkerboard synergy assay (22). Time-kill experiments in the same study using minocycline at 4 and 16 mg/liter in combination with colistin at 0.5 mg/liter showed synergy in all four isolates. Our study revealed similar findings for KPC-producing K. pneumoniae, as polymyxin B and minocycline were synergistic. However, bactericidal activity and synergy were more apparent when polymyxin B concentrations were ≥2 mg/liter against polymyxin-susceptible isolates.
Several hypotheses may explain the efficacy of this combination (20, 22, 23). Polymyxin B might increase membrane permeability and intracellular minocycline concentrations. Polymyxins competitively displace cations from the phosphate groups of lipopolysaccharide and phospholipids of lipid A in the outer membrane of Gram-negative bacteria, leading to leakage of intracellular contents and eventual cell death. Polymyxin B may also alter the function of efflux pumps in bacterial membranes that are responsible for minocycline resistance (20). Tetracycline resistance is often due to the presence of various tet genes that code for upregulation of efflux pumps or ribosomal protection. Unlike that of the other tetracyclines, minocycline resistance is primarily attributable to the tet(B) gene, which upregulates efflux pumps (24). Our results are consistent with both hypotheses, as the pharmacodynamic activity was greatest against the more polymyxin-susceptible strains and at higher polymyxin B concentrations regardless of minocycline susceptibility. However, resistance to polymyxin B is another growing concern. Dysfunction of the mgrB gene has been found to upregulate the PhoP/PhoQ system, which reduces the negative charge of lipid A and subsequently the affinity of polymyxin B (25).
To our knowledge, this is the first report to evaluate polymyxin B and minocycline against KPC-producing K. pneumoniae. Further, we evaluated this combination against clinical isolates with a broad range of polymyxin B and minocycline susceptibilities. The combination performed better than monotherapy against all isolates by prolonging the time to regrowth to control levels from 24 h to 48 h. Bactericidal activity was observed against all polymyxin B-sensitive isolates with MICs of ≤0.5 mg/liter at clinically achievable concentrations. As such, this combination might be a viable treatment option against infections with such isolates. Although synergy was present, this combination did not demonstrate sustained bactericidal activity against these isolates past 8 h at clinically achievable concentrations. We also showed that the combination did not perform well against the highly polymyxin B-resistant BRKP57 isolate. Triple combinations, although infrequently used in the United States, may be necessary for prolonged sustained activity to treat infections due to highly resistant K. pneumoniae isolates. Previous studies have suggested a clinical and microbiological benefit to three-drug combinations (7, 26). Moreover, adding an additional drug such as a carbapenem or fosfomycin to the polymyxin B and minocycline combination may increase efficacy against KPC-producing K. pneumoniae by increasing the log kill rate of polymyxin B with minocycline to theoretically slow any regrowth. Additional studies to assess such combinations are warranted.
This study had limitations. Antibiotic concentrations in time-kill experiments are inherently constant and do not fully simulate the pharmacokinetics of antibiotic drug regimens in the clinical setting. Therefore, these findings may not accurately reflect the activity of polymyxin B and minocycline in vivo in the presence of drug metabolism and elimination and/or in the presence of an active immune system. The pharmacodynamic activity in response to humanized regimens needs to be evaluated, perhaps first employing in vivo infection models, to further our knowledge and enable translation of these combinations to the clinic.
As KPC-producing K. pneumoniae strains continue to spread and treatment options become more limited, it is important to determine which combinations of existing antibiotics are most effective and to prevent the emergence of further resistance. In this study, polymyxin B and minocycline showed synergistic activity at clinically relevant concentrations, although sustained activity was not achieved beyond 24 h and the mechanism has yet to be fully elucidated. Further in vitro and animal studies should be performed to evaluate the efficacy of this drug combination.
MATERIALS AND METHODS
Bacterial isolates, susceptibilities, and characterization.
Six clinical K. pneumoniae strains (BRKP62 [polymyxin B MIC, <0.5 mg/liter; minocycline MIC, 4 mg/liter], BRKP07 [polymyxin B MIC, 0.5 mg/liter; minocycline MIC, 2 mg/liter], BRKP21 [polymyxin B MIC, 0.5 mg/liter; minocycline MIC, 16 mg/liter], BRKP25 [polymyxin B MIC, 0.5 mg/liter; minocycline MIC, 32 mg/liter], BRKP27 [polymyxin B MIC, 1 mg/liter; minocycline MIC, 8 mg/liter], and BRKP57 [polymyxin B MIC, 64 mg/liter; minocycline MIC, 4 mg/liter]) were obtained from the Instituto Dante Pazzanese de Cardiologia, Sao Paulo, Brazil, during KPC outbreaks between June 2009 and June 2013 (27). MICs were determined in triplicate using broth microdilution according to Clinical and Laboratory Standards Institute (CLSI) guidelines (28). Genomic DNA was isolated from bacterial isolates using a DNA isolation kit (E.Z.N.A. bacterial DNA kit; Omega Biotek, Norcross, GA). PCR analyses to evaluate the presence of seven different β-lactamases belonging to Ambler class A (GES and KPC), B (NDM, VIM, and IMP), and D (OXA-48 and OXA-40) (29) and of mgrB (25) and tet(B) (30) were carried out using specific primers (see Table S1 in the supplemental material) commercially synthesized by IDT (Integrated DNA Technologies, Inc., Coralville, IA). Q5 Hi-Fidelity Taq DNA polymerase (NEB, Ipswich, MA) was used in PCRs. The PCR product sequencing was performed at the sequencing facility at Roswell Park Cancer Institute, Buffalo, NY. The NCBI website (www.ncbi.nlm.nih.gov) was used for nucleotide and deduced protein sequence analysis. The insertion sequence (IS) was analyzed using the IS finder website (https://www-is.biotoul.fr/). All isolates were stored at −80°C and subcultured on Mueller-Hinton agar (MHA) 1 day prior to each experiment.
Antibiotics and media.
Stock solutions of polymyxin B (Sigma-Aldrich, USA; catalog no. WXBB4470V) and minocycline (Sigma-Aldrich, USA; catalog no. WXBB4793V) were prepared in water and filter sterilized through 0.20-μm-pore-size syringe filters (Corning Inc., Corning, NY) immediately prior to each experiment. Susceptibility testing and static time-kill studies were performed using cation-adjusted (25.0 mg/liter Ca2+ and 12.5 mg/liter Mg2+) Mueller-Hinton broth (CAMHB) (Becton, Dickinson and Company, Sparks, MD).
Time-kill studies.
The bacterial killing activity of polymyxin B and minocycline as monotherapies and in combination was evaluated against the six isolates by performing time-kill experiments over 48 h using methods previously described (26). Polymyxin B concentrations of 0.5, 1, 2, 4, and 16 mg/liter and minocycline concentrations of 1, 2, 4, 8, and 16 mg/liter were evaluated as monotherapy. A 4-by-5 concentration matrix of polymyxin B (0.5, 1, 2, and 4 mg/liter) in combination with minocycline (1, 2, 4, 8, and 16 mg/liter) and a growth control were also evaluated. The range of concentrations selected included clinically achievable unbound plasma concentrations and supratherapeutic concentrations to evaluate any potential benefits of intensive dosing. Minocycline dosed as a 200-mg intravenous infusion over 30 to 60 min yields maximum concentrations of 3 to 8.75 mg/liter, and the steady-state plasma concentrations following infusion of 100 mg of minocycline every 12 h range between 1 and 4 mg/liter (31–33). The average steady-state plasma concentration of polymyxin B is reported as approximately 2.79 mg/liter for doses ranging from 0.45 to 3.38 mg/kg of body weight/day (34). Levels of protein binding for minocycline and polymyxin B are ∼76% and ∼58%, respectively.
Subcultured bacterial isolates were added to CAMHB to produce a bacterial suspension with an initial inoculum of ∼106 CFU/ml. The antibiotic(s) was added to the bacterial suspension in the logarithmic-growth phase at 37°C. Samples were obtained at 0, 1, 2, 4, 6, 8, 24, 28, 32, and 48 h for bacterial quantification. A ProtoCOL automated colony counter (Synbiosis, Cambridge, United Kingdom) was used to quantify bacteria after 24 h of incubation at 37°C. The limit of quantification was 100 CFU/ml.
Pharmacodynamic analysis.
The evaluation of the pharmacodynamic effect (E) of the individual and combination therapies was performed according to the change in log10 CFU per milliliter at an early time point of 4 h and at later time points of 24 and 48 h compared to the change in log10 CFU per milliliter at baseline (0 h) according to the following equation:
Bactericidal activity was defined as a ≥3 log10 CFU/ml reduction from baseline. Synergy was defined as a ≥2 log10 CFU/ml reduction and additivity as a ≥1 to <2 log10 reduction in CFU/ml caused by the combination of polymyxin B and minocycline compared to the most active single agent in the combination.
Supplementary Material
ACKNOWLEDGMENTS
We thank Alison Vitale for all her help and support in the laboratory.
This study received no funding.
We have no conflicts of interest to declare.
Footnotes
Supplemental material for this article may be found at https://doi.org/10.1128/AAC.00073-17.
REFERENCES
- 1.Podschun R, Ullmann U. 1998. Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin Microbiol Rev 11:589–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tzouvelekis LS, Markogiannakis A, Psichogiou M, Tassios PT, Daikos GL. 2012. Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clin Microbiol Rev 25:682–707. doi: 10.1128/CMR.05035-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Patel G, Huprikar S, Factor SH, Jenkins SG, Calfee DP. 2008. Outcomes of carbapenem-resistant Klebsiella pneumoniae infection and the impact of antimicrobial and adjunctive therapies. Infect Control Hosp Epidemiol 29:1099–1106. doi: 10.1086/592412. [DOI] [PubMed] [Google Scholar]
- 4.Schwaber MJ, Klarfeld-Lidji S, Navon-Venezia S, Schwartz D, Leavitt A, Carmeli Y. 2008. Predictors of carbapenem-resistant Klebsiella pneumoniae acquisition among hospitalized adults and effect of acquisition on mortality. Antimicrob Agents Chemother 52:1028–1033. doi: 10.1128/AAC.01020-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Arnold RS, Thom KA, Sharma S, Phillips M, Johnson JK, Morgan DJ. 2011. Emergence of Klebsiella pneumoniae carbapenemase (KPC)-producing bacteria. South Med J 104:40–45. doi: 10.1097/SMJ.0b013e3181fd7d5a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gupta N, Limbago BM, Patel JB, Kallen AJ. 2011. Carbapenem-resistant Enterobacteriaceae: epidemiology and prevention. Clin Infect Dis 53:60–67. doi: 10.1093/cid/cir202. [DOI] [PubMed] [Google Scholar]
- 7.Munoz-Price LS, Poirel L, Bonomo RA, Schwaber MJ, Daikos GL, Cormican M, Cornaglia G, Garau J, Gniadkowski M, Hayden MK, Kumarasamy K, Livermore DM, Maya JJ, Nordmann P, Patel JB, Paterson DL, Pitout J, Villegas MV, Wang H, Woodford N, Quinn JP. 2013. Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect Dis 13:785–796. doi: 10.1016/S1473-3099(13)70190-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee GC, Burgess DS. 2012. Treatment of Klebsiella pneumoniae carbapenemase (KPC) infections: a review of published case series and case reports. Ann Clin Microbiol Antimicrob 11:32. doi: 10.1186/1476-0711-11-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Falagas ME, Lourida P, Poulikakos P, Rafailidis PI, Tansarli GS. 2014. Antibiotic treatment of infections due to carbapenem-resistant Enterobacteriaceae: systematic evaluation of the available evidence. Antimicrob Agents Chemother 58:654–663. doi: 10.1128/AAC.01222-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Matthaiou DK, Michalopoulos A, Rafailidis PI, Karageorgopoulos DE, Papaioannou V, Ntani G, Samonis G, Falagas ME. 2008. Risk factors associated with the isolation of colistin-resistant gram-negative bacteria: a matched case-control study. Crit Care Med 36:807–811. doi: 10.1097/CCM.0B013E3181652FAE. [DOI] [PubMed] [Google Scholar]
- 11.Liu YY, Wang Y, Walsh TR, Yi LX, Zhang R, Spencer J, Doi Y, Tian G, Dong B, Huang X, Yu LF, Gu D, Ren H, Chen X, Lv L, He D, Zhou H, Liang Z, Liu JH, Shen J. 2016. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 16:161–168. doi: 10.1016/S1473-3099(15)00424-7. [DOI] [PubMed] [Google Scholar]
- 12.Paul M, Carmeli Y, Durante-Mangoni E, Mouton JW, Tacconelli E, Theuretzbacher U, Mussini C, Leibovici L. 2014. Combination therapy for carbapenem-resistant Gram-negative bacteria. J Antimicrob Chemother 69:2305–2309. doi: 10.1093/jac/dku168. [DOI] [PubMed] [Google Scholar]
- 13.Zavascki AP, Bulitta JB, Landersdorfer CB. 2013. Combination therapy for carbapenem-resistant Gram-negative bacteria. Expert Rev Anti Infect Ther 11:1333–1353. doi: 10.1586/14787210.2013.845523. [DOI] [PubMed] [Google Scholar]
- 14.Bishburg E, Shah M, Chan T. 2014. Use of intravenous minocycline for the treatment of methicillin-resistant Staphylococcus aureus (MRSA) and resistant Gram-negative organisms: experience in a tertiary care hospital. Infect Dis Clin Pract (Baltim Md) 22:26–31. doi: 10.1097/IPC.0b013e31828bbb82. [DOI] [Google Scholar]
- 15.Pogue JM, Neelakanta A, Mynatt RP, Sharma S, Lephart P, Kaye KS. 2014. Carbapenem-resistance in Gram-negative bacilli and intravenous minocycline: an antimicrobial stewardship approach at the Detroit Medical Center. Clin Infect Dis 59:S388–S393. doi: 10.1093/cid/ciu594. [DOI] [PubMed] [Google Scholar]
- 16.Ritchie DJ, Garavaglia-Wilson A. 2014. A review of intravenous minocycline for treatment of multidrug-resistant Acinetobacter infections. Clin Infect Dis 59:S374–S380. doi: 10.1093/cid/ciu613. [DOI] [PubMed] [Google Scholar]
- 17.Tan TY, Ng LSY, Tan E, Huang G. 2007. In vitro effect of minocycline and colistin combinations on imipenem-resistant Acinetobacter baumannii clinical isolates. J Antimicrob Chemother 60:421–423. doi: 10.1093/jac/dkm178. [DOI] [PubMed] [Google Scholar]
- 18.Liang W, Liu XF, Huang J, Zhu DM, Li J, Zhang J. 2011. Activities of colistin- and minocycline-based combinations against extensive drug resistant Acinetobacter baumannii isolates from intensive care unit patients. BMC Infect Dis 11:109. doi: 10.1186/1471-2334-11-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rodríguez CH, Nastro M, Vay C, Famiglietti A. 2015. In vitro activity of minocycline alone or in combination in multidrug-resistant Acinetobacter baumannii isolates. J Med Microbiol 64:1196–1200. doi: 10.1099/jmm.0.000147. [DOI] [PubMed] [Google Scholar]
- 20.Bowers DR, Cao H, Zhou J, Ledesma KR, Sun D, Lomovskaya O, Tam VH. 2015. Assessment of minocycline and polymyxin B combination against Acinetobacter baumannii. Antimicrob Agents Chemother 59:2720–2725. doi: 10.1128/AAC.04110-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang Y, Chen F, Sun E, Ma R, Qu C, Ma L. 2013. In vitro antibacterial activity of combinations of fosfomycin, minocycline and polymyxin B on pan-drug-resistant Acinetobacter baumannii. Exp Ther Med 5:1737–1739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang Y-S, Lee Y, Tseng K-C, Huang W-C, Chuang M-F, Kuo S-C, Yang Lauderdale T-L, Chen T-L. 25 April 2016. In vivo and in vitro efficacy of minocycline-based combination therapy for minocycline-resistant Acinetobacter baumannii. Antimicrob Agents Chemother doi: 10.1128/aac.02994-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Barth N, Ribeiro VB, Zavascki AP. 2015. In vitro activity of polymyxin B plus imipenem, meropenem, or tigecycline against KPC-2-producing Enterobacteriaceae with high MICs for these antimicrobials. Antimicrob Agents Chemother 59:3596–3597. doi: 10.1128/AAC.00365-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chopra I, Roberts M. 2001. Tetracycline antibiotics: mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol Mol Biol Rev 65:232–260. doi: 10.1128/MMBR.65.2.232-260.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Poirel L, Jayol A, Bontron S, Villegas MV, Ozdamar M, Turkoglu S, Nordmann P. 2015. The mgrB gene as a key target for acquired resistance to colistin in Klebsiella pneumoniae. J Antimicrob Chemother 70:75–80. doi: 10.1093/jac/dku323. [DOI] [PubMed] [Google Scholar]
- 26.Diep J, Jacobs DM, Sharma R, Covelli J, Bowers DR, Russo TA, Rao GG. 21 November 2016. Polymyxin B in combination with rifampin and meropenem against polymyxin B-resistant KPC-producing Klebsiella pneumoniae. Antimicrob Agents Chemother doi: 10.1128/aac.02121-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Abboud CS, Monteiro J, Stryjewski ME, Zandonadi EC, Barbosa V, Dantas D, Sousa EE, Fonseca MJ, Jacobs DM, Pignatari AC, Kiffer C, Rao GG. 2016. Post-surgical mediastinitis due to carbapenem-resistant Enterobacteriaceae: clinical, epidemiological and survival characteristics. Int J Antimicrob Agents 47:386–390. doi: 10.1016/j.ijantimicag.2016.02.015. [DOI] [PubMed] [Google Scholar]
- 28.CLSI. 2016. Performance standards for antimicrobial susceptibility testing, 26th ed CLSI supplement M100S. Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- 29.Monteiro J, Widen RH, Pignatari AC, Kubasek C, Silbert S. 2012. Rapid detection of carbapenemase genes by multiplex real-time PCR. J Antimicrob Chemother 67:906–909. doi: 10.1093/jac/dkr563. [DOI] [PubMed] [Google Scholar]
- 30.Aminov RI, Chee-Sanford JC, Garrigues N, Teferedegne B, Krapac IJ, White BA, Mackie RI. 2002. Development, validation, and application of PCR primers for detection of tetracycline efflux genes of Gram-negative bacteria. Appl Environ Microbiol 68:1786–1793. doi: 10.1128/AEM.68.4.1786-1793.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Agwuh KN, MacGowan A. 2006. Pharmacokinetics and pharmacodynamics of the tetracyclines including glycylcyclines. J Antimicrob Chemother 58:256–265. doi: 10.1093/jac/dkl224. [DOI] [PubMed] [Google Scholar]
- 32.Saivin S, Houin G. 1988. Clinical pharmacokinetics of doxycycline and minocycline. Clin Pharmacokinet 15:355–366. doi: 10.2165/00003088-198815060-00001. [DOI] [PubMed] [Google Scholar]
- 33.Bishburg E, Bishburg K. 2009. Minocycline–an old drug for a new century: emphasis on methicillin-resistant Staphylococcus aureus (MRSA) and Acinetobacter baumannii. Int J Antimicrob Agents 34:395–401. doi: 10.1016/j.ijantimicag.2009.06.021. [DOI] [PubMed] [Google Scholar]
- 34.Sandri AM, Landersdorfer CB, Jacob J, Boniatti MM, Dalarosa MG, Falci DR, Behle TF, Bordinhão RC, Wang J, Forrest A, Nation RL, Li J, Zavascki AP. 2013. Population pharmacokinetics of intravenous polymyxin B in critically ill patients: implications for selection of dosage regimens. Clin Infect Dis 57:524–531. doi: 10.1093/cid/cit334. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.