Abstract
The protective effect and mechanism of Ginsenoside Rg1 on aging mouse pancreas damaged by D-galactose (D-gal)-induced was studied. Two-month-old male C57BL/6J mice were randomly divided into three groups of 10 mice per group. The D-gal group of mice received hypodermic injection of D-gal (120 mg/kg/day) for 42 days; the Rg1+D-gal group of mice receiving D-gal + intraperitoneal injection Rg1 (40 mg/kg/day) for 27 days from the 16th day of D-gal replication; and the naïve group that constituted the normal control mice receiving the same dose of saline instead of the drug. The related indicators were tested on the second day after modeling and administration. Fasting blood glucose (FBG), oral glucose tolerance test (OGTT) and fasting insulin level were measured by taking peripheral blood. Samples of pancreas were weighed and visceral index was calculated. Paraffin sections were prepared. H&E staining sections were produced to observe pancreatic tissue morphology. Immunohistochemical staining was used to observe advanced glycation end products (AGEs) and integral optical density (IOD) of stained positive tissue in pancreas. Ultrathin slices were used to observe ultrastructural change of pancreatic tissue. Frozen sections were prepared to test the relative optical density of positive cells that were stained by senescence-associated β-galactosidase (SA-β-gal) in pancreatic tissue. Superoxide dismutase (SOD), malonaldehyde (MDA) and total antioxidant capacity (T-AOC) were detected by preparing pancreas tissue homogenates. Compared with the control group, Rg1+D-gal mice had significantly decreased pancreatic wet weight and visceral index and significantly lower FBG; OGTT for 30 and 120 min. There was no significant difference of the blood sugar level between the groups. The area under the curve and the number and size of the nucleated cells within islet were markedly reduced. In addition, SA-β-gal-positive particles in pancreas tissue intracytoplasmic cells significantly decreased and relative optical density also reduced. The IOD of AGEs in pancreas tissue and MDA content decreased. SOD and T-AOC activity significantly increased. Ginsenoside Rg1 can be effective antagonistic structure and function of the pancreas injury induced by D-gal. The mechanism may be associated with reducing oxidative damage.
Keywords: Ginsenoside Rg1, D-galactose, pancreas aging, protective effect
Introduction
Pancreas is a digestive gland in mammals, playing an important role in digestion and absorption of food and constant blood sugar levels by secreting pancreatic juice and hormone. With increased age, structure and function of the pancreas gradually decline, although the underlying mechanism remains to be determined. The theory of traditional Chinese Medicine (TCM) holds that the bodys aging is closely related to the lack of vital Qi, through toning Qi can delay human aging; it has a protective effect on tissues morphology and function of human organs (1,2).
Ginsenoside Rg1 is an important medicine used in TCM clinically, and it can strengthen healthy Qi to eliminate pathogens, repairing damage and prolong life. The present study showed that Ginsenoside Rg1 is an important anti-aging constituent of ginseng, which can delay stem cell aging and antagonism in multiple organ injury caused by the attenuation agent (3,4).
The present study used D-gal to replicate the aging model mice, aiming to examine the protective effects of Ginsenoside Rg1 on pancreas injury by an antagonism attenuation agent and its mechanism of modern biology.
Materials and methods
Animals
Two-month male C57BL/6J mice (19–21 g) were supplied by the Medical and Laboratory Animal Center of Chongqing (qualified no. SCXK yu. 2012-0001). The animals were housed in conditions of controlled natural lighting and temperature (20–25°C) and received a standard free access to water and food. This study was approved by the Animal Ethics Committee of Chongqing Medical University Animal Center.
Reagents
Ginsenoside Rg1 (purity 98.6%) was obtained from Jilin Hongjiu Biological Technology Co., Ltd. (Jilin, China); D-galactose (D-gal) (purity ≥99%) was purchased from Shanghai Shengong Biological Engineering Co., Ltd. (Shanghai, China). Glucometer (One Touch UItra) was supplied by Johnson & Johnson Medical Ltd. (Shanghai, China). Blood sugar test paper (steady house type) was obtained from LifeScan Europe (Zug, Switzerland). The advanced glycation end products (AGEs) test kit was supplied by Abcam Trading (Shanghai) Co., Ltd. (Cambridge, UK). DBA staining reagent was supplied by Zhongshan Golden Bridge Biotechnology Co., Ltd. (Beijing, China); the BAC protein concentration test kit, the superoxide dismutase (SOD), the malonaldehyde (MDA), the total antioxidant capacity (T-AOC) and senescence-associated β-galactosidase (SA-β-gal) kit were supplied by Beyotime Institute of Biotechnology (Shanghai, China).
Replication of the mouse D-gal aging model and group administration
C57BL/6J mice were randomly divided into three groups, of 10 animals each. The mice of D-gal model group received hypodermic injection of D-gal (120 mg/kg/day) (5) for 6 weeks; Rg1+D-gal groups were injected with D-gal, time and dose with D-gal model group, intraperitoneal injection with Rg1 (40 mg/kg/day) for 27 days from the 16th day of D-gal replication; the same dose of saline was administered into the naive control mice at the same time. Related indicators were measured on the second day after modeling and administration.
General observation
In the process of modeling, each group of mice was observed for mental state, coat color, activity and water intake and the weight was measured daily.
Determination of fasting blood glucose (FBG) and oral glucose tolerance test (OGTT)
Mice were fasted for 12–14 h with free access to water. Tail venous blood samples were collected for measurement of FBG the following morning. Thereafter, their stomachs were filled with 20% glucose solution, and the blood glucose levels were measured at 30, 60, 90 and 120 min after lavage. The OGTT curve was drawn and the area under the curve (AUC) calculated.
Observation of morphology of pancreatic tissue structure
The mice were sacrificed by cervical dislocation and the pancreas was collected. The pancreas index was calculated as: pancreas index = pancreas wet weight (mg)/weight (g). The pancreatic tissues were fixed in 5% paraformaldehyde solution for 2 days, embedded in paraffin and sectioned. Serial section of one slice at interval of 10 sections was collected for H&E staining. The morphological change of pancreatic tissue was observed under an Olympus BX43 microscope (Olympus Optical Co., Ltd., Tokyo, Japan). Randomly selected islet data of 5 views and the number of nucleated cells in each islet under 10 times magnification. The area of each islet, cell number within islet and cell areas within a single islet were counted using Image-Pro Plus 6.0 (IPP) software. Frozen sections were prepared to stain according to the kit instructions of SA-β-gal and observe the relative optical density of stained positive cells in pancreatic tissue. Fresh pancreatic tail tissues were taken and cut into small sections and fixed in glutaraldehyde for 4 h and then washed with phosphate-buffered saline 3 times, then fixed for 1 h with 1% osmium tetroxide. Using gradient elution of ethanol and embedded with Epon 812 to prepare the ultra-thin sections was used. Samples were electronically double stained by uranium acetate and lead nitrate before observation under H-600 transmission electron microscope and the taking of images.
Measurement of pancreas oxidation and antioxidant index
The pancreas was extracted for creating 0.1 g of pancreas tissue homogenate, the supernatant was collected after centrifugation as 2,500 × g for 10 min. The supernatant protein concentration was detected using BCA Protein Assay kit for each group. MDA content, SOD and T-AOC were measured following the kit manufacturers instructions.
AGEs were determined
Dewaxing the paraffin section, 3% catalase was added to remove endogenous enzyme and the tissue was sealed with 5% goat serum. The first and second antibodies were added, following by the developing DAB developing agent and hematoxylin for analysis of pancreatic tissue AGEs by immumohistochemical staining. The distribution of AGE-stained positive cell were observed at ×20 magnification, the integral optical density (IOD) of positive cells stained for AGEs. Each section was measured using cell image analysis system IPP.
Statistical analysis
The data were processed by using statistical software SPSS 16.0 (SPSS, Inc., Chicago, IL, USA) and expressed as mean ± standard deviation. Comparison between groups was done using one-way ANOVA test followed by post hoc test (LSD). P<0.05 was considered to indicate a statistically significant difference.
Results
General condition of mice
After the D-gal model mice were dosed continuously with D-gal for 6 weeks, the coat color gradually withered, and had dim luster, and the hair was falling off. Their spirits were down, skin elasticity was reduced, lethargic, food intake significantly reduced and body mass increased slowly, presented the obvious signs of aging, while the control group and Rg1+D-gal group showed no obvious related signs.
Effect of Ginsenoside Rg1 on aging mouse pancreas weight and viscera index
Results showed that pancreatic wet weight of D-gal aging model group increased obviously; pancreatic wet weight of Rg1+D-gal group were not statistically significant compared with normal control group (Table I).
Table I.
Effect of Ginsenoside Rg1 on pancreas wet weight and index of D-gal model in mice (mean ± standard deviation, n=10).
| Groups | No. | Pancreas wet weight (g) | Body weight (g) | Pancreas index |
|---|---|---|---|---|
| Normal control | 10 | 0.2367±0.0306b | 25.7900±1.1692 | 9.1561±0.7960b |
| D-gal model | 10 | 0.4567±0.0058 | 28.0833±1.2907 | 16.2782±0.5578 |
| Rg1+D-gal model | 10 | 0.2133±0.0058b | 24.3767±0.1750a | 8.7516±0.2435b |
P<0.05
P<0.01 compared with the D-gal model group. D-gal, D-galactose.
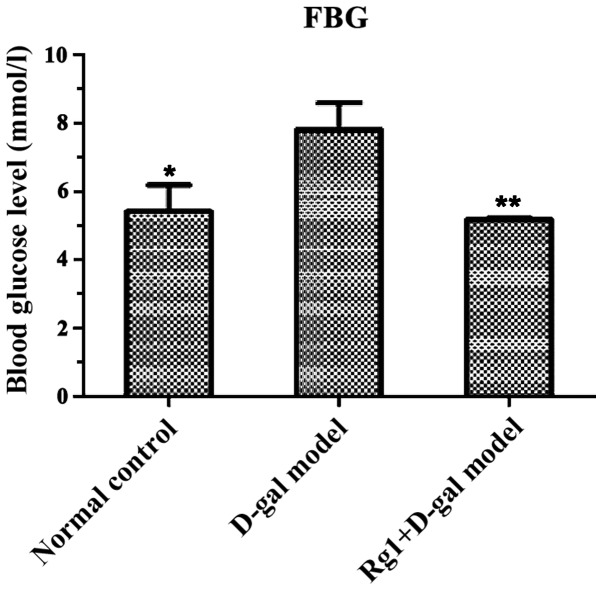
Effect of Rg1 on FBG in D-gal-induced mice
The FBG in D-gal aging model was significantly increased (C57/6J mouse blood sugar normal is 7.5 mmol/l), the FBG were at normal level in Rg1+D-gal group and the normal control group (P<0.01) (Fig. 1).
Figure 1.
Effect of Rg1 on FBG in D-gal-induced mice. *P<0.05, **P<0.01 compared with the D-gal model group. D-gal, D-galactose; FBG, fasting blood glucose.
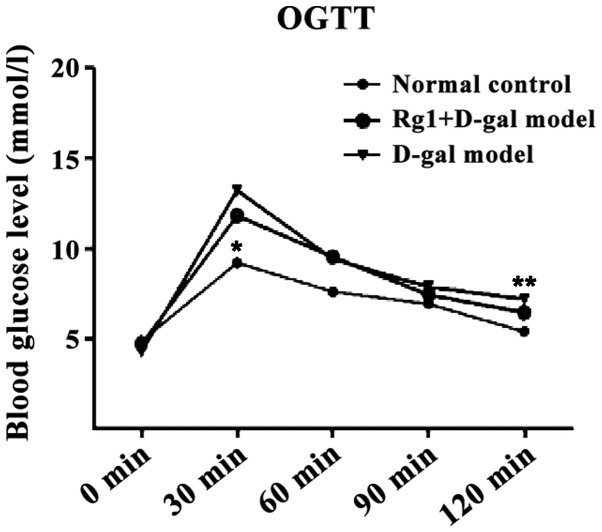
Effect of Rg1 on OGTT in mice
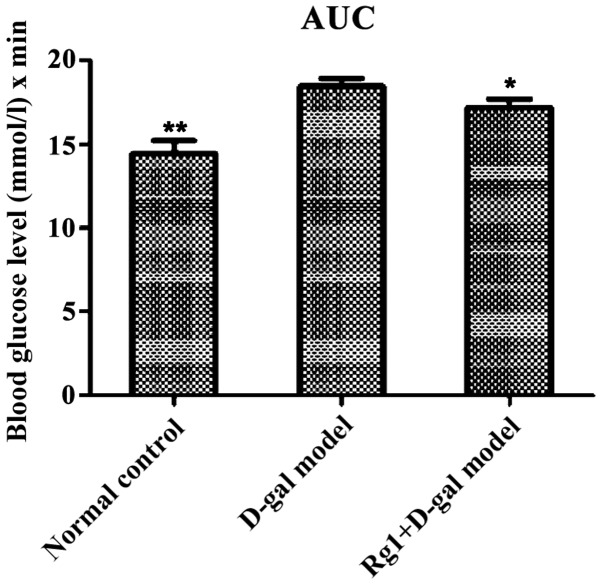
The blood glucose of each group reached the peak in 30 min and thereafter the level of blood glucose in all groups showed a downward trend. D-gal aging model group was still at the maximum during this process (Fig. 2). The AUC calculated were all less in the normal control group (P<0.01) and Rg1+D-gal group (P<0.05)than in the D-gal aging model group (Fig. 3).
Figure 2.
OGTT test in mouse-blood glucose level curve. *P<0.05, **P<0.01 compared with the D-gal model group. D-gal, D-galactose; OGTT, oral glucose tolerance test.
Figure 3.
OGTT test in mice showing the area under the blood glucose level curve. *P<0.05, **P<0.01 compared with the D-gal model group. D-gal, D-galactose, AUC, area under the curve.
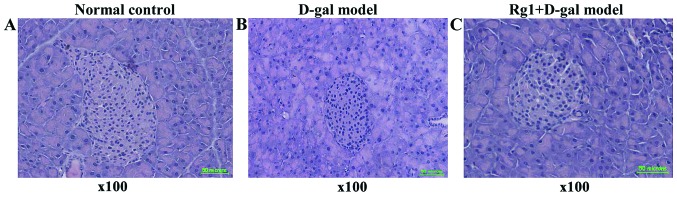
Rg1 on D-gal aging model group effect of pancreatic tissue
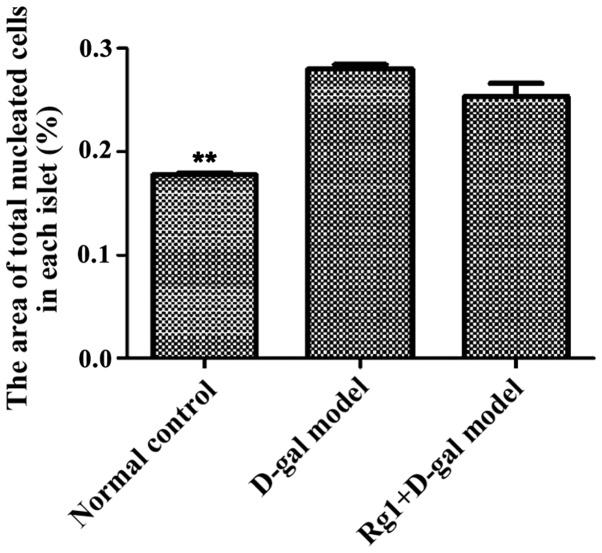
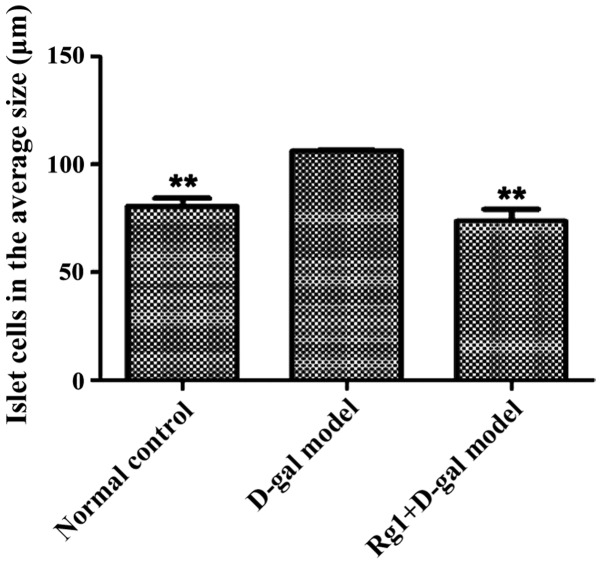
Pancreas islet morphological structures were completed in three groups (Fig. 4). For D-gal model group, the islet cell volume increased; the results showed that the areas of each cell in the islets were significantly increased compared with that of the normal control and the Rg1+D-gal model (Fig. 5); the percentage of total nucleated cells was increased in D-gal model groups than that in normal control (Fig. 6).
Figure 4.
Effect of Rg1 on the pancreas structure of D-gal aging model mice. (A) Normal group; (B) D-gal group; (C) Rg1+D-gal group (H&E stain; magnification, ×100). D-gal, D-galactose.
Figure 5.
The percentage of nucleated cell area in islets from the islet area **P<0.01 compared with the D-gal model group. D-gal, D-galactose.
Figure 6.
Average area of single nucleated cells in islets. **P<0.01 compared with the D-gal model group. D-gal, D-galactose.
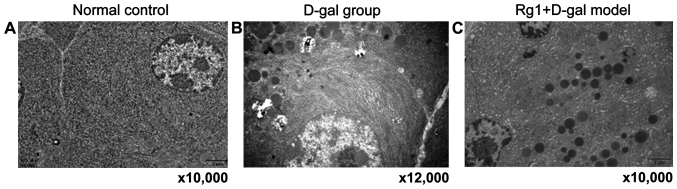
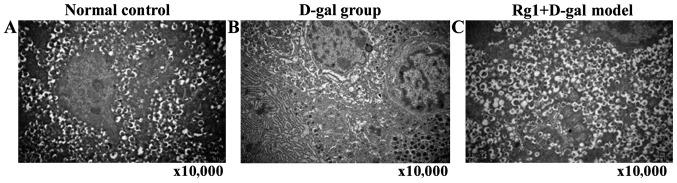
Rg1 on D-gal model group of ultrastructure on the pancreas cells
Endocrine (islets) and exocrine pancreas cells were observed by electron microscopy. D-gal aging model group of external secretion cell ultrastructure showed: Swelling mitochondria, vesicles and medullary structure formation; endoplasmic reticulum and expanded Golgi body; individual cell nuclei appeared with heterochromatin cohesion and perinuclear space broadening. In Rg1+D-gal model group, mitochondria swelled slightly, crista expanded; endoplasmic reticulum and Golgi body expansion reduced obviously. The endocrine β-cell morphology and number of secretory granules in each group had no significant abnormality with normal control, aging characteristic were not obvious (Figs. 7 and 8).
Figure 7.
Rg1 on D-gal model group of ultrastructure of the pancreas exocrine portion. (A) Normal group (magnification, ×10,000); (B) D-gal group (magnification, ×12,000); (C) Rg1+D-gal group. (magnification, ×10,000). D-gal, D-galactose.
Figure 8.
Rg1 on D-gal model group effect of ultrastructure of endocrine portion of pancreas β-cell. (A) Normal group; (B) D-gal group; (C) Rg1+D-gal group (magnification, ×10,000). D-gal, D-galactose.
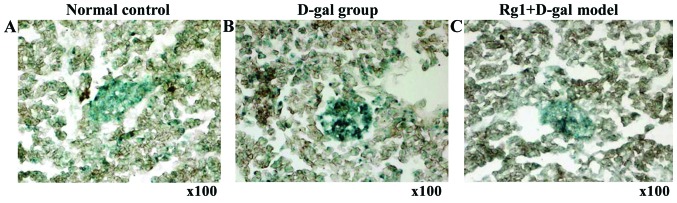
Rg1 on D-gal model group of pancreatic tissue β-gal staining
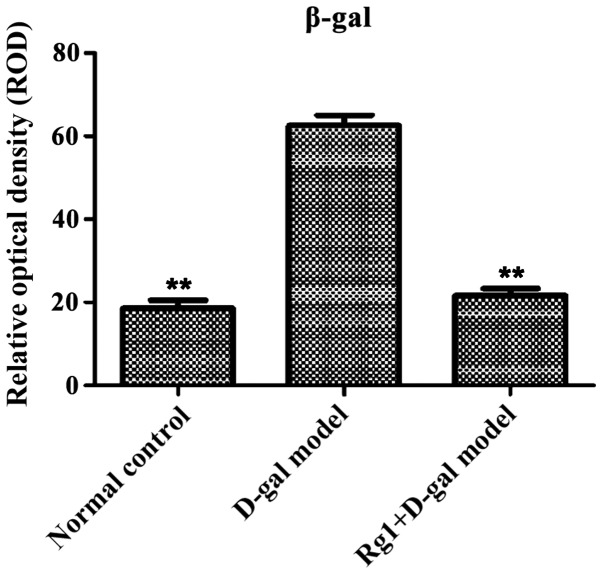
β-galactose glucoside enzyme staining showed aging cells in the cytoplasm with blue particles (Fig. 9). Compared with normal control and Rg1+D-gal model group, pancreas tissue ROD was increased (P<0.01) in D-gal aging model group (Fig. 10).
Figure 9.
Effect of Rg1 on the pancreatic cell senescence of D-gal model mice. (A) Normal group; (B) D-gal group; (C) Rg1+D-gal group (SA-β-gal stain; magnification, ×100). D-gal, D-galactose; SA-β-gal, senescence-associated β-galactosidase.
Figure 10.
Relative optical density of β-galactosidase (β-gal) staining. **P<0.01 compared with the D-gal model group. D-gal, D-galactose; β-gal, β-galactosidase.
Rg1 on D-gal model group mouse pancreas tissue oxidation and oxidative damage resistance
SOD and T-AOC were obviously decreased and MDA was increased markedly in the D-gal aging model group. In the normal control and Rg1+D-gal model group, the quantity of expression of SOD and T-AOC were higher than D-gal aging model group, and the quantity of expression of MDA was significantly decreased (Table II).
Table II.
Effect of Rg1 on the level of SOD, MDA and T-AOC in the pancreas of D-gal model mice (mean ± SD, n=10).
| Group | No. | SOD (U/mg prot) | MDA (nmol/mg) | T-AOC (U/ml) |
|---|---|---|---|---|
| Normal control | 10 | 14.669±0.365b | 3.498±0.470b | 0.266±0.011b |
| D-gal model | 10 | 11.848±0.050 | 7.421±0.293 | 0.117±0.018 |
| Rg1+D-gal model | 10 | 13.751±0.417a | 4.903±0.138b | 0.194±0.015b |
P<0.05
P<0.01 compared with the D-gal model group. D-gal, D-galactose; SOD, superoxide dismutase; MDA, malonaldehyde; T-AOC, total antioxidant capacity; SD, standard deviation.
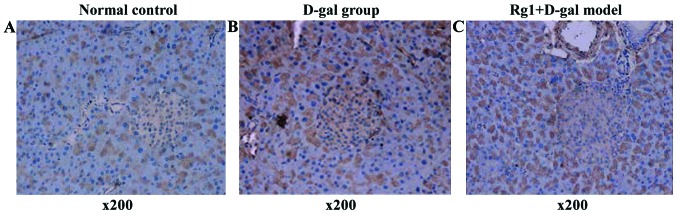
Rg1 on D-gal model group of pancreatic AGEs
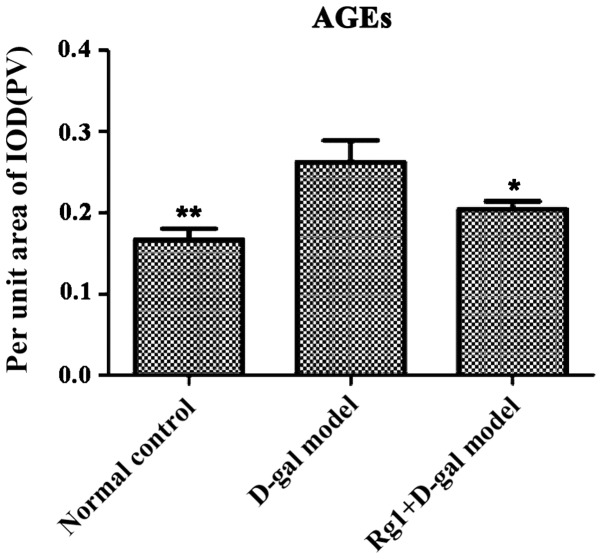
Brown areas represent AGE accumulated positive region. D-gal model group was visible in the cytoplasm of acinar cells and islet positive regional expression increased (Fig. 11) and IOD of pancreas AGEs was obviously higher, with clear difference compared with normal control (P<0.01) and Rg1+D-gal group (P<0.05) (Fig. 12).
Figure 11.
Effect of Rg1 on the AGEs of pancreas of D-gal model mice. AGEs, advanced glycation end products; (A) normal group; (B) D-gal group; (C) Rg1+D-gal group (AGEs immunohistochemical stain; magnification, ×200). D-gal, D-galactose.
Figure 12.
Effect of Rg1 on AGEs in pancreas tissue induced by D-gal in mice. *P<0.05, **P<0.01 compared with the D-gal model group. AGEs, advanced glycation end products; IOD, integral optical density.
Discussion
Pancreas is an important digestive gland to mammalian, particularly in the human body. Pancreas secretes juice that contains a variety of digestive enzymes. Pancreatic juice plays an important role in digestion and absorption of food as well as in the regulation of the constant levels of blood sugar in the body through the secretion of insulin and glucagon and other hormones. Indeed, pancreas is susceptible to the disease factors damage organs, how to prevent and treat injury of the pancreas is worth highly regarding. D-gal is now recognized as an inducer of aging reagents that can be recapitulated in animal models of aging model (6,7). The mechanism by which it induces damage or aging may be related to the attacks of the mitochondrial inner membrane lipids, protein and mitochondrial RNA that may affect the function of mitochondria (8). The aging model of mice made with D-gal have similarities with natural aging mice in the level of oxidative stress-free radical injury, non-enzymatic glycation. It may damage a number of organs including the pancreas (9,10).
The determination of organ weight and viscera indexes and histopathological observation can reflect the change of organ structure to a certain degree. The results of the study showed that the pancreas swelling was significant, wet weight and their visceral index markedly increased in D-gal aging model group, although pancreatic parenchyma cells of obvious damage had not seen in the tissue slice. However, the tissue edema was obvious, inflammatory cell infiltration, showing islet compensatory hypertrophy on the morphology, number of islet cells and unit area of islet cells were increased. Ultrastructure showed that organelles of exocrine portion swelling degeneration and there was lipofuscin deposition. Morphological structure and secretory granule of islet cells especially the β-cell, did not change obviously. These results indicated that the pancreas structure of aging model mice made with D-gal was damaged. Ginsenoside Rg1 was injected to D-gal aging model mice, but there was no evident difference compared with normal control group in pancreatic edema, weight, organ index or histopathology. The curative effects of Tiped Ginsenoside Rg1 suggest antagonism effects on islet morphology structure in aging mice damaged by D-gal.
Blood glucose level is the index to evaluate body glucose metabolism and endurance ability, it is also reflecting pancreatic function, especially the important index of islet function. We observed that the value of D-gal model group FBG exceed C57BL/6J mice maximum normal blood glucose levels, in a state of impaired fasting glucose (IFG). OGTT is an important indicator to determine the bodys glucose tolerance damage degree. The blood glucose values at 30 min can reflect the insulin resistance and pancreatic β-cell function to some extent. Blood sugar level was not significantly different in D-gal aging model group, and 120 min blood sugar of D-gal aging model mice did not return to normal levels. D-gal may cause damage to glucose tolerance in mice. While D-gal aging model mice were injected Ginsenoside Rg1, FBG can be maintained in the normal levels. 0–120 min AUC as well as 0 and 120 min blood glucose difference value displayed that Ginsenoside Rg1 obviously effect the reducing levels of FBG and postprandial glucose of mice. The result indicated that although Ginsenoside Rg1 cannot improve D-gal damage of glucose tolerance in mice, it can, however, effectively lower the level of blood glucose in mice (9).
SA-β-gal is an important marker of cell senescence, which can reflect the cell lysosome function (11). AGEs by increasing ROS generation, damaging the antioxidant system repair and inducing the formation of their own can accelerate cellular aging process (12). Both are important indicators to evaluate cell aging. The early stage of the research found Ginsenoside Rg1 as anti-ageing active ingredient in ginseng inhibited oxidative damage effect. Our data proved that D-gal aging model group in the pancreatic tissue β-gal dyed relative optical density of positive cells increased significantly and AGE positive staining region of IOD increased, thus showing that D-gal can lead to pancreas cell aging. While aging model mice were injected Ginsenoside Rg1, the pancreas significantly reduced aging, the degree of pancreatic aging significantly reduced, suggesting that Ginsenoside Rg1 may counteract aging effects induced by D-gal on mouse pancreas (13).
Oxidative damage is the mainstream theory of cellular senescence. SOD is an important scavenging enzyme of oxygen free radicals and has the function of protecting cellular DNA, proteins and cell membranes (14). Oxidative damage produced more reactive oxygen species (ROS) and disturbed cellular redox signal (15), eventually causing cell DNA, protein, and lipid damage (16,17). MDA is used in the evaluation of biomarkers of oxidative stress being the final products of ROS induced peroxidation. Our data show the reduction of pancreatic tissue SOD, T-AOC expression and the enhancement of MDA expression. Injection of Ginsenoside Rg1 in aging model mice induced SOD, T-AOC expression was enhanced and MDA expression reduced, hinting that Ginsenoside Rg1 can effectively raise the content of antioxidant enzymes in the pancreas, scavenging histiocytic free radicals improving its oxidative stress.
In conclusion, Ginsenosides Rg1 is an effective antagonist of mouse pancreas damage induced with D-gal; reducing the FBG level. The mechanism may be through two aspects of anti-aging and improve blood sugar levels. A tentative inference on this result is that the mechanism may be related to reduced oxidative damage.
Acknowledgements
The authors are grateful to grants from the National Natural Science Foundation of China (no. 30973818).
References
- 1.Li XT, Wei HH, Zhang HL, Sun HB, Su YR. Mitochondrial reactive oxygen species scavenging investigations of Astragalus membranaceus/Codonopsispilosula Qi-invigorating herbal tea. J Dalian Nationalities University. 2015;5:453–457. [Google Scholar]
- 2.Enhu Zhang, Qi Zhang, Rui HU. Review on anti-oxidant effect of Chinese Tonic-Qi herbs. Glob Tradit Chin Med. 2011;6:490–493. [Google Scholar]
- 3.Cheng Y, Shen LH, Zhang JT. Anti-amnestic and anti-aging effects of ginsenoside Rg1 and Rb1 and its mechanism of action. Acta Pharmacol Sin. 2005;26:143–149. doi: 10.1111/j.1745-7254.2005.00034.x. [DOI] [PubMed] [Google Scholar]
- 4.Fan Y, Xia J, Jia D, Zhang M, Zhang Y, Huang G, Wang Y. Mechanism of ginsenoside Rg1 renal protection in a mouse model of d-galactose-induced subacute damage. Pharm Biol. 2016;54:1815–1821. doi: 10.3109/13880209.2015.1129543. [DOI] [PubMed] [Google Scholar]
- 5.Li CP, Zhang MS, Liu J, Geng S, Li J, Zhu JH, Zhang YY, Jia YY, Wang L, Wang SH, et al. Research of anti-aging mechanism of ginsenoside Rg1 on brain. Zhongguo Zhong Yao Za Zhi. 2014;39:4442–4447. (In Chinese) [PubMed] [Google Scholar]
- 6.Ho SC, Liu JH, Wu RY. Establishment of the mimetic aging effect in mice caused by D-galactose. Biogerontology. 2003;4:15. doi: 10.1023/A:1022417102206. [DOI] [PubMed] [Google Scholar]
- 7.Yoon KH, Ko SH, Cho JH, Lee JM, Ahn YB, Song KH, Yoo SJ, Kang MI, Cha BY, Lee KW, et al. Selective beta-cell loss and alpha-cell expansion in patients with type 2 diabetes mellitus in Korea. J Clin Endocrinol Metab. 2003;88:2300–2308. doi: 10.1210/jc.2002-020735. [DOI] [PubMed] [Google Scholar]
- 8.St-Pierre J, Buckingham JA, Roebuck SJ, Brand MD. Topology of superoxide production from different sites in the mitochondrial electron transport chain. J Biol Chem. 2002;277:44784–44790. doi: 10.1074/jbc.M207217200. [DOI] [PubMed] [Google Scholar]
- 9.Song X, Bao M, Li D, Li YM. Advanced glycation in D-galactose induced mouse aging model. Mech Ageing Dev. 1999;108:239–251. doi: 10.1016/S0047-6374(99)00022-6. [DOI] [PubMed] [Google Scholar]
- 10.Brownlow BS, Petro A, Feinglos MN, Surwit RS. The role of motor activity in diet-induced obesity in C57BL/6J mice. Physiol Behav. 1996;60:37–41. doi: 10.1016/0031-9384(95)02210-4. [DOI] [PubMed] [Google Scholar]
- 11.Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, Medrano EE, Linskens M, Rubelj I, Pereira-Smith O. A biomarker that identifies senescent human cells in culture and in aging skin in vivo; Proc Natl Acad Sci USA; 1995; pp. 9363–9367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nowotny K, Jung T, Höhn A, Weber D, Grune T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules. 2015;5:194–222. doi: 10.3390/biom5010194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Thorpe SR, Baynes JW. Maillard reaction products in tissue proteins: New products and new perspectives. Amino Acids. 2003;25:275–281. doi: 10.1007/s00726-003-0017-9. [DOI] [PubMed] [Google Scholar]
- 14.Warner HR. Superoxide dismutase, aging, and degenerative disease. Free Radic Biol Med. 1994;17:249–258. doi: 10.1016/0891-5849(94)90080-9. [DOI] [PubMed] [Google Scholar]
- 15.Sies H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015;4:180–183. doi: 10.1016/j.redox.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Das SK, Vasudevan DM. Alcohol-induced oxidative stress. Life Sci. 2007;81:177–187. doi: 10.1016/j.lfs.2007.05.005. [DOI] [PubMed] [Google Scholar]
- 17.Ashok BT, Ahmad J, Ali R. Immunochemical detection of oxidative DNA damage in cancer and aging using anti-reactive oxygen species modified DNA monoclonal antibody. Int J Biochem Cell Biol. 1998;30:1367–1377. doi: 10.1016/S1357-2725(98)00100-9. [DOI] [PubMed] [Google Scholar]