Abstract
Surfactants, such as triton X-100 (Tx-100), cetylpyridinium chloride (CPC), and sodium dodecyl sulfate (SDS) are known to be toxic to Artemia Franciscana (Artemia) – an organism, frequently used to monitor the health of the aquatic environment. The phospho-metabolite profile of a living organism is often indicative of imbalances that may have been caused by environmental stressors, such as surfactants. This study utilizes i n vivo 31P NMR to monitor temporal changes in the phospho-metabolite profile of Artemia caused by Tx-100, CPC, and SDS and the ability of humic acid (HA) to mitigate the toxicity of these surfactants. It was found that, while Tx-100 does not have any effect on the phospho-metabolite profile, both CPC and SDS cause a complete retardation in growth of the phosphodiester (PDE) peak in the 31P NMR spectrum, which is indicative of the inhibited cell replication. This growth inhibition was independently verified by the decreased guanosine triphosphate (GTP) concentration in the CPC and SDS-exposed Artemia. In addition, upon introduction of HA to the CPC and SDS-exposed Artemia, an increase of PDE peak over time is indicative of HA mitigating toxicity.
Keywords: environmental, NMR, Artemia, surfactants, toxicity
1. Introduction
Surfactants are amphiphilic water soluble compounds often classified as zwitterionic, nonionic, cationic, and anionic. Surfactants used in household and industrial applications are typically nonionic, cationic, or anionic (Ivanković and Hrenović 2010). Due to their heavy use, surfactants inevitably end up in the environment via run-off, waste water treatment plants, remediation treatments, and pesticide formulations (Deese et al. 2016). The potential toxicity of surfactants to aquatic organisms depends on the organism, the surfactant type, and surfactant structure. (Ivanković and Hrenović 2010). Surfactant toxicity has been a focus of many studies (Chen et al. 2014, Cserhati et al. 2002, Czarnota and Thomas 2013, Ivanković and Hrenović 2010) with cationic surfactants being found to generally be more toxic than anionic surfactants and nonionic surfactants being the least toxic (Ivanković and Hrenović 2010).
Furthermore, humic acids (HAs), which are omnipresent in the environment, have been shown to mitigate the toxicity of a range of pollutants, including surfactants (Deese et al. 2016, Koopal et al. 2004). HAs are complex heterogeneous organic molecular assemblies formed by the degradation of organic matter. They are amphiphilic and are made up of a variety of functional groups, mainly carbohydrates, aromatics, and lipids. HAs structure and functionality varies depending on their biogeochemical origin. HA-pollutant interactions can be caused by electrostatic or hydrophobic/hydrophilic interactions and by chemical binding (Stevenson 1994). These interactions are dependent on both the type of the HA and the pollutant. Previous Artemia Franciscana (Artemia) hatching assays have shown that surfactant toxicity can be mitigated by HAs and that mitigation is based on electrostatic interactions, π-π interactions, and the amphiphilic functionality of the HA (Deese et al. 2016).
Artemia, commonly known as brine shrimp, are aquatic crustaceans that are often used in toxicological studies. Pollutant toxicity to Artemia has been studied for a range of toxicants, including (but not limited to) surfactants (Deese et al. 2016), oil dispersants (Rodd et al. 2014), pharmaceuticals (Nunes et al. 2005), pesticides (Venkateswara Rao et al. 2007), metals (Kokkali et al. 2011), and nanoparticles (Arulvasu et al. 2014, Rajabi et al. 2015). Adverse conditions, such as a polluted environment, can cause significant changes and inhibitions of an embryo's development as well as retard growth after hatching. The common approaches utilize Artemia to monitor hatching ability, short-term mortality (≤ 48 h), and long-term mortality (> 48 h). Decreased hatching ability under toxic conditions suggests either i) death of the embryos or ii) a delay of the processes required to hatch. Mortality assays measure the response of Artemia to a toxic condition after they have hatched. Although these methods can provide information on the toxicity of pollutants, they do not provide any mechanistic insight on the causes of toxicity.
Because of the limited information obtained by Artemia hatching assays and the extensive sample preparation required for metabolite extraction, an in vivo method for analyzing embryo development under a variety of conditions is desired. Nuclear magnetic resonance spectroscopy (NMR) is an almost ideal technique for in vivo experiments because of its noninvasive nature. Phosphorous-31 (31P) NMR is of particular interest because 31P is a highly receptive and biologically selective nucleus. Notably, important metabolites containing phosphorus include i) adenosine triphosphate (ATP), ii) adenosine diphosphate (ADP), iii) inorganic phosphate (Pi), iv) sugar phosphates, such as glucose-1-phosphate and nicotinamide adenine dinucleotide phosphate (NADPH), v) phosphodiester bonds (PDE), and vi) phosphocreatine (PCr). The health of the organism in question can be monitored by in vivo 31P NMR by tracking the changes in concentration of these important phospho-metabolites (Busa et al. 1982, Covi et al. 2005, Viant et al. 2002).
It was previously found that triton X-100 (Tx-100) causes mortality after hatching, cetylpyridinium chloride (CPC) and sodium dodecyl sulfate (SDS) inhibit hatching, and HAs can mitigate these toxic effects (Deese et al. 2016); however, questions remained in regards to the toxicity mechanism of CPC and SDS and whether toxicity could be measured in real time. In this study, we utilized 31P NMR to address the following questions: 1) can surfactants measurably change the phospho-metabolite profile of the Artemia embryos, 2) can these changes be measured in real time, 3) are the toxicity mechanisms based on membrane disruption or a growth inhibition, and 4) can the toxicity mitigation by HA be measured by 31P NMR?
2. Materials and Methods
The surfactants Tx-100, CPC and SDS, as well as the sodium chloride, sodium hydrogen carbonate, and guanosine 5′-triphosphate sodium standard, the perchloric acid, the phosphoric acid, HPLC-grade methanol and the deuterium oxide were purchased from Sigma Aldrich (Piscataway, NJ, USA). Sterile 18 MΩ deionized water was sourced from a US filter water purification system. Artemia Franciscana were purchased from Brine Shrimp Direct (Ogdon, UT, USA). The Leonardite humic acid reference material was purchased from the International Humic Substances Society (IHSS, MN, USA) The Perista Pump SJ 1220 peristaltic pump was manufactured by the ATTO Corporation (Tokyo, Japan). Silicone tubing for the peristaltic pump (0.64 mm inner diameter, 1.27 mm outer diameter, 15.3 m length and 1.47 mm inner diameter, 1.97 mm outer diameter, 15.3 m length) was purchased from Fisher Scientific (Pittsburg, PA, USA).
2.1 Experimental Design
In addition to Artemia Franciscana being a well-studied model organism for toxicity studies, it was chosen for the in vivo 31P NMR studies because its phospho-metabolite profile is readily monitored by 31P NMR, its preparation for NMR studies is simple, and its embryogenesis has been well characterized. The ability of Artemia to go into a diapause state, i.e., suspension of development or dormancy, makes them ideal candidates for laboratory studies as they can be stored for long periods of time. Optimal conditions for Artemia involve a saline environment (20 – 40 ppt NaCl), pH 7.5 – 9, and temperatures between 25 and 30°C (Nunes et al. 2006, Warner et al. 1989, Neumeyer et al. 2015).
The surfactants chosen for the 31P NMR studies were Tx-100 (non-ionic), CPC (cationic) and SDS (anionic) as they cover the three common classes of surfactants, are extensively used in households and industries, and responses of the toxic effects of these surfactants on Artemia have been previously studied (Deese et al. 2016). Surfactant concentrations used in this work mirrors those previously established, whereby it was found that Tx-100 at 100 ppm causes 100 % Artemia mortality at 32 hours, while CPC and SDS concentrations of 5 ppm and 35 ppm, respectively, cause lower than 5% hatching success of Artemia.
Leonardite humic acid (LAHA) was chosen for this work because it shows similar trends as other HAs in surfactant toxicity reduction (Deese et al. 2016), is economically viable for the experiments discussed below (each individual in vivo 31P NMR requires between 5 and 35 mg of LAHA), and has been well characterized as well as extensively studied (Deese et al. 2015, IHSS). The chosen concentrations of LAHA provided the 1:1 ratio of LAHA to surfactant concentration by mass, as this ratio was previously shown to demonstrate maximum toxicity mitigation (Deese et al. 2016).
2.2 Surfactant and humic acid stock solution preparation
Stock solutions of the surfactants Tx-100, CPC, and SDS were prepared by dissolving 1 g of the surfactant into 100 mL of 18 MΩ water for a concentration of 10,000 ppm (1%). The stock solution of the LAHA was prepared fresh for each experiment by dissolving approximately 150 mg of LAHA in 18 MΩ water. NaOH was added as necessary to dissolve the LAHA in water and the pH was adjusted to pH 7.8 using HCl and NaOH. The solution was diluted to 250 mL using 18 MΩ water, protected from light, and stirred overnight. The pH was checked and, if necessary, adjusted after the equilibration period.
2.3 Exposure solutions
Artemia exposures took place in a 35 ppt sodium chloride (NaCl) solution at pH 7.8. The 35 ppt NaCl solution was prepared by dissolving 35 g of NaCl in 1 L of 18 MΩ water. Sodium hydrogen carbonate (NaHCO3) was added to the solution until pH 7.8 was reached. The 35 ppt NaCl solution without any pollutants or HA added was used for the control experiments. For surfactant exposure solutions, appropriate amounts of Tx-100, CPC, or SDS stock solutions were added to the 35 ppt NaCl solution for the final surfactant concentration of 100 ppm, 5 ppm, or 35 ppm, respectively. Humic acid exposure solutions were prepared by adding the appropriate amount of the LAHA stock solution to the 35 ppt NaCl solution along with any desired surfactant. The final LAHA concentration of 35 ppm was used for the LAHA control as well as the SDS solution; a 5 ppm LAHA concentration was used for the CPC solution.
2.4 Decapsulation of Artemia Franciscana embryos
Artemia were decapsulated using a bleaching method, as illustrated in Supplemental Material Figure S2 (Stottrup and McEvoy 2003). In preparation for the decapsulation, approximately 5 g of Artemia cysts were hydrated with 18 MΩ water in a 250 mL Erlenmeyer flask equipped with a bubbler for 1.5 h in an ice bath. The ice bath was used to keep the Artemia cysts below 5 °C to prevent any premature development. After complete hydration, the cysts were filtered utilizing a nylon mesh fabric, placed back into the Erlenmeyer flask, and a hypochlorite solution (pure Clorox® bleach) was added with continued aeration.
After ∼35 s, when the Artemia embryos began to turn orange (see Supplemental Material), they were immediately filtered using the nylon mesh fabric, and rinsed with copious amounts of deionized (D.I.) water. For the NMR experiments, the Artemia embryos were packed into a 10 mm NMR tube. For the perchloric acid extraction step required for the high performance liquid chromatography (HPLC) analysis, the Artemia were transferred directly after decapsulation to their exposure solutions containing 35 ppt NaCl and the appropriate surfactant concentrations.
2.5 In vivo 31P NMR
A perfusion system was designed based on previous in vivo 31P NMR studies, see Figure S3 and S4 in Supplemental Material (Covi et al. 2005, Tjeerdema et al. 1993, Viant et al. 2006). The total length of each tube used for the NMR experiments was ∼4 m. Decapsulated Artemia embryos were packed by gravity into a 10 mm NMR tube along with the pump's tubing system. The 10 mm NMR tube was filled with D.I. water and the opening of the “in” tubing of the pump system was placed at the bottom of the NMR tube. Small aliquots of the Artemia embryos (< 0.5 mL) were then added, allowing for settling between each aliquot. This multi-step gravity packing method assured that the Artemia embryos packed tightly in the NMR tube, which limited any movement once the liquid began to flow. Once the Artemia filled ∼4-5 cm of the NMR tube, glass wool was placed on top of the Artemia and gently pressed down to remove any air bubbles and increase packing efficiency, as illustrated in Supplemental Material Figure S5.
It was important not to over-fill the Artemia embryos as too many embryos would consume too much oxygen, leading to anoxia problems. The glass wool also acted as a “cap” to keep the embryos from being removed from the NMR tube via the “out” tubing, placed at the top of the glass wool “cap”. A bottom-to-top flow through the system was achieved by pumping fresh solution into the bottom of the NMR tube and then removing the solution from the top of the Artemia population. A peristaltic pump was utilized to pump in fresh, oxygen rich, 35 ppt NaCl exposure solution at pH 7.8 (adjusted with NaHCO3) to the Artemia through the tubing at a flow rate of 2 mL/min. The pump system was constantly monitored to verify that the “in” and “out” tubes were working properly throughout each 5 h long NMR experiment.
All 31P NMR experiments were performed on a Bruker AVIII HD 400 MHz NMR spectrometer at a controlled temperature of 298 K and equipped with a 10 mm broad band probe with a 31P operating frequency of 202.43 MHz. All spectra were baseline and phase corrected. The NMR spectral shift was calibrated using an 80% H3PO4/20%D2O solution, with the phosphate peak serving as the 0 ppm reference. A deuterium lock was not required. The Artemia embryo samples were interrogated in increments of 12 minutes with 3072 transients, 2048 data points, a spectral width of 82 ppm, a relaxation delay time of 0.1 s, a pulse power of –6 dB, and a pulse angle of 30°. Data were processed utilizing 25 Hz line-broadening prior to Fourier transformation. All experiments (5 h runs) were repeated in triplicate.
To determine the “control” hatching success, Artemia embryos within the spectrometer's detection window were gently removed from the NMR tube and placed into a 250 mL Erlenmeyer flask equipped with a bubbler that contained the 35 ppt NaCl hatching solution. The solution was aerated and hatching was observed after 24 h.
2.6 Extraction of phosphorylated metabolites
The phosphorylated metabolites were extracted from the decapsulated embryos by a perchloric acid extraction method, as required for HPLC analysis, following Viant et al (Viant et al. 2006). After the Artemia were exposed to 35 ppt NaCl and pollutant solutions for 1 h and 5 h (n = 6), aliquots of the Artemia embryos were removed and flash frozen with liquid nitrogen. The frozen embryo samples were lyophilized overnight to remove all water, homogenized with a mortar and pestle, and weighed. All solutions used during the extraction procedure were kept at T ≤ 5 °C in an ice bath throughout the entire experiment. The dry tissue was extracted with ice-cold 1.0 M perchloric acid, vortexed for 30 s, and then put into an ice bath for 10 min. The samples were centrifuged at 10,000 × g for 10 min. The supernatant was then removed and neutralized to pH 7.0 with ice-cold 1 M Na2CO3, kept on ice for 30 min, and then centrifuged again under the same conditions. The samples were diluted to 10 mL with 18 MΩ water and filtered with 0.45 μm polyvinylidene fluoride filters prior to HPLC analysis.
2.7 High performance liquid chromatography
All measurements were obtained utilizing an Agilent 1100 series HPLC with a Zorbax eclipse XDB-C18 column; 4.6 × 150 mm, 5 um column. The HPLC method used to detect guanosine triphosphate (GTP) from the perchloric acid extraction was developed by Veciana-Nogues et al. (Veciana-Nogues et al. 1997) with UV detection at 254 nm. A 24 min gradient was used for each sample. The mobile phase A was 0.05 M phosphate buffer and mobile phase B was HPLC-grade methanol. The flow rate was 1 mL/min. The gradient was as follows: 0-9 min 100% A, 0% B; 9-14 min 70% A, 30% B 14-24 min 100% A, 0% B. Peak assignment was confirmed with the use of standards. A calibration curve for GTP was created with 1, 5, 10, 20, and 30 ppm GTP standard and a R2 = 0.99 was obtained.
3. Results
The 31P NMR profile of Artemia obtained in this study is similar to previous Artemia studies. It is also similar to the phospholipid profile of medaka embryos (Busa et al. 1982, Covi et al. 2005, Kwast et al. 1995, Viant et al. 2006). The detectable peaks (see Figure 1A) are identified here as a phosphomonoester (PME, sugar phosphate; 3.7-4.2 ppm) peak, an inorganic phosphate (Pi; 2.5-3.5 ppm) peak, a phosphodiester (PDE, DNA backbone; 1.2-1.8 ppm) peak, a phosphocreatine (PCr; 0-1 ppm) peak, adenosine triphosphate (ATP; –19.0 - –18.6 ppm) peak, and two peaks attributed to both ATP and adenosine diphosphate (ADP; –11.8 - –9.8 ppm and – 5.5 - –4.5 ppm) that are indistinguishable from each other (further discussion on these peaks can be found in the Supplemental Material).
Figure 1.

A) Representative 31P NMR spectrum of live Artemia Franciscana. Peak identities are 1) phosphomonoesters (PME; 3.7-4.2 ppm), 2) inorganic phosphate (Pi; 2.5-3.5 ppm), 3) phosphodiesters (PDE; 1.2-1.8 ppm), 4) phosphocreatine (PCR; 0-1 ppm), 5, 6, 7, and 8) α-adenosine triphosphate (ATP)/α-adenosine diphosphate (ADP), β-ATP (–5.5 - –4.5 ppm; –11.8 - –9.8 ppm) 9) γ-ATP/β-ATP (–19.0 - –18.6 ppm) and B)31P NMR example spectrum of dead Artemia embryos (embryo's death was induced on purpose for illustrative purposes by leaving the embryos in the hypochlorite solution for too long).
Figure 1B illustrates an example of the 31P NMR profile of dead Artemia embryos
3.1 31P NMR of Artemia in 35 ppt NaCl solution – Control conditions
Representative spectra of the control can be seen in Figure 2.A. The PME, PCr, and ATP/ADP peaks showed some insignificant changes over the course of 5 h and are attributed to the constant variation in the PME, PCr, and ATP/ADP concentrations as the embryos begin to develop. The Artemia's PDE peak for the controls significantly increased after ∼150 min and continued to increase over the course of the experiment.
Figure 2.

Stacked representative 31P NMR spectra for Artemia franciscana embryos in A) 35 ppt NaCl, B)100 ppm Tx-100, C) 5 ppm CPC and D) 35 ppm SDS
3.2 Tx-100
31P NMR spectra were collected for the Artemia with 100 ppm Tx-100 in 35 ppt NaCl. As can be seen in Figure 2B, there was a slight decrease in the [ATP] (specifically peaks at approximately –10.4 ppm and –19 ppm) over the entire course of the 5 h experiment. This decrease in [ATP] is greater than that seen for the control and other surfactant conditions, as can be seen in the data presented in Figure 2 (see Supplemental Material for more detailed 31P NMR spectral representations). Although a significant and steady decrease in [ATP] is indicative of a stressed system, in the case of Tx-100, it is attributable to anoxia rather than stress by the surfactant as it was not possible to fully aerate the Tx-100 solution due to a “foaming” issue, causing the solution to overflow with bubbles. This “foaming” issue did not occur with the other surfactant solutions during aeration and thus, the Artemia did not exhibit anoxia-related problems, as illustrated in Supplemental Material Figure S6. The PCr and PME peaks exhibited no significant changes over the time course of the experiment. Similar to the control Artemia spectra, the PDE peak increased significantly over time after ∼150 min.
3.3 CPC and SDS
The signals of the ATP peaks in the CPC and SDS spectra were similar to those of the control 31P NMR spectra. Some anticipated fluctuation occurred in the [ATP] over time; however, the increase in [PDE] was significantly lower for the CPC- and SDS-exposed Artemia compared to the control and the Tx-100-exposed Artemia. These solutions were constantly aerated to eliminate, or minimize, the effects of anoxia, as illustrated in Supplemental Material Figure S7.
The in vivo 31P NMR intensities were used to calculate a PDE/Pi ratio for each time point obtained. The moving average (n = 3, interval = 2) of the PDE/Pi ratios are shown, with standard error bars in Figure 3. The PDE/Pi increased consistently over time after ∼150 min for both the control and Tx-100 solutions.
Figure 3.

31P NMR intensities of PDE/Pi of Artemia Franciscana embryos with 35 ppt NaCl, 5 ppm CPC in 35 ppt NaCl, 35 ppm SDS in 35 ppt NaCl, and 100 ppm Tx-100 in 35 ppt NaCl.
After ∼150 min, the PDE/Pi ratio was significantly lower for CPC and SDS-exposed Artemia compared to the control and Tx-100-exposed Artemia. The PDE/Pi ratio remained below 1 for the CPC and SDS-exposed Artemia. The PDE/Pi ratio easily exceeded 1.4 from the control and Tx-100-exposed Artemia, especially after 200 min.
3.4 HPLC results
HPLC with UV detection was utilized to measure the concentration of guanosine triphosphate (GTP) in the Artemia embryos exposed to CPC, SDS, and Tx-100, as well as in the control (no surfactant exposure). The concentration of GTP was measured as its decrease is indicative of embryo growth inhibition, while an increase is generally indicative of growth, maturation, and cell replication. Figure 4 shows the HPLC results obtained for the Artemia embryo extracts as the change in μmol/mg of dry Artemia tissue over 5 h. For the Artemia embryos in the control and 100 ppm Tx-100 solutions, the GTP concentration increases, while for SDS and CPC solutions, the GTP concentration decreases. The CPC and SDS-induced changes in [GTP] were significantly different (p < 0.05) compared to the control sample. The HPLC results are in agreement with the 31P NMR results in that, unlike CPC and SDS, Tx-100 does not appear to have a negative effect on cell replication.
Figure 4.

The changes in micromoles of guanosine triphosphate (GTP) per milligram of dry Artemia Franciscana tissue extracts for a 5h exposure to as measured by HPLC (*p < 0.05 versus the control)
3.5 31P NMR of Artemia with addition of LAHA
HAs have been shown to mitigate the toxic effects of surfactants to Artemia (Deese et al. 2016). Specifically, LAHA was able to interact and reduce the bioavailability of CPC, SDS, and Tx-100, resulting in changes in the hatching or mortality rates of Artemia. Since CPC and SDS induced changes in the Artemia's phospho-metabolite profile, LAHA was added to the CPC and SDS toxic solutions in order to determine if LAHA's ability to mitigate the toxicity of these surfactants could be measured by in vivo 31P NMR, resulting in an improved understanding of the toxicity mitigation mechanism.
The PDE/Pi ratios of the control, LAHA alone, CPC alone, and CPC plus LAHA were calculated from the 31P NMR data and plotted in Figure 5. These LAHA data closely mirror those for the control; this was expected as LAHA has been shown in the previous hatching assays to have no effect on the Artemia hatching ability (Deese et al. 2016). The data also show that CPC is less toxic in the presence of LAHA, as illustrated by the increased PDE/Pi ratio versus that for the CPC alone.
Figure 5.
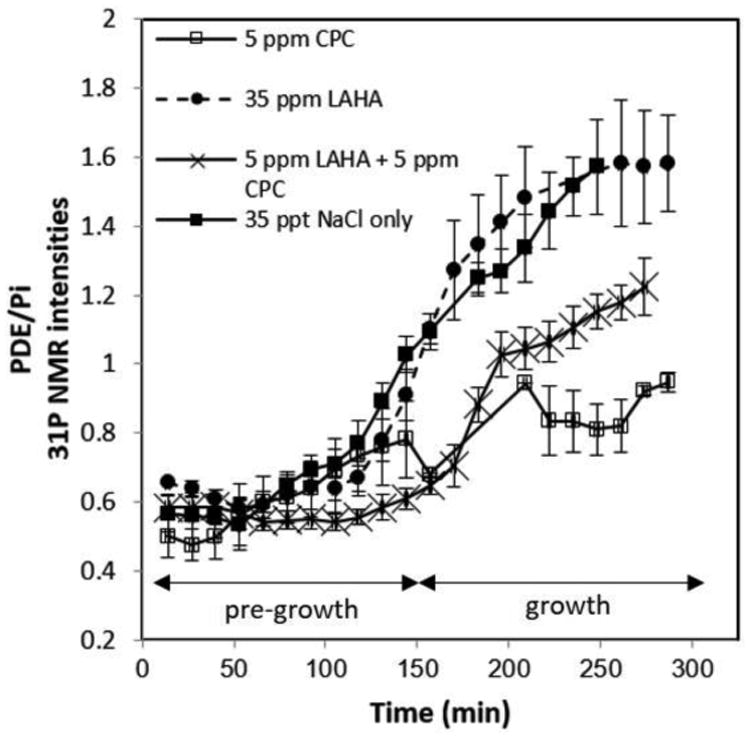
31P NMR intensities of PDE/Pi of Artemia Franciscana embryos under 35 ppt NaCl, 35 ppm LAHA in 35 ppt NaCl, 5 ppm CPC in 35 ppt NaCl, and 5 ppm LAHA and 5 ppm CPC in 35 ppt NaCl
The PDE/Pi of Artemia with CPC and LAHA was above 1 but less than 1.4, falling directly in between the highest ratio obtained for the CPC-only solutions and the lowest ratio for the control solution. Similar results were obtained with SDS conditions, as shown in Figure 6. Based on the PDE/Pi ratio, LAHA mitigates SDS toxicity better than it mitigates the toxicity of CPC. The data in Table 1 further illustrate these points and show that LAHA significantly (p < 0.05) mitigates both CPC and SDS toxicity to Artemia.
Figure 6.
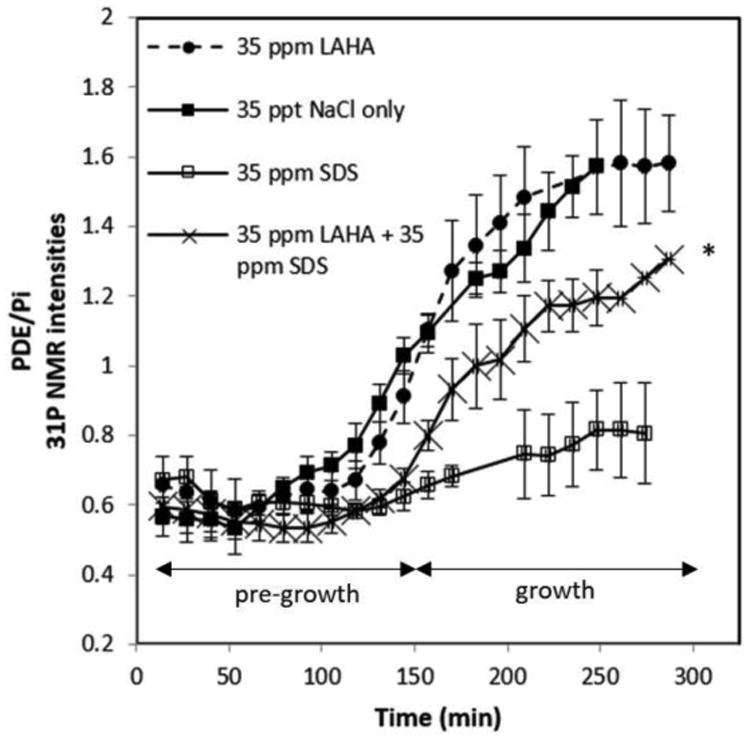
31P NMR intensities of PDE/Pi of Artemia Franciscana embryos under 35 ppt NaCl, 35 ppm LAHA in 35 ppt NaCl, 35 ppm SDS in 35 ppt NaCl, and 35 ppm LAHA and 35 ppm SDS in 35 ppt NaCl (*last three time points only repeated in duplicate).
Table 1. PDE/Pi ratios of Artemia Franciscana embryos measured by in vivo 31P NMR.
| Exposure Solution | |||||||
|---|---|---|---|---|---|---|---|
| Time (min) | 35 ppt NaCl | 5 ppm CPC | 35 ppm SDS | 100 ppm Tx100 | 35 ppm LAHA | 5 ppm LAHA + 5 ppm CPC | 35 ppm LAHA + 35 ppm SDS |
| 15 | 0.57±0.06 | 0.50±0.06 | 0.67±0.07 | 0.56± 0.03 | 0.66±0.01 | 0.58±0.04 | 0.60±0.05 |
| 150 | 1.03±0.05 | 0.78±0.11 | 0.62±0.04* | 0.66±0.02 | 0.91±0.08 | 0.61±0.03* | 0.68±0.03 |
| 200 | 1.34±0.10 | 0.90±0.04* | 0.75±0.13* | 1.24±0.09 | 1.48±0.15 | 1.04±0.06 | 1.11±0.09 |
| 250 | 1.57±0.14 | 0.81±0.07* | 0.82±0.12* | 1.41±0.05 | 1.58±0.18 | 1.15±0.05† | 1.20±0.08† |
Values represent mean ± S.E.M. of three replicates. (
p < 0.05 at the same time point where null hypothesis = control.
p < 0.05 at same time point where null hypothesis = the respective surfactant-only exposure).
4. Discussion
CPC, SDS, and Tx-100 are known to be toxic to Artemia, and HAs have the ability to mitigate the toxicity of these surfactants. This study examines whether these trends could be observed using in vivo methods, namely 31P NMR, and whether a better understanding of the toxicity mechanisms can be obtained. The major advantages of in vivo 31P NMR is that clean, simple, and directly interpretable data can be obtained in real time.
The control 31P NMR spectra indicated some minor variation in [ATP] throughout the experiment. Although changes in [ATP] may indicate stress on an organism, [ATP] has also been shown to fluctuate greatly during embryogenesis in Artemia (Warner and Finamore 1967, Zhu et al. 2009) and other organisms (Moroz and Luzhin 1976). Post-diapause development of Artemia is complex and most of the development events cause changes in energy usage and thus, changes in ATP demands by the embryo (Zhu et al. 2009). Since the degradation and synthesis of ATP is a cyclic process, the [ATP] can depend on the Artemia embryo's point in the cycle at a particular time. Thus, the small fluctuations observed in the 31P NMR ATP/ADP peaks of the control can be attributed to normal fluctuations in energy demands.
The major change observed in the phospho-metabolite profile of Artemia in the control over time is an increase in [PDE] after ∼150 min. Artemia embryos are known to stay in a diapause state until introduced to the proper hatching conditions, such as the right salinity, pH, and temperature (Stappen, 1996). Thus, the time between 0 and 150 min is likely the delay time between when the Artemia are introduced to the appropriate hatching conditions and when the embryos begin to develop. Increases of [PDE] have been shown to occur during tissue growth, maturation, and cell replication (Certaines et al. 1993) and decreases of [PDE] have been attributed to declining rates of cellular replication (Viant et al. 2006). The increase of [PDE] observed in the control spectra of the Artemia can be attributed to the significant cell replication occurring during embryonic development.
The [PDE] increase observed in the Tx-100-exposed Artemia spectra indicates that there is still cell replication occurring under Tx-100 conditions. Because Tx-100 does not affect the Artemia hatching ability (Deese et al. 2016) and since normal processes still need to occur within the cell in order to hatch, it stands to reason that there should not be any significant differences in the phospho-metabolite profile of the Artemia embryos exposed to Tx-100 versus the control. This notion is confirmed by the data in Figures 2, 3, and 4, as well as Table 1.
Because CPC and SDS only affect hatching rates and not mortality rates, it can be assumed that they either 1) disrupt the cellular membranes of the embryos and cause them to die before hatching or 2) inhibit growth of the embryos in some way. Surfactants can lyse cellular membranes and this ability has been considered to play a role in surfactant toxicity to cells (Partearroyo et al. 1990). It has also been shown that cationic surfactants cause narcosis in aquatic organisms, which is a depression in biological activity typically caused by narcotics (Roberts and Costello 2003). Anionic surfactants have been shown to have similar ability as cationic surfactants to depress biological functions by means of binding to bioactive macromolecules, such as peptides, enzymes, and DNA, causing conformational changes and dissociation (Cserháti et al. 2002).
The presence of clear and stable ATP peaks indicates that CPC and SDS do not cause initial mortality of the embryos. If CPC or SDS disrupted the cell membranes and caused embryonic death, the resulting NMR spectrum would resemble that presented in Figure 1B. As this was not observed, as evidenced by the spectra in Figure 2C and D, the second toxicity mechanism presented above is more likely.
While the Artemia's PDE peak increased under CPC and SDS conditions, it remains greatly reduced versus that for the control condition, as illustrated by the 31P NMR data presented in Figure 3. Because the increase in [PDE] is indicative of cell replication and thus growth of the system, it is suggested that CPC and SDS inhibit cell replication.
Cationic and anionic surfactants have been shown to attack different components of cells; anionic surfactants bind to peptides and DNA, while cationic surfactants attack cytoplasmic membranes (Ivanković and Hrenović 2010, Ostroumov 2006). Although these two classes of surfactants may have different mechanisms of toxicity, it has been shown that both cause a decrease in biological activity or narcosis (Cserháti et al. 2002, Roberts and Costello 2003).
To verify the in vivo 31P NMR findings in regards to the reduced [PDE], as induced by CPC and SDS, HPLC was utilized to measure [GTP] changes over time. Embryogenesis studies of Artemia have indicated that an increase in the [GTP] is a major indicator of embryo growth (Warner and Finamore 1967), and a decrease in the [GTP] is indicative of stress on the system and lower hatching rates (Warner and Clegg 2001). When the [GTP] increases, the growth in the system is assumed; however, under CPC and SDS conditions, the [GTP] significantly (p < 0.05) decreased, as shown in Figure 4, indicating an inhibition in the embryo's growth. This decrease in the [GTP] corroborates the inhibition of Artemia embryo growth as seen in the in vivo 31P NMR spectra.
In previous hatching assays, LAHA and, to a lesser extent, Florida Peak humic acid (FPHA) and Suwannee River humic acid (SRHA), were shown to have the ability to reduce the toxicity of Tx-100, CPC and SDS to Artemia (Deese et al. 2016). Although there was significant toxicity mitigation when these humic acids were present, some toxicity was still evident through slightly lower hatching rates. This phenomenon is clearly evident in the NMR data presented in Figures 5 and 6 for CPC and SDS, respectively. While the PDE/Pi ratio increases in time after 150 min, it remains lower than in the controls in the presence of the surfactant and LAHA, indicating that LAHA does not fully mitigate the toxicity of either surfactant. LAHA's ability to mitigate the toxicity of these surfactants is attributed to the interactions between LAHA and CPC or SDS which, in turn, reduce the bioavailability of these surfactants.
As alluded to before, there are a number of components within environmental waters contaminated with surfactants other than simply the surfactant and water. Two such components are metal ions as well as suspended clay and oxide particles. Overall, the roles of metal ions and clays on surfactant binding (or availability from the opposite point of view) will vary depending on the surfactant's ionic classification and can be explained by ionic and hydrophobic considerations, including the hydrophilicity of the sorbent's surface (Ishiguro and Koopal 2016). In grosso modo terms, metal ions block the cationic sorption sites within or on clays, oxides, and natural organic matter, including HAs, and hence decrease the ability of HAs to sorb cationic surfactants, while inducing aggregation and precipitation of anionic surfactants. Depending on the structure and charge of a clay, similar rules to those for metal ions apply. Similarly, for oxide particles, including density of hydroxide sites. In addition, surfactants can sorb either to the clay's surface or interstitially. Finally, clay particles can also sorb different components of the HA, chemically editing it. It has been previously shown that the aliphatic molecular components of a HA sorb to clays (Feng et al. 2005). With this knowledge and our previous study on the role of HA chemical composition on its ability to sorb surfactants (Deese et al. 2016), it can be postulated that the presence of clay particles decreases the ability of a HA to sorb nonionic surfactants.
6. Conclusions
Surfactants pose a threat to the overall health of the environment as they can be toxic to a variety of organisms; however, to fully evaluate this toxicity, deeper insight into the underlying mechanism(s) is needed and the role of other environmental constituents that are known to interact with pollutants, such as HA, must be considered.
Previous hatching assays demonstrated that CPC and SDS surfactants caused Artemia hatching inhibitions; however, the question remained unanswered as to whether this inhibition was caused by membrane disruption or inhibition of development. In this study, an in vivo 31P NMR method was utilized to measure toxicity trends of surfactants and it was able to illustrate growth inhibition of the Artemia embryos in the presence of CPC and SDS. Over the course of time, the phospho-metabolite profile of the 35 ppt NaCl control and Tx-100 conditions, but not the CPC or SDS conditions, show a significant increase in the PDE/Pi ratio for the Artemia embryos. Our results demonstrate that CPC and SDS inhibit cell replication and that in vivo 31P NMR is a powerful, yet direct, tool that provides a noninvasive measure of the growth inhibition, and thus, toxicity, in real time. Additionally, the toxicity mitigation effects as a result of adding LAHA to the CPC and SDS solutions were investigated by in vivo 31P NMR measurements of the PDE content of Artemia. A greater PDE/Pi ratio was obtained with the use of LAHA compared to that for the CPC or SDS alone. This work shows (i) the role of environmental matrix components, such as the natural organic matter and, in particular, HAs in mitigating the toxicity of pollutants, and (ii) the ability of in vivo 31P NMR spectroscopy to monitor the influence of these components.
This study also illustrates a significant advantage of in vivo 31P NMR over other methods. In particular, by offering insights into the mechanisms by which toxicity processes may take place, in vivo, 31P NMR results are more informative than those from hatching assays. For real time systems, it also provides a suitable alternative method to metabolomics, which, by utilizing 1H NMR, requires extensive multivariate analysis and metabolite extractions, extensive sample preparation and time for analysis.
Supplementary Material
Acknowledgments
This study is based on work supported by the National Science Foundation under grant CHE-0547982 and CHE-1411547 and NIEHS Superfund Research program for Louisiana State University under grant 2P42ES013648-03. Rachel D. Deese thanks the Louisiana Board of Regents for financial support afforded by their Graduate Fellowship program.
Footnotes
Disclaimer: The authors declare that there is no conflict of interest regarding the contents of this study.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Arulvasu C, Jennifer SM, Prabhu D, Chandhirasekar D. Toxicity effect of silver nanoparticles in brine shrimp artemia. Scientific World J. 2014 doi: 10.1155/2014/256919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busa WB, Crowe JH, Matson G. Intracellular ph and the metabolic status of dormant and developing artemia embryos. Arch Biochem Biophys. 1982;216(2):711–718. doi: 10.1016/0003-9861(82)90261-2. [DOI] [PubMed] [Google Scholar]
- Certaines JD, Bovee WMMJ, Podo F. Magnetic resonance spectroscopy in biology and medicine. NMR Biomed. 1993;6(3):235–235. doi: 10.1002/nbm.1940060310. [DOI] [Google Scholar]
- Chen Y, Geurts M, Sjollema SB, Kramer NI, Hermens JL, Droge ST. Acute toxicity of the cationic surfactant c12-benzalkonium in different bioassays: How test design affects bioavailability and effect concentrations. Environ Toxicol Chem. 2014;33(3):606–615. doi: 10.1002/etc.2465. [DOI] [PubMed] [Google Scholar]
- Covi JA, Treleavan WD, Hand SC. V-atpase inhibition prevents recovery from anoxia in artemia franciscana embryos: Quiescence signaling through dissipation of proton gradients. J Exp Biol. 2005;(208):2799–2808. doi: 10.1242/jeb.01681. [DOI] [PubMed] [Google Scholar]
- Cserhati T, Forgaces E, Oros G. Biological activity and environmental impact of anionic surfactants. Environ Int. 2002;28:337–348. doi: 10.1016/S0160-4120(02)00032-6. [DOI] [PubMed] [Google Scholar]
- Cserháti T, Forgács E, Oros G. Biological activity and environmental impact of anionic surfactants. Environ Int. 2002;28(5):337–348. doi: 10.1016/S0160-4120(02)00032-6. [DOI] [PubMed] [Google Scholar]
- Czarnota M, Thomas P. Cooperative Extension. The University of Georgia; 2013. Using surfactants, wetting agents, and adjuvants in the greenhouse. [Google Scholar]
- Deese RD, LeBlanc MR, Cook RL. Surfactant toxicity to artemia franciscana and the influence of humic acid and chemical composition. Envir Chem. 2016;13(3):507–516. doi: 10.1071/EN15108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng X, Simpson AJ, Simpson MJ. Chemical and mineralogical controls on humic acid sorption to clay mineral surfaces. Org Geochem. 2005;36:1553–1566. doi: 10.1016/j.orggeochem.2005.06.008. [DOI] [Google Scholar]
- IHSS. Isolation of IHSS samples. Retrieved July 4, 2016, from http://www.humicsubstances.org/isolation.html.
- Ishiguro M, Koopal LK. Surfactant sorption to soil components and soils. Adv Colloid Interface Sci. 2016;231:59–102. doi: 10.1016/j.cis.2016.01.006. [DOI] [PubMed] [Google Scholar]
- Ivanković T, Hrenović J. Surfactants in the environment. Arh Hig Rada Toksikol. 2010;61(1):95. doi: 10.2478/10004-1254-61-2010-1943. [DOI] [PubMed] [Google Scholar]
- Kokkali V, Katramados I, Newman JD. Monitoring the effect of metal ions on the mobility of artemia salina nauplii. Biosensors. 2011;1:36–45. doi: 10.3390/bios1020036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koopal LK, Goloub TP, Davis TA. Binding of ionic surfactants to purified humic acid. J Colloid Interface Sci. 2004;275:360–367. doi: 10.1016/j.jcis.2004.02.061. [DOI] [PubMed] [Google Scholar]
- Kwast KE, Shapiro JI, Rees BB, Hand SC. Oxidative phosphorylation and the realkalinization of intracellular ph during recovery from anoxia in artemia franciscana embryos. Biochim Biophys Acta. 1995;1232:5–12. doi: 10.1016/0005-2728(95)00090-9. [DOI] [Google Scholar]
- Martello LB, Tjeerdema RS, Smith WS, Kauten RJ, Crosby DG. Influence of salinity on the actions of pentachlorophenol in haliotis as measured by in vivo 31P NMR spectroscopy. Aquat Toxicol. 1998;41:229–250. [Google Scholar]
- Moroz IE, Luzhin B. Dynamics of metabolism in the embryonic and postembryonic development of the carp (cyprinus carpio l.) Vopr Ihtiol. 16:1976. 1061–1068. [Google Scholar]
- Neumeyer CH, Gerlach JL, Ruggiero KM, Covi JA. A novel model of early development in the brine shrimp, artemia franciscana, and its use in assessing the effects of environmental variables on development, emergence, and hatching. J Morphol. 2015;276(3):342–360. doi: 10.1002/jmor.20344. [DOI] [PubMed] [Google Scholar]
- Nunes B, Carvalho F, Guilhermino L. Acute toxicity of widely used pharmaceuticals in aquatic species: Gambusia holbrooki, artemia parthenogenetica and tetraselmis chuii. Ecotoxicol Environ Saf. 2005;61(3):413–419. doi: 10.1016/j.ecoenv.2004.08.010. [DOI] [PubMed] [Google Scholar]
- Nunes BS, Carvalho FD, Guilhermino LM, Stappen GV. Use of the genus artemia in ecotoxicity testing. Environ Pollut. 2006;144:453–462. doi: 10.1016/j.envpol.2005.12.037. [DOI] [PubMed] [Google Scholar]
- Ostroumov SA. Biological effects of surfactants. Boca Raton: Florida, CRC Press; 2006. [Google Scholar]
- Partearroyo MA, Ostolaza H, Goñi FM, Barberá-Guillem E. Surfactant-induced cell toxicity and cell lysis. Biochem Pharmacol. 1990;40(6):1323–1328. doi: 10.1016/0006-2952(90)90399-6. [DOI] [PubMed] [Google Scholar]
- Pincetich CA, Viant MR, Hinton DE, Tjeerdema RS. Metabolic changes in japanese medaka (oryzias latipes) during embryogenesis and hypoxia as determined by in vivo 31p nmr. Comp Biochem Physiol. 2005;140:103–113. doi: 10.1016/j.cca.2005.01.010. [DOI] [PubMed] [Google Scholar]
- Rajabi S, Ramazani A, Hamidi M, Naji T. Artemia salina as a model organism in toxicity assessment of nanoparticles. DARU. 2015;23(1):20. doi: 10.1186/s40199-015-0105-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts DW, Costello J. Qsar and mechanism of action for aquatic toxicity of cationic surfactants. QSAR Comb Sci. 2003;22(2):220–225. doi: 10.1002/qsar.200390015. [DOI] [Google Scholar]
- Rodd AL, Creighton MA, Vaslet CA, Rangel-Mendez JR, Hurt RH, Kane AB. Effects of surface-engineered nanoparticle-based dispersants for marine oil spills on the model organism artemia franciscana. Environ Sci Technol. 2014;48(11):6419–6427. doi: 10.1021/es500892m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Safety data sheet: Cetylpyridinium chloride. Sigma-Aldrich; 2015. [Google Scholar]
- Sids initial assessment profile: Sodium dodecyl sulfate, cas no: 151-35-3.
- Stappen GV. Introduction, biology and ecology of artemia. In: Lavens P, Sorgeloos P, editors. FAO Fisheries Technical Paper. Ghent, Belgium: Food and Agriculture Organization of the United Nations; 1996. p. 361. [Google Scholar]
- Stevenson FJ. Humus chemistry: Genesis, composition, reactions. New York, N.Y: John Wiley & Sons, Inc; 1994. [Google Scholar]
- Stottrup J, McEvoy L. Live feeds in marine aquaculture. Blackwell Science Ltd; 2003. [Google Scholar]
- Tjeerdema RS, Kauten RJ, Crosby DG. Interactive effects of pentachlorophenol and temperature in the abalone (haliotis rugescens) as measured by in vivo 31p-nmr spectroscopy. Aquat Toxicol. 1993;26:117–132. doi: 10.101/0166-445X(93)90009-P. [DOI] [PubMed] [Google Scholar]
- Triton surfactants: Fda status of triton surfactants. The Dow Chemical Company; 2010. [Google Scholar]
- Veciana-Nogues MT, Izquierdo-Pulido M, Vidal-Carou MC. Determination of atp related compounds in fresh and canned tuna fish by hplc. Food Chem. 1997;59(1):467–472. doi: 10.1016/S0308-8146(96)00243-9. [DOI] [Google Scholar]
- Venkateswara Rao J, Kavitha P, Jakka NM, Sridhar V, Usman PK. Toxicity of organophosphates on morphology and locomotor behavior in brine shrimp, artemia salina. Arch Environ Contam Toxicol. 2007;53(2):227–232. doi: 10.1007/s00244-006-0226-9. [DOI] [PubMed] [Google Scholar]
- Viant MR, Pincetich CA, Hinton DE, Tjeerdema RS. Toxic actions of dinoseb in medaka (oryzias latipes) embryos as determined by in vivo 31p nmr, hplc-uv and 1h nmr metabolomics. Aquat Toxicol. 2006;76:329–342. doi: 10.1016/j.aquatox.2005.10.007. [DOI] [PubMed] [Google Scholar]
- Viant MR, Pincetich CA, Walton JH, Tjeerdema RS, Hinton DE. Utilizing in vivo nuclear magnetic resonance spectroscopy to study sublethal stress in aquatic organisms. Mar Environ Res. 2002;54:553–557. doi: 10.1016/S0141-1136(02)00193-9. [DOI] [PubMed] [Google Scholar]
- Viant MR, Walton JH, TenBrook PL, Tjeerdema RS. Sublethal actions of copper in abalone (haliotis rufescens) as characterized by in vivo 31P NMR. Aquat Toxicol. 2002;57:139–151. doi: 10.1016/S0166-445X(01)00195-3. [DOI] [PubMed] [Google Scholar]
- Wang W, Meng B, Chen W, Ge X, Liu S, Yu J. A proteomic study on postdiapaused embryonic development of brine shrimp (artemia franciscana) Proteomics. 2007;7 doi: 10.1002/pmic.200700259. [DOI] [PubMed] [Google Scholar]
- Warner AH, Clegg JS. Diguanosine nucleotide metabolism and the survival of artemia embryos during years of continuous anoxia. Eur J Biochem. 2001;268 doi: 10.1046/j.1432-1327.2001.01993.x. [DOI] [PubMed] [Google Scholar]
- Warner AH, Finamore FJ. Nucleotide metabolism during brine shrimp embryogenesis. J Biol Chem. 1967;242(8):1933–1937. [PubMed] [Google Scholar]
- Warner AH, MacRae TH, Bagshaw JC. Cell and molecular biology of artemia development. New York: Plenum Press; 1989. [Google Scholar]
- Warner AH, Perz MJ, Osahan JK, Zielinski BS. Potential role in development of the major cysteine protease in larvae of the brine shrimp artemia franciscana. Cell Tissue Res. 1995;282 doi: 10.1007/BF00319129. [DOI] [PubMed] [Google Scholar]
- Zhu XJ, Dai JQ, Tan X, Zhao Y, Yang WJ. Activation of an amp-activated protein kinase is involved in post-diapause development of artemia franciscana encysted embryos. BMC Dev Biol. 2009;9:21–21. doi: 10.1186/1471-213X-9-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


