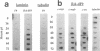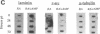Abstract
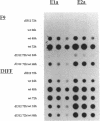
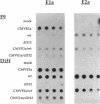
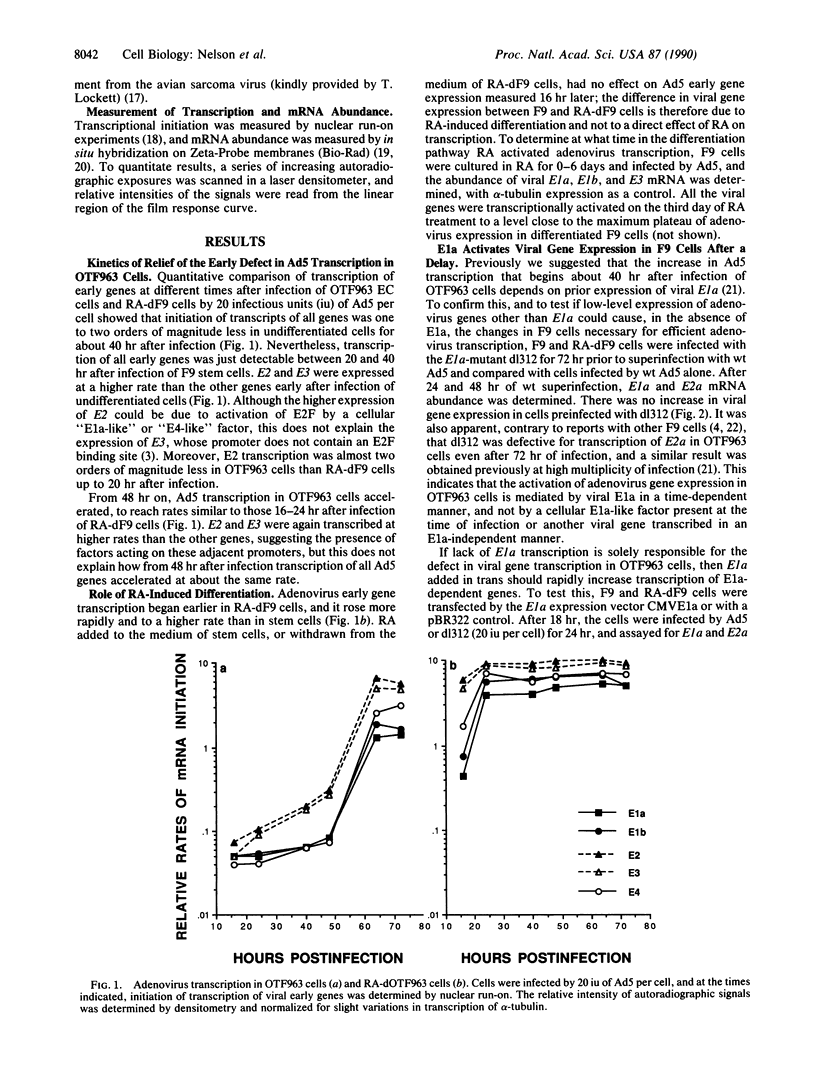
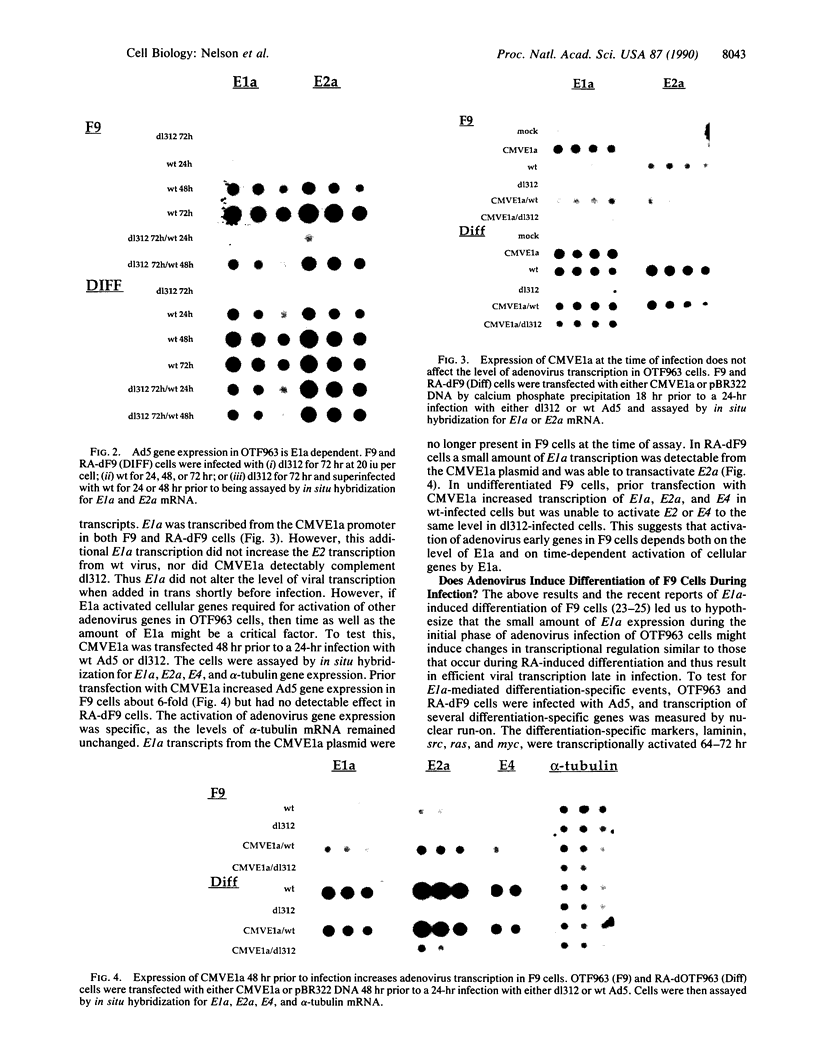
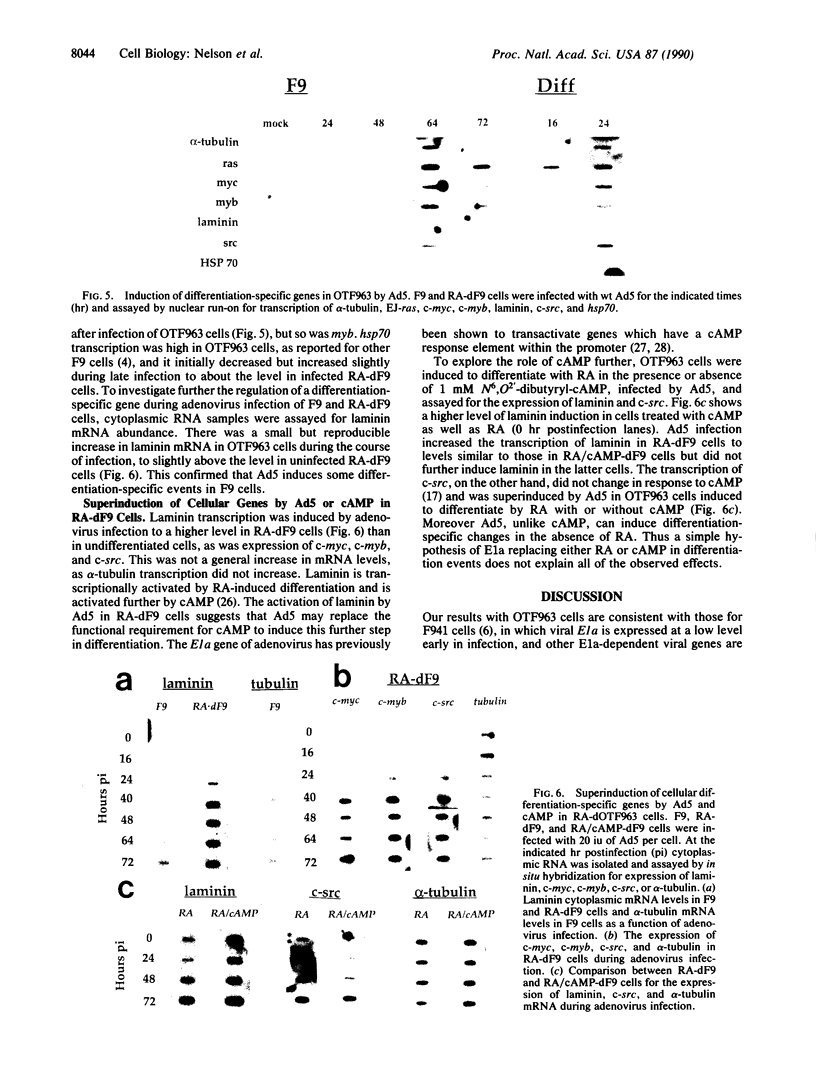
Some undifferentiated F9 embryonal carcinoma cells allow adenovirus genes to be expressed independently of the E1a oncogene normally required for their activation; this has been attributed to a cellular equivalent of E1a in F9 cells. However, transcription of all early genes was low in undifferentiated OTF963 embryonic carcinoma cells during the first 48 hr after infection with adenovirus type 5 (Ad5). Transcription then increased to about the level seen 16 hr after infection of cells induced to differentiate by retinoic acid (RA) (referred to as RA-dF9 cells), but this increase did not occur in cells infected by the E1a deletion mutant dl312. Addition of E1a in trans, or of RA, had no immediate effect on viral transcription in OTF963 cells, but viral transcription increased about 48 hr after these additions. Ad5 induced transcription of several differentiation-specific genes in OTF963 cells with about the same kinetics as their induction by RA. These genes were superinduced in RA-dF9 cells by cAMP or infection by adenovirus. We suggest the small amount of E1a produced early in infection of OTF963 cells activates cellular genes, some of which are differentiation specific and required for efficient transcription of viral genes, so that E1a both induces and is induced by differentiation. The simple hypothesis of a cellular equivalent to E1a does not adequately explain the complex interactions between viral and cellular genes in OTF963 embryonic carcinoma cells.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bellett A. J., Li P., David E. T., Mackey E. J., Braithwaite A. W., Cutt J. R. Control functions of adenovirus transformation region E1A gene products in rat and human cells. Mol Cell Biol. 1985 Aug;5(8):1933–1939. doi: 10.1128/mcb.5.8.1933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boeuf H., Reimund B., Jansen-Durr P., Kédinger C. Differential activation of the E2F transcription factor by the adenovirus EIa and EIV products in F9 cells. Proc Natl Acad Sci U S A. 1990 Mar;87(5):1782–1786. doi: 10.1073/pnas.87.5.1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C., Okayama H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol Cell Biol. 1987 Aug;7(8):2745–2752. doi: 10.1128/mcb.7.8.2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng C., Praszkier J. Regulation of type 5 adenovirus replication in murine teratocarcinoma cell lines. Virology. 1982 Nov;123(1):45–59. doi: 10.1016/0042-6822(82)90293-8. [DOI] [PubMed] [Google Scholar]
- Dean M., Levine R. A., Campisi J. c-myc regulation during retinoic acid-induced differentiation of F9 cells is posttranscriptional and associated with growth arrest. Mol Cell Biol. 1986 Feb;6(2):518–524. doi: 10.1128/mcb.6.2.518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel D. A., Hardy S., Shenk T. cAMP acts in synergy with E1A protein to activate transcription of the adenovirus early genes E4 and E1A. Genes Dev. 1988 Dec;2(12A):1517–1528. doi: 10.1101/gad.2.12a.1517. [DOI] [PubMed] [Google Scholar]
- Grover A., Adamson E. D. Evidence for the existence of an early common biochemical pathway in the differentiation of F9 cells into visceral or parietal endoderm: modulation by cyclic AMP. Dev Biol. 1986 Apr;114(2):492–503. doi: 10.1016/0012-1606(86)90213-7. [DOI] [PubMed] [Google Scholar]
- Imperiale M. J., Kao H. T., Feldman L. T., Nevins J. R., Strickland S. Common control of the heat shock gene and early adenovirus genes: evidence for a cellular E1A-like activity. Mol Cell Biol. 1984 May;4(5):867–874. doi: 10.1128/mcb.4.5.867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones N. C., Rigby P. W., Ziff E. B. Trans-acting protein factors and the regulation of eukaryotic transcription: lessons from studies on DNA tumor viruses. Genes Dev. 1988 Mar;2(3):267–281. doi: 10.1101/gad.2.3.267. [DOI] [PubMed] [Google Scholar]
- Kaddurah-Daouk R., Greene J. M., Baldwin A. S., Jr, Kingston R. E. Activation and repression of mammalian gene expression by the c-myc protein. Genes Dev. 1987 Jun;1(4):347–357. doi: 10.1101/gad.1.4.347. [DOI] [PubMed] [Google Scholar]
- Kovesdi I., Reichel R., Nevins J. R. Role of an adenovirus E2 promoter binding factor in E1A-mediated coordinate gene control. Proc Natl Acad Sci U S A. 1987 Apr;84(8):2180–2184. doi: 10.1073/pnas.84.8.2180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kryszke M. H., Piette J., Yaniv M. Induction of a factor that binds to the polyoma virus A enhancer on differentiation of embryonal carcinoma cells. Nature. 1987 Jul 16;328(6127):254–256. doi: 10.1038/328254a0. [DOI] [PubMed] [Google Scholar]
- La Thangue N. B., Rigby P. W. An adenovirus E1A-like transcription factor is regulated during the differentiation of murine embryonal carcinoma stem cells. Cell. 1987 May 22;49(4):507–513. doi: 10.1016/0092-8674(87)90453-3. [DOI] [PubMed] [Google Scholar]
- Lavi S., Etkin S. Carcinogen-mediated induction of SV40 DNA synthesis in SV40 transformed Chinese hamster embryo cells. Carcinogenesis. 1981;2(5):417–423. doi: 10.1093/carcin/2.5.417. [DOI] [PubMed] [Google Scholar]
- Lee K. A., Hai T. Y., SivaRaman L., Thimmappaya B., Hurst H. C., Jones N. C., Green M. R. A cellular protein, activating transcription factor, activates transcription of multiple E1A-inducible adenovirus early promoters. Proc Natl Acad Sci U S A. 1987 Dec;84(23):8355–8359. doi: 10.1073/pnas.84.23.8355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linial M., Gunderson N., Groudine M. Enhanced transcription of c-myc in bursal lymphoma cells requires continuous protein synthesis. Science. 1985 Dec 6;230(4730):1126–1132. doi: 10.1126/science.2999973. [DOI] [PubMed] [Google Scholar]
- Lockett T. J., Sleigh M. J. Oncogene expression in differentiating F9 mouse embryonal carcinoma cells. Exp Cell Res. 1987 Dec;173(2):370–378. doi: 10.1016/0014-4827(87)90277-1. [DOI] [PubMed] [Google Scholar]
- Montano X., Lane D. P. The adenovirus Ela gene induces differentiation of F9 teratocarcinoma cells. Mol Cell Biol. 1987 May;7(5):1782–1790. doi: 10.1128/mcb.7.5.1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montminy M. R., Bilezikjian L. M. Binding of a nuclear protein to the cyclic-AMP response element of the somatostatin gene. Nature. 1987 Jul 9;328(6126):175–178. doi: 10.1038/328175a0. [DOI] [PubMed] [Google Scholar]
- Nelson C. C., Braithwaite A. W., Silvestro M., Bellett A. J. Defective expression of adenovirus genes during early infection of undifferentiated OTF963 embryonal carcinoma cells. J Virol. 1990 Sep;64(9):4329–4337. doi: 10.1128/jvi.64.9.4329-4337.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onclercq R., Gilardi P., Lavenu A., Cremisi C. c-myc products trans-activate the adenovirus E4 promoter in EC stem cells by using the same target sequence as E1A products. J Virol. 1988 Dec;62(12):4533–4537. doi: 10.1128/jvi.62.12.4533-4537.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paeratakul U., De Stasio P. R., Taylor M. W. A fast and sensitive method for detecting specific viral RNA in mammalian cells. J Virol. 1988 Apr;62(4):1132–1135. doi: 10.1128/jvi.62.4.1132-1135.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pei R., Berk A. J. Multiple transcription factor binding sites mediate adenovirus E1A transactivation. J Virol. 1989 Aug;63(8):3499–3506. doi: 10.1128/jvi.63.8.3499-3506.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinlan M. P. The Ad5 12S growth factor induces F9 cell proliferation and differentiation. Oncogene. 1989 Aug;4(8):1051–1055. [PubMed] [Google Scholar]
- Reichel R., Kovesdi I., Nevins J. R. Developmental control of a promoter-specific factor that is also regulated by the E1A gene product. Cell. 1987 Feb 13;48(3):501–506. doi: 10.1016/0092-8674(87)90200-5. [DOI] [PubMed] [Google Scholar]
- Sassone-Corsi P. Cyclic AMP induction of early adenovirus promoters involves sequences required for E1A trans-activation. Proc Natl Acad Sci U S A. 1988 Oct;85(19):7192–7196. doi: 10.1073/pnas.85.19.7192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sleigh M. J. Differential regulation of viral and cellular genes in F9 mouse embryonal carcinoma cells. Nucleic Acids Res. 1987 Nov 25;15(22):9379–9395. doi: 10.1093/nar/15.22.9379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sleigh M. J., Lockett T. J., Kelly J., Lewy D. Competition studies with repressors and activators of viral enhancer function in F9 mouse embryonal carcinoma cells. Nucleic Acids Res. 1987 May 26;15(10):4307–4324. doi: 10.1093/nar/15.10.4307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speck N. A., Baltimore D. Six distinct nuclear factors interact with the 75-base-pair repeat of the Moloney murine leukemia virus enhancer. Mol Cell Biol. 1987 Mar;7(3):1101–1110. doi: 10.1128/mcb.7.3.1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strickland S., Smith K. K., Marotti K. R. Hormonal induction of differentiation in teratocarcinoma stem cells: generation of parietal endoderm by retinoic acid and dibutyryl cAMP. Cell. 1980 Sep;21(2):347–355. doi: 10.1016/0092-8674(80)90471-7. [DOI] [PubMed] [Google Scholar]
- Velcich A., Kern F. G., Basilico C., Ziff E. B. Adenovirus E1a proteins repress expression from polyomavirus early and late promoters. Mol Cell Biol. 1986 Nov;6(11):4019–4025. doi: 10.1128/mcb.6.11.4019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velcich A., Ziff E. B. The adenovirus-5 12S E1a protein, but not the 13S induces expression of the endoA differentiation marker in F9 cells. Oncogene. 1989 Jun;4(6):707–713. [PubMed] [Google Scholar]
- Wasylyk B., Imler J. L., Chatton B., Schatz C., Wasylyk C. Negative and positive factors determine the activity of the polyoma virus enhancer alpha domain in undifferentiated and differentiated cell types. Proc Natl Acad Sci U S A. 1988 Nov;85(21):7952–7956. doi: 10.1073/pnas.85.21.7952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young K. S., Weigel R., Hiebert S., Nevins J. R. Adenovirus E1A-mediated negative control of genes activated during F9 differentiation. Mol Cell Biol. 1989 Jul;9(7):3109–3113. doi: 10.1128/mcb.9.7.3109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zajchowski D. A., Jalinot P., Kedinger C. EIa-mediated stimulation of the adenovirus EIII promoter involves an enhancer element within the nearby EIIa promoter. J Virol. 1988 May;62(5):1762–1767. doi: 10.1128/jvi.62.5.1762-1767.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]