Abstract
Semaphorin3A is a secreted protein known to be involved in organogenesis, immune responses and cancer. In the kidney, semaphorin3A is expressed in the glomerular podocytes, distal tubules and collecting tubules, and believed to play a role in the regulation of the kidney development and function. We examined the serum and urinary semaphorin3A levels in 72 patients with renal disease and 5 healthy volunteers. The patients had been diagnosed with thin basement membrane disease (n=4), minimal change nephrotic syndrome (MCNS; n=22), IgA nephritis (n=21), membranous nephropathy (n=16) and focal segmental glomerular sclerosis (n=9). The level of urinary semaphorin3A in MCNS patients tended to be relatively high among all disease groups. We also investigated the urinary semaphorin3A level in 7 patients with MCNS from disease onset to remission during the drug therapy. MCNS patients in pre-remission states had higher urinary semaphorin3A levels than those in post-remission states receiving immunosuppressive therapies. These results suggested that the urinary semaphorin3A level correlates with the MCNS activity. Semaphorin3A has the potential as a biomarker for MCNS to clarify the reactivity for therapy and may be useful in examining other glomerular diseases with proteinuria as well.
Keywords: semaphorin3A, biomarker, minimal change nephrotic syndrome
Introduction
The pathological findings in renal diseases with proteinuria, such as minimal change nephrotic syndrome (MCNS), membranous nephropathy (MN), IgA nephritis (IgA-N) and focal segmental glomerular sclerosis (FSGS), as well as other cases of glomerular nephritis are podocyte foot process effacement and slit diaphragm absence.1 The damage to podocytes is believed to cause proteinuria, and long-standing proteinuria leads to the deterioration of the nephron, which results in glomerular sclerosis and renal fibrosis. To evaluate the damage associated with these renal pathological findings, a biomarker capable of determining the precise state of the renal function at a given moment is needed. Urinary podocyte loss and urinary nephrin have been studied as candidate biomarkers of diabetic nephropathy and other nephropathies.2,3 However, no specific biomarker that relates to the activity of glomerular diseases has been identified.
Semaphorins are guidance proteins that influence axonal progression.4 There are seven subclasses that are distinguished by the differences in several domains.5 Semaphorin3A is a member of the semaphorin family that is involved in nerve cell migration, angiogenesis, regulation of the immune system and cancer progression.5–8 With regard to the kidney, semaphorin3A is reported to be an endogenous antagonist of ureteric bud branching and a regulator of endothelial cell number and podocyte differentiation during glomerular development.9,10 Semaphorin3A and its receptors neuropilin-1 and neuropilin-2 are expressed on differentiated podocytes, distal tubules and collecting duct cells.11,12 Recombinant semaphorin3A decreases the expression of podocin with nephrin and CD2-associated protein in vitro.12 Additionally, recombinant semaphorin3A administration causes proteinuria in adult mice.13 The associated pathological findings are podocyte foot process effacement and fusion in the glomeruli.13 Altogether, semaphorin3A is believed to play various roles in the podocyte maintenance of the slit diaphragm.
An interesting report suggested that semaphorin3A might represent a biomarker of acute kidney injury (AKI).14 Another report said that hypertensive patients with albuminuria with or without a reduced estimated glomerular filtration rate had increased urinary semaphorin3A levels.15 Furthermore, urinary semaphorin3A from diabetic patients was reported to increase with the deterioration of nephropathy, and in basic research, the suppression of semaphorin3A was suggested to inhibit the renal damage of diabetic nephropathy.16 From these reports, semaphorin3A might influence the pathophysiology in the course of kidney disease, with respect to not only interstitial damage but also glomerular diseases. Therefore, we hypothesized that semaphorin3A played some roles in glomerular diseases with proteinuria.
In this study, we analyzed the serum and urinary semaphorin3A levels of patients with glomerular disease who were examined and treated at the nephrology department in our hospital and other hospitals. In addition, we followed up several patients who received immunosuppressive therapy. Through these efforts, we clarified the association between MCNS disease states and urinary semaphorin3A.
Materials and methods
Patient population
This study is a multi-institutional, retrospective analysis evaluating patients’ serum and urine samples. We studied 72 Japanese patients admitted to the Okayama University Hospital and 3 other facilities for the investigation of urinary abnormalities or renal failure between January 2011 and January 2016. The patients were 17–90 years of age. At the time of renal biopsy in our hospital, we collected serum and urine samples from the patients. We diagnosed each nephropathy according to the pathological findings (immunofluorescence, light microscopic and electron microscopic analyses) of renal biopsy and the clinical findings. The patients were diagnosed with thin basement membrane disease (TBM), MCNS, IgA-N, MN or FSGS. In addition, we followed seven patients with MCNS from disease onset to remission during drug therapy to examine the changes in the urinary semaphorin3A levels. All the data were retrospectively obtained from electronic medical charts. Five healthy volunteers from Okayama University also participated in this study.
The study was approved by the institutional review board of the Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, Japanese Red Cross Society Himeji Hospital, Shigei Medical Research Hospital and Kurashiki Central Hospital. We performed serum and urine sampling as part of approved protocols (approval numbers 843 and 2063). We obtained written informed consent from all subjects for this study. This study was registered with the Clinical Trial Registry of the University Hospital Medical Information Network (registration number UMIN000013422 and UMIN000010104). All the patient samples were stored at −80°C.
Semaphorin3A quantification by enzyme-linked immunosorbent assay
We measured the level of semaphorin3A in serum and urine using an enzyme-linked immunosorbent assay kit (MyBio-Source, San Diego, CA, USA). Microtiter plates were precoated with an antibody. The serum and urine samples, standards and a buffered solution were added to the plates. After the enzyme conjugate was added and thoroughly mixed, the plates were covered and incubated for 1 h at room temperature. After washing the plates, we added two types of substrates to the plates. The enzyme and substrate were incubated to react for 15 min. Following mixing with the sulfuric acid solution to arrest the reaction, we evaluated the levels of semaphorin3A by measuring the optical density at 450 nm using a microplate reader (Thermo Scientific, Yokohama, Japan).
N-Acetyl-β-d-glucosaminidase (NAG) quantification by a colorimetric analysis
We measured patients’ urinary NAG level by determining the concentration of a chemical substance in a method called colorimetric analysis. We used the β-N-acetylglucosaminidase assay kit (Sigma-Aldrich, St. Louis, MO, USA). The assay is based on the hydrolysis of the NAG substrate, 4-nitrophenl N-acetyl-β-d-glucosaminide by the enzyme. First, the substrate solution and the standard solution were equilibrated to 37°C by incubating for 5 min. After the substrate solution was added to each sample, the samples were incubated for 10 min at 37°C. The stop solution was then added to each well. The absorption was measured at 405 nm.
Immunohistochemistry analysis
Immunohistochemistry was performed on formalin-fixed, paraffin-embedded sections. For the immunohistochemistry of semaphorin3A and neuropilin-1, paraffin-embedded sections (4 μm) were dewaxed and rehydrated. Antigen retrieval was performed in a microwave oven for 5 min. Endogenous peroxidase was quenched with 3% H2O2 for 30 min, after which the sections were blocked with 3% bovine serum albumin (Sigma-Aldrich) for 30 min. The sections were first incubated with primary antibody for semaphorin3A (ab23393; Abcam, Cambridge, MA, USA) and neuropilin-1 (ab81321; Abcam) in a humidity cabinet for 1 h at room temperature, and the negative controls were incubated in the normal serum for the same time. Next, the sections were incubated with biotin-conjugated goat anti-rabbit IgG antibody (Nichirei Bioscience, Tokyo, Japan) for 30 min at room temperature. The sections were then incubated for 30 min using the Vectastain ABC Elite reagent kit (Vector Laboratories, Burlingame, CA, USA) and developed with diaminobenzidine (Vector Laboratories) for 5 min. All slides were counterstained with hematoxylin, and the images were recorded using an FSX-100 microscope (Olympus, Tokyo, Japan).
Statistical analysis
All data are expressed as the means ± SD. Multiple comparisons were performed by the Kruskal–Wallis method. Analyses of the correlation between urinary semaphorin3A and urinary protein were performed by Spearman’s correlation coefficient. The changes in the urinary semaphorin3A level between onset and remission in MCNS patients was analyzed by Wilcoxon signed-rank test. A p-value of <0.05 was regarded as statistically significant. The data were analyzed using the IBM SPSS Statistics software program, version 22 (IBM, Armonk, NY, USA).
Results
Patients’ characteristics
Four patients (5.6%) were diagnosed with TBM, 22 (30.6%) with MCNS, 21 (29.2%) with IgA-N, 16 (22.2%) with MN and 9 (12.5%) with FSGS (Table 1). The diagnosis was established based on the clinical and pathological findings (immunofluorescence, light microscopic and electron microscopic analyses). The TBM, IgA-N and MN patients did not receive any treatment for their renal disorder before sampling. In contrast, 15 patients (68%) in the MCNS group started treatment with steroids and/or immunosuppressive drugs before sampling of the serum and urine, and about half of FSGS patients (55.6%) received immunosuppressive therapy for complicated diseases and a clinical diagnosis of FSGS before sampling. Patients in the MN group tended to be older than in MCNS and IgA-N groups. The degree of proteinuria in the MCNS, MN and FSGS groups was significantly higher than in the healthy control group. Of the 15 patients (68%) in the MCNS group who had already started immunosuppressive therapy, 4 had already achieved remission by the time of sampling, and the remaining 11 exhibited reduced levels of urinary protein. Nephrotic-range proteinuria (≥3.5 g) was evident in 13 patients (59%) in the MCNS group.
Table 1.
Characteristics of the patients and controls
| Characteristic | TBM (n=4) | MCNS (n=22) | IgA-N (n=21) | MN (n=16) | FSGS (n=9) | Control (n=5) |
|---|---|---|---|---|---|---|
| Age (years) | 47.8±8.3 | 45.7±24.0* | 40.7±16.2** | 66.7±17.3 | 49.6±15.8 | 37.0±7.5 |
| Male (%) | 2 (50.0) | 13 (59.1) | 8 (40.1) | 11 (68.8) | 6 (67) | 3 (60) |
| Cr (mg/dL) | 0.65±0.15 | 0.88±0.40 | 1.00±0.58 | 0.96±0.36 | 1.13±0.39 | 0.80±0.15 |
| uTP/uCr (g/g⋅Cr) | 0.34±0.13 | 5.28±4.30†† | 2.00±2.00 | 6.62±6.20†† | 3.11±2.52† | 0.03±0.02 |
| Drug therapy (%) | 0 | 15 (68.0) | 0 | 0 | 5 (55.6) | 0 |
Notes: Mean±SD are reported for the continuous variables. Percentages are reported for the categorical variables.
p<0.05 versus MN,
p<0.01 versus MN,
p<0.05 versus control,
p<0.01 versus control.
Abbreviations: Cr, serum creatinine; FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MCNS, minimal change nephrotic syndrome; MN, membranous nephropathy; TBM, thin basement membrane disease.
Association of urinary semaphorin3A with remission in MCNS
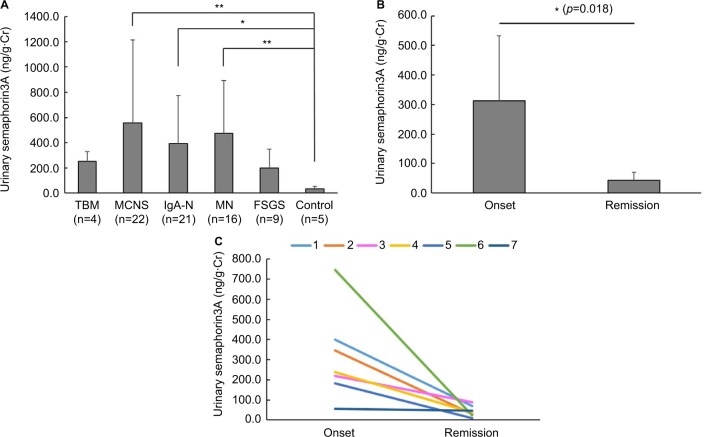
The urinary semaphorin3A level in MCNS was statistically higher than in control (Figure 1A). We suspected there might be a correlation between semaphorin3A and MCNS activity, so we tracked the urinary semaphorin3A level of seven MCNS patients from onset to remission. Time after remission ranged from about 2 weeks to 3 years. The levels of urinary semaphorin3A at onset were significantly higher than those at remission (312.14±220.98 vs 43.67±26.66 ng/g·Cr; Figure 1B and C respectively). This result suggests the urinary semaphorin3A might reflect the acute functional or morphological change of glomeruli of MCNS. Urinary semaphorin3A levels in IgA-N and MN groups were also higher than in control (Figure 1A). Then we also examined the urinary N-acetyl-β-d-glucosaminidase (NAG) of each patient as a biomarker of damage to the proximal tubules. We also observed significant increases in MCNS, IgA-N and MN groups compared with control (Figure S1A). These findings suggest that the difference in the urinary levels of semaphorin3A might reflect the degree of interstitial damage induced by each glomerular disease. The serum semphorin3A levels in MCNS and MN patients were higher than in control (Figure S1B).
Figure 1.
Urinary semaphorin3A levels and the change of the urinary semaphorin3A level in MCNS patients between onset and remission.
Notes: (A) Urinary semaphorin3A levels in the MCNS, IgA-N and MN groups are higher than in control. *p<0.05, **p<0.01. (B) The urinary semaphorin3A level at disease onset (312.1±221.0 ng/mL) was significantly higher than at remission (43.8±26.7 ng/mL) in patients with MCNS (n=7). *p<0.05. (C) The urinary semaphorin3A level was reduced by treatment in MCNS patients (n=7).
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MCNS, minimal change nephrotic syndrome; MN, membranous nephropathy; TBM, thin basement membrane disease.
Localization of semaphorin3A and neuropilin-1 in MCNS
Immunohistochemical examinations revealed semaphorin3A in the glomerular podocytes, distal tubules and collecting tubules (Figure 2A: in glomerulus, Figure 2B: in tubules) in the kidneys of MCNS patients. The distributions exhibited in patients with other glomerular diseases and TBM, which was considered almost normal clinically, were the same as that observed for MCNS kidney (Figure S2A, C, E and G: in glomerulus, Figure 2B, D, F and H: in tubules). Additionally, one of the semaphorin3A receptors, neuropilin-1, was also detected in the podocytes, the luminal side of the distal tubules and the collecting tubules in the MCNS group (Figure 2C: in glomerulus, Figure 2D: in tubules). In glomerular diseased and TBM kidneys, neuropilin-1-positive cells were similarly mainly detected in the glomerular podocytes and in the luminal side of the distal tubular cells (Figure S3A, C, E and G: in glomerulus, Figure 3B, D, F and H: in tubules).
Figure 2.
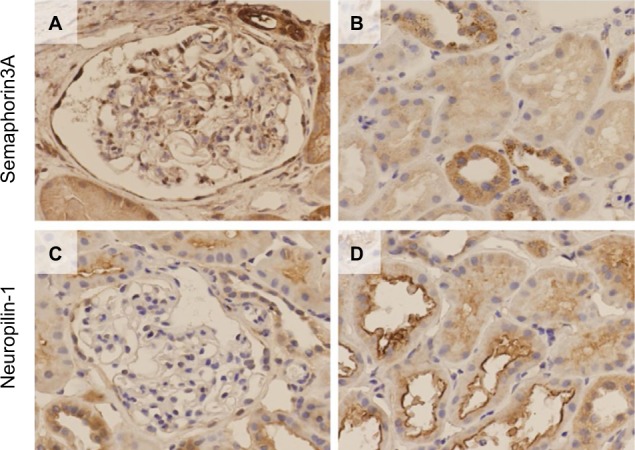
Immunohistochemical localization of semaphorin3A and neuropilin-1 in the glomerulus and renal tubules in patients with MCNS.
Notes: (A) The podocytes exhibit partial positive staining in the glomerulus. (B) The renal tubules, which appear to be distal or collecting tubules in the absence of a brush border, are positively stained. (C) The podocytes exhibit focal positive staining for neuropilin-1 in the glomerulus. (D) The renal tubules are positively stained, particularly at the luminal side for neuropilin-1.
Abbreviation: MCNS, minimal change nephrotic syndrome.
Urinary semaphorin3A levels vs urinary protein levels
Some studies have reported a correlation with urinary semaphorin3A and urinary protein or albuminuria in diabetic nephropathy and chronic kidney disease.15,16 We therefore analyzed the correlation between urinary semaphorin3A and urinary protein. None of the renal disease groups exhibited similar tendencies (Table 2). Nonsignificant correlation was observed in the subjects of the MCNS group not receiving corticosteroid therapy (data not shown).
Table 2.
Correlations of urinary protein levels with urinary semaphorin3A levels
| TBM | MCNS | IgA-N | MN | FSGS | Control | |
|---|---|---|---|---|---|---|
| Number of subjects | 4 | 22 | 21 | 16 | 9 | 5 |
| Correlation coefficient | 0.800 | −0.304 | 0.242 | 0.409 | 0.383 | 0.000 |
| p-value | 0.200 | 0.168 | 0.291 | 0.116 | 0.308 | 1.000 |
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MCNS, minimal change nephrotic syndrome; MN, membranous nephropathy; TBM, thin basement membrane disease; r, correlation coefficient.
Urinary NAG levels vs urinary semaphorin3A levels
We examined the correlation between urinary semaphorin3A and urinary NAG. The IgA-N, MN and FSGS groups showed significant correlations; however, the MCNS group did not show any correlations (Table 3). Generally, most of the IgAN, MN and FSGS patients showed chronic disease progression accompanied by tubulointerstitial damage; therefore, semaphorin3A in the renal tubules might have been excreted into the urine.
Table 3.
Correlations of urinary NAG levels with urinary semaphorin3A levels
| TBM | MCNS | IgA-N | MN | FSGS | Control | |
|---|---|---|---|---|---|---|
| Number of subjects | 4 | 20 | 21 | 14 | 9 | 5 |
| Correlation coefficient | 0.800 | −0.041 | 0.734 | 0.587 | 0.683 | 0.400 |
| p-value | 0.200 | 0.865 | 0.00015* | 0.027* | 0.042* | 0.565 |
Note:
p<0.05.
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MCNS, minimal change nephrotic syndrome; MN, membranous nephropathy; NAG, N-acetyl-β-glucosaminidase; TBM, thin basement membrane disease; r, correlation coefficient.
Discussion
Semaphorin3A is reported to be a candidate predictive biomarker for AKI or chronic kidney disease severity.14,15 We examined the semaphorin3A levels of glomerular disease patients. We also tracked the levels of patients with MCNS who achieved remission. From these analyses, we recognized a drastic decrease in the urinary semaphorin3A from disease onset to remission in MCNS patients, suggesting that urinary semaphorin3A levels are associated with the activity of MCNS.
A previous report showed that AKI induced the excretion of semaphorin3A through urine.14 MCNS often causes tubular ischemia that leads to AKI via circulatory disturbance and impaired glomerular permeability.17,18 In this study, patients with MCNS did not reach an AKI state. We did not observe any obvious tubular damage as pathological findings in the MCNS group. On the other hand, there were significant increase in the urinary NAG level in MCNS, IgA-N and MN patients. However, in MCNS group, neither urinary protein nor urinary NAG had a significant correlation with urinary semaphorin3A. The precise origin of urinary semaphorin3A in MCNS could not be determined from our results. However, given that semaphorin3A expression is found on podocytes and renal tubules, semaphorin3A in urine might represent glomerular podocyte disorder, tubulointerstitial damage or both. Our results suggested that semaphorin3A might be involved in the pathophysiology of MCNS. In addition, the evaluation of urinary semaphorin3A levels in MCNS patients might be useful for the examination of reactivity toward immunosuppressive therapy.
MN and FSGS are also typical glomerular diseases presenting with substantially elevated urinary protein levels, and some patients with IgA-N also show high urinary protein levels. The histologic features of nephrotic syndrome include the loss of integrity of the foot processes and the slit diaphragm, leading to foot process effacement.1 The typical slit diaphragm structures disappear in nephrotic syndrome, regardless of the type of nephropathy.1 Although both MCNS and MN often show an increase in glomerular permeability with few structural abnormalities on light microscopy, the etiological association of immunoglobulin deposits is well documented in MN.19,20 Urinary semaphorin3A levels in MCNS, IgA-N and MN groups were higher than in control, but only IgA-N and MN exhibited the significant correlations between urinary NAG levels. In our study, urinary semaphorin3A in IgA-N and MN might have mostly reflected the tubulointerstitial damage. MN also showed an increase in serum semaphorin3A. At present, there is little report of human serum semaphorin3A; therefore, the significance of serum semaphorin3A is yet to be elucidated. The mechanism of increase of serum semaphorin3A in MN is not clear. With regard to FSGS, certain nonimmunoglobulin-circulating permeability factors have been postulated.21 The difference in these disease mechanisms may induce variation in the findings for podocyte disorder, and the roles of semaphorin3A at each part of the nephron may also differ among diseases. However, this consideration remains speculative, and additional research is required.
Only 72 patients were enrolled in this study, mainly because most samples were incomplete or unavailable. We also did not evaluate any patients with diabetic nephropathy, as we usually do not perform renal biopsies on patients who demonstrate the clinical features typical of diabetic nephropathy. Therefore, we did not collect sufficient number of samples from patients with diabetic nephropathy for this study. In this study, the correlation of urinary semaphorin3A level and urinary protein in the MCNS group was not significant. The small number of subjects might have affected the results. Another limitation of this study is that we were only able to follow patients with MCNS. Besides, in MCNS, we could follow the change of urinary semaphorin3A levels only twice. By examining the urinary semaphorin3A levels of patients with various glomerular diseases in various disease states, we can improve our understanding of the role of semaphorin3A in these diseases.
We exhibited the increase of urinary semaphorin3A in specific glomerular diseases in this study as already reported AKI and CKD. Semaphorin3A has also been reported as a candidate therapeutic target in diabetic nephropathy, because a semaphorin3A inhibitory peptide was reported to ameliorate diabetic nephropathy in an animal model.16 Applying the findings from pharmacological investigations in other glomerular diseases such as MCNS to this disease may therefore be useful. Further study is required to clarify the role of semaphorin3A in patients with MCNS and other renal glomerular diseases in a larger cohort in the future. We conclude that urinary semaphorin3A levels correlate with the MCNS activity and that urinary semaphorin3A has the potential as a biomarker in MCNS treatment for obtaining a differential diagnosis from other renal diseases.
Supplementary materials
Urinary NAG and serum semaphorin3A.
Notes: (A) Urinary NAG levels in the MCNS, IgA-N and MN groups are higher than in control. *p<0.05, **p<0.01. (B) Serum semaphorin3A levels in the MCNS and MN groups are higher than in control. *p<0.05, **p<0.01.
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MCNS, minimal change nephrotic syndrome; MN, membranous nephropathy; NAG, N-acetyl-β-glucosaminidase; TBM, thin basement membrane disease.
Immunohistochemical analysis for semaphorin3A in the glomerulus and renal tubules for each nephropathy.
Notes: (A, C, E, G) Glomerulus, (B, D, F, H) tubules. (A and B) TBM, (C and D) IgA-N, (E and F) MN, (G and H) FSGS. Representative figures for each nephropathy. The podocytes exhibit partial positive staining in the glomerulus. The renal tubules, which appear to be distal or collecting tubules in the absence of a brush border, are positively stained. There are no apparent differences in the staining intensity among the nephropathies.
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MN, membranous nephropathy; TBM, thin basement membrane disease.
Immunohistochemical analysis for neuropilin-1 in the glomerulus and renal tubules for each nephropathy.
Notes: (A, C, E, G) Glomerulus, (B, D, F, H) tubules. (A and B) TBM, (C and D) IgA-N, (E and F) MN, (G and H) FSGS. Representative figures for each nephropathy. The podocytes exhibit 2 partial positive staining in the glomerulus. The renal tubules, which appear to be distal or collecting tubules in the absence of a brush border, are positively stained. There are no apparent differences in the staining intensity among the nephropathies.
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MN, membranous nephropathy; TBM, thin basement membrane disease.
Acknowledgments
We thank Professor Hitoshi Sugiyama, Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences, for his helpful advice.
Footnotes
Disclosure
Shinji Kitamura takes part in a cooperative research about semaphorins with Chiome Bioscience Inc. The other authors report no conflicts of interest in this work.
References
- 1.Somlo S, Mundel P. Getting a foothold in nephrotic syndrome. Nat Genet. 2000;24(4):333–335. doi: 10.1038/74139. [DOI] [PubMed] [Google Scholar]
- 2.Pätäri A, Forsblom C, Havana M, Taipale H, Groop PH, Holthöfer H. Nephrinuria in diabetic nephropathy of type 1 diabetes. Diabetes. 2003;52(12):2969–2974. doi: 10.2337/diabetes.52.12.2969. [DOI] [PubMed] [Google Scholar]
- 3.Yu D, Petermann A, Kunter U, Rong S, Shankland SJ, Floege J. Urinary podocyte loss is a more specific marker of ongoing glomerular damage than proteinuria. J Am Soc Nephrol. 2005;16(6):1733–1741. doi: 10.1681/ASN.2005020159. [DOI] [PubMed] [Google Scholar]
- 4.Kolodkin AL, Matthes DJ, Goodman CS. The semaphorin genes encode a family of transmembrane and secreted growth cone guidance molecules. Cell. 1993;75(7):1389–1399. doi: 10.1016/0092-8674(93)90625-z. [DOI] [PubMed] [Google Scholar]
- 5.Goodman CS, Kolodkin AL, Luo Y, Püschel AW, Raper JA. Unified nomenclature for the semaphorins/collapsins. Cell. 1999;97(5):551–552. doi: 10.1016/s0092-8674(00)80766-7. [DOI] [PubMed] [Google Scholar]
- 6.Capparuccia L, Tamagnone L. Semaphorin signaling in cancer cells and in cells of the tumor microenvironment – two sides of a coin. J Cell Sci. 2009;122(Pt 11):1723–1736. doi: 10.1242/jcs.030197. [DOI] [PubMed] [Google Scholar]
- 7.Bussolino F, Giraudo E, Serini G. Class 3 semaphorin in angiogenesis and lymphangiogenesis. Chem Immunol Allergy. 2014;99:71–88. doi: 10.1159/000353315. [DOI] [PubMed] [Google Scholar]
- 8.Takamatsu H, Kumanogoh A. Diverse roles for semaphorin-plexin signaling in the immune system. Trends Immunol. 2012;33(3):127–135. doi: 10.1016/j.it.2012.01.008. [DOI] [PubMed] [Google Scholar]
- 9.Tufro A, Teichman J, Woda C, Villegas G. Semaphorin3a inhibits ureteric bud branching morphogenesis. Mech Dev. 2008;125(5–6):558–568. doi: 10.1016/j.mod.2007.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Reidy KJ, Villegas G, Teichman J, et al. Semaphorin3a regulates endothelial cell number and podocyte differentiation during glomerular development. Development. 2009;136(23):3979–3989. doi: 10.1242/dev.037267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Villegas G, Tufro A. Ontogeny of semaphorins 3A and 3F and their receptors neuropilins 1 and 2 in the kidney. Mech Dev. 2002;119(Suppl 1):S149–S153. doi: 10.1016/s0925-4773(03)00108-4. [DOI] [PubMed] [Google Scholar]
- 12.Guan F, Villegas G, Teichman J, Mundel P, Tufro A. Autocrine class 3 semaphorin system regulates slit diaphragm proteins and podocyte survival. Kidney Int. 2006;69(9):1564–1569. doi: 10.1038/sj.ki.5000313. [DOI] [PubMed] [Google Scholar]
- 13.Tapia R, Guan F, Gershin I, Teichman J, Villegas G, Tufro A. Semaphorin3a disrupts podocyte foot processes causing acute proteinuria. Kidney Int. 2008;73(6):733–740. doi: 10.1038/sj.ki.5002726. [DOI] [PubMed] [Google Scholar]
- 14.Jayakumar C, Ranganathan P, Devarajan P, Krawczeski CD, Looney S, Ramesh G. Semaphorin 3A is a new early diagnostic biomarker of experimental and pediatric acute kidney injury. PLoS One. 2013;8(3):e58446. doi: 10.1371/journal.pone.0058446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Viazzi F, Ramesh G, Jayakumar C, Leoncini G, Garneri D, Pontremoli R. Increased urine semaphorin-3A is associated with renal damage in hypertensive patients with chronic kidney disease: a nested case-control study. J Nephrol. 2015;28(3):315–320. doi: 10.1007/s40620-014-0097-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mohamed R, Ranganathan P, Jayakumar C, et al. Urinary semaphorin 3A correlates with diabetic proteinuria and mediates diabetic nephropathy and associated inflammation in mice. J Mol Med. 2014;92(12):1245–1256. doi: 10.1007/s00109-014-1209-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stellato T, Cappelleri A, Farina M, et al. Severe reversible acute renal failure in idiopathic nephrotic syndrome. J Nephrol. 2010;23(6):717–724. [PubMed] [Google Scholar]
- 18.Smith JD, Hayslett JP. Reversible renal failure in the nephrotic syndrome. Am J Kidney Dis. 1992;19(3):201–213. doi: 10.1016/s0272-6386(13)80001-7. [DOI] [PubMed] [Google Scholar]
- 19.Tipping PG. Are podocytes passive or provocative in proteinuric glomerular pathology? J Am Soc Nephrol. 2008;19(4):651–653. doi: 10.1681/ASN.2008020156. [DOI] [PubMed] [Google Scholar]
- 20.Wasserstein AG. Membranous glomerulonephritis. J Am Soc Nephorol. 1997;8(4):664–674. doi: 10.1681/ASN.V84664. [DOI] [PubMed] [Google Scholar]
- 21.Mathieson PW. Minimal change nephropathy and focal segmental glomerulosclerosis. Semin Immunopathol. 2007;29(4):415–426. doi: 10.1007/s00281-007-0094-z. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Urinary NAG and serum semaphorin3A.
Notes: (A) Urinary NAG levels in the MCNS, IgA-N and MN groups are higher than in control. *p<0.05, **p<0.01. (B) Serum semaphorin3A levels in the MCNS and MN groups are higher than in control. *p<0.05, **p<0.01.
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MCNS, minimal change nephrotic syndrome; MN, membranous nephropathy; NAG, N-acetyl-β-glucosaminidase; TBM, thin basement membrane disease.
Immunohistochemical analysis for semaphorin3A in the glomerulus and renal tubules for each nephropathy.
Notes: (A, C, E, G) Glomerulus, (B, D, F, H) tubules. (A and B) TBM, (C and D) IgA-N, (E and F) MN, (G and H) FSGS. Representative figures for each nephropathy. The podocytes exhibit partial positive staining in the glomerulus. The renal tubules, which appear to be distal or collecting tubules in the absence of a brush border, are positively stained. There are no apparent differences in the staining intensity among the nephropathies.
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MN, membranous nephropathy; TBM, thin basement membrane disease.
Immunohistochemical analysis for neuropilin-1 in the glomerulus and renal tubules for each nephropathy.
Notes: (A, C, E, G) Glomerulus, (B, D, F, H) tubules. (A and B) TBM, (C and D) IgA-N, (E and F) MN, (G and H) FSGS. Representative figures for each nephropathy. The podocytes exhibit 2 partial positive staining in the glomerulus. The renal tubules, which appear to be distal or collecting tubules in the absence of a brush border, are positively stained. There are no apparent differences in the staining intensity among the nephropathies.
Abbreviations: FSGS, focal segmental glomerular sclerosis; IgA-N, IgA nephritis; MN, membranous nephropathy; TBM, thin basement membrane disease.