Abstract
The aim of this study was to identify genes for which the expression within carotid atherosclerosis was reproducibly associated with the symptoms of cerebral embolization. Two publically available microarray datasets E-MEXP-2257 and GSE21545 were analysed using GeneSpring 11.5. The two datasets utilized a total of 22 and 126 carotid atherosclerosis samples, obtained from patients with and without symptoms of cerebral embolization, respectively. To assess whether the findings were reproducible we analysed carotid atherosclerosis samples from another 8 patients with and 7 patients without symptoms of cerebral embolization using real-time PCR. In vitro studies using VSMC were performed to assess the functional relevance of one of the validated genes. We identified 1624 and 135 differentially expressed genes within carotid atherosclerosis samples of symptomatic compared to asymptomatic patients using the E-MEXP-2257 and GSE21545 datasets, respectively (≥1.15-absolute fold-change, P < 0.05). Only 7 differentially expressed genes or 0.4% (7/1,752) were consistent between the datasets. We validated the differential expression of ARSB which was upregulated 1.15-fold (P = 0.029) in atherosclerosis from symptomatic patients. In vitro incubation of VSMCs with the ARSB inhibitor L-ascorbic acid resulted in marked upregulation of SIRT1 and AMPK. This study suggests that ARSB may represent a novel target to limit carotid embolization.
Introduction
The prevalence of carotid artery stenosis is approximately 4% to 8% in adults aged 50 to 79 years1–3. Carotid atherosclerosis is estimated to be responsible for ~20% of all ischemic strokes4, 5. Atherosclerotic plaque rupture and cerebral embolization is believed to be the mechanism by which carotid atherosclerosis leads to cerebral symptoms, such as transient ischemic attack and stroke1. However, the identification of the so-called “vulnerable plaque” has been elusive. The first whole-genome gene expression study of stroke was published a decade ago6. The authors at that time profiled peripheral blood mononuclear cells of stroke patients and compared them with those of healthy donors6. Almost 200 differentially expressed genes were identified; however, those assessed had limited diagnostic value with estimated specificity and sensitivity less than 80%6. A number of previous studies have examined differential gene expression in symptomatic carotid atherosclerosis; however, findings have not been consistent across the studies7–15. All these results question the value of differential gene expression in representing true molecular determinants of stroke. The current study re-examined previously published microarray datasets of carotid artery atherosclerosis to gain further insight. An attempt has been made to identify consistent and reproducible differentially expressed genes using publically available microarray datasets that utilized carotid endarterectomy samples from symptomatic and asymptomatic patients.
Methods
Data preparation
Two human microarray datasets were included in this study to establish a consensus set of differentially expressed genes in carotid atherosclerosis associated with the recent symptoms of cerebral embolization12, 13. Suitable datasets were required to be publically available, to assess whole-genome gene expression within carotid plaque biopsies from patients both with and without symptoms of cerebral embolization, and to use commercially available microarrays of the same chip platform. The publically available raw array data were downloaded from Gene Expression Omnibus (GEO) provided by the National Center for Biotechnology Information (NCBI) or ArrayExpress provided by the European Molecular Biology Laboratory-European Bioinformatics Institute (EMBL-EBI) public repositories.
Re-analysis of the original array data
In order to identify differentially expressed genes in carotid atherosclerotic tissue between symptomatic and asymptomatic patients, we analysed each original microarray dataset as described previously16. Briefly, the raw data matrix downloaded from GEO or ArrayExpress public repositories was uploaded into GeneSpring GX 11.5 (Agilent Technologies Pty Ltd) and the standard normalization procedures recommended for the Affymetrix GeneChip arrays was followed. Expression values were normalized using percentile shift normalization with default settings. These included normalization to 75th percentile. The expression profile of carotid atherosclerosis samples obtained from participants with symptoms of cerebral embolization was compared to those without symptoms. Since all samples represented advanced atherosclerotic tissue, only small differences in gene expression were expected between patients with and without symptoms of cerebral embolization. Previous evidence suggested that small changes in gene expression are able to induce significant phenotypic differences17. In line with this, genes with ≥1.15-absolute fold differential expression between groups based on P-value < 0.05 by non-parametric Mann–Whitney U-test with no mathematical correction for multiple testing were considered to be differentially expressed.
Validation of microarrays findings
Using total RNA obtained from atherosclerosis removed from the proximal internal carotid (PIC) arteries of 8 patients with and 7 patients without symptoms of cerebral embolization, we assessed the validity of microarray findings (validation group). Total RNA was extracted from PIC biopsies stored in RNAlater (Sigma-Aldrich) at −80 °C using RNeasy Mini Kit (Qiagen) according to manufacturer’s instructions. Symptomatic patients presented with focal neurological symptoms related to their anterior cerebral circulation such as transient ischemic attack (TIA) or stroke within 6 weeks of surgery; asymptomatic patients presented with no history of neurological symptoms1. Quantitative real-time reverse transcription PCR (qRT-PCR) assay was performed to assess the relative expression of arylsulphatase B (ARSB) as this was consistently differential expressed in the array analyses. The relative expression of ARSB in each sample was calculated by using the Concentration-Ct-standard curve method and normalized using the average expression of the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene for each sample using the Rotor-Gene Q operating software version 2.0.24 (Qiagen). GAPDH was chosen as the “housekeeping” gene since analyses showed its expression to be similar in carotid biopsies from symptomatic and asymptomatic patients. The QuantiTect SYBR Green one-step RT-PCR Kit (Qiagen) was used according to the manufacturer’s instructions with 20 ng of total RNA as template. All reactions were independently repeated in duplicate to assess the repeatability of the results and the mean of the two values for each sample was used for analyses. The QuantiTect Primer Assays QT00026684 and QT00079247 (Qiagen) were used for the ARSB and GAPDH assessments, respectively. Mann–Whitney U test was performed to identify differences in expression levels of ARSB between carotid atherosclerosis biopsies of patients with and without symptoms of cerebral embolization. Data were presented as box-and-whisker plots with median and interquartile range with maximum and minimum data points (whiskers) for relative expression. Statistical significance was defined at the conventional 5% level. All computations were performed using the Stata/MP 13.1 statistical software (StataCorp LP, USA). Ethical approval was granted from The Townsville Hospital (TTH) and Health Services Committees, written informed consent was obtained from each participant, and the protocols conformed to the ethical guidelines of the Declaration of Helsinki.
Cell culture
We investigated the effects of chondroitin sulphate, a natural substrate for ARSB, and L-ascorbic acid, an ARSB inhibitor, on the expression of sirtuin 1 (SIRT1) and protein kinase AMP-activated catalytic subunit alpha 1 (PRKAA1 or AMPK, 5′-prime-AMP-activated protein kinase) under inflammatory conditions in vitro. These two ARSB-associated bioactive molecules were specifically selected to test their individual and combined ability to upregulate the SIRT1/AMPK metabolic pathway that exerts strong anti-inflammatory, anti-atherogenic, and plaque-stabilizing effects18, 19. We used human vascular smooth muscle cells (VSMC; Clonetics) that were plated at a seeding density of 1 × 105 cells/well and maintained at 37 °C, 5% CO2, in Dulbecco’s Modified Eagle Medium (DMEM; Sigma-Aldrich) containing 10% fetal bovine serum (FBS; Gibco). Cells were growth arrested at 90% confluency by incubation in DMEM + 0.1% FBS overnight (18 hours). Control cultures (n = 6) were exposed to DMEM + 10% FBS comprising 10% v/v conditioned media generated from human monocytic THP-1 cells exposed to 10 µg/ml endotoxin (Lipopolysaccharide; Sigma-Aldrich) over a period of 24 hours. Experimental cultures were exposed to the same pro-inflammatory media but supplemented with either L-ascorbic acid (Sigma-Aldrich; 400 μM; n = 6 cultures) or chondroitin sulphate sodium salt (Sigma-Aldrich; 300 μM; n = 6 cultures), or a combination of both (n = 6 cultures). All cells were harvested after a 24-hour experimental period and subjected to RNA extraction followed by qRT-PCR using the Qiagen’s QuantiTect Primer Assay QT00009436 (AMPK) and QT00051261 (SIRT1) as outlined above.
Results
Datasets characteristics
Two whole-genome gene expression datasets were included in this study to determine the consensus set of differentially expressed genes in carotid atherosclerosis associated with the symptoms of cerebral embolization (Table 1). The E-MEXP-2257 dataset utilized 22 carotid plaque biopsies obtained from 13 and 9 patients with and without symptoms of cerebral embolization, respectively. The summary characteristics of participants included in the E-MEXP-2257 dataset are presented elsewhere12. In brief, the mean age of patients was 64 ± 8 years, ~27% of participants were females (6/22), and ~27% of participants (6/22) had a positive history of smoking (Table 1). The second dataset included in this study, GSE21545, utilized 126 carotid plaque biopsies obtained from 25 and 101 symptomatic and asymptomatic patients, respectively (Table 1). The summary characteristics of participants included in the GSE21545 dataset are presented elsewhere13. Briefly, the mean age of patients was 71 ± 9 years, ~22% of participants were females (28/126), and ~49% of participants (62/126) had a positive history of smoking (Table 1). No individual patient’s clinical characteristics, including medication and severity of carotid atherosclerosis, were publically available for both datasets.
Table 1.
Characteristics of two microarray datasets included in this study.
| Data set | E-MEXP-2257 | GSE21545 |
|---|---|---|
| Reference | 12 | 13 |
| Technology used | Affymetrix GeneChip HG-U133A | Affymetrix GeneChip HG-U133 Plus 2 |
| Number of transcripts analysed | 18,400 | 47,000 |
| Sample analysed | Carotid plaque biopsies | Carotid plaque biopsies |
| Number of patients | 22 | 126 |
| Number of symptomatic patients | 13 (59%) | 25 (20%) |
| Age (years) | 64 ± 8 | 71 ± 9 |
| Number of females | 6 (27%) | 28 (22%) |
| Number of current or previous smokers | 6 (27%) | 62 (49%) |
Age, calendar age at entry-to-study presented as mean ± standard deviation (SD).
Numerical assessment of differentially expressed genes
The E-MEXP-2257 and GSE21545 microarray datasets were individually re-analysed to identify differentially expressed genes and the overlap between the findings. A total of 1,624 and 135 genes were found to be differentially expressed (≥1.15-absolute fold change, uncorrected P < 0.05) within carotid plaques of symptomatic compared to asymptomatic patients in E-MEXP-2257 and GSE21545, respectively (Fig. 1). Full lists of differentially expressed genes are given in Supplemental Table I and Supplemental Table II for E-MEXP-2257 and GSE21545, respectively. Although E-MEXP-2257 and GSE21545 collectively identified 1,752 differentially expressed individual genes, only 7 genes or 0.4% were consistently differentially expressed in the two datasets (7/1,752; Fig. 1).
Figure 1.
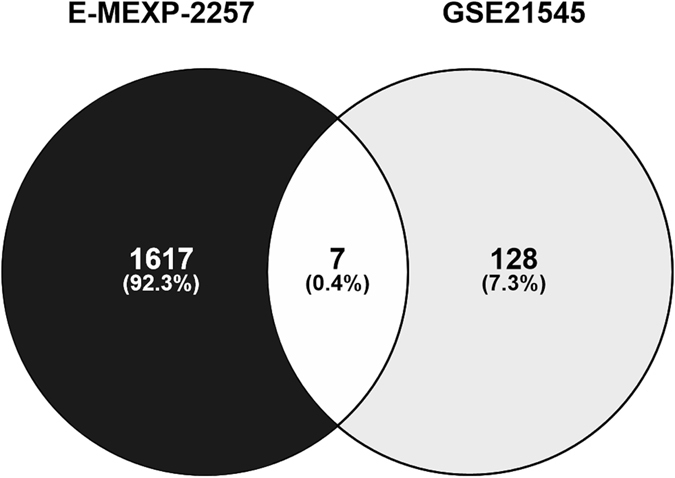
Numerical assessment of genes for which the expression in carotid atherosclerosis was associated with the symptoms of cerebral embolization. Venn diagram illustrating the overlap between the E-MEXP-2257 and GSE21545 microarray datasets profiling differentially expressed genes within carotid plaque biopsies. Samples obtained from patients with symptoms of cerebral embolization were compared with those of patients without the symptoms (≥1.15-absolute fold difference, P < 0.05 without mathematical correction for multiple comparisons calculated with non-parametric Mann-Whitney test).
Assessment of consistently differentially expressed genes
Inspection of the differentially expressed genes revealed that the arylsulfatase B (ARSB) gene was the only gene consistently upregulated in symptomatic compared to asymptomatic patients identified in both E-MEXP-2257 (1.15-fold change, P = 0.039) and GSE21545 (1.15-fold change, P = 0.030) datasets (Table 2). The coagulation factor III, tissue factor (F3) gene and the ubiquitin like modifier activating enzyme 6 (UBA6) gene were found to be upregulated in symptomatic compared to asymptomatic patients in the E-MEXP-2257 dataset but downregulated in symptomatic compared to asymptomatic patients in the GSE21545 dataset (Table 2). The growth arrest specific 6 (GAS6) gene, the uncharacterized LOC730101 (LOC730101), and the secretory leukocyte peptidase inhibitor (SLPI) gene were identified to be downregulated in symptomatic compared to asymptomatic patients in both datasets (Table 2). Finally, the G protein-coupled receptor 135 (GPR135) gene was found to be downregulated in symptomatic compared to asymptomatic patients in the E-MEXP-2257 dataset but upregulated in symptomatic compared to asymptomatic patients in the GSE21545 dataset (Table 2).
Table 2.
Genes for which the expression in carotid atherosclerosis was consistently associated with the symptoms of cerebral embolization in the two microarray datasets included in this study.
| Gene symbol | Gene name | E-MEXP-2257 dataset | GSE21545 dataset | ||||
|---|---|---|---|---|---|---|---|
| Fold change | Regulation | P-value | Fold change | Regulation | P-value | ||
| ARSB | arylsulfatase B | 1.15 | Up | 0.039 | 1.15 | Up | 0.030 |
| F3 | coagulation factor III, tissue factor | 1.26 | Up | 0.019 | −1.20 | Down | 0.035 |
| GAS6 | growth arrest specific 6 | −1.42 | Down | 0.011 | −1.18 | Down | 0.032 |
| GPR135 | G protein-coupled receptor 135 | −1.21 | Down | 0.004 | 1.17 | Up | 0.012 |
| LOC730101 | uncharacterized LOC730101 | −1.16 | Down | 0.006 | −1.20 | Down | 0.025 |
| SLPI | secretory leukocyte peptidase inhibitor | −1.27 | Down | 0.013 | −1.35 | Down | 0.018 |
| UBA6 | ubiquitin like modifier activating enzyme 6 | 1.20 | Up | 0.039 | −1.39 | Down | 0.032 |
P-value, calculated with non-parametric Mann-Whitney test without mathematical correction for multiple comparisons.
Validation of microarray findings
The validity of microarray findings was further assessed using carotid atherosclerosis biopsies obtained from 8 symptomatic and 7 asymptomatic patients undergoing carotid endarterectomy (validation group; Table 3). The risk factors and medications of symptomatic and asymptomatic patients were similar (Table 3). The relative expression of ARSB, the only gene consistently upregulated in symptomatic compared to asymptomatic patients in both microarray datasets, was also found to be significantly increased within the carotid atherosclerotic tissue of symptomatic compared to asymptomatic patients of the validation group using qRT-PCR (*P = 0.029; Fig. 2).
Table 3.
Risk factors and medication of patients with and without symptoms of cerebral embolization included in the validation group.
| Characteristic | Symptomatic patients | Asymptomatic patients | P-value |
|---|---|---|---|
| Number of patients | 8 | 7 | — |
| Age (years) | 72 ± 11 | 72 ± 5 | 0.779 |
| Number of females | 2 (25%) | 1 (14%) | 0.677 |
| Number of current or previous smokers | 7 (88%) | 6 (86%) | 0.933 |
| Type 2 diabetes | 3 (38%) | 1 (14%) | 0.390 |
| Hypertension | 6 (75%) | 5 (71%) | 0.962 |
| Ischemic heart disease | 3 (38%) | 3 (43%) | 0.853 |
| Dyslipidaemia | 5 (63%) | 4 (57%) | 0.853 |
| Statins | 5 (63%) | 3 (43%) | 0.505 |
| Fibrates | 0 (0%) | 2 (29%) | 0.200 |
| Angiotensin converting enzyme inhibitors | 2 (25%) | 5 (71%) | 0.109 |
| Angiotensin receptor blockers | 2 (25%) | 1 (14%) | 0.462 |
Nominal variables are presented as numbers; continuous variables are presented as mean ± standard deviation. Two-sided P value calculated by Mann Whitney U test (continuous variables) or Fisher’s exact test (nominal variables).
Figure 2.
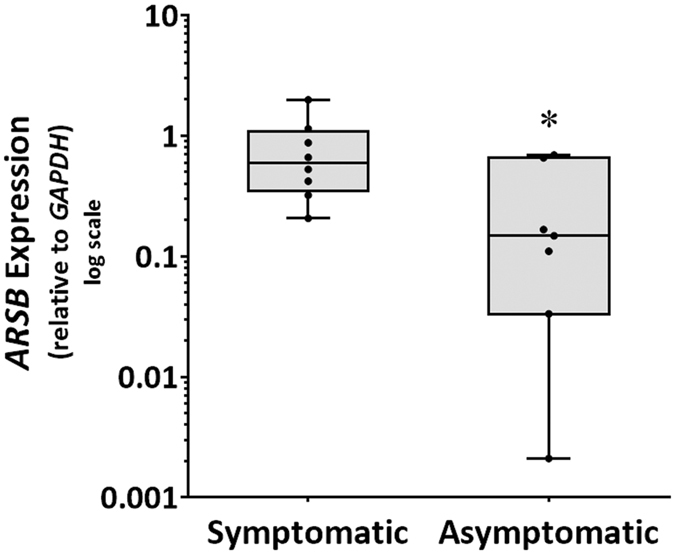
Expression of the ARSB gene within carotid atherosclerosis of patients with and without symptoms of cerebral embolization. Increased expression of ARSB within carotid atheroma biopsies of symptomatic (N = 8) compared to asymptomatic (N = 7) patients (*P = 0.029). Data expressed as median and interquartile range with maximum and minimum data points (whiskers) for relative expression and compared by Mann-Whitney U test.
Cell culture
Finally, we investigated the effect of chondroitin sulphate (ARSB substrate) and L-ascorbic acid (ARSB inhibitor) on important anti-atherogenic pathways represented by the AMPK and SIRT1 genes using human VSMC in vitro under inflammatory conditions. We found that incubation of VSMC with L-ascorbic acid was associated with upregulation of both AMPK (Fig. 3A) and SIRT1 (Fig. 3B). The incubation of VSMC with chondroitin sulphate was associated with upregulation of SIRT1 (Fig. 3B) but not AMPK (Fig. 3A). Importantly, the simultaneous incubation of VSMC with chondroitin sulphate and L-ascorbic acid resulted in synergistic upregulation of AMPK (Fig. 3A) and additive upregulation of SIRT1 (Fig. 3B). These findings suggest that the combination of chondroitin sulphate and L-ascorbic acid may represent a potent activator of the AMPK/SIRT1 pathways.
Figure 3.
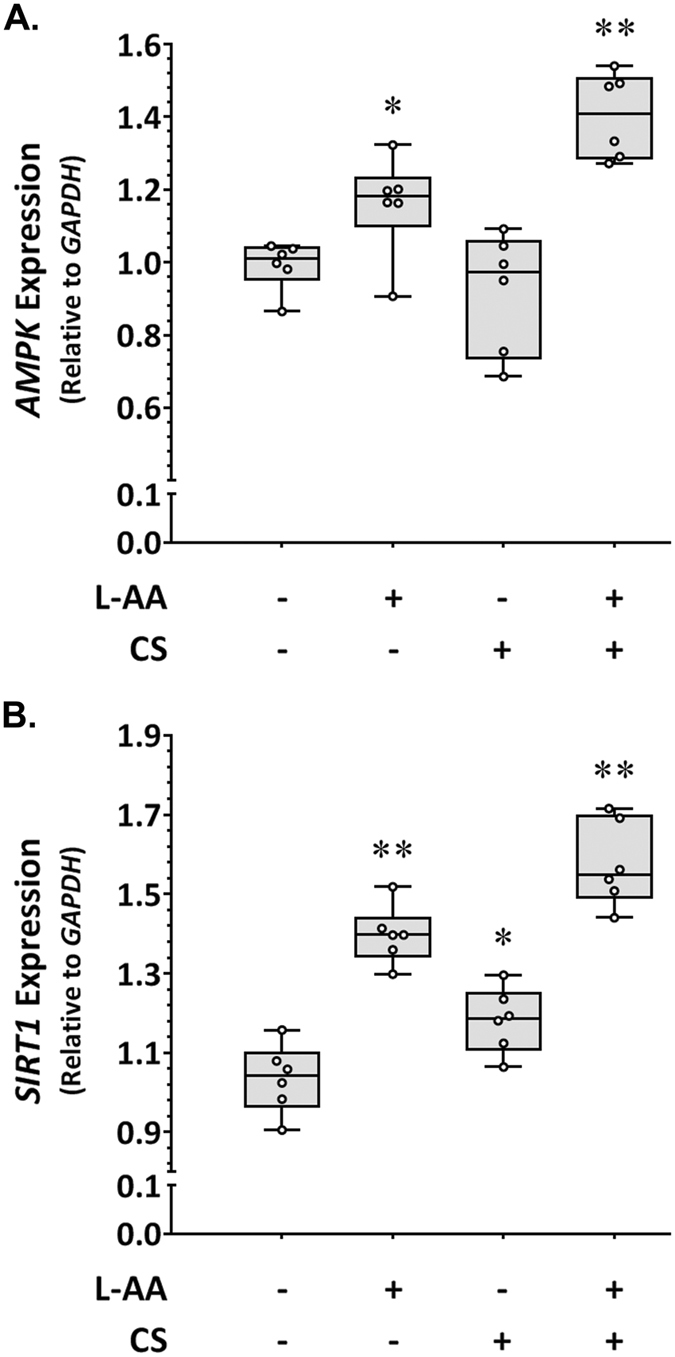
Effect of L-ascorbic acid and chondroitin sulphate on AMPK and SIRT1 expression in inflammation-activated VSMC in vitro. Upregulation of AMPK (A) (*P = 0.041, **P = 0.002) and SIRT1 (B) (*P = 0.015, **P = 0.002) in VSMCs incubated with L-ascorbic acid (L-AA) and chondroitin sulphate (CS) compared to control. Data expressed as median and interquartile range with maximum and minimum data points (whiskers) for relative expression and compared by Mann-Whitney U test.
Discussion
We analysed publically available microarray datasets from previous gene expression studies that utilised carotid plaque biopsies from patients with and without symptoms of cerebral embolization12, 13. By focusing on genes that were simultaneously differentially expressed in the datasets analysed, we were able to discover and validate the upregulation of ARSB in carotid plaques of symptomatic compared to asymptomatic patients, not acknowledged in the original microarray studies. It is important to note that the protein product of this gene is involved in degradative processes of sulphated proteoglycans, the major component of virtually all extracellular matrices (ECMs)20. Furthermore, previous data suggest an inverse association between enzymatic activity of ARSB and the stability of sulphated proteoglycans within the ECM21. In line with this, Koledgie et al. found that plaque rupture sites contain very little proteoglycan content relative to stable lesions22, consistent with a degradative process.
Although the upregulation of ARSB within the carotid plaque biopsies of symptomatic patients identified in both microarray datasets was relatively small, we were able to confirm these findings by qRT-PCR using carotid plaque biopsies obtained from another group of symptomatic and asymptomatic patients. This led us to hypothesize that upregulation of ARSB may represent an important pathological mechanism associated with symptoms of cerebral embolization, consistent with previous findings that even small changes in gene expression can induce major phenotypic differences17. The ARSB enzyme catalyses de-sulphation of ubiquitous glycosaminoglycans such as chondroitin sulphate23. Published evidence suggests that plasma concentration of under-sulphated chondroitin is elevated in patients with carotid artery disease24, while sulphated chondroitin has been long known to exhibit anti-atherogenic properties in rodents, primates, and humans25–28. Several historical studies from the 1960s and 1970s report reduced incidence of coronary events and cardiovascular mortality in atherosclerotic subjects treated with chondroitin sulphate28–30. Recent data suggests that anti-atherogenic actions of chondroitin sulphate may occur through interfering with the pro-inflammatory activation of monocytes and endothelial cells by tumor necrosis factor (TNF) alpha31, a cytokine thought to be crucially involved in the pathogenesis of atherosclerotic plaque32. Although the authors did not elucidate the exact mechanism of action of chondroitin sulphate31, it is possible that upregulation of anti-inflammatory microRNAs, the negative regulators of gene expression, could play a role33–35. Previous studies suggest an inhibitory effect of chondroitin sulphates on gene expression through modification of microRNAs36. Importantly, the ARSB enzyme is inhibited by L-ascorbic acid21, 37. Due to the lack of a well-developed animal model of carotid atherosclerosis associated with cerebral embolization, we further investigated the effect of L-ascorbic acid and chondroitin sulphate on important anti-atherogenic pathways in vitro. We found that chondroitin sulphate and L-ascorbic acid administered together induced a remarkable upregulation in the expression of SIRT1 and AMPK genes in VSMCs exposed to inflammatory conditions in vitro. These findings suggest that chondroitin sulphate formulated with L-ascorbic acid may serve as a potent activator of the SIRT1/AMPK pathway. This may hold promise as a novel therapeutic approach for carotid atherosclerosis since the SIRT1/AMPK pathway is key to a number of vasculoprotective processes. In particular, SIRT1 is the nicotinamide adenosine dinucleotide (NAD)-dependent deacetylase that has been associated with inhibition of the proatherogenic VSMC foam cell formation possibly through the suppressing of the nuclear factor kappa B (NF-κB) signalling pathway38. AMPK is the main energy-sensing kinase in all eukaryotic cells and has been implicated in stabilizing atherosclerotic plaques through the inhibition of the mammalian target of rapamycin (mTOR) signalling pathway39. The downregulation of genes for secretory leukocyte peptidase inhibitor (SLPI), uncharacterized LOC730101, and growth arrest specific 6 (GAS6) in carotid atherosclerosis associated with the symptoms of cerebral embolization in both datasets included in this study was also demonstrated. The role of SLPI and LOC730101 in human carotid atherosclerosis is largely unknown. Some evidence suggests that more severe atherosclerosis in humans is associated with an increase in GAS6 expression40, while similar expression of GAS6 in human carotid arteries with and without atherosclerosis has been reported41. Further investigation of the role of these three genes in carotid atherosclerosis is needed.
The limitations of this study include the relatively small number of patients included in the original microarray datasets. In particular, although both datasets collectively included 148 carotid atherosclerosis tissue samples, the total number of 38 ischemic events was relatively small and findings need to be substantiated by larger studies. We also observed very limited overlap and consistency between the genes differentially expressed in the two datasets suggesting the heterogeneous nature of the patients investigated. Further genome-wide gene expression studies involving histologically standardized sets of patients are needed. In view of these limitations we sought to validate important microarrays finding using another set of carotid artery biopsies obtained from patients with and without symptoms of cerebral embolization. The assessment of independent samples helps to minimize the possibility that selection biases adversely affected the generalizability of the findings. We did, however, only assess mRNA not protein levels due to limited availability of carotid artery biopsies. Finally, the exact mechanism by which chondroitin sulphate combined with L-ascorbic acid upregulated the SIRT1/AMPK pathway as well as the therapeutic doses of these two bioactive molecules remains to be elucidated.
In conclusion, a decade after the first microarrays for stroke, its molecular determinants are still poorly understood. The outcome of this study highlight a potential role for arylsulfatase B in promoting atherosclerosis-related stroke and warrants its further investigation as a therapeutic target that could be of potential clinical benefit.
Electronic supplementary material
Acknowledgements
The Townsville Private Practice Trust Fund (RG04213), Australia, The National Health and Medical Research Council (1000967) and The Queensland Government supported this work. JG holds a Practitioner Fellowship from the National Health and Medical Research Council, Australia (1117061), and a Senior Clinical Research Fellowship from the Health and Medical Research Office, Queensland Government. We would like to acknowledge the Hunter Medical Research Institute Stroke Grant made possible by the Delara Foundation who provided support for this research.
Author Contributions
The authors contributed to the manuscript as follows: E.B., C.L., and J.G. designed the study; E.B., C.S.M., and J.G. wrote the manuscript; E.B. performed microarray and gene expression analysis and contributed to the statistical analysis; J.G. provided tissue samples; C.S.M. performed cell culture work and contributed to the statistical analysis; E.H. contributed to the microarray analysis; J.M., E.H., and C.L. contributed to the preparation of the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-04497-9
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Golledge J, Greenhalgh RM, Davies AH. The symptomatic carotid plaque. Stroke. 2000;31:774–781. doi: 10.1161/01.STR.31.3.774. [DOI] [PubMed] [Google Scholar]
- 2.Dodick DW, Meissner I, Meyer FB, Cloft HJ. Evaluation and management of asymptomatic carotid artery stenosis. Mayo. Clin. Proc. 2004;79:937–944. doi: 10.4065/79.7.937. [DOI] [PubMed] [Google Scholar]
- 3.Lanzino G, Tallarita T, Rabinstein AA. Internal carotid artery stenosis: natural history and management. Semin. Neurol. 2010;30:518–575. doi: 10.1055/s-0030-1268864. [DOI] [PubMed] [Google Scholar]
- 4.Golledge J, Siew DA. Identifying the carotid ‘high risk’ plaque: is it still a riddle wrapped up in an enigma? Eur. J. Vasc. Endovasc. Surg. 2008;35:2–8. doi: 10.1016/j.ejvs.2007.09.004. [DOI] [PubMed] [Google Scholar]
- 5.Mughal MM, et al. Symptomatic and asymptomatic carotid artery plaque. Expert. Rev. Cardiovasc. Ther. 2011;9:1315–1330. doi: 10.1586/erc.11.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Moore DF, et al. Using peripheral blood mononuclear cells to determine a gene expression profile of acute ischemic stroke: a pilot investigation. Circulation. 2005;111:212–221. doi: 10.1161/01.CIR.0000152105.79665.C6. [DOI] [PubMed] [Google Scholar]
- 7.Vemuganti R, Dempsey RJ. Carotid atherosclerotic plaques from symptomatic stroke patients share the molecular fingerprints to develop in a neoplastic fashion: a microarray analysis study. Neuroscience. 2005;131:359–374. doi: 10.1016/j.neuroscience.2004.08.058. [DOI] [PubMed] [Google Scholar]
- 8.Papaspyridonos M, et al. Novel candidate genes in unstable areas of human atherosclerotic plaques. Arterioscler. Thromb. Vasc. Biol. 2006;26:1837–1844. doi: 10.1161/01.ATV.0000229695.68416.76. [DOI] [PubMed] [Google Scholar]
- 9.Ijäs P, et al. Microarray analysis reveals overexpression of CD163 and HO-1 in symptomatic carotid plaques. Arterioscler. Thromb. Vasc. Biol. 2007. 2007;27:154–160. doi: 10.1161/01.ATV.0000251991.64617.e7. [DOI] [PubMed] [Google Scholar]
- 10.Agardh HE, et al. Expression of fatty acid-binding protein 4/aP2 is correlated with plaque instability in carotid atherosclerosis. J. Intern. Med. 2011;269:200–210. doi: 10.1111/j.1365-2796.2010.02304.x. [DOI] [PubMed] [Google Scholar]
- 11.Razuvaev A, et al. Correlations between clinical variables and gene-expression profiles in carotid plaque instability. Eur. J. Vasc. Endovasc. Surg. 2011;42:722–730. doi: 10.1016/j.ejvs.2011.05.023. [DOI] [PubMed] [Google Scholar]
- 12.Saksi J, et al. Gene expression differences between stroke-associated and asymptomatic carotid plaques. J. Mol. Med. (Berl). 2011;89:1015–1026. doi: 10.1007/s00109-011-0773-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Folkersen L, et al. Prediction of ischemic events on the basis of transcriptomic and genomic profiling in patients undergoing carotid endarterectomy. Mol. Med. 2012;18:669–675. doi: 10.2119/molmed.2011.00479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Perisic L, et al. Profiling of atherosclerotic lesions by gene and tissue microarrays reveals PCSK6 as a novel protease in unstable carotid atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2013;33:2432–2443. doi: 10.1161/ATVBAHA.113.301743. [DOI] [PubMed] [Google Scholar]
- 15.Salem MK, et al. Histologically unstable asymptomatic carotid plaques have altered expression of genes involved in chemokine signalling leading to localised plaque inflammation and rupture. Eur. J. Vasc. Endovasc. Surg. 2013;45:121–127. doi: 10.1016/j.ejvs.2012.11.006. [DOI] [PubMed] [Google Scholar]
- 16.Biros E, et al. Differential gene expression in human abdominal aortic aneurysm and aortic occlusive disease. Oncotarget. 2015;6:12984–12996. doi: 10.18632/oncotarget.3848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ruzycki PA, et al. Graded gene expression changes determine phenotype severity in mouse models of CRX-associated retinopathies. Genome Biol. 2015;16:171. doi: 10.1186/s13059-015-0732-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Winnik S, Auwerx J, Sinclair DA, Matter CM. Protective effects of sirtuins in cardiovascular diseases: from bench to bedside. Eur. Heart. J. 2015;36:3404–3412. doi: 10.1093/eurheartj/ehv290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ding Y, et al. AMP-activated protein kinase alpha 2 deletion induces VSMC phenotypic switching and reduces features of atherosclerotic plaque stability. Circ. Res. 2016;119:718–730. doi: 10.1161/CIRCRESAHA.116.308689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yanagishita M. Function of proteoglycans in the extracellular matrix. Acta. Pathol. Jpn. 1993;43:283–293. doi: 10.1111/j.1440-1827.1993.tb02569.x. [DOI] [PubMed] [Google Scholar]
- 21.Schwartz ER, Adamy L. Effect of ascorbic acid on arylsulfatase activities and sulfated proteoglycan metabolism in chondrocyte cultures. J. Clin. Invest. 1977;60:96–106. doi: 10.1172/JCI108774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kolodgie FD, et al. Differential accumulation of proteoglycans and hyaluronan in culprit lesions: insights into plaque erosion. Arterioscler. Thromb. Vasc. Biol. 2002;22:1642–1648. doi: 10.1161/01.ATV.0000034021.92658.4C. [DOI] [PubMed] [Google Scholar]
- 23.Gorham SD, Cantz M. Arylsulphatase B, an exo-sulphatase for chondroitin 4-sulphate tetrasaccharide. Hoppe. Seylers. Z. Physiol. Chem. 1978;359:1811–1814. doi: 10.1515/bchm2.1978.359.2.1811. [DOI] [PubMed] [Google Scholar]
- 24.Zinellu E, et al. Association between human plasma chondroitin sulfate isomers and carotid atherosclerotic plaques. Biochem. Res. Int. 2012;2012:281284. doi: 10.1155/2012/281284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Morrison LM, Bernick S, Alfin-Slater RB, Patek PR, Ershoff BH. Inhibition of coronary atherosclerosis in the x-irradiated, cholesterol-fed rat by chondroitin sulfate A. Proc. Soc. Exp. Biol. Med. 1966;123:904–911. doi: 10.3181/00379727-123-31635. [DOI] [PubMed] [Google Scholar]
- 26.Morrison LM, Murata K, Quilligan JJ, Jr., Schjeide OA, Freeman L. Prevention of atherosclerosis in sub-human primates by chondroitin sulfate. A. Circ. Res. 1966;19:358–363. doi: 10.1161/01.RES.19.2.358. [DOI] [PubMed] [Google Scholar]
- 27.Nakazawa K, Murata K. The therapeutic effect of chondroitin polysulphate in elderly atherosclerotic patients. J. Int. Med. Res. 1978;6:217–225. doi: 10.1177/030006057800600309. [DOI] [PubMed] [Google Scholar]
- 28.Nakazawa K, Murata K. Comparative study of the effects of chondroitin sulfate isomers on atherosclerotic subjects. Z. Alternsforsch. 1979;34:153–159. [PubMed] [Google Scholar]
- 29.Morrison LM. Response of ischemic heart disease to chondroitin sulfate-A. J. Am. Geriatr. Soc. 1969;17:913–923. doi: 10.1111/j.1532-5415.1969.tb02328.x. [DOI] [PubMed] [Google Scholar]
- 30.Morrison LM, Enrick N. Coronary heart disease: reduction of death rate by chondroitin sulfate A. Angiology. 1973;24:269–287. doi: 10.1177/000331977302400503. [DOI] [PubMed] [Google Scholar]
- 31.Melgar-Lesmes P, et al. Treatment with chondroitin sulfate to modulate inflammation and atherogenesis in obesity. Atherosclerosis. 2016;245:82–87. doi: 10.1016/j.atherosclerosis.2015.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kleinbongard P, Heusch G, Schulz R. TNFalpha in atherosclerosis, myocardial ischemia/reperfusion and heart failure. Pharmacol. Ther. 2010;127:295–314. doi: 10.1016/j.pharmthera.2010.05.002. [DOI] [PubMed] [Google Scholar]
- 33.Santulli G, et al. A selective microRNA-based strategy inhibits restenosis while preserving endothelial function. J Clin Invest. 2014;124:4102–4114. doi: 10.1172/JCI76069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Novák J, et al. Mechanistic Role of MicroRNAs in Coupling Lipid Metabolism and Atherosclerosis. Adv Exp Med Biol. 2015;887:79–100. doi: 10.1007/978-3-319-22380-3_5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Santulli G. MicroRNAs and Endothelial (Dys) Function. J Cell Physiol. 2016;231:1638–1644. doi: 10.1002/jcp.25276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang Y, et al. MicroRNAs in the axon locally mediate the effects of chondroitin sulfate proteoglycans and cGMP on axonal growth. Dev Neurobiol. 2015;75:1402–1419. doi: 10.1002/dneu.22292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Selvidge LA, Verlangieri AJ. Inhibition of arylsulfatase B by ascorbic acid. Res. Commun. Chem. Pathol. Pharmacol. 1991;73:253–256. [PubMed] [Google Scholar]
- 38.Zhang MJ, et al. Impaired SIRT1 promotes the migration of vascular smooth muscle cell-derived foam cells. Histochem. Cell. Biol. 2016;146:33–43. doi: 10.1007/s00418-016-1408-9. [DOI] [PubMed] [Google Scholar]
- 39.Martinet W, De Loof H, De Meyer GR. mTOR inhibition: a promising strategy for stabilization of atherosclerotic plaques. Atherosclerosis. 2014;233:601–607. doi: 10.1016/j.atherosclerosis.2014.01.040. [DOI] [PubMed] [Google Scholar]
- 40.Tjwa M, Moons L, Lutgens E. Pleiotropic role of growth arrest-specific gene 6 in atherosclerosis. Curr. Opin. Lipidol. 2009;20:386–392. doi: 10.1097/MOL.0b013e328330982e. [DOI] [PubMed] [Google Scholar]
- 41.Hurtado B, et al. Expression of the vitamin K-dependent proteins GAS6 and protein S and the TAM receptor tyrosine kinases in human atherosclerotic carotid plaques. Thromb. Haemost. 2011;105:873–882. doi: 10.1160/TH10-10-0630. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


