Abstract
Pneumococcal peptidoglycan amidase (N-acetylmuramoyl-L-alanine amidase, EC 3.5.1.28) and phage CPL1 lysozyme degrade a common substrate (choline-containing pneumococcal cell walls); the former hydrolyzes the bond between muramic acid and alanine, whereas the latter breaks down the linkage between muramic acid and glucosamine. The amino acid sequences of their C-terminal domains are homologous. Chimeric genes were constructed by site-directed mutagenesis: a unique SnaBI restriction site in the cpl1 gene, coding for the phage lysozyme, was introduced at a location equivalent to the SnaBI site present in the lytA gene, which codes for the pneumococcal amidase. The resulting genes expressed lytic activities at levels similar to those of the parental genes. The gene products, which have been purified to electrophoretical homogeneity, exhibited unusual combined biochemical properties--e.g., by exchange of protein domains, we have switched the regulatory properties of these enzymes without altering their catalytic activities. Chimeric gene construction in Streptococcus pneumoniae and its bacteriophages is an excellent model to study the modular organization of genes and proteins and to help to establish evolutionary relationships between phage and bacteria. These constructions provide an experimental approach to the molecular processes involved in cassette recruitment during evolution and contribute support to the concept of bacteria as adaptable chimeras.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balganesh T. S., Reiners L., Lauster R., Noyer-Weidner M., Wilke K., Trautner T. A. Construction and use of chimeric SPR/phi 3T DNA methyltransferases in the definition of sequence recognizing enzyme regions. EMBO J. 1987 Nov;6(11):3543–3549. doi: 10.1002/j.1460-2075.1987.tb02681.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berry A. M., Lock R. A., Hansman D., Paton J. C. Contribution of autolysin to virulence of Streptococcus pneumoniae. Infect Immun. 1989 Aug;57(8):2324–2330. doi: 10.1128/iai.57.8.2324-2330.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolivar F. Construction and characterization of new cloning vehicles. III. Derivatives of plasmid pBR322 carrying unique Eco RI sites for selection of Eco RI generated recombinant DNA molecules. Gene. 1978 Oct;4(2):121–136. doi: 10.1016/0378-1119(78)90025-2. [DOI] [PubMed] [Google Scholar]
- Botstein D. A theory of modular evolution for bacteriophages. Ann N Y Acad Sci. 1980;354:484–490. doi: 10.1111/j.1749-6632.1980.tb27987.x. [DOI] [PubMed] [Google Scholar]
- Boyer H. W., Roulland-Dussoix D. A complementation analysis of the restriction and modification of DNA in Escherichia coli. J Mol Biol. 1969 May 14;41(3):459–472. doi: 10.1016/0022-2836(69)90288-5. [DOI] [PubMed] [Google Scholar]
- Briese T., Hakenbeck R. Interaction of the pneumococcal amidase with lipoteichoic acid and choline. Eur J Biochem. 1985 Jan 15;146(2):417–427. doi: 10.1111/j.1432-1033.1985.tb08668.x. [DOI] [PubMed] [Google Scholar]
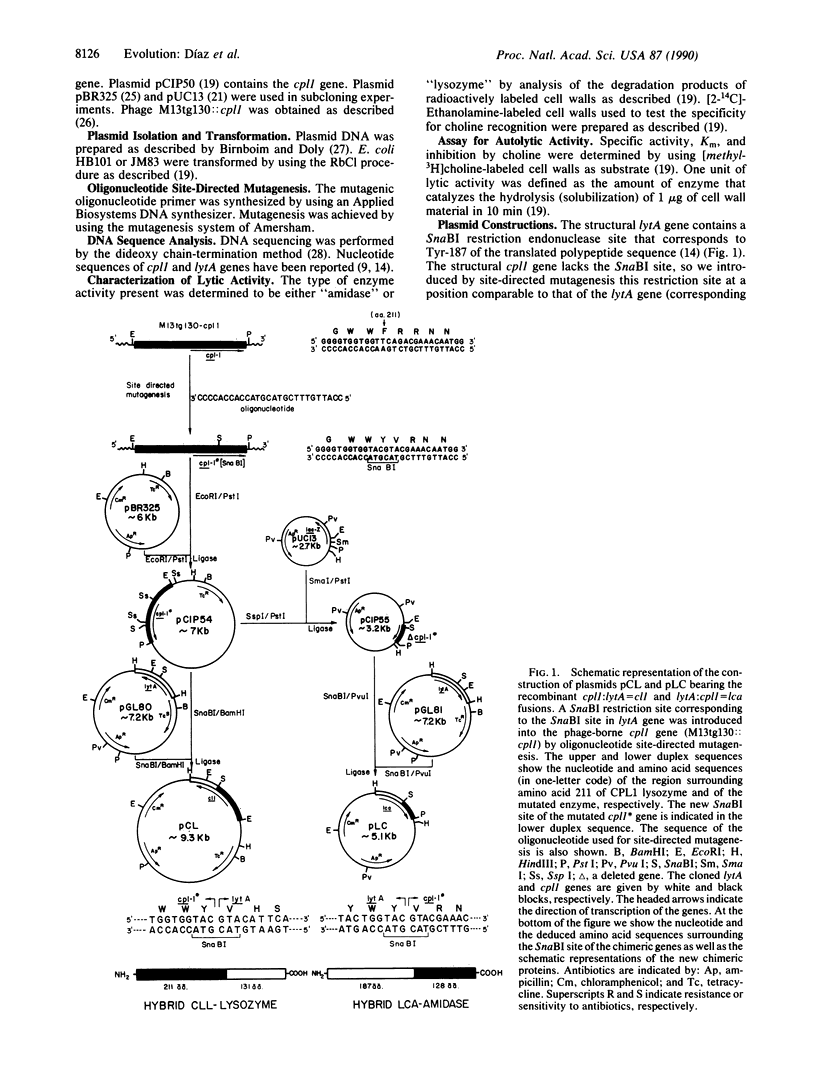
- García E., García J. L., García P., Arrarás A., Sánchez-Puelles J. M., López R. Molecular evolution of lytic enzymes of Streptococcus pneumoniae and its bacteriophages. Proc Natl Acad Sci U S A. 1988 Feb;85(3):914–918. doi: 10.1073/pnas.85.3.914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García J. L., García E., Arrarás A., García P., Ronda C., López R. Cloning, purification, and biochemical characterization of the pneumococcal bacteriophage Cp-1 lysin. J Virol. 1987 Aug;61(8):2573–2580. doi: 10.1128/jvi.61.8.2573-2580.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García J. L., García E., López R. Overproduction and rapid purification of the amidase of Streptococcus pneumoniae. Arch Microbiol. 1987;149(1):52–56. doi: 10.1007/BF00423136. [DOI] [PubMed] [Google Scholar]
- García P., García J. L., García E., López R. Nucleotide sequence and expression of the pneumococcal autolysin gene from its own promoter in Escherichia coli. Gene. 1986;43(3):265–272. doi: 10.1016/0378-1119(86)90215-5. [DOI] [PubMed] [Google Scholar]
- García P., García J. L., García E., López R. Purification and characterization of the autolytic glycosidase of Streptococcus pneumoniae. Biochem Biophys Res Commun. 1989 Jan 16;158(1):251–256. doi: 10.1016/s0006-291x(89)80205-0. [DOI] [PubMed] [Google Scholar]
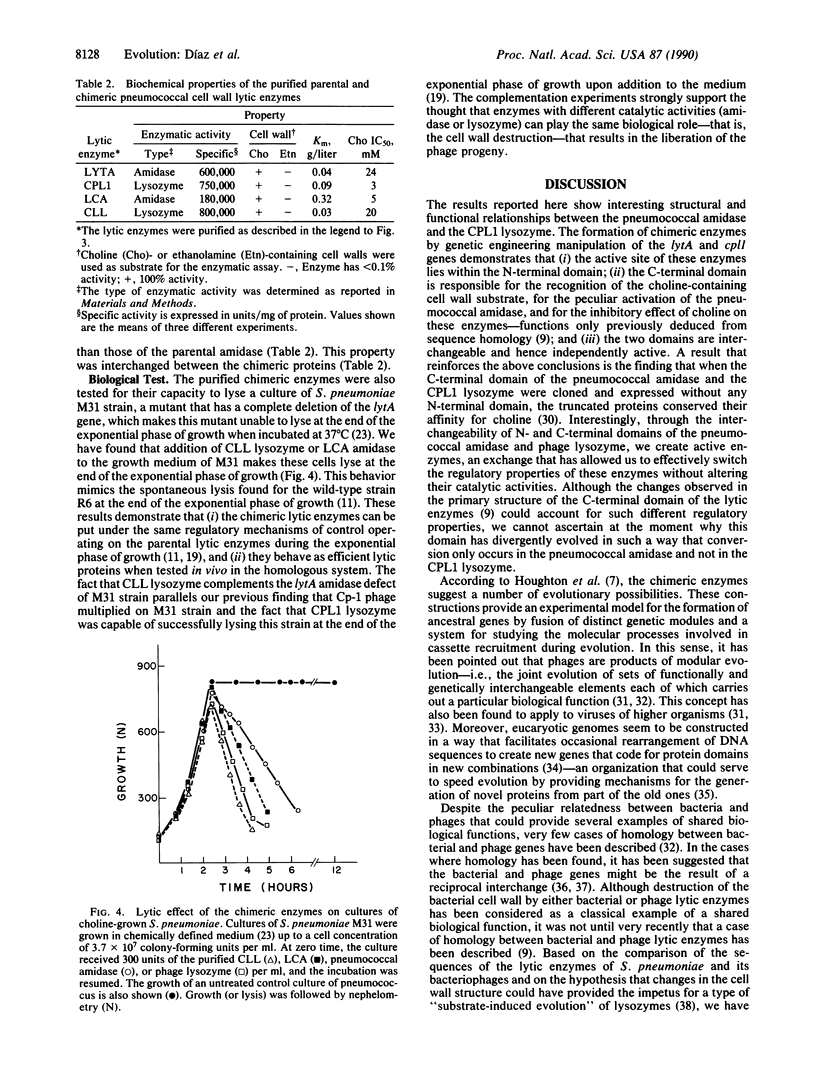
- García P., García J. L., García E., Sánchez-Puelles J. M., López R. Modular organization of the lytic enzymes of Streptococcus pneumoniae and its bacteriophages. Gene. 1990 Jan 31;86(1):81–88. doi: 10.1016/0378-1119(90)90116-9. [DOI] [PubMed] [Google Scholar]
- Gibbs A. Molecular evolution of viruses; 'trees', 'clocks' and 'modules'. J Cell Sci Suppl. 1987;7:319–337. doi: 10.1242/jcs.1987.supplement_7.22. [DOI] [PubMed] [Google Scholar]
- Gilbert W. Why genes in pieces? Nature. 1978 Feb 9;271(5645):501–501. doi: 10.1038/271501a0. [DOI] [PubMed] [Google Scholar]
- Giudicelli S., Tomasz A. Attachment of pneumococcal autolysin to wall teichoic acids, an essential step in enzymatic wall degradation. J Bacteriol. 1984 Jun;158(3):1188–1190. doi: 10.1128/jb.158.3.1188-1190.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houghton J. E., O'Donovan G. A., Wild J. R. Reconstruction of an enzyme by domain substitution effectively switches substrate specificity. Nature. 1989 Mar 9;338(6211):172–174. doi: 10.1038/338172a0. [DOI] [PubMed] [Google Scholar]
- Jollès P., Jollès J. What's new in lysozyme research? Always a model system, today as yesterday. Mol Cell Biochem. 1984 Sep;63(2):165–189. doi: 10.1007/BF00285225. [DOI] [PubMed] [Google Scholar]
- Kobilka B. K., Kobilka T. S., Daniel K., Regan J. W., Caron M. G., Lefkowitz R. J. Chimeric alpha 2-,beta 2-adrenergic receptors: delineation of domains involved in effector coupling and ligand binding specificity. Science. 1988 Jun 3;240(4857):1310–1316. doi: 10.1126/science.2836950. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lauster R., Trautner T. A., Noyer-Weidner M. Cytosine-specific type II DNA methyltransferases. A conserved enzyme core with variable target-recognizing domains. J Mol Biol. 1989 Mar 20;206(2):305–312. doi: 10.1016/0022-2836(89)90480-4. [DOI] [PubMed] [Google Scholar]
- López R., Ronda C., García P., Escarmís C., García E. Restriction cleavage maps of the DNAs of Streptococcus pneumoniae bacteriophages containing protein covalently bound to their 5' ends. Mol Gen Genet. 1984;197(1):67–74. doi: 10.1007/BF00327924. [DOI] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Nelles L., Lijnen H. R., Collen D., Holmes W. E. Characterization of a fusion protein consisting of amino acids 1 to 263 of tissue-type plasminogen activator and amino acids 144 to 411 of urokinase-type plasminogen activator. J Biol Chem. 1987 Aug 5;262(22):10855–10862. [PubMed] [Google Scholar]
- Ronda-Lain C., Lopez R., Tapia A., Tomasz A. Role of the pneumococcal autolysin (murein hydrolase) in the release of progeny bacteriophage and in the bacteriophage-induced lysis of the host cells. J Virol. 1977 Jan;21(1):366–374. doi: 10.1128/jvi.21.1.366-374.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronda C., García J. L., García E., Sánchez-Puelles J. M., López R. Biological role of the pneumococcal amidase. Cloning of the lytA gene in Streptococcus pneumoniae. Eur J Biochem. 1987 May 4;164(3):621–624. doi: 10.1111/j.1432-1033.1987.tb11172.x. [DOI] [PubMed] [Google Scholar]
- Sanchez-Puelles J. M., Ronda C., Garcia J. L., Garcia P., Lopez R., Garcia E. Searching for autolysin functions. Characterization of a pneumococcal mutant deleted in the lytA gene. Eur J Biochem. 1986 Jul 15;158(2):289–293. doi: 10.1111/j.1432-1033.1986.tb09749.x. [DOI] [PubMed] [Google Scholar]
- Sanger F., Coulson A. R., Barrell B. G., Smith A. J., Roe B. A. Cloning in single-stranded bacteriophage as an aid to rapid DNA sequencing. J Mol Biol. 1980 Oct 25;143(2):161–178. doi: 10.1016/0022-2836(80)90196-5. [DOI] [PubMed] [Google Scholar]
- Sanz J. M., Lopez R., Garcia J. L. Structural requirements of choline derivatives for 'conversion' of pneumococcal amidase. A new single-step procedure for purification of this autolysin. FEBS Lett. 1988 May 23;232(2):308–312. doi: 10.1016/0014-5793(88)80759-2. [DOI] [PubMed] [Google Scholar]
- Sonea S. A bacterial way of life. Nature. 1988 Jan 21;331(6153):216–216. doi: 10.1038/331216a0. [DOI] [PubMed] [Google Scholar]
- Stroynowski I. T. Distribution of bacteriophage phi 3T homologous deoxyribonucleic acid sequences in Bacillus subtilis 168, related bacteriophages, and other Bacillus species. J Bacteriol. 1981 Oct;148(1):91–100. doi: 10.1128/jb.148.1.91-100.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sánchez-Puelles J. M., Sanz J. M., García J. L., García E. Cloning and expression of gene fragments encoding the choline-binding domain of pneumococcal murein hydrolases. Gene. 1990 Apr 30;89(1):69–75. doi: 10.1016/0378-1119(90)90207-8. [DOI] [PubMed] [Google Scholar]
- Tomasz A., Westphal M. Abnormal autolytic enzyme in a pneumococus with altered teichoic acid composition. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2627–2630. doi: 10.1073/pnas.68.11.2627. [DOI] [PMC free article] [PubMed] [Google Scholar]