Abstract
The graft-versus-leukemia (GVL) effect in allogeneic hematopoietic stem cell transplantation (alloSCT) is potent against chronic phase chronic myelogenous leukemia (CP-CML), but blast crisis CML (BC-CML) and acute myeloid leukemias (AML) are GVL resistant. To understand GVL resistance, we studied GVL against mouse models of CP-CML, BC-CML, and AML generated by the transduction of mouse BM with fusion cDNAs derived from human leukemias. Prior work has shown that CD4+ T cell–mediated GVL against CP-CML and BC-CML required intact leukemia MHCII; however, stem cells from both leukemias were MHCII negative. Here, we show that CP-CML, BC-CML, and AML stem cells upregulate MHCII in alloSCT recipients. Using gene-deficient leukemias, we determined that BC-CML and AML MHC upregulation required IFN-γ stimulation, whereas CP-CML MHC upregulation was independent of both the IFN-γ receptor (IFN-γR) and the IFN-α/β receptor IFNAR1. Importantly, IFN-γR–deficient BC-CML and AML were completely resistant to CD4- and CD8-mediated GVL, whereas IFN-γR/IFNAR1 double-deficient CP-CML was fully GVL sensitive. Mouse AML and BC-CML stem cells were MHCI+ without IFN-γ stimulation, suggesting that IFN-γ sensitizes these leukemias to T cell killing by mechanisms other than MHC upregulation. Our studies identify the requirement of IFN-γ stimulation as a mechanism for BC-CML and AML GVL resistance, whereas independence from IFN-γ renders CP-CML more GVL sensitive, even with a lower-level alloimmune response.
Keywords: Immunology, Transplantation
Introduction
Allogeneic hematopoietic stem cell transplantation (alloSCT) is the most successful and widely applied form of adoptive T cell immunotherapy. Alloreactive αβ T cells in allografts can recognize and kill recipient leukemia cells, thereby mediating the graft-versus-leukemia (GVL) effect (1). Unfortunately, alloreactive T cells also attack nonmalignant host tissues, causing graft-versus-host disease (GVHD) (2–4). A longstanding and elusive objective has been to develop approaches that preserve GVL while minimizing GVHD. A second and arguably more important goal is to overcome GVL resistance, as relapse of malignant neoplasms is the greatest single cause of post-transplantation mortality (5). GVL resistance and sensitivity are not equal across different types of hematopoietic malignancies. For example, chronic phase chronic myelogenous leukemia (CP-CML) is exquisitely GVL sensitive, whereas blast crisis CML (BC-CML) is relatively GVL resistant, despite the two sharing a common biology, in that both are reliant on breakpoint cluster region/Abelson murine leukemia viral oncogene homolog 1 (BCR/ABL) signaling (6–8). Acute myeloid leukemias (AML) and acute lymphoblastic leukemias are also relatively GVL resistant.
Because GVL resistance and sensitivity track with the identity of the underlying neoplasm, we reasoned that they are leukemia cell–intrinsic properties. To understand the mechanisms of GVL resistance, we used mouse models to evaluate GVL against CP-CML (mCP-CML) and BC-CML (mBC-CML) (9–11). mCP-CML is created via retroviral transfer of the BCR-ABL fusion cDNA, the defining genetic abnormality of CP-CML, into mouse BM cells (12, 13), whereas mBC-CML is created via retroviral transfer of both BCR-ABL and nucleoporin 98–homeobox A9 (NUP98-HOXA9) fusion cDNAs (14, 15). The NUP98-HOXA9 fusion is a second-hit translocation in BC-CML that has also been identified in de novo AML (14–22), as have NUP98 fusions with other class I HOX genes. mCP-CML and mBC-CML are therefore excellent phenocopies and genocopies of their human counterparts, have defined stem cell populations (15, 23), and, importantly, are GVL sensitive and GVL resistant, respectively (11). A powerful advantage of this approach is that, by transducing BM from gene-deficient mice, we can create gene-deficient leukemias as a means to explore mechanisms of GVL resistance (10, 11, 24–26). Using these systems, we found that GVL against mCP-CML and GVL against mBC-CML share essential features: (a) both leukemias must express ICAM1; (b) T cell killing mechanisms are highly redundant; and (c) CD8+ and CD4+ T cell killing requires T cell receptor–MHCI (TCR-MHCI) or TCR-MHCII interactions, respectively (11, 24, 25). Paradoxically, however, despite the fact that MHCII-deficient mCP-CML and mBC-CML were completely resistant to CD4-mediated GVL, WT mCP-CML and mBC-CML leukemia stem cells (LSCs) expressed little to no surface MHCII, as MHCII staining was similar in WT and MHCII-deficient LSCs (11). Here, we report that expression of MHCII and MHCI on mBC-CML and MHCII on mouse AML (mAML; induced by transduction with the MLL-AF9 fusion cDNA) (27) and mCP-CML LSCs is upregulated in the alloimmune environment. Upregulation of MHC on mBC-CML and mAML required IFN-γ receptor (IFN-γR) stimulation, whereas MHC upregulation on mCP-CML LSCs was entirely independent of IFN-γR or IFN-α/β (IFNAR1) signaling. Importantly, IFN-γR–deficient (Ifngr–/–) mBC-CML and mAML were highly resistant to both CD4- and CD8-mediated GVL, whereas Ifngr–/–, Ifngr–/– Ifnar1–/–, and Stat1–/– Stat2–/– mCP-CML were fully GVL sensitive. Our data further suggest that IFN-γ sensitizes myeloblastic leukemias to GVL by mechanisms beyond simply upregulating MHC. The differential requirements for IFN-γ stimulation at least in part explain the exquisite GVL sensitivity of CP-CML and GVL resistance of myeloblastic leukemias and suggest a therapeutic strategy for overcoming the GVL resistance of myeloblastic leukemias.
Results
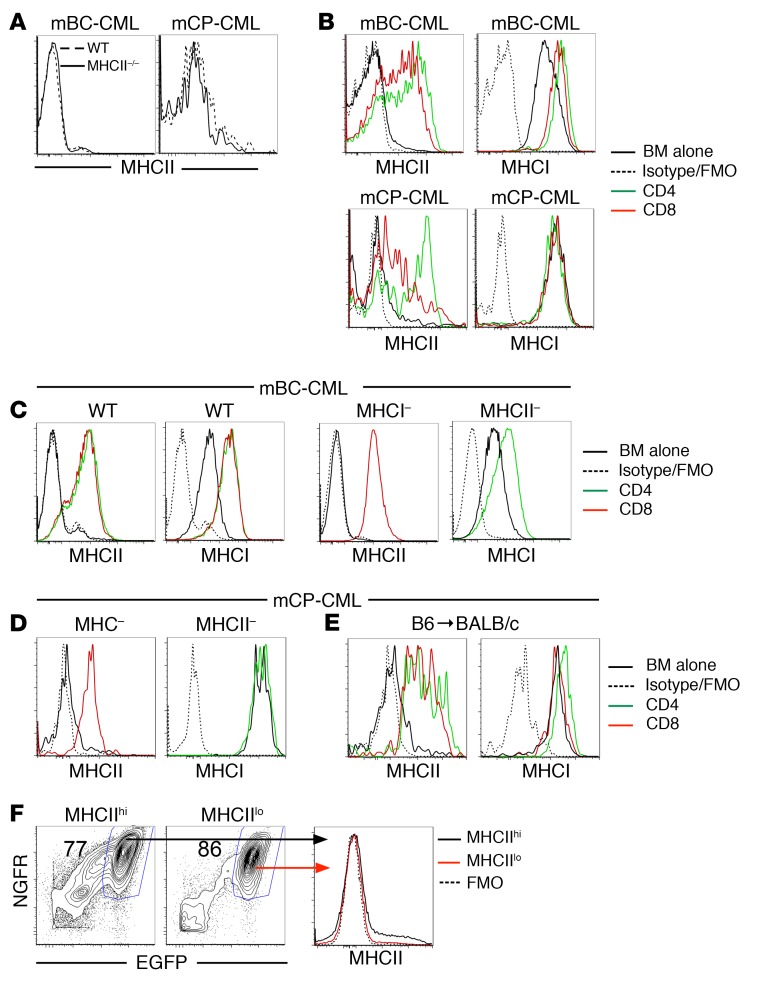
MHCII and MHCI are upregulated on leukemia cells in a GVH environment.
Although MHCII-/- mCP-CML and mBC-CML were completely resistant to CD4-mediated GVL, staining for surface MHCII on WT and MHCII-/- LSCs harvested from sublethally irradiated syngeneic recipients or alloBMT recipients that did not receive donor T cells was similar (ref. 11 and Figure 1A). To account for the MHCII requirement in GVL, we hypothesized that surface MHCII was upregulated in an alloimmune environment. To test this, we analyzed mBC-CML and mCP-CML cells from mice with or without an ongoing GVH response in the C3H.SW→B6 model. MHCII was upregulated on both mBC-CML LSCs (lineage– [lin–]or CD11b–) (ref. 15 and data not shown) and mCP-CML LSCs (lin–sca-1+c-kit+) (ref. 28 and Figure 1B) harvested from mice in which GVH was induced by either CD4 or CD8 cells. We found that MHCI was consistently upregulated on mBC-CML LSCs but minimally and inconsistently so on mCP-CML LSCs (Figure 1B).
Figure 1. Expression of MHC molecules on mCP-CML and mBC-CML LSCs increases in the alloimmune environment, independently of cognate TCR-MHC interactions.
MHCII expression on WT and MHCII–/– mBC-CML (A, left) or mCP-CML (A, right) LSCs harvested from mice transplanted with leukemia cells but without GVH-inducing T cells. (B) Irradiated B6 mice were reconstituted with C3H.SW BM and CD4 or CD8 T cells and either mBC-CML or mCP-CML. Mice were sacrificed between days 10 and 14, and LSCs were analyzed for MHCI and MHCII expression. Representative data from at least 3 independent experiments are shown. (C) Irradiated B6 mice were reconstituted with C3H.SW BM with B6 B2m–/– mBC-CML (MHCI–) and C3H.SW CD8 cells; B6 MHCII–/– mBC-CML (MHCII–) and C3H.SW CD4 cells; or WT B6 mBC-CML and C3H.SW CD4 or CD8 cells. On day 15 after BMT, splenocytes were harvested, and MHCI and MHCII expression on mBC-CML LSCs was assessed. Similar MHC upregulation was noted on LSCs harvested from BM (data not shown). Data are representative of 3 independent experiments. (D) Mice were transplanted as in C, except with B2m–/– or MHCII–/– mCP-CML cells. MHC upregulation was also independent of TCR-MHC interactions. (E) Irradiated BALB/c mice were reconstituted with B6 BM and B6 mCP-CML with no T cells or with B6 CD4 or CD8 cells. MHCII and MHCI were upregulated on splenic mCP-CML LSCs on day 15 after BMT. Similar MHC upregulation was seen in BM LSCs (data not shown). (F) MHCIIhi and MHCIIlo mBC-CML LSCs from mice undergoing a GVHD response (C3H.SW→B6 model with GVH induced by CD4 cells) were sort purified and transferred into sublethally irradiated B6 mice. Both populations transferred disease (F, left panels). Progeny of sorted MHCIIhi and MHCIIlo mBC-CML cells recovered 15 days after transfer were MHCIIlo (F, right panel). FMO, fluorescence minus one.
MHC upregulation does not require TCR-MHC interactions.
Because CD4-mediated GVL absolutely requires both mCP-CML and mBC-CML to express MHCII (10, 11, 24), we reasoned that MHCII upregulation would occur in the GVH environment independently of TCR interactions with MHC on mBC-CML or mCP-CML LSCs. Consistent with this, donor CD8+ T cells induced MHCII upregulation on MHCI-deficient (β-2-microglobulin–deficient, referred to herein as B2m–/–) B6 mBC-CML and mCP-CML, and CD4 cells induced MHCI upregulation on MHCII-deficient B6 mBC-CML cells (Figure 1, C and D). To further investigate the requirement of TCR-MHC interactions for MHC upregulation, we asked whether donor-strain mCP-CML cells in mice undergoing a GVH response would upregulate MHCII. Irradiated BALB/c mice were reconstituted with B6 BM and B6 mCP-CML cells, with or without B6 CD4 or CD8 cells. In this design, donor T cells are activated by host BALB/c cells, but because the T cells are syngeneic with the B6 mCP-CML, they do not mediate GVL and presumably do not have high-avidity TCR-MHC interactions with them. In line with data using MHC-deficient mCP-CML, we observed that MHCII was upregulated on donor mCP-CML LSCs in GVH mice relative to LSCs in mice that did not receive donor T cells (Figure 1E).
MHCIIhi mBC-CML cells can serially transplant leukemia.
To determine whether LSC-phenotype cells induced to express MHCII have functional properties of LSCs, we sort purified MHCIIhiCD11b– and MHCIIloCD11b– mBC-CML cells from mice undergoing a GVH response and transferred them into sublethally irradiated syngeneic B6 mice. Both MHCIIlo and MHCIIhi mBC-CML cells transferred mBC-CML, but the progeny of MHCIIhi cells lost MHCII expression (Figure 1F), indicating that MHCII upregulation was transient.
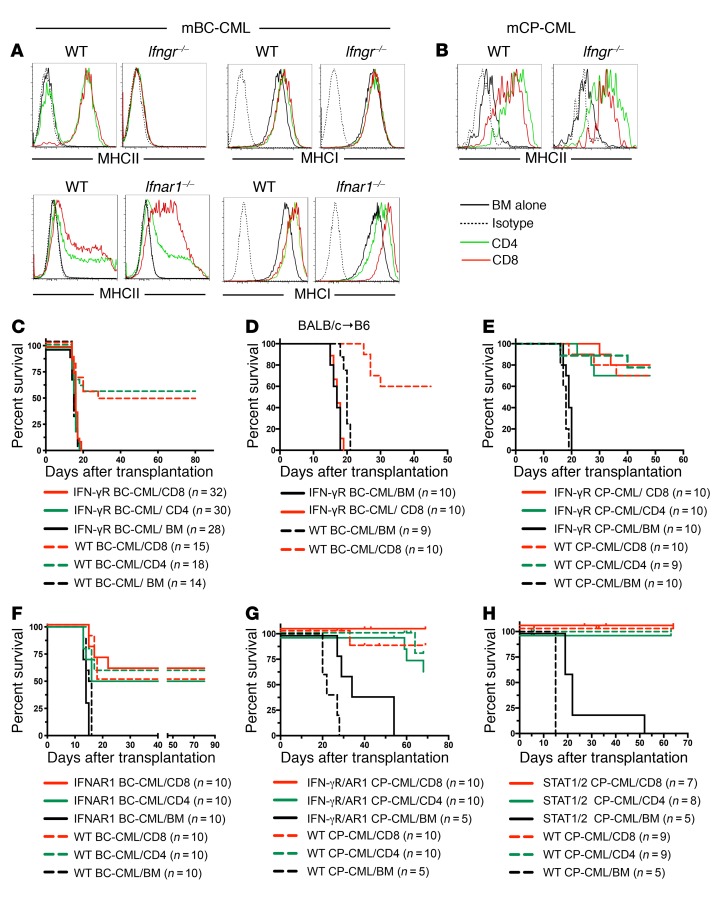
Role of IFNs in leukemia MHC upregulation and GVL.
IFNs upregulate MHC on many cell types. To test the role of IFNs in GVH-induced MHC upregulation on mBC-CML cells, we created type I IFN receptor–deficient (Ifnar1–/–) and IFN-γR–deficient (Ifngr–/–) mBC-CML. Whereas MHC upregulation was intact in Ifnar1–/– mBC-CML cells, we found that it was completely abrogated in Ifngr–/– mBC-CML cells (Figure 2A). In contrast, MHCII upregulation was similar in WT and Ifngr–/– mCP-CML LSCs harvested from GVH mice (Figure 2B). Strikingly, consistent with the MHC upregulation data, Ifngr–/– mBC-CML was completely resistant to CD4- and CD8-mediated GVL in the C3H.SW→B6 strain pairing (Figure 2C), whereas Ifnar1–/– mBC-CML, Ifngr–/– mCP-CML, Ifngr–/– Ifnar1–/–, and Stat1–/– Stat2–/– mCP-CML were as GVL sensitive as their WT counterparts (Figure 2, E–H). It was unexpected that Ifngr–/– mBC-CML was completely resistant to CD8-mediated GVL as there is substantial basal MHCI expression that is only modestly increased with IFN-γ stimulation. We considered the possibility that there is a dominant, targeted minor histocompatibility antigen (miHA) that depends on IFN-γ for its generation and presentation. We therefore performed a GVL experiment in the BALB/c (H-2d)→B6 MHC-mismatched system, in which it would be unlikely that CD8 alloimmunity would rely on a single miHA. Even with an MHCI mismatch, IFN-γR–/– mBC-CML was completely resistant to CD8-mediated GVL (Figure 2D), suggesting that IFN-γ sensitization of mBC-CML cells to CD8-mediated GVL involves more than MHCI upregulation.
Figure 2. The IFN-γR on mBC-CML cells is required for GVH-induced MHC upregulation and effective CD4- and CD8-mediated GVL, whereas MHCII upregulation on mCP-CML cells and GVL are independent of both the IFN-γR and IFNAR1 and STAT1/STAT2.
(A) Irradiated B6 mice were reconstituted with C3H.SW BM and B6 Ifngr–/–, B6 Ifnar1–/–, or control WT B6 mBC-CML cells, with no C3H.SW T cells or C3H.SW CD4 or CD8 cells. MHCI and MHCII upregulation did not occur on Ifngr–/– mBC-CML LSCs but was intact on Ifnar1–/– LSCs. (B) Mice were transplanted as in A, except with B6 WT or Ifngr–/– mCP-CML cells. MHCII upregulation was similar in WT and gene-deficient mCP-CML LSCs. Consistent with the upregulation data, IFN-γR mBC-CML was resistant to CD4- and CD8-mediated GVL in the C3H.SW→B6 model (C) and to CD8-mediated GVL in the BALB/c→B6 model (D). In contrast, Ifngr–/– mCP-CML (E), Ifnar1–/– mBC-CML (F), Ifngr–/– Ifnar1–/– (double-KO [DKO]) mCP-CML (G), and Stat1–/– Stat2–/– mCP-CML (H) cells were GVL sensitive. Experiments shown in E–H were in the C3H.SW→B6 model. AR1, IFNAR1–/–. P < 0.006, comparing any WT mBC-CML or IFNAR mBC-CML CD4 or CD8 recipient group with its BM-alone control; P < 0.002, comparing any mCP-CML T cell recipient group with its BM-alone control. P values determined by 2-tailed Mann-Whitney U test.
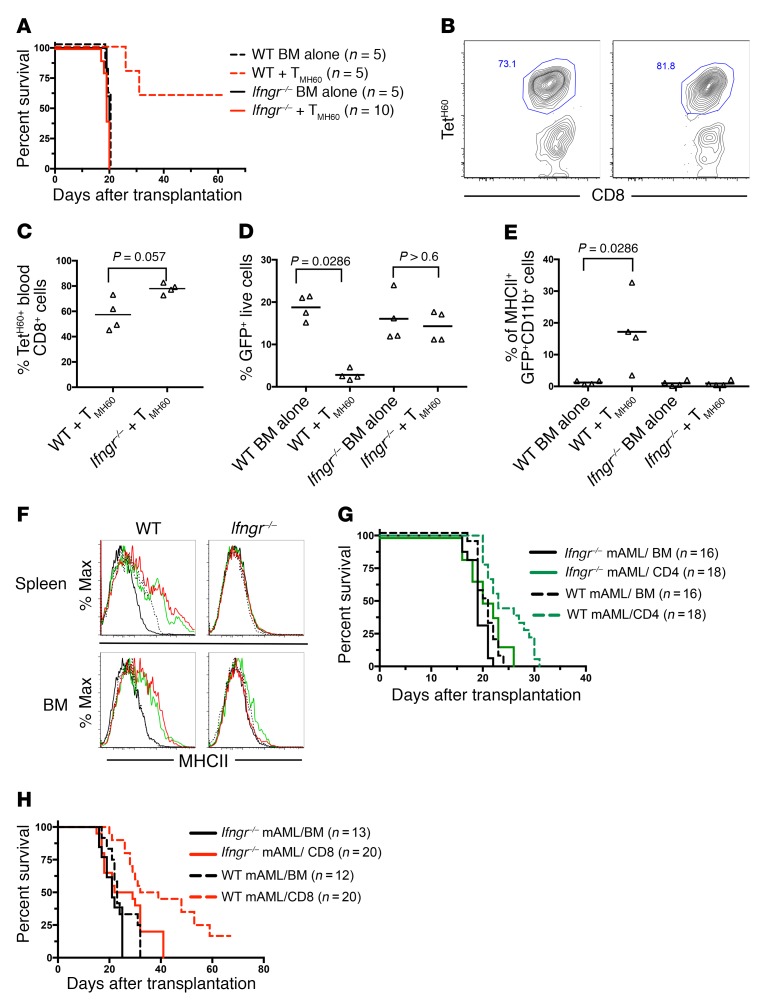
We also compared the GVL effect against WT and Ifngr–/– mBC-CML mediated by memory T cells raised against the miHA H60 (26, 29, 30). C3H.SW mice (H60–) were vaccinated against H60 as previously reported (26). B6.H60 mice (congenic for H60) were irradiated and reconstituted with C3H.SW BM, with WT or Ifngr–/– B6.H60 mBC-CML with no T cells, or with 5 × 104 sort-purified CD8+CD44+ cells from H60-vaccinated mice (TMH60) containing 3,500 H60-reactive cells as determined by H60 tetramer staining (TetH60; data not shown). Ifngr–/– B6.H60 mBC-CML was highly GVL resistant (Figure 3A, survival), despite an expansion of H60-reactive T cells in blood that was at least as strong as that observed in recipients of WT B6.H60 mBC-CML (Figure 3, B and C). Whereas TMH60 reduced blood WT B6.H60 mBC-CML cells, they failed to do the same against Ifngr–/– B6.H60 mBC-CML (Figure 3D). Finally, H60-reactive CD8 cells induced MHCII upregulation (a marker for IFN-γ stimulation) only on WT B6.H60 mBC-CML cells (Figure 3E).
Figure 3. IFN-γ stimulation is required for TMH60-mediated GVL against H60+ mBC-CML and CD4- and CD8-mediated GVL against MLL-AF9–induced AML.
(A–E). Irradiated B6.H60 mice were reconstituted with C3H.SW BM, WT or Ifngr–/– B6.H60 mBC-CML, with no T cells or with and 5 × 104 sort-purified CD8+CD44+ cells from H60-vaccinated donors, which contained 3,500 TetH60+ cells. (A) Post-transplantation survival. (B) Representative TetH60 staining of blood CD8 cells from WT and Ifngr–/– H60+ mBC-CML recipients and their quantitation (C) on day 14 after transplantation. The percentage of blood NGFR+EGFP+ cells on day 14 after transplantation and the percentage of these cells that were MHCII+ are shown in D and E, respectively. For C–E, each symbol represents data from an individual mouse; horizontal lines represent the mean. P = 0.0034, comparing survival of recipients of WT H60+ BC-CML, with or without TMH60. P = 0.378, comparing survival of recipients of Ifngr–/– H60+ mBC-CM with or without TMH60. (F–H) Irradiated B6 mice were reconstituted with C3H.SW BM, WT or Ifngr–/– MLL-AF9 AML with no T cells, or with C3H.SW CD4 or CD8 cells. Spleen and BM cells were harvested on day 13 after transplantation and analyzed for MLL-AF9 AML (GFP+) MHCII expression (F). Survival for CD4-mediated and CD8-mediated GVL is shown in G and H, respectively. P ≤ 0.0016, comparing the survival in the WT mAML BM-alone group with WT CD4 or CD8 recipients; P = 0.024, comparing Ifngr–/– BM alone with CD4 recipients; P = 0.0079 and P = 0.0053, comparing CD4 and CD8 recipients of WT versus Ifngr–/– MLL-AF9 AML, respectively. P values determined by 2-tailed Mann-Whitney U test. Data in G and H were combined from 2 repetitions.
To determine whether the reliance on IFN-γ stimulation for effective GVL is unique to mBC-CML or is a more general property of myeloblastic leukemias, we created WT and Ifngr–/– AML by transducing WT or Ifngr–/– BM with a retrovirus encoding the MLL-AF9 translocation (27) and then tested the sensitivities of these AML cells to CD4- and CD8-mediated GVL in the C3H.SWgB6 model. Cohorts were sacrificed 12 days after BMT for analysis of MHC expression on splenic and BM AML cells. CD4 and CD8 cells induced a modest upregulation of MHCII on WT AML cells but not on Ifngr–/– AML cells (Figure 3F). MHCI was not upregulated (date not shown). Importantly, compared with WT MLL-AF9, we found that Ifngr–/– MLL-AF9 mAML was relatively resistant to both CD4- and CD8-mediated GVL (Figure 3, G and H).
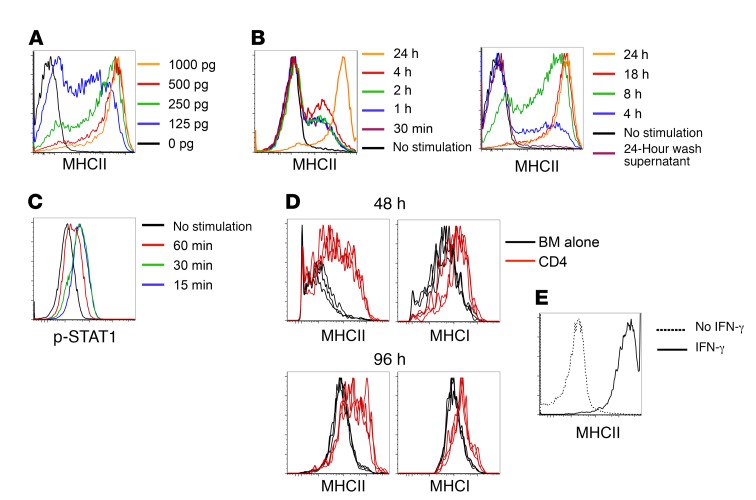
IFN-γ is sufficient to upregulate MHC on mBC-CML cells.
While the IFN-γR was required for MHC upregulation on mBC-CML cells in vivo, it was possible that other unidentified stimuli were also required. To test whether IFN-γ alone is sufficient, we examined the effects of IFN-γ on mBC-CML cells in vitro. Culture in media plus IFN-γ, but not media alone, induced MHCII upregulation. With continuous culture in IFN-γ at concentrations detected in the serum of GVHD mice (31), MHCII upregulation was not detected until 24 to 48 hours and usually peaked between 48 and 72 hours (Figure 4A and data not shown). An IFN-γ pulse of as short as 1 hour induced MHCII upregulation on a minority of cells at 72 hours, but maximum MHCII upregulation required exposure for 8 to 18 hours (Figure 4B). Despite this requirement, in vitro IFN-γ stimulation induced mBC-CML STAT1 phosphorylation within 15 minutes (Figure 4C), indicating that, despite rapid STAT1 phosphorylation, more prolonged IFN-γR signaling is required for maximal MHCII upregulation. To determine whether the in vitro time course of MHCII upregulation parallels what occurs in vivo, we transferred B6 MHCIIlo mBC-CML cells harvested from sublethally irradiated syngeneic B6 mice into irradiated B6 mice that were transplanted with C3H.SW BM, with or without C3H.SW CD4 cells, 7 days earlier. We observed that MHCI and MHCII expression increased on splenic (Figure 4D) and BM (not shown) mBC-CML LSCs in GVH mice at 48 and 96 hours after transfer, but not in mice that did not receive CD4 cells.
Figure 4. In vitro IFN-γ treatment is sufficient to upregulate MHCII on mBC-CML LSCs.
(A) Sort-purified mBC-CML LSCs were continuously cultured with graded concentrations of IFN-γ, and MHCII expression was measured 72 hours later. (B) mBC-CML LSCs were cultured for 30 minutes to 24 hours in IFN-γ (200 pg/ml). Stimulated cells were washed extensively and then further cultured without IFN-γ for a total culture time of 72 hours, followed by measurement of MHCII expression. The final supernatant from each wash was cultured with fresh mBC-CML cells for 72 hours, and MHCII was not upregulated (see “24-Hour wash supernatant” in B). Data are representative of 2 experiments, with 3 replicates per condition. (C) mBC-CML cells were cultured in IFN-γ, and p-STAT1 was measured by flow cytometry at the indicated times. (D) Irradiated B6 mice were reconstituted with C3H.SW BM with or without C3H.SW CD4 cells. On day 7 after alloBMT, sort-purified mBC-CML LSCs were injected. mBC-CML cells were recovered 48 and 96 hours later and MHCI and MHCII expression was analyzed by flow cytometry. Shown are data from splenic mBC-CML cells; data in BM were similar (not shown). Each line represents data from an individual mouse. (E) Irradiated B6 mice were transplanted with mBC-CML cells, C3H.SW BM, and CD4 cells. On day 21 after transplantation, sort-purified splenic MHCII– CD11b– mBC-CML cells were cultured with or without IFN-γ for 72 hours. IFN-γ induced MHCII expression.
These results identify a key mechanism that at least in part explains the resistance of mBC-CML and AML GVL against GVL compared with the GVL that develops against CP-CML. Effective GVL against mBC-CML and mAML requires sufficient and sustained IFN-γ to sensitize LSCs to T cell killing, whereas effective GVL against mCP-CML does not require IFN-γ stimulation . mBC-CML LSCs recovered from mice that were dying from leukemia at later times after transplantation, despite being transplanted with donor T cells, were mostly MHCIIlo. This decline in MHCII expression was not due to selection for cells that could not respond to IFN-γ, as MHCIIlo cells harvested from mice with progressive mBC-CML, despite receiving donor T cells, uniformly upregulated MHCII in vitro with IFN-γ stimulation (Figure 4E). This suggests that leukemia progression at later points after transplantation is due to both a contraction of the alloreactive T cell response and a drop in IFN-γ levels.
mBC-CML and mCP-CML gene expression changes in the GVH environment.
To better understand potential mechanisms of action of IFN-γ, we performed gene expression analyses on WT and Ifngr–/– mBC-CML and WT mCP-CML LSCs sorted from mice that were or were not undergoing a GVH response. For mBC-CML LSCs, irradiated B6 mice were reconstituted with WT or Ifngr–/– mBC-CML cells, C3H.SW T cell–depleted BM, with or without C3H.SW CD4 cells. For mCP-CML, to facilitate recovery of sufficient mCP-CML cells, we again used the B6gBALB/c GVH system with B6 mCP-CML cells, with or without B6 CD4 cells. mBC-CML and LSK mCP-CML cells (lin–) were harvested between days 12 and 14 after BMT. LSCs from 3 individual mice were analyzed from each group.
We saw a clear IFN-γ gene signature when comparing mRNA from Ifngr–/– mBC-CML cells from the BM-alone group with mRNA from WT mBC-CML cells harvested from CD4 recipients (Figure 5). Relative to the Ifngr–/– groups, there was a background IFN-γ signature in WT mBC-CML cells harvested from the B6 BM-alone group, probably due to IFN-γ produced by the host-versus-graft response. In addition to a classic IFN-γ signature, in WT BC-CML cells from GVH mice, we observed changes in genes linked to antigen presentation, ubiquitination, proteasome function, and apoptosis. As expected, there was no IFN-γ signature in the IFN-γR mBC-CML cells. By volcano plot analysis, we found that gene expression was similar in Ifngr–/– mBC-CML LSCs harvested from the BM-alone and CD4 groups, and pathway analyses were unrevealing as to what drove the few observed differences (data not shown).
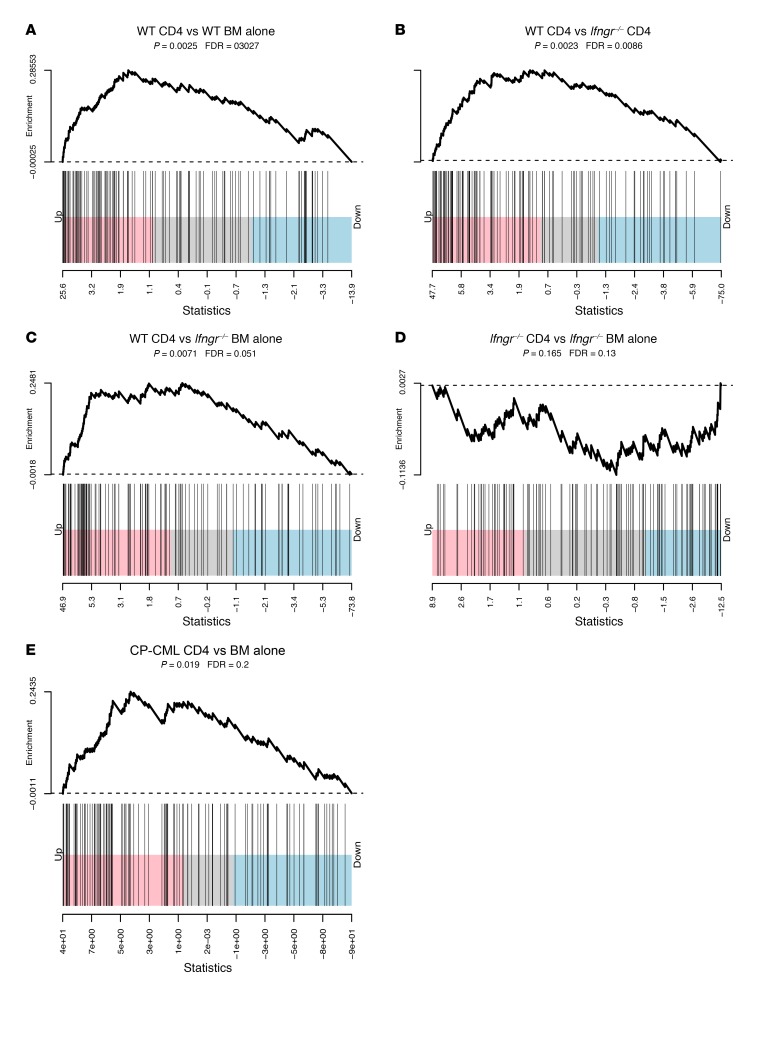
Figure 5. Gene expression analyses of mBC-CML and mCP-CML LSCs.
(A–D) WT or Ifngr–/– mBC-CML cells were harvested from C3H.SW→B6 recipients of C3H.SW CD4 cells. Shown are Gene Set Enrichment Analysis–style (GSEA-style) barcode plots for IFN-γ pathway gene expression using hallmark data. Note the significant upregulation of IFN-γR pathway genes comparing WT mBC-CML cells harvested from CD4 recipients with WT mBC-CML LSCs from BM-alone recipients (A), Ifngr–/– mBC-CML LSCs from CD4 recipients (B), and Ifngr–/– mBC-CML LSCs from BM-alone recipients (C). There was no IFN-γR signature comparing Ifngr–/– mBC-CML LSCs harvested from CD4 recipients compared with those from BM-alone recipients (D), and few differentially expressed genes distinguished these groups (Supplemental Figure 1). (E) WT B6 mCP-CML LSCs were harvested from BALB/c recipients that did or did not receive CD4 cells (BM alone). An IFN-γR signature was observed in RNA from LSCs harvested from CD4 recipients as compared to RNA from LSCs from BM alone controls.
mCP-CML gene expression analysis.
We also detected an IFN-γ signature in mCP-CML LSCs harvested from CD4 recipients relative to LSCs harvested from mice treated with BM alone (Supplemental Figure 1; supplemental material available online with this article; https://doi.org/10.1172/JCI85736DS1). Therefore, the fact that WT mCP-CML and Ifngr–/– mCP-CML were equivalently GVL sensitive was not due to WT mCP-CML being unresponsive to IFN-γ.
T cells are the critical source of IFN-γ.
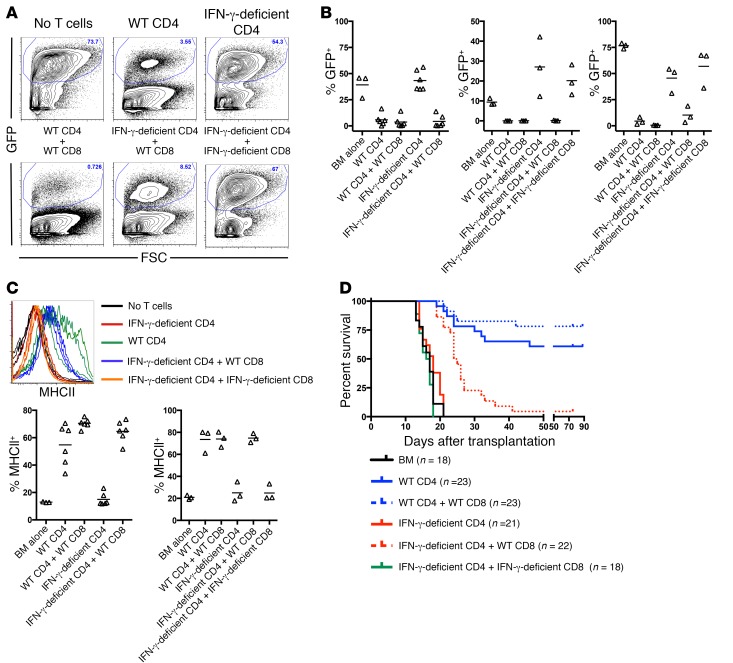
T cells are the major, but not exclusive, producers of IFN-γ. To determine whether donor T cell–derived IFN-γ is required for MHC upregulation and GVL, we compared mBC-CML MHC upregulation and GVL in recipients of WT or IFN-γ–deficient CD4+ T cells. Because TCR-MHC interactions were not required for MHC upregulation, we also considered the possibility that T cells unable to directly kill leukemia cells could provide IFN-γ in trans, thereby rescuing the inability of IFN-γ–deficient T cells to mediate GVL. Irradiated B6 mice were reconstituted with B6 B2m–/– mBC-CML (MHCI–) and BALB/c BM with: a) no T cells; b) WT or IFN-γ–deficient BALB/c CD4 cells; or c) WT or IFN-γ–deficient CD4 cells with WT or IFN-γ–deficient BALB/c CD8 cells. B2m–/– mBC-CML was used to prevent donor CD8 cells from directly killing leukemia cells. Whereas WT CD4 cells induced MHCII expression and reduced the number of splenic and BM mBC-CML cells by day 14 after BMT, IFN-γ–deficient CD4 cells failed to do so (Figure 6, A–C, and data not shown), indicating that donor T cell–derived IFN-γ is critical. The addition of WT, but not IFN-γ–deficient, CD8 cells restored IFN-γ–induced MHCII upregulation and diminished the number of mBC-CML cells in spleen and BM by day 14 (Figure 6, A–C, and not shown), indicative of a GVL-promoting effect. Consistent with this early GVL effect, the addition of WT, but not IFN-γ–deficient, CD8 cells to IFN-γ–deficient CD4 cells prolonged the survival of mice in 2 of 3 repeated experiments (Figure 6D). The rescue of IFN-γ–deficient CD4 cells by WT CD8 cells may have been incomplete because of differences in the kinetics and magnitude of IFN-γ production by CD4 and CD8 cells, which is consistent with the lower mean fluorescence intensity (MFI) of MHCII in the IFN-γ–deficient CD4 plus WT CD8 group (mean of 443 vs. 167). That the addition of WT CD8 cells to IFN-γ–deficient CD4 cells restored GVL also confirms that the inability of IFN-γ–deficient CD4 cells to mediate GVL was not due to a failure to generate cytolytic effectors.
Figure 6. T cell–derived IFN-γ delivered in cis or in trans is required for CD4-mediated GVL.
Irradiated B6 mice were reconstituted with BALB/c BM and B6 B2m–/– mBC-CML with no T cells or with the following BALB/c background T cells: (i) WT CD4 cells; (ii) IFN-γ–deficient CD4 cells; (iii) WT CD4 cells plus WT CD8 cells; (iv) IFN-γ–deficient CD4 cells plus WT CD8 cells; or (v) IFN-γ–deficient CD4 cells plus IFN-γ–deficient CD8 cells. Cohorts were sacrificed 10–14 days after transplantation, and mBC-CML cells were analyzed by flow cytometry. (A) Representative flow cytometry from splenocytes. (B) Percentage of leukemic splenocytes from individual mice (n = 3 independent experiments). Each symbol represents data from an individual mouse; horizontal lines indicate mean values. (C) Upper panel: WT, but not IFN-γ–deficient, CD4 or CD8 cells induced MHCII upregulation on mBC-CML LSCs. The upper panel shows LSC MHCII expression. Each line represents data from an individual mouse. Lower panel: Percentage of LSCs that were MHCII+ from 2 of 3 experiments. Each symbol represents data from an individual mouse; horizontal lines indicate the mean values. Insufficient numbers of LSCs were present to analyze in the third experiment (see bottom panel in Figure 5B). (D) In 2 of 3 experiments, the addition of WT CD8 cells, but not IFN-γ–deficient CD8 cells, to IFN-γ–deficient CD4 cells prolonged survival (data were combined from 2 experiments). P = 0.0037 comparing the KO CD4 + WT CD8 group to either the KO CD4 or KO CD4 + KO CD8 groups.
IFN-γ alone is insufficient for GVL.
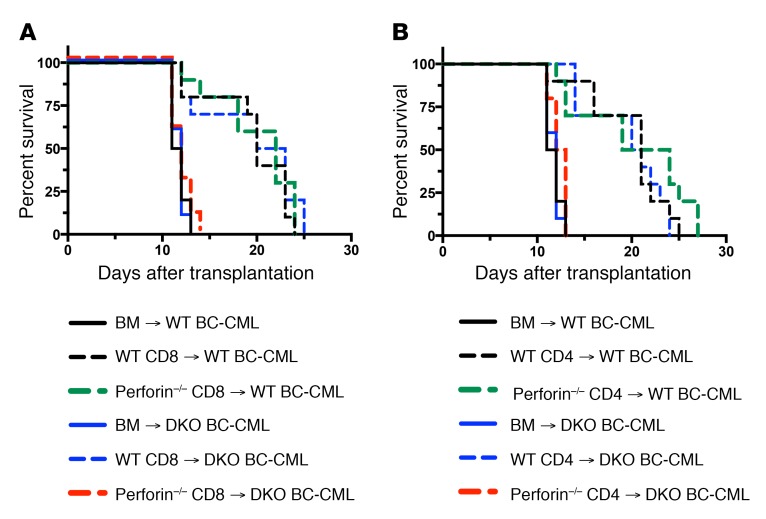
Given the complete GVL resistance of Ifngr–/– mBC-CML, we considered the possibility that IFN-γ alone has direct and meaningful antileukemia activity. Arguing against this is that for effective GVL, CD4 and CD8 cells require leukemia expression of MHCII and MHCI, respectively (10, 11, 24) despite alloreactive T cells activated in response to recipient alloantigens generating substantial IFN-γ. However, sufficiently high local IFN-γ levels may only be achieved when IFN-γ–producing T cells make cognate TCR-MHC contacts. To test this hypothesis, we engineered a situation in which cognate TCR-MHC interactions occurred, but perforin and death receptor–dependent T cell–induced apoptosis and necroptosis were prevented. We first created Fas (TNFRSF6) via Fas-associated protein with death domain (Fadd–/– Ripk3–/–) mBC-CML cells. FADD is required for all death receptor signaling; however, Fadd–/– mice are embryonically lethal unless they are crossed with Ripk3–/– mice due to the role of FADD in inhibiting RIPK3-dependent necroptosis (32). We then compared GVL mediated by WT or perforin–/– CD4 or CD8 cells against Fadd–/– Ripk3–/– and control Fadd+/– Ripk3+/– mBC-CML. Effective GVL only required either WT T cells or FADD/RIPK3-intact mBC-CML (Figure 7). In contrast, GVL was completely abrogated when donor T cells were perforin–/– and mBC-CML cells were Fadd–/– Ripk3–/–, despite T cells being capable of producing IFN-γ and making TCR-MHC contacts with mBC-CML cells. Therefore, CD4- and CD8-dependent GVL depends on T cell perforin and/or mBC-CML death receptor signaling, and T cell–derived IFN-γ alone is insufficient.
Figure 7. CD4 and CD8-mediated GVL require intact T cell perforin or leukemia FADD/RIPK3, and T cell–derived IFN-γ alone is insufficient for GVL.
Irradiated B6 mice were reconstituted with BALB/c BM and B6 Fadd–/– Ripk3–/– (DKO) or control B6 Fadd+/– Ripk3+/– mBC-CML (WT) with no BALB/c T cells or with WT or perforin–/– CD8 cells (A) or CD4 cells (B). Data show survival rates from 1 of 2 similar experiments with 10 mice per group. P < 0.0003, comparing any WT or DKO BM-alone group with any WT T cell group. P > 0.07, comparing DKO BM-alone versus perforin–/– CD4 or CD8 groups. P values determined by 2-tailed Mann-Whitney U test.
Discussion
Relapsed acute leukemia after alloSCT remains a major unmet medical need. Much of the early and exciting efficacy of alloSCT as a form of adoptive T cell immunotherapy was observed in patients with CP-CML (33); however, in the tyrosine kinase inhibitor era, these patients infrequently undergo transplantation. While GVL is active against myeloblastic leukemias, it is less potent than GVL against CP-CML. This difference in potency is reflected in the higher rates of BC-CML and AML relapse after alloSCT. Unfortunately, there has been little progress in decreasing or treating relapses, in which the withdrawal of immunosuppression and donor leukocyte infusions infrequently result in durable remissions. A major barrier to progress has been a lack of understanding of the resistance mechanisms. A central paradox, evident since the early days of clinical alloSCT, is why an alloimmune response sufficient to cause GVHD, promote 100% donor hematopoietic chimerism, and mediate GVL against CP-CML is so much less effective against AML. Here, we report a mechanism that may explain this: the alloimmune T cell response against AML and BC-CML must generate sufficient IFN-γ to sensitize myeloblast LSCs to T cell killing. In contrast, a smaller and more smoldering alloreactive response, which does not generate high levels of IFN-γ, is sufficient for GVL against mCP-CML and for GVHD.
Ifngr–/– mBC-CML and MLL-AF9 mAML were GVL resistant, and, conversely, IFN-γ–deficient T cells mediated no GVL against mBC-CML, despite their expansion and induction of clinical GVHD (data not shown). In mouse models without immunosuppression, there is an early wave of IFN-γ production coincident with rapid alloreactive T cell expansion (34–36). However, in clinical alloSCT, in which pharmacologic immunosuppression is given, such a rise in IFN-γ may only infrequently occur, and this may in part contribute to the more global GVL resistance of myeloblastic leukemias. Chronic GVHD (cGVHD) has been associated with a lower risk for AML relapse, and recent data suggest that cGVHD is associated with the action of IFN-γ (37). Perhaps the sustained IFN-γ associated with cGVHD contributes to GVL, along with alloreactive cytolytic T cells. In contrast, GVL against mCP-CML was independent of type I and II IFNs or their downstream signaling components STAT1 and STAT2, even though gene expression analysis indicated that mCP-CML LSCs are IFN-γ responsive.
In an effort to understand the cell-intrinsic properties of mBC-CML cells that render them GVL resistant relative to CP-CML cells, we have tested in GVL models mouse blast crisis and chronic phase leukemias deficient in numerous genes including those encoding β2M, MHCII, Fas, TRAIL, TNFR1/R2, PD-L1, PD-L2, ICAM-1, and IFNAR1 (10, 11, 24, 25). Until the present study, we found no deficiency that selectively diminished GVL against mBC-CML without equivalently reducing GVL against mCP-CML.
Other factors in addition to the requirement for IFN-γR signaling may also contribute to the relative GVL resistance of myeloblastic leukemias as compared with that of CP-CML. Nonetheless, the present results argue that GVL could be meaningfully augmented by exposing malignant myeloblasts to IFN-γ after transplantation, timed to coincide with the presence of alloreactive cytolytic T cells. The key translational question is how to achieve this. IFN-γ–induced MHC upregulation and GVL sensitization do not require TCR-MHC contacts between IFN-γ–producing T cells and leukemia cells (see Figures 1 and 5). That is, IFN-γ can be delivered to leukemia cells in trans by T cells that do not target the leukemia cells. This suggests a feasible clinical strategy. CMV reactivation early after transplantation and the use of CMV seropositive donors have been associated with lower rates of AML relapse (38–41). CMV reactivation induces the expansion of CMV-reactive T cells and NKG2C+ NK cells (42), both of which can produce IFN-γ and raise serum IFN-γ levels (43). Although other explanations are possible, our data suggest that anti-CMV immune responses could be promoting GVL through the elaboration of IFN-γ. Approaches to further promote the anti-CMV immune response, perhaps by donor and recipient anti-CMV vaccination (44, 45), might augment GVL without an increase in GVHD. This strategy is analogous to our use of IFN-γ–positive CD8 cells to promote GVL by IFN-γ–deficient CD4 cells.
That IFN-γ was required for CD4-mediated GVL is easy to understand, given its importance in upregulating MHCII. However, it was surprising that Ifngr–/– mBC-CML and MLL-AF9 mAML were resistant to CD8-mediated GVL, as these leukemias are MHCI+ at baseline, and IFN-γ–induced MHCI upregulation is modest or does not occur in the case of MLL-AF9 AML. It is possible that MHCI upregulation crosses a threshold of MHCI-miHA peptide complexes to sufficiently engage alloreactive T cells. However, the fact that Ifngr–/– mBC-CML was also resistant to CD8-mediated GVL in the MHC-mismatched setting argues against this being the sole explanation, considering the much higher frequency of T cells that recognize allogeneic MHC (46, 47) and the diminished likelihood that GVL in this model would rely on a single targeted peptide. IFN-γ upregulates ICAM-1 (data not shown), immunoproteasome components, and chemokines that attract T cells and modifies pro- and antiapoptotic pathways (Figure 5 and ref. 48), all of which could promote effective CD8 cell killing. Discovering precisely how IFN-γR signaling sensitizes mBC-CML and AML cells to killing will require substantial further investigation, and given the pleiotropic effects of IFN-γR signaling, it is possible that no single downstream mechanism will explain its dominant role. Another intriguing and unanswered question is what signal(s) in the alloimmune environment induce MHCII upregulation on mCP-CML LSCs. Given that TCR-MHC interactions are also not required, a soluble factor is most likely responsible.
We used Fadd–/– Ripk3–/– mBC-CML and perforin–/– T cells to demonstrate that, despite IFN-γ being critical for GVL, alone it is insufficient, as there was no GVL when T cells lacked perforin and leukemias lacked FADD and RIPK3. These data also highlight the idea that cytolytic pathways engaged by both CD4 and CD8 cells are redundant, in that either perforin or death receptor FADD/RIPK3-dependent killing is sufficient for GVL.
We studied models of CP-CML, BC-CML, and AML, which share essential genetic and phenotypic features with their human counterparts (49). It will be important to determine the importance of IFN-γ in GVL against other mouse myeloid leukemias and against human AML samples. Available data suggest that IFN-γ could broadly impact GVL against human AML. Human AML stem cells include MHCII– cells (50), and acute promyelocytic AML is classically MHCII–. Other leukemias reported to be MHCII+ usually have cell populations that are MHCII–, as the MHCII expression threshold for calling an AML MHCII+ does not require all cells to express MHCII (51–54). Therefore, if alloreactive CD4 cells are to directly mediate GVL against these MHCII– cells, MHCII expression must be induced. Primary human AML cells and AML-like cell lines express the IFN-γR and are IFN-γ responsive (48, 55–62), with both basal and IFN-γ–induced STAT1 phosphorylation (58) and MHCI and MHCII upregulation. We hope our results demonstrating a critical role for IFN-γ in GVL responses against two clinically relevant mouse models of myeloblastic leukemias will both stimulate a broader evaluation of the IFN-γ responsiveness of additional mouse AML models and of human AML specimens classified by molecular features, and the development of approaches to safely deliver IFN-γ in the clinic.
Methods
Mice
C57BL6 (B6; H-2b) and BALB/c (H-2d) mice were purchased from Harlan Laboratories. C3H.SW mice were purchased from The Jackson Laboratory and bred at Yale University. IFN-γ–deficient (63) and perforin–/– mice backcrossed with BALB/c mice (64, 65) were bred at the University of Iowa and at Yale University. Ifngr–/– mice were purchased from the Jackson Laboratory. Ifnar1–/– mice were obtained from H. Rosenberg (66) (National Cancer Institute [NCI], Bethesda, Maryland, USA) and bred at Yale University. Ifnar1–/– Ifngr–/– mice were obtained from William Klimstra (University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA). Stat1–/– Stat2–/– mice were obtained from John Alcorn (University of Pittsburgh). B6.H60 mice were originally obtained from Derry Roopenian (The Jackson Laboratory) and were bred at the University of Pittsburgh. B6 Fadd–/– Ripk3–/– and control B6 Fadd–/+ Ripk3–/+ mice were maintained at St. Jude Children’s Research Hospital (Memphis, Tennessee, USA) (32).
Leukemia induction
mBC-CML and mCP-CML were created as previously described (11, 12). Briefly, mBC-CML was created by spin-infection of BM from 5FU-treated mice with two MSCV2.2-based retroviruses, one expressing BCR-ABL (along with a truncated and nonsignaling human nerve growth factor receptor [NGFR]) and a second expressing NUP98 and HOXA9 (coexpressing GFP) (11). MLL-AF9 AML was induced by transduction with an MSCV2.2-based retrovirus encoding the MLL-AF9 fusion cDNA and GFP (27) (gift of Scott Armstrong, Memorial Sloan Kettering Institute, New York, New York, USA). Cells were passaged in vivo and frozen. For each experiment mCP-CML was created by spin-infection with BCR-ABL retrovirus alone as described previously (24).
BMT
Four different strain pairings were used in alloBMT experiments. All irradiation was delivered by a cesium irradiator. All BM was T cell depleted and is referred to herein as BM.
C3H.SW (H-2b)→B6 (H-2b).
B6 mice received 900 cGy irradiation and were reconstituted with 5 × 106 to 7 × 106 C3H.SW BM, with or without bead-purified C3H.SW CD4 or CD8 cells.
C3H.SW→B6.H60.
B6.H60 mice received 900cGy irradiation and were reconstituted with 7 × 106 C3H.SW BM cells and CD8+CD44+ cells from C3H.SW mice vaccinated against H60 as previously described (26).
BALB/c (H-2d)→B6.
B6 mice were irradiated (900 cGy) and reconstituted with 107 BALB/c BM cells, with or without purified BALB/c (WT or gene-deficient) CD4 or CD8 cells.
B6→BALB/c.
Irradiated BALB/c mice (900 cGy) were reconstituted with 107 B6 BM cells, with or without purified B6 CD4 or CD8 cells. mBC-CML and AML cells were either from frozen stocks or fresh cells harvested from sublethally irradiated B6 mice seeded 14 to 21 days beforehand. Two hundred micrograms anti-NK1.1 (PKC136; lab-prepared) was given i.p. on days –2 and –1 in experiments with the BALB/c→B6 strain pair and on days –2, –1, and +7 in experiments with β2M–/– mBC-CML to prevent NK cell–mediated killing of MHCI– or H-2d+b– cells.
Antibodies and flow cytometry
mCP-CML LSCs were identified by excluding lin+ cells using staining with biotin-conjugated antibodies against TER-119, Gr-1, CD19, and CD11b (all from BD Pharmingen; clones TER-119, RB6-8C5, 1D3, and M1/70, respectively); CD4 and CD8 (clones GK1.5 and TIB120; lab-prepared); CD117 phycoerythrin (PE) and Sca-1 FITC (BD Pharmingen; clones 2B8 and Ly-6a/E); and anti-NGFR Alexa 647 (clone HB8737; lab-prepared). mBC-CML cells (lin–) could be identified by only staining for CD11b. Staining for Kb and I-A/I-E (MHCII) was done with clones AF6-88.5 and M5/114.15.2, respectively (BioLegend). Phosphorylated STAT1 (p-STAT1) staining was performed with or without IFN-γ stimulation, followed by fixation in 2% paraformaldehyde in PBS. The cells were permeabilized in BD Phosflow Perm Buffer III for 30 minutes on ice, washed twice in BD Pharmingen Stain Buffer, and stained with PE-conjugated p-STAT1 antibody (pY701; BD). H60-reactive T cells, which detect the LTFNYRNL peptide bound to Kb, were identified by MHCI tetramer staining (TetH60+). H60 tetramers were created at the NIH’s tetramer facility (Atlanta, Georgia, USA).
Cell purifications
Lymph node (LN) and splenic cells were prepared as previously described (11). T cell purifications were performed using EasySep negative selection reagents according to the manufacturer’s instructions (STEMCELL Technologies). Cell purities were greater than 88%, with less than 2% of contaminating CD4 or CD8 cells. CD8 memory T cells were isolated from H60-vaccinated mice by first using a CD8 EasySep negative selection kit. Cells were then stained with antibodies against CD8 and CD44, followed by sorting on a FACSAria cell sorter (BD Biosciences).
In vitro mBC-CML stimulation
Splenocytes from sublethally irradiated B6 mice injected with mBC-CML cells approximately 2 to 3 weeks earlier (at least 80% EGFP+NGFR+) were cultured for different lengths of time in DMEM with 10% FCS with graded amounts of IFN-γ (Peprotech). In experiments in which IFN-γ was removed and cells were recultured, cells were washed 4 times in 50 cc of media. The final supernatant was cultured with fresh mBC-CML cells for another 72 hours as a confirmation that IFN-γ had been removed. At the end of the culture period, cells were more than 80% viable.
Gene expression profiling
mCP-CML.
Irradiated BALB/c mice were reconstituted with B6 BM, 7 × 105 BCR-ABL spin-infected B6 BM (see leukemia induction) with no B6 T cells, or with 5 × 105 B6 CD4 cells. On day 12 after BMT, NGFR+ LSK cells were separately sorted from the spleens of 3 individual mice from each group.
mBC-CML.
Irradiated B6 mice were reconstituted with C3H.SW BM, B6 WT or Ifngr–/– mBC-CML, with no T cells, or with 4 × 106 C3H.SW CD4 cells. On day 14 after BMT, lin– mBC-CML cells were sorted from the spleens of 3 individual mice from each group. RNA was isolated using a QIAGEN RNA Extraction Kit, followed by library preparation using an Epicentre kit. Expression was analyzed by quantitation using an Illumina WG-6 v2.0 Expression BeadChip. Differential expression was computed using the Bioconductor package (67), and pathway analysis was performed using the limma “camera” function (68), which controls for intercorrelation among genes, and the “canonical pathways” gene sets provided by mSigDB (69).
Statistics
Survival differences were calculated using the log-rank test (Mantel-Cox) (GraphPad Prism; GraphPad Software version 7). In some CD8-mediated GVL against mCP-CML experiments, some mice reached predetermined criteria for sacrifice due to GVHD. If spleens were small and the most recent analysis of peripheral blood showed, at most, only a few NGFR+ CP-CML cells, these mice were scored as having GVHD and were censored (see Figure 2, G and H). Differences in cell percentages were determined by 2-tailed Mann-Whitney U test.
Study approval
All animal studies were approved by the Yale University School of Medicine IACUC and the University of Pittsburgh School of Medicine IACUC.
Author contributions
CMM designed and executed experiments, analyzed data, and created figures. JL and MZ designed and executed experiments. WDS designed experiments, analyzed data, created figures, and wrote the manuscript. MC analyzed gene expression data. DRG provided intellectual input and provided key reagents. JTH provided key reagents.
Supplementary Material
Acknowledgments
This work was supported by NIH grants R01 HL117855 and NIH R01CA09643. We also thank the excellent staff of the Yale Animal Resources Center.
Footnotes
CMM’s present address is: Department of Medicine, Yale University School of Medicine, New Haven, Connecticut, USA.
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference information:J Clin Invest. 2017;127(7):2765–2776.https://doi.org/10.1172/JCI85736.
Contributor Information
Catherine Matte-Martone, Email: catherine.matte@yale.edu.
Jinling Liu, Email: Julia_Liu@pitt.edu.
Meng Zhou, Email: meng.zhou@pitt.edu.
Maria Chikina, Email: mchikina@gmail.com.
References
- 1.Vincent K, Roy DC, Perreault C. Next-generation leukemia immunotherapy. Blood. 2011;118(11):2951–2959. doi: 10.1182/blood-2011-04-350868. [DOI] [PubMed] [Google Scholar]
- 2.Blazar BR, Murphy WJ, Abedi M. Advances in graft-versus-host disease biology and therapy. Nat Rev Immunol. 2012;12(6):443–458. doi: 10.1038/nri3212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Markey KA, MacDonald KP, Hill GR. The biology of graft-versus-host disease: experimental systems instructing clinical practice. Blood. 2014;124(3):354–362. doi: 10.1182/blood-2014-02-514745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shlomchik WD. Graft-versus-host disease. Nat Rev Immunol. 2007;7(5):340–352. doi: 10.1038/nri2000. [DOI] [PubMed] [Google Scholar]
- 5.Alyea EP, et al. NCI First International Workshop on The Biology, Prevention and Treatment of Relapse after Allogeneic Hematopoietic Cell Transplantation: report from the committee on prevention of relapse following allogeneic cell transplantation for hematologic malignancies. Biol Blood Marrow Transplant. 2010;16(8):1037–1069. doi: 10.1016/j.bbmt.2010.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Collins RH, Jr, et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol. 1997;15(2):433–444. doi: 10.1200/JCO.1997.15.2.433. [DOI] [PubMed] [Google Scholar]
- 7.McGlave P. Bone marrow transplants in chronic myelogenous leukemia: an overview of determinants of survival. Semin Hematol. 1990;27(3 suppl 4):23–30. [PubMed] [Google Scholar]
- 8.Sawyers CL, et al. Imatinib induces hematologic and cytogenetic responses in patients with chronic myelogenous leukemia in myeloid blast crisis: results of a phase II study. Blood. 2002;99(10):3530–3539. doi: 10.1182/blood.V99.10.3530. [DOI] [PubMed] [Google Scholar]
- 9.Matte CC, et al. Donor APCs are required for maximal GVHD but not for GVL. Nat Med. 2004;10(9):987–992. doi: 10.1038/nm1089. [DOI] [PubMed] [Google Scholar]
- 10.Matte-Martone C, Liu J, Jain D, McNiff J, Shlomchik WD. CD8+ but not CD4+ T cells require cognate interactions with target tissues to mediate GVHD across only minor H antigens, whereas both CD4+ and CD8+ T cells require direct leukemic contact to mediate GVL. Blood. 2008;111(7):3884–3892. doi: 10.1182/blood-2007-11-125294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Matte-Martone C, et al. Graft-versus-leukemia (GVL) against mouse blast-crisis chronic myelogenous leukemia (BC-CML) and chronic-phase chronic myelogenous leukemia (CP-CML): shared mechanisms of T cell killing, but programmed death ligands render CP-CML and not BC-CML GVL resistant. J Immunol. 2011;187(4):1653–1663. doi: 10.4049/jimmunol.1100311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pear WS, et al. Efficient and rapid induction of a chronic myelogenous leukemia-like myeloproliferative disease in mice receiving P210 bcr/abl-transduced bone marrow. Blood. 1998;92(10):3780–3792. [PubMed] [Google Scholar]
- 13.Li S, Ilaria RL, Jr, Million RP, Daley GQ, Van Etten RA. The P190, P210, and P230 forms of the BCR/ABL oncogene induce a similar chronic myeloid leukemia-like syndrome in mice but have different lymphoid leukemogenic activity. J Exp Med. 1999;189(9):1399–1412. doi: 10.1084/jem.189.9.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dash AB, et al. A murine model of CML blast crisis induced by cooperation between BCR/ABL and NUP98/HOXA9. Proc Natl Acad Sci U S A. 2002;99(11):7622–7627. doi: 10.1073/pnas.102583199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Neering SJ, et al. Leukemia stem cells in a genetically defined murine model of blast-crisis CML. Blood. 2007;110(7):2578–2585. doi: 10.1182/blood-2007-02-073031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Borrow J, et al. The t(7;11)(p15;p15) translocation in acute myeloid leukaemia fuses the genes for nucleoporin NUP98 and class I homeoprotein HOXA9. Nat Genet. 1996;12(2):159–167. doi: 10.1038/ng0296-159. [DOI] [PubMed] [Google Scholar]
- 17.Nakamura T, et al. Fusion of the nucleoporin gene NUP98 to HOXA9 by the chromosome translocation t(7;11)(p15;p15) in human myeloid leukaemia. Nat Genet. 1996;12(2):154–158. doi: 10.1038/ng0296-154. [DOI] [PubMed] [Google Scholar]
- 18.Yamamoto K, Nakamura Y, Saito K, Furusawa S. Expression of the NUP98/HOXA9 fusion transcript in the blast crisis of Philadelphia chromosome-positive chronic myelogenous leukaemia with t(7;11)(p15;p15) Br J Haematol. 2000;109(2):423–426. doi: 10.1046/j.1365-2141.2000.02003.x. [DOI] [PubMed] [Google Scholar]
- 19.Ahuja HG, Popplewell L, Tcheurekdjian L, Slovak ML. NUP98 gene rearrangements and the clonal evolution of chronic myelogenous leukemia. Genes Chromosomes Cancer. 2001;30(4):410–415. doi: 10.1002/1098-2264(2001)9999:9999<::AID-GCC1108>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 20.Nakamura T, Yamazaki Y, Hatano Y, Miura I. NUP98 is fused to PMX1 homeobox gene in human acute myelogenous leukemia with chromosome translocation t(1;11)(q23;p15) Blood. 1999;94(2):741–747. [PubMed] [Google Scholar]
- 21.Wong KF, So CC, Kwong YL. Chronic myelomonocytic leukemia with t(7;11)(p15;p15) and NUP98/HOXA9 fusion. Cancer Genet Cytogenet. 1999;115(1):70–72. doi: 10.1016/S0165-4608(99)00085-0. [DOI] [PubMed] [Google Scholar]
- 22.Hussey DJ, Dobrovic A. Recurrent coiled-coil motifs in NUP98 fusion partners provide a clue to leukemogenesis. Blood. 2002;99(3):1097–1098. doi: 10.1182/blood.V99.3.1097. [DOI] [PubMed] [Google Scholar]
- 23.Huntly BJ, Gilliland DG. Leukaemia stem cells and the evolution of cancer-stem-cell research. Nat Rev Cancer. 2005;5(4):311–321. doi: 10.1038/nrc1592. [DOI] [PubMed] [Google Scholar]
- 24.Matte CC, et al. Graft-versus-leukemia in a retrovirally induced murine CML model: mechanisms of T-cell killing. Blood. 2004;103(11):4353–4361. doi: 10.1182/blood-2003-10-3735. [DOI] [PubMed] [Google Scholar]
- 25.Zheng H, et al. Effector memory CD4+ T cells mediate graft-versus-leukemia without inducing graft-versus-host disease. Blood. 2008;111(4):2476–2484. doi: 10.1182/blood-2007-08-109678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li N, et al. Memory T cells from minor histocompatibility antigen-vaccinated and virus-immune donors improve GVL and immune reconstitution. Blood. 2011;118(22):5965–5976. doi: 10.1182/blood-2011-07-367011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Krivtsov AV, et al. Transformation from committed progenitor to leukaemia stem cell initiated by MLL-AF9. Nature. 2006;442(7104):818–822. doi: 10.1038/nature04980. [DOI] [PubMed] [Google Scholar]
- 28.Huntly BJ, et al. MOZ-TIF2, but not BCR-ABL, confers properties of leukemic stem cells to committed murine hematopoietic progenitors. Cancer Cell. 2004;6(6):587–596. doi: 10.1016/j.ccr.2004.10.015. [DOI] [PubMed] [Google Scholar]
- 29.Wang X, Roopenian D, Martone C, Li N, Li H, Shlomchik WD. Mechanisms of cross-presentation in graft-vs-host disease. Blood. 2009;114(22):687 [Google Scholar]
- 30.Choi EY, et al. Immunodominance of H60 is caused by an abnormally high precursor T cell pool directed against its unique minor histocompatibility antigen peptide. Immunity. 2002;17(5):593–603. doi: 10.1016/S1074-7613(02)00428-4. [DOI] [PubMed] [Google Scholar]
- 31.Li H, et al. Graft-versus-host disease is independent of innate signaling pathways triggered by pathogens in host hematopoietic cells. J Immunol. 2011;186(1):230–241. doi: 10.4049/jimmunol.1002965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dillon CP, et al. RIPK1 blocks early postnatal lethality mediated by caspase-8 and RIPK3. Cell. 2014;157(5):1189–1202. doi: 10.1016/j.cell.2014.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Horowitz MM, et al. Graft-versus-leukemia reactions after bone marrow transplantation. Blood. 1990;75(3):555–562. [PubMed] [Google Scholar]
- 34.Teshima T, et al. Acute graft-versus-host disease does not require alloantigen expression on host epithelium. Nat Med. 2002;8(6):575–581. doi: 10.1038/nm0602-575. [DOI] [PubMed] [Google Scholar]
- 35.Hill GR, Crawford JM, Cooke KR, Brinson YS, Pan L, Ferrara JL. Total body irradiation and acute graft-versus-host disease: the role of gastrointestinal damage and inflammatory cytokines. Blood. 1997;90(8):3204–3213. [PubMed] [Google Scholar]
- 36.Li H, et al. Graft-versus-host disease is independent of innate signaling pathways triggered by pathogens in host hematopoietic cells. J Immunol. 2011;186(1):230–241. doi: 10.4049/jimmunol.1002965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hakim FT, et al. Upregulation of IFN-iducible and damage-response pathways in chronic graft-versus-host disease. J Immunol. 2016;197(9):3490–3503. doi: 10.4049/jimmunol.1601054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nachbaur D, Clausen J, Kircher B. Donor cytomegalovirus seropositivity and the risk of leukemic relapse after reduced-intensity transplants. Eur J Haematol. 2006;76(5):414–419. doi: 10.1111/j.1600-0609.2005.00625.x. [DOI] [PubMed] [Google Scholar]
- 39.Elmaagacli AH, et al. Early human cytomegalovirus replication after transplantation is associated with a decreased relapse risk: evidence for a putative virus-versus-leukemia effect in acute myeloid leukemia patients. Blood. 2011;118(5):1402–1412. doi: 10.1182/blood-2010-08-304121. [DOI] [PubMed] [Google Scholar]
- 40.Lönnqvist B, Ringdèn O, Ljungman P, Wahren B, Gahrton G. Reduced risk of recurrent leukaemia in bone marrow transplant recipients after cytomegalovirus infection. Br J Haematol. 1986;63(4):671–679. doi: 10.1111/j.1365-2141.1986.tb07551.x. [DOI] [PubMed] [Google Scholar]
- 41.Green ML, et al. CMV reactivation after allogeneic HCT and relapse risk: evidence for early protection in acute myeloid leukemia. Blood. 2013;122(7):1316–1324. doi: 10.1182/blood-2013-02-487074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Foley B, et al. Cytomegalovirus reactivation after allogeneic transplantation promotes a lasting increase in educated NKG2C+ natural killer cells with potent function. Blood. 2012;119(11):2665–2674. doi: 10.1182/blood-2011-10-386995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rhodes-Feuillette A, Canivet M, Champsaur H, Gluckman E, Mazeron MC, Peries J. Circulating interferon in cytomegalovirus infected bone-marrow-transplant recipients and in infants with congenital cytomegalovirus disease. J Interferon Res. 1992;Spec No:91–98. doi: 10.1089/jir.1992.1992.91. [DOI] [PubMed] [Google Scholar]
- 44.Griffiths PD, et al. Cytomegalovirus glycoprotein-B vaccine with MF59 adjuvant in transplant recipients: a phase 2 randomised placebo-controlled trial. Lancet. 2011;377(9773):1256–1263. doi: 10.1016/S0140-6736(11)60136-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Plotkin SA, et al. Effect of Towne live virus vaccine on cytomegalovirus disease after renal transplant. A controlled trial. Ann Intern Med. 1991;114(7):525–531. doi: 10.7326/0003-4819-114-7-525. [DOI] [PubMed] [Google Scholar]
- 46.Aosai F, et al. Different types of allospecific CTL clones identified by their ability to recognize peptide loading-defective target cells. Eur J Immunol. 1991;21(11):2767–2774. doi: 10.1002/eji.1830211118. [DOI] [PubMed] [Google Scholar]
- 47.Crumpacker DB, Alexander J, Cresswell P, Engelhard VH. Role of endogenous peptides in murine allogenic cytotoxic T cell responses assessed using transfectants of the antigen-processing mutant 174xCEM.T2. J Immunol. 1992;148(10):3004–3011. [PubMed] [Google Scholar]
- 48.Varela N, Munoz-Pinedo C, Ruiz-Ruiz C, Robledo G, Pedroso M, López-Rivas A. Interferon-gamma sensitizes human myeloid leukemia cells to death receptor-mediated apoptosis by a pleiotropic mechanism. J Biol Chem. 2001;276(21):17779–17787. doi: 10.1074/jbc.M100815200. [DOI] [PubMed] [Google Scholar]
- 49.Sykes DB, et al. Inhibition of dihydroorotate dehydrogenase overcomes differentiation blockade in acute myeloid leukemia. Cell. 2016;167(1):171–186.e15. doi: 10.1016/j.cell.2016.08.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Blair A, Hogge DE, Sutherland HJ. Most acute myeloid leukemia progenitor cells with long-term proliferative ability in vitro and in vivo have the phenotype CD34(+)/CD71(–)/HLA–DR–. Blood. 1998;92(11):4325–4335. [PubMed] [Google Scholar]
- 51.Syampurnawati M, et al. DR negativity is a distinctive feature of M1/M2 AML cases with NPM1 mutation. Leuk Res. 2008;32(7):1141–1143. doi: 10.1016/j.leukres.2007.11.017. [DOI] [PubMed] [Google Scholar]
- 52.Syampurnawati M, et al. HLA-DR-negative AML (M1 and M2): FLT3 mutations (ITD and D835) and cell-surface antigen expression. Leuk Res. 2007;31(7):921–929. doi: 10.1016/j.leukres.2006.09.017. [DOI] [PubMed] [Google Scholar]
- 53.Kussick SJ, et al. A distinctive nuclear morphology in acute myeloid leukemia is strongly associated with loss of HLA-DR expression and FLT3 internal tandem duplication. Leukemia. 2004;18(10):1591–1598. doi: 10.1038/sj.leu.2403458. [DOI] [PubMed] [Google Scholar]
- 54.Wetzler M, et al. HLA-DR antigen-negative acute myeloid leukemia. Leukemia. 2003;17(4):707–715. doi: 10.1038/sj.leu.2402865. [DOI] [PubMed] [Google Scholar]
- 55.Oliveira G, et al. Modeling antileukemic adoptive immunotherapy in mouse-humans chimeras to identify novel mechanisms of cancer immunoediting. Blood. 2013;122(21):2017 [Google Scholar]
- 56.Nguyen S, et al. HLA-E upregulation on IFN-γ-activated AML blasts impairs CD94/NKG2A-dependent NK cytolysis after haplo-mismatched hematopoietic SCT. Bone Marrow Transplant. 2009;43(9):693–699. doi: 10.1038/bmt.2008.380. [DOI] [PubMed] [Google Scholar]
- 57.Eljaafari A, et al. Alloreaction increases or restores CD40, CD54, and/or HLA molecule expression in acute myelogenous leukemia blasts, through secretion of inflammatory cytokines: Dominant role for TNFβ, in concert with IFNγ. Leukemia. 2006;20(11):1992–2001. doi: 10.1038/sj.leu.2404375. [DOI] [PubMed] [Google Scholar]
- 58.Irish JM, et al. Single cell profiling of potentiated phospho-protein networks in cancer cells. Cell. 2004;118(2):217–228. doi: 10.1016/j.cell.2004.06.028. [DOI] [PubMed] [Google Scholar]
- 59.Mizuno S, Emi N, Kasai M, Ishitani A, Saito H. Aberrant expression of HLA-G antigen in interferon γ-stimulated acute myelogenous leukaemia. Br J Haematol. 2000;111(1):280–282. doi: 10.1046/j.1365-2141.2000.02345.x. [DOI] [PubMed] [Google Scholar]
- 60.Munker R, Andreeff M. Induction of death (CD95/FAS), activation and adhesion (CD54) molecules on blast cells of acute myelogenous leukemias by TNF-α and IFN-γ. Cytokines Mol Ther. 1996;2(3):147–159. [PubMed] [Google Scholar]
- 61.Ferm M, Grönberg A, Tally M. IFN-γ treatment increases insulin binding and MHC class I expression in erythroleukemia cells. Immunol Invest. 1996;25(1–2):37–47. doi: 10.3109/08820139609059289. [DOI] [PubMed] [Google Scholar]
- 62.Lecchi M, Lovisone E, Genetta C, Peruccio D, Resegotti L, Richiardi P. Gamma-IFN induces a differential expression of HLA-DR, DQ and DP antigens on peripheral blood myeloid leukemic blasts at various stages of differentiation. Leuk Res. 1989;13(3):221–226. doi: 10.1016/0145-2126(89)90015-5. [DOI] [PubMed] [Google Scholar]
- 63.Dalton DK, Pitts-Meek S, Keshav S, Figari IS, Bradley A, Stewart TA. Multiple defects of immune cell function in mice with disrupted interferon-γ genes. Science. 1993;259(5102):1739–1742. doi: 10.1126/science.8456300. [DOI] [PubMed] [Google Scholar]
- 64.Badovinac VP, Harty JT. Adaptive immunity and enhanced CD8+ T cell response to Listeria monocytogenes in the absence of perforin and IFN-γ. J Immunol. 2000;164(12):6444–6452. doi: 10.4049/jimmunol.164.12.6444. [DOI] [PubMed] [Google Scholar]
- 65.White DW, Harty JT. Perforin-deficient CD8+ T cells provide immunity to Listeria monocytogenes by a mechanism that is independent of CD95 and IFN-γ but requires TNF-α. J Immunol. 1998;160(2):898–905. [PubMed] [Google Scholar]
- 66.Garvey TL, et al. Inflammatory responses to pneumovirus infection in IFN-αβR gene-deleted mice. J Immunol. 2005;175(7):4735–4744. doi: 10.4049/jimmunol.175.7.4735. [DOI] [PubMed] [Google Scholar]
- 67. Smyth GK. 23 limma: linear models for microarray data. In: Gentleman RC, Carey V, Dudoit S, Irizarry R, Huber W, eds. Bioinformatics And Computational Biology Solutions Using R And Bioconductor. New York, New York, USA: Springer; 2005:397–420. [Google Scholar]
- 68.Wu D, Smyth GK. Camera: a competitive gene set test accounting for inter-gene correlation. Nucleic Acids Res. 2012;40(17):e133. doi: 10.1093/nar/gks461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Subramanian A, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A. 2005;102(43):15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.