Abstract
The efficacy of bortezomib monotherapy in desensitizing kidney transplant candidates with preformed donor-specific antibodies remains unclear. We evaluated the effect of bortezomib on preformed antibodies and upstream components of the B cell response in a primate model sensitized by fully mismatched allogeneic skin transplants to provide mechanistic insights regarding the use of bortezomib as a means of desensitization. Bortezomib treatment given intravenously twice weekly for 1 month (1.3 mg/m2 per dose) clearly reduced the numbers of antibody-producing cells and CD38+CD19+CD20− plasma cells in the bone marrow (P<0.05), but donor-specific alloantibody levels did not decrease. We observed a rapid but transient induction of circulating IgG+ B cells and an increased number of proliferating B cells in the lymph nodes after 1 month of treatment. Notably, bortezomib treatment induced germinal center B cell and follicular helper T cell expansion in the lymph nodes. These data suggest that bortezomib-induced plasma cell depletion triggers humoral compensation.
Keywords: Bortezomib, Desensitization, alloantibody, nonhuman primate, antibody mediated rejection
Preformed donor-specific antibody (DSA) is not only a barrier to successful allocation of solid organ and bone marrow transplants but also, a major limitation for long-term graft survival.1 Widely accepted desensitization protocols include plasmapheresis (removal of Ig), IVIg (neutralizing donor-reactive Igs), and B cell depletion with rituximab. These approaches have been effective in facilitating crossmatch-positive living donor renal transplantation, thereby reducing the mortality of sensitized patients compared with similar patients who remain on dialysis.2,3 Despite these efforts, sensitized patients with a positive crossmatch have higher rates of post-transplant antibody-mediated rejection compared with negative crossmatch counterparts.4 In this regard, none of the currently used treatments achieve long-term, stable desensitization.5 It has been hypothesized that plasma cells (PCs), which are not targeted by current methods of desensitization, contribute to rebound humoral responses.
Bortezomib, a proteasome inhibitor, has been approved for treatment of multiple myeloma, a malignant PC disorder.6 Proteasome inhibition induces apoptosis of PCs.7,8 Its effectiveness in treating antibody-related diseases, such as chronic graft-versus-host disease and lupus-like autoimmune disease, has been documented in murine models and clinical settings. It has also been shown that normal human bone marrow–derived PCs are susceptible to bortezomib in vitro.9 Furthermore, bortezomib monotherapy in highly sensitized patients depletes DSA-producing cells,10 leading to application of bortezomib as a strategy to reduce alloantibody levels in patients awaiting a kidney transplant. Despite promising murine data, the efficacy of bortezomib to desensitize transplant candidates before transplantation is not convincing,2,11–16 and mechanistic studies of bortezomib in humans are naturally limited due to difficulties in sampling lymphoid compartments. In a primate model, we show that bortezomib reduces PCs in bone marrow but shows limited effect on alloantibody due to rapid humoral compensation via a robust germinal center (GC) response.
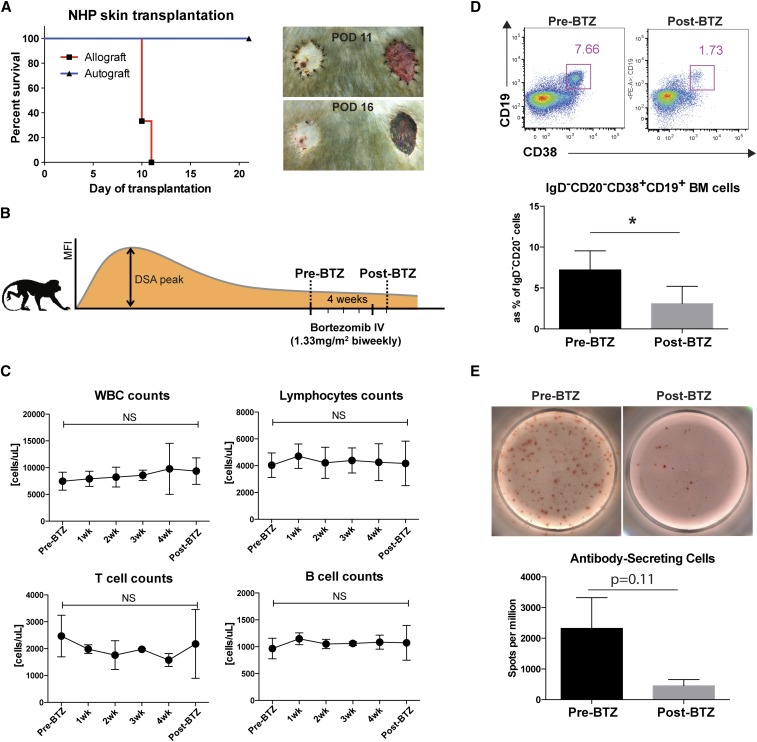
The effect of bortezomib on desensitization in a large animal model17 was assessed in four rhesus macaques sensitized by fully mismatched allogeneic skin transplants. As shown in Figure 1A, the allograft (Figure 1A, right panel) was fully rejected by post-transplant day 14, whereas the autograft was fully embedded without signs of rejection (Figure 1A). After the DSA level stabilized, we administered bortezomib twice weekly for 4 weeks at a dose of 1.3 mg/m2 (Figure 1B). On the basis of complete blood counts, biochemistry, and clinical parameters, this bortezomib dose was well tolerated. Previously, it has been reported that bortezomib depletes not only PCs but also, alloreactive T18 and activated B cells19 of humans in vitro. However, with the dosing strategy used, we did not observe global cell depletion of white blood cells, lymphocytes, monocytes, or T and B cells (Figure 1C). As expected, CD20−IgD−CD19+CD38+ cells in bone marrow were significantly reduced compared with pretreatment (Figure 1D), suggesting reduction of long-lived PCs after treatment. More importantly, we observed a reduction in the frequency of IgG-secreting antibody secreting cells (ASCs) in bone marrow post-treatment (Figure 1E). We concluded that bortezomib treatment in sensitized rhesus monkeys results in significant reduction of bone marrow PCs.
Figure 1.
Bortezomib treatment promotes bone marrow PC reduction in the skin graft of sensitized rhesus macaque. (A) Skin sensitization. Recipient animals were transplanted with abdominal skin from recipient (left panel) and donor (right panel) with no treatment. Syngeneic skin grafts (left panel) were fully accepted, whereas allogeneic skin grafts (right panel) were rejected with a mean survival time of <2 weeks. (B) Dosing strategy for desensitization. Rhesus macaques after skin grafting were treated with desensitization regimens for 4 weeks after their DSA levels stabilized. (C) Absolute white blood cells (WBCs), lymphocytes, and T and B cell counts before and after bortezomib treatment. There was no significant difference in absolute counts of WBCs, lymphocytes, and CD3 and CD20 cells between pre- and postbortezomib treatment. (D) Representative flow plot showing IgD−CD20− cells in bone marrow aspirates by flow cytometry. The frequency of the gated population (CD19+CD38+) is shown as a bar graph. *P<0.05. (E) Representative total IgG-specific ELISPOT assays from pre- and postbortezomib treatment. The frequency of IgG-producing cells in pre- and postbortezomib treatment in bone marrow was evaluated. Lower frequency of ASCs was seen after bortezomib treatment. Data are expressed as the mean±SEM of four independent experiments. BM, bone marrow; BTZ, bortezomib; IV, intravenous; MFI, mean fluorescence intensity; POD, post-transplant day.
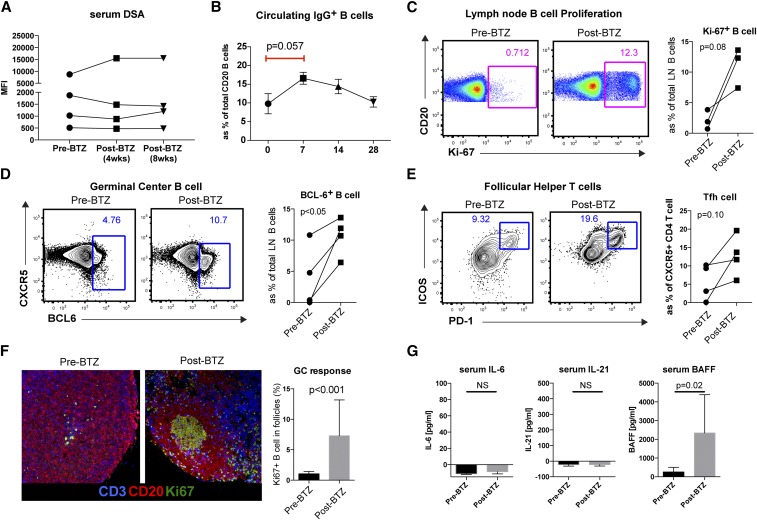
In a recent clinical trial, a bortezomib-based desensitization induced long-term DSA reduction.20 However, it is hard to identify the effect of bortezomib, because the regimen includes IVIg, rituximab, and plasmaphoresis, which are known to reduce DSA as well. Countering this, Stegall and colleagues21 reported only a modest reduction of HLA antibody with no significant change in cPRA or flow crossmatch, despite prolonged bortezomib treatment. However, it should be considered that the lack of antibody titers and the small size of the sample number might preclude the ability to detect a significant change. To assess the level of desensitization with bortezomib, circulating DSA was measured in serum by flow crossmatch. It is believed that the half-life of circulating IgG in NHP is typically around 3–4 weeks.22 Despite a reduction in ASC frequency after 4 weeks of bortezomib treatment, DSA levels did not significantly decrease in this timeframe, even when followed up to 8 weeks (Figure 2A). It is still possible that the clinical dose of bortezomib (1.33 mg/m2) is inadequate for nonhuman primates. However, the current dosing strategy clearly reduced PCs and ASCs from BM (Figure 1). Again, suboptimal dose of bortezomib might spare some PCs in LNs and spleen, while effectively reducing those in the BM. Higher doses of bortezomib could induce stable DSA reduction; however, our use of bortezomib had a very narrow safety window: high-dose use of bortezomib (0.33 mg/kg) induced acute cardiopulmonary complications in rhesus macaque (data not shown). The second generation of proteasome inhibitor, carfilzomib, might be more attractive, because it is mechanistically irreversible.23 Nevertheless, we observed a transient increase in isotype-switched circulating IgG+ B cells 7 days after bortezomib treatment (Figure 2B), which may be due to either a rapid homeostatic repopulation of memory B cells or a T cell–dependent GC-driven clonal B cell expansion. To evaluate the level of memory B cell induction, IgD−CD27+CD20+ memory B cells and IgD+CD27−CD20+ naïve B cells were measured in blood and lymph nodes. After bortezomib treatment, increased memory B cells were identified in the lymph node tissue, but this was not observed in the circulation (data not shown). To identify B cell proliferation in the secondary lymphoid organs, Ki67 staining of all B cells (CD20+), memory B cells (IgD−CD27+CD20+), and naïve B cells (IgD+CD27−CD20+) was performed. We observed an increase in proliferation of all B cells, including both naïve and memory B cell phenotypes (P=0.08).
Figure 2.
Bortezomib did not alter donor-specific alloantibody levels but increased B cell proliferation, GC B cells, Tfh cells, and GC activation in the sensitized rhesus macaques. (A) DSA level was evaluated by T cell crossmatch. Serum was assayed for IgG alloantibody level pre- and postbortezomib treatment at 4 and 8 weeks. Shown is the mean fluorescence intensity (MFI) of the entire population of the 1:100 serum dilution expressed as the mean±SEM. NS indicates >0.05. (B) Frequency of circulating IgG+CD20+ B cells at 7 days post-treatment was increased. Error bar indicates SEM. (C) B cell proliferation was evaluated with Ki67 intracellular staining. There was a trend of increased total B cells, naïve (CD27+IgD+) B cells, and memory B cells (CD27+IgD−) after bortezomib treatment compared with pretreated time points. (D) More GC-related B cells were identified with BCL-6+ intracellular staining in the lymph nodes. BCL-6+CD20+ cells in the lymph nodes increased 4 weeks after bortezomib treatment. P<0.05. (E) Flow cytometry contour plots show the ICOS+PD-1hi cell population among CXCR5+CD4+ T cells in draining lymph nodes before and after bortezomib treatment. PD-1+ICOS+ CXCR5+CD4 T cells in the lymph node showed an increasing trend after bortezomib treatment. (F) In situ GC staining with CD20, Ki67, and CD3 staining. B cell clonal expansion in GC was increased after bortezomib treatment. Original magnification, ×200. (G) The serum levels of IL-6, IL-21, and BAFF were measured with ELISA. Serum B-cell activating factor (BAFF) level was significantly increased after bortezomib treatment. Increased BAFF level after PC depletion could be a mediator for the humoral compensation. BTZ, bortezomib; LN, lymph node.
Because the induction of IgG B cell proliferation may have occurred by both homeostatic repopulation and GC responses, we tested the level of BCL-6+ B cells in the lymph nodes (transcription factor Bcl-6 is increased selectively in GC B cells). Interestingly, we did observe a significant increase in Bcl-6+ B cells in the lymph nodes after bortezomib treatment (Figure 2D). Taken together, the results of Ki67 staining and BCL-6+ B cells suggest a clonal B cell expansion by somatic hypermutation in the GC after bortezomib treatment. To confirm this observation of GC activation by bortezomib, we evaluated the follicular helper T (Tfh) cells. We observed the induction of GC-related Tfh cells (CXCR5+PD-1hiICOS+CD4+ T cells in the LN) after bortezomib treatment (Figure 2E). Finally, we confirmed in situ induction of GC (CD20/Ki67/CD3) response after bortezomib treatment through immunohistochemistry (Figure 2F).
Increased GC response after bortezomib treatment is presumably a compensatory mechanism to replace PCs depleted by bortezomib treatment and may represent a PC depletion–specific response rather than simple homeostatic repopulation. At present, the mediator responsible for this mechanism is unknown. We initially hypothesized that unconsumed IL-6 as a result of PC depletion might affect the differentiation of Tfh cells, because IL-6 is a survival mediator for PCs, while a great mediator for Tfh cell differentiation. However, we were unable to identify changes in systemic levels of IL-6 after bortezomib treatment (data not shown). Similarly, we also observed no change in serum IL-21 levels, which also have potential to be affective on Tfh cell. The lack of systemic changes of IL-6 or IL-21 does not rule out the possibility of changes in the local milieu. It may be useful to evaluate in situ expression of these cytokines, and others could mediate the response. Interestingly, serum BAFF level was significantly increased after bortezomib treatment (Figure 2G). These data suggest that BAFF is a potential mediator for this PC depletion–mediated humoral compensation. An alternative explanation for our observations might be that bortezomib-induced PC reduction abolishes the usual PC feedback inhibition of Tfh cells.24 More recently, BAFF is believed to be involved in antigen-experienced B cell survival/differentiation, whereas it used to be known to control immature B cell survival.25 The hypothesis would be that rapid PC depletion (or plasmablast) would increase intra-GC BAFF level, and this unconsumed BAFF promotes GC B cells and Tfh cells. In this study, unlike IL-6, serum BAFF level was significantly elevated after bortezomib treatment (Figure 2G). Therefore, targeting BAFF with proteasome inhibitor with anti-BAFF agents (i.e., atacicept, blimumab, or tabalumab) is a logical step to follow in testing this theory.
On the basis of the effect of bortezomib on PC and in vivo compensation, we suggest that bortezomib should not be used alone for desensitization, because bortezomib depletion of PCs will lead to a compensatory response, which is likely to be deleterious to an allograft.26 We suggest that desensitization regimens incorporating the unique effect of bortezomib should be designed on the basis of its mechanistic effects and limitations and that adjuvant B cell depletion, blocking of GC initiation, or strategies to prevent GC compensation should be considered.
Concise Methods
Male rhesus macaques (Macaca mulatta) were housed in Yerkes National Primate Research Center (Atlanta, GA). All experiments were in compliance with the Emory Institutional Animal Care and Use Committee. Full thickness abdominal skin (approximately 1-in diameter) was transplanted to the dorsal area of the recipient. DSAs in serum were assessed by flow cytometric crossmatch as previously described.17,27,28 Cells from spleen, lymph nodes, bone marrow, and blood were processed and stained with fluorochrome-conjugated antibodies as discussed in Supplemental Material. Cytometric analysis was performed using an LSRII and analyzed with FlowJo software. Total IgG secreting ASCs were visualized with ELISPOT from bone marrow cells, and serum IL-6, IL-21, and BAFF were measured by ELISA. A paired t test was used. A P value <0.05 was considered to be statistically significant.
Disclosures
None.
Supplementary Material
Acknowledgments
We acknowledge the Yerkes National Primate Research Center staff and the expert assistance of Dr. Elizabeth Strobert and Dr. Joe Jenkins for animal care.
This study was supported by National Institutes of Health grant U19AI051731 (to S.K.).
A portion of this article was presented at the 2016 American Transplant Congress held June 11–15, 2016 in Boston, Massachusetts.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
See related editorial, “Targeting Plasma Cells with Proteasome Inhibitors: Principles from Primates,” on pages 1951–1953.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2016070727/-/DCSupplemental.
References
- 1.Loupy A, Lefaucheur C, Vernerey D, Prugger C, Duong van Huyen JP, Mooney N, Suberbielle C, Frémeaux-Bacchi V, Méjean A, Desgrandchamps F, Anglicheau D, Nochy D, Charron D, Empana JP, Delahousse M, Legendre C, Glotz D, Hill GS, Zeevi A, Jouven X: Complement-binding anti-HLA antibodies and kidney-allograft survival. N Engl J Med 369: 1215–1226, 2013 [DOI] [PubMed] [Google Scholar]
- 2.Montgomery RA, Lonze BE, King KE, Kraus ES, Kucirka LM, Locke JE, Warren DS, Simpkins CE, Dagher NN, Singer AL, Zachary AA, Segev DL: Desensitization in HLA-incompatible kidney recipients and survival. N Engl J Med 365: 318–326, 2011 [DOI] [PubMed] [Google Scholar]
- 3.Jordan SC, Tyan D, Stablein D, McIntosh M, Rose S, Vo A, Toyoda M, Davis C, Shapiro R, Adey D, Milliner D, Graff R, Steiner R, Ciancio G, Sahney S, Light J: Evaluation of intravenous immunoglobulin as an agent to lower allosensitization and improve transplantation in highly sensitized adult patients with end-stage renal disease: Report of the NIH IG02 trial. J Am Soc Nephrol 15: 3256–3262, 2004 [DOI] [PubMed] [Google Scholar]
- 4.Bentall A, Cornell LD, Gloor JM, Park WD, Gandhi MJ, Winters JL, Chedid MF, Dean PG, Stegall MD: Five-year outcomes in living donor kidney transplants with a positive crossmatch. Am J Transplant 13: 76–85, 2013 [DOI] [PubMed] [Google Scholar]
- 5.Jackson AM, Kraus ES, Orandi BJ, Segev DL, Montgomery RA, Zachary AA: A closer look at rituximab induction on HLA antibody rebound following HLA-incompatible kidney transplantation. Kidney Int 87: 409–416, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.San Miguel JF, Schlag R, Khuageva NK, Dimopoulos MA, Shpilberg O, Kropff M, Spicka I, Petrucci MT, Palumbo A, Samoilova OS, Dmoszynska A, Abdulkadyrov KM, Schots R, Jiang B, Mateos MV, Anderson KC, Esseltine DL, Liu K, Cakana A, van de Velde H, Richardson PG; VISTA Trial Investigators : Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N Engl J Med 359: 906–917, 2008 [DOI] [PubMed] [Google Scholar]
- 7.Everly JJ, Walsh RC, Alloway RR, Woodle ES: Proteasome inhibition for antibody-mediated rejection. Curr Opin Organ Transplant 14: 662–666, 2009 [DOI] [PubMed] [Google Scholar]
- 8.Mujtaba T, Dou QP: Advances in the understanding of mechanisms and therapeutic use of bortezomib. Discov Med 12: 471–480, 2011 [PMC free article] [PubMed] [Google Scholar]
- 9.Perry DK, Burns JM, Pollinger HS, Amiot BP, Gloor JM, Gores GJ, Stegall MD: Proteasome inhibition causes apoptosis of normal human plasma cells preventing alloantibody production. Am J Transplant 9: 201–209, 2009 [DOI] [PubMed] [Google Scholar]
- 10.Diwan TS, Raghavaiah S, Burns JM, Kremers WK, Gloor JM, Stegall MD: The impact of proteasome inhibition on alloantibody-producing plasma cells in vivo. Transplantation 91: 536–541, 2011 [DOI] [PubMed] [Google Scholar]
- 11.Wahrmann M, Haidinger M, Körmöczi GF, Weichhart T, Säemann MD, Geyeregger R, Kikić Z, Prikoszovich T, Drach J, Böhmig GA: Effect of the proteasome inhibitor bortezomib on humoral immunity in two presensitized renal transplant candidates. Transplantation 89: 1385–1390, 2010 [DOI] [PubMed] [Google Scholar]
- 12.Guthoff M, Schmid-Horch B, Weisel KC, Häring HU, Königsrainer A, Heyne N: Proteasome inhibition by bortezomib: Effect on HLA-antibody levels and specificity in sensitized patients awaiting renal allograft transplantation. Transpl Immunol 26: 171–175, 2012 [DOI] [PubMed] [Google Scholar]
- 13.Morrow WR, Frazier EA, Mahle WT, Harville TO, Pye SE, Knecht KR, Howard EL, Smith RN, Saylors RL, Garcia X, Jaquiss RD, Woodle ES: Rapid reduction in donor-specific anti-human leukocyte antigen antibodies and reversal of antibody-mediated rejection with bortezomib in pediatric heart transplant patients. Transplantation 93: 319–324, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Patel J, Everly M, Chang D, Kittleson M, Reed E, Kobashigawa J: Reduction of alloantibodies via proteasome inhibition in cardiac transplantation. J Heart Lung Transplant 30: 1320–1326, 2011 [DOI] [PubMed] [Google Scholar]
- 15.Schmidt N, Alloway RR, Walsh RC, Sadaka B, Shields AR, Girnita AL, Hanseman DJ, Woodle ES: Prospective evaluation of the toxicity profile of proteasome inhibitor-based therapy in renal transplant candidates and recipients. Transplantation 94: 352–361, 2012 [DOI] [PubMed] [Google Scholar]
- 16.Snyder LD, Gray AL, Reynolds JM, Arepally GM, Bedoya A, Hartwig MG, Davis RD, Lopes KE, Wegner WE, Chen DF, Palmer SM: Antibody desensitization therapy in highly sensitized lung transplant candidates. Am J Transplant 14: 849–856, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Burghuber CK, Kwun J, Page EJ, Manook M, Gibby AC, Leopardi FV, Song M, Farris AB 3rd, Hong JJ, Villinger F, Adams AB, Iwakoshi NN, Knechtle SJ: Antibody-mediated rejection in sensitized nonhuman primates: Modeling human biology. Am J Transplant 16: 1726–1738, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Blanco B, Pérez-Simón JA, Sánchez-Abarca LI, Carvajal-Vergara X, Mateos J, Vidriales B, López-Holgado N, Maiso P, Alberca M, Villarón E, Schenkein D, Pandiella A, San Miguel J: Bortezomib induces selective depletion of alloreactive T lymphocytes and decreases the production of Th1 cytokines. Blood 107: 3575–3583, 2006 [DOI] [PubMed] [Google Scholar]
- 19.Mulder A, Heidt S, Vergunst M, Roelen DL, Claas FH: Proteasome inhibition profoundly affects activated human B cells. Transplantation 95: 1331–1337, 2013 [DOI] [PubMed] [Google Scholar]
- 20.Woodle ES, Shields AR, Ejaz NS, Sadaka B, Girnita A, Walsh RC, Alloway RR, Brailey P, Cardi MA, Abu Jawdeh BG, Roy-Chaudhury P, Govil A, Mogilishetty G: Prospective iterative trial of proteasome inhibitor-based desensitization. Am J Transplant 15: 101–118, 2015 [DOI] [PubMed] [Google Scholar]
- 21.Moreno Gonzales MA, Gandhi MJ, Schinstock CA, Moore NA, Smith BH, Braaten NY, Stegall MD: 32 Doses of bortezomib for desensitization is not well tolerated and is associated with only modest reductions in anti-HLA antibody [published online ahead of print July 1, 2016]. Transplantation doi:10.1097/TP.0000000000001330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mezo AR, McDonnell KA, Hehir CA, Low SC, Palombella VJ, Stattel JM, Kamphaus GD, Fraley C, Zhang Y, Dumont JA, Bitonti AJ: Reduction of IgG in nonhuman primates by a peptide antagonist of the neonatal Fc receptor FcRn. Proc Natl Acad Sci USA 105: 2337–2342, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kortuem KM, Stewart AK: Carfilzomib. Blood 121: 893–897, 2013 [DOI] [PubMed] [Google Scholar]
- 24.Pelletier N, McHeyzer-Williams LJ, Wong KA, Urich E, Fazilleau N, McHeyzer-Williams MG: Plasma cells negatively regulate the follicular helper T cell program. Nat Immunol 11: 1110–1118, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Goenka R, Scholz JL, Sindhava VJ, Cancro MP: New roles for the BLyS/BAFF family in antigen-experienced B cell niches. Cytokine Growth Factor Rev 25: 107–113, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sberro-Soussan R, Zuber J, Suberbielle-Boissel C, Candon S, Martinez F, Snanoudj R, Rabant M, Pallet N, Nochy D, Anglicheau D, Leruez M, Loupy A, Thervet E, Hermine O, Legendre C: Bortezomib as the sole post-renal transplantation desensitization agent does not decrease donor-specific anti-HLA antibodies. Am J Transplant 10: 681–686, 2010 [DOI] [PubMed] [Google Scholar]
- 27.Kim EJ, Kwun J, Gibby AC, Hong JJ, Farris AB 3rd, Iwakoshi NN, Villinger F, Kirk AD, Knechtle SJ: Costimulation blockade alters germinal center responses and prevents antibody-mediated rejection. Am J Transplant 14: 59–69, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kwun J, Page E, Hong JJ, Gibby A, Yoon J, Farris AB, Villinger F, Knechtle S: Neutralizing BAFF/APRIL with atacicept prevents early DSA formation and AMR development in T cell depletion induced nonhuman primate AMR model. Am J Transplant 15: 815–822, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.