Abstract
The necrosis- and ethylene-inducing protein 1 (Nep1)-like proteins (NLPs) are a class of microbe-associated molecular patterns widely distributed across diverse groups of plant-associated microorganisms. In spite of the cytotoxic activity in dicot plants, the role of most NLPs in the virulence of plant pathogens is still largely unknown. We showed that the MoNLP family of rice blast fungus varied very little in amino acid sequence, transient expression of three MoNLPs induced cell death and the production of reactive oxygen species in Nicotiana benthamiana, and the expression of MoNLPs was induced during infection of susceptible rice plants. To further investigate the biological role of the MoNLP family, a marker-free gene replacement vector was developed and used to knock out the whole family in Magnaporthe oryzae. Results showed no significant difference in disease levels caused by wild type and the quadruple ΔMoNLP mutant strains. Likewise, the sporulation and radial growth of the two strains were similar under various unfavorable cultural conditions including malnutrition and abiotic stresses. These observations demonstrated that the MoNLP family is dispensable for the fungal tolerance to the tested adverse cultural conditions, and more importantly, for the virulence of blast fungus on susceptible rice plants.
Introduction
Plants have developed a complex and multilayered immune system to detect and ward off invasions by diverse microbial pathogens. The first line of a plant’s surveillance system invokes the recognition of conserved molecules derived from diverse groups of microbes, also known as microbe-associated molecular patterns (MAMPs), by pattern recognition receptors (PRRs) distributed at the surface of plant cells1. An array of defense responses are subsequently induced including the production of reactive oxygen species, fortification of cell walls, upregulation of defense-related genes, and accumulation of antimicrobial compounds2, 3, which collectively confer plant resistance to adapted and non-adapted pathogens (also referred to as MAMP-triggered immunity, MTI).
A variety of microbial patterns or components have been described as MAMPs from bacteria, oomycetes, and fungi4. MAMPs from different microbial groups are normally distinct; however, a class of necrosis and ethylene-inducing protein 1 (Nep1)-like proteins (NLPs) have been reported as MAMPs from mostly plant-associated microbes across all three of the taxonomic groups5–7. The first described member of NLPs is Nep1, a 24-kDa protein that was purified from Fusarium oxysporum culture filtrates and capable of inducing necrosis and ethylene biosynthesis in dicot but not monocot plants8. The NLPs share a conserved NPP1 domain7, 9, and over 500 NLP-encoding genes have been identified based on sequence similarity analysis from microorganisms of diverse taxonomy and lifestyles10. Interestingly, the number of NLP family members can vary significantly among microorganisms. For instance, the wheat pathogen Mycosphaerella graminicola only has a single NLP gene in the genome11, whereas up to 33 copies of NLP genes have been identified from the genome of soybean pathogen Phytophthora sojae 12. In addition, studies have also demonstrated the versatile variation in sequence feature and necrosis-inducing activity across NLP family members10, 13. These observations indicate the existence of functional diversification of NLPs during complex biological processes in a broad range of microorganisms.
Apart from the ability to elicit MTI responses, the role of NLP in plant-microbe interaction is not fully understood, although the cytotoxicity of NLPs has been shown associated with the virulence of some pathogens on their dicot host plants13–15. Several observations indicate that NLPs may have roles independent of cytotoxicity: many NLPs are unable to cause plant cell death12, 13; multiple NLPs have been identified from obligate biotrophic pathogens16 as well as pathogens colonizing monocot host plants11, 17, 18. The impact of NLPs on the virulence of the pathogen under these non-cytotoxic conditions is still largely unknown. Motteram et al. (2009) have demonstrated that the only NLP gene of M. graminicola is dispensable for the fungal pathogen to cause disease on wheat plants but the role of a multi-membered NLP family in host colonization has not been reported.
Here, we describe our study on the potential biological role of NLP family in Magnaporthe oryzae, the causal agent of rice blast disease, which has four MoNLPs genes in the genome7, 17. Through a gain-of-function genetic screening, we initially identified a MoNLP gene that encoded an elicitor triggering typical immune responses in Nicotiana benthamiana. Subsequently, we investigated several aspects of the MoNLP family of the fungus including the amino acid sequence variation among lab strains and field isolates, expression profiles during infection, and the cytotoxicity of each member of the protein family. More importantly, we developed a pop-in/pop-out gene replacement vector and successfully deleted all four MoNLP genes from the genome of M. oryzae. The potential roles of MoNLPs in fungal virulence and tolerance to various adverse cultural conditions were further evaluated.
Results
Activation tagging identified an MoNLP gene from M. oryzae that elicited cell death when overexpressed in N. benthamiana plants
To search for proteinaceous elicitors from M. oryzae that can trigger immune responses in non-host plants, we modified an activation tagging vector previously used for generation of gain-of-function mutation in Arabidopsis 19, and obtained the binary vector pCB260-M (see Supplementary Fig. S1) used for construction of a genomic library of M. oryzae in Agrobacterium tumefaciens. We screened about 15,000 clones of the library by Agrobacterium-mediated transient expression and identified four clones that were capable of inducing either cell death or chlorosis in N. benthamiana plants (see Supplementary Table S1). Sequencing of the insert ends of G7, a cell death-inducing clone, revealed that the insert contained two annotated fungal genes MGG_10531 and MGG_10532 (see Supplementary Fig. S2A). To further determine which of these two genes was responsible for the cell death, restriction endonucleases that specifically disrupted the individual gene were used to truncate the G7 plasmid clone, and the transient expression assay showed that MGG_10532, which encodes the MoNLP417, was both necessary and sufficient for the G7 induced cell death in N. benthamiana plants (see Supplementary Fig. S2B). Genes responsible for the elicitor activity of other three clones were subsequently identified with a similar approach, and they were predicted to encode two hypothetical proteins and a Ras-2 protein, respectively (see Supplementary Table S1). Among these candidate elicitors, MoNLP proteins have the predicted signal peptide for protein secretion, and the function of NLPs in the microorganism is still obscure; we therefore chose the MoNLP family for further investigation.
The MoNLP family is highly conserved across strains of M. oryzae
Multiple studies have revealed that NLP family members can vary significantly in number among different microbial species (see Supplementary Table S2) although they share the highly conserved domain7, indicating the potential differentiation in NLP function associated with distinct microorganisms. Previous studies have identified four NLP-encoding genes in the blast fungus: MGG_08454 (MoNLP1), MGG_00401 (MoNLP2), MGG_02332 (MoNLP3) and MGG_10532 (MoNLP4)7, 17. We investigated the sequence variation of MoNLPs among three sequenced M. oryzae strains20 and a panel of field isolates collected from different geographic regions of China. The results showed that the MoNLP family rarely varied: MoNLP1, MoNLP2, and MoNLP4 were individually identical, and MoNLP3 shared 99.6 ~ 100% of identity across the 21 tested strains and isolates (see Supplementary Fig. S3), implying that MoNLPs may have important roles in the biology of M. oryzae.
Expression of MoNLPs triggered oxidative burst and cell death in N. benthamiana
Several NLPs have been shown to be able to induce cell death preferentially in dicotyledonous plants9, 12, 14. To investigate the MoNLP-induced cellular responses on N. benthamiana plants, cDNAs of individual MoNLP genes were cloned and engineered downstream of the inducible promoter of a modified vector of pER821, and the resulting constructs were used for Agrobacterium-mediated transient expression assay. Tissue staining of Agrobacterium-infiltrated leaves showed the production of reactive oxygen species (ROS) and the cell death of plant cells at 24 hours after treatment with the chemical inducer for constructs expressing MoNLP1, MoNLP2 and MoNLP4, but not for MoNLP3 and the GFP control (Fig. 1), indicating that MoNLPs differed in cytotoxic activity on N. benthamiana.
Figure 1.
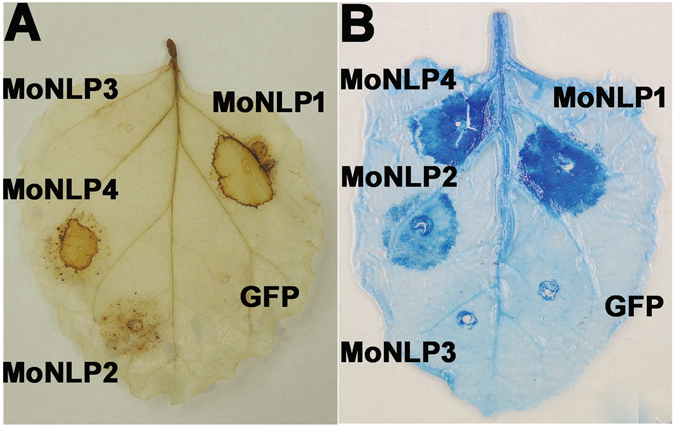
Transient expression of genes encoding necrosis and ethylene-inducing protein 1 (Nep1)-like protein (NLP) from Magnaporthe oryzae in Nicotiana benthamiana. MoNLP1, MoNLP2 and MoNLP4 trigger the accumulation of reactive oxygen species (A) and cell death (B), but not MoNLP3 and GFP control. Agrobacteria harboring individual MoNLP or GFP genes under the control of estrogen-inducible promoter were infiltrated into plant leaves. The estradiol solution (5 μM) was infiltrated into the inoculated area 24 hours post inoculation (hpi). Leaves were stained with DAB or trypan blue at 48 hpi.
Members of the MoNLP family were induced during infection of rice plant
To investigate the potential role of MoNLPs during the interaction between M. oryzae and its host plant, we inoculated susceptible rice seedlings with wild type blast fungus strain P131, and analyzed levels of MoNLP transcripts throughout the infection process by quantitative real-time PCR. Results showed that all four members of the MoNLP family were up-regulated during the infection process but with seemingly distinct induction profiles. MoNLP2 was strongly induced at 8 hours post inoculation (hpi) but subsided quickly afterward. Transcript levels of MoNLP4 were significantly up-regulated at multiple time points in both early and late stage of the infection. The up-regulation of MoNLP1 initiated from 48 hpi, when the symptom of chlorosis started to develop, whereas a significant transient induction of MoNLP3 could be observed at a rather late stage of infection (96 hpi), when infected leaf tissue had already collapsed and fungal sporulation was visible on the abaxial surface of the inoculated leaf (Fig. 2). The diversified patterns of transcripts accumulation indicated that MoNLPs were likely involved in both biotrophic and necrotrophic phases of the infection.
Figure 2.
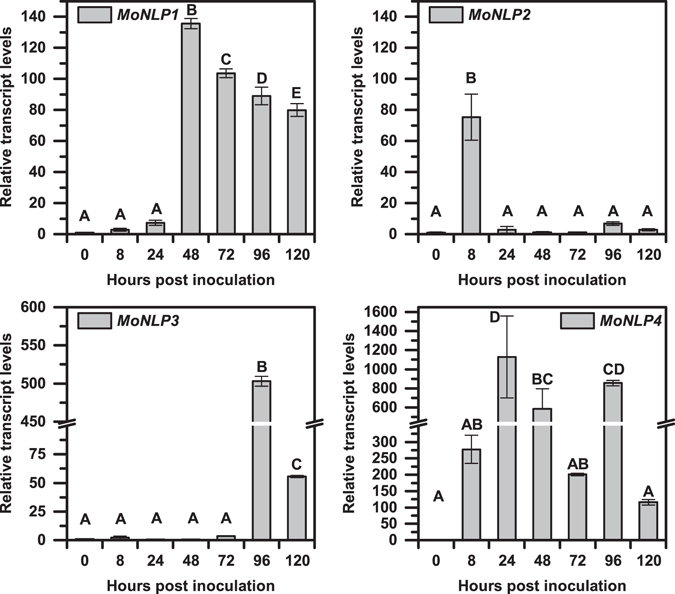
The relative transcript levels of MoNLP family are induced during infection of rice plants. Four-week-old rice plants were inoculated with M. oryzae, and samples were collected at indicated time points. The relative transcript levels of four MoNLP genes were determined by quantitative real-time polymerase chain reaction (q-RTPCR) using Actin and 40S ribosomal protein S3aE of M. oryzae as reference genes and normalized against levels at 0 hpi. The conidial inoculum was used as the 0-hour sample. Data shown are means ± sd of three replicates. Statistical differences among the samples are labeled with different letters (ANOVA, P < 0.01). The experiment was repeated twice with similar results.
Knocking out the MoNLP family through pop-in/pop-out gene replacement
To further elucidate whether the infection-associated activation of MoNLPs contributed to the virulence of blast fungus, we attempted to knock out all four members of the MoNLP family from the genome of M. oryzae. To perform the gene replacement, we firstly constructed a plasmid vector, pMFKO-DONR, which contained a hygromycin B resistance gene for positive selection, a conditional lethal HSVtk gene22 for negative selection, and the attP cassette of a pDONR vector (Gateway technology, Invitrogen) for cloning of DNA fragments required for in situ homologous recombination (Fig. 3). Subsequently, upstream and downstream DNA fragments of individual MoNLP coding regions were amplified and fused together by PCR and recombined with the vector by BP recombinase to produce pMFKO-NLP. These plasmids were used to sequentially delete all four MoNLP genes from the genome of P131 strain (Fig. 4). Interestingly, while we were constructing the quadruple mutant strainΔMoNLP 1–4, we found that knocking out either MoNLP1 or MoNLP4 had little impact on the expression of MoNLP3 during infection but simultaneous disruption of both genes strongly enhanced transcript levels of MoNLP3 at 96 hpi (Fig. 5). This synergistic effect on gene expression between MoNLPs indicated that certain levels of redundancy may exist between members of the MoNLP family during disease.
Figure 3.
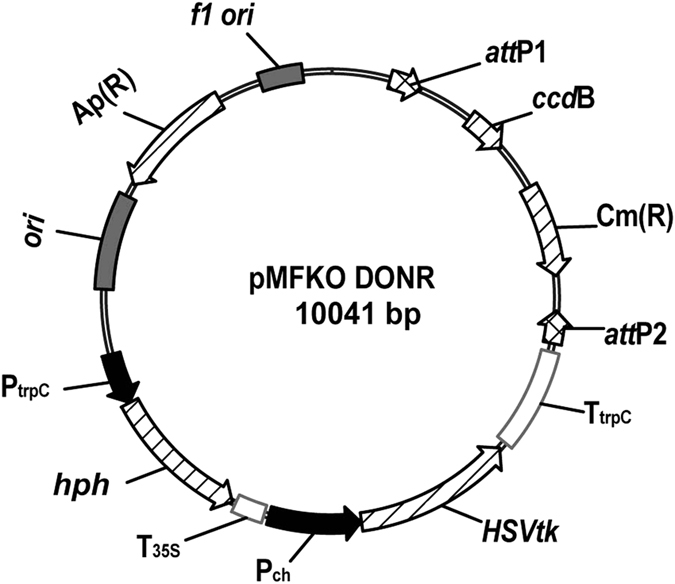
Schematic diagram of the vector pMFKO-DONR. f1 ori, phage f1 region; attP1 and attP2, Gateway sites; ccdB, coupled cell division B; Cm(R), chloramphenicol resistance gene; TtrpC, terminator of trpC gene; HSVtk, thymidine kinase of herpes simplex virus; Pch, promoter region of Cochliobolus heterostrophus unknown protein presented in GenBank Accession Number M17304; T35S, CaMV 3’UTR (polyA signal); hph, hygromycin B phosphotransferase gene; PtrpC, promoter region of trpC gene. ori, ColE1 origin of replication; Ap(R), ampicillin resistance gene.
Figure 4.
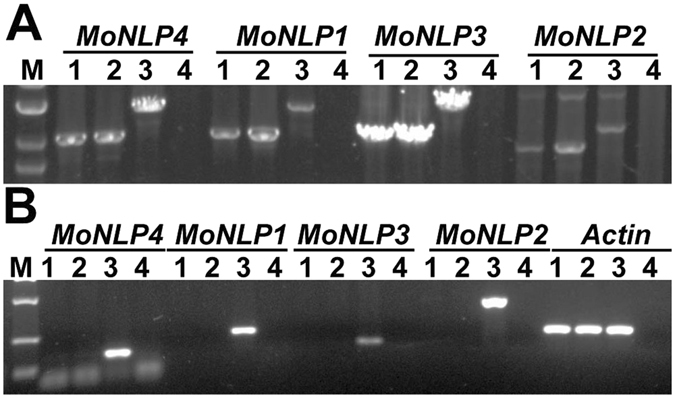
Validation of the loss-of MoNLP family in quadruple knockout strains by PCR. Genomic DNA of MoNLP knocked-out strains was amplified with specific primers annealing at sequences flanking the target regions subjected to homologous recombination (A) and with primers annealing at MoNLP coding regions (B). M indicates the molecular marker. Two independent knockout strains (1, 2), wild type M. oryzae (3), and water control (4) were assayed.
Figure 5.
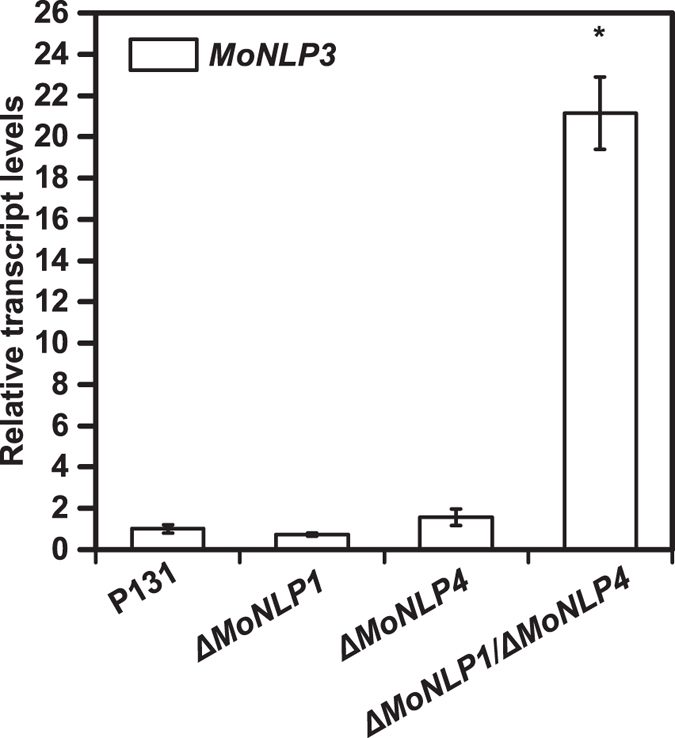
The expression of MoNLP3 is significantly enhanced in ΔMoNLP1/ΔMoNLP4 mutant during infection of rice. Leaves of 10-day-old rice seedlings were inoculated with wild type and mutants of M. oryzae, and samples were harvested at 96 hpi. Transcript levels of MoNLP3 were determined by real-time PCR using Actin as the reference gene. Data shown are means ± sd of three replicates. Asterisk indicates a significant difference from P131 (Student’s t test, P < 0.05). The experiment was repeated at least twice with similar results.
The MoNLP family is dispensable for the infection of rice plants and the growth under various stress conditions
Susceptible rice seedlings were spray-inoculated with the quadruple mutant and wild type P131 strains to investigate the potential role of MoNLP genes in fungal virulence. Five days after inoculation, similar levels of disease symptom in terms of the size and density of lesions were observed on plant leaves for both treatments (Fig. 6A). Likewise, the biomass of the quadruple mutant and wild type strains in the inoculated leaves hardly differed as indicated by the quantitative PCR analysis of the fungal DNA in diseased samples (Fig. 6B). We also inoculated detached rice leaves with conidial droplets and observed no significant difference in disease symptom and fungal biomass between quadruple mutants and wild type strains (see Supplementary Fig. S4). Moreover, we examined the growth of infectious hyphae around infection sites on the leaf sheath of rice plants. Penetration sites of single spores were classified into three categories based on the number of plant cells (1, 2–9 and > 10 cells) that the infectious hyphae had colonized. At 72 hpi, the percentages of each category of the infection sites were similar between wild type P131 and the quadruple mutant strain (Fig. 6C), indicating that deletion of the MoNLP gene family did not affect the fungal growth on leaf sheath as well. These results collectively demonstrated that the MoNLP gene family is not required for M. oryzae to infect the rice plant.
Figure 6.
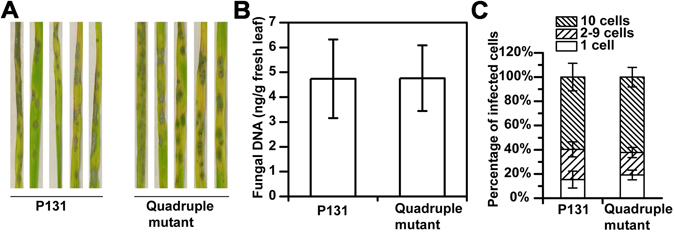
The MoNLP family is dispensable for virulence of M. oryzae on rice plants. (A) Disease symptoms caused by wild type and mutant fungal strains are similar on rice leaves. Ten-day-old susceptible rice seedlings (cv. LTH) were sprayed with conidial suspensions (1 × 105 conidia ml−1) and incubated at room temperature with high relative humidity. Leaves were photographed at 5 days post inoculation. (B) The fungal growth of quadruple mutant is similar to that of wild type during infection. Samples were collected and weighted at 120 hpi, and DNA was extracted for qPCR assay of the fungal Actin. The fungal growth was quantitatively determined by the amount of fungal DNA in fresh leaves. (C) Percentages of types of infection sites classified by number of cells occupied by infectious hyphae on leaf sheath. The hollow space of sheaths (6.5 leaf stage) was injected with conidial suspension (5 × 103 conidia ml−1). The infected tissues were stained with lactophenol cotton blue at 72 hpi. At least 40 infection sites were examined for each of the four plants used. Data shown in (B) are means ± sd (n = 4), and in (C) are means ± se (n = 4). Experiments were repeated at least twice with similar results.
To determine whether the MoNLP family was required for the growth of M. oryzae, wild type and the quadruple mutant strains were grown on CM medium, 1.2% agar, Czapek-Dox medium, and nitrogen or carbon starvation medium. Results showed that the radial growth of the quadruple mutant was similar to that of wild type strain on these media (see Supplementary Fig. S5A). In addition, both strains produced similar levels of conidia when cultured on the oatmeal-tomato agar (OTA) medium (see Supplementary Fig. S5B). These findings suggested that the MoNLP family was not central to the growth and sporulation of M. oryzae under these conditions.
To examine the role of MoNLP family in the fungal response to diverse stress conditions, wild type and the quadruple mutant strains were grown on rich medium supplemented with SDS, congo red, 1.2 M sorbitol, and the iron chelator diethylenetriamine pentaacetic acid (DTPA), respectively. Radial growth of the quadruple mutant was found similar to that of the wild type strain (see Supplementary Fig. S6A), indicating that disruption of the MoNLP family did not compromise the integrity of fungal cell wall and membrane, and the tolerance to osmotic stress or iron limitation. Likewise, the growth of the two strains showed no difference under diverse pH (5.0 and 8.0) (see Supplementary Fig. S6A) or temperatures (15 °C and 32 °C) (see Supplementary Fig. S6B), suggesting that the gene family is not required for tolerance to these adverse conditions.
Discussion
Current understanding of the biological role of NLPs during disease process is very limited, although genes encoding this class of proteins have been found in a wide range of microorganisms. Previous studies have shown that four genes encoding NLP exist in the genome of M. oryzae 7, 17. Our analysis revealed a remarkable conservation of MoNLPs among all the tested field isolates and lab strains (see Supplementary Fig. S3), implying that these genes may be essential for the survival of M. oryzae in nature. Interestingly, we observed that transcript levels of all four MoNLPs were induced in either or both of the biotrophic and necrotrophic phases during the infection of susceptible rice plants (Fig. 2). The up-regulation of NLP gene expression during infection has been described in other plant-pathogen interactions as well. In necrotrophic fungal pathogen Botrytis cinerea, the induction of BcNep1 and BcNep2 is at early and late stage of infection, respectively23. In addition, transcript levels of most of the expressed PsNLPs in P. sojae 12, 24 and two VdNLPs in V. dahliae 13 are highly induced at the late stage of infection; in contrast, the induction of four HaNLPs in the obligate biotroph oomycete Hyaloperonospora arabidopsidis occurs at the early stage of infection16. Notably, in hemibiotroph M. graminicola, the expression of the only NLP gene is prominently enhanced during a specific period near the end of the symptomless stage of the infection process11. These observations indicate that NLPs are naturally involved in the interactions between microbial pathogens and their host plants, but the complexity of gene induction profiles implies that these proteins may have diversified roles during infections.
NLPs are often regarded as virulence factors of pathogens on dicots due to the cytotoxic activity25, 26. For instance, ectopic expression of an NLP gene from F. oxysporum in Colletotrichum coccdes, a fungal pathogen used for biological control of weeds, strongly enhances the virulence of C. coccdes on the weed Abutilon theophrasti 27; silencing several PcNLPs in P. capsici leads to reduced virulence on pepper leaves15. However, the impact of some cytotoxic NLPs on pathogen virulence on dicots appears to be host dependent. For example, VdNLP1 and VdNLP2 are two genes encoding cytotoxic NLPs from V. dahliae, and disruption of either VdNLP compromises the fungal virulence on tomato and Arabidopsis, whereas, on N. benthamiana plants, the disruption of only VdNLP1 but not VdNLP2 affects the virulence13; the fungal virulence is not affected on cotton plants even when both genes were disrupted28. Likewise, the EccNip from Erwinia carotovora subsp. carotovora is required for full virulence of bacteria on potato tuber but not in potato stem or on other tested host plants14. Moreover, other studies show that deletion of a member of NLP family in B. cinerea or F. oxysporum does not impair the fungal virulence on tomato, N. benthamiana 23, or coca plants29, suggesting that NLPs may be dispensable for the virulence of pathogens. It is also possible that in these studies additional NLP genes may compensate the loss of a member of the family, which may mask the potential phenotype of mutant strains.
Gene family expansion is a common phenomenon in nature that leads to gene redundancy and functional diversification, which have also been reported for NLPs12, 13, 16, 30. In our study, the expression of MoNLP3 was significantly upregulated at 96 hpi in the ΔMoNLP1/ΔMoNLP4 double mutant compared to single knock-out or wild type strains (Fig. 5), implying that MoNLPs may be functionally complementary during infection of rice plants. To eliminate the potential redundancy between family members, we therefore, for the first time, disrupted all four MoNLP genes in the blast fungus, and results showed that the loss of entire multi-membered MoNLP family did not compromise the fungal virulence on rice (Fig. 6). This is unexpected especially considering the extensive upregulation of MoNLPs’ expression during the infection process. However, it cannot be ruled out that other components or systems in M. oryzae may compensate the impact of the loss of MoNLP family on fungal virulence, and mask the phenotype of the quadruple mutant; alternatively, the enhanced MoNLPs’ expression may be responsive to unknown stimuli generated during the infection process rather than boosting the fungal virulence per se. Previous studies also show that deletion of the only MgNLP from M. graminicola does not affect fungal virulence on wheat11. These results collectively demonstrate that the NLP family are not essential for fungal infection of monocot plants.
Results of previous research show that NLPs from monocot-colonizing pathogens do not trigger necrosis on monocots11, 18, NLP genes are activated in obligate biotrophic pathogens during infection16, and notably, insect pathogenic fungi, non-pathogens, and other animal-related fungi also carry NLP genes in their genomes7, 10. Hence, it is reasonable to assume that NLPs may have essential roles independent of phytotoxicity during certain fundamental biological processes. It has been reported that VdNLP1 is required for the vegetative growth and production of conidiospore in V. dahliae 13, and several VdNLPs are induced at a later stage of cultivation in liquid Czapek-Dox medium or by supplementing the medium with cotton root28. However, vegetative growth and sporulation were not affected in the quadruple mutant of M. oryzae in this study (see Supplementary Fig. S4). Many NLPs have a signal peptide and are secreted outside of the cells8, 9, and evidence also shows that the expression of SsNep2 in S. sclerotiorum is upregulated by the physical properties of the contact surface31. Thus, the rich variation in NLP expression profiles may reflect the complexity of environmental cues encountered by microbes during their life cycle. Further investigation of the nature of these cues may provide helpful clues for dissecting the biological roles of MoNLPs.
In this study, we developed the vector pMFKO-DONR to delete four MoNLP genes in M. oryzae via the pop-in/pop-out method. This marker-free approach is commonly used for homologous replacement of genes in bacteria32, and is especially convenient when multiple genes are targeted for deletion. The efficiency of homologous recombination depends on where the target gene is located in the genome, and it has been reported that the average rate of targeted gene replacement is only about 7% in M. grisea 33. Our results demonstrated that this approach works well in the blast fungus. Thus, the vector pMFKO-DONR and related experimental protocols may be useful for reverse genetic study in other haploid fungi where potential gene redundancy is involved as well.
Materials and Methods
Isolation of genomic DNA
For PCR screening of in situ transformants, the genomic DNA of M. oryzae was isolated from mycelia grown in liquid CM medium for 3 days according to Cenis34. To construct the genomic library, mycelia were harvested and ground in liquid nitrogen, and DNA was extracted with cetyltrimethylammonium bromide (CTAB) according to the protocols35, 36.
Construction of M. oryzae genomic library for activation tagging
The binary vector pJFAT260 used for generation of activation tagging lines of Arabidopsis 19 was digested with SpeI and ligated to reverse the orientation of the TAIL-PCR anchor and 4 × 35 S enhancer repeats. The resulting plasmid, designated as pCB260-M, was digested with BamHI to remove part of the T-DNA region unnecessary for transient expression assay. The BamHI digested vector was ligated to size fractionated genomic DNA (about 4–8 kb) of M. oryzae derived from partial digestion with Sau3AI. The ligation products were transformed into Agrobacterium AGL1 strain by electroporation. Individual clones grown on agar plates supplemented with kanamycin (50 μg ml−1) were cultured overnight in 96-well plates at 28 °C, and stored in 40% glycerol at −80 °C for further study.
Sequence similarity analyses
Amino acid sequences of MoNLP family were aligned by Clustal W, and the percentage of identity was calculated by DNASTAR software.
Agrobacterium-mediated expression on N. benthamiana
A. tumefaciens clones were grown at 28 °C overnight in LB medium supplemented with kanamycin (50 μg ml−1) until OD600 reached 2.0. Bacterial cells were collected by centrifugation and resuspended in distilled water. The bacterial suspensions were adjusted to OD600 = 0.5 and infiltrated with a needleless syringe into the leaves of N. benthamiana.
Histochemical staining
Trypan blue staining was performed as described37. For DAB staining, samples were incubated in the DAB staining solution (1 mg ml−1, pH 3.8) overnight, and destained 5–10 min subsequently in boiling ethanol38.
The inoculated sheath was fixed using formaldehyde (ethanol-formaldehyde-acetic acid, 80:3.5:5, by vol.) as described39. The plant material was stained with lactophenol cotton blue for 6 hours at room temperature40.
Vector construction
To construct the vector pCBER DEST used for inducible expression of MoNLP genes, the attR cassette of a Destination vector compatible with the GATEWAY™ cloning technology was cloned into XhoI and SpeI sites of the target expression cassette of the pER8 vector21. This modified inducible transcription unit and the G10–90-XVE cassette of pER8 were amplified by PCR, and cloned into the HindIII and XbaI (blunted) sites, respectively, of pCB30241 to obtain pCBER DEST.
To construct pMFKO-DONR, the attP cassette of pDONR201 (Invitrogen, USA) was amplified by PCR and cloned into the pGEM-T Easy vector (Promega, USA) carrying the hygromycin B phosphotransferase gene. The resulting plasmid was linearized by PCR with primer pair pTGD For and pTGD Rev (see Supplementary Table S3), and further ligated with the negative selection marker HSVtk gene that was amplified from the plasmid pHH-542 to obtain pMFKO-DONR (Fig. 3).
To disrupt MGG_10531 in the G7 plasmid, SmaI and HindIII were used to digest the G7 plasmid, and the resulting fragment was blunted and self-ligated to obtain the plasmid clone ΔMGG_10531. Likewise, ClaI and PstI were used to digest G7 plasmid to disrupt MGG_10532, and the resulting fragment was blunted and self-ligated to obtain the plasmid cloneΔMGG_10532.
Targeted disruption of four MoNLP genes
Genomic DNA fragments over 1 kb in length flanking the coding region of the targeted MoNLP gene were amplified and fused together by PCR with gene-specific primers bearing attB sites at 5′-ends (see Supplementary Table S3). This DNA product was introduced into the pMFKO-DONR by BP recombinase to produce the vector pMFKO-NLP, which was used for the PEG/CaCl2 mediated transformation of the protoplasts of blast fungus43, 44. The transformed protoplasts were selected on 1% top agar supplemented with 250 μg ml−1 hygromycin B, and the resulting transformants were further screened for in situ homologous recombination by PCR analysis. Colonies bearing targeted integrations were transferred to OTA medium containing 5 μM 5-fuoro-2′-deoxyuridine (F2dU) to enrich cells having lost the counter-selection marker HSVtk gene. Conidia were harvested and grown on CM medium supplemented with 100 μM F2dU, and resistant candidates were subjected to PCR analysis to obtain mutants with the deletion of targeted MoNLP gene. The primers used for knocking out MoNLP genes are listed in Supplementary Table S3.
Gene expression analysis
Total RNA was extracted from about 100 mg inoculated leaves by Trizol (Generay), and cDNA was synthesized by M-MLV (Takara). Transcript levels of MoNLP family were determined by quantitative real-time RT-PCR (Takara). The genes encoding Actin and 40 S ribosomal protein S3aE of M. oryzae were used as reference genes. Each sample has three technical repeats. The experiments were repeated at least twice. Primer pairs used in this study were listed in Supplementary Table S3.
Plate assay of fungal growth, sporulation and stress tolerance
Radial growth of fungal mycelia was determined by placing 5 μl conidial suspension (1 × 104 conidia ml−1 in water) on the center of an agar plate supplemented with various ingredients45. The inoculated plates were incubated at 28 °C in darkness. The diameter of colonies was measured at 6 days. The media used for assays include the CM medium46, CM medium supplemented with 1.2 M sorbitol or 200 μg/ml congo red47, 1.2% agar, Czapek-Dox medium, nitrogen or carbon starvation medium48 and rice medium with pH 8.0 or 5.049, 1/10 CM medium supplemented with 8 μM DTPA50.
For quantification of sporulation, M. oryzae strains were cultured on OTA for 12 days51. Each strain has three repeats. Four discs (φ5 mm) harvested about 1 cm from the edge of plate were put into 2 ml tube, and 0.5 ml sterile water was added. Then, each sample was shaken vigorously for 1 minute. The conidial suspension was counted by haemocytometer.
Plant inoculation
Fresh conidia were harvested from OTA medium with sterile water containing 0.02% Tween-20 as described51. For virulence detection, about ten-day-old rice seedlings were spray inoculated with conidial suspension51. Four pots (about 20 seedlings in a pot) were treated with each strain. For microscopic observation, the hollow space of rice leaf sheath from 5-week-old plants was inoculated with conidial suspension52, 53. In order to examine the expression of MoNLP genes during infection of rice, the detached leaves of about 4-week-old rice were placed in plates with moist filter paper, and 20 μl conidial suspension (1 × 106 conidia ml−1 or 5 × 104 conidia ml−1) was dropped on the leaves. All pots and plates were placed in a moist plastic box at 25 °C for the first 24 hours in darkness, and then transferred to a chamber with a photoperiod of 16 hours under fluorescent light.
For fungal biomass assay, genomic DNA of infected leaves was isolated, and the amount of fungal DNA was quantified with qPCR as previously described54.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Electronic supplementary material
Acknowledgements
We thank Z-S Xiao and X-M Zheng for the help with genetic screen of the library of M. oryzae, and Prof. M-G Zhou from Nanjing Agricultural University for the kind gift of the pHH-5 plasmid. This work is funded by The National Natural Science Foundation of China (31571946) and The Specialized Research Fund for the Doctoral Program of Higher Education of China (20130008110005) to JF.
Author Contributions
J.F. and Y.–L.P. designed the research; Y.–L.F. performed the research, collected and analysed the data; All authors wrote the manuscript.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-04430-0
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Dodds PN, Rathjen JP. Plant immunity: towards an integrated view of plant-pathogen interactions. Nature reviews Genetics. 2010;11:539–548. doi: 10.1038/nrg2812. [DOI] [PubMed] [Google Scholar]
- 2.Tsuda K, Katagiri F. Comparing signaling mechanisms engaged in pattern-triggered and effector-triggered immunity. Curr. Opin. Plant Biol. 2010;13:459–465. doi: 10.1016/j.pbi.2010.04.006. [DOI] [PubMed] [Google Scholar]
- 3.Bigeard J, Colcombet J, Hirt H. Signaling mechanisms in pattern-triggered immunity (PTI) Mol. Plant. 2015;8:521–539. doi: 10.1016/j.molp.2014.12.022. [DOI] [PubMed] [Google Scholar]
- 4.Zipfel C. Plant pattern-recognition receptors. Trends Immunol. 2014;35:345–351. doi: 10.1016/j.it.2014.05.004. [DOI] [PubMed] [Google Scholar]
- 5.Oome S, et al. Nep1-like proteins from three kingdoms of life act as a microbe-associated molecular pattern in Arabidopsis. Proc. Natl. Acad. Sci. USA. 2014;111:16955–16960. doi: 10.1073/pnas.1410031111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Böhm H, et al. A conserved peptide pattern from a widespread microbial virulence factor triggers pattern-induced immunity in Arabidopsis. PLoS Path. 2014;10:e1004491. doi: 10.1371/journal.ppat.1004491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gijzen M, Nürnberger T. Nep1-like proteins from plant pathogens: recruitment and diversification of the NPP1 domain across taxa. Phytochemistry. 2006;67:1800–1807. doi: 10.1016/j.phytochem.2005.12.008. [DOI] [PubMed] [Google Scholar]
- 8.Bailey BA. Purification of a protein from culture filtrates of Fusarium oxysporum that induces ethylene and necrosis in leaves of Erythroxylum coca. Phytopathology. 1995;85:1250–1255. doi: 10.1094/Phyto-85-1250. [DOI] [Google Scholar]
- 9.Fellbrich G, et al. NPP1, a Phytophthora-associated trigger of plant defense in parsley and Arabidopsis. The Plant Journal. 2002;32:375–390. doi: 10.1046/j.1365-313X.2002.01454.x. [DOI] [PubMed] [Google Scholar]
- 10.Oome S, Van den Ackerveken G. Comparative and functional analysis of the widely occurring family of Nep1-Like Proteins. Mol. Plant-Microbe Interact. 2014;27:1081–1094. doi: 10.1094/MPMI-04-14-0118-R. [DOI] [PubMed] [Google Scholar]
- 11.Motteram J, et al. Molecular characterization and functional analysis of MgNLP, the sole NPP1 domain-containing protein, from the fungal wheat leaf pathogen Mycosphaerella graminicola. Mol. Plant-Microbe Interact. 2009;22:790–799. doi: 10.1094/MPMI-22-7-0790. [DOI] [PubMed] [Google Scholar]
- 12.Dong S, et al. The NLP toxin family in Phytophthora sojae includes rapidly evolving groups that lack necrosis-inducing activity. Mol. Plant-Microbe Interact. 2012;25:896–909. doi: 10.1094/MPMI-01-12-0023-R. [DOI] [PubMed] [Google Scholar]
- 13.Santhanam P, et al. Evidence for functional diversification within a fungal NEP1-like protein family. Mol. Plant-Microbe Interact. 2013;26:278–286. doi: 10.1094/MPMI-09-12-0222-R. [DOI] [PubMed] [Google Scholar]
- 14.Mattinen L, Tshuikina M, Mäe A, Pirhonen M. Identification and characterization of Nip, necrosis-inducing virulence protein of Erwinia carotovora subsp. carotovora. Mol. Plant-Microbe Interact. 2004;17:1366–1375. doi: 10.1094/MPMI.2004.17.12.1366. [DOI] [PubMed] [Google Scholar]
- 15.Feng B, et al. Characterization of necrosis-inducing NLP proteins in Phytophthora capsici. BMC Plant Biol. 2014;14:126. doi: 10.1186/1471-2229-14-126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cabral A, et al. Nontoxic Nep1-like proteins of the downy mildew pathogen Hyaloperonospora arabidopsidis: repression of necrosis-inducing activity by a surface-exposed region. Mol. Plant-Microbe Interact. 2012;25:697–708. doi: 10.1094/MPMI-10-11-0269. [DOI] [PubMed] [Google Scholar]
- 17.Mogga, V. et al. Magnaporthe oryzae effectors MoHEG13 and MoHEG16 interfere with host infection and MoHEG13 counteracts cell death caused by Magnaporthe-NLPs in tobacco. Plant Cell Rep. 1–17 (2016). [DOI] [PubMed]
- 18.Staats M, van Baarlen P, Schouten A, van Kan JA. Functional analysis of NLP genes from Botrytis elliptica. Mol. Plant Pathol. 2007;8:209–214. doi: 10.1111/j.1364-3703.2007.00382.x. [DOI] [PubMed] [Google Scholar]
- 19.Fan J, Hill L, Crooks C, Doerner P, Lamb C. Abscisic acid has a key role in modulating diverse plant-pathogen interactions. Plant Physiol. 2009;150:1750–1761. doi: 10.1104/pp.109.137943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xue M, et al. Comparative analysis of the genomes of two field isolates of the rice blast fungus Magnaporthe oryzae. PLoS Genet. 2012;8:e1002869. doi: 10.1371/journal.pgen.1002869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zuo J, Niu QW, Chua NH. An estrogen receptor-based transactivator XVE mediates highly inducible gene expression in transgenic plants. The Plant Journal. 2000;24:265–273. doi: 10.1046/j.1365-313x.2000.00868.x. [DOI] [PubMed] [Google Scholar]
- 22.Khang CH, Park SY, Lee YH, Kang S. A dual selection based, targeted gene replacement tool for Magnaporthe grisea and Fusarium oxysporum. Fungal Genet. Biol. 2005;42:483–492. doi: 10.1016/j.fgb.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 23.Arenas YC, et al. Functional analysis and mode of action of phytotoxic Nep1-like proteins of Botrytis cinerea. Physiol. Mol. Plant Pathol. 2010;74:376–386. doi: 10.1016/j.pmpp.2010.06.003. [DOI] [Google Scholar]
- 24.Qutob D, Kamoun S, Gijzen M. Expression of a Phytophthora sojae necrosis-inducing protein occurs during transition from biotrophy to necrotrophy. The Plant Journal. 2002;32:361–373. doi: 10.1046/j.1365-313X.2002.01439.x. [DOI] [PubMed] [Google Scholar]
- 25.Baroncelli R, et al. Gene family expansions and contractions are associated with host range in plant pathogens of the genus. Colletotrichum. BMC Genomics. 2016;17:555. doi: 10.1186/s12864-016-2917-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ottmann C, et al. A common toxin fold mediates microbial attack and plant defense. Proc. Natl. Acad. Sci. USA. 2009;106:10359–10364. doi: 10.1073/pnas.0902362106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Amsellem Z, Cohen BA, Gressel J. Engineering hypervirulence in a mycoherbicidal fungus for efficient weed control. Nat. Biotechnol. 2002;20:1035–1039. doi: 10.1038/nbt743. [DOI] [PubMed] [Google Scholar]
- 28.Zhou BJ, Jia PS, Gao F, Guo HS. Molecular characterization and functional analysis of a necrosis-and ethylene-inducing, protein-encoding gene family from Verticillium dahliae. Mol. Plant-Microbe Interact. 2012;25:964–975. doi: 10.1094/MPMI-12-11-0319. [DOI] [PubMed] [Google Scholar]
- 29.Bailey B, Apel-Birkhold PC, Luster DG. Expression of NEP1 by Fusarium oxysporum f. sp. erythroxyli after gene replacement and overexpression using polyethylene glycol-mediated transformation. Phytopathology. 2002;92:833–841. doi: 10.1094/PHYTO.2002.92.8.833. [DOI] [PubMed] [Google Scholar]
- 30.Soanes DM, et al. Comparative genome analysis of filamentous fungi reveals gene family expansions associated with fungal pathogenesis. PLOS ONE. 2008;3:e2300. doi: 10.1371/journal.pone.0002300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dallal Bashi Z, Hegedus DD, Buchwaldt L, Rimmer SR, Borhan MH. Expression and regulation of Sclerotinia sclerotiorum necrosis and ethylene-inducing peptides (NEPs) Mol. Plant Pathol. 2010;11:43–53. doi: 10.1111/j.1364-3703.2009.00571.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lenz O, Schwartz E, Dernedde J, Eitinger M, Friedrich B. The Alcaligenes eutrophus H16 hoxX gene participates in hydrogenase regulation. J. Bacteriol. 1994;176:4385–4393. doi: 10.1128/jb.176.14.4385-4393.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Talbot NJ, Foster AJ. Genetics and genomics of the rice blast fungus Magnaporthe grisea: developing an experimental model for understanding fungal diseases of cereals. Adv. Bot. Res. 2001;34:263–287. doi: 10.1016/S0065-2296(01)34011-9. [DOI] [Google Scholar]
- 34.Cenis J. Rapid extraction of fungal DNA for PCR amplification. Nucleic Acids Res. 1992;20:2380. doi: 10.1093/nar/20.9.2380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zolan ME, Pukkila PJ. Inheritance of DNA methylation in Coprinus cinereus. Mol. Cell. Biol. 1986;6:195–200. doi: 10.1128/MCB.6.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xu JR, Leslie JF. A genetic map of Gibberella fujikuroi mating population A (Fusarium moniliforme) Genetics. 1996;143:175–189. doi: 10.1017/S0016672300034066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ma, L., Lukasik, E., Gawehns, F. & Takken, F. L. In Plant Fungal Pathogens: Methods and Protocols (eds Melvin D. Bolton & Bart P. H. J. Thomma) 61–74 (Humana Press, 2012).
- 38.Jambunathan, N. in Plant Stress Tolerance: Methods and Protocols (ed Ramanjulu Sunkar) 291–297 (Humana Press, 2010).
- 39.Coca M, et al. Enhanced resistance to the rice blast fungus Magnaporthe grisea conferred by expression of a cecropin A gene in transgenic rice. Planta. 2006;223:392–406. doi: 10.1007/s00425-005-0069-z. [DOI] [PubMed] [Google Scholar]
- 40.Marques JPR, Soares MKM, Appezzato-Da-Gloria B. New staining technique for fungal-infected plant tissues. Turk. J. Bot. 2013;37:784–787. [Google Scholar]
- 41.Xiang C, Han P, Lutziger I, Wang K, Oliver DJ. A mini binary vector series for plant transformation. Plant Mol. Biol. 1999;40:711–717. doi: 10.1023/A:1006201910593. [DOI] [PubMed] [Google Scholar]
- 42.Zheng Z, et al. FgFim, a key protein regulating resistance to the fungicide JS399-19, asexual and sexual development, stress responses and virulence in Fusarium graminearum. Mol. Plant Pathol. 2014;15:488–499. doi: 10.1111/mpp.12108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Leung H, et al. Transformation of the rice blast fungus Magnaporthe grisea to hygromycin B resistance. Curr. Genet. 1990;17:409–411. doi: 10.1007/BF00334519. [DOI] [PubMed] [Google Scholar]
- 44.Sweigard JA, Chumley FG, Valent B. Cloning and analysis of CUT1, a cutinase gene from Magnaporthe grisea. Mol. Gen. Genet. 1992;232:174–182. doi: 10.1007/BF00279994. [DOI] [PubMed] [Google Scholar]
- 45.Santhanam, P. et al. Rhamnose synthase activity is required for pathogenicity of the vascular wilt fungus Verticillium dahliae. Mol. Plant Pathol., doi:10.1111/mpp.12401 (2016). [DOI] [PMC free article] [PubMed]
- 46.Chen, X. L., Yang, J. & Peng, Y. L. in Fungal Genomics: Methods and Protocols (eds Jin-Rong Xu & Burton H. Bluhm) 213–224 (Humana Press, 2011).
- 47.Guo M, et al. The bZIP transcription factor MoAP1 mediates the oxidative stress response and is critical for pathogenicity of the rice blast fungus Magnaporthe oryzae. PLoS Path. 2011;7:e1001302. doi: 10.1371/journal.ppat.1001302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Talbot NJ, Ebbole DJ, Hamer JE. Identification and characterization of MPG1, a gene involved in pathogenicity from the rice blast fungus Magnaporthe grisea. The Plant Cell. 1993;5:1575–1590. doi: 10.1105/tpc.5.11.1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Landraud P, Chuzeville S, Billon-Grande G, Poussereau N, Bruel C. Adaptation to pH and role of PacC in the rice blast fungus Magnaporthe oryzae. PLOS ONE. 2013;8:e69236. doi: 10.1371/journal.pone.0069236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nedialkova LP, et al. Temperate phages promote colicin-dependent fitness of Salmonella enterica serovar Typhimurium. Environ. Microbiol. 2016;18:1591–1603. doi: 10.1111/1462-2920.13077. [DOI] [PubMed] [Google Scholar]
- 51.Peng YL, Shishiyama J. Temporal sequence of cytological events in rice leaves infected with Pyricularia oryzae. Can. J. Bot. 1988;66:730–735. doi: 10.1139/b88-107. [DOI] [Google Scholar]
- 52.Kankanala P, Czymmek K, Valent B. Roles for rice membrane dynamics and plasmodesmata during biotrophic invasion by the blast fungus. The Plant Cell. 2007;19:706–724. doi: 10.1105/tpc.106.046300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Koga H, Dohi K, Nakayachi O, Mori M. A novel inoculation method of Magnaporthe grisea for cytological observation of the infection process using intact leaf sheaths of rice plants. Physiol. Mol. Plant Pathol. 2004;64:67–72. doi: 10.1016/j.pmpp.2004.07.002. [DOI] [Google Scholar]
- 54.Qi M, Yang Y. Quantification of Magnaporthe grisea during infection of rice plants using real-time polymerase chain reaction and northern blot/phosphoimaging analyses. Phytopathology. 2002;92:870–876. doi: 10.1094/PHYTO.2002.92.8.870. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.


