Abstract
Purpose of Review
To highlight the range of methodological approaches used to objectively measure hedonic responses to taste stimuli during the first year of life and how these behavioral responses change with experience. Challenges inherent to this type of research are discussed.
Recent Findings
Although newborns display characteristic orofacial reactivity to four of the five basic tastes, the facial expressions made and the amount of food consumed can be modified by experience: children learn to like what they are fed. In some cases changes in facial responses are concordant with infant consumption, whereas in other cases facial reactivity follows changes in intake.
Summary
Together with ingestive measurements, precise and objective measurements of orofacial reactivity provide an understanding of how early experiences shift the hedonic tone of the taste of foods, the foundation of dietary preferences.
Keywords: distaste, pleasure, liking, taste, flavor, facial expressions
1. Introduction
As suggested by Darwin over a century ago, “We can learn much about humans from the microstructure of their behavioral affective reactions” [1]. Over the past fifty years research has indeed demonstrated that spontaneous facial expressions speak an unequivocal language that provides a window into emotional experiences [2, 3]. Paul Ekman’s research has shown that, by manifesting characteristic facial expressions, humans universally communicate the basic emotions of fear, anger, sadness, surprise, happiness (which includes sensory pleasure), and disgust [4]
Disgust, which has been considered a basic emotion since the second century [5], is defined as a feeling of revulsion or strong disapproval aroused by something unpleasant or offensive [6]. According to Paul Rozin and colleagues, the basic emotion of “core disgust” represents a culturally based conceptual rejection of an item that is associated with contamination. It is believed to originate from distaste, a basic biological motivational system that serves to reject offensive-tasting foods from the body [7]. In humans, the characteristic facial expressions that coincide with the experience of disgust and distaste include behaviors such as gaping and nose wrinkling, which are usually elicited by nausea or revulsion. These negative expressions are typically evoked by unpalatable tastes, such as bitter, both in children [e.g., 8, 9, 10, 11] and in adults [12]. Palatable tastes, such as sucrose, are thought to induce sensory pleasure, which elicits less frequently expressed appetitive reactions, such as facial relaxation and smiling [8, 9, 10] and sucking movements [8, 9, 10, 12].
While a variety of methodological tools are available to measure hedonic responses in older children and adults, many of these measures are not available for young children, who have limited language and cognitive abilities. Thus, orofacial displays to chemosensory taste stimuli have been especially useful in assessing affective responses in human infants, as well as in nonhuman animals [13]. In this article, we review the literature on the ontogeny of hedonic responses, as determined by orofacial reactivity, to the taste component of foods, which is a major determinant of food choice and acceptance, especially among children [14, 15, 16]. To demonstrate the important role that early sensory experiences play in shifting hedonic responses, we highlight research that focuses on orofacial reactivity in infants from within hours after birth (hereafter referred to as newborns) until twelve months of age. Although infants younger than one year have not yet learned to control and mask their facial expressions to conform to societal norms [17], methodological approaches nonetheless need to control for orofacial imitation, which is evident early in life [18]. These and other methodological issues that should be considered when measuring and coding orofacial reactivity among human infants will also be highlighted.
1. Ontogeny of Taste Perception and Its Evolutionary Significance
Taste, a powerful determinant of human ingestive behavior throughout the life span, is mediated by taste buds in the periphery and in multiple brain areas that are phylogenetically well conserved. Relative to other sensory capacities, the sense of taste emerges early in the human fetus. Just eight weeks after conception taste buds begin to appear, and by the thirteenth to fourteenth week they begin to morphologically resemble those of adults. Behavioral studies suggest that by the last trimester taste buds are capable of detecting tastes and communicating information to structures within the central nervous system responsible for organizing and controlling affective behaviors [19, 20].
The sensation of taste, which can be categorized into the five basic tastes of sweet, sour, salt, bitter, and umami, has taken on great interest in recent years as a major determinant of food acceptance patterns among children. Taste serves as a powerful stimulus for eliciting affective responses because it plays a critical role as the gatekeeper of the body, guarding against consumption of dangerous substances (e.g., bitter) while encouraging consumption of mother’s milk and other energy-containing foods (e.g., sweet) [21]. Similarly, preference for salty tastes (which develops during infancy) and for savory tastes is thought to attract us to foods such as salty-tasting minerals and foods rich in vitamins and protein that are important for growth and development. Although children are born with an inborn dislike for sour tastes, for some this initial negative response transforms into a preference, related to intake of sour-tasting foods such as fruit [22].
From an evolutionary perspective, inborn hedonic facial expressions to tastes and flavors play an important adaptive role, allowing infants to convey information to caretakers about the sensory characteristics of foods [23]. Displays of gaping in response to bitter tastes are visually striking and are readily identified by caregivers [24, 25]. Positive responses of sucking and facial relaxation reflect preferences and encourage the feeding of energy-producing nutrients that are important for growth and development [26].
3. Orofacial Reactivity to Taste in the Newborn
A. Measuring Orofacial Reactivity in Infants
Jacob Steiner, Judy Ganchrow, and colleagues were among the first to systematically describe orofacial reactivity to tastes in human infants and non-human animals. Although Steiner’s early studies did not provide fine-grained analyses of infants’ behaviors, after the development of the Facial Action Coding System (FACS) in the late 1970s [27], researchers began to analyze the microstructure of infants’ facial expressions in response to chemosensory stimuli [11]. With this coding system, virtually any visible facial expression can be dissected into its constituent action units (AUs), which correspond to contractions or relaxations of facial muscles that lead to characteristic movements of the face. For example, orofacial displays of distaste may involve movements in the upper part of the face, such as brow lowering (AU 4), brow raising (AU 1 and/or AU 2), and cheek raisers (AU 6) hereafter referred to as squints; midface movements, such as nose wrinkling (AU 9); and lower face movements, such as upper lip raising (AU 10), lip puckers (AU 18), and gapes (AU 26 + 27) (see Figure 1). In contrast, sensory displays of pleasure may involve lower face movements such as smiles (AU 12).
Figure 1.
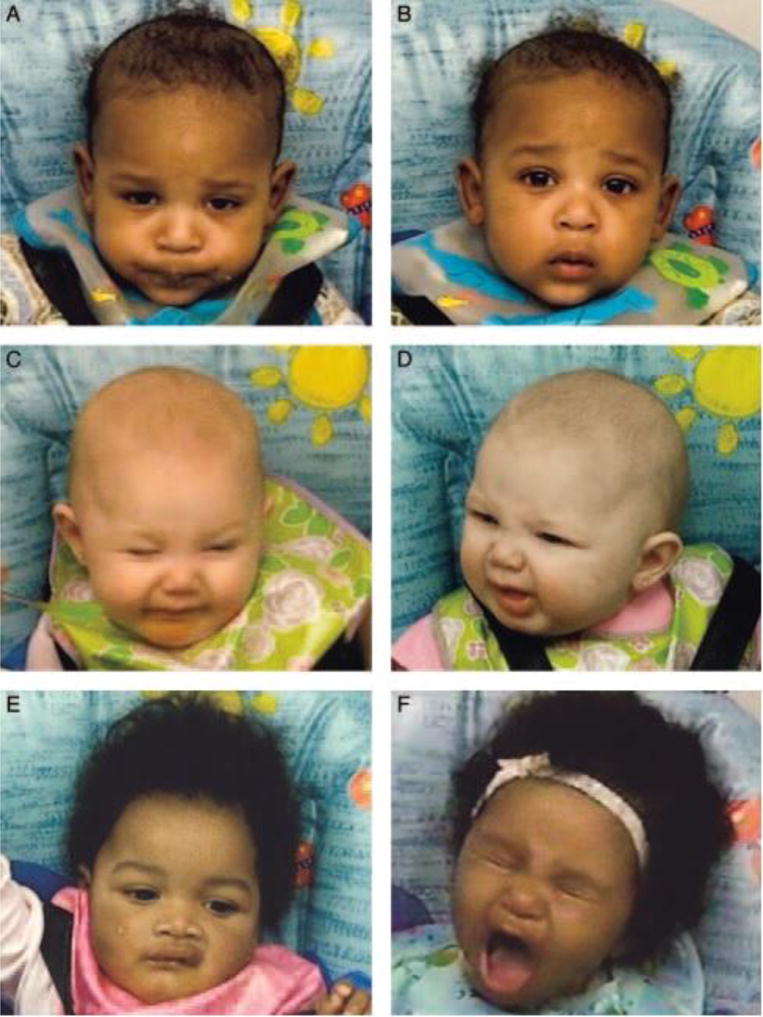
Facial expressions of distaste: brow lowerer; AU 4 (A), inner brow raise AU 1 (B), cheek raiser AU 6 (C), nose wrinkle AU 9 (D), upper lip raise AU 10 (E), and gape AU 26 + AU 27(F).
Reproduced with permission from Pediatrics, Volume 120, Pages 1247–54, Copyright © 2007 by the American Academy of Pediatrics (AAP).
There is considerable variation in methods to assess infants’ hedonic responses and in reporting of results. While early studies provided global descriptions of infants’ facial expressions, such as “smiling,” “gaping,” and “squinting” [e.g., 8, 9, 10, 28], later studies used video analyses to quantify orofacial reactivity with FACS. These studies either reported the frequency of infants who displayed each AU either alone or in combination with other AUs [e.g., 11] or reported the mean numbers of each type of AU separately or in combination by summing orofacial displays of distaste or pleasure [e.g., 29].
B. Descriptions of Orofacial Reactions to Tastes in Infants
Similar to other primates [13], human infants do not enter the world with a taste palette that is a blank slate. Rather, they can distinguish between and differentially respond to the five basic tastes with distinctive orofacial responses. Given the extensive prenatal development of the taste systems, it is not surprising that newborns are sensitive and responsive to taste stimuli after birth. In Steiner’s pioneering studies, when a 0.5 ml drop of sweet-, sour-, bitter-, or umami-tasting solution was placed on a newborn’s tongue, the infant responded with characteristic and differential facial responses [8, 9, 10, 28]. When tasting sweet (0.73 M sucrose), infants’ faces relaxed and they began suckling and smiling, consistent with greater intake in newborns of sweet-tasting solutions (0.05–0.30 M sucrose, glucose, lactose, and fructose) relative to water [30].
Later work demonstrated that, when tasting soup broth containing the basic taste of umami (0.1% and 0.5% monosodium glutamate (MSG)), newborns responded in a manner similar to that for sweet solutions: increased sucking, mouthing responses, and facial relaxation [28]. Later research demonstrated that infants preferentially consumed umami taste (0.05–0.40% MSG) when presented in soup broth relative to broth alone [31, 32]. However, they rejected MSG when it was presented in water [reviewed in 33]. Thus, it appears that, unlike sweet tastes, the taste of umami substances must be experienced in the context of other chemosensory stimuli to be considered palatable by infants. It has been suggested that MSG is a “flavor enhancer,” increasing the palatability of flavors it is mixed with [33].
Steiner found that, in contrast to their reactions to sweet and umami tastes, newborns gaped when a bitter solution (0.0003 M quinine sulfate) was presented. Moreover, as the concentrations of bitter solutions increased (0.15–0.25 M urea), the intensity of gaping increased [34]. However, intake studies revealed that newborns consumed similar amounts of 0.18–0.48 M urea in a weak sucrose solution when compared to the weak sucrose solution alone—rejection of this bitter substance does not appear until infants are approximately 2 weeks of age [35]. Thus, there may be postnatal maturation in the ability to regulate intake of urea solutions.
Steiner [10] also found that, in response to sour solutions (0.12 M citric acid), infants squinted and pursed their lips. When citric acid (0.003–0.024 M) was added to a weak sweet diluent (0.07 M sucrose), consumption of the solution was reduced when compared to the diluent alone [36], suggesting that at these concentrations of citric acid are unpalatable to newborns. However, we have all witnessed the young infant make these facial expressions while avidly sucking a lemon; whether there are individual differences in avidity for extreme sour, like there is for older infants [22] and children [37] remains unexplored.
Differential responses to sweet, bitter, sour, and umami solutions similar to those observed in normal full-term infants were also observed in anencephalic infants (i.e., those with a neural tube defect in which they are missing the cerebrum and cerebellum). These findings suggest that these orofacial responses to taste stimuli are mediated in the hindbrain and not in the cerebral cortex, where voluntary movement is controlled [8, 9, 10, 28]. Steiner and his colleagues additionally demonstrated that similar responses are observed across a wide range of species [38, 39, 40, 41, 13], suggesting that certain affective reaction components to taste may have developed early in vertebrate evolution [13].
C. Quantification of Orofacial Reactions to Tastes in Infants by FACS
More than a decade after Steiner first reported his findings with newborn infants, Diana Rosenstein and Harriet Oster [11] employed a variation of FACS, called Baby FACS which was developed by Oster, to objectively quantify neonates’ facial responses. This study revealed that, when initially tasting a sweet substance (0.73 M sucrose), infants transiently showed negative midface actions, such as cheek raising (AU 6) or nose wrinkling (AU 9). This was followed by more positive and sustained responses of facial relaxation and sucking, similar that reported by Steiner. However, Rosenstein and Oster did not observe smiling (AU 12) in response to sweet tastes. When tasting sour solutions (0.12 M citric acid) and bitter solutions (0.0003 M quinine sulfate), infants reacted mainly with actions of the lower face region. For example, sour solutions elicited lip pursing (AU 18), and bitter solutions elicited gaping (AU 26 and AU 27).
Unlike for sweet, sour, and bitter, the story for salt was more complex. Rosenstein and Oster reported no distinctive facial expression in response to salt (0.73 M NaCl), which elicited only diffuse mouth and lip movements, such as mouth gaping (AU 26 and 27) and lip pursing (AU 18), and occasional negative upper- and midface actions. In contrast, a later study reported that normal infants displayed both positive and negative orofacial reactions to 0.1–0.2 M NaCl solutions, and those who had been prenatally exposed to maternal dehydration, as a result of morning sickness, showed fewer negative orofacial reactions [42]. Consistent with Rosenstein and Oster’s findings, newborns do not differentially ingest salty solutions (0.05–0.20 M NaCl) when presented in a weak (0.07 M) sucrose diluent [36], but preferences for salty solutions develop by 6 months of age [43, 44].
D. Summary: Orofacial Reactivity to Taste in Infants
Taken together, these findings demonstrate that newborns can discriminate the basic tastes of sweet, sour, bitter, and umami and that the lack of reactivity to salt is consistent with a postnatal maturation of salt taste. The convergence of research findings in this area supports the conclusion that the inborn preference for sweets and umami and rejection of bitter and sour tastes reflect the basic biology of human infants. These preferences and aversions, which are expressed through orofacial and consummatory responses, are consequences of evolutionary selection that encourages consumption of high-nutrient foods and discourages consumption of poisonous plants.
4. Early Sensory Experiences Modify Orofacial Reactivity and Acceptance
As will be reviewed below, dietary experiences during early life are an essential part of learning to like and accept the tastes and flavors of foods inherent to one’s food environment and culture.
A. Effect of Early Milk Feedings
The early postnatal diet is unique in that it is typically solely milk based, consisting of breast milk, artificial milk (formula), or both. However, infant formulas are not homogeneous; a main difference between the types of formula available on the market (e.g., cow milk formula (CMF) extensively protein hydrolyzed formula (EHF)) is the form of their protein. Unlike the intact protein found in CMF, the milk proteins in EHF are treated with enzymes to break down peptide bonds to lessen the burden of digestion, resulting in higher concentrations of small peptides and free amino acids [45]. We have used the striking differences in taste among the different formulas as a model system to understand how the earliest feeding experiences modify orofacial reactivity to and intake of the basic tastes. In particular, we focused on extensively hydrolyzed protein formula (EHF), which is often fed to infants with cow’s milk protein allergies or intolerance. The higher levels of small peptides and free amino acids found in EHF result in prominent savory, bitter, and sour taste sensations when compared to CMF [29]. Based on these pronounced flavor differences in the milk infants feed, we hypothesized that repeated exposure to EHF versus CMF would differentially modify infants’ acceptance of the basic tastes of sour, bitter, and umami. We also compared responses of both groups of formula-fed infants to those of infants fed breast milk (BM).
In one study, 4- to 9-month-old infants who were either exclusively fed BM, CMF, or EHF were tested on six occasions to measure their acceptance of the basic tastes in a cereal matrix: sweet (0.56 M D-lactose), salty (0.1 M NaCl), bitter (0.24 M urea), savory (0.02 M MSG), sour (0.006 M citric acid), and plain cereals on separate days (Mennella, Forestell, Morgan, & Beauchamp, 2009). As hypothesized, EHF-fed infants ate significantly more savory-, bitter-, and sour-tasting and plain cereals and displayed fewer facial expressions of distaste during the feeding. They squinted (AU 6) less and tended to make fewer facial responses of distaste overall, compared with the BM-fed infants while they were fed the bitter- and savory-flavored cereals. Although 38% of the BM-fed infants and 25% of the CMF-fed infants gaped (AU 26 and AU 27) while eating the bitter-flavored cereal, none of the EHF-fed infants made this facial response of distaste. Moreover, the BM- and EHF-fed infants were more likely than the CMF-fed infants to smile (AU 12) while eating the savory cereal, which likely reflects their exposure to the high concentrations of free glutamate found in human breast milk [46, 47] and EHF [45]. Taken together, these data reveal that the tastes to which infants are exposed during formula feedings will depend on the type and brand of formula they are fed, which will in turn affect infants’ liking and acceptance of foods at weaning.
B. Repeated Exposure to Solid Foods
The convergence of findings from several experimental studies indicates that repeated exposures to a food (i.e., eight to ten tastes familiarize infants to that food and increase their willingness to consume it [24, 48, 49, 50]. Merely looking at the food is not sufficient; rather, the infants must taste the food to learn to like it [51].
To date few studies have reported on how early exposure to fruits and vegetables changes infants’ hedonic orofacial responses to these foods at weaning [e.g. 24]. In one study, one group of infants was fed only green beans (Group GB) and another was fed peaches after the green beans (Group GB-P) each day for eight days. Although both groups increased their intake of green beans, only those in Group GB-P displayed fewer facial expressions of distaste after just eight exposures. Thus, increased intake does not always coincide with increased liking, and how quickly infants learn to like a target food depends on the other foods with which it is presented—it might take longer to “change the face” when a food is presented alone. Another study that assessed maternal ratings of infants’ hedonic responses suggested that ten presentations may be sufficient to increase liking [50].
Based on this research, it seems that mothers may give up too soon when introducing foods that are initially disliked because they react to infants’ facial expressions of distaste made during feeding. Instead, upon initial exposure to a food they should focus on their infant’s willingness to eat the food (e.g., does their infant open their mouth when a spoonful of food is offered). As they continue to expose their infant to the food, they will see shifts in facial expressions that mirror changes in intake—exposure needs to be of sufficient duration to produce shifts in liking.
5. Methodological Issues
Individual AUs and global facial expressions are objective measures of infants’ hedonic responses to tastes and reflect infants’ initial responses to these foods, as well as changes in those responses through flavor learning. Recent studies that measure orofacial responses to tastes typically involve frame-by-frame video analyses [52] to quantify the actual number of affective reactions that infants express over the first two minutes of feeding, as a measure of the valence and intensity of affective reactions [16]. In our research, we have controlled for individual differences in rates of feeding and orofacial expression by focusing on the total number of facial expressions of distaste made for each spoonful of food offered, as well as the incidence of specific facial responses. This often involves multiple observations of the videos to fully capture the rich array of transient facial expressions that may occur on different parts of the face simultaneously. Individuals who are certified in FACS analyze the videos, and the reliability between individuals’ scores must be established. As a result, this approach can be time-consuming. Although the FACS manual [53] has been designed to be self-instructional, typically it takes 50–100 hours to prepare for the final FACS certification test.
Most of the studies we have conducted to measure orofacial responses in infants have involved multiple trials conducted in experimental settings. It is therefore important for test sessions to occur at approximately the same time of day, and optimally at a time when the infant is hungry. To ensure that testing objectively measures infants’ behavioral responses to a food, our test procedures allow infants to determine the pace and duration of each meal and the amount consumed (infant-led feeding). Testing procedures that allow mothers to determine when to end the feeding session (mother-led feeding) do not accurately measure infants’ food acceptance because some mothers may either under- or overfeed their infant by not attending to their infant’s satiety cues [e.g. 54, 55, 56].
Because infants are sensitive to and imitate orofacial responses of adults [18], we required mothers to wear a fabric mask over the lower part of their face and to not talk or express emotions while feeding. This practice ensures that infants’ facial responses accurately reveal their reactivity to the flavor of the food rather than merely imitate their mother’s responses. Prior to testing, mothers are asked to use the mask at home while feeding to ensure that their infants acclimatize to it. Despite this, the use of the mask may be construed as a limitation because it does not reflect the daily feeding environment experienced by the child. However, we caution that testing procedures that allow mothers to freely interact and display emotional expressions while feeding are potentially biased. Therefore, studies that fail to control for mothers’ behaviors during the session should, at the very least, objectively measure mothers’ orofacial reactivity behaviors and control for them in the final analyses.
While orofacial responses are especially useful as a reliable measure of preverbal infants’ hedonic responses to tastes [reviewed 16], we caution that orofacial reactivity responses to tastes may not be as reliable for older children, or adults, because as children mature they learn to control and manage their facial expressions to satisfy rules of display consistent with societal norms [17, 57, 58]. Because of such emotional masking, attempts by older children to conceal or exaggerate their actual responses to particular tastes may lead to biased or unreliable data [59].
Although individuals attempt to manage their facial responses, transient expressions (or microexpressions) that reflect their true emotions often “leak” into their overall expression [57]. These microexpressions are difficult to observe because they are often subtle and transient; however, they can be detected using facial electromyography, which measures the electrical activity of facial muscles and can detect movements that are too discreet for the eye. This procedure has been used to measure responses to tastes in older children [60, 61, 62, 63].
6. Conclusions
Because we are what we eat and we eat what we like, understanding how children learn to like the flavor of foods is an important aspect of infant nutrition [64]. The convergence of findings from studies that employ precise and detailed measurements of orofacial responses and infant-led measures of intake provides scientists with a rich understanding of the factors involved in the development of learned flavor preferences, which have their origin during infancy. Like adults, newborn infants are well equipped to convey a wide range of hedonic responses to tastes and flavors [65]. As reviewed herein, while these initial responses are primarily inborn and are a function of infants’ basic biology, the inherent plasticity of the chemosensory system interacts with early experiences to ensure children are not restricted to a narrow range of foodstuffs. The flavors of milk, whether from formula or from breast milk, and the flavors of complementary foods expose young children to the foods and flavors that are part of their cultural cuisine, facilitating acceptance. These early sensory experiences establish food patterns during the first years of life that set the stage for lifelong dietary habits [66].
Human and Animal Rights and Informed Consent.
All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards (including the Helsinki declaration and its amendments, institutional/national research committee standards, and international/national/institutional guidelines).
Footnotes
Compliance with Ethics Guidelines
Conflict of Interest
Catherine A. Forestell and Julie A. Mennella declare they have no conflict of interest.
References
- 1.Darwin C. The expression of the emotions in man and animals. Oxford, UK: Harper Collins/Oxford University Press; 1998. originally published in 1872. [Google Scholar]
- 2.Fridlund AJ, Gilbert AN, Izard CE, Burdett AN. Emotions and facial expression. Science. 1985;230(4726):607–8. doi: 10.1126/science.4048950. [DOI] [PubMed] [Google Scholar]
- 3.Ekman P, Friesen WV. Constants across cultures in the face and emotion. J Pers Soc Psychol. 1971;17(2):124–9. doi: 10.1037/h0030377. [DOI] [PubMed] [Google Scholar]
- 4.Ekman P, Rosenberg EL. What the face reveals: Basic and applied studies of spontaneous expression using the Facial Action Coding System (FACS) New York: Oxford University Press; 1997. [Google Scholar]
- 5.Masson JL, Patwardhan MV. Aesthetic rapture: the rasadhyaya of the natasastra. Pune, India: Deccan College; 1970. [Google Scholar]
- 6.English Oxford Living Dictionaries [internet]. Disgust [cited 2016 Dec. 15] available from: https://en.oxforddictionaires.com
- 7.Rozin P, Haidt J, McCauley CR. Disgust: The body and soul emotion. In: Dalgleish T, Power M, editors. Handbook of Cognition and Emotion. Chichester, England: John Wiley & Sons Ltd; 1999. [Google Scholar]
- 8.Steiner JE. The gustofacial response: observation on normal and anencephalic newborn infants. In: Bosma JF, editor. Fourth symposium on oral sensation and perception. Bethesda, MD: US Department of Health, Education and Welfare; 1973. [PubMed] [Google Scholar]
- 9.Steiner JE. Taste and development: the genesis of sweet preference. Washington, DC: U.S. Government Printing Office; 1977. Facial expressions of the neonate infant indicating the hedonics of food-related chemical stimuli. [Google Scholar]
- 10.Steiner JE. Human facial expressions in response to taste and smell stimulation. Adv Child Dev Behav. 1979;13:257–95. doi: 10.1016/s0065-2407(08)60349-3. [DOI] [PubMed] [Google Scholar]
- 11.Rosenstein D, Oster H. Differential facial responses to four basic tastes in newborns. Child Dev. 1988;59:1555–68. [PubMed] [Google Scholar]
- 12.Weiland R, Ellgring H, Macht M. Gustofacial and olfactofacial responses in human adults. Chem Senses. 2010;35(9):841–53. doi: 10.1093/chemse/bjq092. [DOI] [PubMed] [Google Scholar]
- 13.Steiner JE, Glaser D, Hawilo ME, Berridge KC. Comparative expression of hedonic impact: affective reactions to taste by human infants and other primates. Neurosci Biobehav Rev. 2001;25(1):53–74. doi: 10.1016/S0149-7634(00)00051-8. [DOI] [PubMed] [Google Scholar]
- 14.Birch LL. Psychological influences on the childhood diet. J Nutr. 1998;128:407S–10S. doi: 10.1093/jn/128.2.407S. [DOI] [PubMed] [Google Scholar]
- 15.Birch LL. Development of food preferences. Annu Rev Nutr. 1999;19:41–62. doi: 10.1146/annurev.nutr.19.1.41. [DOI] [PubMed] [Google Scholar]
- 16••.Forestell CA, Mennella JA. The ontogeny of taste perception and preference throughout childhood. In: Doty RL, editor. Handbook of olfaction and gustation. 3rd. Wilmington, DE: John Wiley & Sons; 2015. Provides a detailed overview of the approaches and discoveries that have contributed to our current knowledge of human development of the five basic tastes and the impact they have had on our understanding of the sensory world of children. [Google Scholar]
- 17.Hiatt SW, Campos JJ, Emde RN. Facial patterning and infant emotional expression: Happiness, surprise, and fear. Child Dev. 1979;50(4):1020–35. [PubMed] [Google Scholar]
- 18.Meltzoff AN, Moore MK. Imitation of facial and manual gestures by human neonates. Science. 1977;198(4312):75–8. doi: 10.1126/science.198.4312.75. [DOI] [PubMed] [Google Scholar]
- 19.Liley AW. Disorders of amniotic fluid. In: Assali NS, editor. Pathophysiology of gestation: fetal placental disorders. New York: Academic Press; 1972. [Google Scholar]
- 20.Maone TR, Mattes RD, Bernbaum JC, Beauchamp GK. A new method for delivering a taste without fluids to preterm and term infants. Dev Psychobiol. 1990;13:179–91. doi: 10.1002/dev.420230208. [DOI] [PubMed] [Google Scholar]
- 21.Cowart BJ. Taste, our body’s gustatory gatekeeper. Cerebrum. 2005;7:7–22. [Google Scholar]
- 22.Blossfeld I, Collins A, Boland S, Baixauli R, Kiely M, Delahunty C. Relationships between acceptance of sour taste and fruit intakes in 18-month-old infants. Br J Nutr. 2007;98:1084–91. doi: 10.1017/S0007114507749231. [DOI] [PubMed] [Google Scholar]
- 23.Plutchik R. Emotions: A Psychoevolutionary Synthesis. New York: Harper & Row; 1980. [Google Scholar]
- 24.Forestell CA, Mennella JA. Early determinants of fruit and vegetable acceptance. Pediatrics. 2007;120:1247–54. doi: 10.1542/peds.2007-0858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Forestell CA, Mennella JA. More than just a pretty face. The relationship between infant’s temperament, food acceptance, and mothers’ perceptions of their enjoyment of food. Appetite. 2012;58:1136–42. doi: 10.1016/j.appet.2012.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Coldwell SE, Oswald TK, Reed DR. A marker of growth differs between adolescents with high vs. low sugar preference. Physiol Behav. 2009;96:574–80. doi: 10.1016/j.physbeh.2008.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ekman P, Friesen W. The facial action coding system. Palo Alto, CA: Consulting Psychologists Press; 1978. [Google Scholar]
- 28.Steiner JE. What the neonate can tell us about umami. In: Kawamura Y, Kare MR, editors. Umami: A Basic Taste. New York: Marcel Dekker; 1987. [Google Scholar]
- 29.Mennella JA, Forestell CA, Morgan LK, Beauchamp GK. Early milk feeding influences taste acceptance and liking during infancy. Am J Clin Nutr. 2009;90:780–788S. doi: 10.3945/ajcn.2009.27462O. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Desor J, Maller O, Turner RE. Taste in acceptance of sugars by human infants. J Comp Physiol Psychol. 1973;84:496–501. doi: 10.1037/h0034906. [DOI] [PubMed] [Google Scholar]
- 31.Beauchamp GK, Pearson P. Human development and umami taste. Physiol Behav. 1991;49:1009–12. doi: 10.1016/0031-9384(91)90215-a. [DOI] [PubMed] [Google Scholar]
- 32.Vazquez M, Pearson PB, Beauchamp GK. Flavor preferences in malnourished Mexican infants. Physiol Behav. 1982;28:513–9. doi: 10.1016/0031-9384(82)90148-2. [DOI] [PubMed] [Google Scholar]
- 33.Beauchamp GK. Sensory and receptor responses to umami: an overview of pioneering work. Am J Clin Nutr. 2009;90(3):723S–7S. doi: 10.3945/ajcn.2009.27462E. [DOI] [PubMed] [Google Scholar]
- 34.Ganchrow JR, Steiner JE, Daher M. Neonatal facial expressions in response to different qualities and intensities of gustatory stimuli. Infant Behav Dev. 1983;6:189–200. [Google Scholar]
- 35.Kajuira H, Cowart BJ, Beauchamp GK. Early developmental change in bitter taste responses in human infants. Dev Psychobiol. 1992;25:375–86. doi: 10.1002/dev.420250508. [DOI] [PubMed] [Google Scholar]
- 36.Desor JA, Maller O, Andrews K. Ingestive responses of human newborns to salty, sour, and bitter stimuli. J Comp Physiol Psychol. 1975;89:966–70. doi: 10.1037/h0077171. [DOI] [PubMed] [Google Scholar]
- 37.Liem DG, Mennella JA. Heightened sour preferences during childhood. Chem Senses. 2003;28:173–80. doi: 10.1093/chemse/28.2.173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ganchrow JR, Oppenheimer M, Steiner JE. Displays to gustatory stimuli in newborn rabbit pups. Chem Senses. 1979;4(1):49–61. [Google Scholar]
- 39.Ganchrow JR, Steiner JE, Canetto S. Behavioral Displays to gustatory stimuli in newborn rat pups. Dev Psychobiol. 1986;19:163–74. doi: 10.1002/dev.420190303. [DOI] [PubMed] [Google Scholar]
- 40.Ganchrow JR, Steiner JE, Bartana A. Behavioral reactions in young chicks (Gallus gallus domesticus) Dev Psychobiol. 1990;23:103–17. doi: 10.1002/dev.420230202. [DOI] [PubMed] [Google Scholar]
- 41.Steiner JE, Glaser D. Differential behavioral responses to taste stimuli in nonhuman primates. J Hum Evol. 1984;13:709–23. [Google Scholar]
- 42.Crystal SR, Bernstein IL. Infant salt preference and mother’s morning sickness. Appetite. 1998;30:297–307. doi: 10.1006/appe.1997.0144. [DOI] [PubMed] [Google Scholar]
- 43.Schwartz C, Issanchou S, Nicklaus S. Developmental changes in the acceptance of the five basic tastes in the first year of life. Br J Nutr. 2009;102:1375–1385. doi: 10.1017/S0007114509990286. [DOI] [PubMed] [Google Scholar]
- 44.Stein LJ, Cowart BJ, Beauchamp GK. The development of salty taste acceptance is related to dietary experience in human infants: a prospective study. Am J Clin Nutr. 2012;95:123–9. doi: 10.3945/ajcn.111.014282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ventura AK, San Gabriel A, Hirota M, Mennella JA. Free amino acid content in infant formulas. Nutr Food Sci. 2012;42:271–278. doi: 10.1108/00346651211248638. [DOI] [Google Scholar]
- 46.Agostoni C, Carratu B, Boniglia C, Riva E, Sanzini E. Free amino acid content in standard infant formulas: comparison with human milk. J Am Coll Nutr. 2000;19(4):434–438. doi: 10.1080/07315724.2000.10718943. [DOI] [PubMed] [Google Scholar]
- 47.Baldeon ME, Mennella JA, Flores N, Fornasini M, San Gabriel A. Free amino acid content in breast milk of adolescent and adult mothers in Ecuador. Springerplus. 2014;3:104. doi: 10.1186/2193-1801-3-104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gerrish CJ, Mennella JA. Flavor variety enhances food acceptance in formula-fed infants. Am J Clin Nutr. 2001;73(6):1080–5. doi: 10.1093/ajcn/73.6.1080. [DOI] [PubMed] [Google Scholar]
- 49••.Mennella JA, Reiter AR, Daniels LM. Vegetable and fruit acceptance during infancy: impact of ontogeny, genetics, and early experiences. Adv Nutr. 2016;7(1):211S–9S. doi: 10.3945/an.115.008649. Considers the ontogeny and sources of individual differences in taste perception, such as the role of genetic variation in bitter taste perception, in order to understand the behavioral phenomena surrounding the introduction of vegetables and fruits into infants’ diets. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sullivan SA, Birch LL. Infant dietary experience and acceptance of solid foods. Pediatrics. 1994;93:271–7. [PubMed] [Google Scholar]
- 51.Birch LL, McPhee L, Shoba BC, Pirok E, Steinberg L. What kind of exposure reduces children’s food neophobia?: Looking vs. tasting. Appetite. 1987;9(3):171–8. doi: 10.1016/s0195-6663(87)80011-9. [DOI] [PubMed] [Google Scholar]
- 52.Berridge KC. Food reward: brain substrates of wanting and liking. Neurosci Biobehav Rev. 1996;20:1–25. doi: 10.1016/0149-7634(95)00033-b. [DOI] [PubMed] [Google Scholar]
- 53.Ekman P, Friesen WV, Hagar JC. Facial Action Coding System on CD-ROM. Salt Lake City, UT: Network Information Research; 2002. [Google Scholar]
- 54.Blissett J, Meyer C, Haycraft E. Maternal and paternal controlling feeding practices with male and female children. Appetite. 2006;47:212–9. doi: 10.1016/j.appet.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 55.Crow RA, Fawcett JN, Wright P. Maternal behavior during breast- and bottle-feeding. J Behav Med. 1980;3:259–77. doi: 10.1007/BF00845051. [DOI] [PubMed] [Google Scholar]
- 56.Li R, Fein SB, Grummer-Strawn LM. Do infants fed from bottles lack self-regulation of milk intake compared with directly breastfed infants? Pediatrics. 2010;125(6):e1386–93. doi: 10.1542/peds.2009-549. [DOI] [PubMed] [Google Scholar]
- 57.Ekman P, Friesen WV. Unmasking the face: A guide to recognizing emotions from facial clues. Los Altos, CA: Malor Books; 2003. [Google Scholar]
- 58.Larochette AC, Chambers CT, Craig KD. Genuine, suppressed and faked facial expressions of pain in children. Pain. 2006;126(1):64–71. doi: 10.1016/j.pain.2006.06.013. [DOI] [PubMed] [Google Scholar]
- 59.Soussignan R, Schaal B. Forms and social signal value of smiles associated with pleasant and unpleasant sensory experience. Ethology. 1996;102(8):1020–41. [Google Scholar]
- 60.Armstrong JE, Hutchinson I, Laing DG, Jinks AL. Facial electromyography: responses of children to odor and taste stimuli. Chem Senses. 2007;32:611–21. doi: 10.1093/chemse/bjm029. [DOI] [PubMed] [Google Scholar]
- 61.Epstein LH, Paluch RA. Habituation of facial muscle responses to repeated food stimuli. Appetite. 1997;29:213–24. doi: 10.1006/appe.1997.0102. [DOI] [PubMed] [Google Scholar]
- 62.Hu S, Player KA, McChesney KA, Dalistan MD, Tyner CA, Scozzafava JE. Facial EMG as an indicator of palatability in humans. Physiol Behav. 1999;68:31–5. doi: 10.1016/s0031-9384(99)00143-2. [DOI] [PubMed] [Google Scholar]
- 63.Hu S, Luo YJ, Hui L. Preliminary study of associations between objective parameters of facial electromyography and subjective estimates of taste palatability. Percept Mot Skills. 2000;91:741–7. doi: 10.2466/pms.2000.91.3.741. [DOI] [PubMed] [Google Scholar]
- 64.Mennella JA. Ontogeny of taste preferences: basic biology and implications for health. Am J Clin Nutr. 2014;99(3):704S–11S. doi: 10.3945/ajcn.113.067694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ekman P, Oster H. Facial expressions of emotion. An Rev Psych. 1979;301:527–54. [Google Scholar]
- 66.Saavedra JM, Deming D, Dattilo A, Reidy K. Lessons from the feeding infants and toddlers study in North America: What children eat and implications for obesity prevention. Ann Nutr Metab. 2013;62(S3):27–36. doi: 10.1159/000351538. [DOI] [PubMed] [Google Scholar]


