Abstract
In this study, the chemical composition and antioxidant and anti-inflammatory activities of sweet basil (Ocimum basilicum L. Lamiaceae family) were evaluated. Sweet basil is a food-related plant that is widely used in traditional Chinese medicine. Sweet basil crude oil was processed via molecular distillation and further characterized using gas chromatography–mass spectrometry (GC–MS) to screen for new compounds. The GC–MS analysis identified thirty-eight compounds. The major constituents of the residue fraction were estragole (17.06%), methyl eugenol (11.35%) and linoleic acid (11.40%), while the distillate fraction primarily contained methyl eugenol (16.96%), α-cadinol (16.24%) and α-bergamotene (11.92%). The antioxidant (DPPH and ABTS assays) and anti-inflammatory (in Raw264.7 cells) activities were evaluated. The residue fraction markedly scavenged the DPPH (IC50 = 1.092 ± 0.066 mg/mL) and ABTS (IC50 = 0.707 ± 0.042 mg/mL) radicals. Meanwhile, the distillate fraction distinctly suppressed the production of cytokines (TNF-α, IL-β, IL-6) and their gene expression in LPS-induced Raw264.7 cells and suppressed NO and iNOS in an in vitro model when compared with the crude oil. In conclusion, the fractions obtained from sweet basil crude oil showed different antioxidant and anti-inflammatory properties, and they could be used as an effective source of natural antioxidant and anti-inflammatory agents after molecular distillation. Thus, the properties of essential oils in natural herbal medicines may be maximized to provide a valuable therapeutic strategy for treating various disorders caused by extreme oxidative stress.
Electronic supplementary material
The online version of this article (doi:10.1007/s13197-017-2620-x) contains supplementary material, which is available to authorized users.
Keywords: Sweet basil, Molecular distillation, Antioxidant, Anti-inflammatory
Introduction
Oxidative stress is defined as the imbalance between the occurrence of reactive oxygen species (ROS) and the organism’s capacity to offset their action through antioxidative protection systems (Persson et al. 2014). It is certain that free radical-induced damage during oxidative stress gives rise to the pathogenesis and pathophysiology of many chronic health problems, such as Parkinson’s and Alzheimer’s diseases and cardiovascular and inflammatory diseases (López-Alarcón and Denicola 2013; Maulik et al. 2013; Toda 2011). The connection between chronic inflammation and oxidative stress is already known. Basically, it has been found that reactive species and antioxidants influence the immune system and that oxidative stress disrupts cell signaling, damages the metabolism of arachidonic acid, and enhances airway and systemic inflammation (Moreno-Macias and Romieu 2014). ROS enhance the inflammatory response when active phagocytic cells induce cellular oxidative phosphorylation and then exceed the capacity of the antioxidant defense system.
The beneficial role of plant species and their constituents is not a new advance, as the benefits of medicinal plants have been known for a very long time. Essential oils from different plants possess many biological properties, such as antioxidant and anti-inflammatory properties. Traditionally, sweet basil is a species used for commercial seasoning and is widely used in cooking, such as in meat and soups (Özcan and Chalchat 2002; Machale et al. 1997). Additionally, sweet basil is well known in many countries as an herb with abundant medicinal value. Especially, the essential oil is useful for first aid in snake bites, colds and for the treatment of rhinitis.
It was previously reported in some studies that molecular distillation (MD) demonstrated an excellent performance in separating or purifying natural products, such as fatty acids (Solaesa et al. 2016) and bio-oil (Wang et al. 2015). For thermo sensitive essential oils, MD is generally regarded as an effective and mild method for separating and purifying heat-sensitive compounds (Isbell and Cermak 2004). MD is characterized by low pressure and temperature in the evaporating space, a short residence time of the liquid and a small distance between the evaporator and condenser, which allows essential oil to retain its original properties. Furthermore, several technologies based on the enrichment properties of essential oil compounds have been applied to upgrade different essential oil fractions obtained from molecular distillation, and then, the fractions are utilized at a high efficiency (Wang et al. 2013, 2014). The available information suggests that MD has been used for essential oil separation previously, but the potential activities of the obtained fractions have been less well studied (Martins et al. 2012; Borgarello et al. 2015).
The objective of this study was to explore the antioxidant and anti-inflammatory activity of both fractions separated from sweet basil essential oil by molecular distillation. Furthermore, the chemical composition of both fractions was evaluated to better understand the role of their composition with respect to their activities.
Materials and methods
Chemicals and reagents
Sweet basil crude oil (Co) was provided by Fengsheng Herb Garden (Haikou, Hainan, China), which was dried with anhydrous sodium sulfate (Na2SO4), filtered and stored in sealed vials at 4 °C before use. Potassium persulfate (K2S2O4), 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), dimethylsulfoxide (DMSO), methyl eugenol (Me) and estragole (Es) were purchased from Macklin Chemical Company (Shanghai, China). Lipopolysaccharide (LPS, from Escherichia coli, 055:B5) and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were obtained from Realtime Co. (Beijing, China). Phosphoric acid, penicillin–streptomycin and Dulbecco’s modified Eagle’s medium (DMEM) were supplied by Boster Biological Technology Company (Wuhan, China). The primers for β-actin, IL-1β, IL-6 and TNF-α were bought from SinoGenoMax Company (Beijing, China). SYBR Select Master Mix was offered by Invitrogen Life Technologies Company (USA). The total RNA isolation kit was bought from Foregene Biotechnology (Chengdu, China).
Fractions obtained by molecular distillation
The distillate (Df) and residue fractions (Rf) of crude oil were obtained by molecular distillation of crude oil using a KDL-5 molecular distillation apparatus (UIC Company, Germany) (Fig. S1). The evaporation temperature and operation pressure were 4 °C and 80 Pa, respectively. The feeding rate was 2 mL/min, and the rotational speed of the roller wiper was 200 rpm.
Chemical composition of sweet basil crude oil and both fractions
The chemical composition of the crude oil, distillate and residue fractions was determined on an Agilent GC–MS-HP6890/5973 system. A DB-5 MS silicon column (30 m × 0.25 mm × 0.25 μm) (J&W co. USA) was used for the analysis. The operating conditions of the column were as follows: oven temperature program from 80 to 150 °C (5 °C/min), withhold time of 10 min, and then from 150 to 220 °C at 10 °C/min (maintained for 9 min). The sample was injected (volume = 0.6 μL) in split mode (1:100), and helium was used as the carrier gas at a flow rate of 1 mL/min. The injector temperature was maintained at 220 °C. The mass spectrometer was used in TIC mode, and the samples were scanned from 30 to 550 amu, with an ion source temperature of 230 °C. The compounds methyl eugenol and estragole were identified by comparison with standard mass spectra obtained under the same conditions and compared with the same mass spectra. Identification of the remaining compounds was performed by comparing their mass spectra with data from NIST05 (National Institute of Standards and Technology, US) and WILEY 275 libraries based on their retention times. Quantification of each peak was achieved using the mass reported by the mass detector, and the results are expressed as a percentage (w/w).
Antioxidant activity determination
Free radical scavenging activity: DPPH test
The free radical scavenging activity of samples was determined using the DPPH method with some alteration (Zheng et al. 2015). Two milliliters of various dilutions of samples (0.2, 0.4, 0.6, 0.8, 1.2, 1.6, 2.0 mg/mL) was mixed with 2 mL of 0.04% DPPH methanol solution. After an incubation period of 30 min at 25 °C in the dark, the absorbance at 517 nm was measured and recorded as Asample. A blank experiment was also performed by applying the same procedure to a solution without the test material, and the absorbance was recorded as Ablank. Inhibition of the DPPH free radicals was calculated as percent inhibition (I %) according to the following formula:
The antioxidant activity of samples is expressed as the IC50 and defined as the concentration of the test material required to cause a 50% decrease in the initial DPPH concentration. All measurements were performed in triplicate.
ABTS assay
An ABTS assay was used to determine the free radical scavenging capacity using ABTS·+ cations (Yu et al. 2015). The ABTS·+ cations were generated by mixing 7 mM ABTS stock solution (in methanol) with 2.45 mM K2S2O4 in a volume ratio of 1:1. Then, the mixture was stored in the dark at room temperature overnight prior to use. The mixture was diluted with water to give an absorbance of 0.7 ± 0.02 at 734 nm. Five-hundred microliters of various dilutions (0.2, 0.4, 0.6, 0.8, 1.2, 1.6, 2.0 mg/mL) of samples (in methanol) was reacted with 2 mL of fresh ABTS solution. After incubating the reaction mixture for 6 min at room temperature, the absorbance was measured at 734 nm, and the percent inhibition was the same formula used in the DPPH assay.
Raw264.7 cell culture and treatments
Murine Raw264.7 macrophages were obtained from Boster Biotechnology Company (Wuhan, China). The cells were cultured in DMEM supplemented with penicillin (100 U/mL), streptomycin (100 μg/mL) and 10% fetal bovine serum (FBS) (Gibco BRL Life Technologies, USA) in an incubator with 5% CO2 at 37 °C. For experimental purposes, the cells were harvested in log phase and then seeded in a medium-sized sterile plate. The supernatants and cells were collected for enzyme-linked immune sorbent assays (ELISAs) and reverse transcription-polymerase chain reaction (RT-PCR).
Cell viability test
Raw264.7 cells in log phase (2 × 105 cells/well) were cultivated in 96-well plates, incubated for 12 h and then treated with fresh medium containing different samples. Control cells were incubated with 0.1% DMSO. After 24 h, 10 μL of MTT (5.0 μg/mL) was added to each well, and the plates were incubated for another 4 h. The medium was then discarded, and the cells were dissolved in 100 μL of DMSO and shaken for 10 min. The optical density at 490 nm was measured with a microplate reader (Bio-Tek, EL-808, USA). The assays were performed in triplicate, and the mean ± SD was calculated.
Measurement of nitrite oxide (NO) production
The production of NO was determined by assaying for nitrite (NO2 −) concentrations in the culture supernatant. Briefly, the obtained culture supernatant was mixed with the same volume of Griess reagent (0.1% N-(1-naphthyl) ethylenediamine dihydrochloride in water and 1% sulfanilamide in 5% H3PO4) (Promega, USA) in a 96-well plate and then incubated at room temperature for 10 min. Indomethacin (In) was the positive control in this assay, and the absorbance of the mixture at 540 nm was measured in a microplate reader (Bio-Tek, Eon, USA). The level of NO was calculated with a sodium nitrite standard curve.
iNOS assay
Cells were treated with LPS l μg/mL for 12 h and then washed twice with PBS and incubated in different experimental samples. After another 12 h, the cells were disrupted with RIPA cell lysate solution, and then centrifuged at 12,000 rpm/min for 30 min. The level of iNOS was measured using a photocolorimetrickit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer’s instructions. Indomethacin was the positive control in the iNOS production assay.
Cytokine assay
The concentrations of IL-1β, IL-6 and TNF-α in the cell culture supernatant were determined using IL-1β, IL-6 and TNF-α ELISA kits (Boster, Wuhan, China) according to the manufacturer’s instructions. The color generated was measured at 450 nm using a microplate reader (Bio-Tek, EL-808, USA). Meanwhile, indomethacin was the positive control in the cytokine production assay. All the analytes were assayed in duplicate, and equivocal results were repeated.
RNA isolation and RT-PCR analysis
Total RNA was extracted with a kit according to the manufacturer’s protocol. The RNA pellet was dissolved in nuclease-free water, and the RNA was quantified using a GeneQuant Pro UV spectrophotometer (Eppendorf, BioPhotometer plus). The integrity of the RNA was confirmed with agarose gel electrophoresis. Total RNA (2.5 μg) was converted to cDNA using a commercially available first strand cDNA synthesis kit (Thermo Scientific). The tubes were incubated at 30 °C for 30 min and then at 42 °C for 30 min followed by heating at 95 °C for 2 min. The samples were then stored at −20 °C until further use. The expression of the selected genes was quantified using quantitative real-time PCR performed in a One Step SYBR Select Master Mix Kit as previously described (Kim et al. 2009). The gene-specific primer sequences for β-actin, IL-1β, IL-6, and TNF-α are shown in Table 1. The RT-reaction and semiquantitative RT-PCR were performed in a thermal cycler (Stratagene Mx3005P Agilent Technologies, USA); each of the thermal cycle conditions included denaturation at 95 °C for 30 s. Annealing was performed at 55 °C for 30 s and elongation at 72 °C for 30 s. The relative expression of IL-1β, IL-6, and TNF-α mRNA levels was normalized to β-actin, a stable housekeeping gene.
Table 1.
Primers used for real-time PCR analysis
| Genes | Sense sequence | Anti-sense sequence |
|---|---|---|
| β-actin | 5′-TGG ATTCCT GTG GCA TTC ATG AAA C-3′ | 5-TAA AAC GCA GCT CAG TTA CAG TCC G-3′ |
| IL-1β | 5′-TGA AGG GCT GCT TCC AAA CCT TTG ACC-3′ | 5′-TGT CCA TTG AGG TGG AGA GCT TTC AGC-3′ |
| IL-6 | 5′-TGG AGT CAC AGA AGG AGT GGC TAA G-3′ | 5′-TCT GAC CAC AGT GAG GAA TGT CCA C-3′ |
| TNF-α | 5′-GCG ACG TGG AAC TGG CAG AAG-3′ | 5′-TCC ATG CCG TTG GCC AGG AGG-3′ |
Statistical analysis
The results of all the experiments are expressed as the mean ± SD of three replicates. Statistical analysis was performed with ANOVA followed by a t test using SPSS 13.0 software. Statistical significance was set at *p < 0.05 and **p < 0.01.
Results
The molecular distilled crude oil and chemical composition
The crude oil (yellow) was divided into two parts: small molecular weight chemicals that have a low boiling point (with a larger free path) reached the cold surface and were obtained in the distillate stream, with golden color. Large molecular weight compounds returned to the heated surface and were enriched in the residue stream, which had a dark-brown color (Fig. S2).
As shown in Table 2, the GC–MS analysis indicated that the main components were linalool (5.51%), estragole (43.92%), methyl eugenol (7.34%) and α-bergamotene (12.31%). The sum was 69.08% of the total composition in the crude oil. The residue fraction contained estragole (17.06%), methyl eugenol (11.35%), α-cadinol (10.01%), hexadecanoic acid (10.84%) and linoleic acid (11.40%) and represented 60.66% of the total composition. The distillate fraction contained estragole (13.00%), methyl eugenol (16.96%), α-cadinol (16.24%), α-bergamotene (11.92%) and carotol (5.75%) and represented 63.87% of the total composition. These data were in agreement with results reported by Arranz et al. (2015) and Rodrigues et al. (2016), who also reported estragole, eugenol, α-bergamotene and linaloolas the main constituents. However, the percentages of these compounds in the total oil were slightly different than those reported here. Estragole (17.06%) was determined to be the main component of the residue fraction based on the high content, while methyl eugenol (16.96%) was found to be the main component of the distillate fraction.
Table 2.
The natural volatile components identified in each fraction of sweet basil oil after molecular distillation
| Peak | R. time | Constituents | Peak area (%) | ||
|---|---|---|---|---|---|
| Df | Rf | Co | |||
| 1 | 3.65 | α-Pinene | – | – | 0.27 |
| 2 | 3.90 | Camphene | – | – | 0.11 |
| 3 | 4.10 | 2,6,6-Trimethyl-2-vinyl-tetrahydropyran | – | – | 0.08 |
| 4 | 4.17 | Sabinene | – | – | 0.08 |
| 5 | 4.33 | β-Myrcene | – | – | 1.35 |
| 6 | 5.03 | 1-Methyl-2-(1-methylethyl)-benzene | – | – | 0.23 |
| 7 | 5.09 | Limonene | – | 0.08 | 0.79 |
| 8 | 5.17 | Eucalyptol | 0.65 | 0.30 | 3.32 |
| 9 | 5.30 | Trans-β-ocimene | 0.36 | 0.15 | 1.79 |
| 10 | 5.60 | γ-Terpinene | – | – | 0.11 |
| 11 | 6.15 | α-Terpinolene | – | – | 0.42 |
| 12 | 6.46 | Linalool | 0.97 | 1.49 | 5.51 |
| 13 | 7.60 | Camphor | 0.69 | 0.11 | 0.59 |
| 14 | 8.41 | 4-Terpineol | – | – | 0.19 |
| 15 | 8.89 | Estragole | 13.00 | 17.06 | 43.92 |
| 16 | 10.76 | (–)-Bornyl acetate | 0.43 | 0.50 | 1.01 |
| 17 | 12.64 | Eugenol | 0.89 | 0.86 | 0.44 |
| 18 | 13.21 | γ-Elemene | 0.70 | – | 0.16 |
| 19 | 13.30 | β-Bourbonene | 0.83 | – | 0.17 |
| 20 | 13.43 | β-Elemene | 1.71 | 1.30 | 2.25 |
| 21 | 13.86 | Methyl eugenol | 16.96 | 11.35 | 7.34 |
| 22 | 14.61 | α-Bergamotene | 11.92 | 8.74 | 12.31 |
| 23 | 15.98 | Germacrene | 1.98 | 0.99 | 1.45 |
| 24 | 17.31 | Calamenene | 1.84 | 1.02 | 0.78 |
| 25 | 18.05 | α-Calacorene | 0.96 | 0.59 | – |
| 26 | 18.88 | Nerolidol | 0.72 | 0.43 | – |
| 27 | 19.32 | Veridiflorol | 0.57 | 0.27 | – |
| 28 | 19.84 | Spathulenol | 2.75 | 3.75 | 0.48 |
| 29 | 20.58 | Epiglobulol | 0.87 | 0.47 | – |
| 30 | 21.54 | Carotol | 5.75 | 1.71 | 0.45 |
| 31 | 23.37 | α-Cadinol | 16.24 | 10.01 | 2.63 |
| 32 | 24.08 | t-Muurolol | 2.47 | 2.23 | – |
| 33 | 30.15 | Caryophyllene oxide | 1.35 | 1.98 | – |
| 34 | 31.32 | Isophytol | 0.89 | 0.35 | – |
| 35 | 32.12 | Hexadecanoic acid | 0.64 | 10.84 | 0.76 |
| 36 | 34.25 | Phytol | – | 0.78 | – |
| 37 | 35.49 | Linoleic acid | – | 11.40 | 0.77 |
| 38 | 35.85 | Octadecanoic acid | – | 0.99 | – |
| Total | 86.14 | 89.32 | 89.76 | ||
Df distillate fraction, Rf residue fraction, Co sweet basil crude oil
Antioxidant activity
The model of DPPH and ABTS radical scavenging is a widely used method to evaluate the free radical scavenging ability of natural substances. The antioxidant activity of a compound can be presented as its ability to scavenge the radicals by measuring the decline in the absorbance (Zhou et al. 2016). As seen in Table 3, the results indicated that the scavenging activity was in the order Rf > Me > Df > Co > Es. The ABTS radicals were generated by potassium persulfate and have a blue-green chromophore with a maximum absorption at 734 nm, which decreases in intensity in the presence of antioxidants. The results also demonstrated that the scavenging activity was in the order Rf > Df > Me > Co > Es. Based on these results, the residue fraction showed the highest ability to scavenge DPPH and ABTS radicals, an indicator of its potency as a radical scavenger; However, estragole showed less antioxidant activity. This result was consistent with that of Lee’s study (Lee et al. 2005).
Table 3.
Various radical scavenging activities (DPPH, ABTS) of the sweet basil oil fractions obtained from molecular distillation
| IC50 value | Co (mg/mL) | Df (mg/mL) | Rf (mg/mL) | Me (mg/mL) | Es (mg/mL) |
|---|---|---|---|---|---|
| Scavenging ability on DPPH radicals | 2.008 ± 0.128 | 1.729 ± 0.056 | 1.092 ± 0.066 | 1.287 ± 0.037 | – |
| Scavenging ability on ABTS radicals | 2.136 ± 0.030 | 1.390 ± 0.058 | 0.707 ± 0.042 | 1.675 ± 0.028 | – |
Values are given as the mean ± SD (n = 3)
Df distillate fraction, Rf residue fraction, Co sweet basil crude oil, Me methyl eugenol, Es estragole
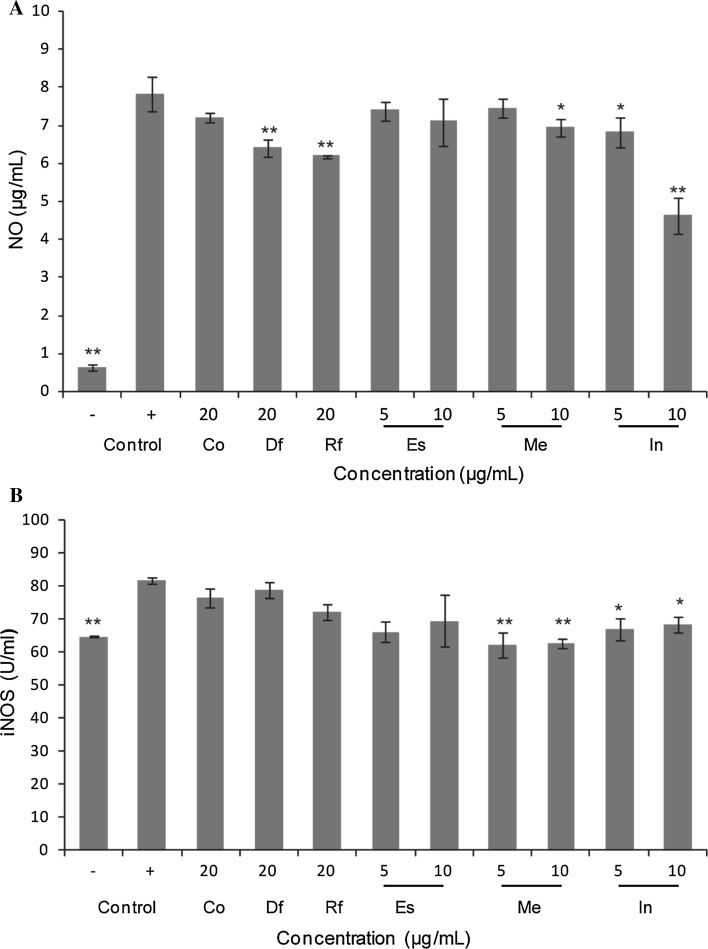
Effect on LPS-induced production of NO and iNOS
The MTT assay results indicated that low concentrations (2.5–50 μg/mL) of the samples had a small effect on the viability of Raw264.7 cells (Fig. 1). Therefore, non-cytotoxic concentrations of the crude oil (20 μg/mL) and the main pure compounds (10 μg/mL) (Arranz et al. 2015) were used for subsequent studies. Modulation of iNOS-mediated NO release is a major contributing factor during the inflammation process (Laskin and Pendino 1995), and the levels of iNOS were determined using a spectrophotometric method. As shown in Fig. 2, after stimulation with LPS, the production of NO and the expression of iNOS in Raw264.7 cells were markedly increased when compared to normal cells. Pretreatment with the Df and Rf and Meat the indicated amounts inhibited the NO production in cells (Fig. 2a) while affecting iNOS activity slightly (Fig. 2b), but estragole had no effect on NO or iNOS (Fig. 2a, b).
Fig. 1.
Effect of different samples on the viability of Raw264.7 cells. Cells were cultured with samples for 12 h, followed by LPS treatment (0.1 μg/mL) for 12 h. Cell viability was measured with MTT assays. The results are reported as the mean ± SD of three independent experiments. Df distillate fraction, Rf residue fraction, Co sweet basil crude oil, Me methyl eugenol, Es estragole
Fig. 2.
Levels of a NO and b iNOS production stimulated by LPS in Raw264.7 cells in the presence of test samples. The values are expressed as the mean ± SD of three individual experiments. *p < 0.05; **p < 0.01 indicate significant differences compared with the LPS-treated group. Df distillate fraction, Rf residue fraction, Co sweet basil crude oil, Me methyl eugenol, Es estragole
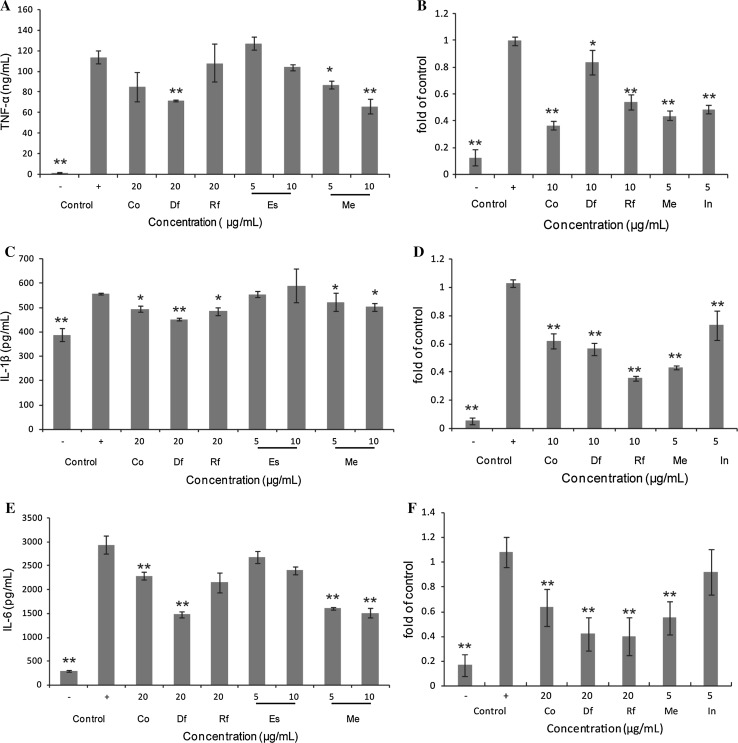
Effects on pro-inflammatory cytokines
The effect of test samples on the production of LPS-induced inflammatory cytokines was investigated in Raw264.7 cells. The Raw264.7 cells were treated with LPS in the presence or absence of samples, and the levels of TNF-α, IL-1β and IL-6 were measured via ELISA and RT-PCR. The results showed that estragole was less effective on these cytokines, which was not in agreement with Rodrigues’s report (Rodrigues et al. 2016). The difference may be caused by the different concentration. However, the Co, Df, and Rf and Me suppressed the production of TNF-α, IL-1β and IL-6 (Fig. 3a, c, e). To confirm whether the decrease in cytokine production was correlated with the cytokine transcriptional levels, the expression of these genes was analyzed with RT-PCR. The results also showed that the Co, Df, and Rf and Me down-regulated the LPS-induced transcription of TNF-α, IL-1β and IL-6 mRNA (Fig. 3b, d, f), which was in accordance with their effects on the cytokines and consistent with Arranz’s results (Arranz et al. 2015). Basically, the anti-inflammatory effect of the distillate fraction was the highest of the three essential oils (referring to crude oil and both fractions), and the activity utilized the same mechanism as indomethacin (Antonisamy et al. 2016).
Fig. 3.
The effects of analytes on TNF-α, IL-1β and IL-6 at the transcriptional (a, c, e) and translational levels (b, d, f) in LPS-induced Raw264.7 cells in the presence of test samples for 24 h (n = 3).*p < 0.05; **p < 0.01 indicate significant differences compared with the LPS-treated group. Df distillate fraction, Rf residue fraction, Co sweet basil crude oil, Me methyl eugenol, Es estragole
Discussion
Medicinal plants have played important roles in human health for a long time. Sweet basil is a traditional medicine, and its crude oil presents high antioxidant, antihypertensive, and anti-inflammatory activities (Rodrigues et al. 2016; Chalchat and Özcan 2008). Meanwhile, molecular distillation is an efficient method for separating heat-sensitive crude oil according to differences in the boiling point of the constituents, and the sweet basil crude oil could be distilled into two parts. According to the GC–MS results, the residue fraction holds a high concentration of methyl eugenol, linoleic acid and α-cadinol, which are known to be natural antioxidants and show diverse roles in reducing free radicals and alleviating oxidative stress. Generally, natural antioxidants are applied for clinical treatment of atherosclerosis, diabetes and hypertension (Pisoschi and Pop 2015) and are easily accepted by consumers due to fewer side effects (Valenzuela et al. 2003),while synthetic antioxidants often have side effects that render them unsuitable for long-term use. According to a previous work, sweet basil crude oil can be used as a natural antioxidant in food products, which are sensitive to lipid oxidation, even organic food (Arranz et al. 2015). However, in this work, it was further confirmed that the residue fraction possessed higher antioxidant activity, which could be used as a potential antioxidant agent.
Similarly, to investigate the anti-inflammatory activity of both fractions in LPS-stimulated Raw264.7 macrophages, several important inflammation mediators were examined. Inflammatory stimulation can induce macrophages to express iNOS, and then iNOS can produce high concentrations of NO after stimulation with bacterial endotoxins, or a variety of pro-inflammatory cytokines (such as TNF-α, IL-1β and IL-6). Generally, suppressing the production of TNF-α and IL-1β has been widely applied to screen anti-inflammatory agents (Yoon et al. 2010; Dinarello 2010), while blocking IL-6 signaling is considered to be therapeutic in some diseases (Zhou et al. 2009), and the agents that can reduce production of IL-6 are thought to be anti-inflammatory. Taking all of the results into account, both fractions (10 μg/mL) were found to adjust the NO and iNOS production induced by LPS in Raw264.7 cells, which can significantly reduce the release of TNF-α, IL-1β and IL-6 by down-regulating their gene expression. In addition, the decrement of IL-1β, IL-6 and TNF-α was correlated with their mRNA reduction. Thus, modulation of the production of these cytokines was the major mechanism in the control of inflammation for the Rf and Df. Furthermore, 5 μg/mL of Me showed high suppression of the production of TNF-α, IL-1β and IL-6 and their gene expression. Therefore, Me might act as the main mediator of the anti-inflammatory activity of both fractions. The Df exhibited a better ability to diminish the immune-mediated inflammatory cytokines, and this result provides further evidence that the distillate fractions may contain potent anti-inflammatory agents.
Conclusion
In this study, the results indicated that the residue fraction of sweet basil had a higher antioxidant activity, while the distillate fraction exhibited better anti-inflammatory activity after molecular distillation. Thus, molecular distillation presents an alternative to separate fractions from essential oils with different abilities that may be maximized for natural herbal medicine.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
This work was financially supported by the National Science Foundation of Hainan Province (No. 20168269), and the Natural Science Foundation of China (No. 81573391) and the National Key Projects of China (No. 81227802).
Compliance with ethical standards
Conflict of interest
The authors declare no conflicts of interest.
Footnotes
Electronic supplementary material
The online version of this article (doi:10.1007/s13197-017-2620-x) contains supplementary material, which is available to authorized users.
Hailong Li and Yanhui Ge have contributed equally to this work.
Contributor Information
Junqing Zhang, Phone: +86 029-82655382, Email: jqzhang2011@163.com.
Qiang Fu, Phone: +86 029-82655382, Email: fuqiang@mail.xjtu.edu.cn.
References
- Antonisamy P, Arasu MV, Dhanasekaran M, Choi KC, Aravinthan A, Kim NS, Kang CW, Kim JH. Protective effects of trigonelline against indomethacin-induced gastric ulcer in rats and potential underlying mechanisms. Food Funct. 2016;7:398–408. doi: 10.1039/C5FO00403A. [DOI] [PubMed] [Google Scholar]
- Arranz E, Jaime L, Hazas MC, Reglero G, Santoyo S. Supercritical fluid extraction as an alternative process to obtain essential oils with anti-inflammatory properties from marjoram and sweet basil. Ind Crops Prod. 2015;67:121–129. doi: 10.1016/j.indcrop.2015.01.012. [DOI] [Google Scholar]
- Borgarello AV, Mezza GN, Pramparo MC, Gayol MF. Thymol enrichment from oregano essential oil by molecular distillation. Sep Purif Technol. 2015;153:60–66. doi: 10.1016/j.seppur.2015.08.035. [DOI] [Google Scholar]
- Chalchat J-C, Özcan MM. Comparative essential oil composition of flowers, leavesand stems of basil (Ocimum basilicum L.) used as herb. Food Chem. 2008;110:501–503. doi: 10.1016/j.foodchem.2008.02.018. [DOI] [PubMed] [Google Scholar]
- Dinarello CA. Anti-inflammatory agents: present and future. Cell. 2010;140:935–950. doi: 10.1016/j.cell.2010.02.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isbell TA, Cermak SC. Purification of meadowfoam monoestolide from polyestolide. Ind Crops Prod. 2004;19:113–118. doi: 10.1016/j.indcrop.2003.07.006. [DOI] [Google Scholar]
- Kim EH, Shim B, Kang S, Jeong G, Lee J-s YuY-B, Chun M. Anti-inflammatory effects of Scutellaria baicalensis exbitract via suppression of immune modulators and MAP kinase signaling molecules. J Ethnopharmacol. 2009;126:320–331. doi: 10.1016/j.jep.2009.08.027. [DOI] [PubMed] [Google Scholar]
- Laskin DL, Pendino KJ. Macrophages and inflammatory mediators in tissue injury. Annu Rev Pharmacol. 1995;35:655–677. doi: 10.1146/annurev.pa.35.040195.003255. [DOI] [PubMed] [Google Scholar]
- Lee SJ, Umano K, Shibamoto T, Lee KG. Identification of volatile components in basil (Ocimum basilicum L.) and thyme leaves (Thymus vulgaris L.) and their antioxidant properties. Food Chem. 2005;91:131–137. doi: 10.1016/j.foodchem.2004.05.056. [DOI] [Google Scholar]
- López-Alarcón C, Denicola A. Evaluating the antioxidant capacity of natural products: a review on chemical and cellular-based assays. Anal Chim Acta. 2013;763:1–10. doi: 10.1016/j.aca.2012.11.051. [DOI] [PubMed] [Google Scholar]
- Machale KW, Niranjan K, Pangarkar VG. Recovery of dissolved essential oils from condensate waters of basil and Mentha arvensis distillation. J Chem Technol Biotechnol. 1997;69:362–366. [Google Scholar]
- Martins PF, Carmona C, Martinez EL, Sbaite P, Maciel Filho R, Wolf Maciel MR. Short path evaporation for methyl chavicol enrichment from basil essential oil. Sep Purif Technol. 2012;87:71–78. doi: 10.1016/j.seppur.2011.11.024. [DOI] [Google Scholar]
- Maulik N, McFadden D, Otani H, Thirunavukkarasu M, Parinandi NL. Antioxidants in longevity and medicine. Oxid Med Cell Longev. 2013 doi: 10.1155/2013/820679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreno-Macias H, Romieu I. Effects of antioxidant supplements and nutrients on patients with asthma and allergies. J Allergy Clin Immun. 2014;133:1237–1244. doi: 10.1016/j.jaci.2014.03.020. [DOI] [PubMed] [Google Scholar]
- Özcan M, Chalchat JC. Essential oil composition of Ocimum basilicum L. and Ocimum minimum L. in Turkey Czech. J Food Sci. 2002;20:223–228. [Google Scholar]
- Persson T, Popescu BO, Cedazo-Minguez A. Oxidative stress in Alzheimer’s disease: why did antioxidant therapy fail? Oxid Med Cell Longev. 2014;2014:1–11. doi: 10.1155/2014/427318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pisoschi AM, Pop A. The role of antioxidants in the chemistry of oxidative stress: a review. Eur J Med Chem. 2015;97:55–74. doi: 10.1016/j.ejmech.2015.04.040. [DOI] [PubMed] [Google Scholar]
- Rodrigues LB, Martins AOBPB, Cesário FRAS, Castro FF, Albuquerque TR, Fernandes MNM, Barbosa R. Anti-inflammatory and antiedematogenic activity of the Ocimum basilicum essential oil and its main compound estragole: in vivo mouse models. Chem Biol Interact. 2016;257:14–25. doi: 10.1016/j.cbi.2016.07.026. [DOI] [PubMed] [Google Scholar]
- Solaesa ÁG, Sanz MT, Falkeborg M, Beltrán S, Guo Z. Production and concentration of monoacylglycerols rich in omega-3 polyunsaturated fatty acids by enzymatic glycerolysis and molecular distillation. Food Chem. 2016;190:960–967. doi: 10.1016/j.foodchem.2015.06.061. [DOI] [PubMed] [Google Scholar]
- Toda S. Polyphenol content and antioxidant effects in herb teas. Chin Med. 2011;02:29–31. doi: 10.4236/cm.2011.21005. [DOI] [Google Scholar]
- Valenzuela BA, Sanhueza J, Nieto S. Natural antioxidants in functional foods: from food safety to health benefits. Grasas Aceites. 2003;54:295–303. doi: 10.3989/gya.2003.v54.i3.245. [DOI] [Google Scholar]
- Wang S, Li X, Zhang F, Cai Q, Wang Y, Luo Z. Bio-oil catalytic reforming without steam addition: application to hydrogen production and studies on its mechanism. Int J Hydrog Energy. 2013;38:16038–16047. doi: 10.1016/j.ijhydene.2013.10.032. [DOI] [Google Scholar]
- Wang S, Cai Q, Wang X, Zhang L, Wang Y, Luo Z. Biogasoline production from the co-cracking of the distilled fraction of bio-oil and ethanol. Energy Fuel. 2014;28:115–122. doi: 10.1021/ef4012615. [DOI] [Google Scholar]
- Wang Y, Wang S, Leng F, Chen J, Zhu L, Luo Z. Separation and characterization of pyrolytic lignins from the residue fraction of bio-oil by molecular distillation. Sep Purif Technol. 2015;152:123–132. doi: 10.1016/j.seppur.2015.08.011. [DOI] [Google Scholar]
- Yoon WJ, Ham YM, Lee WJ, Lee NH, Hyun CG. Brown alga Sargassum muticum inhibits proinflammatory cytokines, iNOS, and COX-2 expression in macrophage RAW 264.7 cells. Turk J Biol. 2010;34:25–34. [Google Scholar]
- Yu XM, Zhu P, Zhong QP, Li MY, Ma HR. Subcritical water extraction of antioxidant phenolic compounds from XiLan olive fruit dreg. J Food Sci Technol. 2015;52:5012–5020. doi: 10.1007/s13197-014-1551-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng L, Liu M, Zhai GY, Ma Z, Wang LQ, Jia L. Antioxidant and anti-ageing activities of mycelia zinc polysaccharide from Pholiota nameko SW-03. J Sci Food Agric. 2015;95:3117–3126. doi: 10.1002/jsfa.7048. [DOI] [PubMed] [Google Scholar]
- Zhou H, Lutterodt H, Cheng Z, Yu L. Anti-inflammatory and antiproliferative activities of trifolirhizin, a Flavonoid from Sophora flavescens roots. J Agric Food Chem. 2009;57:4580–4585. doi: 10.1021/jf900340b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou D, Wang D, Yang L, Liu Z, Zhang Y. A Modified and improved assay based on microbial test system (MTS) to evaluate antioxidant activity. Food Anal Methods. 2016;9:895–904. doi: 10.1007/s12161-015-0266-8. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.