Abstract
Oxaliplatin, in combination with 5-fluorouracil plus folinate (or capecitabine), has increased survival substantially in stage III colorectal cancer and prolonged life in stage IV patients, but its use is compromised because of severe toxicity. Chemotherapy-induced peripheral neuropathy (CIPN) is the most problematic dose-limiting toxicity of oxaliplatin. Oncologists included for years calcium and magnesium infusion as part of clinical practice for preventing CIPN. Results from a phase III prospective study published in 2014, however, overturned this practice. No other treatments have been clinically proven to prevent this toxicity. There is a body of evidence that CIPN is caused by cellular oxidative stress. Clinical and preclinical data suggest that the manganese chelate and superoxide dismutase mimetic mangafodipir (MnDPDP) is an efficacious inhibitor of CIPN and other conditions caused by cellular oxidative stress, without interfering negatively with the tumoricidal activity of chemotherapy. MnPLED, the metabolite of MnDPDP, attacks cellular oxidative stress at several critical levels. Firstly, MnPLED catalyzes dismutation of superoxide (O2•−), and secondly, having a tremendous high affinity for iron (and copper), PLED binds and disarms redox active iron/copper, which is involved in several detrimental oxidative steps. A case report from 2009 and a recent feasibility study suggest that MnDPDP may prevent or even cure oxaliplatin-induced CIPN. Preliminary results from a phase II study (PLIANT) suggest efficacy also of calmangafodipir, but these results are according to available data obscured by a surprisingly low number of adverse events and a seemingly lower than expected efficacy of FOLFOX.
Background
Colorectal cancer (CRC) is the second or third most common cancer in Europe and the United States. About half of the CRC patients in Europe will ultimately die of the disease, corresponding annually to about 200,000 people [1], whereas about one third of the patients in the United States will die of the disease, corresponding to about 50,000 [2].
In about one third of the diagnosed CRCs, the disease is locally advanced to one or more lymph nodes (stage III). Postoperative adjuvant chemotherapy in stage III colon cancer patients, for many years with 5-fluorouracil (5-FU) plus levamisole and later folinate, increased 5-year survival from about 47% to 65% [3], [4], [5]. When 5-FU (+ folinate) was combined with oxaliplatin [FOLinate, 5-FU, and OXliplatin (FOLFOX)], 5-year survival alternatively 3-year disease-free survival was further increased in this group to more than 70% [6], [7]. The objective response rate (ORR) and the progression-free survival in palliative treatment of stage IV patients with the combination have increased from about 27% to 45% and from about 7 to 9 months, respectively, compared to 5-FU alone [2], [8], [9], [10]. Sørbye and coworkers [11] reported an ORR of 46% in metastatic CRC patients treated with Nordic FLOX, an oxaliplatin-containing regimen similar to FOLFOX. Addition of oxaliplatin is, however, compromised by vigorous increase in dose-limiting toxicity.
Oxaliplatin-associated chemotherapy-induced peripheral neuropathy (CIPN) is the most frequent cause of complete discontinuation of an otherwise successful therapy [12]. About 12% to 18% of the patients experience grade 3 CIPN on the The National Cancer Institute's Common Terminology Criteria for Adverse Events (NCI-CTCAE) grade 1 to 3 scale [6], [8], where persistent grade ≥ 2 neuropathy usually results in complete discontinuation of oxaliplatin therapy. Furthermore, almost every CRC patient treated with FOLFOX experiences some grade of neutropenia, and 40% to 50% of the patients experience grade 3/4 neutropenia (according to the NCI CTCAE 1-4 scale) [6], [8], [9], [10], [13], [14], in addition to a multitude of other adverse events (AEs). Sørbye and coworkers [11] reported 58% grade 3/4 neutropenia in their study using Nordic FLOX.
Oxaliplatin-associated CIPN differs from cisplatin-associated CIPN in one particular sense. Both cause a peripheral stocking-glove neuropathy that worsens with the cumulative dose, but oxaliplatin is also associated with an acute neuropathic problem that generally occurs after each oxaliplatin dose and often resolves within a few days [15]. Although the acute form can be quite bothersome, it is the chronic CIPN that is the general dose-limiting problem and the main cause of complete discontinuation of oxaliplatin treatment. It has been suggested that oxaliplatin may directly alter axonal voltage-gated sodium channels, inducing an acute neurotoxicity manifested by peripheral nerve hyperexcitability [16].
The chronic neuropathy is characterized by bilaterally symmetrical sensory symptoms (e.g., numbness, tingling, and pain) appearing in the feet, or in both the feet and hands, and occurs with chemotherapeutics across drug classes with distinctly different antitumor mechanisms, such as taxanes (e.g., paclitaxel) and platinum compounds (e.g., oxaliplatin). Despite potential diverse mechanisms underlying the development of CIPN, common degenerative pathways may be triggered when the normal processes and energy delivery mechanisms of the peripheral nervous system become disrupted [16]. Experimental studies in rats suggest that the common underlying mechanism in the development of these neuropathies is oxidative stress [17], [18] and subsequent mitochondrial toxicity in primary nerve sensory axons arising from reduced mitochondrial bioenergetics, e.g., adenosine triphosphate production deficits due to compromised respiratory complex I and II activity [19], [20], [21], [22]. Contrary to the central nerve system, the peripheral nerve system lacks a blood-brain barrier, a draining lymph system, and cerebrospinal fluid. This makes potentially dangerous substances substance, such as chemotherapy drugs, to accumulate in the peripheral nerve system and cause oxidative stress [22], as schematically illustrated in Figure 1. In fact, oxidative stress is identified to be responsible for the neuronal damage in different models of neuropathies such as diabetic neuropathy, acrylamide-induced neuropathy, and Charcot-Marie-Tooth neuropathy [22]. Furthermore, gene therapy in rats with mitochondrial superoxide dismutase (SOD) protects the retinal ganglion cells from chronic intraocular pressure elevation–induced injury via attenuating oxidative stress and improving mitochondrial function [23].
Figure 1.
The susceptibility of the peripheral nerve system to chemotherapy drugs is schematically illustrated. The central nerve system lacks a blood-brain barrier, a draining lymph system, and cerebrospinal fluid. This makes potentially dangerous substances, such as chemotherapy drugs, to accumulate in the peripheral nerve system and cause oxidative stress and detrimental nerve injuries (modified from [22]).
Numerous compounds to prevent CIPN have been tested in patients. A recent Cochrane review [24] shows, however, that those tested so far, more or less, lack efficacy in human patients. Intravenous infusion of calcium and magnesium (CaMg), given before and after FOLFOX therapy, represent the most thoroughly studied and, up to 2014, the most commonly used clinical regimen for the prevention of oxaliplatin-associated CIPN [25], [26], [27], [28]. However, a phase III clinical trial (N08CB/Alliance) did not demonstrate any significant effects of CaMg in preventing oxaliplatin-associated CIPN [15]. The SOD mimetic mangafodipir (MnDPDP) has shown protective efficacy against oxaliplatin-associated CIPN in both rats and humans [29].
As both preclinical and clinical studies suggest that CIPN is associated with cellular oxidative stress and a subsequent mitochondrial toxicity, the present review will focus on oxaliplatin-associated CIPN in CRC patients and its possible relationship to cellular oxidative stress and MnDPDP as a selective cytoprotective agent, i.e., an agent that protects normal tissue but not tumor tissue.
Cellular Oxidative Stress
Aerobic organisms exist in a catch-22 situation. Oxygen and transition metals sustain them, but they also poison them, primarily, via reactive oxygen species (ROS) and, secondarily, via reactive nitrogen species (RNS). The mitochondrial route of O2 reduction to H2O is by a series of univalent electron transfers. Hence, ROS intermediates will be encountered on this univalent pathway, and these are, by turn, superoxide (O2•−), hydrogen peroxide (H2O2), and hydroxyl radicals (•OH). It is these intermediates that are primarily responsible for the toxicity of O2. Furthermore, transition metals are essential components for all living organisms, as they participate in key biological processes such as cellular respiration, gene transcription, and many enzymatic reactions. Their ability to oscillate between different redox states is the main feature that allows them to act as cofactors in cellular enzymes. The most abundant transition metals in human cells are, by turn, iron, copper, and manganese. They can be found in various forms: mainly bound to proteins, but also in association with low–molecular weight species, and as “free” ions. Under certain conditions, these transition metals can catalyze generation of secondary ROS and RNS that leads to oxidative damage of proteins, lipids, and DNA [30].
Oxidative stress can be defined as a situation where the production of ROS and RNS outcompete the endogenous cellular defense. Endogenous SODs, in particular, the mitochondrial manganese-containing SOD (MnSOD), attack and disarm O2•−, the first ROS in the route of O2 reduction to H2O. Except for peroxidases, according to our knowledge, cells lack direct defense against •OH and other secondarily produced ROS and RNS, such as •NO2. SODs are hence essential in the defense against detrimental oxidative stress.
ROS are generated during chemotherapy with several structurally dissimilar anticancer drugs [31], [32], [33]. The resulting increase in cellular oxidative stress is proposed to be the main cause of toxicity to normal cells, including CIPN [17], [18], [19], [21], [22]. However, it may also be a contributing factor to death of cancer cells. Consequently, to be clinically useful, a chemotherapy protectant should selectively protect normal tissue from chemotherapy-induced toxicity but not tumor tissue. So far, two antioxidants, amifostine (a noncatalytic antoxidant) and dexrazoxane (an iron chelator), have reached the clinic. However, their uses have been restricted due to suspicions of negative influence on the tumoricidal activity of chemotherapy and/or adverse events [34].
The mechanisms behind platinum-associated CIPN seem to result from accumulation of platinum in dorsal root ganglion cells [35] and a subsequent increase in oxidative stress. The oxidative stress mechanisms involve peroxynitrite (ONOO−)-mediated nitration and irreversible inactivation of the mitochondrial manganese-containing SOD [18].
SODs and Their Relation to Cellular Oxidative Stress
Irwin Fridovich and Joe McCord discovered the first SOD enzyme, containing copper (Cu) and zinc (Zn), in the late part of the 1960s [36], [37]. A few years later, Fridovich together with Richard A. Weisiger discovered the mitochondrial MnSOD [38], [39]. SOD enzymes catalyze dismutation of O2•− to H2O2 and molecular oxygen, and are the most essential parts of the cellular defense against ROS. Mammals have three different types of SODs: an extracellular CuZnSOD, a cytosolic CuZnSOD, and a mitochondrial MnSOD [34]. These metal (M)-containing enzymes dismutate O2•− [36], as follows:
Under normal conditions, endogenous SODs keep ROS in check, and H2O2 is subsequently decomposed into water and oxygen by peroxidases (hydrogen peroxidase and glutathione peroxidase). Nitric oxide (•NO) is an endogenously synthesized free radical with many physiological functions, like vasodilation and neurotransmission. During situations of more extreme oxidative stress, the production of O2•− and •NO increases to levels where O2•− escapes the endogenous SODs. Superoxide then reacts with •NO and gives rise to highly toxic peroxynitrite (ONOO−) [40], [41], which subsequently nitrates tyrosine (Tyr) residues of proteins through a transition metal (Mn+)–driven process [30], [41], [42]:
| ONOO− + Mn+X - > ONOO − Mn+X - > •NO2 + •O − Mn+X - > NO2 + O = M(n+1)+X |
As a result of this reaction, •NO2 and oxometal complexes, e.g., O = Fe4+X and O = Mn4+X, are generated. These two species are able to oxidize and nitrate Tyr residues. So, in the presence of transition metal centers (X; low–molecular weight complexes or metalloproteins) that react with ONOO− by the above-described reaction, NO2Tyr is produced. Peroxynitrite-dependent Tyr nitration is mediated mainly by the abovementioned two oxometal complexes [30], [42], [43]. Interestingly, nitration of Tyr34 in the mitochondrial MnSOD is catalyzed by intraenzymatic manganese, as follows [43]:
| Tyr34 + O = Mn4+SOD - > •Tyr34 + Mn3+SOD + OH− |
| NO2− + •OH - > •NO2 + OH− |
| •Tyr34 + •NO2 - > NO2Tyr34 |
Tyr nitration can also be achieved by ONOO−-independent routes as well, depending on the transition of metal-catalyzed oxidation, like that of nitrite (NO2−) to •NO2 in the presence H2O2. These processes can be achieved either by hemeperoxidase-dependent reactions or by ferrous (Fe2+) and cuprous (Cu+) ions through Fenton-type chemistry. The nitration of Tyr residues represents an oxidative modification that causes disruption of normal •NO signaling and metabolism and severely exacerbates oxidative stress further.
Peroxynitrite-dependent nitration in mitochondrial MnSOD occurs site-specifically at Tyr34 located 5 Å from the active site, a process catalyzed by SOD-coordinated manganese [43]. By nitrating human Tyr34, ONOO− irreversibly inactivates endogenous SOD activity and hence causes a vicious circle that enhances the production of ONOO− further.
Crystallographic data and molecular dynamics simulations indicate that Tyr34 nitration leads to enzyme inactivation via generation of a large energy barrier for the entry of O2•− through the access channel of the enzyme [43]. Cytochrome c is another mitochondrial target for ONOO−, resulting in nitration of Tyr74 that triggers a conformational change in the protein, resulting in an alternative conformation lacking its normal electron transport capacities and increased capacity to react with peroxides and promote oxidation of a second substrate [43].
Nitrated proteins are found in several pathological conditions, including inflammatory process [42], chronic rejection of human renal allografts [44], tumor-associated immunosuppression [45], paracetamol (acetaminophen) intoxication [46], and oxaliplatin (and paclitaxel)-associated CIPN [18]. Interestingly, CIPN is associated with mitochondrial Tyr34 nitration of mitochondrial MnSOD and a subsequent inactivation of enzyme activity, an effect prevented by the SOD mimetic compound [47]/ONOO− decomposition catalyst, MnTE-2-PyP(5+) [18]. Such conditions seem to encompass opening of the mitochondrial membrane permeability transition pore, resulting in a collapse of mitochondrial membrane potential and cessation of adenosine triphosphate synthesis, which in turn trigger apoptotic/necrotic cell death [48].
Oxidative stress promotes metal mobilization from proteins by mechanisms that include O2•−-mediated oxidation of labile iron-sulfur clusters, redox-dependent metal release from storage or transport proteins (transferrin, ferritin, and ceruloplasmin), oxidative modifications of heme proteins that result in heme release and/or degradation (myoglobin and cytochrome c), and histidine oxidation with disruption of metal coordination sites of, e.g., CuZn and MnSODs, among others [42].
Metal Chelation and SOD to Combat Oxidative Stress
Metal chelators with high affinity for iron (and copper) may be helpful during the above-described conditions. Importantly, iron can coordinate six ligands in an octahedral arrangement; thus, iron chelators with the highest affinity will normally be hexadentate, binding iron in a 1:1 ratio (chelator:iron), with one example being deferoxamine [49]. By contrast, iron chelators that do not bind in this ratio may be redox active and extremely cytotoxic by catalyzing free radical generation.
It was early realized that SOD enzymes might be clinically useful for treating conditions of cellular oxidative stress, such as those seen during conditions of ischemia-reperfusion, and doxorubicin toxicity. However, their use is restricted owing to their large molecular weight and poor cellular uptake. A clinical study published as late as 2014 demonstrated no efficacy of human recombinant SOD against doxorubicin toxicity [50]. A more successful approach is the development of low–molecular weight SOD mimetics. Kensler et al. [51] described anticarcinogenic activity of the first generation of a CuSOD mimetic in 1983. They were followed by MnSOD mimetics, particularly of the so-called porphyrin, salen, cyclic polyamine, and MnPLED types [34]. The MnPLED type has among other things demonstrated borderline statistical efficacy in a small feasibility study in patients with acute myocardial infarction going through percutaneous coronary invention [34], a procedure known to cause severe oxidative stress. This type has also shown efficacy against doxorubicin cardiotoxicity in mice (see below).
The SOD Mimetics MnDPDP and Calmangafodipir [Ca4Mn(DPDP)5] as Selective Cytoprotective Agents
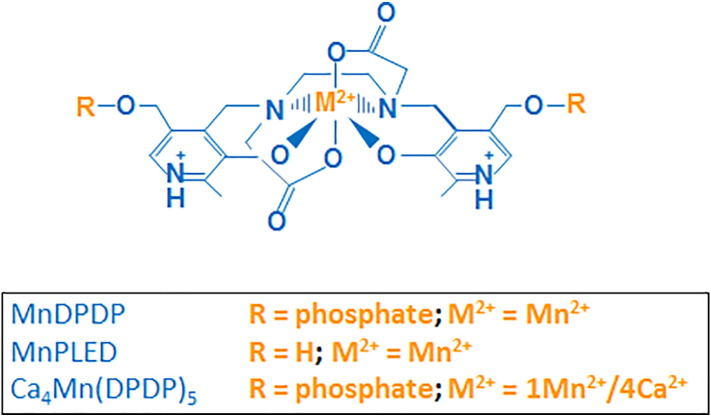
During the development of MnDPDP (Figure 2) as a magnetic resonance imaging (MRI) contrast agent at the beginning of the 1990s, clinical studies showed that rapid intravenous injection into patients caused cardiovascular effects and facial flushing. A mechanistic study was conducted in isolated arteries with and without endothelial cells. This study demonstrated that MnDPDP protected endothelium-derived •NO from reacting with O2•− [52]. The results from this study further suggested that this effect was the result of an SOD mimetic activity of MnDPDP, which was later confirmed by electron spin resonance spectroscopy (Figure 3). After injection, MnDPDP is rapidly dephosphorylated to the much more lipophilic and cell-permeable MnPLED (Figure 2). MnPLED also possesses SOD activity, whereas ZnDPDP and free Mn2+ are deprived of such activity (Figure 3) [53]. Paradoxically, Asplund et al. [52] showed similar vasodilator effects of MnCl2 as those of MnDPDP. This in vitro effect is presumably caused by Mn2+ complexed to phosphoric acid and carbonic acid of the used Krebs' buffer. These metal complexes are known to possess SOD activity. In fact, the prokaryote Lactobacillus plantarum and related lactic acid bacteria lack SOD enzymes. In these organisms, Mn2+ forms complexes with various organic acids, including phosphoric acid, lactic acid, and carbonic acid [34]. However, this effect of MnCl2 is not expected to occur under in vivo conditions in multicellular eukaryotes.
Figure 2.
Chemical structure of MnDPDP (manganese dipyridoxyl diphosphate; generic name mangafodipir), MnPLED (manganese pyridoxyl ethyldiamine), and Ca4Mn(DPDP)5 [tetracalcium monomanganese penta(dipyridoxyl diphosphate); calmangafodipir].
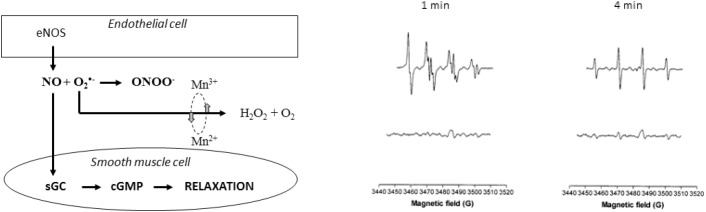
Figure 3.
The proposed vasodilator mechanism for MnDPDP is illustrated at the left [52]. Vasodilator nitric oxide (•NO) is produced by endothelial NO synthase (eNOS) in the innermost (endothelial) cellular layer of the artery, and NO stimulates guanylate cyclase (sGC) in smooth muscle cells to produce increasing amounts of cGMP which cause relaxation and hence vasodilation. Under in vivo conditions, a considerable fraction of •NO reacts with superoxide (O2•−), forming peroxynitrite (ONOO−). MnDPDP preserves vasodilator •NO by dismutating O2•− into H2O2 and O2. Electron spin resonance spectra are shown in the right part of the figure; O2•− was generated in the xanthine oxidase reaction in the presence of the spin trap DMPO. Control reaction (upper panel) revealed that the superoxide adduct DMPO-OOH evolved rapidly (within 1 minute) toward the hydroxyl adduct DMPO-OH (4 minutes). Ten micromolars of MnDPDP (lower panel) markedly reduced the signal intensity, indicating SOD mimetic activity [53].
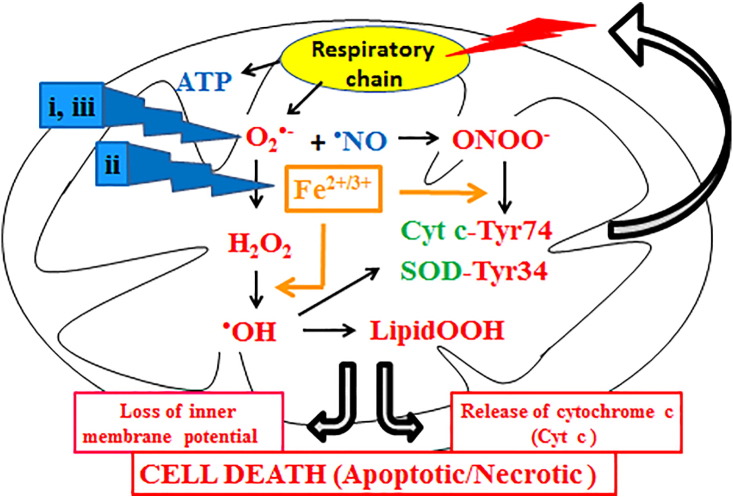
MnDPDP and Ca4Mn(DPDP)5 (Figure 2) have in preclinical models been shown to protect normal cells against chemotherapy-induced toxicity without interfering negatively with the tumoricidal activity of several chemotherapeutic drugs, including oxaliplatin [34]. Findings from a case report [54] and a feasibility study [55] reported to the Swedish Medical Product Agency in 2010 (Table 1) suggested that MnDPDP, through its SOD mimetic activity, protects blood, nervous, and mucous tissues in patients during chemotherapy with FOLFOX. Other important properties of DPDP and its metabolite PLED are their high affinities for iron and copper. The 10log formation constants [10log(KML)] of DPDP and PLED for Fe3+are 33.5 and 36.9, respectively. The corresponding constants of DPDP and PLED for Cu2+ are 22.1 and 21.5, respectively [56], whereas the formation constants of DPDP and PLED for Mn2+ are 15.1 and 12.6. Theoretically, MnDPDP, MnPLED, and DPDP are hence expected to inhibit the iron- or copper-driven Fenton reaction. In fact, these compounds inhibit the iron-driven Fenton reaction in a one to one manner, that is, an equimolar concentration of MnDPDP, MnPLED, or DPDP to that of iron in the reaction mixture fully quenches the Fenton reaction [57]. This finding quite clearly suggests that the resulting iron complexes are not redox active. Hence, MnDPDP and MnPLED, through their combined SOD mimetic activities and iron-binding properties, are expected to be efficacious inhibitors of cellular oxidative stress, including the above-mentioned incorporation of nitrate to the Tyr34 residue of mitochondrial MnSOD and nitration of other proteins. Interestingly, MnDPDP has been shown to inhibit the mitochondrial membrane permeability transition pore and, as indicated in Figure 4, subsequent loss of inner membrane potential and cell death [58].
Table 1.
AEs Occurring in the Placebo Group and the MnDPDP (Mangafodipir) Group According to NCI-CTCAE Version 3 or Sanofi-NCI Criteria (No Effect = 0, Mild = 1, Moderate = 2, Severe = 3, and Life Threatening = 4)
| Placebo (7 Patients; 18 Cycles) |
Mangafodipir (7 Patients; 20 Cycles) |
|||||||
|---|---|---|---|---|---|---|---|---|
| Adverse, Event | Gr 1 | Gr 2 | Gr 3 | Gr 4 | Gr 1 | Gr 2 | Gr 3 | Gr 4 |
| Neutropenia | 3 | 1 | 1 | 1 | 4 | 1** | 0 | 0 |
| Other hematological toxicity | 15 | 1 | 0 | 0 | 13 | 0 | 0 | 0 |
| Neurosensory toxicity | 9 | 0 | 1 | 0 | 9 | 0 | 0 | 0 |
| Oral mucocitis | 2 | 2 | 0 | 0 | 0 | 0 | 0 | 0 |
| Nausea | 7 | 3 | 0 | 0 | 9 | 1** | 0 | 0 |
| Vomiting | 1 | 1 | 1 | 0 | 2 | 0 | 0 | 0 |
| Diarrhea | 0 | 0 | 0 | 0 | 8 | 0 | 0 | 0 |
| Fatigue | 4 | 2 | 0 | 0 | 4 | 0 | 0 | 0 |
| Other AE | 3*, †, § | 1‡ | 1¶ | 0 | 1# | 0 | 0 | 0 |
| Σ AE | 44 | 11 | 4 | 1 | 50 | 2 | 0 | 0 |
Chest wall pain.
Dyspnea.
Pain in lower extremities.
Skin reaction (face).
Ileus.
Nasal mucositis.
Patient received a 10% too high dose during the first FOLFOX6 cycle.
Figure 4.
The proposed cytoprotective mechanisms for the MnDPDP metabolite MnPLED are schematically illustrated. i) It lowers the intracellular level of O2•− directly through its MnSOD mimetic activity; ii) it binds and disarms transition metals, particularly iron and copper, and thus arrests incorporation of •NO2 into Tyr34 and Tyr74 of mitochondrial MnSOD and cytochrome c, respectively, and inhibits production of highly toxic OH• radicals; and iii) it replaces endogenous mitochondrial MnSOD that already has been irreversible inactivated SOD mimetic activity and preserves the NO signaling.
An in vitro mouse model was employed by Towart et al. [59] to test the cardioprotective potential of MnDPDP against doxorubicin toxicity, seen as a decrease in contractility of an electrically paced mouse left atrium. MnDPDP did not protect when added directly into the organ bath. However, after having been intravenously injected 30 minutes before the mouse was killed and the left atrium was dissected out, 1 to 10 μmol/kg MnDPDP displayed cardioprotective efficacy. MnDPDP was shown to be about 100 times more efficacious than clinically available dexrazoxane, i.e., 1 μmol/kg MnDPDP gave similar protection to that of 93 μmol/kg dexrazoxane. Most importantly, the fully dephosphorylated metabolite of MnDPDP, MnPLED, was about 100 times more efficacious than MnDPDP (unpublished data). Furthermore, when added directly into the organ bath, MnPLED but not MnDPDP protected against doxorubicin cardiotoxicity [57]. Indirectly, these results suggested that MnDPDP undergoes in vivo dephosphorylation before it can exert the therapeutic effect, i.e., MnDPDP should be looked upon as a prodrug to MnPLED. Whereas the prodrug with polar phosphate groups is readily soluble in water, its dephosphorylated metabolite is nonpolar and lipid soluble. MnPLED, in contrast to MnDPDP, is therefore able to enter target cells and act at the intracellular site, like in mitochondria.
In vivo and in vitro experiments, utilizing human breast cancer cells (MX-1) and ovarian cancer cells (A2780), suggested that cardioprotection takes place without interfering negatively with the anticancer activity of doxorubicin [57]. In vitro experiments in fact suggested the opposite, i.e., MnDPDP, and in particular DPDP, exerted an anticancer activity of its own. In addition to the above-described potential use of MnPLED derivatives in anthracycline-induced cardiotoxicity, preclinical studies have demonstrated that MnDPDP protects against myelosuppressive effects of paclitaxel [32]. Furthermore, coadministration of MnDPDP with paclitaxel dramatically improved the survival rate of mice infected with Staphylococcus aureus [32], revealing a possible preventive effect on febril neutropenia. Importantly, the cytoprotective effect of MnDPDP was obtained without diminishing the anticancer efficacy. Contrary, MnDPDP was shown to enhance the anticancer effect of oxaliplatin toward colon cancer cells under in vitro and in vivo conditions, and it displayed a distinct anticancer effect of its own [31], [32]. The reason why MnDPDP exerts anticancer activity is poorly understood. However, cancer cells require more iron in order to support their increased rates of proliferation [60]. So “iron starvation” through chelation to PLED may explain this phenomenon.
When an MRI dose (i.e., 5-10 μmol/kg) is injected into humans or rats, about 80% of the Mn2+ is released from DPDP or its dephosphorylated counterparts, and only about 20% stays bound to the chelator. Release of paramagnetic Mn2+ is in fact a prerequisite for the diagnostic MRI properties of MnDPDP [34], whereas the in vivo SOD mimetic activity fully depends on the intact manganese complex [53]. Manganese is an essential as well as potentially neurotoxic metal [61]. It has been known for many years that, under conditions of chronic exposure to manganese, a syndrome of extrapyramidal dysfunction similar to Parkinson's disease may occur. The neurological symptoms correlate with accumulation of manganese in the basal ganglia, seen as hyperintensity on a T1-weighted MRI [61], [62]. For MRI purposes and for occasional therapeutic use, dissociation of Mn2+ from MnDPDP does not result in an increase in T1-weighted MRI signal of the basal ganglia in humans [63]. However, for more frequent use, as in therapeutic use, cumulative manganese toxicity can represent a problem.
Manganese bound to DPDP or PLED is most probably nontoxic and is readily excreted through the kidneys [64]. A considerable part of Mn2+ release from MnDPDP is governed by the presence of a limited amount of free or loosely bound plasma Zn2+ [64]. Replacement of 80% of Mn2+with Ca2+, generating a compound known as Ca4Mn(DPDP)5, is enough for binding a considerable amount of the readily available plasma Zn2+, resulting in considerably less Mn2+ release and retention in the brain and other organs. Karlsson et al. [64] reported that replacement of 80% of the Mn2+ with Ca2+ more than doubles the in vivo stability of the Mn complex and results in significantly (∼40%) less retention of manganese in the brain compared with MnDPDP (at equimolar manganese doses) after repeated dosing (39 doses) at more than 30 times the assumed clinical dose (per dose) in rats. At equivalent Mn2+ doses, Ca4Mn(DPDP)5 is significantly more efficacious than MnDPDP to protect BALB/c mice against myelosuppressive effects of the chemotherapy drug oxaliplatin [64], and it displayed distinct tumoricidal effects [64].
Clinical Evidence That MnDPDP Protects Against Oxidative Stress and CIPN
In a recent feasibility study, a relatively small number of stage III colon cancer patients were followed throughout the first 3 of 12 scheduled FOLFOX cycles [55] and randomized to a 5-minute infusion of either MnDPDP or placebo (7 in each group). AEs were evaluated according to the NCI-CTCAE and the Sanofi-NCI criteria. There were four AEs of grade 3 (severe) and one AE of grade 4 (life threatening) in four patients in the placebo group, whereas there were none in the MnDPDP group (Table 1). Of the grade 3 and 4 events, two were neutropenia and one was neurosensory toxicity (CIPN). There were 16 AEs grade ≥ 2 in the placebo group, whereas there were only 2 in the MnDPDP group. Furthermore, white blood cell counts were significantly higher in the MnDPDP group than in the placebo group (P < .01) after treatment with FOLFOX.
There were eight events (in four patients) of mild (grade 1) diarrhea in the MnDPDP group but none in the placebo group (Table 1). When MnDPDP is used as an MRI contrast agent, mild diarrhea has been reported to occur. Interestingly, a phase III randomized trial of adding topical nitroglycerin to first-line carboplatin chemotherapy for advanced non–small cell lung cancer showed a statistical higher frequency of diarrhea in the nitroglycerin group compared to the placebo group, 23% versus 14% [65]. Both MnDPDP and nitroglycerin increase the intracellular content of cGMP through an NO-mediated activation of guanylate cyclase (see Figure 3), which may stimulate intestinal secretion and hence strengthen FOLFOX-induced diarrhea. Addition of topical nitroglycerin to carboplatin-based chemotherapy in non–small cell lung cancer did not offer any tumoricidal benefit.
Findings from a case report [54] and the above-mentioned feasibility/translational study in stage III colon cancer patients [55] suggest that MnDPDP, through its SOD mimetic activity, protects blood, nervous, and mucous tissues in patients during chemotherapy with Nordic FLOX/FOLFOX (oxaliplatin + 5-FU) (Table 1). Coriat and coworkers [29] have also published promising results with MnDPDP in patients with preexisting oxaliplatin-associated CIPN.
The case report of Yri et al. [54] described a patient who received 15 palliative cycles of oxaliplatin plus 5-FU/LV (Nordic FLOX regimen). It suggested that MnDPDP protects against oxaliplatin-associated CIPN. In 14 of the cycles, the patient received pretreatment with MnDPDP. The patient received an accumulated dose of 1275 mg/m2 oxaliplatin, which is a dose likely to give cumulative CIPN symptoms. No such symptoms were detected except during the fifth cycle, when MnDPDP was deliberately left out and the patient experienced an acute grade 2 CIPN. After five cycles, the performance status for the patient was drastically improved, and the demand for analgesics was significantly reduced. Neutropenia did not occur during any of the chemotherapy cycles. This patient however over 7 months received a cumulative MnDPDP dose as high as 140 μmol/kg and showed Parkinson's disease–like symptoms and increased MRI signal intensity in the basal ganglia. From diagnostic MRI, it is known that a cumulative dose of 15 μmol/kg, corresponding to 15 μmol/kg Mn2+ (5 + 10), does not cause any increase in the MRI signal (Wang et al., 1997). However, the upper limit is not known. Karlsson et al. [55] used 2 μmol/kg MnDPDP in three cycles. Coriat et al. [29] used 5 μmol/kg in up to eight cycles, giving rise to a cumulative dose of 40 μmol/kg. They reported that the mean plasma manganese content increased from 11.8 ± 5.5 nM to 19.8 ± 4.3 nM after eight cycles of MnDPDP cotreatment, all within normal values. However, plasma manganese is considered a weak predictor of manganese neurotoxicity. A much more reliable predictor is T1-weighted MRI [62]. When it comes to Ca4Mn(DPDP)5, it seems realistic to reduce the dose down to 1 μmol/kg Mn2+, which should be low enough to allow at least 15 repeated treatments without causing manganese toxicity. However, this is a theoretical assumption that has to be proven clinically by brain MRI.
In the Coriat study, 22 cancer patients with grade ≥ 2 oxaliplatin-associated CIPN received intravenous MnDPDP following oxaliplatin. Neuropathic effects were monitored for up to eight cycles of oxaliplatin and MnDPDP. In 77% of the patients treated with oxaliplatin and MnDPDP, CIPN improved or stabilized after four cycles. After eight cycles, CIPN was downgraded in six of seven patients. Prior to enrollment, patients received an average of 880 ± 239 mg/m2 oxaliplatin. Patients treated with MnDPDP tolerated an additional dose of 458 ± 207 mg/m2 oxaliplatin despite preexisting CIPN. MnDPDP responders managed a cumulative dose of 1426 ± 204 mg/m2 oxaliplatin. The clinical findings were further backed up by relevant preclinical tests in mice. These tests demonstrated convincingly that MnDPDP prevented motor and sensory dysfunction [29]. As an indicator of oxidative stress, advanced oxidized protein products (AOPPs) were monitored both in the patients and in the mice. In mice, serum AOPPs decreased after 4 weeks of MnDPDP cotreatment, and serum AOPPs were lower in human responders compared with those in nonresponders.
In opposite to MnDPDP, no preclinical data are present showing efficacy of Ca4Mn(DPDP)5 against CIPN. However, preliminary results from a phase II study (PLIANT) on Ca4Mn(DPDP)5 (ClinicalTrials.gov Identifier: NCT01619423) may suggest efficacy also of this compound against oxaliplatin-associated CIPN (PledPharma's web site; Devalingam Mahalingam at the MASCC meeting in Copenhagen 2015; and Bengt Glimelius at the ASCO meeting in Chicago 2016). However, these results are obscured by a surprisingly low number of grade 3 to 4 neutropenias and a seemingly lower than expected efficacy of FOLFOX in the placebo group (Mahalingam D; MASCC meeting in Copenhagen, June 26, 2015). The reported ORR of 27% in the placebo group is more or less identical to what one expects from 5-FU alone. As stated earlier in the review, addition of oxaliplatin to 5-FU has increased the ORR from about 27% to 45% [2], [8], [9], [10].
Nevertheless, there is little or nothing suggesting any major difference in pharmacological activity between MnDPDP and Ca4Mn(DPDP)5, except that the latter has a considerably higher therapeutic index at equivalent manganese doses [64]. The therapeutic index of Ca4Mn(DPDP)5 probably allows use in at least 15 cycles of chemotherapy.
Conclusion
MnDPDP, after being metabolized into MnPLED, attacks cellular oxidative stress at three levels: 1) it lowers the intracellular level of O2•− directly through its MnSOD mimetic activity; 2) it binds and disarms transition metals, particularly iron and copper, and thus arrests incorporation of •NO2 into Tyr-34 and Tyr-74 of mitochondrial MnSOD and cytochrome c, respectively, and production of highly toxic OH• radicals; and 3) it replaces endogenous mitochondrial MnSOD that already has been irreversibly inactivated (Figure 4). Through its combined MnSOD mimetic activities and iron- and copper-binding properties, MnDPDP and Ca4Mn(DPDP)5 are highly efficacious inhibitors of CIPN and other pathological conditions caused by oxidative stress.
Footnotes
This work was supported by a grant from Medical Research Council of Southeast Sweden (FORSS-85191).
Jan Olof G. Karlsson and Per Jynge are two of the founders of PledPharma AB. Karlsson and Jynge are inventors on two granted patent families (e.g., US6258828 and US6147094) covering the therapeutic use of mangafodipir, which are owned by GE Healthcare. Karlsson owns shares in PledPharma AB. Rolf G.G. Andersson and Karlsson are inventors on two granted patent families (e.g., US8377969, US8633174, and US9187509) covering the therapeutic use of calmangafodipir, which are owned by PledPharma AB. Karlsson is a former employee of GE Healthcare and PledPharma AB. Andersson is former Board member of PledPharma.
References
- 1.Ferlay J, Autier P, Boniol M, Heanue M, Colombet M, Boyle P. Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol. 2007;18:581–592. doi: 10.1093/annonc/mdl498. [DOI] [PubMed] [Google Scholar]
- 2.O'Neil BH, Goldberg RM. Innovations in chemotherapy for metastatic colorectal cancer: an update of recent clinical trials. Oncologist. 2008;13:1074–1083. doi: 10.1634/theoncologist.2008-0083. [DOI] [PubMed] [Google Scholar]
- 3.Moertel CG, Fleming TR, Macdonald JS, Haller DG, Laurie JA, Tangen CM, Ungerleider JS, Emerson WA, Tormey DC, Glick JH. Fluorouracil plus levamisole as effective adjuvant therapy after resection of stage III colon carcinoma: a final report. Ann Intern Med. 1995;122:321–326. doi: 10.7326/0003-4819-122-5-199503010-00001. [DOI] [PubMed] [Google Scholar]
- 4.O'Connell MJ, Laurie JA, Kahn M, Fitzgibbons RJ, Jr., Erlichman C, Shepherd L, Moertel CG, Kocha WI, Pazdur R, Wieand HS. Prospectively randomized trial of postoperative adjuvant chemotherapy in patients with high-risk colon cancer. J Clin Oncol. 1998;16:295–300. doi: 10.1200/JCO.1998.16.1.295. [DOI] [PubMed] [Google Scholar]
- 5.Haller DG, Catalano PJ, Macdonald JS, O'Rourke MA, Frontiera MS, Jackson DV, Mayer RJ. Phase III study of fluorouracil, leucovorin, and levamisole in high-risk stage II and III colon cancer: final report of Intergroup 0089. J Clin Oncol. 2005;23:8671–8678. doi: 10.1200/JCO.2004.00.5686. [DOI] [PubMed] [Google Scholar]
- 6.André T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, Topham C, Zaninelli M, Clingan P, Bridgewater J. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004;350:2343–2351. doi: 10.1056/NEJMoa032709. [DOI] [PubMed] [Google Scholar]
- 7.André T, Iveson T, Labianca R, Meyerhardt JA, Souglakos I, Yoshino T, Paul J, Sobrero A, Taieb J, Shields AF. The IDEA (International Duration Evaluation of Adjuvant Chemotherapy) Collaboration: prospective combined analysis of phase III trials investigating duration of adjuvant therapy with the FOLFOX (FOLFOX4 or Modified FOLFOX6) or XELOX (3 versus 6 months) regimen for patients with stage III colon cancer: trial design and current status. Curr Colorectal Cancer Rep. 2013;9:261–269. doi: 10.1007/s11888-013-0181-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18:2938–2947. doi: 10.1200/JCO.2000.18.16.2938. [DOI] [PubMed] [Google Scholar]
- 9.Goldberg RM, Sargent DJ, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Pitot HC, Alberts SR. A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol. 2004;22:23–30. doi: 10.1200/JCO.2004.09.046. [DOI] [PubMed] [Google Scholar]
- 10.Ducreux M, Bennouna J, Hebbar M, Ychou M, Lledo G, Conroy T, Adenis A, Faroux R, Rebischung C, Bergougnoux L. Capecitabine plus oxaliplatin (XELOX) versus 5-fluorouracil/leucovorin plus oxaliplatin (FOLFOX-6) as first-line treatment for metastatic colorectal cancer. Int J Cancer. 2011;128:682–690. doi: 10.1002/ijc.25369. [DOI] [PubMed] [Google Scholar]
- 11.Sørbye H, Glimelius B, Berglund A, Fokstuen T, Tveit KM, Braendengen M, Øgreid D, Dahl O. Multicenter phase II study of Nordic fluorouracil and folinic acid bolus schedule combined with oxaliplatin as first-line treatment of metastatic colorectal cancer. J Clin Oncol. 2004;22:31–38. doi: 10.1200/JCO.2004.05.188. [DOI] [PubMed] [Google Scholar]
- 12.Grisold W, Cavaletti G, Windebank AJ. Peripheral neuropathies from chemotherapeutics and targeted agents: diagnosis,treatment, and prevention. Neuro Oncol. 2012;14(Suppl. 4):iv45–iv54. doi: 10.1093/neuonc/nos203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cassidy J, Clarke S, Díaz-Rubio E, Scheithauer W, Figer A, Wong R, Koski S, Rittweger K, Gilberg F, Saltz L. XELOX vs FOLFOX-4 as first-line therapy for metastatic colorectal cancer: NO16966 updated results. Br J Cancer. 2011;105:58–64. doi: 10.1038/bjc.2011.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hong YS, Nam BH, Kim KP, Kim JE, Park SJ, Park YS, Park JO, Kim SY, Kim TY, Kim JH. Oxaliplatin, fluorouracil, and leucovorin versus fluorouracil and leucovorin as adjuvant chemotherapy for locally advanced rectal cancer after preoperative chemoradiotherapy (ADORE): an open-label, multicentre, phase 2, randomized controlled trial. Lancet Oncol. 2014;15:1245–1253. doi: 10.1016/S1470-2045(14)70377-8. [DOI] [PubMed] [Google Scholar]
- 15.Loprinzi CL, Qin R, Dakhil SR, Fehrenbacher L, Flynn KA, Atherton P, Seisler D, Qamar R, Lewis GC, Grothey A. Phase III randomized, placebo-controlled, double-blind study of intravenous calcium and magnesium to prevent oxaliplatin-induced sensory neurotoxicity (N08CB/Alliance) J Clin Oncol. 2014;32:997–1005. doi: 10.1200/JCO.2013.52.0536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Park SB, Goldstein D, Krishnan AV, Lin CS, Friedlander ML, Cassidy J, Koltzenburg M, Kiernan MC. Chemotherapy-induced peripheral neurotoxicity: a critical analysis. CA Cancer J Clin. 2013;63:419–437. doi: 10.3322/caac.21204. [DOI] [PubMed] [Google Scholar]
- 17.Carozzi VA, Marmiroli P, Cavaletti G. The role of oxidative stress and anti-oxidant treatment in platinum-induced peripheral neurotoxicity. Curr Cancer Drug Targets. 2010;10:670–782. doi: 10.2174/156800910793605820. [DOI] [PubMed] [Google Scholar]
- 18.Janes K, Doyle T, Bryant L, Esposito E, Cuzzocrea S, Ryerse J, Bennett GJ, Salvemini D. Bioenergetic deficits in peripheral nerve sensory axons during chemotherapy-induced neuropathic pain resulting from peroxynitrite-mediated post-translational nitration of mitochondrial superoxide dismutase. Pain. 2014;154:2432–2440. doi: 10.1016/j.pain.2013.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zheng H, Xiao WH, Bennett GJ. Functional deficits in peripheral nerve mitochondria in rats with paclitaxel- and oxaliplatin-evoked painful peripheral neuropathy. Exp Neurol. 2011;232:154–161. doi: 10.1016/j.expneurol.2011.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xiao WH, Zheng H, Bennett GJ. Characterization of oxaliplatin-induced chronic painful peripheral neuropathy in the rat and comparison with the neuropathy induced by paclitaxel. Neuroscience. 2012;203:194–206. doi: 10.1016/j.neuroscience.2011.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xiao WH, Bennett GJ. Effects of mitochondrial poisons on the neuropathic pain produced by the chemotherapeutic agents, paclitaxel and oxaliplatin. Pain. 2012;153:704–709. doi: 10.1016/j.pain.2011.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Areti A, Yerra VG, Naidu V, Kumar A. Oxidative stress and nerve damage: role in chemotherapy induced peripheral neuropathy. Redox Biol. 2014;2:289–295. doi: 10.1016/j.redox.2014.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jiang W, Tang L, Zeng J, Chen B. Adeno-associated virus mediated SOD gene therapy protects the retinal ganglion cells from chronic intraocular pressure elevation induced injury via attenuating oxidative stress and improving mitochondrial dysfunction in a rat model. Am J Transl Res. 2016;8:799–810. [PMC free article] [PubMed] [Google Scholar]
- 24.Albers JW, Chaudhry V, Cavaletti G, Donehower RC. Interventions for preventing neuropathy caused by cisplatin and related compounds. Cochrane Database Syst Rev. 2014:CD005228. doi: 10.1002/14651858.CD005228.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gamelin L, Boisdron-Celle M, Delva R, Guérin-Meyer V, Ifrah N, Morel A, Gamelin E. Prevention of oxaliplatin-related neurotoxicity by calcium and magnesium infusions: a retrospective study of 161 patients receiving oxaliplatin combined with 5-fluorouracil anleucovorin for advanced colorectal cancer. Clin Cancer Res. 2004;10:4055–4061. doi: 10.1158/1078-0432.CCR-03-0666. [DOI] [PubMed] [Google Scholar]
- 26.Hochster HS, Grothey A, Childs BH. Use of calcium and magnesium salts to reduce oxaliplatin-related neurotoxicity. J Clin Oncol. 2007;25:4028–4029. doi: 10.1200/JCO.2007.13.5251. [DOI] [PubMed] [Google Scholar]
- 27.Gamelin L, Boisdron-Celle M, Morel A, Poirier AL, Berger V, Gamelin E, Tournigand C, de Gramont A. Oxaliplatin-related neurotoxicity: interest of calcium-magnesium infusion and no impact on its efficacy. J Clin Oncol. 2008;26:1188–1189. doi: 10.1200/JCO.2007.15.3767. [DOI] [PubMed] [Google Scholar]
- 28.Grothey A, Nikcevich DA, Sloan JA, Kugler JW, Silberstein PT, Dentchev T, Wender DB, Novotny PJ, Chitaley U, Alberts SR. Intravenous calcium and magnesium for oxaliplatin-induced sensory neurotoxicity in adjuvant colon cancer: NCCTG N04C7. J Clin Oncol. 2011;29:421–427. doi: 10.1200/JCO.2010.31.5911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Coriat R, Alexandre J, Nicco C, Quinquis L, Benoit E, Chéreau C, Lemaréchal H, Mir O, Borderie D, Tréluyer JM. Treatment of oxaliplatin-induced peripheral neuropathy by intravenous mangafodipir. J Clin Invest. 2014;124:262–272. doi: 10.1172/JCI68730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Campolo N, Bartesaghi S, Radi R. Metal-catalyzed protein tyrosine nitration in biological systems. Redox Rep. 2014;19:221–231. doi: 10.1179/1351000214Y.0000000099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Laurent A, Nicco C, Chéreau C, Goulvestre C, Alexandre J, Alves A, Lévy E, Goldwasser F, Panis Y, Soubrane O. Controlling tumor growth by modulating endogenous production of reactive oxygen species. Cancer Res. 2005;65:948–956. [PubMed] [Google Scholar]
- 32.Alexandre J, Nicco C, Chéreau C, Laurent A, Weill B, Goldwasser F, Batteux F. Improvement of the therapeutic index of anticancer drugs by the superoxide dismutase mimic mangafodipir. J Natl Cancer Inst. 2006;98:236–244. doi: 10.1093/jnci/djj049. [DOI] [PubMed] [Google Scholar]
- 33.Doroshow JH. Redox modulation of chemotherapy-induced tumor cell killing and normal tissue toxicity. J Natl Cancer Inst. 2006;98:223–225. doi: 10.1093/jnci/djj065. [editorial] [DOI] [PubMed] [Google Scholar]
- 34.Karlsson JO, Ignarro LJ, Lundström I, Jynge P, Almén T. Calmangafodipir [Ca4Mn(DPDP)5], mangafodipir (MnDPDP) and MnPLED with special reference to their SOD mimetic and therapeutic properties. Drug Discov Today. 2015;20:411–421. doi: 10.1016/j.drudis.2014.11.008. [DOI] [PubMed] [Google Scholar]
- 35.Holmes J, Stanko J, Varchenko M, Ding H, Madden VJ, Bagnell CR, Wyrick SD, Chaney SG. Comparative neurotoxicity of oxaliplatin, cisplatin, and ormaplatin in a Wistar rat model. Toxicol Sci. 1998;46:342–351. doi: 10.1006/toxs.1998.2558. [DOI] [PubMed] [Google Scholar]
- 36.McCord JM, Fridovich I. The reduction of cytochrome c by milk xanthine oxidase. J Biol Chem. 1968;243:5753–5760. [PubMed] [Google Scholar]
- 37.McCord JM, Fridovich I. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein) J Biol Chem. 1969;244:6049–6055. [PubMed] [Google Scholar]
- 38.Weisiger RA, Fridovich I. Superoxide dismutase. Organelle specificity. J Biol Chem. 1973;248:3582–3592. [PubMed] [Google Scholar]
- 39.Weisiger RA, Fridovich I. Mitochondrial superoxide dismutase. Site of synthesis and intramitochondrial localization. J Biol Chem. 1973;248:4793–4796. [PubMed] [Google Scholar]
- 40.Beckman JS. Oxidative damage and tyrosine nitration from peroxynitrite. Chem Res Toxicol. 1996;9:836–844. doi: 10.1021/tx9501445. [DOI] [PubMed] [Google Scholar]
- 41.Beckman JS, Koppenol WH. Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and ugly. Am J Phys. 1996;271:C1424–C1437. doi: 10.1152/ajpcell.1996.271.5.C1424. [DOI] [PubMed] [Google Scholar]
- 42.Radi R. Nitric oxide, oxidants, and protein tyrosine nitration. Proc Natl Acad Sci. 2004;101:4003–4008. doi: 10.1073/pnas.0307446101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Radi R. Protein tyrosine nitration: biochemical mechanisms and structural basis of functional effects. Acc Chem Res. 2013;46:550–559. doi: 10.1021/ar300234c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.MacMillan-Crow LA, Crow JP, Kerby JD, Beckman JS, Thompson JA. Nitration and inactivation of manganese superoxide dismutase in chronic rejection of human renal allografts. Proc Natl Acad Sci U S A. 1996;93:11853–11858. doi: 10.1073/pnas.93.21.11853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bronte V, Kasic T, Gri G, Gallana K, Borsellino G, Marigo I, Battistini L, Iafrate M, Prayer-Galetti T, Pagano F. Boosting antitumor responses of T lymphocytes infiltrating human prostate cancers. J Exp Med. 2005;201:1257–1268. doi: 10.1084/jem.20042028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Agarwal R, MacMillan-Crow LA, Rafferty TM, Saba H, Roberts DW, Fifer EK, James LP, Hinson JA. Acetaminophen-induced hepatotoxicity in mice occurs with inhibition of activity and nitration of mitochondrial manganese superoxide dismutase. J Pharmacol Exp Ther. 2011;337:110–116. doi: 10.1124/jpet.110.176321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Batinic-Haberle I, Benov L, Spasojevic I, Fridovich I. The ortho effect makes manganese(III) meso-tetrakis(N-methylpyridinium-2-yl)porphyrin a powerful and potentially useful superoxide dismutase mimic. J Biol Chem. 1998;273:24521–24528. doi: 10.1074/jbc.273.38.24521. [DOI] [PubMed] [Google Scholar]
- 48.Rasola A, Bernardi P. The mitochondrial permeability transition pore and its adaptive responses in tumor cells. Cell Calcium. 2014;56:437–445. doi: 10.1016/j.ceca.2014.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Richardson DR, Ponka P. The molecular mechanisms of the metabolism and transport of iron in normal and neoplastic cells. Biochim Biophys Acta. 1997;1331:1–40. doi: 10.1016/s0304-4157(96)00014-7. [DOI] [PubMed] [Google Scholar]
- 50.Broeyer FJ, Osanto S, Suzuki J, de Jongh F, van Slooten H, Tanis BC, Bruning T, Bax JJ, Ritsema van Eck HJ, de Kam ML. Evaluation of lecithinized human recombinant superoxide dismutase as cardioprotectant in anthracycline-treated breast cancer patients. Br J Clin Pharmacol. 2014;78:950–960. doi: 10.1111/bcp.12429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kensler TW, Bush DM, Kozumbo WJ. Inhibition of tumor promotion by a biomimetic superoxide dismutase. Science. 1983;221:75–77. doi: 10.1126/science.6857269. [DOI] [PubMed] [Google Scholar]
- 52.Asplund A, Grant D, Karlsson JO. Mangafodipir (MnDPDP)- and MnCl2-induced endothelium-dependent relaxation in bovine mesenteric arteries. J Pharmacol Exp Ther. 1994;271:609–614. [PubMed] [Google Scholar]
- 53.Brurok H, Ardenkjaer-Larsen JH, Hansson G, Skarra S, Berg K, Karlsson JO, Laursen I, Jynge P. Manganese dipyridoxyl diphosphate: MRI contrast agent with antioxidative and cardioprotective properties? Biochem Biophys Res Commun. 1999;254:768–772. doi: 10.1006/bbrc.1998.0131. [DOI] [PubMed] [Google Scholar]
- 54.Yri OE, Vig J, Hegstad E, Hovde O, Pignon I, Jynge P. Mangafodipir as a cytoprotective adjunct to chemotherapy—a case report. Acta Oncol. 2009;48:633–635. doi: 10.1080/02841860802680427. [DOI] [PubMed] [Google Scholar]
- 55.Karlsson JO, Adolfsson K, Thelin B, Jynge P, Andersson RG, Falkmer UG. First clinical experience with the magnetic resonance imaging contrast agent and superoxide dismutase mimetic mangafodipir as an adjunct in cancer chemotherapy-a translational study. Transl Oncol. 2012;5:32–38. doi: 10.1593/tlo.11277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rocklage SM, Cacheris WP, Quay SC, Hahn FE, Raymond KN. Manganese(II) N,N’-dipyridoxylethylenediamine-N,N’-diacetate 5,5-bis(phosphate). Synthesis and characterization of a paramagnetic chelate for magnetic resonance imaging enhancement. Inorg Chem. 1989;28:477–485. [Google Scholar]
- 57.Kurz T, Grant D, Andersson RG, Towart R, De Cesare M, Karlsson JO. Effects of MnDPDP and ICRF-187 on doxorubicin-induced cardiotoxicity and anticancer activity. Transl Oncol. 2012;5:252–259. doi: 10.1593/tlo.11304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Laskar A, Miah S, Andersson RG, Li W. Prevention of 7β-hydroxycholesterol-induced cell death by mangafodipir is mediated through lysosomal and mitochondrial pathways. Eur J Pharmacol. 2010;640:124–128. doi: 10.1016/j.ejphar.2010.04.046. [DOI] [PubMed] [Google Scholar]
- 59.Towart R, Jynge P, Refsum H, Karlsson JOG. Manganese dipyridoxyl diphosphate protects against acute anthracycline induced cardiotoxicity mice. Arch Pharmacol. 1998;358(Suppl. 2):R626. [Google Scholar]
- 60.Wang J, Pantopoulos K. Regulation of cellular iron metabolism. Biochem J. 2011;434:365–381. doi: 10.1042/BJ20101825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Aschner M, Erikson KM, Dorman DC, Aschner M. Manganese dosimetry: species differences and implications for neurotoxicity. Crit Rev Toxicol. 2005;35:1–32. doi: 10.1080/10408440590905920. [DOI] [PubMed] [Google Scholar]
- 62.Takagi Y, Okada A, Sando K, Wasa M, Yoshida H, Hirabuki N. Evaluation of indexes of in vivo manganese status and the optimal intravenous dose for adult patients undergoing home parenteral nutrition. Am J Clin Nutr. 2002;75:112–118. doi: 10.1093/ajcn/75.1.112. [DOI] [PubMed] [Google Scholar]
- 63.Wang C., Gordon P.B., Hustvedt S.O., Grant D., Sterud A.T., Martinsen I., Ahlström H., Hemmingsson A. MR imaging properties and pharmacokinetics of MnDPDP in healthy volunteers. Acta Radiol. 1997;38:665–676. doi: 10.1080/02841859709172399. [DOI] [PubMed] [Google Scholar]
- 64.Karlsson JO, Kurz T, Flechsig S, Näsström J, Andersson RG. Superior therapeutic index of calmangafodipir in comparison to mangafodipir as a chemotherapy adjunct. Transl Oncol. 2012;5:492–502. doi: 10.1593/tlo.12238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Davidson A, Veillard AS, Tognela A, Chan MM, Hughes BG, Boyer M, Briscoe K, Begbie S, Abdi E, Crombie C. A phase III randomized trial of adding topical nitroglycerin to first-line chemotherapy for advanced nonsmall-cell lung cancer: the Australasian lung cancer trials group NITRO trial. Ann Oncol. 2015;26:2280–2286. doi: 10.1093/annonc/mdv373. [DOI] [PubMed] [Google Scholar]