Abstract
Pulmonary complications, including acute respiratory distress syndrome (ARDS), are well described in P. falciparum (PF) and to a lesser extent in other malaria species. In non-endemic areas, malaria diagnosis may be overlooked; if a thorough travel history is not obtained on all patients with acute febrile illness. Three patients with malaria associated respiratory distress were admitted to our intensive care unit. The diagnosis was delayed; however, all patients received artesunate and intensive therapy with a satisfactory outcome. One patient presented with respiratory disease while the others developed ARDS during or following appropriate therapy. Similarly, level of parasitemia was variable ranging from undetectable to over 5%. Variability in timing and severity of illness is exciting and gives emphasis to the different pathological processes contemplated in this complication.
Keywords: Malaria, Falciparum, Pulmonary, ARDS
1. Introduction
More than 10% of adults with severe falciparum malaria (SFM) requiring intensive care unit (ICU) admission will die from the disease [1]. Altered pulmonary physiology in Plasmodium falciparum infection includes airflow obstruction, impaired ventilation, reduced gas transfer, and increased pulmonary phagocytic activity [2]. Adults with SFM may develop non-cardiogenic pulmonary oedema and acute respiratory distress syndrome (ARDS) that leads to a high mortality rate. This is seen most frequently in non-immune individuals with P. falciparum infection. The diagnosis of ARDS is based on the Berlin criteria, and includes bilateral pulmonary infiltrates, PaO2/FiO2 ratio ranging between 100 and 300 and normal left ventricular function (LVF) on echocardiogram (ECHO) [3]. Notably, ARDS has been reported in 5–25% of adults with SFM and in 1–10% of patients with severe P. vivax infection. The associated attributable mortality could reach up to 20% in developed countries [4].
1.1. Case patient 1
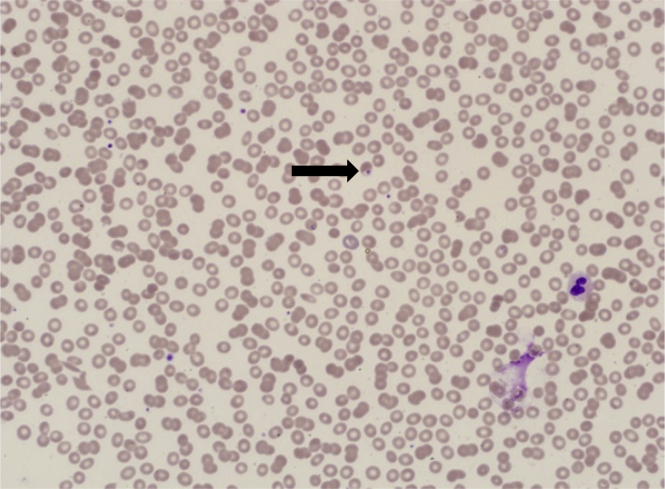
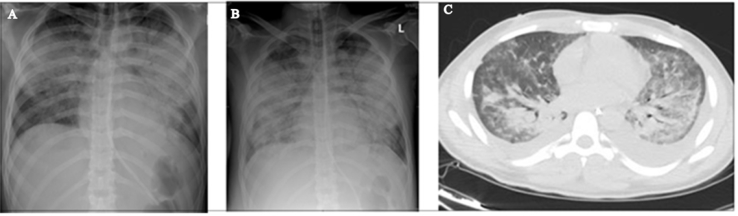
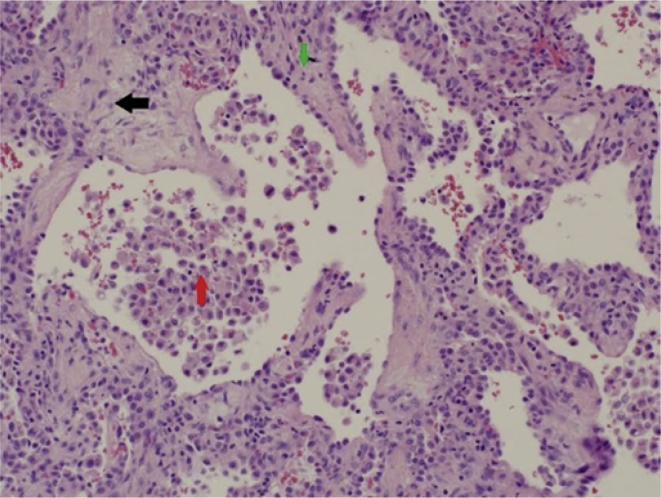
A 29-year-old patient presented with fever, flu-like illness, cough, and shortness of breath (SOB) for 5 days. He was assessed in different private clinics where he was given antibiotics for upper respiratory tract infection. When admitted to the hospital, a chest radiograph (CXR) showed bilateral basal and perihilar patchy consolidation with bilateral diffuse alveolar infiltration. A computerised tomography scan (CT scan) of the chest showed bilateral ground-glass opacities consistent with acute ARDS. Nasopharyngeal aspirate (NPA) and sputum yielded negative results for Middle East respiratory syndrome coronavirus (MERS-CoV) and influenza viruses. He was subsequently referred to our centre where he was immediately intubated and connected to a ventilator. On arrival he was conscious, in respiratory distress, had a pulse rate (PR) of 125 beats/min, blood pressure (BP) of 80/60 mmHg, respiratory rate (RR) of 30 breaths/min, temperature of 39 °C, and an oxygen saturation (SaO2) of 80% on room air. There were bilateral diffuse crepitations. His arterial blood gas (ABG) results showed pH 7.33, PaCO2 50 mmHg, PaO2 52.9 mmHg, SaO2 90%, HCO3− 26 mEq/L, and alveolar-arterial gradient 155 mmHg. The PO2/FiO2 ratio was 106. An ECHO revealed normal LVF and an ejection fraction (EF) of 55%. His initial laboratory investigations revealed white blood count (WBC) 10.0 × 109/L, haemoglobin 14.9 g/dL, platelet count 47 × 109/L, urea 12 mmol/L, creatinine 137 μmol/L, albumin 25 g/L, bilirubin 83 μmol/L, alanine aminotransferase (ALT) 115 U/L, lactate dehydrogenase (LDH) 733 U/L, markedly raised creatine kinase (CK) 6640 U/L, and C-reactive protein (CRP) 318 mg/L. Polymerase chain reaction (PCR) for MERS-CoV, respiratory virus panel test and atypical pneumonia screen all yielded negative results, and his sputum and blood were sterile. He was started on piperacillin/tazobactam (4.5 g q6h), oseltamivir (75mg BID), and azithromycin (500mg OD). On further inquiry, the patient had paid a visit to Jizan near the borders of Yemen. He was not on malaria prophylaxis. An urgent blood smear showed ring forms of P. falciparum (PF) with a parasitaemia of 1.3% (Fig. 1). Intravenous artesunate, 2.4 mg/kg (per dose) was given at times 0, 12 hours, 24 hours, 48 hours, and 72 hours. Parasitaemia disappeared by the third day. Two weeks later he continued to worsen with fever, had worsening infiltrates on CXR (Fig. 2A and B), and there was difficulty in weaning him off the ventilator. A CT scan of the chest showed bilateral pleural effusion, airspace shadowing (consolidation) and ground-glass appearance involving both lungs (Fig. 2C). The findings were suggestive of interstitial pneumonia, acute (adult) respiratory distress syndrome or pulmonary haemorrhage. He was commenced on high-dose prednisolone. Bronchoalveolar lavage (BAL) yielded negative results for MERS-CoV and influenza, but isolated extended-spectrum beta-lactamase (ESBL) Klebsiella pneumoniae. Piperacillin/tazobactam was changed to imipenem/cilastatin. A lung biopsy was reported to show moderate thickening of the alveolar walls with fibroblastic plug in both the alveolar spaces and bronchioles and marked intra-alveolar accumulation of histiocytes was noted. The features were consistent with the resolving phase of ARDS or cryptogenic organising pneumonia (Fig. 3). The steroids were continued. He started to improve and was weaned after three weeks of ventilation.
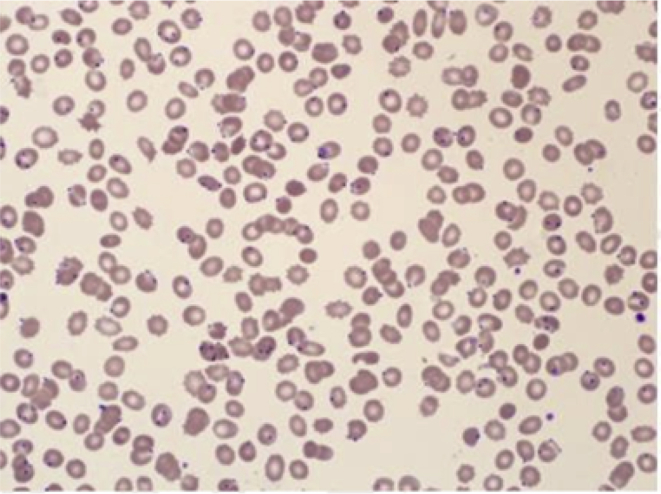
Fig. 1.
Peripheral blood film showing PF ring forms (Arrow).
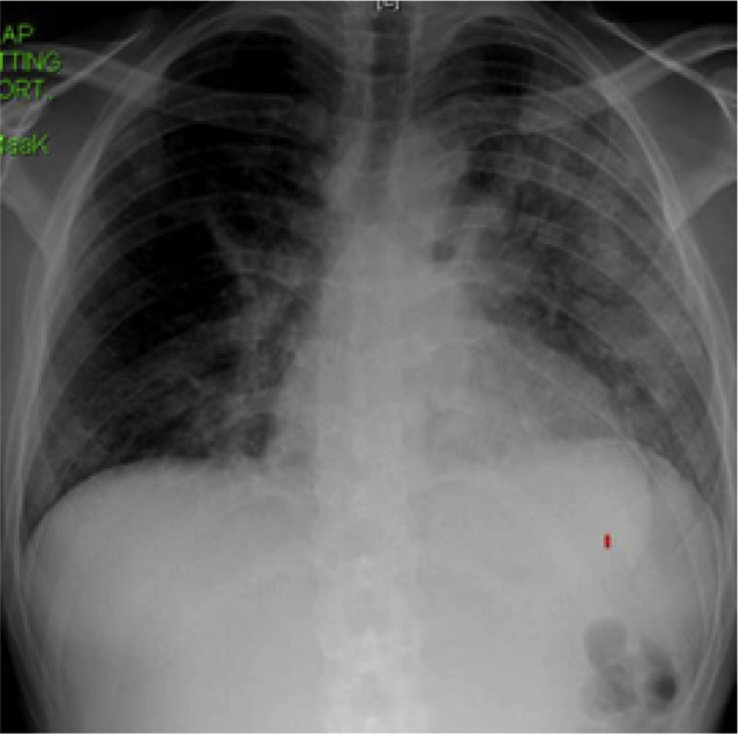
Fig. 2.
A. A PA CXR on admission to the hospital. B. PA CXR 2 weeks following admission. C. CT scan of the chest 2 weeks following admission.
Fig. 3.
Haemotoxylin and eosin (H&E) stain of lung tissue showing diffuse moderate thickening of the alveolar walls 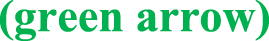 with fibroblastic plugs in both alveolar spaces and bronchioles (black arrow), and marked intra-alveolar accumulation of histiocytes
with fibroblastic plugs in both alveolar spaces and bronchioles (black arrow), and marked intra-alveolar accumulation of histiocytes  .
.
1.2. Case patient 2
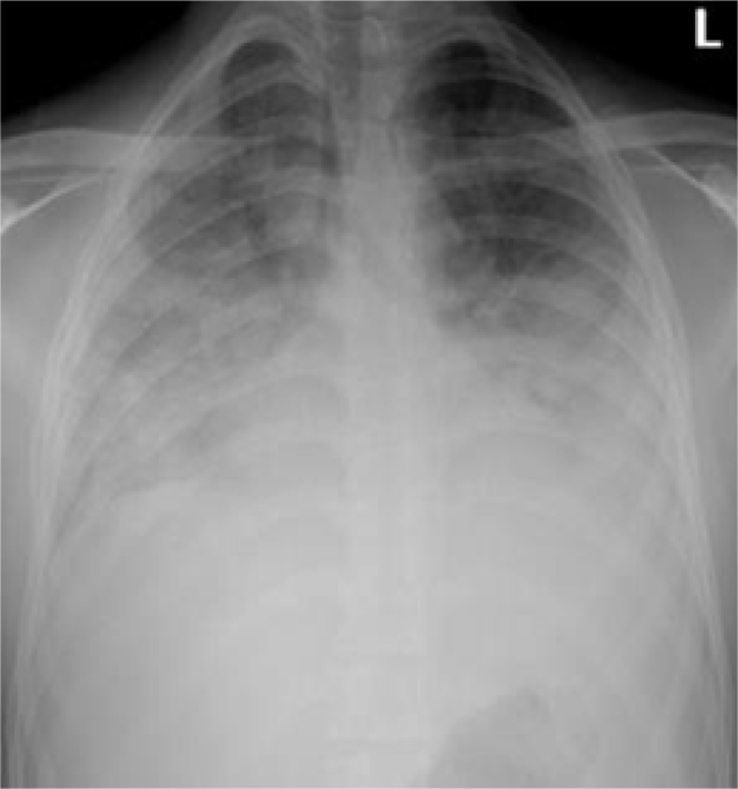
A 23-year-old man was admitted with fever, headache, anorexia, nausea, and vomiting for 5 days. Examination showed a temperature of 39.4 °C, PR 78 beats/min, RR 20 breaths/min, BP 100/50 mmHg, and SaO2 100% on room air. The examination was otherwise normal. Blood investigations revealed a WBC of 8.6 × 109/L, haemoglobin 15.5 g/dL, platelets 21 × 109/L, glucose 5 mmol/L, urea 11 mmol/L, creatinine 163 μmol/L, ALT 99 U/L, total bilirubin 50 μmol/L, and albumin 26 g/L. A CXR was normal and blood film showed PF with parasitaemia of 0.14%. He was started on oral artemether/lumefantrine, at an initial dose of 80/480 mg, followed 8 hours later by a repeat dose, then at the same dose twice daily for two days. A repeated blood film showed negative results for malaria. On the third day of admission and while being considered for discharge, he developed a cough, hemoptysis, breathlessness, and a fall in oxygen saturation. The RR was 35 breaths/min, SaO2 90–95% on 15 L of oxygen with a non-rebreather mask. His ABG results revealed a pH 7.48, PaO2 61 mmHg, PaCO2 32.8 mmHg, HCO3− 23.9 mEq/L, and lactate 1.9 mmol/L. The PaO2/FiO2 ratio was 192. A CXR displayed extensive bilateral shadowing consistent with pulmonary oedema or ARDS (Fig. 4). He was moved to the ICU where he received intravenous furosemide and bilevel positive airway pressure (BPAP) breathing support. An ECHO showed a normal LVF with EF of 55%. An NPA and sputum yielded negative results for influenza, respiratory syncytial virus (RSV) and MERS-CoV. His condition improved on supportive treatment, and he was discharged home 10 days after hospital admission.
Fig. 4.
AP CXR 3 days after admission showing extensive bilateral airspace disease.
1.3. Case patient 3
A 29-year-old patient presented to the hospital with a 7-day history of fever. He attended a private clinic on two occasions where antibiotics were prescribed. On the day of admission, he developed confusion, restlessness and urinary incontinence. There was a history of travel to the southern part of Saudi Arabia. He looked pale, jaundiced, and had a Glasgow coma scale (GCS) score of 12/15, but there were no signs of meningitis. The temperature was 36.9 °C, PR 120 beats/min, BP 90/50 mmHg, and RR 20 breaths/min. Apart from left-sided crepitation on the chest, the rest of the examination was normal. The initial blood investigations revealed a WBC of 9.9 × 109/L, platelets 12 × 109/L, and a peripheral blood film showed PF ring forms >5% (Fig. 5). There was evidence of hemolysis with bilirubin 208 μmol/L, reticulocyte count of 375 × 109/L (11%), LDH 2197 U/L, and low haptoglobin 0.07 g/L (normal range: 0.3–2.0 g/L). His haemoglobin dropped to 4 g/dL, requiring a blood transfusion. His serum albumin was low at 25 g/L, while CRP was high at 215 mg/L, and ferritin 2852 ng/mL. Two days following admission his blood sugar remained normal, but creatinine rose to 167 μmol/L and urea to 17.6 mmol/L. Both CK and myoglobin were raised at 1124 U/L and 2.74 nmol/L, respectively (normal range for myoglobin: <1.11 nmol/L). He received intravenous artesunate. On the third day of admission, he developed a cough and SOB. An initially normal CXR showed bilateral infiltrates more visible on the left lung (Fig. 6). An ABG analysis after 3 days on admission showed pH 7.44, PCO2 29.3 mmHg, PaO2 55.6 mmHg, HCO3− 19.5 mEq/L, FiO2 32%, and PaO2/FiO2 174. An ECHO revealed a normal LVF and EF, as well as a normal sized left atrium. Screening for viral pneumonia and bacterial cultures yielded negative results. He responded to high flow oxygen support with slow clearance of parasitaemia extending over a 10-day duration. Follow-up investigations one month later showed normalisation of haemoglobin level to 13.2 g/dL, platelets 255 × 109/L, CRP 5 mg/L and negative smears for malaria.
Fig. 5.
Blood film for malaria showing high degree PF parasitaemia.
Fig. 6.
PA CXR 3 days after admission.
2. Discussion
Initially, these patients were clinically suspected to have viral pneumonia, since all of the physicians considered the possibility of the patients being infected with MERS-CoV. This is largely because widespread circulation of MERS-CoV remains active throughout the Arabian Peninsula, primarily in Saudi Arabia, where the majority of cases (>85%) have been reported since 2012 [5]. However, the symptoms of malaria overlap considerably with those of MERS-CoV infection, and consequently, malaria can be misdiagnosed, especially if a patient's proper travel record is not obtained, which was the case in our study. A Canadian study showed that 59% of malaria cases were initially misdiagnosed in travellers. Of note, in 16% of the cases, three or more physician visits occurred before blood smears were ordered and malaria diagnosed [6], [7]. This underscores the importance of emergency physicians enhancing their capacity to detect P. falciparum malaria by obtaining a thorough travel history. In a non-immune patient, P. falciparum malaria can progress to life-threatening disease, with 20%–35% of the cases resulting in mortality even with adequate treatment [8]. Further, the three patients had thrombocytopenia, which may be present in 72%–84.9% of malaria-infected patients [9]. Therefore, the presence of thrombocytopenia should alert physicians to pursue a diagnosis of malaria.
In our patients, the onset of pulmonary symptoms was unpredictable. While the first patient presented with respiratory failure, the second developed symptoms three days following treatment. Typically, patients present with acute breathlessness at disease presentation or more interestingly, following therapy [10], [11]. In many instances, ARDS commences one to five days after treatment begins, when peripheral parasitaemia has decreased or nearly disappeared.
Similar to previous studies, our patients' levels of parasitaemia was inconsistent. While the third patient had a parasitaemia level greater than 5%, the other two patients had low or undetectable malaria parasites. ARDS can develop after successful treatment and parasite clearance. In these cases, ARDS may reflect the presence of inflammatory cytokines in the absence of infected erythrocytes. Emerging evidence suggests that even after treatment, free parasite antigens may persist, which could be stimuli for inflammation [12]. Moreover, this new evidence indicates that microvascular disease caused by parasite cytoadherence to endothelial cells or rosetting with uninfected erythrocytes is not the only explanation for ARDS. In addition, severe disease and ARDS are increasingly reported in Plasmodium vivax malaria, in which cytoadherence is not typically seen.
Although macrovascular disease with hypovolaemia may affect the disease outcome, liberal fluid replacement adversely affects pulmonary circulation. Adults with SFM have a generalised increase in vascular permeability [13], and so fluid loading may lead to complications and pulmonary oedema. Fluid boluses significantly increased 48-h mortality in critically ill children with impaired perfusion [14]. Thus, to correct hypovolaemia in patients with severe malaria, the consensus upholds the old malariologist's adage to “run them dry” [1]. In addition to overload, ARDS is often associated with hypoalbuminaemia [15]. All our patients with ARDS displayed hypoalbuminaemia with albumin levels ranging between 25 and 28 g/L.
Patient 1 had a protracted course with the chest CT scan showing extensive pulmonary infiltrates. The lung histopathology findings were interesting; even though the features were more consistent with the fibrotic phase of ARDS, cryptogenic organising pneumonia (COP) could not be excluded. COP has rarely been reported in malaria patients, predominantly appearing in vivax malaria and quinine-treated patients [16], [17]. He responded to a pulse methylprednisolone therapy with improved oxygenation and regression of pulmonary infiltrates. Corticosteroids have no proven benefits and may be harmful in severe malaria; however, the use of corticosteroids in unresolving ARDS (>7 days) remains controversial [18]. COP may resolve spontaneously; albeit, corticosteroids are the present established therapy [19].
Currently, the only effective therapeutic measure to decrease mortality in severe ARDS is low tidal volume and prone mechanical ventilation [20]. In a systematic review of the literature for the use of non-invasive ventilation (NIV) in cases of acute lung injury/ARDS related to P. vivax, Agarwal et al. found it to be associated with a good outcome [21]. If confirmed in further studies, NIV will become more accessible to district hospitals with limited facilities.
In conclusion, pulmonary complications of malaria can be misdiagnosed as acute respiratory illnesses, especially if cases develop in non-endemic areas and a patient's thorough travel history is not taken. This has been noted to be more prevalent in the background of national or international emerging diseases. The early recognition and effective intensive care support can greatly reduce mortality and improve outcomes.
Funding source
The author(s) received no financial support for the research, authorship and/or publication of this article.
Financial disclosure
The authors have no financial relationships relevant to this case report to disclose.
Conflicts of interest
None.
References
- 1.Anstey N.M., Jacups S.P., Cain T., Pearson T., Ziesing P.J., Fisher D.A. Pulmonary manifestations of uncomplicated falciparum and vivax malaria: cough, small airways obstruction, impaired gas transfer, and increased pulmonary phagocytic activity. J. Infect. Dis. 2002:1326–1334. doi: 10.1086/339885. [DOI] [PubMed] [Google Scholar]
- 2.Rahman M., HR, AS Quinine-induced bronchiolitis obliterans and organizing pneumonia. J. Anaesthesiol. Clin. Pharmacol. 2012 Oct;28(4):505. doi: 10.4103/0970-9185.101942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kim W.-Y., H S.B. Sepsis and acute respiratory distress syndrome: recent update. Tuberc. Respir. Dis. 2016:53–57. doi: 10.4046/trd.2016.79.2.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Agarwal R., Nath A., Gupta D. Noninvasive ventilation in Plasmodium vivax related ALI/ARDS. Intern. Med. 2007:2007–2012. doi: 10.2169/internalmedicine.46.0401. [DOI] [PubMed] [Google Scholar]
- 5.Alves C., Chen J.T., Patel N., Abrams D., Figueiredo P., Santos L., Sarmento A., Paiva J.A., Bacchetta M., Wilgus M.L., Roncon-Albuquerque R. Extracorporeal membrane oxygenation fo refractory acute respiratory distress syndrome in severe malaria. Malar. J. 2013:360. doi: 10.1186/1475-2875-12-306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Davis T.M., Suputtamongkol Y., Spencer J.L., Ford S., Chienkul N., Schulenburg Measures of capillary permeability in acute falciparum malaria: relation to severity of infection and treatment. Clin. Infect. Dis. 1992:256–266. doi: 10.1093/clinids/15.2.256. [DOI] [PubMed] [Google Scholar]
- 7.Dshmukh S., Singh V.B., Meena B., Saini V.K., Beniwal S., Thaned A.P. Vivax malaria causing BOOP: a rare complication. Int. J. Med. Res. Rev. 2015 May;3(04) 31. [Google Scholar]
- 8.Hanson J., Anstey N.M., Bihari D., White N.J., Day N.P., Dondorp A.M. The fluid management of adults with severe malaria. Crit. Care. 2014:1. doi: 10.1186/s13054-014-0642-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maitland K., Kiguli S., Opoka R.O., Engoru C., Olupot-Olupot P., Akech S.O., Nyeko R., Mtove G., Reyburn H., Lang T., Brent B. Mortality after fluid bolus in African children with severe infection. N. Engl. J. Med. 2011:2483–2495. doi: 10.1056/NEJMoa1101549. [DOI] [PubMed] [Google Scholar]
- 10.Kotepui M., Phunphuech B., Phiwklam N., Chupeerach C., Duangmano S. Effect of malarial infection on haematological parameters in population near Thailand-Myanmar border. Malar. J. 2014:218. doi: 10.1186/1475-2875-13-218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Marks M.1, Gupta-Wright A.1, Doherty J.F.2, Singer M.3, Walker D.4. Managing malaria in the intensive care unit. Br. J. Anaesth. 2014:910–921. doi: 10.1093/bja/aeu157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Y Koh. Update in acute respiratory distress syndrome. J. Intensive Care. 2014:1. doi: 10.1186/2052-0492-2-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Van den Steen P.E., Geurts N., Deroost K., Van Aelst I., Verhenne S., Heremans H., Van Damme J., Opdenakker G. Immunopathology and dexamethasone therapy in a new model for malaria-associated acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2010:957–968. doi: 10.1164/rccm.200905-0786OC. [DOI] [PubMed] [Google Scholar]
- 14.Goldman-Yassen Adam E., Mony Vidya K., Arguin Paul M., Daily Johanna P. Higher rates of misdiagnosis in pediatric patients versus adults hospitalized with imported malaria. Pediatr. Emerg. Care. 2016:227–231. doi: 10.1097/PEC.0000000000000251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cheng M.P., Yansouni C.P. Management of severe malaria in the intensive care unit. Crit. Care Clin. 2013:865–885. doi: 10.1016/j.ccc.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 16.Taylor W.R., Canon V., White N.J. Pulmonary manifestations of malaria : recognition and management. Treat. Respir. Med. 2006:419–428. doi: 10.2165/00151829-200605060-00007. [DOI] [PubMed] [Google Scholar]
- 17.Taylor W.R., Hanson J., Turner G.D., White N.J. Dondorp AM.l. Respiratory manifestations of malaria. CHEST J. 2012:492–505. doi: 10.1378/chest.11-2655. [DOI] [PubMed] [Google Scholar]
- 18.Sara A.-G., Hamdan A.-J., Hanaa B., Nawaz K.A. Bronchiolitis obliterans organizing pneumonia: pathogenesis, clinical features, imaging and therapy review. Ann. Thorac. Med. 2008:67–75. doi: 10.4103/1817-1737.39641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.organisation wh. world health organization. [Online].; 2015 [cited 2016 september 3. Available from: www.who.int/emergencies/mers-cov/en/.
- 20.Kain K.C., Harrington M.A., Tennyson S., Keystone J.S. Imported malaria: prospective analysis of problems in diagnosis and management. Clin. Infect. Dis. 1998:142–149. doi: 10.1086/514616. [DOI] [PubMed] [Google Scholar]
- 21.Antonelli M., Bonten M., Cecconi M., Chastre J., Citerio G., Conti G., Curtis J.R., Hedenstierna G., Joannidis M., Macrae D., Maggiore S.M. Year in review in Intensive Care Medicine 2012: I. Neurology and neurointensive care, epidemiology and nephrology, biomarkers and inflammation, nutrition, experimentals. Intensive Care Med. 2013 Feb 1;39(2):232–246. doi: 10.1007/s00134-012-2774-y. [DOI] [PMC free article] [PubMed] [Google Scholar]