Abstract
Purpose
The persistence of leukemia stem cells (LSC)-containing cells after induction therapy may contribute to minimal residual disease (MRD) and relapse in acute myeloid leukemia (AML). We investigated the clinical relevance of CD34+CD123+ LSC-containing cells and anti-leukemia potency of a novel antibody-conjugate SL-101 in targeting CD123+ LSCs.
Experimental Methods and Results
In a retrospective study on 86 newly diagnosed AML patients, we demonstrated that a higher proportion of CD34+CD123+ LSC-containing cells in remission was associated with persistent MRD, and predicted shorter relapse-free survival in patients with poor-risk cytogenetics. Using flow cytometry, we explored the potential benefit of therapeutic targeting of CD34+CD38−CD123+ cells by SL-101, a novel antibody-conjugate comprising an anti-CD123 single-chain Fv fused to Pseudomonas exotoxin A. The anti-leukemia potency of SL-101 was determined by the expression levels of CD123 antigen in a panel of AML cell lines. Colony-forming assay established that SL-101 strongly and selectively suppressed the function of leukemic progenitors while sparing normal counterparts. The internalization, protein synthesis inhibition and flow cytometry assays revealed the mechanisms underlying the cytotoxic activities of SL-101 involved rapid and efficient internalization of antibody, sustained inhibition of protein synthesis, induction of apoptosis, and blockade of IL-3–induced p-STAT5 and p-AKT signaling pathways. In a patient-derived xenograft model using NSG mice, the repopulating capacity of LSCs pretreated with SL-101 in vitro was significantly impaired.
Conclusions
Our data define the mechanisms by which SL-101 targets AML and warrant further investigation of the clinical application of SL-101 and other CD123-targeting strategies in AML.
Keywords: acute myeloid leukemia, CD123, leukemia stem cell, SL-101, minimal residual disease
Introduction
Acute myeloid leukemia (AML) can be considered as a group of genetically and clinically heterogeneous neoplasms. Despite advances in the treatment of AML, the clinical outcomes have improved only minimally over the past three decades, especially among elderly patients, who have a long-term disease-free survival rate of less than 10% and an average overall survival duration of less than 1 year (1). Therefore the development of novel therapeutic agents to improve outcome is warranted. An emerging focus is the potential of targeting the leukemic stem cells (LSCs) that can regenerate AML in mouse xenotransplantation models, and are believed to initiate and maintain AML in patients. In their quiescent state, LSCs may contribute to AML resistance to conventional chemotherapy (2–4). The persistence of LSCs has been postulated to contribute towards minimal residual disease (MRD) and relapse (5).
Assessment of MRD improves stratification of patient prognostic profiles and selection of patients for directed treatment in AML (6–8). However, about 20% of AML patients experience relapse despite having no detectable MRD after induction. Data on the association between putative LSCs and pathologic detection of MRD are lacking. A higher proportion of CD34+CD38− stem cells was associated with an increased frequency of MRD and poorer survival (9). The Lin−CD34+CD45RA+CD123+ LSCs were persistent in remission in patients with resistant AML (10). The persistence of residual LSCs may explain why patients with no detectable MRD experiences relapse. These residual LSCs may not only have prognostic value but also may allow identification of patients who may benefit from targeted therapeutics.
The plasma membrane molecules preferentially expressed in subsets of AML would be ideal LSC targets. In many AML cases, LSC capable of xenotransplantation are enriched among CD34+CD38− cells, which lack lineage markers (11). Interleukin (IL)-3 receptor alpha (IL-3Rα, CD123) has been identified to be highly expressed on AML blasts and stem cells in most cases, but expressed at lower levels on normal hematopoietic stem cells and other more mature CD34+ subsets including common myeloid precursors and granulocyte-macrophage precursors (12–15). The IL-3R beta subunit (βc, CD131) is shared by receptors for IL-3, granulocyte-macrophage colony-stimulating factor (GM-CSF) and IL-5 (16, 17). Preclinical data demonstrated that targeting CD123 eliminates AML stem cells, indicating that CD123 plays a pivotal role in repopulating AML (18).
In the past decade, monoclonal antibody (mAb) therapy targeting cells with specific antigens has been proven feasible. However, unconjugated mAbs show limited efficacy and can rarely induce durable remission (19, 20). Delivery of highly cytotoxic substances via the binding domains of mAbs would facilitate killing activity specific to the cells that express the cognate antigens. Because of the potency of their enzymatic domains, immunotoxin conjugates are promising anticancer therapeutics which can elicit high cytotoxic activity against tumor cells (21). Several such conjugates, utilizing Pseudomonas exotoxin A (PE), an enzymatic inhibitor of protein synthesis, have been developed for treating leukemia (21). As an example, a recombinant immunotoxin BL22, a fusion of a 38-kd fragment of PE38 to the variable region (Fv) of the anti-CD22 antibody (22, 23), induces high complete remission (CR) rates in hairy cell leukemia and other CD22-positive leukemias (22–24).
Immunotoxin conjugate potency is dependent on sufficient expression and efficient internalization of the target cell-surface antigen. CD123 is highly expressed in the majority of AML cases, making it an excellent target for directed immunotoxin–mAb treatment (18, 25). SL-101 is a novel anti-CD123 antibody-conjugate comprising the anti-CD123 single-chain Fv (scFv) fused to a truncated and optimized PE38 lacking its native targeting domain but containing its translocation and ADP-ribosylation domains (26). It is a high-affinity antibody that reacts with the alpha subunit of IL-3R without depending on the presence of the beta subunit (26).
In this study, we aimed to determine whether the presence of CD34+CD123+ LSC-containing cells is associated with MRD and relapse risk in high-risk AML patients and tested the binding and internalization of the anti-CD123 conjugate in leukemia cells and evaluated its cytotoxicity against AML cell lines and primary AML, especially the primitive stem/progenitor cells, both in vitro and in vivo. Our data indicate that SL-101 is a promising anti-leukemia agent in CD123-expressing AML.
Methods
Patients
This retrospective portion of the study was approved by the Institutional Review Board (IRB) of The University of Texas MD Anderson Cancer Center (MDACC). From July 2011 to November 2012, 153 patients with newly diagnosed AML were treated with either high-dose cytarabine based regimens or low-dose regimens including low-dose cytarabine or hypomethylating agents. Bone marrow samples from the patients at diagnosis and in remission were analyzed at the time of collection by multicolor flow cytometry in the Department of Hematopathology at MDACC, by comparing with the known patterns of antigen expression by normal maturing myeloid precursors and monocytes. A distinct cluster of at least 20 cells showing altered expression of at least two antigens was regarded as an aberrant population, yielding an optimal sensitivity of 1 in 104 cells, or 0.01% (8, 27).
Construction and purification of SL-101
SL-101 [26292(Fv)-PE38-KDEL] was provided by Stemline Therapeutics, Inc (New York, NY). The conjugate was constructed by replacing the PE38 portion of the expression plasmid with PE38 containing KDEL at the C-terminus scFv-PE38, and was expressed and purified as described elsewhere (26).
Human AML cell lines
TF-1/HRAS was provided by Stemline Therapeutics, Inc.; K562GFP-CD123 by Dr. JF DiPersio (Washington University) and generated as previously described (28); and Mo7e by Dr. Z Estrov (MDACC). The other AML cell lines were purchased from the American Type Culture Collection or Deutsche Sammlung von Mikroorganismen und Zellkulturen. All cells were routinely propagated in RPMI-1640 medium (Cat. 10-040-CV; Mediatech, Inc. Manassas, VA) containing 10% fetal bovine serum (FBS, Cat. F4135; Sigma-Aldrich) and 1% each penicillin and streptomycin. For TF-1 and Mo7e cells, GM-CSF (cat. NDC 58406-002-01; Immunex Corporation, Seattle, WA) was added to the medium (2 ng/mL for TF-1, and 20 ng/mL, respectively).
Apoptosis of AML cell lines and primary AML stem/progenitor cells
Peripheral blood and bone marrow (BM) samples were obtained from patients with AML or healthy donors after informed consent was obtained in accordance with MDACC IRB regulations (under protocol LAB08-0740). AML cells were treated with SL-101 at indicated concentrations and incubated for 48 hours in RPMI-1640 medium supplemented with 10% FBS. Apoptosis in AML cell lines was detected by flow cytometry using Annexin-V–APC (Cat. 550475; BD Biosciences, San Jose, CA) and 4′6-diamidino-2-phenylindole (DAPI). Viable AML cells were enumerated by using CountBright counting beads (Cat. C36950; Invitrogen, Carlsbad, CA) with concurrent Annexin-V and DAPI detection on a Gallios Flow Cytometer (Beckman Coulter, Indianapolis, IN). Data were analyzed by using Flowjo software (Tree Star, Ashland, OR). IC50 values were calculated by using Calcusyn software (Biosoft, Cambridge, MA) based on the number of Annexin-V−/DAPI− viable cells.
MNCs isolated from primary AML specimens were stained with a cocktail of antibodies comprising CD34-FITC (Cat. 555821), CD38-PE-Cy7 (Cat. 335790), CD45-APC-Cy7 (Cat. 557833), CD123-PerCP-Cy5.5 (Cat. 558714; all from BD Biosciences), and Annexin-V–APC for 30 minutes at room temperature in dark. The cells were then washed, resuspended in PBS with DAPI, and analyzed on a Gallios Flow Cytometer. Results are presented as percentage of specific apoptosis: 100 × (% apoptosis of treated cells − % apoptosis of control cells)/(100 − % apoptosis of control cells). IC50 values were calculated by using Calcusyn software based on the number of CD45dim/Annexin-V−/DAPI− viable cells.
Colony-forming cell assay
MNCs isolated from patients with AML (100,000) or healthy donors (50,000) were plated in methylcellulose medium (1 mL/well; Cat. 04435; STEMCELL Technologies Inc., Vancouver, BC, Canada) in triplicate per condition. Colonies were scored after 2 weeks of culture.
Internalization assays
Cells were incubated with SL-101–DyLight 680 (1×106/mL) for 30 minutes on ice to facilitate binding, followed by incubation at 37°C for the indicated time points. Cells were then washed with PBS and the membrane-bound SL-101 was stripped by suspending the cells in ice-cold glycine (0.2 M, pH 2.5; 500 μL) supplemented with BSA (1 mg/mL) on ice for 10 minutes. The reaction was quenched with Tris-HCL (1 M, pH 8.0; 1 mL). Cells were washed and resuspended in 1% BSA/PBS supplemented with DAPI.
Protein synthesis inhibition assay
To detect the inhibitory effects of SL-101 on protein synthesis, L-azidohomoalanine (AHA) reaction cocktails were applied to detect AHA signaling according to the manufacturer’s protocol and as published elsewhere (29). Briefly, MV4-11 and MOLM13 cells were treated with SL-101 (1 μg/mL) in a 96-well plate for 24 hours or with cycloheximide (100 μM) as a positive control. After treatment, the drug-containing medium was removed, and L-methionine–free medium containing AHA was added to the cell cultures. After incubation for 30 minutes, cells were washed, fixed with 3.7% formaldehyde, and permeabilized with 0.5% Triton X-100 (Cat. 28314; Thermo Fisher Scientific) for 20 min. The AHA reaction cocktails were added to detect the AHA signal that was measured by a fluorescence microplate reader (485/520 nm) and the percentages of inhibition were calculated compared with the control group.
Surface and intracellular flow cytometry staining
The AML cells were stained with CD123 and CD131-PE (Cat. 306104; BioLegend, San Diego, CA) for 30 minutes at room temperature in dark and then washed and resuspended in PBS with DAPI before analysis on a Gallios Flow Cytometer.
For intracellular staining, Mo7e cells were starved overnight at 1 million cells/mL in a 37°C incubator in RPMI-1640 medium containing 1% FBS and penicillin/streptomycin and supplemented with SL-101 (1 μg/mL). Cells were then stimulated with IL-3 (100 ng/mL; Cat. 200-03; Peprotech, Rocky Hill, NJ) for 10 minutes and fixed with paraformaldehyde (Cat. 15710; Electron Microscopy Sciences, Hatfield, PA) at a final concentration of 1.6% at room temperature for 10 minutes. Cells were spun down, washed twice with wash buffer comprising PBS supplemented with 0.5% bovine serum albumin (BSA; Cat. A7906; Sigma-Aldrich), and permeabilized by suspending in 1 mL 80% cold methanol (Cat. 67-56-1; Thermo Fisher Scientific, Inc., Waltham, MA) overnight at −20°C. Cells were washed twice with wash buffer and stained with p-STAT5-Alexa Fluor 647 (1:10, Cat. 612599; BD Biosciences), p-ERK1/2-Alexa Fluor 488 (3:100, Cat. 4374), or pAKT-Alexa Fluor 647 (3:100, Cat. 4075; both from Cell Signaling Technology, Beverly, MA) in 100-μL final reaction volumes for 30 minutes at room temperature in darkness. After being washed twice, cells were analyzed on a Gallios Flow Cytometer. Data were analyzed using Flowjo software.
In vivo study of SL-101 efficacy in AML patient-derived xenograft (PDX) mouse models
The animal studies were performed in accordance with guidelines approved by the Institutional Animal Care and Use Committee at MDACC. Primary AML peripheral blood cells were left untreated or pretreated with SL-101 (1.0 μg/mL) and incubated in RPMI-1640 medium supplemented with 10% FBS overnight at 37°C. Cells were washed and resuspended in PBS before transplantation into nonobese diabetic/severe combined immunodeficient gamma (NSG) mice. Ten-week old female NSG mice (17.1–22.7g) were purchased from Jackson lab and sublethally irradiated (250 cGy) 1 day before intravenous injection of primary AML cells (0.9 × 106 viable cells per mouse). Engraftment (>1% human CD45) was confirmed in peripheral blood samples by flow cytometry. Leukemia burden was monitored weekly by flow cytometry using anti-human CD45 and CD123 antibodies. The mice were sacrificed when they became moribund, and the spleen and BM were harvested to determine the extent of engraftment and to identify LSC phenotypes, defined as human CD45dimSSClowCD34+CD38−CD123+, using the antibodies CD45-APC-Cy7, CD38-PE-Cy7, CD123-PerCP-Cy5.5, CD34-APC (Cat. 555824; all from BD Biosciences). Additional PDX study was performed using AML11 injected into NSG-SGM3 (NSGS) mice (9–11-week old) breed in house (0.6 × 106 viable cells per mouse). Once human CD45 engraftment was confirmed by flow cytometry, mice were treated intravenously with PBS or SL-101 at 0.1 mg/kg every other day for 6 doses.
Statistical analyses
A univariate logistic regression model was used to analyze the association between MRD negativity and factors potentially predicting relapse. Relapse-free survival (RFS) was measured from the date of CR until relapse. Patients without relapse at their last follow-up monitoring were censored on that date. The Kaplan-Meier method was used to estimate the median RFS. Univariate Cox proportional hazards regression was performed to identify the association between each of the variables and RFS. Multivariate Cox proportional hazards regression was used to model all the variables in the univariate setting. The backward selection method was used to remove variables that did not remain significant in the multivariate model (p=0.15). Hazard ratios (HRs) were generated with 95% confidence intervals (95% CI). Data were analyzed with STATA/SE version 14.1 statistical software (Stata Corp. LP, College Station, TX). The Student t-test was used to analyze the statistical significance of differences between groups, both in vitro and in vivo. All statistical tests were two-sided, and the results are expressed as the mean ± standard deviation. A p value ≤ 0.05 was considered statistically significant.
Results
Residual CD34+CD123+ LSC-containing blasts associate with MRD and relapse in high-risk AML patients
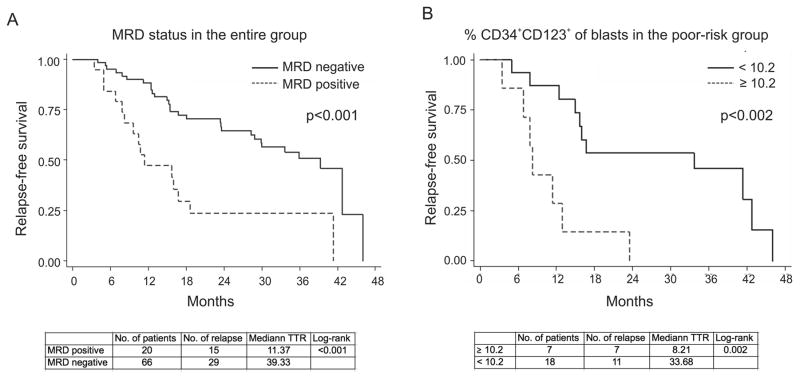
From July 2011 to November 2012, 106 out of 153 patients (69.3%) with newly diagnosed AML achieved complete remission and 86 patients had flow cytometry data on BM specimens at diagnosis and in the first remission. To determine the association between CD34+CD123+ blasts, MRD, and relapse in AML, we re-analyzed flow cytometric data collected on BM specimens at diagnosis and at the time of achieving response from 86 patients. The clinical characteristics are shown in Table S1. The MRD antibody panels containing CD34 and CD123 were presented in Table S2 and the gating strategy was shown in Figure S1. CD34+CD123+ cells accounted for 41.0±33.3% of CD45dim blasts at diagnosis and 10.5±13.1% in remission (Table S3). Percentages of CD34+ and CD34+CD123+ cells within blast gate at diagnosis were significantly higher in patients with poor-risk cytogenetics, wild-type FLT3 and wild-type NPM1 (Table 1).
Table 1.
Association between proportions of CD34+, CD123+, and CD34+/CD123+ cells and cytogenetics and gene mutations
| Cytogenetics
|
P value | ||
|---|---|---|---|
| Fav/Int | Poor | ||
| CD34+, % | 40.5±36.7 | 62.3±32 | 0.011 |
| CD34+CD123+, % | 34.6±33 | 56.5±29 | 0.004 |
| CD34+CD123+%/CD34+% | 0.8±0.3 | 0.9±0.1 | 0.020 |
|
| |||
|
FLT3
|
P value | ||
| WT | ITD | ||
|
| |||
| CD34+, % | 53.3±36.1 | 26.7±30.7 | 0.011 |
| CD34+CD123+, % | 46.3±33.2 | 23.4±26.9 | 0.004 |
|
| |||
|
NPM1
|
P value | ||
| WT | Mutant | ||
|
| |||
| CD34+, % | 60.9±31.6 | 9.7±15.1 | <0.001 |
| CD34+CD123+, % | 53.1±29.8 | 8.9±14.5 | <0.001 |
Fav, favorable; Int, intermediate; WT, wild type; ITD, internal tandem duplication
Sixty-six patients (77.9%) in this cohort were negative for MRD determined by flow cytometry. To determine whether CD123 expression is associated with persistent MRD, we performed logistic regression models. None of the CD123/CD34 values at diagnosis correlated with MRD. However, persistence of a higher fraction of CD123-expressing cells within CD34 progenitor cells in remission, represented by a ratio of CD34+CD123+/CD34+ greater than 0.7, was significantly associated with MRD positivity (p=0.035, Table S4). As expected, patients with poor-risk cytogenetics, and those treated with low dose (LD) regimens, were at higher risk for MRD positivity than those with favorable- or intermediate-risk cytogenetics and those treated with high-dose (HD) regimens (p=0.022 and 0.004, respectively, Table S4).
To determine associations between the CD123+ blasts and rate of relapse, we performed Cox proportional hazard models. Forty-four patients (51.2%) experienced relapse. In the univariate analysis, cytogenetics (poor vs. others, p=0.06), regimen (HD vs LD, p=0.003), and achievement of negative MRD (p=0.001, Figure 1A) were factors predictive for a better outcome for relapse-free survival. Multivariate analyses show that cytogenetics (poor vs others, p=0.15) has a borderline significant effect; regimen [HD vs LD, p=0.03] and MRD status (p=0.03) were significant predictors of RFS (Table S5A). Since CD34+ and CD34+CD123+ expression levels are highly correlated, if they were included in the same multivariate model, they may cause a problem of collinearity and give false-positive or -negative results. Therefore, they were analyzed by two separate models in Tables S5A&B. These results show that neither CD34+ nor CD34+CD123+ cell frequencies predicted relapse in the entire patient cohort. However, in patients with poor-risk cytogenetics (n=25), a higher percentage of CD34+CD123+ cells in remission significantly correlated with occurrence of relapse in both univariate (HR: 1.05; 95% CI: 1.02–1.10; p=0.005) and multivariate analysis (HR: 1.08, 95% CI: 1.02–1.15; p=0.013). Using Martingale residual analysis, a cut-off of 10.2% of CD34+CD123+ cells of blasts was selected, and patients with more than 10.2% of CD34+CD123+ in remission were more likely to undergo relapse (p<0.002, Fig. 1B). These findings support the utility of CD123 expressed on residual progenitor cells persisting after completion of induction chemotherapy, as a potential therapeutic target in AML.
Figure 1. MRD status and proportion of CD34+CD123+ blasts in remission are associated with relapse-free survival.
RFS as analyzed by the Cox proportional hazards regression model was significantly reduced by the presence of MRD (all patients, A) and higher percentage of CD34+CD123+ cells as a percentage of blasts in remission in the poor-risk cytogenetics patients (B). TTR, time to relapse (months).
Cytotoxicity of SL-101 against myeloid leukemia cell lines in vitro
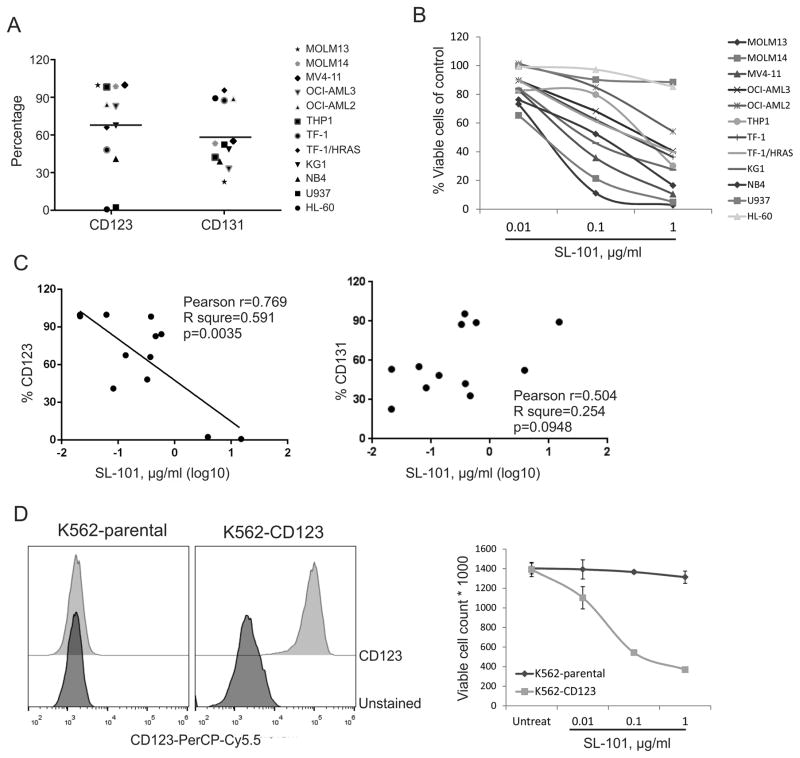
The high binding ability, selectivity, and stability of SL-101 in the CD123-expressing TF-1 AML cell line were previously reported.(26) To assess the anti-leukemia activity of SL-101, we studied its effects on cell proliferation in 12 myeloid leukemia cell lines (Table S6). Cells expressed various levels of CD123 (median 75.2%, range 0.9%–99.9%) and CD131 (median 52.8%, range 22.7%–95.7%) (Fig. 2A). SL-101 at tested doses was active against 10 AML cell lines after treatment for 48 hours, with a median IC50 of 0.232 μg/mL (range 0.021–0.584 μg/mL) (Fig. 2B). CD123-negative U937 and HL-60 cells were resistant to SL-101. Notably, SL-101 was highly active against all three cell lines harboring FLT3-ITD mutations (median IC50, 0.021 μg/mL). The anti-leukemia activity of SL-101 positively correlated with expression levels of CD123 (Pearson r = 0.769, p=0.0035) but not IL-3R beta subunit CD131 (Fig. 2C). To further assess the specificity of SL-101, we tested the cytotoxicity of SL-101 on CD123-overexpressing K562 leukemia cells (K562GFP-CD123). While SL-101 had no activity against parental CD123− K562 cells, it potently inhibited cellular growth of K562GFP-CD123 cells, indicating that the cytotoxicity of SL-101 is dependent on the expression level of CD123 (Fig. 2D).
Figure 2. Cytotoxicity of SL-101 against myeloid leukemia cell lines in vitro.
(A) Percentages of CD123+ and CD131+ cells are shown for myeloid leukemia cell lines. (B) Annexin-V−/DAPI− viable cells were measured by flow cytometry after treatment with SL-101 at indicated doses for 48 hours. The cell counts were normalized to those of untreated samples. (C) Correlation analysis was performed between expression of IL-3 receptor subunits and IC50 values of SL-101 calculated by using Calcusyn software. (D) Expression of CD123 was measured in parental and CD123-overexpressing (K562CG-CD123) K562 cells. Cells were left untreated or exposed to indicated concentrations of SL-101 for 48 hours, and viable cell counts were determined as described in (B).
SL-101 efficiently kills primary AML blasts and LSCs
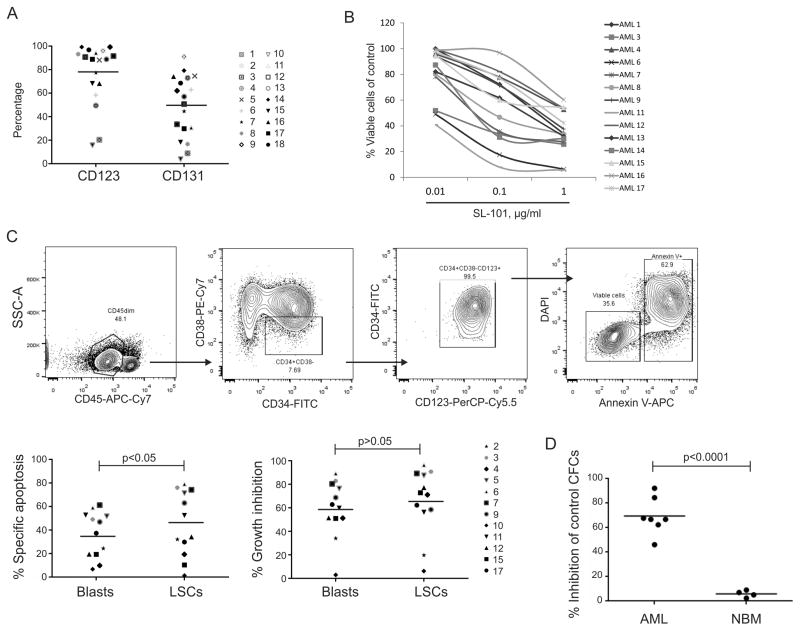
We next tested SL-101 in 17 genetically diverse primary AML samples (Table S7). Most samples expressed high levels of CD123 (median 88.9%, range 20.4–99.3%) and intermediate levels of CD131 (median 54.0%, range 9.0–91.1%, Fig. 3A). AML blasts from patient peripheral blood were exposed to SL-101 for 48 hours. SL-101 was highly active against these AML samples, with a median IC50 of 0.33 μg/mL (range 0.002–0.98 μg/mL, Table S7), and demonstrated cell-killing activity in a dose-dependent manner in 14 of the 17 AML samples (Fig. 3B). No significant correlation was found between SL-101 activity and expression levels of CD123 or CD131 (Figure S2).
Figure 3. SL-101 efficiently kills primary AML blasts and stem/progenitor cells.
(A) Percentages of CD123+ and CD131+ fractions within CD45dim blast gate on primary AML samples are shown. (B) Samples were treated with SL-101 at indicated doses for 48 hours. Normalized viable cell counts (left) were determined in a cohort of primary AML samples. (C) Gating scheme for the LSC population (CD45dimCD34+CD38−CD123+). Specific apoptosis was calculated based on percentage of Annexin-V+ cells using the following formula: percentage of specific apoptosis = 100 × (% apoptosis of treated cells − % apoptosis of control cells)/(100 − % apoptosis of control cells). Percentage of growth inhibition was calculated based on Annexin-V−/DAPI− viable cells using the following formula: 100 – 100 × % viable cells of treated cells/% of viable cells of control cells in both CD45dim blasts and LSC-enriched blasts. (D) Colony-forming cell assay was performed in primary AML samples and normal bone marrow (NBM) cells. Data were normalized to those from untreated control groups and represent the mean values of triplicate assays. The absolute colony numbers were shown in Supplementary Figure 1.
To test whether SL-101 is capable of inducing cell death in the LSC-enriched (LSCe) subset with a CD34+CD38−CD123+ immunophenotype (13), 12 AML samples were treated with SL-101 (1.0 μg/mL) for 48 hours, and apoptosis induction was determined in both AML blasts (CD45dimSSClow) and LSCe cells by flow cytometry (Fig. 3C). SL-101 induced apoptotic cell death in both bulk AML (36.5% ± 19.6%) and LSCe cells (45.3% ± 27.5%, n=12; p<0.05). Quantification of the viable (AnnexinV−/DAPI−) cells using counting beads also demonstrated further reduction of cell numbers by SL-101 (54.1% ± 29.2% in blasts and 60.6% ± 33.7% in LSCe cells), indicating additional mechanisms of cell growth inhibition (Fig. 3C and Figure S3). To assess the effect of SL-101 on function of leukemia progenitors, we performed a colony-forming assay with primary AML and normal BM samples. SL-101 selectively and significantly suppressed AML colony formation (69.5% ± 15.0% inhibition of total colonies, n=7) while sparing normal BM (5.6% ± 3.3% inhibition, n=4; p<0.0001) (Fig. 3D and Figure S4).
Mechanisms of the cytotoxic activity of SL-101
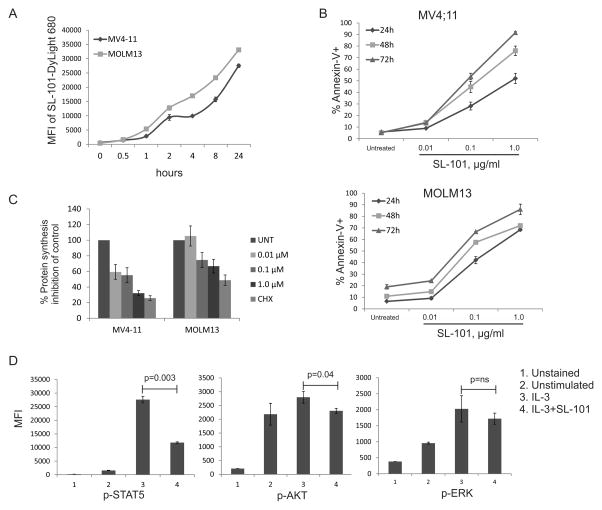
It was recently demonstrated that, upon internalization, PE traffics to the cytosol to inactivate protein synthesis by catalyzing ADP ribosylation of elongation factor 2, causing growth inhibition and apoptotic cell death.(21, 30) To function as a cytotoxic agent, SL-101 must be efficiently internalized upon binding CD123. We first studied the internalization of SL-101 by exposing CD123-expressing AML cell lines to DyLight 680-labeled SL-101 at different time points and analyzing DyLight 680 signal by flow cytometry. The intracellular median intensity of DyLight 680 signal increased after 1 hour in both MV4-11 and MOLM13 cells, demonstrating rapid and efficient cellular uptake of SL-101 (Fig. 4A). The signal did not reach a plateau even after 24 hours (data not shown), indicating a continuous recruitment and internalization of SL-101 over time.
Figure 4. Mechanisms of the cytotoxic activity of SL-101.
(A) MOLM13 and MV4-11 cells were incubated (1 million/mL) with SL-101–DyLight 680 overnight, and internalized SL-101 was detected by flow cytometry on DyLight 680 signal after removing the membrane-bound SL-101. MFI, mean fluorescence intensity. (B) MOLM13 and MV4-11 cells were left untreated (untreat, or UNT) or treated with SL-101 at indicated concentrations for 24, 48, or 72 hours. Annexin-V+ apoptotic cells are shown. (C) MV4-11 and MOLM13 cells were treated with SL-101 (1 μg/mL) for 24 hours. Cycloheximide (100 μM) was used as a positive control. AHA (L-azidohomoalanine) reaction cocktails were applied to detect active protein synthesis. The signal was measured by a fluorescence microplate reader (485/520 nm) and the inhibition percentages were calculated in comparison with control group. (D) Mo7e cells (1 million/mL) were starved overnight and then stimulated with IL-3 (100 ng/mL) for 10 min. Cells were stained with antibodies after fixation and permeabilization.
Because recent studies demonstrated apoptosis induction by PE and other protein synthesis inhibitors,(31) we tested apoptosis induction by SL-101 in AML cell lines. MV4-11 and MOLM13 cells were exposed to SL-101 at various concentrations for 24, 48, or 72 hours. Apoptotic cell death was determined by Annexin-V/DAPI flow cytometry. SL-101 induced dose- and time-dependent cell killing in both cell lines (Fig. 4B). Apoptotic cell death in primary AML samples is shown in Fig. 3C.
Next, we examined the efficacy of SL-101 in inhibiting nascent protein synthesis in AML cell lines using an AHA Alexa Fluor 488 protein synthesis assay. SL-101 (1.0 μg/mL) significantly reduced protein synthesis in MV4-11 cells (67.8% ± 3.4% inhibition, p=0.001) and MOLM13 cells (33.3% ± 8.7% inhibition, p<0.01) within 24 hours, comparable with cycloheximide used as a positive control (Fig. 4C). These findings confirmed the potency of SL-101 to internalize and promote cell death through protein synthesis blockade.
To further investigate the ability of SL-101 to inhibit intracellular signaling in response to IL-3, we studied cytokine-dependent Mo7e leukemia cells which were serum starved and pretreated with SL-101 (1.0 μg/mL) overnight, followed by stimulation with IL-3. SL-101 significantly suppressed IL-3–induced activation of p-STAT5 (57.1% ± 2.6% inhibition, p=0.003) and modestly inhibited p-AKT (17.4% ± 5.4% inhibition, p=0.04) but not p-ERK signaling (Fig. 4D).
SL-101 targets AML leukemia-initiating cells
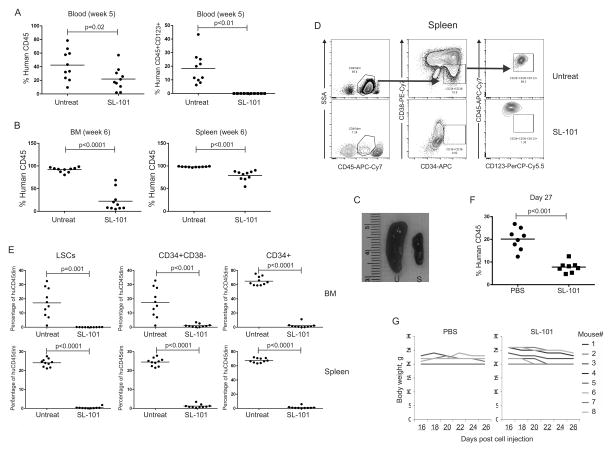
To evaluate the effect of SL-101 on leukemia-initiating cells in vivo, primary AML cells from two patients (AML11 and AML18, Table S7) were either left untreated or exposed in vitro to SL-101, followed by transplantation into NSG mice. Engraftment of human AML11 was confirmed by flow cytometry analysis of peripheral blood 3 weeks post injection. In week 5, the circulating leukemia burden was significantly reduced in the mice that received SL-101–treated cells (42.5% ± 22.4% in control versus 22.0% ± 16.6% in SL-101 group, p=0.02). Strikingly, the human CD45+CD123+ cells were undetectable at week 5 (Fig. 5A). All mice were sacrificed at week 6 to assess the leukemia burden determined by human CD45 flow cytometry in BM and spleen. Pretreatment with SL-101 significantly reduced leukemia engraftment in both BM (91.8% ± 5.5% in control group versus 22.3% ± 22.8% in SL-101 group, p<0.0001) and less so in the spleen (98.2% ± 0.9% in control versus 79.0% ± 11.0% in SL-101 group, p<0.01) (Fig. 5B). The splenomegaly observed in the control mice was largely abrogated by SL-101 treatment (Fig. 5C). We further analyzed the effects of SL-101 on AML cells highly enriched for LSCs (CD45dimSSClowCD34+CD38−CD123+). Representative flow plots are shown in Fig. 5D. Ex vivo exposure of transplanted AML cells to SL-101 reduced the fraction of AML blasts (CD45dimSSClow), CD34+ progenitor, CD34+CD38− cells, and importantly almost completely ablated CD45dimSSClowCD34+CD38−CD123+ cells in both BM (17.2% ± 10.3% in control versus 0.06% ± 0.06% in the SL-101 group, p<0.001) and spleen (24.1% ± 2.1% versus 0.37% ± 0.51%, respectively, p<0.0001) (Fig. 5E). In mice transplanted with AML18, until week 10, only three mice from each group demonstrated over 1% human CD45 engraftment (33.7% ± 18.7% in the untreated group versus 4.6% ± 1.8% in the SL-101–treated group, p>0.05, Figure S5). The same PDX model established using AML11 was applied to study the anti-leukemia efficacy of in vivo administration of SL-101. A significant decrease on leukemia burden was observed after the last dose (20.2% ± 4.8% in PBS- versus 7.8% ± 2.4% in the SL-101-treated group, p<0.001, Fig. 5F). The dose was tolerable as demonstrate by the stable body weight over treatment (Fig. 5G). These data demonstrate that treatment with SL-101 significantly impaired the stem cell–repopulating capacity of AML LSCs and reduced tumor burden in peripheral blood, BM, and spleen.
Figure 5. SL-101 targets AML leukemia-initiating cells in vivo.
The primary AML patient sample 11 (4030094) was left untreated or pre-treated with SL-101 (1.0 μg/mL) overnight prior to transplantation via intravenous injection into NSG mice (1×106 viable cells for each mouse). (A) Engraftment of human AML cells was measured by flow cytometry in blood by using anti-human CD45 antibody at week 5 after injection (left). CD123+ cells were gated on the human CD45+ fraction (right). (B) At week 6, all mice were sacrificed and engraftment of human AML was measured in both bone marrow (BM) and spleen. (C) Representative spleens from untreated (U) and SL-101-treated (S) mice are shown. (D) The gating scheme is shown for the CD45dimCD34+CD38−CD123+ LSC-enriched population in untreated and SL-101–treated mice. (E) Percentages of LSCe in human CD45+ cells were determined in both bone marrow and spleen by flow cytometry. AML11 cells (0.6×106) were injected into NSGS) mice that were treated intravenously with PBS or SL-101 at 0.1 mg/kg every other day for 6 doses, after confirming human CD45 engraftment. (F) Leukemia burden was measured after treatment (G) Body weight was monitored over treatment.
Discussion
The prognostic power of MRD in AML has been shown in several studies; moreover, at least 20-30% of AML patients eventually experience relapse despite having no detectable MRD after induction chemotherapy. LSCs, considered as the cause of relapse, are thus an excellent target for novel therapies because of their unique phenotypes. LSC persistence in remission has been documented in patients with resistant AML, by quantifying phenotypically defined LSCs (Lin−CD34+CD45RA+CD123+) over time (10). Recent studies using next-generation sequencing delineated a complex genetic landscape of leukemia and demonstrated persistence of pre-LSCs in remission, which survive chemotherapy and may contribute to relapse (32–34). In our cohort, although high-intensity regimens were used in the majority patients (67.4%), they failed to eradicate the LSC-containing CD34+CD123+ population. Higher percentages of CD34+CD123+ blasts in remission significantly correlated with poor-risk cytogenetics and wild-type NPM1. Of importance, persistence of CD34+CD123+ blasts was associated with a shorter time to relapse in poor-risk cytogenetics patients. These data provide a basis to identify patients who may benefit from CD123-targeted therapeutics.
We next demonstrated the anti-leukemia efficacy of SL-101. It showed profound cytotoxicity in a panel of AML cell lines, through rapid internalization, induction of apoptosis, inhibition of protein synthesis, and downregulation of IL-3–induced STAT5 signaling. In vitro studies in primary AML samples demonstrated cytotoxicity in both bulk and primitive cell fractions. Notably, SL-101 was highly active in three AML cell lines with FLT3-ITD mutations. Our previous studies and other reports demonstrated that CD123 was highly enriched in LSC-immunophenotypic blasts of FLT3-mutant AML (35–37), making SL-101 an attractive therapeutic agent alone or in combination with tyrosine kinase inhibitors in this poor-risk cohort of AML patients. Further, ex vivo pre-treatment of SL-101 significantly reduced human engraftment and eliminated CD34+CD38−CD123+ LSCs in an NSG mouse xenograft model. Reduced leukemia burden was observed by in vivo administration of SL-101 at a tolerable dose.
CD123 has been recognized as an LSC marker for AML over a decade (13, 17). Monoclonal antibody 7G3 against CD123 profoundly reduced CD34+CD38− cells and engraftment by inhibiting homing of human leukemia cells to BM in a xenotransplantation model (18). However, a phase I clinical study with CSL360, a 7G3-derived IgG1 recombinant chimeric monoclonal antibody, showed limited anti-leukemia activity in patients with advanced AML (20). These results suggest that blockade of target antigen function using a naked mAb might not be sufficient as a therapeutic strategy, consistent with findings from other hematological malignancies and solid tumors (19, 38).
Immunotoxin conjugates are attractive as anti-leukemia agents, adding more potent cytotoxicity to the targeting antibody (21). The efficacy of immunotoxin conjugates is determined by the binding affinity of the Ab to its target, the expression levels of target antigens on tumor cells, and the efficiency of internalization to deliver the toxin (21, 38). Previous work from the Pastan group has demonstrated the high affinity of SL-101 to CD123 on AML cells (26). In this study, we demonstrated that most AML patient samples express high levels of CD123, both in total blasts and CD34+CD38− LSCe cells. The immunotoxin effects strongly correlated with CD123 expression in AML cell lines. This indicates that SL-101 may act differently than ligand-based toxins which may depend on both CD123 and CD131 for their cytotoxicity (15, 39). This correlation was not observed in primary AML samples, which may have been due to the generally high level of CD123 expression in most of these cases (15/18). Finally, the internalization rate affects the activity of the immunotoxin. A previous study revealed that a PE38 conjugate targeting CD22 was more active than one targeting CD19 in lymphoma cells because of the faster internalization rate of CD22, although both conjugates had similar cell-binding affinity. Our data indicate that SL-101 has internalization kinetics similar to that of anti-CD22 immunotoxin.
The anti-leukemia activity of SL-101 was further proven in primary AML specimens across the entire spectrum of cytogenetic risk and diversity of genetic alterations. The median IC50 value was 0.33 μg/mL, with 9 of 14 tested AML samples demonstrating an IC50 value less than 0.5 μg/mL, the plasma concentration level that can be achieved clinically by several different PE38 immunotoxins (22, 40). A critical question is whether the CD123-targeted therapeutics cause toxicity against normal hematopoietic stem/progenitor cell, although these cells are believed to express lower levels of CD123. Targeting of CD123 via chimeric antigen receptor-engineered T cells showed profound anti-leukemia efficacy; however this markedly impaired normal hematopoietic stem/progenitor cell function, as demonstrated by colony-forming assay and mouse models (25, 41). An additional argument for the CD123 CAR-T marrow toxicity could be the longer persistence of these cells in the circulation compared to the SL-101 immunoconjugate, i.e. area under the time-exposure curve, rather than just antigen expression density. Further, one would expect that use of an active cell exerting cytotoxicity can lead to a more pronounced effect than administration of an immunoconjugate. Transient expression of CARs to induce self-limited activity has been utilized in CART33 and has been suggested in CART123 therapy for AML (25, 42). To test the selectivity of SL-101 for leukemia versus normal stem/progenitor cells, we evaluated the effects of SL-101 in a colony-forming assay and found that SL-101 preferentially eliminated leukemia progenitors and spared their normal counterparts. It is plausible that immunoconjugate-antibody unlike CARTs could be less toxic against normal BM cells with low CD123 expression.
Further in vivo toxicology studies are required to fully examine potentially deleterious effects of this toxin conjugate against normal BM function. Expression of CD123 has been also reported on human endothelial cells and male germ cells (43–45). However, considering the low expression of CD123 on those cells and the safety profile of SL-401, a recombinant fusion protein comprising diptheria toxin fused to IL3, as a clinical-stage targeted therapy directed to CD123 (46–48), SL-101 is not expected to have significantly adverse effects on these organs.
It has been demonstrated that the antitumor mechanisms of PE38 involve inactivation of signal transduction pathways and inhibition of protein synthesis (38). Our mechanistic studies revealed that SL-101 could induce apoptosis in both AML blasts and CD123+ LSCe subsets. Nascent protein synthesis was efficiently inhibited, consistent with known mechanisms of action of PE38 immunotoxin. Surprisingly, we also observed significant inhibition of IL-3–induced p-STAT5 and p-AKT pathways, which may have been due to the antibody portion causing blockade of CD123 function. This pathway blockade may contribute towards anti-leukemia efficacy, in combination with immunotoxin-mediated cytotoxicity.
In conclusion, our data demonstrate that CD123 is a legitimate target for AML therapies aimed at elimination of residual leukemia stem/progenitor cells persistent in remission. We showed that the novel anti-CD123 antibody-toxin conjugate SL-101 is highly active in AML, inducing growth inhibition and apoptosis in AML blasts and LSCs by inhibiting protein synthesis and interfering with IL-3 signal transduction pathways. Our findings warrant further development of CD123 antibody-toxin conjugate for clinical application with the goal to improve outcomes of AML therapy.
Supplementary Material
Statement of translational relevance.
Although assessment of minimal residual disease (MRD) improves risk stratification and directs treatment in acute myeloid leukemia (AML), about 20% AML patients without detectable MRD experience relapse, which may be due to the persistence of residual leukemia stem cells (LSC)-containing cells. To study these residual LSCs may aid in identification of patients who may benefit from targeted therapeutics. In this study, we determined the association of CD34+CD123+ LSC-containing cells with MRD and relapse risk in AML patients. In order to target the CD123+ LSCs, we tested the binding and internalization of the anti-CD123 conjugate SL-101 in leukemia cells and evaluated its cytotoxicity against AML cell lines and primary AML, especially the primitive stem/progenitor cells, both in vitro and in vivo. Our data strongly suggest that SL-101 is a promising anti-leukemia agent in CD123-expressing AML.
Acknowledgments
Grant support: This work was supported in part by Stemline, the NIH/NCI through The MD Anderson Cancer Center Support Grant (P30 CA016672), the National Natural Science Foundation of China (NSFC 81200357, to L Han) and R01 CA194552-01 (To JF DiPersio).
Footnotes
Conflict-of-interest disclosure: M.K., research funding from Stemline; C.B. is an employee of Stemline Therapeutics. All authors reviewed and approved the manuscript.
Authors’ Contributions
Conception and design: L. Han, M. Konopleva
Development of methodology: L. Han, C. Brooks, M. Konopleva
Acquisition of data: L. Han, J.L. Jorgensen, S.A. Wang, C. Shi, Q. Zhang, A. Cavazos, R. Pan, H. Mu
Analysis and interpretation of data: L. Han, G.M. Nogueras González, X. Huang, M. Konopleva
Writing, review, and/or revision of the manuscript: L. Han, J.L. Jorgensen, C. Brooks, R. Pan, M. Andreeff, F. Ravandi, M. Konopleva
Administrative, technical, or material support: G. AI-Atrash, S.O. Ciurea, J. Zhou, M. Retting, J.F. DiPersio, J. Cortes, H. M. Kantarjian
Study supervision: M. Konopleva
References
- 1.DiNardo CD, Cortes JE. New treatment for acute myelogenous leukemia. Expert Opin Pharmacother. 2015;16:95–106. doi: 10.1517/14656566.2015.981527. [DOI] [PubMed] [Google Scholar]
- 2.Felipe Rico J, Hassane DC, Guzman ML. Acute myelogenous leukemia stem cells: from Bench to Bedside. Cancer Lett. 2013;338:4–9. doi: 10.1016/j.canlet.2012.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hope KJ, Jin L, Dick JE. Acute myeloid leukemia originates from a hierarchy of leukemic stem cell classes that differ in self-renewal capacity. Nat Immunol. 2004;5:738–43. doi: 10.1038/ni1080. [DOI] [PubMed] [Google Scholar]
- 4.Horton SJ, Huntly BJ. Recent advances in acute myeloid leukemia stem cell biology. Haematologica. 2012;97:966–74. doi: 10.3324/haematol.2011.054734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Terwijn M, Zeijlemaker W, Kelder A, Rutten AP, Snel AN, Scholten WJ, et al. Leukemic stem cell frequency: a strong biomarker for clinical outcome in acute myeloid leukemia. PLoS One. 2014;9:e107587. doi: 10.1371/journal.pone.0107587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Boumber Y, Kantarjian H, Jorgensen J, Wen S, Faderl S, Castoro R, et al. A randomized study of decitabine versus conventional care for maintenance therapy in patients with acute myeloid leukemia in complete remission. Leukemia. 2012;26:2428–31. doi: 10.1038/leu.2012.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kohnke T, Sauter D, Ringel K, Hoster E, Laubender RP, Hubmann M, et al. Early assessment of minimal residual disease in AML by flow cytometry during aplasia identifies patients at increased risk of relapse. Leukemia. 2015;29:377–86. doi: 10.1038/leu.2014.186. [DOI] [PubMed] [Google Scholar]
- 8.Ouyang J, Goswami M, Tang G, Peng J, Ravandi F, Daver N, et al. The clinical significance of negative flow cytometry immunophenotypic results in a morphologically scored positive bone marrow in patients following treatment for acute myeloid leukemia. Am J Hematol. 2015;90:504–10. doi: 10.1002/ajh.23988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.van Rhenen A, Feller N, Kelder A, Westra AH, Rombouts E, Zweegman S, et al. High stem cell frequency in acute myeloid leukemia at diagnosis predicts high minimal residual disease and poor survival. Clin Cancer Res. 2005;11:6520–7. doi: 10.1158/1078-0432.CCR-05-0468. [DOI] [PubMed] [Google Scholar]
- 10.Craddock C, Quek L, Goardon N, Freeman S, Siddique S, Raghavan M, et al. Azacitidine fails to eradicate leukemic stem/progenitor cell populations in patients with acute myeloid leukemia and myelodysplasia. Leukemia. 2013;27:1028–36. doi: 10.1038/leu.2012.312. [DOI] [PubMed] [Google Scholar]
- 11.Will B, Steidl U. Multi-parameter fluorescence-activated cell sorting and analysis of stem and progenitor cells in myeloid malignancies. Best Pract Res Clin Haematol. 2010;23:391–401. doi: 10.1016/j.beha.2010.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Florian S, Sonneck K, Hauswirth AW, Krauth MT, Schernthaner GH, Sperr WR, et al. Detection of molecular targets on the surface of CD34+/CD38− stem cells in various myeloid malignancies. Leuk Lymphoma. 2006;47:207–22. doi: 10.1080/10428190500272507. [DOI] [PubMed] [Google Scholar]
- 13.Jordan CT, Upchurch D, Szilvassy SJ, Guzman ML, Howard DS, Pettigrew AL, et al. The interleukin-3 receptor alpha chain is a unique marker for human acute myelogenous leukemia stem cells. Leukemia. 2000;14:1777–84. doi: 10.1038/sj.leu.2401903. [DOI] [PubMed] [Google Scholar]
- 14.Manz MG, Miyamoto T, Akashi K, Weissman IL. Prospective isolation of human clonogenic common myeloid progenitors. Proc Natl Acad Sci U S A. 2002;99:11872–7. doi: 10.1073/pnas.172384399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yalcintepe L, Frankel AE, Hogge DE. Expression of interleukin-3 receptor subunits on defined subpopulations of acute myeloid leukemia blasts predicts the cytotoxicity of diphtheria toxin interleukin-3 fusion protein against malignant progenitors that engraft in immunodeficient mice. Blood. 2006;108:3530–7. doi: 10.1182/blood-2006-04-013813. [DOI] [PubMed] [Google Scholar]
- 16.Martinez-Moczygemba M, Huston DP. Biology of common beta receptor-signaling cytokines: IL-3, IL-5, and GM-CSF. J Allergy Clin Immunol. 2003;112:653–65. doi: 10.1016/S0091. quiz 66. [DOI] [PubMed] [Google Scholar]
- 17.Testa U, Riccioni R, Diverio D, Rossini A, Lo Coco F, Peschle C. Interleukin-3 receptor in acute leukemia. Leukemia. 2004;18:219–26. doi: 10.1038/sj.leu.2403224. [DOI] [PubMed] [Google Scholar]
- 18.Jin L, Lee EM, Ramshaw HS, Busfield SJ, Peoppl AG, Wilkinson L, et al. Monoclonal antibody-mediated targeting of CD123, IL-3 receptor alpha chain, eliminates human acute myeloid leukemic stem cells. Cell Stem Cell. 2009;5:31–42. doi: 10.1016/j.stem.2009.04.018. [DOI] [PubMed] [Google Scholar]
- 19.Schrama D, Reisfeld RA, Becker JC. Antibody targeted drugs as cancer therapeutics. Nat Rev Drug Discov. 2006;5:147–59. doi: 10.1038/nrd1957. [DOI] [PubMed] [Google Scholar]
- 20.He SZ, Busfield S, Ritchie DS, Hertzberg MS, Durrant S, Lewis ID, et al. A Phase 1 study of the safety, pharmacokinetics and anti-leukemic activity of the anti-CD123 monoclonal antibody CSL360 in relapsed, refractory or high-risk acute myeloid leukemia. Leuk Lymphoma. 2014:1–10. doi: 10.3109/10428194.2014.956316. [DOI] [PubMed] [Google Scholar]
- 21.Wayne AS, Fitzgerald DJ, Kreitman RJ, Pastan I. Immunotoxins for leukemia. Blood. 2014;123:2470–7. doi: 10.1182/blood-2014-01-492256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kreitman RJ, Squires DR, Stetler-Stevenson M, Noel P, FitzGerald DJ, Wilson WH, et al. Phase I trial of recombinant immunotoxin RFB4(dsFv)-PE38 (BL22) in patients with B-cell malignancies. J Clin Oncol. 2005;23:6719–29. doi: 10.1200/JCO.2005.11.437. [DOI] [PubMed] [Google Scholar]
- 23.Kreitman RJ, Wilson WH, Bergeron K, Raggio M, Stetler-Stevenson M, FitzGerald DJ, et al. Efficacy of the anti-CD22 recombinant immunotoxin BL22 in chemotherapy-resistant hairy-cell leukemia. N Engl J Med. 2001;345:241–7. doi: 10.1056/NEJM200107263450402. [DOI] [PubMed] [Google Scholar]
- 24.Kreitman RJ, Pastan I. BL22 and lymphoid malignancies. Best Pract Res Clin Haematol. 2006;19:685–99. doi: 10.1016/j.beha.2006.06.009. [DOI] [PubMed] [Google Scholar]
- 25.Gill S, Tasian SK, Ruella M, Shestova O, Li Y, Porter DL, et al. Preclinical targeting of human acute myeloid leukemia and myeloablation using chimeric antigen receptor-modified T cells. Blood. 2014;123:2343–54. doi: 10.1182/blood-2013-09-529537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Du X, Ho M, Pastan I. New immunotoxins targeting CD123, a stem cell antigen on acute myeloid leukemia cells. J Immunother. 2007;30:607–13. doi: 10.1097/CJI.0b013e318053ed8e. [DOI] [PubMed] [Google Scholar]
- 27.Jaso JM, Wang SA, Jorgensen JL, Lin P. Multi-color flow cytometric immunophenotyping for detection of minimal residual disease in AML: past, present and future. Bone Marrow Transplant. 2014;49:1129–38. doi: 10.1038/bmt.2014.99. [DOI] [PubMed] [Google Scholar]
- 28.Al-Hussaini M, Rettig MP, Ritchey JK, Karpova D, Uy GL, Eissenberg LG, et al. Targeting CD123 in acute myeloid leukemia using a T-cell-directed dual-affinity retargeting platform. Blood. 2016;127:122–31. doi: 10.1182/blood-2014-05-575704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lai CY, Pan SL, Yang XM, Chang LH, Chang YL, Yang PC, et al. Depletion of 4E-BP1 and regulation of autophagy lead to YXM110-induced anticancer effects. Carcinogenesis. 2013;34:2050–60. doi: 10.1093/carcin/bgt146. [DOI] [PubMed] [Google Scholar]
- 30.Weldon JE, Pastan I. A guide to taming a toxin--recombinant immunotoxins constructed from Pseudomonas exotoxin A for the treatment of cancer. FEBS J. 2011;278:4683–700. doi: 10.1111/j.1742-4658.2011.08182.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Du X, Youle RJ, FitzGerald DJ, Pastan I. Pseudomonas exotoxin A-mediated apoptosis is Bak dependent and preceded by the degradation of Mcl-1. Mol Cell Biol. 2010;30:3444–52. doi: 10.1128/MCB.00813-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shlush LI, Zandi S, Mitchell A, Chen WC, Brandwein JM, Gupta V, et al. Identification of pre-leukaemic haematopoietic stem cells in acute leukaemia. Nature. 2014;506:328–33. doi: 10.1038/nature13038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shlush LI, Mitchell A. AML evolution from preleukemia to leukemia and relapse. Best Pract Res Clin Haematol. 2015;28:81–9. doi: 10.1016/j.beha.2015.10.004. [DOI] [PubMed] [Google Scholar]
- 34.Corces-Zimmerman MR, Hong WJ, Weissman IL, Medeiros BC, Majeti R. Preleukemic mutations in human acute myeloid leukemia affect epigenetic regulators and persist in remission. Proc Natl Acad Sci U S A. 2014;111:2548–53. doi: 10.1073/pnas.1324297111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Angelini DF, Ottone T, Guerrera G, Lavorgna S, Cittadini M, Buccisano F, et al. A Leukemia-Associated CD34/CD123/CD25/CD99+ Immunophenotype Identifies FLT3-Mutated Clones in Acute Myeloid Leukemia. Clin Cancer Res. 2015 doi: 10.1158/1078-0432.CCR-14-3186. [DOI] [PubMed] [Google Scholar]
- 36.Han L, Qiu P, Zeng Z, Jorgensen JL, Mak DH, Burks JK, et al. Single-cell mass cytometry reveals intracellular survival/proliferative signaling in FLT3-ITD-mutated AML stem/progenitor cells. Cytometry A. 2015;87:346–56. doi: 10.1002/cyto.a.22628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Han L, Zeng Z, Qiu P, Jorgensen JL, Mak DH, Burks JK, et al. Single-Cell Mass Cytometry Reveals Phenotypic and Functional Heterogeneity In Acute Myeloid Leukemia At Diagnosis and In Remission. 2013 [Google Scholar]
- 38.Gao W, Tang Z, Zhang YF, Feng M, Qian M, Dimitrov DS, et al. Immunotoxin targeting glypican-3 regresses liver cancer via dual inhibition of Wnt signalling and protein synthesis. Nat Commun. 2015;6:6536. doi: 10.1038/ncomms7536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Testa U, Riccioni R, Biffoni M, Diverio D, Lo-Coco F, Foa R, et al. Diphtheria toxin fused to variant human interleukin-3 induces cytotoxicity of blasts from patients with acute myeloid leukemia according to the level of interleukin-3 receptor expression. Blood. 2005;106:2527–9. doi: 10.1182/blood-2005-02-0540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kreitman RJ, Wilson WH, White JD, Stetler-Stevenson M, Jaffe ES, Giardina S, et al. Phase I trial of recombinant immunotoxin anti-Tac(Fv)-PE38 (LMB-2) in patients with hematologic malignancies. J Clin Oncol. 2000;18:1622–36. doi: 10.1200/JCO.2000.18.8.1622. [DOI] [PubMed] [Google Scholar]
- 41.Mardiros A, Dos Santos C, McDonald T, Brown CE, Wang X, Budde LE, et al. T cells expressing CD123-specific chimeric antigen receptors exhibit specific cytolytic effector functions and antitumor effects against human acute myeloid leukemia. Blood. 2013;122:3138–48. doi: 10.1182/blood-2012-12-474056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kenderian SS, Ruella M, Shestova O, Klichinsky M, Aikawa V, Morrissette JJ, et al. CD33-specific chimeric antigen receptor T cells exhibit potent preclinical activity against human acute myeloid leukemia. Leukemia. 2015;29:1637–47. doi: 10.1038/leu.2015.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rauch MC, Brito M, Zambrano A, Espinoza M, Perez M, Yanez A, et al. Differential signalling for enhanced hexose uptake by interleukin (IL)-3 and IL-5 in male germ cells. Biochem J. 2004;381:495–501. doi: 10.1042/BJ20031615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lim LH, Burdick MM, Hudson SA, Mustafa FB, Konstantopoulos K, Bochner BS. Stimulation of human endothelium with IL-3 induces selective basophil accumulation in vitro. J Immunol. 2006;176:5346–53. doi: 10.4049/jimmunol.176.9.5346. [DOI] [PubMed] [Google Scholar]
- 45.Korpelainen EI, Gamble JR, Vadas MA, Lopez AF. IL-3 receptor expression, regulation and function in cells of the vasculature. Immunol Cell Biol. 1996;74:1–7. doi: 10.1038/icb.1996.1. [DOI] [PubMed] [Google Scholar]
- 46.Cohen KA, Liu TF, Cline JM, Wagner JD, Hall PD, Frankel AE. Safety evaluation of DT388IL3, a diphtheria toxin/interleukin 3 fusion protein, in the cynomolgus monkey. Cancer Immunol Immunother. 2005;54:799–806. doi: 10.1007/s00262-004-0643-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cohen KA, Liu TF, Cline JM, Wagner JD, Hall PD, Frankel AE. Toxicology and pharmacokinetics of DT388IL3, a fusion toxin consisting of a truncated diphtheria toxin (DT388) linked to human interleukin 3 (IL3), in cynomolgus monkeys. Leuk Lymphoma. 2004;45:1647–56. doi: 10.1080/10428190410001663572. [DOI] [PubMed] [Google Scholar]
- 48.Frankel AE, Woo JH, Ahn C, Pemmaraju N, Medeiros BC, Carraway HE, et al. Activity of SL-401, a targeted therapy directed to interleukin-3 receptor, in blastic plasmacytoid dendritic cell neoplasm patients. Blood. 2014;124:385–92. doi: 10.1182/blood-2014-04-566737. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.