Abstract
Multimodality treatment including macroscopic complete resection (MCR) is currently the most effective therapy for patients with malignant pleural mesothelioma (MPM). However, the allocation to one of the possible combinations has to be discussed individually since various factors influence the decision making process. In the past years, relevant new information about the disease and the outcome of different treatment modalities have been generated, which allows a more individual allocation to the best treatment. For many years extrapleural pneumonectomy (EPP)—the most aggressive procedure to achieve a MCR—was the first choice in combination with adjuvant or neo-adjuvant chemotherapy and radiation therapy for all patients except those with minimal disease. However, mortality and morbidity of an EPP and the fact, that even with the most radical procedure only MCR and not a complete resection can be achieved, led to shift towards pleurectomy/decortication in most of the centers. But also today, EPP is still a valuable option in selected cases and the entire treatment concept requires a nuanced view, which will be discussed in this review.
Keywords: Extrapleural pneumonectomy (EPP), malignant pleural mesothelioma (MPM), multimodality treatment, outcome
Introduction
The best individual treatment for patients with malignant pleural mesothelioma (MPM) is discussed controversially in the last decades although increasing knowledge has been generated over time. In particular many “expert opinions” are currently circulating upon the role of extrapleural pneumonectomy (EPP) for MPM (1-5). The lack of evidence for answering this specific topic is mainly due to the fact that MPM is a rare disease and characterized by its heterogeneity in clinical presentation and biological behavior. This makes it difficult to perform studies addressing these surgical questions within a realistic time frame. Furthermore, the reporting of outcome data after surgical interventions especially morbidity is less standardized than after medical treatment. The present article aims to review the current literature focusing on short- and long-term outcome after EPP embedded in multimodality treatment concepts. It presents our perspective regarding the role of EPP, which is based on clinical experience accompanied with critical analysis of our own data as well as the literature and our interaction with other specialists worldwide.
Short-term outcome: 30- and 90-day mortality, morbidity
The value of surgical resection of MPM even in a multimodality concept is debated since no randomized trials have answered this question until now. Under these circumstances of a debatable benefit it is particularly important, that this intervention is as harmless as possible for the patient. The criticism of an exceedingly high mortality rate of EPP came up after publication of the results of the Mesothelioma and Radical Surgery (MARS) trial where surgical mortality in a small case series of 19 patients was 10.5%. However, this is not supported by recently reported EPP trials for trimodality therapy showing a 30-day mortality in a range of 2–5% in experienced centres (Table 1) (21). But taking into account all studies published between 1985–2010 a wide range of 0 to a maximum of 11.8% mortality is reported (22) at 30-day. This means that an EPP can be performed relatively safely by experienced surgeons in a well selected patient population. Reporting mortality at 30-day in these often very sick patients with multiple co-morbidities and after a complex operation might not be sufficient, whereas 90-day mortality seems to be more representative. Table 1 summarizes mortality rates reported in studies with more than 100 patients undergoing EPP. It has to be taken into account, that in these larger series also patients with higher risk profiles were accepted for surgery.
Table 1. Overview of reported mortality and morbidity and OS in extrapleural pneumonectomy (including studies with 100 or more EPPs completed).
| Publication | Study design | No. of patients in study | Modalities | No. of EPPs completed | Mortality | Overall morbidity | Median OS (months) |
|---|---|---|---|---|---|---|---|
| Unpub. Data; Opitz, Weder | 148 | 148 | In-hospital: 5%, 30 d: 5%, 90 d: 11% | Major: 38% | 21 | ||
| Mordant et al., 2016 (6) | Retro | 126 | 64 ind. CTX, 62 ind. IMRT | 126 | CTX: 6.3%, IMRT:3.2% | CTX: 29.7%, IMRT: 35.5% | NR |
| Sharkey et al., 2016 (7) | Retro | 362 | Mixed | 133 | In-hospital: 5.3%, 30 d: 6%, 90 d: 13.5% | 37.15 | 12.9 |
| Stahel et al., 2015 (8) | Phase II trial | 151 | Ind. CTX, ± adj. RT | 113 | 30 d: 4.4% | NR | NR |
| Bovolato et al., 2014 (9) | Retro | 1,365 | ± CTX | 301 | 30 d: 4.1%, 90 d: 6.9% | 21.6% | 18.8 |
| Lauk et al., 2014 (10) | Retro | 251 | Ind. CTX, ± adj. RT | 251 | 30 d: 5%, 90 d: 8% | major: 30% | NR |
| Nakas et al., 2014 (11) | Retro | 252 | ± CTX | 112 | NR | NR | 19.2 |
| Spaggiari et al., 2014 (12) | Retro | 518 | Mixed | 518 | In hospital: 3.9%, 30 d: 0.2%, 90 d: 6.9% | major: 26.3% | 18 |
| Sugarbaker et al., 2014 (13) | Retro | 1,258 | 529 | 30 d: 5%, 90 d: 8% | NR | 18 | |
| Nakas et al., 2012 (14) | Retro | 212 | NR | 127 | NR | NR | 15.6 |
| Rusch et al., 2012 (15) | Retro | 3,101 | Mixed | 1,225 | NR | NR | NR |
| Edwards et al., 2007 (16) | Retro | 105 | Mixed | 105 | 6.7% | NR | 14.5 |
| Flores et al., 2007 (17) | Retro | 945 | Mixed, ± ind. CTX, ± adj. RT, ± other | 208 | 30 d: 5% | NR | 14.3 |
| Rice, 2007 (18) | Retro | 100 | ± ind. CTX, ± adj. RT | 100 | 8% | 73% | 10.2 |
| Rusch et al., 1999 (19) | Retro | 231 | ± adj. therapy | 115 | 30 d: 5.2% | NR | NR |
| Sugarbaker et al., 1999 (20) | Retro | 183 | adj. CTX + RT | 183 | 3.8% | 50%, major: 24.5%, minor: 41% | 19 |
EPP, extrapleural pneumonectomy; OS, overall survival; IMRT, intensity-modulated radiotherapy; RT, radiotherapy; CTX, chemotherapy; NR, not reported.
Not surprisingly, morbidity after an EPP is very frequent and the prevention but also the efficiency of its management depends highly on the centers’ experience (Table 2). Surgery is performed often after an induction therapy in patients with usually a higher tumor load, sometimes with infiltration into the chest wall. The procedure harbors a relevant risk for technical complications. This includes risk of postoperative hemorrhage due to a large resection surface, empyema because the procedure takes many hours, failure at the reconstructed diaphragm or pericardium sites, but also, atrial fibrillation, frequently seen after lung resection, pneumonia or ARDS, and many more. Morbidity rates reported after EPP are extremely variable because of variation in definition of morbidity, being major or minor or life-threatening or other. In the following paragraph, the morbidity profile after EPP will be discussed. Table 3 summarizes the most frequent complications reported after EPP representing only studies with more than 100 patients after EPP.
Table 2. Morbidity and mortality after EPP—STS-GTD (n=225) [adapted from Burt et al., JTCVS 2014 (23)].
| Event | Univariate | Multivariate | |||
|---|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | ||
| Procedure (EPP) | 6.99 (2.73–17.90) | <0.001 | 6.51 (2.07–20.47) | 0.001 | |
| Center volume <5 procedures/y | 3.42 (1.52–7.70) | 0.002 | 1.38 (0.49–3.93) | 0.54 | |
EPP, extrapleural pneumonectomy.
Table 3. Common complications after EPP [adapted from Taioli et al. (24). Meta-analysis of survival after pleurectomy decortication versus extrapleural pneumonectomy in mesothelioma] (including studies with 100 or more EPPs completed).
| Publication | No. of patients in study | No. of EPPs completed | Overall morbidity | Complications [n] |
|---|---|---|---|---|
| Mordant et al., 2016 (6) | 126 | 126 | CTX: 29.7%, IMRT: 35.5% | AF [21] |
| Sharkey et al., 2016 (7) | 362 | 133 | 37.15% | AF [29], BPF [9], PE [3], PF [11] |
| Stahel et al., 2015 (8) | 151 | 113 | NR | BPF [4], PE [3] |
| Bovolato et al., 2014 (9) | 1,365 | 301 | 21.6% | AF [32], ARDS [1], BPF [3], DVT [2], PE [3], PF [3] |
| Lauk et al., 2014 (10) | 251 | 251 | major: 30% | AF [67], ARDS [3], BPF [20], PE [9], PF [12] |
| Spaggiari et al., 2014 (12) | 518 | 518 | major: 26.3% | AF [18.7%], BPF [14] |
| Flores et al., 2008 (25) | 663 | 385 | NR | DVT [3], PE [6] |
| Rice, 2007 (18) | 100 | 100 | 73% | ARDS [8], BF [2], DVT [3], PE [1], PF [3] |
| Sugarbaker et al., 1999 (20) | 183 | 183 | 50%, major: 24.5%, minor: 41% | AF [68], DVT [4], PE [3], PF [2] |
AF, atrial fibrillation; ARDS, adult respiratory distress syndrome; BPF, bronchopleural fistula; DVT, deep vein thrombosis; PE, pulmonary embolism; PF, patch failure; EPP, extrapleural pneumonectomy; IMRT, intensity-modulated radiotherapy; CTX, chemotherapy; NR, not reported.
Whereas atrial fibrillation represents a very frequent, reversible and relatively easily manageable problem, bronchopleural fistula and subsequent empyema, ARDS, or patch failure resulting in gastric herniation are severe postoperative complications. The frequency of empyema varies considerably in the literature between 3–30%. This might be related to the fact, that some series do not report about late empyema, which is to our experience the most frequent form of empyema occurring several weeks after the operations when patients already returned home or were transferred to rehabilitation institutions. Over the past decades we can report positive outcome after our accelerated empyema treatment with repetitive debridements and subsequent VAC treatment, where the chest was definitively closed within 8 days in 95% of the patients (26). Whereas bleeding complication and patch failures are more technical complications and should be avoided, pulmonary embolism and ARDS are difficult to predict or to avoid and contribute in most series to the lethal complications. For this reason, some centers recommend preoperative screening for deep venous thrombosis (27), or temporary postoperative therapeutic anticoagulation (10). For those centers offering induction chemotherapy to their patients, the perioperative anesthesiological protocol is critical as it has been well documented that high intraoperative FiO2 triggers postoperative ARDS (28). The management can be very challenging in a pneumonectomized patient and—as depicted in Table 4—it has been demonstrated that the centers’ experience plays a crucial role here. Centers with less than 5 EPP per year have a significant higher incidence of postoperative ARDS (23). Centers offering ECLS programs provide certainly more experience in handling these patients.
Table 4. Morbidity and Mortality after EPP—STS-GTD (n=225) [adapted from Burt et al., JTCVS 2014 (23)].
| Characteristic | EPP | ||
|---|---|---|---|
| ≥5/y | <5/y | P value | |
| Cases | 31 (32.6) | 64 (67.4) | <0.001 |
| ARDS | 0 (0.0) | 8 (12.5) | 0.050 |
ARDS, adult respiratory distress syndrome; EPP, extrapleural pneumonectomy.
In general, it has been reported from the same STS database, that the center volume influences significantly morbidity and mortality after MPM surgery in univariate analysis (Table 2).
Quality of life (QoL)
For obvious reasons, deterioration of QoL after this operation is a big concern for the patient as well as the physicians. This is especially important because life expectancy is limited in MPM patients and the QoL in this remaining time should be as good as possible. However, tumor progression is also substantially interfering with QoL and this has to be taken into account when data are interpreted. In Table 5 three studies are summarized reporting QoL after EPP.
Table 5. QoL reported in literature.
| Publication | Study design | No. of patients in study | Modalities | No. of EPPs completed | Mortality | Overall morbidity | QoL |
|---|---|---|---|---|---|---|---|
| Rena et al., 2012 (5) | Retro | 77 | ind. CTX (n=64), adj. CTX (n=13) | 40 | 30 d: 5% | 62% | Below baseline after 6 & 12 months |
| Treasure et al., 2011 (21) | Prospective | 50 | ind. CTX ± adj. therapy | 19 | 30 d: 10.5%, in-hospital: 15.8% | 69% | Below baseline after 6 weeks, 3, 6, 9, 12, 18 & 19 months |
| Weder et al., 2007 (29) | Prospective | 61 | ind. CTX ± adj. RT | 45 | 30 d: 2.2% | Major: 35% | Overall QoL was less impaired after surgery, though it did not reach the baseline level 6 months thereafter |
EPP, extrapleural pneumonectomy; QoL, quality of life; CTX, chemotherapy.
All studies report QoL below baseline after EPP, some until after 12 months after the operation (5), or even 19 months (21), whereas others report return to baseline after 6 months (29). In a recent analysis of our own cohort of patients undergoing induction chemotherapy in a 2-year period of time, patients filled out the EORTC QLC-C30 and -C15 and SF-36 self-rating questionnaires preoperatively, 6 weeks, and 4 months after the operation. Interestingly, some symptoms such as general health score, vitality, social functioning, and mental health were even better 4 months after the operation compared to preoperatively (unpublished data). This might be related to the fact, that entrapped lung is resected.
Long-term outcome
The most important parameter for evaluating an oncological treatment concept in MPM is overall survival (OS). It is well known, that the biological behavior of MPM is very heterogeneous and that clinical staging is much less accurate and reproducible than in other tumor entities. These two facts make it very difficult to describe precisely a patient cohort which undergoes treatment. Therefore, a comparison between case series and interventions should be done cautiously. However, the different reports in the literature describe a realistic scenario which allows some conclusions.
The role of EPP in treatment of MPM has been heavily criticized after the release of the MARS I trial (21). The MARS I trial concluded that “EPP within trimodal therapy offers no benefit and possibly harms patients” although only 16 patients of 24 patients assigned to the EPP arm received radical surgery. The study was not designed to answer the question of benefit or not of EPP but rather of the feasibility of such a trial. A definitive answer to this question would need an accrual of 670 patients to identify a survival benefit, as we have addressed these critical facts together with other experts (30).
OS data are within a comparable range in most series of multimodality treatment and reach usually a plateau at about 18–22 months median OS. Again it depends substantially on patient selection and subset analysis creates much more favorable outcome data. Table 1 summarizes the results from several centers reporting OS of patients undergoing multimodality treatment including EPP.
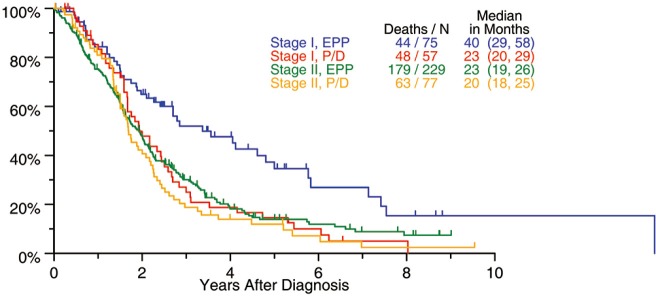
The biggest surgical database from the IASLC including 1,494 patients treated with surgery with curative intent, suggested a possible OS benefit in patients with stage I undergoing EPP with 40 months compared with 23 months after P/D (Figure 1). Based on these data, we should recommend EPP for stage I MPM. However, this has not gained support in current practice where most centers perform P/D in patients with little tumor load and reserve EPP for patients with advanced disease especially with fissure involvement.
Figure 1.
Survival after EPP (IASLC database) (15). EPP, extrapleural pneumonectomy; P/D, pleurectomy/decortication.
Progression free survival (PFS) is rarely reported in the literature and difficult to assess in MPM patients because the interpretation of a follow-up CT or PET CT after the surgical intervention is highly variable (as are the follow-up algorithms), as the differentiation between a regular postoperative change or a tumor recurrence can only be documented with repetitive scans and clear growth over time.
Recommendations
For many years, EPP was considered to be the only surgical procedure which achieves a macroscopic complete resection (MCR) and it was applied in operable patients independent of stage and histology. In some individuals with very early disease, a P/D was performed in order to preserve the lungs. Since OS of patients after P/D is not different compared to EPP, most groups started a transition from EPP to P/D in the majority of patients over the last years. However, as in other malignancies, one procedure does not fit for all and there are situations, in which a patient may require an EPP in order to obtain a MCR and still have a “functioning” lung left behind.
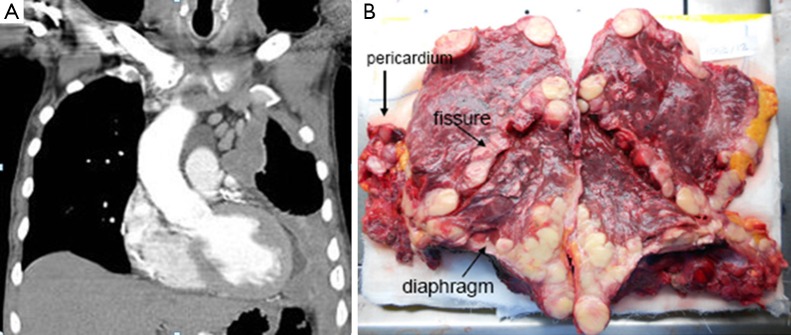
This can be anticipated from the CT scan, but final decision is sometimes been made during surgery only. This is typically the case in tumors with relevant lung parenchyma infiltration (as depicted in Figure 2) and preferentially on the left side were an EPP is usually better tolerated. Another argument for EPP is the concept applied in the SMART trial (1) with neoadjuvant hemithoracic radiotherapy followed by EPP reporting surprisingly good OS which cannot be explained by patient selection alone.
Figure 2.
CT scan staging imaging and pathological specimen of a patient undergoing EPP for MPM. (A) Staging contrast-enhanced CT scan: left-sided mesothelioma with suspicion of infiltration of the lung tissue; (B) post-resectional EPP pathological specimen with infiltration of mesothelioma into the lung parenchyma from the surface and via the fissures (31). EPP, extrapleural pneumonectomy; MPM, malignant pleural mesothelioma.
Therefore, patients foreseen for a P/D should be prepared to undergo EPP in case of extensive lung infiltration discovered during surgery. Not only during patients informed consent discussion, the patient should be informed about EPP but also preoperative functional assessments have to be performed accordingly. Pulmonary function testing showing a forced expiratory volume in 1 second (FEV1) of greater than 2 L is generally adequate for pneumonectomy for nearly all patients (32). Quantitative ventilation/perfusion scanning should be performed and predicted postoperative (PPO) FEV1 (ideally more than 1.2 L) be calculated in all patients. If both, FEV1 and DLCO are above 80%, resection up to pneumonectomy is feasible without any further investigation (32). Cardiac assessment should be performed in function of the patients’ comorbidities, some centres advise routine echography for all patients undergoing EPP in order to rule out pulmonary hypertension.
If early disease should be treated by EPP remains an open question, although data from IASLC are suggestive.
In conclusion, patients with MPM who are treated in a multidisciplinary concept including MCR, should be discussed with a nuanced view in which lung preservation should be achieved whenever possible, but an EPP may be a valuable solution in selected cases.
Acknowledgements
We thank Dr. Martina Friess and Dr. Chloé Spichiger for supporting us for literature review for this article.
Footnotes
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- 1.de Perrot M, Feld R, Leighl NB, et al. Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma. J Thorac Cardiovasc Surg 2016;151:468-73. 10.1016/j.jtcvs.2015.09.129 [DOI] [PubMed] [Google Scholar]
- 2.Cardillo G, Treasure T. Extrapleural Pneumonectomy Is Not Shown to Be Clinically Effective in the Treatment of Malignant Pleural Mesothelioma. Ann Surg 2017;265:e53. 10.1097/SLA.0000000000001266 [DOI] [PubMed] [Google Scholar]
- 3.Bertoglio P, Waller DA. The role of thoracic surgery in the management of mesothelioma: an expert opinion on the limited evidence. Expert Rev Respir Med 2016;10:663-72. 10.1586/17476348.2016.1171147 [DOI] [PubMed] [Google Scholar]
- 4.Baud M, Bobbio A, Lococo F, et al. Should we continue to offer extrapleural pneumonectomy to selected mesothelioma patients? A single center experience comparing surgical and non-surgical management. Jpn J Clin Oncol 2014;44:1127-9. 10.1093/jjco/hyu134 [DOI] [PubMed] [Google Scholar]
- 5.Rena O, Casadio C. Extrapleural pneumonectomy for early stage malignant pleural mesothelioma: A harmful procedure. Lung Cancer 2012;77:151-5. 10.1016/j.lungcan.2011.12.009 [DOI] [PubMed] [Google Scholar]
- 6.Mordant P, McRae K, Cho J, et al. Impact of induction therapy on postoperative outcome after extrapleural pneumonectomy for malignant pleural mesothelioma: does induction-accelerated hemithoracic radiation increase the surgical risk? Eur J Cardiothorac Surg 2016;50:433-8. 10.1093/ejcts/ezw074 [DOI] [PubMed] [Google Scholar]
- 7.Sharkey AJ, Tenconi S, Nakas A, et al. The effects of an intentional transition from extrapleural pneumonectomy to extended pleurectomy/decortication. Eur J Cardiothorac Surg 2016;49:1632-41. 10.1093/ejcts/ezv403 [DOI] [PubMed] [Google Scholar]
- 8.Stahel RA, Riesterer O, Xyrafas A, et al. Neoadjuvant chemotherapy and extrapleural pneumonectomy of malignant pleural mesothelioma with or without hemithoracic radiotherapy (SAKK 17/04): a randomised, international, multicentre phase 2 trial. Lancet Oncol 2015;16:1651-8. 10.1016/S1470-2045(15)00208-9 [DOI] [PubMed] [Google Scholar]
- 9.Bovolato P, Casadio C, Bille A, et al. Does surgery improve survival of patients with malignant pleural mesothelioma?: a multicenter retrospective analysis of 1365 consecutive patients. J Thorac Oncol 2014;9:390-6. 10.1097/JTO.0000000000000064 [DOI] [PubMed] [Google Scholar]
- 10.Lauk O, Hoda MA, de Perrot M, et al. Extrapleural pneumonectomy after induction chemotherapy: perioperative outcome in 251 mesothelioma patients from three high-volume institutions. Ann Thorac Surg 2014;98:1748-54. 10.1016/j.athoracsur.2014.05.071 [DOI] [PubMed] [Google Scholar]
- 11.Nakas A, Waller D. Predictors of long-term survival following radical surgery for malignant pleural mesothelioma. Eur J Cardiothorac Surg 2014;46:380-5; discussion 5. 10.1093/ejcts/ezt664 [DOI] [PubMed] [Google Scholar]
- 12.Spaggiari L, Marulli G, Bovolato P, et al. Extrapleural pneumonectomy for malignant mesothelioma: an Italian multicenter retrospective study. Ann Thorac Surg 2014;97:1859-65. 10.1016/j.athoracsur.2014.01.050 [DOI] [PubMed] [Google Scholar]
- 13.Sugarbaker DJ, Richards WG, Bueno R. Extrapleural pneumonectomy in the treatment of epithelioid malignant pleural mesothelioma: novel prognostic implications of combined N1 and N2 nodal involvement based on experience in 529 patients. Ann Surg 2014;260:577-80; discussion 80-2. 10.1097/SLA.0000000000000903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nakas A, Waller D, Lau K, et al. The new case for cervical mediastinoscopy in selection for radical surgery for malignant pleural mesothelioma. Eur J Cardiothorac Surg 2012;42:72-6; discussion 6. 10.1093/ejcts/ezr251 [DOI] [PubMed] [Google Scholar]
- 15.Rusch VW, Giroux D, Kennedy C, et al. Initial analysis of the international association for the study of lung cancer mesothelioma database. J Thorac Oncol 2012;7:1631-9. 10.1097/JTO.0b013e31826915f1 [DOI] [PubMed] [Google Scholar]
- 16.Edwards JG, Martin-Ucar AE, Stewart DJ, et al. Right extrapleural pneumonectomy for malignant mesothelioma via median sternotomy or thoracotomy? Short- and long-term results. Eur J Cardiothorac Surg 2007;31:759-64. 10.1016/j.ejcts.2007.01.061 [DOI] [PubMed] [Google Scholar]
- 17.Flores RM, Zakowski M, Venkatraman E, et al. Prognostic factors in the treatment of malignant pleural mesothelioma at a large tertiary referral center. J Thorac Oncol 2007;2:957-65. 10.1097/JTO.0b013e31815608d9 [DOI] [PubMed] [Google Scholar]
- 18.Rice DC, Stevens CW, Correa AM, et al. Outcomes After Extrapleural Pneumonectomy and Intensity-Modulated Radiation Therapy for Malignant Pleural Mesothelioma. Ann Thorac Surg 2007;84:1685. 10.1016/j.athoracsur.2007.04.076 [DOI] [PubMed] [Google Scholar]
- 19.Rusch VW, Venkatraman ES. Important prognostic factors in patients with malignant pleural mesothelioma, managed surgically. Ann Thorac Surg 1999;68:1799-804. 10.1016/S0003-4975(99)01038-3 [DOI] [PubMed] [Google Scholar]
- 20.Sugarbaker DJ, Flores RM, Jaklitsch MT, et al. Resection margins, extrapleural nodal status, and cell type determine postoperative long-term survival in trimodality therapy of malignant pleural mesothelioma: results in 183 patients. J Thorac Cardiovasc Surg 1999;117:54-63; discussion-5. [DOI] [PubMed]
- 21.Treasure T, Lang-Lazdunski L, Waller D, et al. Extra-pleural pneumonectomy versus no extra-pleural pneumonectomy for patients with malignant pleural mesothelioma: clinical outcomes of the Mesothelioma and Radical Surgery (MARS) randomised feasibility study. Lancet Oncol 2011;12:763-72. 10.1016/S1470-2045(11)70149-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cao CQ, Yan TD, Bannon PG, et al. A systematic review of extrapleural pneumonectomy for malignant pleural mesothelioma. J Thorac Oncol 2010;5:1692-703. 10.1097/JTO.0b013e3181ed0489 [DOI] [PubMed] [Google Scholar]
- 23.Burt BM, Cameron RB, Mollberg NM, et al. Malignant pleural mesothelioma and the Society of Thoracic Surgeons Database: an analysis of surgical morbidity and mortality. J Thorac Cardiovasc Surg 2014;148:30-5. 10.1016/j.jtcvs.2014.03.011 [DOI] [PubMed] [Google Scholar]
- 24.Taioli E, Wolf AS, Flores RM. Meta-analysis of survival after pleurectomy decortication versus extrapleural pneumonectomy in mesothelioma. Ann Thorac Surg 2015;99:472-80. 10.1016/j.athoracsur.2014.09.056 [DOI] [PubMed] [Google Scholar]
- 25.Flores RM, Pass HI, Seshan VE, et al. Extrapleural pneumonectomy versus pleurectomy/decortication in the surgical management of malignant pleural mesothelioma: results in 663 patients. J Thorac Cardiovasc Surg 2008;135:620-6, 6 e1-3. [DOI] [PubMed]
- 26.Schneiter D, Grodzki T, Lardinois D, et al. Accelerated treatment of postpneumonectomy empyema: a binational long-term study. J Thorac Cardiovasc Surg 2008;136:179-85. 10.1016/j.jtcvs.2008.01.036 [DOI] [PubMed] [Google Scholar]
- 27.Sugarbaker DJ, Jaklitsch MT, Bueno R, et al. Prevention, early detection, and management of complications after 328 consecutive extrapleural pneumonectomies. J Thorac Cardiovasc Surg 2004;128:138-46. 10.1016/j.jtcvs.2004.02.021 [DOI] [PubMed] [Google Scholar]
- 28.Muraoka M, Oka T, Akamine S, et al. Postoperative complications of pulmonary resection after platinum-based induction chemotherapy for primary lung cancer. Surg Today 2003;33:1-6. 10.1007/s005950300000 [DOI] [PubMed] [Google Scholar]
- 29.Weder W, Stahel RA, Bernhard J, et al. Multicenter trial of neo-adjuvant chemotherapy followed by extrapleural pneumonectomy in malignant pleural mesothelioma. Ann Oncol 2007;18:1196-202. 10.1093/annonc/mdm093 [DOI] [PubMed] [Google Scholar]
- 30.Weder W, Stahel RA, Baas P, et al. The MARS feasibility trial: conclusions not supported by data. Lancet Oncol 2011;12:1093-4; author reply 4-5. 10.1016/S1470-2045(11)70307-2 [DOI] [PubMed] [Google Scholar]
- 31.Bedirhan MA, Cansever L, Demir A, et al. Which type of surgery should become the preferred procedure for malignant pleural mesothelioma: extrapleural pneumonectomy or extended pleurectomy? J Thorac Dis 2013;5:446-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Brunelli A, Charloux A, Bolliger CT, et al. ERS/ESTS clinical guidelines on fitness for radical therapy in lung cancer patients (surgery and chemo-radiotherapy). Eur Respir J 2009;34:17-41. 10.1183/09031936.00184308 [DOI] [PubMed] [Google Scholar]