Abstract
Lysophosphatidylcholine (LPC) is a major phospholipid component of oxidized low-density lipoprotein (ox-LDL) and is implicated in its atherogenic activity. This study investigated the effects of LPC on cell viability, intracellular calcium homeostasis, and the protective mechanisms of chlorogenic acid (CGA) in human umbilical vein endothelial cells (HUVECs). LPC increased intracellular calcium ([Ca2+]i) by releasing Ca2+ from intracellular stores and via Ca2+ influx through store-operated channels (SOCs). LPC also increased the generation of reactive oxygen species (ROS) and decreased cell viability. The mRNA expression of Transient receptor potential canonical (TRPC) channel 1 was increased significantly by LPC treatment and suppressed by CGA. CGA inhibited LPC-induced Ca2+ influx and ROS generation, and restored cell viability. These results suggested that CGA inhibits SOC-mediated Ca2+ influx and ROS generation by attenuating TRPC1 expression in LPC-treated HUVECs. Therefore, CGA might protect endothelial cells against LPC injury, thereby inhibiting atherosclerosis.
Keywords: Calcium, Chlorogenic acid, Lysophosphatidylcholine, Store-operated channel, Transient receptor potential canonical channel 1
INTRODUCTION
Lysophosphatidylcholine (LPC) is generated by the hydrolysis of phosphatidylcholine by phospholipase A2, and 1-palmitoylsn-glycero-3-phosphocholine is the main component of phospholipids. LPC is a major component of oxidized low-density lipoprotein (ox-LDL), which causes atherosclerosis (1). LPC induces endothelial cell dysfunction by generating reactive oxygen species (ROS) in the vascular endothelium and increases oxidative stress by increasing the free Ca2+ concentration in the cytoplasm of leukocytes, macrophages, and muscle cells (2).
Intracellular calcium ions (Ca2+) are important secondary messengers and mediators of signal transduction in cells (3), which play important roles in various physiological processes and metabolic signaling (4). Usually, the intracellular Ca2+ concentration ([Ca2+]i) is low at 100 nmol/L, and this is maintained by protein binding or movement through ion channels (5). If the [Ca2+]i is increased by specific stimulation, cellular responses, including inflammation, cell growth, cell proliferation, thrombosis, oxidative stress, and calcification, can occur (6). The increase in [Ca2+]i induced by LPC is observed in various cell types associated with atherosclerosis and inflammatory diseases, such as endothelial cells, monocytes, macrophages, and myocardial cells (7). LPC increases [Ca2+]i through store-operated Ca2+ channels (SOC) in various cells. Functional analysis via mRNA expression of transient receptor potential canonical (TRPC) channels has led to it being identified as a molecular component of SOC (6).
Chlorogenic acid (CGA) is a polyphenol that is abundant in Crataegus monogyna, Eucalyptus globules, Eupatorium perfoliatum, Vaccinium angustifolium, and, especially, coffee (8–11). CGA is one of the many powerful phenolic antioxidant compounds in coffee (12). CGA has myocardial protective effects via the inhibition of lipid peroxidation, has anti-tumor effects, and produces antioxidant effects by removing free radicals (13). Dietary CGA also improves endothelial cell dysfunction by reducing the free radical generation of nitric oxide (12).
In this study, we demonstrated the function of LPC in [Ca2+]i-induced cell damage and investigated the protective effects of CGA against oxidative stresses in human umbilical vein endothelial cells (HUVECs).
RESULTS
Effect of CGA on LPC-induced reductions in cell viability
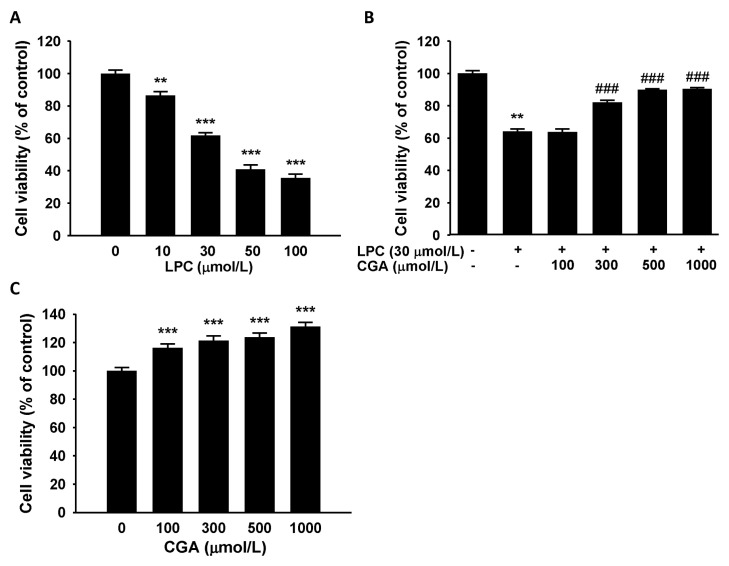
To determine the cytotoxic effects of LPC, cell viability was measured in HUVECs using the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. Treatment with LPC (10–100 μmol/L) for 1 h reduced cell viability significantly in a dose-dependent manner (Fig. 1A). CGA was then assessed for its inhibitory effect on LPC-induced cytotoxicity in HUVECs. The cytotoxic effect of LPC (30 μmol/L) challenge for 1 h was blocked by pretreatment with CGA (100–1,000 μmol/L) for 24 h. Moreover, at CGA concentrations greater than 300 μmol/L, the blocking effect was marked (Fig. 1B). We also observed the effect of CGA treatment without LPC on the HUVECs’ viability. CGA increased the cell viability significantly compared with vehicle-treated control cells after 24 h of incubation (Fig. 1C). These results indicated that CGA could ameliorate the LPC-induced reduction in cell viability.
Fig. 1.
Effects of chlorogenic acid (CGA) on lysophosphatidylcholine (LPC)-induced cell death in human umbilical vein endothelial cells (HUVECs). (A) Effects of LPC on cell viability in HUVECs. LPC treatment for 1 h decreased cell viability in a dose-dependent manner. (B) CGA attenuated the LPC-induced decrease in cell viability. Data are the mean ± SEM of three separate experiments. (C) Effects of CGA on HUVECs viability without LPC treatment. Treatment by CGA alone for 24 h significantly increases the cell viability. **P < 0.01, ***P < 0.001 compared with the corresponding vehicle only control; ###P < 0.001 compared with the corresponding single treatment with 30 μmol/L LPC.
Intracellular Ca2+ concentration changes after LPC treatment
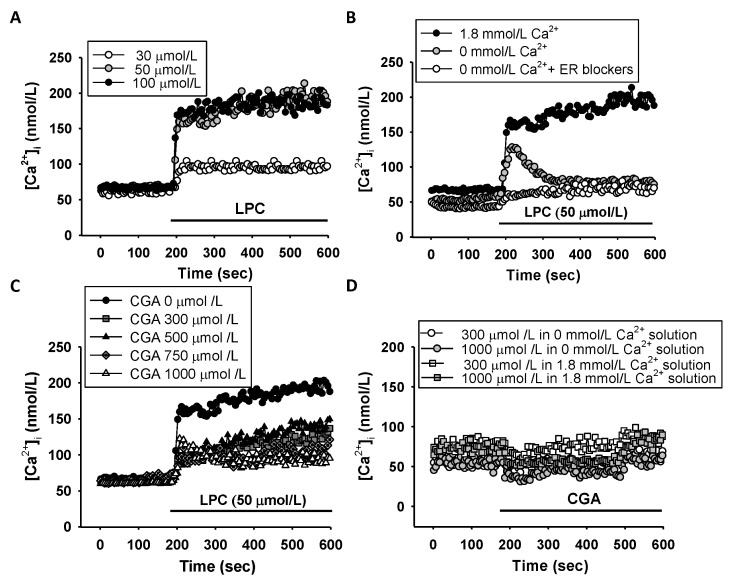
To investigate the effect of LPC on changes in [Ca2+]i in HUVECs, [Ca2+]i was measured in the cells before and after LPC treatment. [Ca2+]i increased with increasing concentrations of LPC (10–50 μmol/L), administered with physiological saline solution (PSS) containing 1.8 mmol/L Ca2+ (Fig. 2A). To determine whether the increased [Ca2+]i was caused by Ca2+ release from the endoplasmic reticulum (ER), [Ca2+]i was measured after blocking Ca2+ secretion from the ER. The increase in [Ca2+]i induced by LPC was suppressed by ER inhibitors (Fig. 2B). To assess the effect of CGA on the increased [Ca2+]i induced by LPC, changes in Ca2+ concentration after LPC (50 μmol/L) challenge, following pretreatment with CGA (300–1,000 μmol/L), were measured. CGA inhibited both Ca2+ release from the ER and intake from outside the cells in a dose-dependent manner (Fig. 2C). The effect of CGA treatment without LPC on [Ca2+]i was tested in the presence and the absence of external Ca2+. CGA alone did not cause any significant changes in [Ca2+]i in either the presence or the absence of external Ca2+ (Fig. 2D). These results suggested that LPC increases [Ca2+]i in the cells by releasing Ca2+ from ER, and that CGA might be involved in inhibiting intracellular Ca2+ release from the ER.
Fig. 2.
Effects of lysophosphatidylcholine (LPC) on intracellular calcium concentration ([Ca2+]i) in human umbilical vein endothelial cells (HUVECs). (A) LPC increased [Ca2+]i dose-dependently in a 1.8 mmol/L Ca2+ bathed-external solution. (B) The effects of LPC on [Ca2+]i depend on the external Ca2+ in HUVECs. LPC induced only an initial transient peak of [Ca2+]i in the absence of external Ca2+ and did not elicit the increase of [Ca2+]i under treatment with endoplasmic reticulum blockers: 50 μmol/L dantrolene and 10 μmol/L U73122. The sustained [Ca2+]i increase represents Ca2+ influx from the extracellular solution. (C) Effects of chlorogenic acid (CGA) on LPC-induced [Ca2+]i in HUVECs. The CGA blocked the LPC-induced sustained [Ca2+]i increase. (D) No effect of CGA without LPC in HUVECs. CGA (300 μmol/L and 1,000 μmol/L) was treated without Ca2+ and with 1.8 mM Ca2+ in HUVECs. Data are the mean values of three independent experiments.
Effect of CGA and SOC inhibitors on the LPS-induced increase in the intracellular Ca2+ concentration
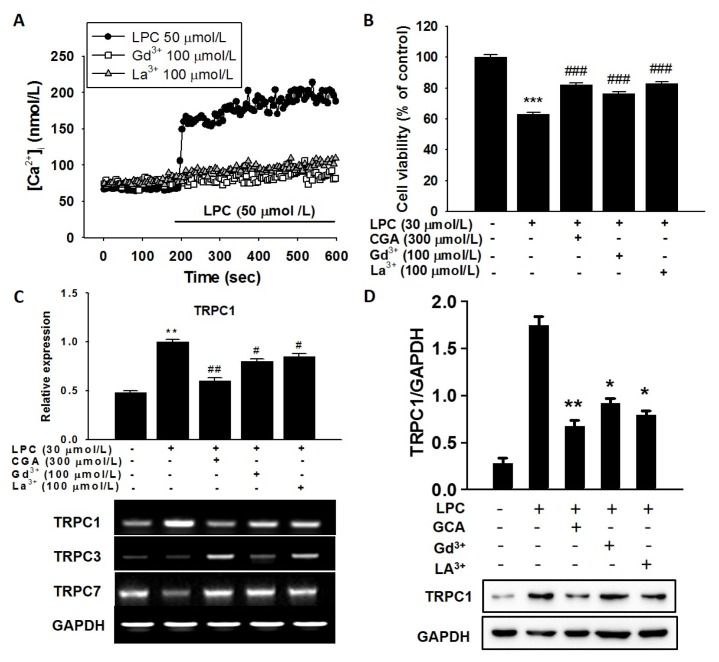
Increased [Ca2+]i might be induced by depletion of Ca2+ in the ER (14). To test this, cells were pretreated with the SOC blockers, Gd3+ (100 μmol/L) and La3+ (100 μmol/L), and then incubated with LPC (50 μmol/L) to measure [Ca2+]i. Gd3+ and La3+ completely ameliorated the increase in [Ca2+]i induced by LPC (Fig. 3A). These results suggest that the [Ca2+]i increase is induced by LPC in cells via SOCs and is suppressed by CGA treatment. To demonstrate the effect of the SOC inhibitors on cell viability, cells were pretreated with Gd3+ and La3+ for 30 minutes and then 30 μmol/L LPC was added. Both Gd3+ and La3+ prevented the LPS-induced reduction in cell viability and these blocking effects were similar to those of CGA (Fig. 3B). SOC channels are functionally involved in TRPC mRNA expression; therefore, we measured the expression levels of TRPC genes using reverse transcription polymerase chain reaction (RT-PCR) in HUVECs. TRPC1 mRNA expression was increased by LPC treatment, and both CGA and SOC blockers suppressed TRPC1 expression significantly (Fig. 3C). However, the expressions of other TRPC isoforms, TRPC3 and TRPC7, were not changed significantly by LPC and SOC blockers (Fig. 3C). We then measured TRPC1 protein levels under the same conditions. The TRPC1 protein level was increased by LPC and decreased by SOC blockers in HUVECs (Fig. 3D). These findings indicated that CGA and SOC blockers ameliorated the expression of TRPC1 induced by LPC in HUVECs.
Fig. 3.
Effects of store-operated channel (SOC) blockers on lysophosphatidylcholine (LPC)-induced [Ca2+]i in human umbilical vein endothelial cells (HUVECs). (A) SOC blockers inhibited the LPC-induced sustained [Ca2+]i increase. (B) Comparative effects of chlorogenic acid (CGA) and SOC blockers on LPC-induced cell death in HUVECs. CGA increased cell survival to the same levels as the SOC blockers. (C) Inhibitory effects of CGA on LPC-induced expressions of transient receptor potential canonical (TRPC) channels 1, 3, and 7 in HUVECs. (D) Immunoblotting for the TRPC1 protein under treatment by CGA and SOC blockers in HUVECs. The bar graph shows that CGA significantly suppressed LPC-induced TRPC1 mRNA expression with similar potency to gadolinium (Gd3+) and lanthanum (La3+). PCR data normalized to GAPDH are expressed as the mean values of three separate experiments. **P < 0.01, ***P < 0.001 compared with a corresponding single treatment with the vehicle control; #P < 0.05, ##P < 0.01, ###P < 0.001 compared with a corresponding single treatment with 30 μmol/L LPC.
Effect of CGA and SOC inhibitors on ROS generated by LPC
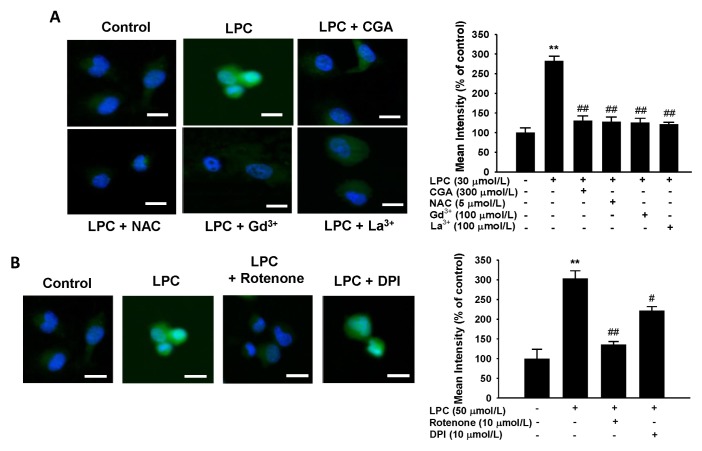
To evaluate whether LPC is involved in generating ROS in HUVECs, and whether this effect is suppressed by CGA, LPC (50 μmol/L) was administered to HUVECs and ROS levels were measured by fluorescent staining. ROS generation increased drastically in LPC-treated cells compared with that in the control group (Fig. 4). The LPC-mediated increases in ROS levels were suppressed by Gd3+ (100 μmol/L) and La3+ (100 μmol/L) compared with that in the LPC-treated cells. Moreover, CGA, as well as the ROS scavenger, N-acetyl-L-cysteine (NAC), decreased the ROS levels significantly (Fig. 4A). Finally, because ROS was induced by LPC, cells exposed to 50 μmol/L LPC were treated with 10 μmol/L diphenyleneiodonium (DPI), an NADPH oxidant inhibitor, and 10 μmol of rotenone, known as an inhibitor of phosphorylation in the mitochondrial electron transport system (ETS), respectively (Fig. 4B). Rote-none caused a more significant decrease in ROS than was observed using DPI. These data demonstrated that LPC might stimulate ROS generation in the mitochondria rather than in the cytoplasm.
Fig. 4.
Effects of chlorogenic acid (CGA) and store-operated channel (SOC) blockers on lysophosphatidylcholine (LPC)-induced ROS generation in human umbilical vein endothelial cells (HUVECs). (A) LPC increased ROS generation. CGA or SOC blockers decreased the LPC-induced ROS generation with a similar potency to N-acetyl-L-cysteine (NAC). The bar graph shows that CGA decreased the LPC-induced ROS generation with the same efficiency as a ROS generation inhibitor or SOC blockers gadolinium (Gd3+) and lanthanum (La3+) (B) Effects of ROS generation inhibitors on LPC-induced ROS generation in HUVECs. Rotenone inhibited LPC-induced ROS generation. The bar graph shows that rotenone decreased LPC-induced ROS generation to a greater extent than diphenyleneiodonium (DPI). Data are the mean ± SEM of three separate experiments. **P < 0.01 compared with a corresponding single treatment with the vehicle control; #P < 0.05, ##P < 0.01 compared with a corresponding single treatment with 50 μmol/L LPC.
DISCUSSION
Atherosclerosis is caused by hypercholesterolemia, smoking, hypertension, and diabetes. Increased levels of oxidized LDL (ox-LDL) in the cell is the most important factor in the development of atherosclerosis (15). LPC is a major component of ox-LDL, and is involved in the inflammatory response in cells, playing an important role in the development of atherosclerosis (16). The aim of this study was to evaluate the effects of LPC on [Ca2+]i, cell viability, and ROS generation in HUVECs, and to investigate the protective effect of CGA, including its mechanism of action. LPC treatment, known to induce cytotoxicity in endothelial cells, reduced the viability of HUVECs in a dose-dependent manner, and CGA blocked this LPC-induced cytotoxicity.
LPC is a major causal factor in atherosclerosis, which triggers malfunction or inflammation of endothelial cells (17). Dysfunction of endothelial cells is closely related to oxidative stress caused by increased ROS production (18). Moreover, LPC causes endothelial cell dysfunction by increasing ROS-mediated oxidative stress (19). The increased [Ca2+]i induced by LPC plays a central role in the initial process of cell injury (20). LPC challenge involves two phases in endothelial cells. The first is the initial rapid increase in [Ca2+]i and the second is the continued maintenance of a high [Ca2+]i concentration (21). U73122 and dantrolene-Na+ treatment (inhibitors of IP3R and ryanodine receptors in the ER) reduced [Ca2+]i; therefore, the initial increase in [Ca2+]i might be caused by release of Ca2+ from the ER (22). In addition, when extracellular Ca2+ was removed using up to 2 mmol/L EGTA (ethylene glycol-bis(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid), the increased Ca2+ levels induced by LPC were not maintained and were restored to the basal state, indicating that the [Ca2+]i might be dependent on extracellular Ca2+ (23). Thus, CGA suppresses Ca2+ influx from both the ER and the extracellular environment.
The non-selective cation channels SOCs are the main channels for Ca2+ influx in non-excitable cells during Ca2+ depletion from the ER (24). Gd3+ and La3+ are used as blocking agents of SOCs and transient receptor potential (TRP) pathways (25). Among TRPs, the canonical types of TRP (TRPC) channels are activated by phospholipase C (PLC) (26). If the signal molecules bind to G-protein-coupled receptors, the activity of PLC stimulates the decomposition of the phospholipid phosphatidylinositol 4,5-bisphosphate in the cell membrane into diacylglycerol and 1,4,5-tris-phosphate (IP3) (27). Gd3+ and La3+ reduced the expression of TRPC1 (Fig. 3); therefore, SOC blockers might inhibit the activity of PLC, which inhibits IP3-mediated release of Ca2+ from the ER by inhibiting the generation of IP3. Similarly, like Gd3+ and La3+, CGA might also inhibit extracellular Ca2+ influx and LPC-induced release of Ca2+ from the ER.
Antioxidants comprise a variety of compounds that prevent damage to cells and tissues caused by generation of ROS and oxidative stress. The most well-known antioxidant is NAC, a synthetic precursor of glutathione (GSH) containing thiol (28). NAC produces an antioxidant effect by removing ROS through the increased generation of intracellular GSH (29). Among a number of antioxidants in nature, CGA is an polyphenol-containing antioxidant that has myocardial protective and anti-tumor effects; inhibits the oxidation of LDL and DNA through chelating metal ions; and can remove ROS (13). CGA also represses ROS production, the expression of adhesion molecules, and the adhesion of monocytes in HUVECs (30). In our study, LPC-induced increases in ROS were reduced by CGA treatment, as well as by NAC challenge (Fig. 4). Moreover, SOC blockers also suppressed ROS production in a manner similar to CGA. Thus, these results suggested that CGA acts as an antioxidant by inhibiting free radicals in HUVECs.
Usually, the generation of ROS occurs via three major pathways. The first is oxidative phosphorylation in the mitochondrial electron transport system (ETS), the second is the activation of NADPH oxidase, and the third is via the action of lipoxygenase (31). In this study, rotenone, rather than DPI, was shown to generate oxidative phosphorylation of the mitochondrial ETS in HUVECs. Thus, our data suggested that LPC induces ROS production through increased oxidative phosphorylation in the mitochondrial ETS. Ca2+ influx into cells is increased via SOC by ROS generated in hypoxic conditions (32). Moreover, ROS produced by mitochondria is associated with Ca2+ channels in the cardiovascular system (33). Thus, ROS are generated by an increase in [Ca2+]i, whereas the production of ROS affects Ca2+ influx into the cells. It has been hypothesized that there is a correlation between the increase in Ca2+ concentration and the generation of ROS in cells (33). If the increases in Ca2+ and ROS levels cause inflammation of the cells, CGA might prevent atherosclerosis efficiently via its anti-inflammatory effect in vascular endothelial cells.
In this study, the Ca2+ level in LPC-treated cells was increased by releasing Ca2+ from the ER and by increasing Ca2+ influx via SOCs in HUVECs. In addition, we confirmed that TRPC1 was involved in the increased [Ca2+]i. LPC induced cytotoxicity and ROS generation in the mitochondrial ETS. CGA blocked production of ROS and Ca2+ intake via SOCs through decreased TRPC1 gene expression, resulting in inhibition of cell damage from ROS in HUVECs. A previous study demonstrated that TRPC1 and TRPC3 expression levels were regulated by activating Bax and caspase-3 and inhibiting Bcl-2 and pAkt in human coronary artery smooth muscle cells (SMCs) (34). However, they also suggested that TRPC3 did not participate in calcium-sensing receptor mediated calcium influx in HUVECs (11). Even though we did not demonstrate the molecular mechanism of CGA on TRPC1 gene expression, previous report suggested that caveolin-1, as a plasma membrane protein for the channel, interacts with TRPC channels to recruit and stabilize them in the plasma membrane (35). Thus, caveolin-1 might be a TRPC1 channel modulator.
In summary, we demonstrated that CGA suppresses the generation of ROS and may be considered as a pharmaceutical agent to ameliorate atherosclerosis by preventing cell damage from ROS in HUVECs.
MATERIALS AND METHODS
Detailed information is provided in the Supplementary Material.
Supplementary Information
ACKNOWLEDGEMENTS
This study was supported financially by a National Research Foundation (NRF) Grant funded by the Korean Government (MSIP) (No. 2014R1A5A2010008 and NRF-2016R1A2B4008516) and the Korea New faculty, Korea Research Foundation (NRF-2013R1A1A1006606) to SS.
Footnotes
CONFLICTS OF INTEREST
The authors have no conflicting financial interests.
REFERENCES
- 1.Grandl M, Schmitz G. Fluorescent high-content imaging allows the discrimination and quantitation of E-LDL-induced lipid droplets and Ox-LDL-generated phospholipidosis in human macrophages. Cytometry A. 2010;77:231–242. doi: 10.1002/cyto.a.20828. [DOI] [PubMed] [Google Scholar]
- 2.Steinbacher P, Eckl P. Impact of oxidative stress on exercising skeletal muscle. Biomolecules. 2015;5:356–377. doi: 10.3390/biom5020356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fedrizzi L, Lim D, Carafoli E. Calcium and signal transduction. Biochem Mol Biol Educ. 2008;36:175–180. doi: 10.1002/bmb.20187. [DOI] [PubMed] [Google Scholar]
- 4.Brini M, Cali T, Ottolini D, et al. Intracellular calcium homeostasis and signaling. Met Ions Life Sci. 2013;12:119–168. doi: 10.1007/978-94-007-5561-1_5. [DOI] [PubMed] [Google Scholar]
- 5.Korkiamaki T, Yla-Outinen H, Leinonen P, et al. The effect of extracellular calcium concentration on calcium-mediated cell signaling in NF1 tumor suppressor-deficient keratinocytes. Arch Dermatol Res. 2005;296:465–472. doi: 10.1007/s00403-004-0538-4. [DOI] [PubMed] [Google Scholar]
- 6.Mak S, Sun H, Acevedo F, et al. Differential expression of genes in the calcium-signaling pathway underlies lesion development in the LDb mouse model of atherosclerosis. Atherosclerosis. 2010;213:40–51. doi: 10.1016/j.atherosclerosis.2010.06.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xu F, Ji J, Li L, et al. Activation of adventitial fibroblasts contributes to the early development of atherosclerosis: a novel hypothesis that complements the “Response-to-Injury Hypothesis” and the “Inflammation Hypothesis”. Med Hypotheses. 2007;69:908–912. doi: 10.1016/j.mehy.2007.01.062. [DOI] [PubMed] [Google Scholar]
- 8.Bernatoniene J, Masteikova R, Majiene D, et al. Free radical-scavenging activities of Crataegus monogyna extracts. Medicina (Kaunas) 2008;44:706–712. [PubMed] [Google Scholar]
- 9.Maas M, Petereit F, Hensel A. Caffeic acid derivatives from Eupatorium perfoliatum L. Molecules. 2008;14:36–45. doi: 10.3390/molecules14010036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hicks JM, Muhammad A, Ferrier J, et al. Quantification of chlorogenic acid and hyperoside directly from crude blueberry (Vaccinium angustifolium) leaf extract by NMR spectroscopy analysis: single-laboratory validation. J AOAC Int. 2012;95:1406–1411. doi: 10.5740/jaoacint.11-415. [DOI] [PubMed] [Google Scholar]
- 11.Liang N, Kitts DD. Role of Chlorogenic Acids in Controlling Oxidative and Inflammatory Stress Conditions. Nutrients. 2016;8:16. doi: 10.3390/nu8010016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bonita JS, Mandarano M, Shuta D, et al. Coffee and cardiovascular disease: in vitro, cellular, animal, and human studies. Pharmacol Res. 2007;55:187–198. doi: 10.1016/j.phrs.2007.01.006. [DOI] [PubMed] [Google Scholar]
- 13.Kwon SH, Lee HK, Kim JA, et al. Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via anti-acetylcholinesterase and anti-oxidative activities in mice. Eur J Pharmacol. 2010;649:210–217. doi: 10.1016/j.ejphar.2010.09.001. [DOI] [PubMed] [Google Scholar]
- 14.Bogeski I, Bozem M, Sternfeld L, et al. Inhibition of protein tyrosine phosphatase 1B by reactive oxygen species leads to maintenance of Ca2+ influx following store depletion in HEK 293 cells. Cell Calcium. 2006;40:1–10. doi: 10.1016/j.ceca.2006.03.003. [DOI] [PubMed] [Google Scholar]
- 15.Choi KM. The Impact of Organokines on Insulin Resistance, Inflammation, and Atherosclerosis. Endocrinol Metab (Seoul) 2016;31:1–6. doi: 10.3803/EnM.2016.31.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Matsumoto T, Kobayashi T, Kamata K. Role of lysophosphatidylcholine (LPC) in atherosclerosis. Curr Med Chem. 2007;14:3209–3220. doi: 10.2174/092986707782793899. [DOI] [PubMed] [Google Scholar]
- 17.Schmitz G, Ruebsaamen K. Metabolism and atherogenic disease association of lysophosphatidylcholine. Atherosclerosis. 2010;208:10–18. doi: 10.1016/j.atherosclerosis.2009.05.029. [DOI] [PubMed] [Google Scholar]
- 18.Diano S. Role of reactive oxygen species in hypothalamic regulation of energy metabolism. Endocrinol Metab (Seoul) 2013;28:3–5. doi: 10.3803/EnM.2013.28.1.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liang GH, Park S, Kim MY, et al. Modulation of nonselective cation current by oxidized LDL and lysophosphatidylcholine and its inhibitory contribution to endothelial damage. Life Sci. 2010;86:733–739. doi: 10.1016/j.lfs.2010.03.005. [DOI] [PubMed] [Google Scholar]
- 20.Chen L, Liang B, Froese DE, et al. Oxidative modification of low density lipoprotein in normal and hyperlipidemic patients: effect of lysophosphatidylcholine composition on vascular relaxation. J Lipid Res. 1997;38:546–553. [PubMed] [Google Scholar]
- 21.Kim MS, Hong JH, Li Q, et al. Deletion of TRPC3 in mice reduces store-operated Ca2+ influx and the severity of acute pancreatitis. Gastroenterology. 2009;137:1509–1517. doi: 10.1053/j.gastro.2009.07.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mogami H, Lloyd Mills C, Gallacher DV. Phospholipase C inhibitor, U73122, releases intracellular Ca2+, potentiates Ins(1,4,5)P3-mediated Ca2+ release and directly activates ion channels in mouse pancreatic acinar cells. Biochem J. 1997;324(Pt 2):645–651. doi: 10.1042/bj3240645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ogita T, Tanaka Y, Nakaoka T, et al. Lysophosphatidylcholine transduces Ca2+ signaling via the platelet-activating factor receptor in macrophages. Am J Physiol. 1997;272:H17–24. doi: 10.1152/ajpheart.1997.272.1.H17. [DOI] [PubMed] [Google Scholar]
- 24.Burdakov D, Petersen OH, Verkhratsky A. Intraluminal calcium as a primary regulator of endoplasmic reticulum function. Cell Calcium. 2005;38:303–310. doi: 10.1016/j.ceca.2005.06.010. [DOI] [PubMed] [Google Scholar]
- 25.Pena F, Ordaz B. Non-selective cation channel blockers: potential use in nervous system basic research and therapeutics. Mini Rev Med Chem. 2008;8:812–819. doi: 10.2174/138955708784912166. [DOI] [PubMed] [Google Scholar]
- 26.Plant TD, Schaefer M. TRPC4 and TRPC5: receptor-operated Ca2+-permeable nonselective cation channels. Cell Calcium. 2003;33:441–450. doi: 10.1016/S0143-4160(03)00055-1. [DOI] [PubMed] [Google Scholar]
- 27.McLaughlin S, Murray D. Plasma membrane phosphoinositide organization by protein electrostatics. Nature. 2005;438:605–611. doi: 10.1038/nature04398. [DOI] [PubMed] [Google Scholar]
- 28.Chen N, Aleksa K, Woodland C, et al. The effect of N-acetylcysteine on ifosfamide-induced nephrotoxicity: in vitro studies in renal tubular cells. Transl Res. 2007;150:51–57. doi: 10.1016/j.trsl.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 29.Yamanaka T, Hishinuma A. Different effects of anticancer drugs on two human thyroid cell lines with different stages of differentiation. Nihon Naibunpi Gakkai Zasshi. 1995;71:73–86. doi: 10.1507/endocrine1927.71.1_73. [DOI] [PubMed] [Google Scholar]
- 30.Liao Y, Dong S, Kiyama R, et al. Flos lonicerae extracts and chlorogenic acid protect human umbilical vein endothelial cells from the toxic damage of perfluorooctane sulphonate. Inflammation. 2013;36:767–779. doi: 10.1007/s10753-013-9603-5. [DOI] [PubMed] [Google Scholar]
- 31.Salmeen A, Barford D. Functions and mechanisms of redox regulation of cysteine-based phosphatases. Antioxid Redox Signal. 2005;7:560–577. doi: 10.1089/ars.2005.7.560. [DOI] [PubMed] [Google Scholar]
- 32.Wang YX, Zheng YM. ROS-dependent signaling mechanisms for hypoxic Ca(2+) responses in pulmonary artery myocytes. Antioxid Redox Signal. 2010;12:611–623. doi: 10.1089/ars.2009.2877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fearon IM. OxLDL enhances L-type Ca2+ currents via lysophosphatidylcholine-induced mitochondrial reactive oxygen species (ROS) production. Cardiovasc Res. 2006;69:855–864. doi: 10.1016/j.cardiores.2005.11.019. [DOI] [PubMed] [Google Scholar]
- 34.Wang Y, Wang Y, Li GR. TRPC1/TRPC3 channels mediate lysophosphatidylcholine-induced apoptosis in cultured human coronary artery smooth muscles cells. Oncotarget. 2016;7:50937–50951. doi: 10.18632/oncotarget.10853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ong HL, Ambudkar IS. Molecular determinants of TRPC1 regulation within ER-PM junctions. Cell Calcium. 2015;58:376–386. doi: 10.1016/j.ceca.2015.03.008. [DOI] [PubMed] [Google Scholar]
- 36.Yi FX, Magness RR, Bird IM. Simultaneous imaging of [Ca2+]i and intracellular NO production in freshly isolated uterine artery endothelial cells: effects of ovarian cycle and pregnancy. Am J Physiol Regul Integr Comp Physiol. 2005;288:R140–148. doi: 10.1152/ajpregu.00302.2004. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.