Abstract
Beauveria bassiana, an entomopathogenic fungus, is the alternative biocontrol agent exploited against major economic crop pests. Pieris brassicae L. is an emerging pest of the Brassicaceae family. Therefore, in the present study, fungal isolates of Beauveria bassiana, viz. MTCC 2028, MTCC 4495, MTCC 6291, and NBAII-11, were evaluated for their virulence against third instar larvae of P. brassicae. Among all these fungal isolates, maximum mortality (86.66%) was recorded in B. bassiana MTCC 4495 at higher concentration of spores (109 conidia/ml), and the minimum mortality (30.00%) was recorded in B. bassiana MTCC 6291 at a lower concentration (107 conidia/ml) after ten days of treatment. The extracellular cuticle-degrading enzyme activities of fungal isolates were measured. Variability was observed both in the pattern of enzyme secretion and the level of enzyme activities among various fungal isolates. B. bassiana MTCC 4495 recorded the maximum mean chitinase (0.51 U/ml), protease (1.12 U/ml), and lipase activities (1.36 U/ml). The minimum mean chitinase and protease activities (0.37 and 0.91 U/ml, respectively) were recorded in B. bassiana MTCC 6291. The minimum mean lipase activity (1.04 U/ml) was recorded in B. bassiana NBAII-11. Our studies revealed B. bassiana MTCC 4495 as the most pathogenic isolate against P. brassicae, which also recorded maximum extracellular enzyme activities, suggesting the possible roles of extracellular enzymes in the pathogenicity of B. bassiana against P. brassicae.
Keywords: Chitinase, Entomopathogenic fungi, Lipase, Mortality, Protease
Introduction
Pieris brassicae (Lepidoptera: Pieridae), which is commonly known as cabbage butterfly, affects cole crops and causes damage at all its growth stages.1 This caterpillar feeds only on plants that releases glucosinolate, which is a characteristic feature of Brassicaceae that includes cabbages, cauliflower, and mustards.2 P. brassicae is a widely distributed lepidopteran pest of crucifers and is the most destructive pest of Brassicaceae,3 causing up to 40% yield loss per year.1 Chemical insecticides are usually applied to control P. brassicae, but high tolerance to most insecticides and associated environmental problems have jeopardized their continued use.4
The cosmopolitan anamorphic fungus Beauveria bassiana (Balsamo) Vuillemin (Ascomycota: Hypocreales) is a well-recognized microbial agent known to infect hundreds of host insect species. It is also considered as the most appropriate biological control agent in temperate agriculture regions.5 This ability has promoted extensive research on the potential of B. bassiana for biological control of many agriculturally important pests.6 The success of B. bassiana as biopesticides depends not only on its high efficacy against insect pests but also on its non-pathogenic behavior toward non-target pests.7 B. bassiana attaches its fungal spore on the hydrophobic cuticle that subsequently develops into an infectious structure called appressorium and releases an array of cuticle solubilizing enzymes.6 The fungus infects the host by breaching its cuticle, which is composed of chitin fibrils embedded in a matrix of proteins, lipids, and N-acylcatecholamines.8 B. bassiana secretes extracellular enzymes such as protease, chitinase, and lipase to degrade the major constituents of the cuticle of the insect that allow hyphal penetration into the cuticle. These enzymes play an important role in the pathogenesis and other physiological processes of B. bassiana.9, 10 So, the present study aimed to evaluate the pathogenicity of B. bassiana isolates against P. brassicae and compare their extracellular enzyme activities.
Materials and methods
Sources of fungal isolates and preparation of conidial suspension
Three fungal isolates, viz. B. bassiana MTCC 2028, MTCC 4495 and MTCC 6291, obtained from IMTECH, Chandigarh, India and one B. bassiana NBAII-11 fungal isolate procured from NBAII, Bangalore, India was used in the present study. The four isolates were maintained and grown in the Sabouraud Dextrose Agar Yeast Extract (SDAY) medium for 10–14 days in flasks, and aqueous spore suspensions were prepared. Spores were inoculated into 100 ml of SDAY medium in 250-ml Erlenmeyer flasks. The seeded flasks were incubated for 15 days at 25 ± 1 °C. The conidia were harvested by scrapping and suspended in sterilized 0.1% Tween 80 solution.11 The suspension was vortexed for 5 min to produce uniform conidial suspension and filtered through one layer of sterile muslin cloth. A sample of the suspension was quantified in a Neubauer chamber according to Alves.12 Then, the suspension containing 109 conidia/ml was prepared, and concentrations of 108 and 107 conidia/ml were obtained through serial dilutions. Sterilized water without fungal inoculation was used as a control.
Rearing of P. brassicae in laboratory
P. brassicae larvae were collected from cauliflower fields and reared on its leaves in the laboratory. The pupae formed were transferred to other battery jars having 2.5 cm thick moist sponge at the bottom covered with a circular paper of diameter just fitting jar to avoid their contact with a moist sponge. The open end of the jar was covered with a piece of muslin cloth and tied with a rubber band. The adults that emerged from the pupae were collected, and pairs of male and female butterflies were transferred to a cage having a pot of cauliflower plant for obtaining egg masses. A cotton swab dipped in liquid protein diet was hung from the roof of the cage. The leaves containing egg masses were taken out and kept separately in another clean battery jar. These egg masses constituted the initial culture, which was further used for the rearing of P. brassicae. The different instars at particular stages were kept in different battery jars to obtain synchronized culture of the pest for study.
Bioassays against third instar larvae of Pieris brassicae
B. bassiana isolates were evaluated for the pathogenicity against third instar larvae of P. brassicae. The viability of conidia was tested at different concentrations as per the methodology of Ibrahim et al.13 The bioassays were conducted as per the methodology of Akmal et al.14 with a few modifications. For each fungal isolate, three different fungal concentrations (107, 108 and 109 conidia/ml) were evaluated, and each concentration was replicated thrice. Larvae to be tested were allowed to crawl on different concentrations of fungal suspension in petri plates. For control, larvae were not treated. There were three replicates per treatment and ten larvae per replicate. After the initial treatment, the larvae were fed with fresh and untreated leaves of cauliflower. The cumulative percentage of mortality was recorded on successive days. Dead larvae were shifted to petri dishes with moist filter paper for fungal development and sporulation in order to confirm death due to fungal infection.
Production of cuticle-degrading enzymes
The extracellular cuticle-degrading enzymes (CDEs) such as chitinase, protease, and lipase activities of fungal isolates were evaluated as described by Nahar et al.14 with some modifications. These fungal isolates at a concentration of 107 spores/ml were inoculated into 250 ml Erlenmeyer flasks containing 100 ml of chitin medium, and the flasks were incubated on a rotary shaker at 150 rpm for 12 days at 25 ± 1 °C. The medium composition was (g/L): KH2PO4, 3.0; K2HPO4, 1.0; MgSO4, 0.7; (NH4)2SO4, 1.4; NaCl, 0.5; CaCl2, 0.5; yeast extract, 0.5; bacto-peptone, 0.5; chitin, 5.0; and olive oil, 5 ml/l. The enzyme extractions and assays were carried out after 24 h of incubation up to 12 days. Flasks were removed from incubation at alternative days, and the broth was centrifuged at 8000 rpm for 25 min at 4 ± 1 °C to extract clear supernatant. The supernatant was considered as crude enzyme extract and used for enzyme assays.
Enzyme assays
Chitinase activity was estimated in the culture supernatant by using acid-swollen chitin as the substrate.16 To prepare acid-swollen chitin, 10 g of chitin powder (Hi media) was suspended in 300 ml of chilled o-phosphoric acid (88%, w/v) at 4 °C for 1 h with occasional stirring. The mixture was then poured into ice cold distilled water and filtered through Whatman filter paper. Acid swollen chitin was repeatedly washed with 1% (w/v) sodium bicarbonate solution, and the pH was adjusted to 7. The solution was homogenized in a Waring blender (1 min), and the concentration of acid swollen chitin was adjusted to 7 mg/ml by adding 50 mM acetate buffer, pH 5.0. The reaction mixture for chitinase assay contained 1 ml of 0.7% swollen chitin, 1 ml of 50 mM acetate buffer, pH 5.0, and 1 ml of crude enzyme extract that was incubated at 50 °C for 1 h. The N-acetylglucosamine residues (GlcNAc) produced was estimated at 520 nm according to the methodology of Somogyi.17 One unit of enzyme activity was expressed as 1 μmol of GLcNAc per min.
Protease activity was measured using casein (10 g casein in 100 ml of 0.2 mM sodium carbonate buffer, pH 9.7) as a substrate.16 The reaction mixture contained 1 ml of casein, 1 ml of sodium carbonate buffer, pH 9.7, and 1 ml of crude enzyme extract. Moreover, the reaction mixture was incubated at 35 °C for 20 min. The reaction was terminated by adding 3 ml trichloroacetic acid (2.6 ml 5% TCA + 0.4 ml 3.3 N HCl). The absorbance of the TCA soluble fraction was measured at 280 nm. One unit of enzyme corresponds to 1 μmol of tyrosine per min.
Lipase activity was determined by using olive oil and gum acacia as substrate according to the methodology of Pignede et al.18 The substrate emulsion was prepared with olive oil (50 ml) and gum arabic (50 ml, 10% (w/v), Himedia) in the ratio of 1:1. The reaction mixture contained 1 ml of crude enzyme extract, 5 ml of substrate emulsion and 2 ml of 50 mM phosphate buffer, pH 6.8. The reaction mixture was incubated for 1 h at 37 °C with constant shaking, and the reaction was terminated with 4 ml of acetone–ethanol (1:1) containing 0.09% phenolphthalein as an indicator. Enzyme activity was determined by titration of the fatty acids released with 50 mM sodium hydroxide. One unit of lipase is the amount of enzyme that released of 1 μmol of fatty acids per min. All the enzyme assays were carried out in triplicates at alternative days.
Data analysis
One-way analysis of variance (ANOVA) was used to compare the results by using CPCS1 software. Enzymatic activities means were compared by Duncan's MRT post hoc test in SPSS 16.00 statistical software. Means with P < 0.05 were considered to be significantly different.
Results and discussion
Bioassay with P. brassicae
After three days of treatment, the higher and middle concentrations (109 and 108 conidia/ml, respectively) of B. bassiana NBAII-11 recorded maximum cumulative percentage mortality (26.66%). Comparatively, the higher concentration (109 conidia/ml) of B. bassiana 6291 recorded the least mortality (3.34%) and was not significantly different from the untreated control. After five days of treatment, the cumulative mortality ranged from 10.00% to 53.33%. The maximum mortality (53.33%) was recorded in B. bassiana NBAII-11 at a higher concentration of spores, which was at par with B. bassiana MTCC 4495 that recorded 50.00% mortality at the same concentration (Table 1). Moreover, B. bassiana NBAII-11 at middle and lower concentrations recorded 43.33% and 33.33% mortality, respectively, and these were at par with each other. The minimum mortality (10.00%) was recorded in MTCC 6291 and MTCC 2028 at a lower concentration (107 conidia/ml). However, this was at par with B. bassiana MTCC 4495 (13.34%) and MTCC 6291 (16.67%) at lower and higher concentrations, respectively. After seven days of treatment, the maximum mortality (60.00%) was recorded in B. bassiana NBAII-11 and MTCC 4495 at higher concentrations, which was at par with B. bassiana MTCC 4495 and NBAII-11 at the middle concentration that recorded 53.33% mortality (Table 1). This was followed by B. bassiana MTCC 2028, which recorded 43.33% mortality at higher concentration and was at par with MTCC 4495 and NBAII-11 with 60.00% and 53.33% mortality at higher and middle concentrations, respectively. Minimum mortality (16.66%) was recorded in MTCC 6291 at lower concentration (107 conidia/ml) and was at par with 26.66% mortality recorded in B. bassiana MTCC 2028 at the same concentration. All treatments at different concentrations were statistically significant compared to the untreated control after seven days of treatment.
Table 1.
Bio-efficacy of B. bassiana isolates against P. brassicae Linn.
| Treatments | Concentrations (conidia/ml) | Days of observations |
|||
|---|---|---|---|---|---|
| 3rd | 5th | 7th | 10th | ||
| MTCC 2028 | 107 | 3.34 (6.68)c | 10.00 (18.42)defg | 26.66 (30.77)defg | 43.33 (41.13)efh |
| 108 | 10.00 (18.42)bc | 20.00 (26.05)cdef | 36.66 (37.20)dfe | 50.00 (44.98)cdefg | |
| 109 | 16.66 (23.84)ab | 23.33(28.76)bcdef | 43.33 (41.05)bcde | 66.66 (54.76)bcde | |
| MTCC 4495 | 107 | 3.34 (6.68)c | 13.34 (17.97)defg | 33.33 (35.20)cdef | 60.00 (50.83)bcdef |
| 108 | 16.66 (23.84)ab | 36.66 (37.20)abcde | 53.33 (46.90)abcd | 70.00 (56.97)bcd | |
| 109 | 23.33 (28.76)ab | 50.00 (44.98)abc | 60.00 (50.74)ab | 86.66 (71.99)a | |
| MTCC 6291 | 107 | 3.34 (6.68)c | 10.00 (15.24)defg | 16.66 (23.84)fg | 30.00 (32.98)fgh |
| 108 | 13.33 (21.13)ab | 16.66 (23.84)cdef | 33.33 (34.99)cef | 43.33 (41.13)dg | |
| 109 | 3.34 (6.68)c | 16.67 (20.18)defg | 36.66 (37.20)cdef | 46.66 (43.05)defg | |
| NBAII-11 | 107 | 16.66 (23.84)ab | 33.33 (35.20)abcde | 40.00 (39.21)bcdef | 40.00 (39.21)efgh |
| 108 | 26.66 (30.98)a | 43.33 (41.13)abcd | 53.33 (46.90)abcd | 60.00 (50.83)bcdef | |
| 109 | 26.66 (30.98)a | 53.33 (46.90)abc | 60.00 (50.74)ab | 60.00 (50.74)bcdef | |
| CONTROL | 3.34 (6.68)c | 3.34(6.68)fg | 6.67 (12.55)h | 6.67 (12.55)i | |
| CD(0.05) | 11.85 | 14.42 | 8.54 | 10.93 | |
Values represent means of three replicates.
Values in parentheses represent arc sine transformations.
Means followed by the same letter (a, b, c) in vertical column are not significantly different at 0.05% level of probability.
After ten days of treatment, the mortality ranged from 30.00% to 86.66%. The maximum mortality (86.66%) was recorded in B. bassiana MTCC 4495 at a higher concentration of spores and was significantly better than all other treatments at various concentrations. This was followed by MTCC 4495, which recorded 70.00% mortality at middle concentration and was at par with MTCC 2028 and NBAII-11 isolates that recorded 66.66% and 60.00% mortality at higher concentrations (Table 1). These were further at par with NBAII-11 (60.00%) at the middle concentration (108 conidia/ml). The minimum mortality (30.00%) was recorded in B. bassiana MTCC 6291 at a lower concentration and was at par with NBAII-11 (40.00%) and MTCC 2028 (43.33%) at the same concentration. All treatments were significantly better than the untreated control.
Chitinase assay
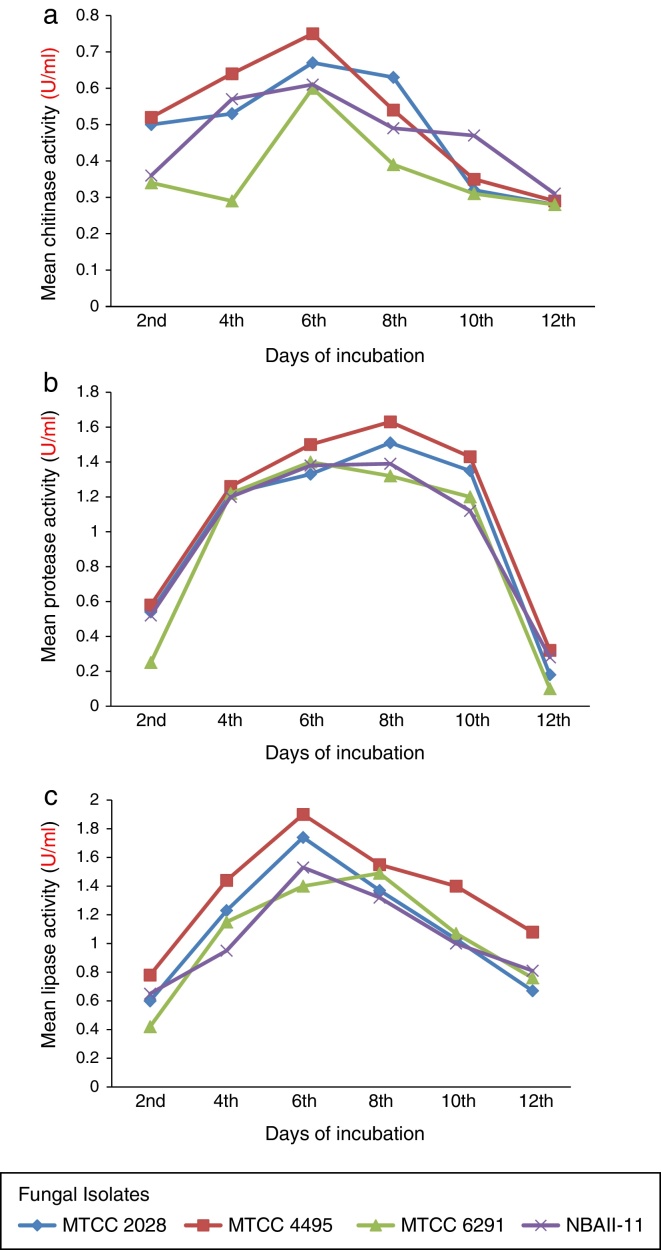
The maximum mean chitinase activity (0.51 U/ml) was recorded in B. bassiana MTCC 4495 followed by B. bassiana MTCC 2028 (0.49 U/ml). In contrast, the minimum mean chitinase activity (0.37 U/ml) was recorded in B. bassiana MTCC 6291 (Table 2). A significant difference was recorded between the chitinase activities at alternative days. The maximum chitinase activity (0.65 U/ml) was recorded on the sixth day that was significantly better than that on all other days. Among all days of observations, the minimum mean chitinase activity (0.29 U/ml) was recorded on the 12th day. The chitinase activity increased from 2nd to 6th day and decreased from 8th to 12th day (Fig. 1a).
Table 2.
Chitinase activity of fungal isolates at alternative days.
| Chitinase activity on different days after inoculation (μg of N-acetylglucosamine produced min/ml of the broth) |
|||||||
|---|---|---|---|---|---|---|---|
| Isolate | 2nd | 4th | 6th | 8th | 10th | 12th | Mean |
| MTCC 2028 | 0.50 ± 0.014dB | 0.53 ± 0.019cC | 0.67 ± 0.004aB | 0.63 ± 0.004bA | 0.32 ± 0.005eC | 0.28 ± 0.002fC | 0.49 |
| MTCC 4495 | 0.52 ± 0.013dA | 0.64 ± 0.06bA | 0.75 ± 0.010aA | 0.54 ± 0.011cB | 0.35 ± 0.006eB | 0.29 ± 0.002fB | 0.51 |
| MTCC 6291 | 0.34 ± 0.005cD | 0.29 ± 0.008fD | 0.60 ± 0.006aD | 0.39 ± 0.006bD | 0.31 ± 0.005dD | 0.28 ± 0.016eC | 0.37 |
| NBAII-11 | 0.36 ± 0.004eC | 0.57 ± 0.002bB | 0.61 ± 0.01aC | 0.49 ± 0.004cC | 0.45 ± 0.025dA | 0.31 ± 0.005fA | 0.46 |
| Mean | 0.43 | 0.51 | 0.65 | 0.51 | 0.36 | 0.29 | |
| CD(5%) isolates = 0.008 CD (5%) days = 0.01 CD(5%) isolates × days = 0.02 |
|||||||
Values are mean ± standard deviation of three replicates.
Means followed by the same letter (a, b, c) in horizontal column are not significantly different at 0.05% level of probability using Duncan LSD post hoc test.
Means followed by the same letter (A, B, C) in vertical column are not significantly different at 0.05% level of probability using Duncan LSD post hoc test.
Fig. 1.
(a) Effect of incubation days on Chitinase activity of B. bassiana isolates. (b) Effect of incubation days on protease activity of B. bassiana isolates. (c) Effect of incubation days on lipase activity of B. bassiana isolates.
Protease assay
The maximum mean protease activity (1.12 U/ml) was recorded in B. bassiana MTCC 4495, which was significantly better than all other isolates of B. bassiana. This was followed by B. bassiana MTCC 2028 with 1.02 U/ml (Table 3). The minimum mean protease activity (0.91 U/ml) was recorded in B. bassiana MTCC 6291. A significant difference was recorded in the mean protease activities at various days. The maximum protease activity (1.46 U/ml) was recorded on the 8th day of incubation, and the minimum mean protease activity (0.22 U/ml) was recorded on the 12th day of incubation. Protease activity increased from day 2nd to 8th, and thereafter, it gradually decreased (Fig. 1b).
Table 3.
Protease activity of fungal isolates at alternative days.
| Protease activity on different days after inoculation (μg of tyrosine produced min/ml of the broth) |
|||||||
|---|---|---|---|---|---|---|---|
| Isolate | 2nd | 4th | 6th | 8th | 10th | 12th | Mean |
| MTCC 2028 | 0.54 ± 0.023dB | 1.22 ± 0.021cAB | 1.33 ± 0.016bC | 1.51 ± 0.016aB | 1.35 ± 0.021bB | 0.18 ± 0.008eB | 1.02 |
| MTCC 4495 | 0.58 ± 0.009eA | 1.26 ± 0.028dA | 1.50 ± 0.021bA | 1.63 ± 0.016aA | 1.43 ± 0.016cA | 0.32 ± 0.001fA | 1.12 |
| MTCC 6291 | 0.25 ± 0.017dC | 1.22 ± 0.020cAB | 1.40 ± 0.016aB | 1.32 ± 0.016bD | 1.20 ± 0.021cC | 0.10 ± 0.073eB | 0.91 |
| NBAII-11 | 0.52 ± 0.009dB | 1.20 ± 0.021bB | 1.38 ± 0.046aBC | 1.39 ± 0.016aC | 1.12 ± 0.020cD | 0.28 ± 0.009eA | 0.98 |
| Mean | 0.47 | 1.22 | 1.40 | 1.46 | 1.27 | 0.22 | |
| CD(5%) isolates = 0.02 CD (5%) days = 0.02 CD(5%)isolates × days = 0.04 |
|||||||
Values are mean ± standard deviation of three replicates.
Means followed by the same letter (a, b, c) in horizontal column are not significantly different at 0.05% level of probability using Duncan LSD post hoc test.
Means followed by the same letter (A, B, C) in vertical column are not significantly different at 0.05% level of probability using Duncan LSD post hoc test.
Lipase assay
A significant difference was recorded between the mean lipase activities of various isolates of B. bassiana. The maximum mean lipase activity (1.36 U/ml) was recorded in B. bassiana MTCC 4495 followed by B. bassiana MTCC 2028 (1.11 U/ml). The minimum mean lipase activity (1.04 U/ml) was recorded in B. bassiana NBAII-11 (Table 4). The mean lipase activities of various days were significantly different from each other. The maximum mean lipase activity (1.64 U/ml) was recorded at 6th day, and the minimum mean lipase activity (0.61 U/ml) was recorded on the 2nd day of incubation (Fig. 1c).
Table 4.
Lipase activity of fungal isolates at alternative days.
| Lipase activity on different days after inoculation (μmol of fatty acids per min/ml of the broth) |
|||||||
|---|---|---|---|---|---|---|---|
| Isolate | 2nd | 4th | 6th | 8th | 10th | 12th | Mean |
| MTCC 2028 | 0.60 ± 0.12dAB | 1.23 ± 0.08bB | 1.74 ± 0.08aA | 1.37 ± 0.04bA | 1.03 ± 0.04cB | 0.67 ± 0.08dB | 1.11 |
| MTCC 4495 | 0.78 ± 0.12dA | 1.44 ± 0.12bA | 1.90 ± 0.08aA | 1.55 ± 0.09bA | 1.40 ± 0.08bA | 1.08 ± 0.08cA | 1.36 |
| MTCC 6291 | 0.42 ± 0.08dB | 1.15 ± 0.05bB | 1.40 ± 0.08aB | 1.49 ± 0.08aA | 1.07 ± 0.08bB | 0.76 ± 0.16cB | 1.05 |
| NBAII-11 | 0.65 ± 0.08dA | 0.95 ± 0.04bC | 1.53 ± 0.12aB | 1.32 ± 0.25aA | 1.00 ± 0.08bB | 0.81 ± 0.16bcB | 1.04 |
| Mean | 0.61 | 1.19 | 1.64 | 1.43 | 1.13 | 0.83 | |
| CD(5%) isolates = 0.72 CD(5%) days = 0.89 CD(5%)isolates × days = 0.17 |
|||||||
Values are mean ± standard deviation of three replicates.
Means followed by the same letter (a, b, c) in horizontal column are not significantly different at 0.05% level of probability using Duncan LSD post hoc test.
Means followed by the same letter (A, B, C) in vertical column are not significantly different at 0.05% level of probability using Duncan LSD post hoc test.
Discussion
Mortality variation was observed among various isolates of B. bassiana; this could be related to the ability to counter the insect immune response, molecular, and physiological mechanisms such as excretion of extracellular enzymes.19 In B. bassiana, various proteins and enzymes have been characterized that are essential for virulence. Proteins of the P450 family have been identified as important virulence factors involved in cuticle-degradation.20 It was also reported that Bbslt2, a novel mitogen-activated protein (MAP) kinase has a pivotal role in regulating fungal development, growth, and pathogenicity.21 Bbgas1, a gene encoding a putative transferase (Glycosylphosphatidyl inositol-anchored β-1,3-Glucanosyl transferase) is involved in virulence and thermal regulation.22 Variation in mortality among different strains of B. bassiana can be correlated with differential expression of these virulence governing genes. Study of genetic, molecular and physiological mechanisms of B. bassiana can underpin the exact reason responsible for the variation among different isolates of B. bassiana.
The cumulative percentage mortality enhanced with the increase in spore concentration. Maximum mortality was recorded at higher concentrations of spores (109 Spores/ml) on all days of observations. Butt and Goettel23 reported a positive correlation between the number of infective spores and mortality by mycosis. They found that susceptibility of most insects depends on spore dosage.
Wright et al.,24 also reported that susceptibility of target insect to fungal infection is dose dependent and the mortality increases with time. Akmal et al.14 evaluated different entomopathogenic fungal strains against different species of aphids under laboratory conditions and found that their mortality enhanced with the increase in spore concentration. Tefera and Pringle25 reported the mortality in their target insect (Chilo partellus) increased when treated with B. bassiana and M. anisopliae. They reported 100% mortality after ten days of the treatment. Similar observations were recorded in our present study against third instar larvae of P. brassicae. These larvae recorded higher mortality at higher concentrations that increased with time. Akbari et al.26 also reported that the cumulative percentage mortality was lower in the first two days but increased thereafter.
The extracellular enzyme activity was considered as an important factor for their virulence. Chitinase is the most important enzyme to degrade the chitin polymer of the insect cuticle. Virulence of entomopathogenic fungi can be correlated with the chitinase activity. Among four isolates of B. bassiana, the highest mean chitinase activity was recorded in B. bassiana MTCC 4495, which showed maximum mortality against third instar larvae of P. brassicae. Pelizza et al.27 also reported that B. bassiana isolate with highest levels of chitinase activity was more pathogenic against Tropida criscollaris. Perinotto et al.28 found that the most virulent strain of entomopathogenic fungi against Rhipicephalus microplus showed the maximum chitinase activity. So, chitinase activity can be correlated with the virulence of B. bassiana. Protease was considered as an important enzyme in the infective process of entomopathogenic fungi.29 Entomopathogenic fungi produced large quantities of protease that degraded the proteinaceous material, and the solubilized proteins were then degraded by amino peptidases and exopeptidases to form amino acids that served as nutrients for the fungi. Extracellular protease produced by B. bassiana is suspected to be involved in the pathogenesis. B. bassiana MTCC 4495, which was evaluated as the most pathogenic strain against third instar larvae of P. brassicae, showed maximum protease activity. Biodochka and Khachatourians30 recorded marked proteolytic activity associated with virulent isolates of Beauveria. B. bassiana MTCC 6291 recorded the least mortality and showed the minimum protease activity. Perinotto et al.28 showed that the isolates of entomopathogenic fungi, which were more virulent against R. microplus, exhibited the highest levels of proteolytic activity. Thus, proteases can be considered as the important factor that governs virulence in B. bassiana against P. brassicae.
Lipases hydrolyze the ester bonds of lipoproteins, fats, and waxes at the interior of the insect integument and significantly contribute to the cuticle penetration and initial release of nutrients.31 Lipase activity and virulence of entomopathogenic fungi can be correlated. B. bassiana MTCC 4495 strain was found to be pathogenic against third instar larvae of P. brassicae, which showed the maximum mean lipase activity. The isolate with minimum mean lipase activity was found to be the least effective against P. brassicae. Hence, lipases can be considered as an important virulence factor in B. bassiana.
The chitinase, protease and lipase activities were found to increase from the 2nd day of incubation up to six days of incubation. After the 8th day of incubation, the extracellular enzyme activities were found to decrease. Proteolytic activity of entomopathogenic fungi decreased with the increase in culture age probably due to nutrient limitation or autolysis of the culture. Braga et al.32 reported that high caseinolytic activity of M. anisopliae during four to six days of culture and a steep decrease in the activity up to 16 days of culture. Ziabeeet et al.33 reported maximum lipase activity at 6th day and subsequent decrease in incubation lipase activity.
The present results are in corroboration with that of other studies. Nahar et al.15 reported chitinase activity (0.001 U/ml) in Metarhizium anisopliae after 120 h of incubation in chitin-containing medium. Supakdamrongkul et al.34 reported specific lipase activity (9.41 U/mg) in purified form of lipase after four days of culture. Dhar and Kaur35 evaluated protease activities of different isolates of B. bassiana at different days of incubation and recorded that the protease activity ranged from 0.0804 to 0.7583 U/ml at sixth day of incubation. They also reported variability in enzyme activities and found higher protease activity comparative to other fungal isolates.
Moreover, colloidal chitin, peptone, and olive oil in minimal medium induced the production of chitinase, proteases, and lipase, respectively. Fernandes et al.36 reported the production of proteases by the entomopathogenic fungi only in the presence of gelatin as the substrate in the medium. In contrast, St. Legar et al.37 exploited colloidal chitin as the sole source of carbon and nitrogen to induce Pr1 activity and reported that polymeric substances like cellulose and insect cuticle enhance the Pr1 activity either by inducing the protease production. They also reported that colloidal chitin was unable to produce catabolite repression. Our studies suggest that colloidal chitin with peptone may induce the production of chitinase as well as protease. Chitinase and protease act concurrently in these enzymes; this was supported by the fact that the highest levels of chitinolytic and proteolytic activities were recorded on 4th, 6th and 8th day of incubation.
However, the mean lipase activity on the 2nd day of incubation was found to be more than the mean chitinase and protease activities on the same day; thus, this may be considered as the first enzyme secreted by B. bassiana for the initiation of infection. Silva et al.38 also reported that the lipase secreted by entomopathogenic fungi were involved in the initial stages of the adhesion and penetration. In addition, extracellular lipase activity was found to be more as compared to protease and chitinase; hence, lipase may be considered as an important enzyme in metabolic activities of B. bassiana. Supakdamrongkul and his coworkers34 found that fungal isolates amended with purified lipase showed higher virulence against Spodoptera litura. Lipase can be considered as the most important enzyme in the physiology and pathogenesis of B. bassiana. The difference in enzyme activities and level of secretion can be correlated with the genetic architecture of B. bassiana. In future, these contradictions can be explained by the genomics and proteomics analysis of B. bassiana. Moreover, the relationship between enzyme activities and virulence may be useful in developing enzyme-based screening methods to select the most virulent fungal isolates against P. brassicae.
Conflicts of interest
The authors declare no conflicts of interest.
Associate Editor: Welington Luiz de Araújo
References
- 1.Hasan F., Ansari M.S. Population growth of Pieris brassicae (L.) (Lepidoptera: Pieridae) on different cole crops under laboratory conditions. J Pest Sci. 2011;84:179–186. [Google Scholar]
- 2.Hopkins R.J., Van Dam N.M., Van Loon J.J.A. Role of glucosinolates in insect–plant relationships and multitrophic interactions. Ann Rev Entomol. 2009;54:57–83. doi: 10.1146/annurev.ento.54.110807.090623. [DOI] [PubMed] [Google Scholar]
- 3.Ansari M.S., Hasan F., Ahmad N. Influence of various host plants on the consumption and utilization of food by Pieris brassicae (Linn.) Bull Entomol Res. 2012;102(2):231–237. doi: 10.1017/S000748531100054X. [DOI] [PubMed] [Google Scholar]
- 4.Grisakova M., Metspalu L., Jogar K., Hiiesaar K., Kuusik A., Poldama P. Effect of biopesticides neem EC on the large white butterfly Pieris brassicae L. (Lepidoptera: Pieridae) Agron Res. 2006;4:181–186. [Google Scholar]
- 5.Glare T.R. Molecular characterization in the entomopathogenic fungal genus Beauveria. Laimburg J. 2004;1:286–298. [Google Scholar]
- 6.Inglis G.D., Goettel M.S., Butt T.M., Strasser H. Use of Hyphomycetous fungus for managing insect pests. In: Butt T.M., Jackson C., Magan N., editors. Fungi AS biocontrol Agents: Progress, Problems and Potential. CABI Publishing; Wallingford, UK: 2001. pp. 23–69. [Google Scholar]
- 7.Thungrabeab M., Tongma S. Effect of entomopathogenic Fungi, Beauveria bassiana (Balsamo) and Metarhizium anisopliae (Metsch) on non target insects. J Sci Tech. 2007;7:8–12. [Google Scholar]
- 8.Richard J.S., Neal T.D., Karl J.K., Michael R.K. Model reactions for insect cuticle sclerotization: participation of amino groups in the cross-linking of Manducasexta cuticle protein MsCP36. Insect Biochem Mol Biol. 2010;40:252–258. doi: 10.1016/j.ibmb.2010.02.008. [DOI] [PubMed] [Google Scholar]
- 9.Wang C., Hu G., St. Leger R.J. Differential gene expression by Metarhizium anisopliae growing in root exudate and host (Manducasexta) cuticle or hemolymph reveals mechanisms of physiological adaptation. Fungal Genet Biol. 2005;42(8):704–718. doi: 10.1016/j.fgb.2005.04.006. [DOI] [PubMed] [Google Scholar]
- 10.Cho E.M., Boucias D., Keyhani N.O. EST analysis of cDNA libraries from the entomopathogenic fungus Beauveria(Cordyceps) bassiana II. Fungal cells sporulating on chitin and producing oosporein. Microbiology. 2006;152:2855–2864. doi: 10.1099/mic.0.28845-0. [DOI] [PubMed] [Google Scholar]
- 11.Luz C., Tigano M.S., Silva I.G., Cordeiro C.M.T., Alijanabi S.M. Selection of Beauveria bassiana and Metarhizium anisopliae isolates to control Triatomainfestans. Mem Inst Oswaldo Cruz. 1998;93:839–846. doi: 10.1590/s0074-02761998000600026. [DOI] [PubMed] [Google Scholar]
- 12.Alves S.B., Fungosentomopatogenicos . In: Controle Microbaino de Insectos. Alves S.B., editor. FEALQ; Piracicaba, Brazil: 1998. pp. 289–382. [Google Scholar]
- 13.Ibrahim L., Butt T.M., Beckett A., Clarke S.J. The germination of oil-formulated conidia of the insect pathogen, Metarhizium anisopliae. Mycol Res. 1999;103:901–907. [Google Scholar]
- 14.Akmal M., Freed S., Malik M.N., Gul H.T. Efficacy of Beauveria bassiana (Deuteromycotina: Hypomycetes) against different aphid species under laboratory conditions. Pakistan J Zool. 2013;45(1):71–78. [Google Scholar]
- 15.Nahar P., Ghormade V., Deshpande M.V. The extracellular constitutive production of chitin deacetylase in Metarhizium anisopliae: possible edge to entomopathogenic fungi in the biological control of insect pests. J Invertebr Pathol. 2004;85:80–88. doi: 10.1016/j.jip.2003.11.006. [DOI] [PubMed] [Google Scholar]
- 16.Vyas P.R., Deshpande M.V. Chitinase production by Myrotheciumverrucaria and its significance for fungal mycelia degradation. J Gen Appl Microbiol. 1989;35:343–350. [Google Scholar]
- 17.Somogyi M. Notes on sugar determination. J Biol Chem. 1952;195:19–23. [PubMed] [Google Scholar]
- 18.Pignede G., Wang H., Fudalei F., Gaillardia C., Semon M., Nicaud J.M. Characterisation of an extra lipase encoded by L1P2 in Yarrowia lipolytica. J Bacteriol. 2000;182:2802–2810. doi: 10.1128/jb.182.10.2802-2810.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Valero-Jimenez C.A., Debets A.J.M., Van Kan J.A.L. Natural variation in virulence of the Entomopathogenic fungus Beauveria bassiana against malaria mosquitoes. Malaria J. 2014;13:479–487. doi: 10.1186/1475-2875-13-479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang S., Widemann E., Bernard G. CYP52X1, representing new cytochrome P450 subfamily, displays fatty acid hydroxylase activity and contributes to virulence and growth on insect cuticular substrates in entomopathogenic fungus Beauveria bassiana. J Biol Chem. 2012;287:13477–13486. doi: 10.1074/jbc.M111.338947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Luo X., Keyhani N.O., Yu X. The MAP kinase Bbslt2 controls growth, conidiation, cell wall integrity, and virulence in the insect pathogenic fungus Beauveria bassiana. Fungal Genet Biol. 2012;49:544–555. doi: 10.1016/j.fgb.2012.05.002. [DOI] [PubMed] [Google Scholar]
- 22.Zhang S., Xia Y., Keyhani N.O. Contribution of the gas1 gene of the entomopathogenic fungus Beauveria bassiana, encoding a putative glycosylphosphatidylinositol-anchored beta-1,3-glucanosyltransferase, to conidialthermotolerance and virulence. Appl Environ Microbiol. 2011;77:2676–2684. doi: 10.1128/AEM.02747-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Butt T.M., Goettel M.S. 2000 Bioessays of entomopathogenous fungi. In: Navon A., Ascher K.R.S., editors. Bioessays of Entomopathogenic Microbes and Nematodes. CAB International; Wallingford, Oxon, UK: 2000. pp. 141–191. [Google Scholar]
- 24.Wright M.S., Raina A.K., Lax A.R. A strain of the fungus Metarhizium anisopliae for controlling subterranean termites. J Econ Entomol. 2005;98:1451–1458. doi: 10.1603/0022-0493-98.5.1451. [DOI] [PubMed] [Google Scholar]
- 25.Tefera T., Pringle K.L. Mortality and maize leaf consumption of Chilopartellus (Lepidoptera: Pyralidae) larvae treated with Beauveriabassiana and MetarhiziumanisopliaeI. Int J Pest Manage. 2004;50(1):29–34. [Google Scholar]
- 26.Akbari S., Safavi S.A., Ghosta Y. Efficacy of Beauveria bassiana (Blas) Vuill against cabbage aphid Brevicoryne brassicae L (Hem: Aphididae) in laboratory condition. Arch Phytopathol Plant Prot. 2014;47(12):1454–1458. [Google Scholar]
- 27.Pelizza S.A., Eliades S.A., Scorsetti A.C., Cabello M.N. Entomopathogenic fungi from Argentina for the control of Schistocercacancellata (Orthoptera: Acrididae) nymphs: fungal pathogenicity and enzyme activity. Biocont Sci Technol. 2012;22:1119–1129. [Google Scholar]
- 28.Perinotto W.M.S., Golo P.S., Rodrigues C.J.B.C. Enzymatic activities and effects of mycovirus infection on the virulence of Metarhizium anisopliae in Rhipicephalus microplus. Veter Parasitol. 2014;203:189–196. doi: 10.1016/j.vetpar.2014.02.011. [DOI] [PubMed] [Google Scholar]
- 29.Mustafa U., Kaur G. Extracellular enzyme production in Metarhizium anisopliae isolates. Folia Microbiol. 2009;54(6):499–504. doi: 10.1007/s12223-009-0071-0. [DOI] [PubMed] [Google Scholar]
- 30.Bidochka M.J., Khachatourians G.G. Identification of Beauveriabassiana extracellular protease as a virulent factor in pathogenicity toward the migratory grasshopper Melanoplus sanguinipes. Entomol Exp Appl. 1990;45:151–156. [Google Scholar]
- 31.Ali S., Huang Z., Ren S.X. Production and extraction of extracellular lipase from the entomopathogenic fungus Isaria fumosoroseus (Cordycipitaceae: Hypocreales) Biocont Sci Technol. 2009;19:81–89. [Google Scholar]
- 32.Braga G.U.L., Destefano R.H.R., Messias C.L. Protease production during growth and autolysis of submerged Metarhizium anisopliae cultures. Rev Microbiol. 1999;30:107–113. [Google Scholar]
- 33.Zibaee A., Bandani A.R. Effects of Artemisia annua L. (Asteracea) on digestive enzymes profiles and cellular immune reactions of sunn pest, Eurygaster integriceps (Heteroptera: Scutellaridae), against Beauvaria bassiana. Bull Entomol Res. 2010;100:185–196. doi: 10.1017/S0007485309990149. [DOI] [PubMed] [Google Scholar]
- 34.Supakdamrongkul P., Bhumiratana A., Wiwat C. Characterization of an extracellular lipase from the biocontrol fungus, Nomuraea rileyi MJ, and its toxicity toward Spodoptera litura. J Invertebr Pathol. 2010;105:228–235. doi: 10.1016/j.jip.2010.06.011. [DOI] [PubMed] [Google Scholar]
- 35.Dhar P., Kaur G. Production of cuticle – degrading proteases by Beauveria bassiana and their induction in different media. African J Biochem. 2010;4(3):65–72. doi: 10.1007/s12088-011-0098-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fernandes E.G., Valerio H.M., Feltrin T., Sand S.T.V.D. Variability in the production of extracellular enzymes by entomopathogenic fungi grown on different substrates. Brazil J Microbiol. 2012:827–833. doi: 10.1590/S1517-83822012000200049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.St. Leger R.J., Charmley A.K., Cooper R.M. Production of polyphenol pigments and phenoloxidase by the entomopathogen, Metarhizium anisopliae. J Invertebr Pathol. 1988;53:211–215. [Google Scholar]
- 38.Silva W.O.B., Santi L., Schrank A., Vainstein M.H. Metarhizium anisopliae lipolytic activity plays a pivotal role in Rhipicephalus (Boophilus) microplus infection. Fungal Biol. 2010;114:10–15. doi: 10.1016/j.mycres.2009.08.003. [DOI] [PubMed] [Google Scholar]