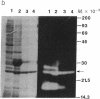
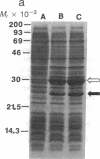
Abstract
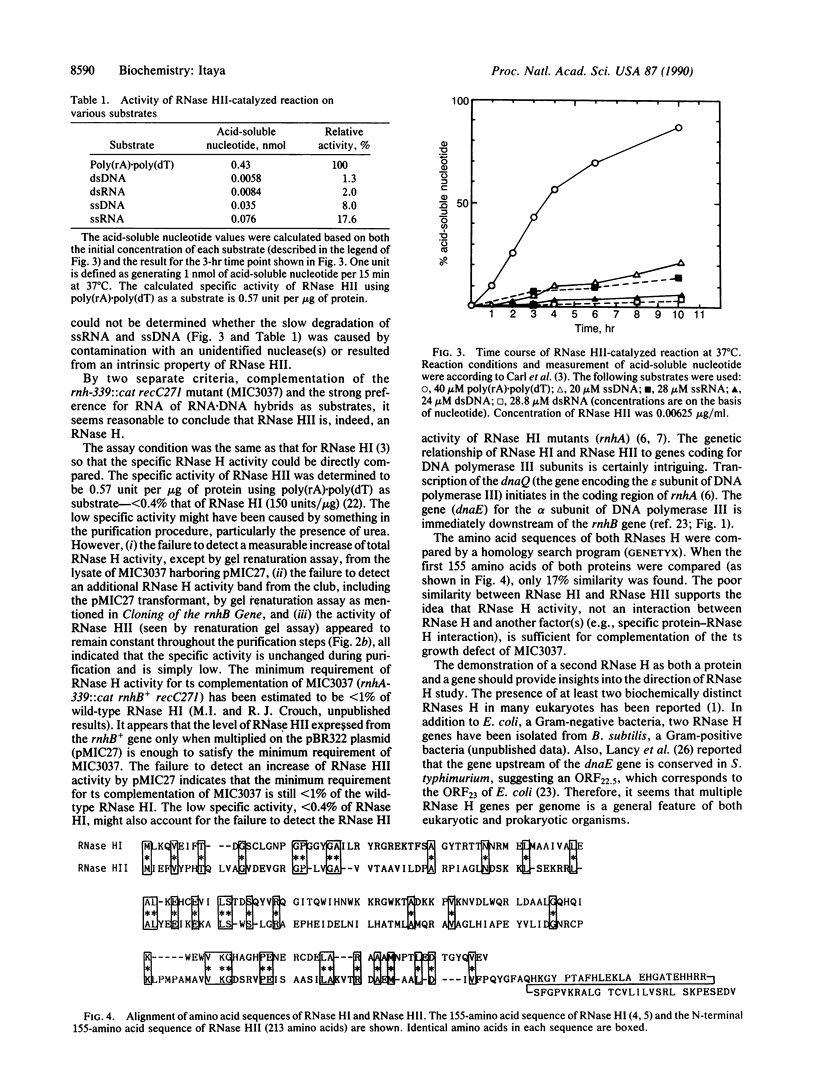
An additional RNase H (EC 3.1.26.4), RNase HII, has been isolated from Escherichia coli K-12. By screening a library of E. coli DNA for clones that suppressed RNase H deficiency of an E. coli rnh mutant, a clone was obtained that produced a protein with RNase H activity. The overexpressed RNase HIII protein in E. coli was purified to near homogeneity and exhibited a strong preference for the ribonucleotide moiety of RNA-DNA hybrid as substrate. The terminal 11 amino acids were determined and were identical to those predicted from the nucleotide sequence. The rnhB gene, which encodes RNase HII, was distinct from rnhA by its map position (4.5 min on E. coli genetic map, between lpxB and dnaE) and by the lack of significant amino acid sequence similarity. The presence of a second RNase H in E. coli indicates that multiple RNase H genes per genome is a general feature of a general feature of a wide variety of organisms.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bockrath R., Wolff L., Farr A., Crouch R. J. Amplified RNase H activity in Escherichia coli B/r increases sensitivity to ultraviolet radiation. Genetics. 1987 Jan;115(1):33–40. doi: 10.1093/genetics/115.1.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carl P. L., Bloom L., Crouch R. J. Isolation and mapping of a mutation in Escherichia coli with altered levels of ribonuclease H. J Bacteriol. 1980 Oct;144(1):28–35. doi: 10.1128/jb.144.1.28-35.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowell D. N., Reznikoff W. S., Raetz C. R. Nucleotide sequence of the Escherichia coli gene for lipid A disaccharide synthase. J Bacteriol. 1987 Dec;169(12):5727–5734. doi: 10.1128/jb.169.12.5727-5734.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dasgupta S., Masukata H., Tomizawa J. Multiple mechanisms for initiation of ColE1 DNA replication: DNA synthesis in the presence and absence of ribonuclease H. Cell. 1987 Dec 24;51(6):1113–1122. doi: 10.1016/0092-8674(87)90597-6. [DOI] [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene. 1984 Jun;28(3):351–359. doi: 10.1016/0378-1119(84)90153-7. [DOI] [PubMed] [Google Scholar]
- Horiuchi T., Maki H., Sekiguchi M. RNase H-defective mutants of Escherichia coli: a possible discriminatory role of RNase H in initiation of DNA replication. Mol Gen Genet. 1984;195(1-2):17–22. doi: 10.1007/BF00332717. [DOI] [PubMed] [Google Scholar]
- Itaya M., Yamaguchi I., Kobayashi K., Endo T., Tanaka T. The blasticidin S resistance gene (bsr) selectable in a single copy state in the Bacillus subtilis chromosome. J Biochem. 1990 Jun;107(6):799–801. doi: 10.1093/oxfordjournals.jbchem.a123128. [DOI] [PubMed] [Google Scholar]
- Itoh T., Tomizawa J. Formation of an RNA primer for initiation of replication of ColE1 DNA by ribonuclease H. Proc Natl Acad Sci U S A. 1980 May;77(5):2450–2454. doi: 10.1073/pnas.77.5.2450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanaya S., Crouch R. J. DNA sequence of the gene coding for Escherichia coli ribonuclease H. J Biol Chem. 1983 Jan 25;258(2):1276–1281. [PubMed] [Google Scholar]
- Kanaya S., Kohara A., Miyagawa M., Matsuzaki T., Morikawa K., Ikehara M. Overproduction and preliminary crystallographic study of ribonuclease H from Escherichia coli. J Biol Chem. 1989 Jul 15;264(20):11546–11549. [PubMed] [Google Scholar]
- Keller W., Crouch R. Degradation of DNA RNA hybrids by ribonuclease H and DNA polymerases of cellular and viral origin. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3360–3364. doi: 10.1073/pnas.69.11.3360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kogoma T., Subia N. L., von Meyenburg K. Function of ribonuclease H in initiation of DNA replication in Escherichia coli K-12. Mol Gen Genet. 1985;200(1):103–109. doi: 10.1007/BF00383320. [DOI] [PubMed] [Google Scholar]
- Kohara Y., Akiyama K., Isono K. The physical map of the whole E. coli chromosome: application of a new strategy for rapid analysis and sorting of a large genomic library. Cell. 1987 Jul 31;50(3):495–508. doi: 10.1016/0092-8674(87)90503-4. [DOI] [PubMed] [Google Scholar]
- Lancy E. D., Lifsics M. R., Munson P., Maurer R. Nucleotide sequences of dnaE, the gene for the polymerase subunit of DNA polymerase III in Salmonella typhimurium, and a variant that facilitates growth in the absence of another polymerase subunit. J Bacteriol. 1989 Oct;171(10):5581–5586. doi: 10.1128/jb.171.10.5581-5586.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindahl G., Lindahl T. Initiation of DNA replication in Escherichia coli: RNase H-deficient mutants do not require the dnaA function. Mol Gen Genet. 1984;196(2):283–289. doi: 10.1007/BF00328061. [DOI] [PubMed] [Google Scholar]
- Maki H., Horiuchi T., Sekiguchi M. Structure and expression of the dnaQ mutator and the RNase H genes of Escherichia coli: overlap of the promoter regions. Proc Natl Acad Sci U S A. 1983 Dec;80(23):7137–7141. doi: 10.1073/pnas.80.23.7137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller H. I., Riggs A. D., Gill G. N. Ribonuclease H (hybrid) in Escherichia coli. Identification and characterization. J Biol Chem. 1973 Apr 10;248(7):2621–2624. [PubMed] [Google Scholar]
- Ogawa T., Okazaki T. Function of RNase H in DNA replication revealed by RNase H defective mutants of Escherichia coli. Mol Gen Genet. 1984;193(2):231–237. doi: 10.1007/BF00330673. [DOI] [PubMed] [Google Scholar]
- Ogawa T., Pickett G. G., Kogoma T., Kornberg A. RNase H confers specificity in the dnaA-dependent initiation of replication at the unique origin of the Escherichia coli chromosome in vivo and in vitro. Proc Natl Acad Sci U S A. 1984 Feb;81(4):1040–1044. doi: 10.1073/pnas.81.4.1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudd K. E., Miller W., Ostell J., Benson D. A. Alignment of Escherichia coli K12 DNA sequences to a genomic restriction map. Nucleic Acids Res. 1990 Jan 25;18(2):313–321. doi: 10.1093/nar/18.2.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Studier F. W., Moffatt B. A. Use of bacteriophage T7 RNA polymerase to direct selective high-level expression of cloned genes. J Mol Biol. 1986 May 5;189(1):113–130. doi: 10.1016/0022-2836(86)90385-2. [DOI] [PubMed] [Google Scholar]
- Tanaka T., Kawata M. Cloning and characterization of Bacillus subtilis iep, which has positive and negative effects on production of extracellular proteases. J Bacteriol. 1988 Aug;170(8):3593–3600. doi: 10.1128/jb.170.8.3593-3600.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomasiewicz H. G., McHenry C. S. Sequence analysis of the Escherichia coli dnaE gene. J Bacteriol. 1987 Dec;169(12):5735–5744. doi: 10.1128/jb.169.12.5735-5744.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]