Abstract
Background
Psoriasis is an immune-mediated, chronic inflammatory disease affecting multiple aspects of patients' lives. Its epidemiology varies regionally; however, nationwide epidemiologic data on psoriasis depicting profile of Korean patients has not been available to date.
Objective
To understand nationwide epidemiologic characteristics and clinical features of adult patients with psoriasis visited university hospitals in Korea.
Methods
This multicenter, non-interventional, cross-sectional study recruited 1,278 adult patients with psoriasis across 25 centers in Korea in 2013. Various clinical data including PASI, BSA, DLQI, SF-36 and PASE were collected.
Results
A total of 1,260 patients completed the study (male:female=1.47:1). The mean age was 47.0 years with a distribution mostly in the 50s (24.9%). Early onset (<40 years) of psoriasis accounted for 53.9% of patients. The mean disease duration was 109.2 months; mean body mass index was 23.9 kg/m2; and 12.7% of patients had a family history of psoriasis. Plaque and guttate types of psoriasis accounted for 85.8% and 8.4%, respectively. Patients with PASI ≥10 accounted for 24.9%; patients with body surface area ≥10 were 45.9%. Patients with DLQI ≥6 accounted for 78.8%. Between PASI <10 and PASI ≥10 groups, significant difference was noted in age at diagnosis, disease duration, blood pressure, waist circumference of female, and treatment experiences with phototherapy, systemic agents, and biologics.
Conclusion
This was the first nationwide epidemiologic study of patients with psoriasis in Korea and provides an overview of the epidemiologic characteristics and clinical profiles of this patient population.
Keywords: Epidemiologic studies, Korea, Psoriasis
INTRODUCTION
Psoriasis is an immune-mediated, chronic inflammatory disease affecting various aspects of patients' lives far beyond the skin and challenging clinicians in its management1. While psoriasis is prevailing worldwide, there are regional differences in its epidemiological characteristics. Local differences in genetic, environmental, cultural, and geographic characteristics may be related. Although outstanding progress have achieved in various fields of psoriasis research, understanding epidemiologic profiles of the disease is still important. A more elaborate understanding of regional epidemiology is essential to refining the management of psoriasis. Unlike many western countries, where data from many large-scale epidemiologic studies are available, epidemiologic study data of nationwide scale were unavailable in Korea. In 2013, the Korean Society for Psoriasis (KSP) conducted a cross-sectional, multicenter study to delineate the clinical profile of adult patients with psoriasis visiting university hospitals in Korea. Here we report the results of this study.
MATERIALS AND METHODS
Study design
This was a multicenter, non-interventional, cross-sectional study. Patients were recruited for approximately 4 months, from February to June 2013. The 25 centers participating in the study were mainly university hospitals, located across the country (with the exception of the Jeju island region). This study was reviewed and approved by the Institutional Review Board of each center. To extract a representative sample reflecting the distribution of psoriasis in Korea, numbers and locations of study centers were selected on the basis of population distribution and health insurance reimbursement data of the patients with psoriasis in Korea.
Because half of the total population of Korea lives near the capital, 13 of 25 centers were selected from the capital area. To avoid selection bias, study patients were examined and enrolled consecutively by visiting orders. After obtaining informed consent, all procedures of the study were performed on 1 day. The study patients were adult patients aged 20 or older diagnosed with psoriasis.
Clinical data were collected by interviewing the patients on demographic factors (age, sex, height, weight, waist circumference, disease duration, age at diagnosis, family history in first-degree relative, drinking/smoking history, and both past and ongoing concomitant therapies). Physical examination for disease-severity measures (psoriasis area and severity index [PASI], body surface area [BSA]), morphological characteristics (clinical types and location) of psoriasis and blood pressure were carried out for all enrolled patients. Quality of life was evaluated by the dermatology life quality index (DLQI) and MOS 36-item short-form health survey (SF-36). Screening for psoriatic arthritis was done using the psoriatic arthritis screening and evaluation (PASE) tool. In addition, data on treatment-related factors (past and current treatment history, patient satisfaction for treatment) were collected. In order to evaluate patient satisfaction for various therapies, questions from the Medication Satisfaction Questionnaire (MSQ)2, which is a validated tool for evaluating treatment satisfaction among patients with schizophrenia, were used in this study. Patients scored their response to the question, “In general, how satisfied are you with your current treatment?” on a 7-point scale ranging from 1 to 7 (a score of 4 means neutral).
Statistical analysis
Descriptive statistics for continuous variables are presented as means with standard deviation, and dichotomous variables are presented as frequencies with percentages in parentheses. Types of psoriasis, location of involved lesions, and clinical/demographic characteristics were compared to assess the statistical significance of any differences among the groups by disease severity (PASI <10, PASI ≥10). We used, where appropriate, the Wilcoxon rank-sum and t tests for continuous variables, and analyses for dichotomous variables that were based on Pearson's chi-square test and Fisher's exact test, without missing data imputation. In addition, a multiple logistic regression analysis was used to assess the adjusted effect of characteristics on PASI ≥10. Multicollinearity between independent variables had been checked by computing correlation coefficient and variance inflation factors (VIF) before the variables to be included in the regression model were selected. All statistical tests were performed using two-sided and p-values less than 0.05 were considered statistically significant. Analyses were performed using the statistical software package SAS 9.2 (SAS Institute, Cary, NC, USA).
RESULTS
Demographics
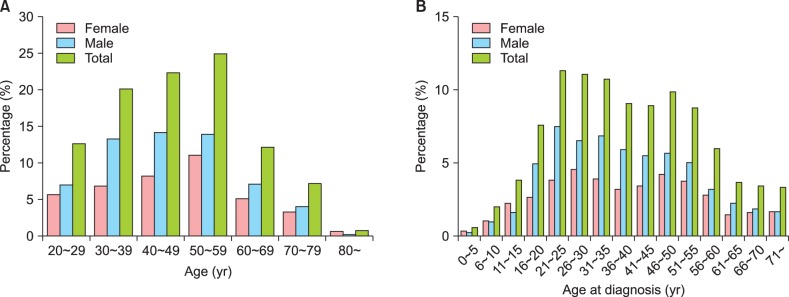
A total of 1,278 patients were enrolled in the study, and 1,260 (98.6%) completed the study. Eighteen patients (1.4%) were excluded from analysis, 16 (1.3%) for violation of the inclusion/exclusion criteria, and two (0.2%) for lack of data for the primary assessment. Demographic data from the 1,260 patients are presented in Table 1. A total of 749 patients (59.4%) were male and 511 (40.6%) were female; the male to female ratio was 1.47:1. The mean age of the study patients was 47.0±14.5 years (range, 20~89 years). Most study patients were in their 50s (24.9%), with a gradual increase from 20s to 50s and a sharp decrease after 60s. The distribution pattern of age at diagnosis revealed dual peaks at 21~25 years and 46~50 years (Fig. 1). A ratio of early onset (<40 years) versus late onset (≥40 years) at diagnosis was 53.9% versus 46.1% (33.8% versus 66.2% when divided by age of 30 years old). The mean disease duration was 109.2±122.0 months. Mean height and weight were 166.5±8.4 cm and 66.4±12.4 kg, respectively, and mean body mass index (BMI) was 23.9±3.5 kg/m2. At the time of the study, 48.3% (n=609) of the total patients had a history of smoking and 64.0% (n=806) had a history of drinking. Male showed higher rate in smoking/drinking history than female. Patients who had a family history of psoriasis in a first-degree relative accounted for 12.7% (n=160) of the total.
Table 1. Demographics.
| Characteristics | Total | Overall | PASI<10 | PASI≥10 | p-value |
|---|---|---|---|---|---|
| Gender | 1,260 | 0.215 | |||
| Male | 749 (59.4) | 553 (58.5) | 196 (62.4) | ||
| Female | 511 (40.6) | 393 (41.5) | 118 (37.6) | ||
| Age (yr) | 1,260 | 47.0±14.5 | 47.3±14.3 | 46.3±15.0 | 0.310 |
| Age at diagnosis (yr) | 1,257 | 38.6±16.5 | 39.3±16.2 | 36.4±17.0 | 0.008* |
| <40 | 677 (53.9) | 492 (52.1) | 185 (59.1) | 0.032* | |
| ≥40 | 580 (46.1) | 452 (47.9) | 128 (40.9) | ||
| Disease duration (mo) | 1,257 | 109.2±122.0 | 103.3±118.2 | 126.9±131.5 | 0.003* |
| Family history | 1,260 | 160 (12.7) | 128 (13.5) | 32 (10.2) | 0.124 |
| Height (cm) | 1,259 | 166.5±8.4 | 166.5±8.5 | 166.5±8.3 | 0.925 |
| Weight (kg) | 1,259 | 66.4±12.4 | 66.1±12.2 | 67.4±12.9 | 0.116 |
| BMI (kg/m2) | 1,259 | 23.9±3.5 | 23.8±3.4 | 24.2±3.8 | 0.059 |
| Male | 748 | 24.5±3.2 | 24.5±3.1 | 24.6±3.5 | 0.699 |
| Female | 511 | 23.0±3.7 | 22.8±3.5 | 23.6±4.2 | 0.046* |
| Waist circumference (cm) | 1,113 | 84.3±11.1 | 83.8±11.0 | 85.6±11.4 | 0.022* |
| Male (≥90 cm) | 659 | 254 (38.5) | 193 (38.6) | 61 (38.4) | 0.958 |
| Female (≥80 cm) | 454 | 199 (43.8) | 138 (40.0) | 61 (56.0) | 0.003* |
| SBP ≥140 mmHg or DBP ≥90 mmHg | 1,164 | 231 (19.8) | 158 (17.8) | 73 (26.5) | 0.001* |
| Smoking history | 1,260 | 609 (48.3) | 449 (47.5) | 160 (51.0) | 0.283 |
| Male | 749 | 538 (71.8) | 394 (71.2) | 144 (73.5) | 0.552 |
| Female | 511 | 71 (13.9) | 55 (14.0) | 16 (13.6) | 0.905 |
| Drinking history | 1,260 | 806 (64.0) | 591 (62.5) | 215 (68.5) | 0.055 |
| Male | 749 | 595 (79.4) | 431 (77.9) | 164 (83.7) | 0.088 |
| Female | 511 | 211 (41.3) | 160 (40.7) | 51 (43.2) | 0.628 |
Values are presented as total number only, number (%), or mean±standard deviation. Total number does not always represent 100% of the study population because of missing values. Percentages of subgroup (by gender) were based on the total number of subjects in each PASI group per subgroup. PASI: psoriasis area and severity index, BMI: body mass index, SBP: systolic blood pressure, DBP: diastolic blood pressure. *p-value<0.05.
Fig. 1. Age distribution of patient (A) and distribution of age at diagnosis (B).
Clinical type of psoriasis
Plaque-type psoriasis was the most prevalent, occurring in 1,081 patients (85.8%), followed by guttate type in 106 (8.4%), pustular type (including localized form) in 66 (5.2%), erythrodermic type in 44 (3.5%), annular type in 24 (1.9%), and inverse type in 4 (0.3%) (Table 2).
Table 2. Types of psoriasis.
| Classification | Overall (n=1,260) |
|---|---|
| Plaque | 1,081 (85.8) |
| Guttate | 106 (8.4) |
| Pustular | 66 (5.2) |
| Erythrodermic | 44 (3.5) |
| Annular | 24 (1.9) |
| Inverse | 4 (0.3) |
Values are presented as number (%).
Location of lesions
Psoriasis lesions were most frequently found in the lower leg (72.6%, n=913), followed by the back (60.9%, n=766), upper leg (60.8%, n=764), elbow (58.0%, n=729), knee (57.1%, n=718), lower arm (56.8%, n=714), abdomen (55.7%, n=700), upper arm (53.5%, n=672), and scalp (53.1%, n=668). Nail involvement was noted in 12.3% of patients and genitalia involvement in 8.8% of patients. Details of lesion location are listed in Table 3.
Table 3. Location of involved lesions.
| Classification | Overall (n=1,257)* | PASI<10 (n=943) | PASI≥10 (n=314) |
|---|---|---|---|
| Scalp | 668 (53.1) | 453 (48.0) | 215 (68.5) |
| Face | 349 (27.8) | 218 (23.1) | 131 (41.7) |
| Neck | 205 (16.3) | 104 (11.0) | 101 (32.2) |
| Chest | 536 (42.6) | 334 (35.4) | 202 (64.3) |
| Abdomen | 700 (55.7) | 460 (48.8) | 240 (76.4) |
| Back | 766 (60.9) | 497 (52.7) | 269 (85.7) |
| Upper arm | 672 (53.5) | 425 (45.1) | 247 (78.7) |
| Elbow | 729 (58.0) | 484 (51.3) | 245 (78.0) |
| Lower arm | 714 (56.8) | 462 (49.0) | 252 (80.3) |
| Palm† | 164 (13.0) | 115 (12.2) | 49 (15.6) |
| Back of hand | 295 (23.5) | 178 (18.9) | 117 (37.3) |
| Nail | 155 (12.3) | 89 (9.4) | 66 (21.0) |
| Upper leg | 764 (60.8) | 499 (52.9) | 265 (84.4) |
| Knee | 718 (57.1) | 472 (50.1) | 246 (78.3) |
| Lower leg | 913 (72.6) | 620 (65.7) | 293 (93.3) |
| Foot sole† | 146 (11.6) | 103 (10.9) | 43 (13.7) |
| Back of foot | 217 (17.3) | 127 (13.5) | 90 (28.7) |
| Buttock | 471 (37.5) | 280 (29.7) | 191 (60.8) |
| Genitalia | 111 (8.8) | 59 (6.3) | 52 (16.6) |
| Other area† | 8 (0.6) | 4 (0.4) | 4 (1.3) |
Values are presented as number (%). PASI: psoriasis area and severity index. *3 patients are missing. †No significant p-value (other involved areas showed p-value<0.0001).
Disease severity
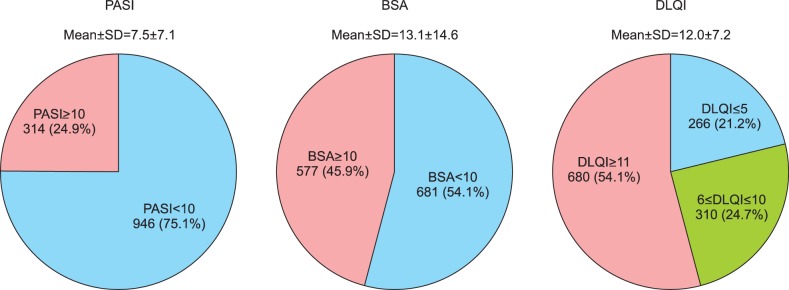
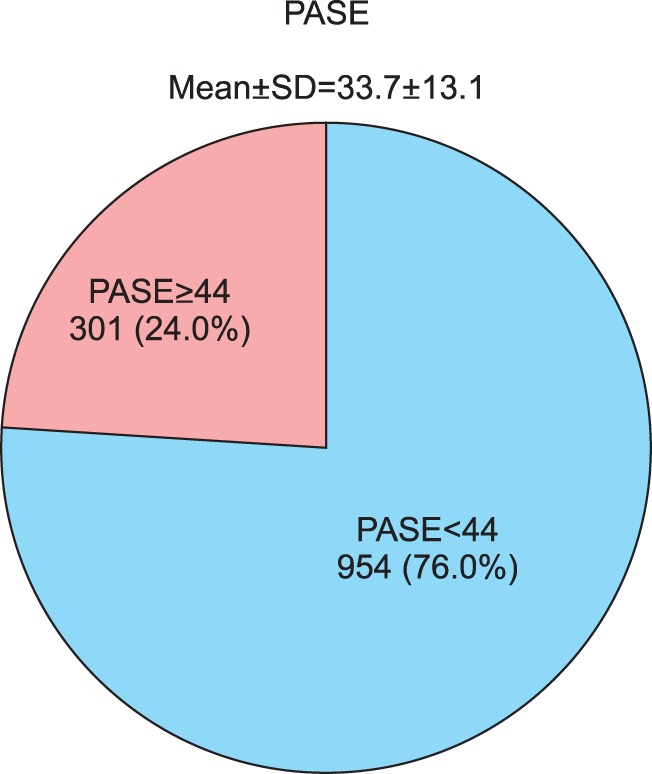
Of the total study patients, 75.1% (n=946) had PASI <10 and 24.9% (n=314) scored PASI ≥10 (19.0% had 10≤PASI<20, 5.9% had PASI ≥20). The mean PASI score was 7.5±7.1. The mean BSA (%) measurement was 13.1±14.6. 54.1% (n=681) of the total patients had BSA <10, 45.9% (n=577) had BSA ≥10 (14.3% had BSA <3%, 39.8% had 3≤BSA<10, 25.4% had 10≤BSA<20, and 20.4% had BSA ≥20). On the other hand, with regards to the DLQI, results showed that of the 1,256 study patients whose DLQI data were available, 21.2% (n=266) scored DLQI ≤5 (no/small effect), 24.7% (n=310) scored 6≤DLQI≤10 (moderate effect), and 54.1% (n=680) scored DLQI ≥11 (very/extremely large effect). The mean value of DLQI was 12.0±7.2 (Fig. 2). The mean SF-36 results were 48.8±8.0 for the physical component summary score and 42.6±11.2 for the mental component summary score (data not shown). PASE data were available for 1,255 patients. The mean PASE score was 33.7±13.1; 301 (24.0%) patients had a PASE score of 44 points or higher (Fig. 3).
Fig. 2. Disease severity classified by psoriasis area and severity index (PASI), body surface area (BSA), and dermatology life quality index (DLQI). Overall mean±standard deviation (SD) are presented below the title. Each n (%) of the classified group is presented.
Fig. 3. Psoriatic arthritis screening and evaluation (PASE). SD: standard deviation.
Past and concomitant therapies
Of all past therapies utilized by the study patients (n=1,260), topical agents were the most common (93.6%, n=1,179), followed by systemic agents (61.2%, n=771), phototherapy (50.8%, n=640), and biologics (5.7%, n=72). The order of current therapy maintained at the time of enrollment was topical agent (86.0%, n=1,083), oral systemic agents (45.9%, n=578), phototherapy (30.6%, n=385), and biologics (4.6%, n=58). Although herbal medicine was used at some point by a considerable number of patients, it was seldom maintained (Table 4).
Table 4. Past and current therapies.
| Category | Overall (n=1,260) | PASI<10 (n=946) | PASI≥10 (n=314) | p-value | |
|---|---|---|---|---|---|
| Past and current | Current only | ||||
| Topical | 1,179 (93.6) | 1,083 (86.0) | 884 (93.4) | 295 (93.9) | 0.753 |
| Glucocorticosteroid | 937 (74.4) | 774 (61.4) | 697 (73.7) | 240 (76.4) | 0.333 |
| Calcipotriol/tacalcitol | 986 (78.3) | 893 (70.9) | 746 (78.9) | 240 (76.4) | 0.367 |
| Phototherapy | 640 (50.8) | 385 (30.6) | 463 (48.9) | 177 (56.4) | 0.023* |
| UVA/UVB | 35 (2.8) | 19 (1.5) | 32 (3.4) | 3 (1.0) | 0.023* |
| Narrowband UVB | 564 (44.8) | 338 (26.8) | 404 (42.7) | 160 (51.0) | 0.011* |
| Broadspectrum UVB | 10 (0.8) | 6 (0.5) | 7 (0.7) | 3 (1.0) | 0.717 |
| PUVA (systemic) | 42 (3.3) | 26 (2.1) | 31 (3.3) | 11 (3.5) | 0.847 |
| Systemic agents | 771 (61.2) | 578 (45.9) | 561 (59.3) | 210 (66.9) | 0.017* |
| Fumaric acid esters | 4 (0.3) | 0 (0.0) | 2 (0.2) | 2 (0.6) | 0.260 |
| Methotrexate | 280 (22.2) | 174 (13.8) | 205 (21.7) | 75 (23.9) | 0.413 |
| Acitretin | 300 (23.8) | 185 (14.7) | 206 (21.8) | 94 (29.9) | 0.003* |
| Cyclosporine | 366 (29.1) | 221 (17.5) | 253 (26.7) | 113 (36.0) | 0.002* |
| Oral corticosteroid | 100 (7.9) | 26 (2.1) | 79 (8.4) | 21 (6.7) | 0.345 |
| Biologics | 72 (5.7) | 58 (4.6) | 45 (4.8) | 27 (8.6) | 0.011* |
| Adalimumab | 6 (0.5) | 4 (0.3) | 5 (0.5) | 1 (0.3) | 1.000 |
| Etanercept | 13 (1.0) | 2 (0.2) | 7 (0.7) | 6 (1.9) | 0.102 |
| Infliximab | 7 (0.6) | 5 (0.4) | 5 (0.5) | 2 (0.6) | 0.687 |
| Ustekinumab | 53 (4.2) | 47 (3.7) | 32 (3.4) | 21 (6.7) | 0.012* |
| Others | 181 (14.4) | 42 (3.3) | 137 (14.5) | 44 (14.0) | 0.837 |
| Diet therapy | 28 (2.2) | 19 (1.5) | 27 (2.9) | 1 (0.3) | 0.008* |
| Herbal medicine | 106 (8.4) | 9 (0.7) | 81 (8.6) | 25 (8.0) | 0.740 |
| Exercise therapy | 8 (0.6) | 8 (0.6) | 7 (0.7) | 1 (0.3) | 0.688 |
| Spa | 14 (1.1) | 11 (0.9) | 11 (1.2) | 3 (1.0) | 1.000 |
| Supplements | 13 (1.0) | 9 (0.7) | 10 (1.1) | 3 (1.0) | 1.000 |
| None | 19 (1.5) | - | 18 (1.9) | 1 (0.3) | 0.058 |
Values are presented as number (%). PASI: psoriasis area and severity index, UV: ultraviolet, PUVA: psoralen and ultraviolet A. *p-value<0.05.
Comparison of factors related to disease based on disease severity
The patients were divided into two groups of PASI <10 and PASI ≥10 and factors that could be related with disease severity were compared. Gender, age, family history, height and weight, smoking/drinking history, and male gender's waist circumference and BMI did not showed any significant difference between two groups. However, there was significant difference between two groups regarding BMI in female, age at diagnosis, disease duration, waist circumference of female, blood pressure. The group with PASI ≥10 had longer disease duration (p=0.003) and earlier age of diagnosis (p=0.008) than the group with PASI <10 and comprised a significantly higher proportion of patients with systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg (p=0.001). BMI in female was significantly higher (p=0.046) and the proportion of waist circumference with ≥80 cm in female was higher in PASI ≥10 group (p=0.003, Table 1). Most of body parts were involved more as disease severity increased except for palm, sole (Table 3). After adjusting for variables in demographic characteristics except age at diagnosis, height and weight due to multicollinearity, younger age, longer disease duration, absence of family history, longer waist circumference and higher blood pressure were significantly associated with severe disease (PASI ≥10, Table 5). More patients with PASI ≥10 had experienced treatment with phototherapy, systemic agents, and biologics compared to patients with PASI <10 (Table 4).
Table 5. Adjusted effect of characteristics on PASI ≥10 based on multiple logistic regression model (n=1,086).
| Characteristics | Ref. | OR | 95% CI | p-value |
|---|---|---|---|---|
| Gender | Male | 1.182 | 0.810~1.725 | 0.386 |
| Age | 0.987 | 0.977~0.998 | 0.020* | |
| Disease duration | 1.002 | 1.001~1.003 | 0.002* | |
| Family history | No | 0.571 | 0.361~0.905 | 0.017* |
| BMI | 0.992 | 0.943~1.044 | 0.763 | |
| Waist circumference | 1.018 | 1.001~1.036 | 0.036* | |
| SBP ≥140 mmHg or DBP ≥90 mmHg | No | 1.535 | 1.090~2.162 | 0.014* |
| Smoking history | No | 1.006 | 0.705~1.435 | 0.975 |
| Drinking history | No | 1.061 | 0.758~1.485 | 0.731 |
One hundred seventy-four patients with missing data were excluded from the logistic regression analysis. PASI: psoriasis area and severity index, Ref.: reference for odds ratio, OR: odds ratio, CI: confidence interval, BMI: body mass index, SBP: systolic blood pressure, DBP: diastolic blood pressure. *p-value<0.05.
Treatment satisfaction
Patients were divided into three treatment satisfaction groups: dissatisfied (from extremely dissatisfied to somewhat dissatisfied), neutral, or satisfied (from somewhat satisfied to extremely satisfied). The satisfied group included 733 patients (58.5%); neutral group, 317 (25.3%); and dissatisfied group, 202 (16.1%). In the PASI <10 group, satisfied proportion was significantly higher than the PASI ≥10 group (p=0.002, Table 6).
Table 6. Comparing the degree of satisfaction with treatment.
| Category | Overall (n=1,252)* | PASI<10 (n=939) | PASI≥10 (n=313) | p-value |
|---|---|---|---|---|
| Degree of satisfaction | 0.002† | |||
| Extremely satisfied | 42 (3.4) | 34 (3.6) | 8 (2.6) | |
| Very satisfied | 229 (18.3) | 192 (20.4) | 37 (11.8) | |
| Somewhat satisfied | 462 (36.9) | 350 (37.3) | 112 (35.8) | |
| Neutral | 317 (25.3) | 226 (24.1) | 91 (29.1) | |
| Somewhat dissatisfied | 125 (10.0) | 89 (9.5) | 36 (11.5) | |
| Very dissatisfied | 58 (4.6) | 36 (3.8) | 22 (7.0) | |
| Extremely dissatisfied | 19 (1.5) | 12 (1.3) | 7 (2.2) |
Values are presented as number (%). PASI: psoriasis area and severity index. *8 patients are missing. †p-value<0.05.
DISCUSSION
Prior to this study, epidemiologic data for the patients with psoriasis in Korea were limited to small-scale or single-center studies using accumulated patient pools over long periods of time3. For this reason, it was difficult to obtain an overview of nationwide epidemiologic profile of psoriasis that is necessary for further research and better policy making for the patients with psoriasis in Korea. Hence, the KSP designed and conducted a nationwide study in 2013 to delineate the epidemiologic profile of Korean patients with psoriasis. To minimize regional sampling bias and improve temporal focusing, we matched the number and location of study centers with general population distribution data. As a result, the distributed proportion of recruited patients was very similar to that of the distributed proportion of claims for psoriasis in the Health Insurance Review and Assessment (HIRA) database of 2013.
The mean age of patients in our study (47.0 years) was higher than that observed in hospital based Japanese studies (45.5 years4 and 44.7 years5) as well as a Chinese population-based study (41.8 years6), but was similar to a Taiwanese cohort study (46.4 years7). Unlike classical bimodal peaks in distribution patterns of age at diagnosis in western studies, which showed peaks located in the late teens and late 50s, Asian studies4,6, including ours, showed dual peaks located in the 20s and 40s. Additionally, early-onset (type 1, <40 years) psoriasis accounted for more than 75% of patients in western studies8 and 67.6% of patients in the Chinese study6, but only 53.9% in our study.
The prevalence of psoriasis has been reported to be equal between men and women in western studies9,10,11. However, in Asian studies4,5,7,12, male predominance has been reported repeatedly. Our study also revealed the predominance of this disease in males (male to female ratio 1.47:1). This tendency was also noted in data from the Korean health insurance database in 2012 (1.36:1). But the degree of male predominance was somewhat lower than studies from other Asian countries such as Japan and Taiwan, which reported a male to female ratio of 1.6~2.1:15,7. Although this phenomenon may be unique to the study population, the cause of this interesting observation needs to be elucidated.
Although it is well known that obesity is closely related to psoriasis in western populations8,13,14, the mean BMI of 23.9±3.5 kg/m2 noted in our study was considerably lower than the mean BMI of 28.0 kg/m2 observed in western populations13. In addition, Kim and Lee15 reported no statistical difference in BMI between the patients with psoriasis and a control group in one study of a Korean population. This implies that further research is necessary to clarify the association between obesity and psoriasis in the Korean population.
Plaque-type psoriasis was the predominant clinical type in Korea, consistent with observations in Caucasian and other Asian populations5,16,17,18. Interestingly, although the proportion of plaque psoriasis is similar, the proportion of guttate psoriasis is relatively high in Korean patients compared with Japanese patients (8.4% versus 2.8%/3.9%)4,5. It was difficult to compare the involved site with other published studies, as each uses different criteria to distinguish the location in the body. For future studies, it is necessary to define the boundary of anatomical regions in a standardized manner and use common protocol to express involved areas. Hernánz et al.19 reported that patients with more severe psoriasis had greater skin involvement in most of area including the palms and soles. However, our data showed no significant differences in the involvement of palms and soles by disease severity divided by PASI score 10.
When comparing disease severity as measured by PASI and BSA with quality of life as measured by DLQI, it was found that the impact of psoriasis on quality of life was not fully explained by the severity or extent of skin disease. Therefore, an independent assessment of quality of life should be considered in the management of psoriasis.
Some studies20,21 reported a higher disease severity in male patients than in female patients. Although there was a higher proportion of males in the severe group, it was not statistically significant in our study (p=0.215).
Smoking has been known to be associated with the clinical severity of psoriasis22 but the ratio of smoking history between two groups by disease severity did not differ statistically. Some studies have reported that patients with more severe disease had an earlier age of onset and a longer disease duration19,23. Those results are in line with our findings. The more severe group presented with significantly higher blood pressure, as reported by Cohen et al.24.
Interestingly, the patients with family history showed significantly milder disease severity than the patients without family history after adjusting for relevant demographic characteristics. Although there are some studies25,26 that family history is associated with early onset age, but it was difficult to find a study to explain a relationship between family history and disease severity. However, López-Estebaranz et al.27 recently presented that the severity of psoriasis was not affected by the presence of a family history. Therefore, further studies are necessary to determine the association between family history and disease severity.
Finally, it was remarkable that significantly more patients in the PASI ≥10 group were dissatisfied with treatment results than the PASI <10 group. This result indicates that more effective and satisfactory treatment is needed for patients with PASI ≥10.
Although we attempted to minimize the bias from regional and temporal factors and reveal multilateral aspects of the clinical profiles of Korean patients with psoriasis, due to the recruitment of adult patients mostly from university hospitals, the representation of the general psoriasis population in Korea is limited. Further, because this study was cross-sectional, it is necessary to use caution when interpreting results for causal relationships between disease-related factors and outcomes over time. Therefore, additional research in the form of prospective longitudinal cohort studies is needed to further understanding of psoriasis in the Korean population.
ACKNOWLEDGMENT
This study was sponsored by Janssen Korea Ltd.
Footnotes
CONFLICTS OF INTEREST: Dr. Hae Jun Song has participated as an advisory board member and has conducted clinical trials for Janssen and has received speaking fees or research support from Janssen. Dr. Chul Jong Park, Dr. Tae Yoon Kim, Dr. Yong Beom Choe, Dr. Seok-Jong Lee, Dr. Nack In Kim, Dr. Jae We Cho, Dr. Jie Hyun Jeon, Dr. Min Soo Jang have no conflict of interest to declare. Dr. Jai Il Youn has conducted clinical trial for Pfizer, Janssen, Lilly, Leo. Dr. Myung Hwa Kim, Dr. Joonsoo Park, Dr. Ki Ho Kim, Dr. Byung Soo Kim have no conflict of interest to declare. Dr. Sang Woong Youn has served as an advisor, received speaker honoraria, and participated in clinical trials for Janssen. Dr. Joo-Heung Lee has no conflict of interest to declare. Dr. Min-Geol Lee has conducted clinical trials for Pfizer, Novartis, Eli Lilly, and Janssen and has received speaking fees from Janssen. Dr. Sung Ku Ahn, Dr. Young Ho Won, Dr. Seok Kweon Yun, Dr. Bong Seok Shin have no conflict of interest to declare. Dr. Seong Jun Seo has conducted clinical trials for Amore, Regeneron, Novartis, Roche. Dr. Ji Yeoun Lee, Dr. Kwang Joong Kim have no conflict of interest to declare. Dr. Young Suck Ro has conducted post-marketing surveillance for Janssen. Mr. Dae Young Yu and Mr. Youngdoe Kim are employee of Janssen Korea. Dr. Jee-Ho Choi has conducted clinical trials for Janssen.
References
- 1.Schön MP, Boehncke WH. Psoriasis. N Engl J Med. 2005;352:1899–1912. doi: 10.1056/NEJMra041320. [DOI] [PubMed] [Google Scholar]
- 2.Vernon MK, Revicki DA, Awad AG, Dirani R, Panish J, Canuso CM, et al. Psychometric evaluation of the Medication Satisfaction Questionnaire (MSQ) to assess satisfaction with antipsychotic medication among schizophrenia patients. Schizophr Res. 2010;118:271–278. doi: 10.1016/j.schres.2010.01.021. [DOI] [PubMed] [Google Scholar]
- 3.Na SJ, Jo SJ, Youn JI. Clinical study on psoriasis patients for past 30 years (1982-2012) in Seoul National University Hospital Psoriasis Clinic. J Dermatol. 2013;40:731–735. doi: 10.1111/1346-8138.12224. [DOI] [PubMed] [Google Scholar]
- 4.Kawada A, Tezuka T, Nakamizo Y, Kimura H, Nakagawa H, Ohkido M, et al. A survey of psoriasis patients in Japan from 1982 to 2001. J Dermatol Sci. 2003;31:59–64. doi: 10.1016/s0923-1811(02)00142-1. [DOI] [PubMed] [Google Scholar]
- 5.Takahashi H, Nakamura K, Kaneko F, Nakagawa H, Iizuka H Japanese Society for Psoriasis Research. Analysis of psoriasis patients registered with the Japanese Society for Psoriasis Research from 2002-2008. J Dermatol. 2011;38:1125–1129. doi: 10.1111/j.1346-8138.2010.01145.x. [DOI] [PubMed] [Google Scholar]
- 6.Ding X, Wang T, Shen Y, Wang X, Zhou C, Tian S, et al. Prevalence of psoriasis in China: a population-based study in six cities. Eur J Dermatol. 2012;22:663–667. doi: 10.1684/ejd.2012.1802. [DOI] [PubMed] [Google Scholar]
- 7.Tsai TF, Wang TS, Hung ST, Tsai PI, Schenkel B, Zhang M, et al. Epidemiology and comorbidities of psoriasis patients in a national database in Taiwan. J Dermatol Sci. 2011;63:40–46. doi: 10.1016/j.jdermsci.2011.03.002. [DOI] [PubMed] [Google Scholar]
- 8.Henseler T, Christophers E. Disease concomitance in psoriasis. J Am Acad Dermatol. 1995;32:982–986. doi: 10.1016/0190-9622(95)91336-x. [DOI] [PubMed] [Google Scholar]
- 9.Gelfand JM, Weinstein R, Porter SB, Neimann AL, Berlin JA, Margolis DJ. Prevalence and treatment of psoriasis in the United Kingdom: a population-based study. Arch Dermatol. 2005;141:1537–1541. doi: 10.1001/archderm.141.12.1537. [DOI] [PubMed] [Google Scholar]
- 10.Christophers E. Psoriasis--epidemiology and clinical spectrum. Clin Exp Dermatol. 2001;26:314–320. doi: 10.1046/j.1365-2230.2001.00832.x. [DOI] [PubMed] [Google Scholar]
- 11.Gudjonsson JE, Elder JT. Psoriasis: epidemiology. Clin Dermatol. 2007;25:535–546. doi: 10.1016/j.clindermatol.2007.08.007. [DOI] [PubMed] [Google Scholar]
- 12.Lin XR. Psoriasis in China. J Dermatol. 1993;20:746–755. doi: 10.1111/j.1346-8138.1993.tb01378.x. [DOI] [PubMed] [Google Scholar]
- 13.Jacobi A, Langenbruch A, Purwins S, Augustin M, Radtke MA. Prevalence of obesity in patients with psoriasis: results of the national study PsoHealth3. Dermatology. 2015;231:231–238. doi: 10.1159/000433528. [DOI] [PubMed] [Google Scholar]
- 14.Herron MD, Hinckley M, Hoffman MS, Papenfuss J, Hansen CB, Callis KP, et al. Impact of obesity and smoking on psoriasis presentation and management. Arch Dermatol. 2005;141:1527–1534. doi: 10.1001/archderm.141.12.1527. [DOI] [PubMed] [Google Scholar]
- 15.Kim CR, Lee JH. An observational study on the obesity and metabolic status of psoriasis patients. Ann Dermatol. 2013;25:440–444. doi: 10.5021/ad.2013.25.4.440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ferrándiz C, Pujol RM, García-Patos V, Bordas X, Smandía JA. Psoriasis of early and late onset: a clinical and epidemiologic study from Spain. J Am Acad Dermatol. 2002;46:867–873. doi: 10.1067/mjd.2002.120470. [DOI] [PubMed] [Google Scholar]
- 17.Sampogna F, Gisondi P, Melchi CF, Amerio P, Girolomoni G, Abeni D. Prevalence of symptoms experienced by patients with different clinical types of psoriasis. Br J Dermatol. 2004;151:594–599. doi: 10.1111/j.1365-2133.2004.06093.x. [DOI] [PubMed] [Google Scholar]
- 18.Fan X, Yang S, Sun LD, Liang YH, Gao M, Zhang KY, et al. Comparison of clinical features of HLA-Cw*0602-positive and -negative psoriasis patients in a Han Chinese population. Acta Derm Venereol. 2007;87:335–340. doi: 10.2340/00015555-0253. [DOI] [PubMed] [Google Scholar]
- 19.Hernánz JM, Sánchez-Regaña M, Izu R, Mendiola V, García-Calvo C. Clinical and therapeutic evaluation of patients with moderate to severe psoriasis in Spain: the secuence study. Actas Dermosifiliogr. 2012;103:897–904. doi: 10.1016/j.adengl.2012.04.017. [DOI] [PubMed] [Google Scholar]
- 20.Farber EM, Nall ML. The natural history of psoriasis in 5,600 patients. Dermatologica. 1974;148:1–18. doi: 10.1159/000251595. [DOI] [PubMed] [Google Scholar]
- 21.Hägg D, Eriksson M, Sundström A, Schmitt-Egenolf M. The higher proportion of men with psoriasis treated with biologics may be explained by more severe disease in men. PLoS One. 2013;8:e63619. doi: 10.1371/journal.pone.0063619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fortes C, Mastroeni S, Leffondré K, Sampogna F, Melchi F, Mazzotti E, et al. Relationship between smoking and the clinical severity of psoriasis. Arch Dermatol. 2005;141:1580–1584. doi: 10.1001/archderm.141.12.1580. [DOI] [PubMed] [Google Scholar]
- 23.Youn JI, Park BS, Park SB, Kim SD, Suh DH. Characterization of early and late onset psoriasis in the Korean population. J Dermatol. 1999;26:647–652. doi: 10.1111/j.1346-8138.1999.tb02066.x. [DOI] [PubMed] [Google Scholar]
- 24.Cohen AD, Weitzman D, Dreiher J. Psoriasis and hypertension: a case-control study. Acta Derm Venereol. 2010;90:23–26. doi: 10.2340/00015555-0741. [DOI] [PubMed] [Google Scholar]
- 25.Chularojanamontri L, Kulthanan K, Suthipinittharm P, Jiamton S, Wongpraparut C, Silpa-Archa N, et al. Clinical differences between early- and late-onset psoriasis in Thai patients. Int J Dermatol. 2015;54:290–294. doi: 10.1111/ijd.12515. [DOI] [PubMed] [Google Scholar]
- 26.Altobelli E, Petrocelli R, Marziliano C, Fargnoli MC, Maccarone M, Chimenti S, et al. Family history of psoriasis and age at disease onset in Italian patients with psoriasis. Br J Dermatol. 2007;156:1400–1401. doi: 10.1111/j.1365-2133.2007.07906.x. [DOI] [PubMed] [Google Scholar]
- 27.López-Estebaranz JL, Sánchez-Carazo JL, Sulleiro S. Effect of a family history of psoriasis and age on comorbidities and quality of life in patients with moderate to severe psoriasis: Results from the ARIZONA study. J Dermatol. 2016;43:395–401. doi: 10.1111/1346-8138.13157. [DOI] [PubMed] [Google Scholar]