Abstract
Purpose
To evaluate the incidence, characteristics, and the progression of epiretinal membrane (ERM) remnant edge seen by optical coherence tomography (OCT) after ERM peeling.
Methods
A retrospective chart review was conducted for 86 eyes of 85 consecutive patients who diagnosed with ERM and underwent pars plana vitrectomy (PPV) for epiretinal membrane peeling between 2013 and 2014. Data collected and analyzed included age, gender, pre- and post-operative visual acuity, use of indocyanine green (ICG) dye to stain internal limiting membrane (ILM), tamponade used after vitrectomy, ERM edge boundaries, presence of cystoid macular edema, and central foveal thickness.
Results
An ERM remnant edge was detected in 33/86 study eyes (38.4%) at the first post-operative OCT scan. Compared to those without an ERM remnant, patients with an ERM remnant after surgery were significantly older at baseline and had a higher incidence of ERM recurrence at their last visit. They were not significantly different in terms of gender, pre- and post-operative visual acuity, reduction of central foveal thickness from baseline, proportion of eyes with pre-operative ERM elevation on OCT, presence of macular edema before surgery, intra-operative use of ICG staining for ILM peeling, or tamponade used. Based on the edge morphology, we classified the ERM remnant into three types: type 1 was flat and blended with the retina (14/33 eyes, 42.4%), type 2 was flat but stepped (17/33 eyes, 51.5%), and type 3 was elevated (2/33 eyes, 6.0%). A significantly higher risk of ERM recurrence was seen in type 2 and type 3 ERM remnants (75% and 100%, respectively) than type 1 ERM remnants (10%).
Conclusion
An ERM remnant edge was detected by OCT after ERM peeling in 38.4% of eyes. The presence of a post-operative ERM edge was associated with a higher risk of ERM recurrence, particularly in type 2 and type 3 ERM remnants.
Keywords: epiretinal membrane, vitrectomy, optical coherence tomography
INTRODUCTION
Epiretinal membrane (ERM) is a common cause of retinal pathology that may cause reduction in vision and metamorphopsia,1-3 , which may be treated by pars plana vitrectomy (PPV) with ERM peeling in symptomatic patients.4 However, ERM recurrence occurs in 10% to 21% of cases and may require additional surgical intervention.4-9 Some surgeons advocate internal limiting membrane (ILM) peeling at the time of ERM removal to prevent ERM recurrence, while others advise against this because of histopathologic evidence of retinal damage associated with ILM removal and use of ICG dye.5-9
Spectral domain-OCT (SD-OCT) has an axial depth resolution of 3–7μm and a lateral resolution of 11μm 10-11, and has become an essential in the diagnosis and monitoring of ERMs12. Previous studies have demonstrated an association between the external limiting membrane, and ellipsoid zone with the post-operative visual acuity outcomes and metamorphosia.12-13 Post operative complications of ERM removal such as swelling of the arcuate nerve fiber layer, cystoid macular edema, delayed new-onset inner nuclear layer cystic changes, and extrafoveal macular holes after ERM surgery have been reported and can now be followed by OCT.14, 15,16
Interestingly, we have observed that the edge of an ERM may often be detected by SD-OCT after surgery. In this study, we reviewed the frequency at which this occurred, the characteristics of the ERM edge remnants, and its prognostic implications.
METHODS
A retrospective chart review study was conducted according to the Helsinki declaration and the Health Insurance Portability and Accountability Act of 1996. Written informed consent was obtained for each patient prior to surgery. Institutional Review Board (IRB) approval was acquired from University of California San Diego for the review and analysis of patients’ data.
A retrospective chart review was conducted for patients with a diagnosis of ERM who underwent pars plana vitrectomy between 2013 and 2014 at the Jacobs Retina Center Shiley Eye Institute, University of California, San Diego. We studied only idiopathic ERMs and excluded ERM secondary to retinal tear or detachment or associated with trauma, vitreous hemorrhage, retinovascular disease, or uveitis. Data collected and analyzed included gender, age, types of tamponade used after vitrectomy, usage of indocyanine green (ICG) staining, internal limiting membrane (ILM) peeling, and visual acuity at each visit. The pre-and post- operative SD-OCT images were analyzed for ERM characteristics, presence of macular edema, and central foveal thickness.
The ophthalmic examinations included ETDRS measurement of the visual acuity, applanation tonometry, slit-lamp biomicroscopy, indirect ophthalmoscopy, fundus photography, fluorescein angiography, and SD-OCT (Spectralis HRA+OCT, Heidelberg, Germany). The central foveal thickness was measured from the internal limiting membrane to the Bruch's membrane. The edge of an ERM remnant was measured from the center of the fovea. ERM recurrence was defined as a regrowth of epiretinal membrane covering the fovea.
All patients underwent fluorescein angiography and SD-OCT using the Heidelberg Spectralis system. Both horizontal and vertical SD-OCT scans were performed with a scanning line length of 6mm. In some cases, a horizontal 49-line raster scan centered on the fovea was performed. Imaging was performed preoperatively and at all follow up visits.
The indication for the ERM surgery was a visual disturbance, including specifically a decrease in visual acuity or metamorphopsia. All of the surgery was performed by the same experienced vitreoretinal surgeon (WRF). Standard transconjunctival three-port pars plana vitrectomy with membrane peeling was performed using 25-gauge instruments. If a complete posterior vitreous detachment was not present, a posterior vitreous detachment was created using aspiration. With the assistance of decanted triamcinolone staining 40 mg/ml (Kenalog, Bristol-Myer squibb, NJ, USA), ERM was peeled circularly and tangentially to the retinal surface using end-gripping intraocular forceps. Special attention was paid to remove the membrane in a large sheet from the macular area. In all cases, the ERM was successfully removed from the fovea and macular area. The internal limiting membrane (ILM) was removed with indocyanine green (ICG) assisted staining 6.25/0.1ml (Akron, Inc, USA) with ILM forceps (Alcon, Forth worth, USA) if the ERM was patchy, or extremely adherent and/or appeared fused to the ILM on preoperative raster OCT scans, or in cases of recurrent ERM.17 In such cases, indocyanine green staining was used to facilitate ILM peeling as previously described.18 We use gas tamponade in cases of thin cystic retina or in cases of inner lamellar macular hole secondary to ERM. The retinal periphery was checked for breaks and peripheral laser photocoagulation was performed to any retinal breaks and in addition three rows of laser spots were placed anterior to the equator.19
Statistical analyses were conducted using SAS software version 9.4 (SAS Institute, Cary, North Carolina, USA). Descriptive statistics included mean, standard deviation (SD), median, range, and percentages where appropriate. The Student's T test for independent samples was used to compare mean vision and central foveal thickness between eyes with and without ERM remnants. The paired Student T test was used to compare mean vision and central foveal thickness at baseline and the final visit. Chi-square testing was used to compare the proportion of gender, ICG staining/ILM peeling, tamponade used and recurrence rate between eyes with and without ERM remnants after surgery. P-values represent results for 2-sided tests, with values less than 0.05 considered statistically significant.
RESULTS
Eighty-six eyes of 85 patients with ERM were reviewed, twenty-one of which (24%) were recurrent ERM. The mean follow-up period after surgery was 9.8 ± 6.8 months (range 1-30 months). The mean age of the patients at baseline was 69.0±10.1 years (range 34-88) and 55.8% of them were female. The mean visual acuity before surgery was logMAR 0.36 ±0.21 (range 0.03-1.0, logMAR, median 0.32, logMAR) (Snellen equivalent: mean 20/63, range 20/600- 20/20, median 20/63). On pre-operative OCT scanning, macular edema was detected in 73.3% of eyes with ERM remnants after the surgery and in 85.1% of eyes without ERM remnants after the surgery (P=0.23). The mean central foveal thickness before surgery was 402±176 μm.
During surgery 34 of the 86 eyes (39.5%) required ICG staining and ILM peeling. Gas tamponade was used for 27/86 eyes, while 59/86 eyes were left with BSS fill after surgery without any tamponade
The first OCT scan was done on average at a mean of 3.3 months (range 2 weeks to 18 months, median 3 month) after surgery. Eighty percent (69/86) of patients had the first scan in the first 2 months. Post-operatively, an ERM remnant edge was detected in 33 out of the 86 (38.4%) operated eyes at the first OCT scan after surgery. Thirty-one out of the 33 ERM edges (94%) were located in the extrafoveal area, and only 2 (6%) of them had ERM edges located within the fovea, defined as within 1000 μm area of the foveola. Macular edema by OCT was present postoperatively in 9/33 eyes (27%) with an ERM remnant. There was no difference in central foveal thickness in the early postoperative period in eyes with ERM remnants compared to eyes without ERM remnants (290.0 ± 71.4 um versus 303.6 ± 127.6 um, P=0.55).
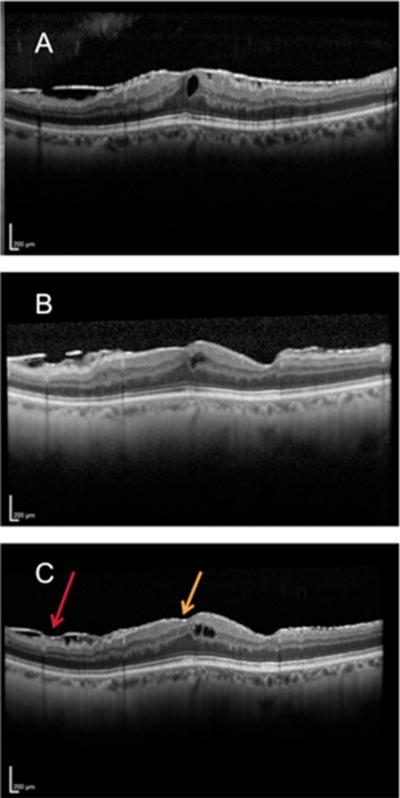
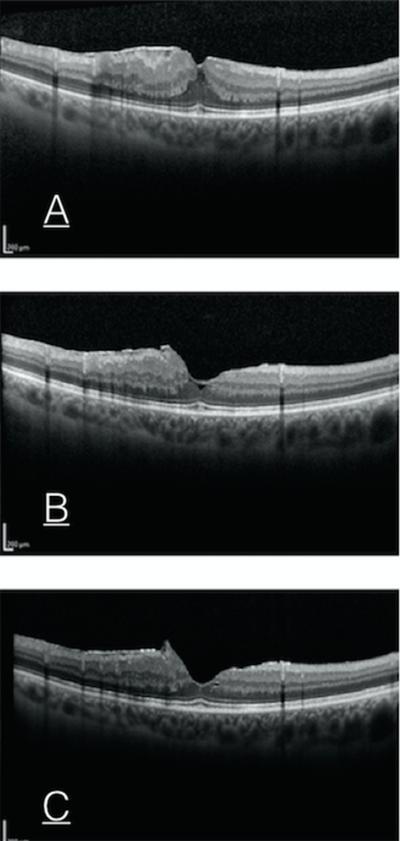
Postoperative serial OCT scans were performed for 19 out of 33 eyes (57%) with ERM remnants, for which the dynamic changes of the ERM remnant edge was observed. Among these 19 eyes, ERM recurred centrally (recurrent ERM covered the entire macular area at the final visit) in 3 eyes (16%) (Figure 1). In 8 eyes (42%), the edge of the ERM progressed to the foveola on average 420 ± 393 μm (range 67-1074 μm, median 280 μm), 6 eyes (31.5%) remained stable, and 2 eye (10.5 %) regressed from foveola (Figure 2) on average 216 ± 7 μm ( range 211-221 μm ) .
Figure 1.
Preoperative OCT of ERM (A), OCT two weeks postop (B), and OCT 18 months postop (C) demonstrating thin recurrent ERM (red arrow demonstrates recurrence with more confluent ERM, yellow arrow demonstrates early thin recurrence of ERM up to the fovea)
Figure 2.
Preoperative OCT of ERM (A), 2 months postop (B) , 4 months postop (C) demonstrating regression of ERM remnant.
Eyes with a detectable ERM remnant in the early post operative period were significantly older at baseline (71.7±8.9 versus 67.3±10.5 years old, P=0.04) and had a higher incidence of recurrence (42.1 % versus 9.4%, P=0.003). There was no significant difference between the two groups in terms of gender, visual acuity and central foveal thickness before surgery or at the final visit, presence of ERM edge elevation before surgery, preoperative macular edema, ICG staining or ILM peeling, tamponade usage, follow-up period, or reduction of central foveal thickness from baseline at the final visit (Table 1), also there was no association between ILM peeling and ERM recurrence (P=0.59).
Table 1.
Comparison of variables between eyes with and without ERM remnant after surgery.
| Variables compared | with ERM remnant | without ERM remnant | P value |
|---|---|---|---|
| Age before surgery (years) | 71.7±8.9 | 67.3±10.5 | 0.04 |
| Gender male/female) | 20/13 | 28/25 | 0.48 |
| Visual acuity before surgery (Log MAR) (Snellen) | 0.55±0.34 (≈20/70) | 0.53±0.31 (≈20/70) | 0.81 |
| Central foveal thickness before surgery (um) | 374.7±127.6 | 418.9±199.6 | 0.29 |
| Proportion of ERM edge elevated before surgery | 13.3% | 14.9% | 0.56 |
| Proportion of macular edema (IRF/SRF) before surgery | 73.3% | 85.1% | 0.23 |
| Proportion of ICG staining and ILM peeling | 36.7% | 43.4% | 0.65 |
| Proportion of temponade usage | 34.4% | 29.6% | 0.81 |
| Follow-up period (months) | 10.6±6.5 | 9.3±7.0 | 0.43 |
| Visual acuity at the final visit (Log MAR) (Snellen) | 0.35±0.31 (≈20/40) | 0.40±0.31 (20/50) | 0.51 |
| Central foveal thickness at the final visit (um) | 274.8±89.3 | 288.6±95.4 | 0.51 |
| Proportion of ERM recurrence | 42.1% | 9.4% | 0.003 |
| Reduction of central foveal thickness (um) | 95.0 ±146.8 | 130.5 ± 155.4 | 0.33 |
ERM: epiretinal membrane; IRF/SRF: intraretinal fluid/subretinal fluid; ICG: indocyanine green; ILM: internal limiting membrane; continuous parameters are represented as mean value ± standard deviation
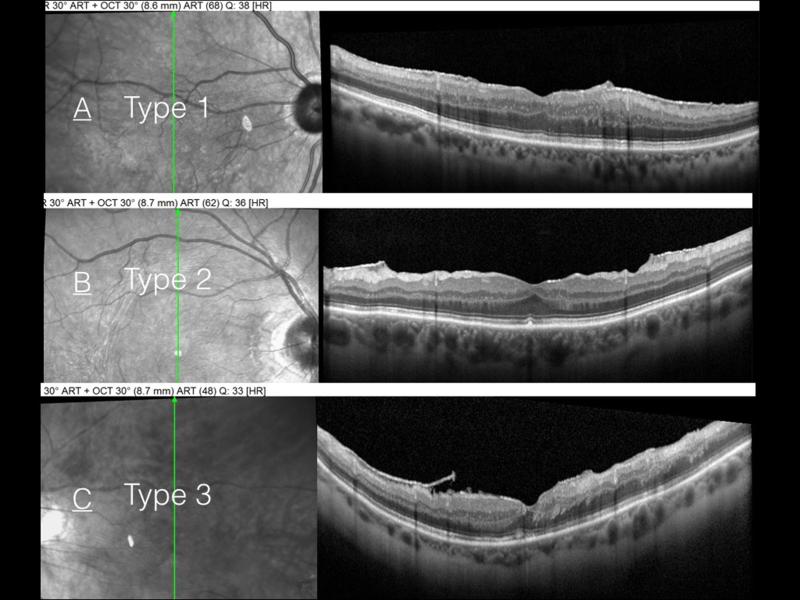
Based on the edge morphology, we further classified the ERM remnant into three types: type 1 was flat and blended with the retina (42.4%), type 2 was flat but stepped (51.5%) and type 3 was elevated (6.0%, Figure 3). A significantly higher risk of ERM recurrence (not necessarily requiring surgical intervention) was seen in type 2 and type 3 ERM remnants (75% and 100% of eyes, respectively) than those with a type 1 ERM remnant (10% of type 1 eyes).
Figure 3.
Classification of epiretinal membrane remnant edges: Type 1 flat and blended with retina (A), Type 2 flat and stepped (B), and Type 3 elevated (C).
DISCUSSION
In this single center retrospective study, ERM remnant edges were detected in the post operative OCT scan in 38.4% of eyes after TPPV and ERM peeling. The OCT detected ERM remnant edges were associated with a significantly higher incidence of ERM recurrence during a mean follow-up period of 10 months. Based on the edge morphology, we further classified the ERM remnant edges into three types: type 1 flat and edge blended with the retina, type 2 flat but edge stepped, type 3 elevated edge. We found a significantly higher incidence of ERM recurrence in eyes with type 2 and type 3 ERM remnants (75% and 100% of eyes, respectively) than those with a type 1 ERM remnant (10% of type 1 eyes).
The clinical significance of ERM is related to its impact on visual function. 1-3 An ERM on the macular surface might cause macular edema and photoreceptor layer disruption, leading to visual symptoms such as visual acuity decrease or metamorphopsia, which are often indications for surgical removal. During surgery for ERM removal, it is important to remove the ERM over the entire macula including the foveal surface to improve macular anatomy and visual function. Extrafoveal or extramacular parts of the ERMs are clinically less significant as they may have a minimal effect on visual function. For this reason, and because it may be more difficult to remove a more peripheral ERM, it is common that some extra macular areas of ERM is allowed to remain after surgery. Our study shows that ERM remnants were detected in 38% of the eyes that underwent ERM removal. In addition, our results show that these extrafoveal ERM remnants appear to have no or minimal effect on fovea thickness and visual acuity.
Although extrafoveal ERM remnants may not affect visual acuity or distortion, the presence of these remnants are associated with a higher recurrence rate of ERM after surgery. In eyes with ERM remnants, the recurrence rate was significantly higher than those eyes without remnants (42% vs 9%, respectively). Although most of the recurrences were asymptomatic and did not require additional surgery, our study suggests that it may be beneficial to remove ERM as extensively as possible including outside of the macula.
ERM recurrence rates have been reported to vary from 2% to 21% in previous studies,4-9 and it has been suggested that ILM peeling might reduce ERM recurrence.5-8 Our study suggests that examination of the morphology of the retina after ERM peel to look for ERM remnant is a definite new predictor of recurrence and indeed ICG use had a minimal predictive value. We note however that ICG use was not randomized in the current study so we do not have a good way to determine it's predictive value.
The limitations and strength of the study should be mentioned. As a retrospective study of a relatively small number of patients treated by a single surgeon, possible selection bias might exist. Future studies with a larger sample size from multiple centers should be performed to confirm our findings. The strength of our study is this is the first study discussing the presence, characteristics, classification and associations of ERM edge remnant detected by OCT after surgical removal.
In summary, ERM remnant edges can be seen by OCT in about one third of eyes after TPPV with ERM peeling. While the edge itself is not associated with post-operative foveal thickness or visual function, it does appear to be correlated with a significantly higher ERM recurrence rate. Based on the morphology of the ERM remnant edge, we can classify the remnants into 3 categories, type 1 flat and blended with retina, type 2 flat and stepped and type 3 elevated, and we find that type 2 and 3 edges are associated with a higher recurrence than type 1 edge.
Summary Statement.
An ERM remnant edge was detected by OCT after ERM peeling in 38.4% of eyes and was associated with a higher risk of ERM recurrence, particularly when the edge is flat and stepped or elevated.
Acknowledgments
Supported in part by UCSD Vision Research Center Core Grant P30EY022589, an unrestricted grant from Research to Prevent Blindness, NY (WRF) and ICO-Retina Research Foundation Helmerich Fellowship, Beijing Talents Scholarship (2015000021223ZK22) and National Natural Science Foundation of China (No. 81400422) (QSY)
REFERENCES
- 1.Mitchell P, Smith W, Chey T, et al. Prevalence and associations of epiretinal membranes. The Blue Mountains Eye Study, Australia. Ophthalmology. 1997;104:1033–1040. doi: 10.1016/s0161-6420(97)30190-0. [DOI] [PubMed] [Google Scholar]
- 2.You Q, Xu L, Jonas JB. Prevalence and associations of epiretinal membranes in adult Chinese: the Beijing eye study. Eye (Lond) 2008;22:874–879. doi: 10.1038/sj.eye.6702786. [DOI] [PubMed] [Google Scholar]
- 3.Gass JDM. Macular dysfunction caused by epiretinal membrane contraction. Stereoscopic Atlas of Macular Diseases: Diagnosis and Treatment. 1997:938–950. [Google Scholar]
- 4.Grewing R, Mester U. Results of surgery for epiretinal membranes and their recurrences. Br J Ophthalmol. 1996;80:323–326. doi: 10.1136/bjo.80.4.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Park DW, Dugel PU, Garda J, et al. Macular pucker removal with and without internal limiting membrane peeling: pilot study. Ophthalmol. 2003;110:62–64. doi: 10.1016/s0161-6420(02)01440-9. [DOI] [PubMed] [Google Scholar]
- 6.Bovey EH, Uffer S, Achache F. Surgery for epimacular membrane: impact of retinal internal limiting membrane removal on functional outcome. Retina. 2004;24:728–735. doi: 10.1097/00006982-200410000-00007. [DOI] [PubMed] [Google Scholar]
- 7.Kwok A, Lai TY, Yuen KS. Epiretinal membrane surgery with or without internal limiting membrane peeling. Clin Experiment Ophthalmol. 2005;33:379–385. doi: 10.1111/j.1442-9071.2005.01015.x. [DOI] [PubMed] [Google Scholar]
- 8.Gandorfer A, Haritoglou C, Scheler R, et al. Residual cellular proliferation on the internal limiting membrane in macular pucker surgery. Retina. 2012;32:477–485. doi: 10.1097/IAE.0b013e3182246e2a. [DOI] [PubMed] [Google Scholar]
- 9.Jackson TL, Donachie PH, Williamson TH, et al. The Royal College of Ophthalmologists’ National Opthalmology Database Study of Vitreoretinal Surgery: Report 4, Epiretinal Membrane. Retina. 2015;35:1615–1621. doi: 10.1097/IAE.0000000000000523. [DOI] [PubMed] [Google Scholar]
- 10.Kozak I, Bartsch DU, Cheng L, Freeman WR. In vivo histology of cotton-wool spots using high-resolution optical coherence tomography. Am J Ophthalmol. 2006;141:748–750. doi: 10.1016/j.ajo.2005.10.048. [DOI] [PubMed] [Google Scholar]
- 11.Wang H, Chhablani J, Freeman WR, et al. Characterization of diabetic microaneurysms by simultaneous fluorescein angiography and spectral-domain optical coherence tomography. Am J Ophthalmol. 2012;153:861–867. doi: 10.1016/j.ajo.2011.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Goldberg RA, Waheed NK, Duker JS. Optical coherence tomography in the preoperative and postoperative management of macular hole and epiretinal membrane. Br J Ophthalmol. 2014;98:20–23. doi: 10.1136/bjophthalmol-2013-304447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Oster SF, Mojana F, Brar M, et al. Disruption of the photoreceptor inner segment/outer segment layer on spectral domain-optical coherence tomography is a predictor of poor visual acuity in patients with epiretinal membranes. Retina. 2010;30:713–718. doi: 10.1097/IAE.0b013e3181c596e3. [DOI] [PubMed] [Google Scholar]
- 14.Kozak I, 1, Freeman WR. Non-progressive extrafoveal retinal hole after foveal epiretinal membrane removal. Am J Ophthalmol. 2006;141:769–771. doi: 10.1016/j.ajo.2005.11.018. [DOI] [PubMed] [Google Scholar]
- 15.Hartmann KL, Schuster AK, Bartsch DU, et al. Restoration of retinal layers after epiretinal membrane peeling. Retina. 2014;34:647–654. doi: 10.1097/IAE.0000000000000003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dolz-Marco R, Hoang QV, Gallego-Pinazo R, Chang S. Assessment of the significance of cystic changes after epiretinal membrane surgery with internal limiting membrane removal. Retina. 2016;36:727–732. doi: 10.1097/IAE.0000000000000780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim JS, Chhablani J, Chan CK, et al. Retinal adherence and fibrillary surface changes correlate with surgical difficulty of epiretinal membrane removal. Am J Ophthalmol. 2012;153:692–697. doi: 10.1016/j.ajo.2011.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yuson RM, Nigam N, Mojana F, Brar M, et al. The use of intraoperative indocyanine green dye to assist in epiretinal membrane removal: a novel application of indocyanine green surgical use. Retina. 2009;29:1367–1370. doi: 10.1097/IOP.0b013e3181b80d7a. [DOI] [PubMed] [Google Scholar]
- 19.Koh HJ, Cheng L, Kosobucki B, Freeman WR. Prophylactic intraoperative 360 degrees laser retinopexy for prevention of retinal detachment. Retina. 2007;27:744–749. doi: 10.1097/IAE.0b013e318030ebd7. [DOI] [PubMed] [Google Scholar]