Abstract
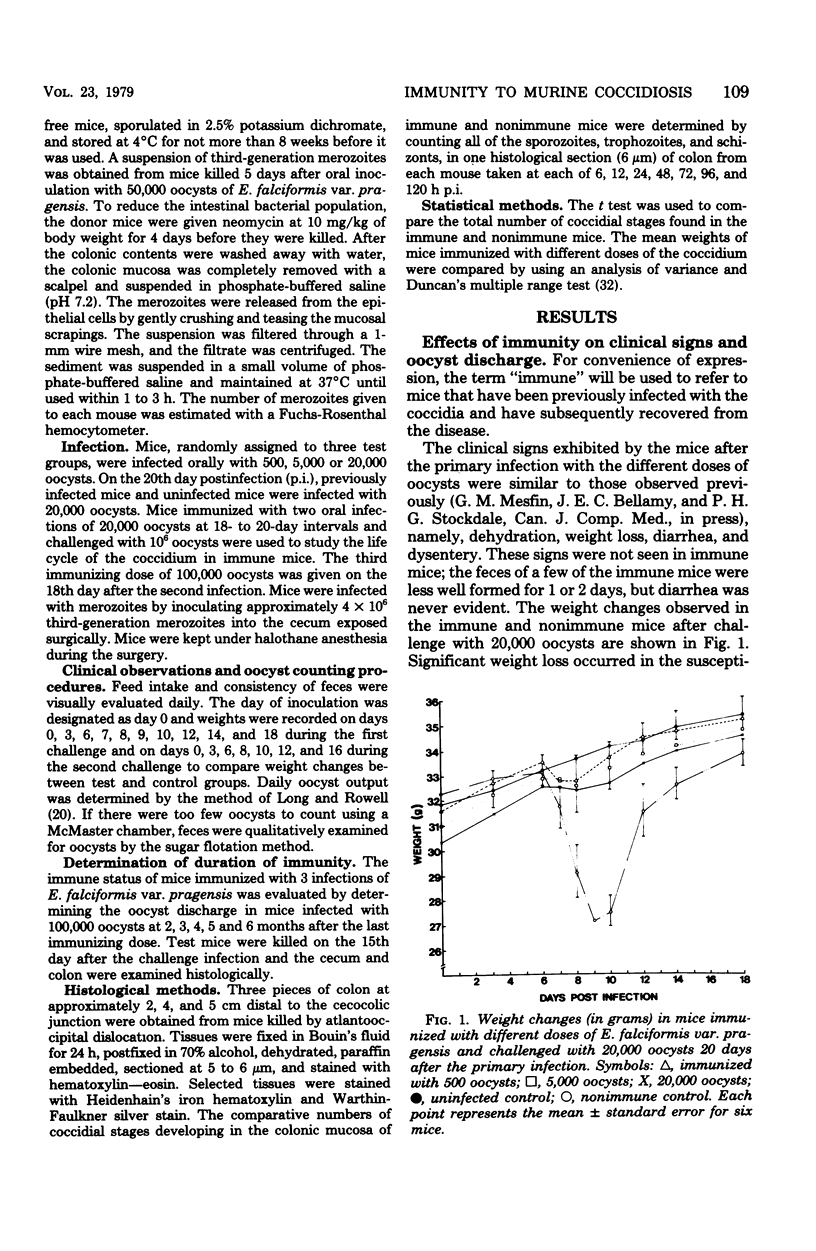
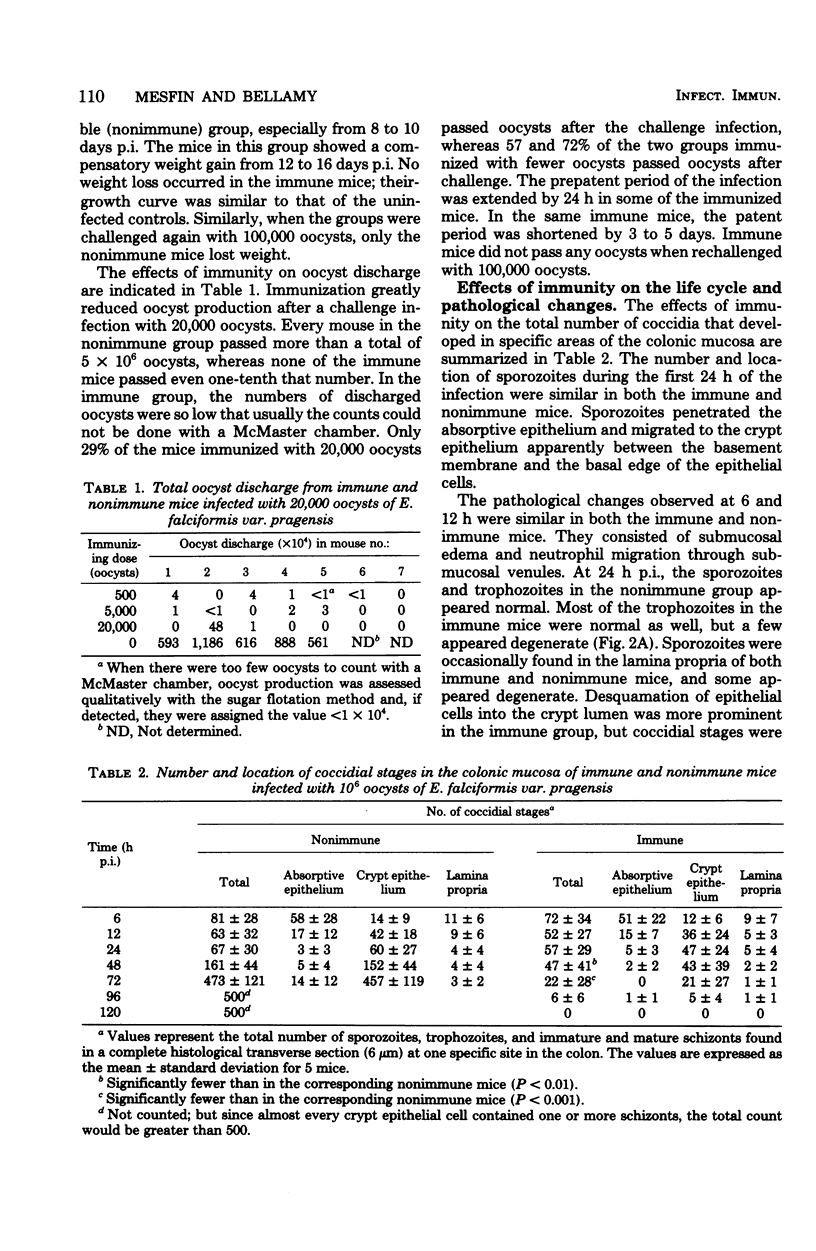
Mice immunized with infections of 500, 5,000, or 20,000 oocysts of E. falciformis var. pragensis were reinfected with 20,000 and 100,000 oocysts at 20 and 38 days, respectively, after the initial infection. After the first challenge infection, none of the immunized mice showed clinical signs of coccidiosis; a few mice passed very low numbers of oocysts, and oocyst discharge seemed to correlate negatively with immunizing dose. None of the mice immunized twice passed oocysts after challenge. Mice immunized with three infections were completely immune to challenge for 4 months. The effect of the immune response on the life cycle of the coccidium was determined by histological examination of the intestines of immune and nonimmune mice infected with the parasite. In both the immune and nonimmune groups, sporozoites penetrated absorptive epithelial cells and migrated to crypt epithelial cells during the first 6 to 24 h postinfection. At 48 to 72 h postinfection, the sporozoites developed into mature first-generation schizonts in the nonimmune mice, whereas the developing first-generation schizonts degenerated within the crypt epithelial cells of the immune mice. In nonimmune mice, third-generation merozoites, inoculated intracecally, developed into mature fourth-generation schizonts, whereas in immune mice the developing fourth-generation schizonts degenerated before maturing. The possibility that a cellmediated immune mechanism is responsible for the arrest in schizogony is discussed.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bienenstock J., Perey D. Y. Immune mechanisms of mucosal resistance. Med Clin North Am. 1972 Mar;56(2):391–402. doi: 10.1016/s0025-7125(16)32403-8. [DOI] [PubMed] [Google Scholar]
- Cerná Z., Sénaud J., Mehlhorn H., Scholtyseck E. Etude comparée des rélations morphologiques et immunologiques chez les coccidies de la souris: Eimeria falciformis et Eimeria pragensis (Coccidia, Eimeriidae) Folia Parasitol (Praha) 1974;21(4):301–309. [PubMed] [Google Scholar]
- Cerottini J. C., Brunner K. T. Cell-mediated cytotoxicity, allograft rejection, and tumor immunity. Adv Immunol. 1974;18:67–132. doi: 10.1016/s0065-2776(08)60308-9. [DOI] [PubMed] [Google Scholar]
- Chinchilla M., Frenkel J. K. Mediation of immunity to intracellular infection (Toxoplasma and Besnoitia) within somatic cells. Infect Immun. 1978 Mar;19(3):999–1012. doi: 10.1128/iai.19.3.999-1012.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark I. A., Allison A. C., Cox F. E. Protection of mice against Babesia and Plasmodium with BCG. Nature. 1976 Jan 29;259(5541):309–311. doi: 10.1038/259309a0. [DOI] [PubMed] [Google Scholar]
- Clark I. A., Richmond J. E., Wills E. J., Allison A. C. Intra-erythrocytic death of the parasite in mice recovering from infection with Babesia microti. Parasitology. 1977 Oct;75(2):189–196. doi: 10.1017/s0031182000062338. [DOI] [PubMed] [Google Scholar]
- Fitzgerald P. R. Results of continuous low-level inoculations with Eimeria bovis in calves. Am J Vet Res. 1967 May;28(124):659–665. [PubMed] [Google Scholar]
- HORTON-SMITH C., PIERCE A. E. BEHAVIOR OF INVASIVE STAGES OF EIMERIA TENELLA IN THE IMMUNE FOWL (GALLUS DOMESTICUS). Exp Parasitol. 1963 Aug;14:66–74. doi: 10.1016/0014-4894(63)90011-0. [DOI] [PubMed] [Google Scholar]
- Hein H. E. Eimeria acervulina, E. brunetti, E. maxima, and E. necatrix: low doses of oocysts to immunize young chickens. Exp Parasitol. 1976 Oct;40(2):250–260. doi: 10.1016/0014-4894(76)90088-6. [DOI] [PubMed] [Google Scholar]
- Klesius P. H., Kramer T. T., Frandsen J. C. Eimeria stiedai: delayed hypersensitivity response in rabbit coccidiosis. Exp Parasitol. 1976 Feb;39(1):59–68. doi: 10.1016/0014-4894(76)90010-2. [DOI] [PubMed] [Google Scholar]
- Klesius P. H., Kramer T., Burger D., Malley A. Passive transfer of coccidian oocyst antigen and diphtheria toxoid hypersensitivity in calves across species barriers. Transplant Proc. 1975 Sep;7(3):449–452. [PubMed] [Google Scholar]
- Klesius P. H., Kristensen F., Elston A. L., Williamson O. C. Eimeria bovis: evidence for a cell-mediated immune response in bovine coccidiosis. Exp Parasitol. 1977 Apr;41(2):480–490. doi: 10.1016/0014-4894(77)90120-5. [DOI] [PubMed] [Google Scholar]
- Kouwenhoven B., van der Horst C. J. Histological observations with respect to the immune mechanism in Eimeria acervulina infection in the domestic fowl. Z Parasitenkd. 1973 Aug 3;42(1):11–21. doi: 10.1007/BF00329006. [DOI] [PubMed] [Google Scholar]
- Leathem W. D., Burns W. C. Effects of the immune chicken on the endogenous stages of Eimeria tenella. J Parasitol. 1967 Feb;53(1):180–185. [PubMed] [Google Scholar]
- Liburd E. M., Pabst H. F., Armstrong W. D. Transfer factor in rat coccidiosis. Cell Immunol. 1972 Nov;5(3):487–489. doi: 10.1016/0008-8749(72)90075-5. [DOI] [PubMed] [Google Scholar]
- Mackaness G. B. Resistance to intracellular infection. J Infect Dis. 1971 Apr;123(4):439–445. doi: 10.1093/infdis/123.4.439. [DOI] [PubMed] [Google Scholar]
- Morita C., Tsutsumi Y., Soekawa M. Migration inhibition test of splenic cells of chickens infected with Eimeria tenella. J Parasitol. 1973 Feb;59(1):199–200. [PubMed] [Google Scholar]
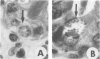
- Niilo L. Acquired resistance to reinfection of rabbits with Eimeria magna. Can Vet J. 1967 Sep;8(9):201–208. [PMC free article] [PubMed] [Google Scholar]
- Rose M. E. Eimeria tenella: skin hypersensitivity to injected antigen in the fowl. Exp Parasitol. 1977 Jun;42(1):129–141. doi: 10.1016/0014-4894(77)90070-4. [DOI] [PubMed] [Google Scholar]
- Rose M. E., Hesketh P. Immunity to coccidiosis: stages of the life-cycle of Eimeria maxima which induce, and are affected by, the response of the host. Parasitology. 1976 Aug;73(1):25–57. doi: 10.1017/s0031182000051295. [DOI] [PubMed] [Google Scholar]
- Rose M. E., Long P. L., Bradley J. W. Immune responses to infections with coccidia in chickens: gut hypersensitivity. Parasitology. 1975 Dec;71(3):357–368. doi: 10.1017/s0031182000047132. [DOI] [PubMed] [Google Scholar]
- Rose M. E. Protective antibodies in infections with Eimeria maxima: the reduction of pathogenic effects in vivo and a comparison between oral and subcutaneous administration of antiserum. Parasitology. 1974 Jun;68(3):285–292. doi: 10.1017/s0031182000045819. [DOI] [PubMed] [Google Scholar]
- Walker W. A., Isselbacher K. J. Intestinal antibodies. N Engl J Med. 1977 Oct 6;297(14):767–773. doi: 10.1056/NEJM197710062971407. [DOI] [PubMed] [Google Scholar]