Abstract
The reconstitution of immune function after hematopoietic cell transplant (HCT) plays an important role in the control of viral infections. Both donor and recipient cytomegalovirus (CMV) serostatus has been shown to contribute to effective immune function; however, the influence of a nonmyeloablative preparative (NMA) regimen using total lymphoid irradiation (TLI) and antithymocyte globulin (ATG) on antiviral immune reconstitution has not yet been described. In 117 recipients of NMA HCT patients following ATG and TLI, not unexpectedly, CMV viremia was seen in approximately 60% of the seropositive patients regardless of donor serostatus, and recipient seropositivity significantly increased the odds of CMV viremia after transplant in a multivariate analysis. The administration of ATG and TLI resulted in a strikingly earlier viremia in the posttransplant period when compared to the previously reported timing of viremia following myeloablative preparative regimens, especially for transplant recipients who were seropositive for CMV with seronegative donors. Furthermore, early viremia in the setting of a CMV naïve donor was associated with a delay in functional antiviral control. These observations demonstrate the dynamic nature of immunity in relation to CMV antigen exposure in the complex environment resulting from NMA conditions where both donor and residual recipient immune response affect viral control.
Keywords: Cytomegalovirus, Bone marrow transplantation, Nonmyeloablative, Immune reconstitution, Antithymocyte globulin
INTRODUCTION
The study of posttransplant infection and disease caused by common viral pathogens such as cytomegalovirus (CMV) offers a unique window into the development of functional immunity after hematopoietic cell transplantation (HCT). The development of CMV-specific immunity is known to be influenced by a number of factors including the genetic relatedness of the transplant donor to the patient, immunomodulatory factors such as T cell depletion or graft-versus-host disease (GVHD) and its treatment, as well as the serologic status of the transplant donor and recipient [1,2]. The relative influence of each of these factors has been delineated for patients undergoing transplant following myeloablative (MA) conditioning regimens [3,4]. However, the factors influencing the reconstitution of effective antiviral response following nonmyeloablative (NMA) preparative regimens, especially those involving modulation of recipient immunity, have not been well defined.
A preparative regimen utilizing total lymphoid irradiation (TLI) and antithymocyte globulin (ATG) has been shown to result in a decreased incidence of acute GVHD (aGVHD) as well as a decrease in transplant-associated toxicity [5,6]. We have observed that viremia occurred earlier in CMV seropositive patients, regardless of their donor serostatus, compared to seronegative patients with seropositive donors. In addition, there was a trend toward earlier viremia in CMV seropositive patients with CMV naive donors compared with patients with CMV seropositive donors, and this early viremia was associated with a delay in sustained viral control. Our novel observations regarding the reconstitution of functional antiviral immunity following a TLI-ATG preparative regimen suggest that, in addition to the recognized influence of the donor experience with CMV, the timing of viral exposure appears to have a significant impact on the nascent immune system.
METHODS
Transplant Protocol
A total of 177 patients who underwent a Stanford program for NMA allogeneic HCT following a TLI and ATG-based preparative regimen between December 2001 and December 2007 in 4 similar transplant protocols were reviewed for CMV seropositivity of donor and recipient. Of these, 126 patients had either donor or recipient seropositivity and 51 were negative for both donor and recipient; 9 patients were not evaluated because of insufficient data or participation in a CMV prophylaxis trial resulting in 117 patients for further analysis. Patients were eligible for transplant under these 4 protocols if they (1) had a diagnosis of malignant lymphoid or myeloid disease including Hodgkin and non-Hodgkin lymphoma, and (2) were not eligible for conventional MA conditioning regimen because of age >55 years, significant medical comorbidities, or extensive exposure to prior chemotherapy regimens. Patients’ disease status at transplant ranged from complete remission to relapse as evaluated by consensus guidelines. The transplant protocol has been previously described [5,6] and included TLI at 0.8 Gy per day administered daily on days 11 through 7 and again from day 4 through day 1 prior to the day of cell infusion as well as ATG (thymoglobulin; Genzyme, Boston, MA) administered intravenously at 1.5 mg/kg/day on days 11 through 7 prior to cell transplant. Donor cells were not T cell depleted or otherwise manipulated prior to infusion. Immunosuppressive regimens included cyclosporine and mycophenolate mofetil. Antimicrobial prophylaxis included fluconazole 400 mg by mouth once daily through day 100 for most patients, acyclovir 400 mg by mouth twice daily through day 100 for herpes simplex virus (HSV) and/or varicella zoster virus (VZV) seropositive patients, and ciprofloxacin 500 mg by mouth twice daily during periods when absolute neutrophil count was <500 cells/microliter.
Posttransplant Monitoring
Chimerism, the percentage of donor DNA in bone marrow or peripheral blood leukocytes, was determined for all patients at days 28, 56, and 90 posttransplant using sequence-length polymorphic markers encoding short tandem repeats [5]. Flow cytometry was used to analyze the absolute number of CD3+, CD4+, CD8+, CD19+, and NK cells at the same time points.
If the patient, donor, or both were seropositive for CMV, quantitative polymerase chain reaction (PCR) testing of serum was performed on a weekly basis using the Amplicor® CMV test (Roche Molecular Diagnostics, Pleasanton, CA) beginning at the time of TLI and ATG administration, for the first 100 days posttransplantation. The detection of virus led to preemptive treatment with intravenous ganciclovir or oral valganciclovir with a minimum of 2 weeks of induction therapy according to an established on-site protocol. Evaluation for aGVHD was performed on a routine basis; grade II to IV disease was treated with immunosuppression typically beginning with a corticosteroid at a dose of 1–2 mg/kg/day of a prednisone equivalent.
Statistical Analysis
CMV PCR results and other laboratory data were obtained by review of the electronic medical record and combined with a preexisting database containing patient characteristics. CMV reactivation was defined as any positive CMV PCR result. An episode of reactivation was considered resolved after 2 consecutive CMV PCR tests were negative. Statistical analysis was performed using SAS Enterprise Guide (version 9). Two-tailed tests were used, with P < .05 connoting statistical significance. The Fisher’s exact test was used to analyze associations between categoric variables, and continuous variables that did not display a normal distribution were analyzed using the Mann-Whitney U test. Correction for multiple comparisons was not performed as few differences between groups were detected.
Logistic regression for the occurrence of CMV reactivation was initially performed by univariate analysis. Following this, backward stepwise regression was used to determine which variables would be included in the multivariate model to generate adjusted odds ratios. Myelodysplastic syndrome (MDS) was used as the comparator group for disease type because that group had the lowest incidence of CMV reactivation. All continuous variables were first analyzed for linearity in the logit; variables not meeting this criterion were transformed into categoric variables using clinically relevant cutpoints (eg, age >55 years, chimerism >95%). Nonbinary categoric variables, such as serogroup and donor type, were analyzed using dummy coding; the group least associated with CMV reactivation was used as the reference group. All analyses satisfied convergence criterion, and colinearity was not observed between variables. The area under the curve (AUC) for CMV PCR results was calculated for each patient by multiplying the time between sequential positive PCR values by the PCR copy number, and summing the total of areas for each patient. Values for each group were compared for statistical significance using the Mann-Whitney U test.
Retrospective analysis of outcomes in patients undergoing NMA HCT was reviewed with the approval of the local institutional review board as administered by the Stanford University Research Compliance Office.
RESULTS
Pretransplantation Patient Characteristics and Outcomes
A total of 117 patients with either donor or recipient seropositivity to CMV who underwent NMA HCT with TLI-ATG preconditioning at Stanford University between 2001 and 2007 were analyzed. Fifty-seven had both donor and recipient CMV seropositivity (R+/D+) and 60 had divergent seropositivity status between donor and recipient, with 15 R−/D+ patients and 45 R+/D− patients.
Patient information divided by seropositivity group is shown in Table 1. There were no differences between the groups with respect to demographic or transplant-related characteristics, nor did they differ significantly in type of underlying hematologic disease, disease status at time of transplant, relatedness of transplant donor, or length of follow-up. Following transplantation, recipient/donor chimerism, incidence of aGVHD, and incidence of death in the first 100 days did not differ between the 3 seropositivity groups (data not shown). The incidence of aGVHD of grade II or greater was <10% in all serogroups. The overall incidence of death in all 3 groups in the first 100 days after transplant was 6% and occurred at a median of 64 days after transplantation. Cause of death was primarily because of relapse of underlying hematologic disease, with no cases of death because of CMV disease in the first 100 days. Kaplan-Meier analysis showed no significant difference in survival by serogroup, even when compared to R−/D− patients (data not shown).
Table 1.
Baseline Characteristics of Patients Studied
| R−/D+ n = 15 |
R+/D+ n = 57 |
R+/D− n = 45 |
|
|---|---|---|---|
| Gender | |||
| Men | 11(73.3) | 26 (45.6) | 24 (53.3) |
| Women | 4 (26.7) | 31 (54.4) | 21 (46.7) |
| Underlying disease type | |||
| MDS | 2 (13.3) | 9 (15.8) | 7 (15.6) |
| CLL/CML | 3 (20.0) | 11 (19.3) | 7 (15.6) |
| HD/NHL | 7 (46.7) | 16 (28.1) | 14 (31.1) |
| ALL/ANLL | 3 (20.0) | 21 (36.8) | 17 (37.8) |
| Disease state at transplant* | |||
| Relapse | 3 (21.4) | 9 (15.8) | 8 (18.2) |
| Partial remission | 5 (35.7) | 17 (29.8) | 16 (36.4) |
| Complete remission | 6 (42.9) | 31 (54.4) | 20 (45.4) |
| Donor type | |||
| MRD | 8 (53.3) | 35 (61.4) | 19 (42.2) |
| Haplo/PMRD | 1 (6.7) | 5 (8.8) | 2 (4.4) |
| URD | 6 (40.0) | 17 (29.8) | 24 (53.3) |
| Age | 55 (27–64) | 56 (23–66) | 58 (21–67) |
| Follow-up time (days) | 437 | 518 | 504 |
MDS indicates myelodysplastic syndrome; CLL, chronic lymphocytic leukemia; CML, chronic myelogenous leukemia; HD, Hodgkin’s disease; NHL, non-Hodgkin’s lymphoma; ALL, acute lymphocytic leukemia; ANLL, acute nonlymphocytic leukemia; MRD, matched related donor; PMRD, partially matched related donor: URD, unrelated donor. Patients undergoing HCT following a TLI/ATG-based, nonmyeloablative preparative regimen were analyzed for pretransplant characteristics by CMV serogroup. For each variable presented the number of patients in each group is followed by parenthesis indicating percentage for each serogroup category. Age is expressed in median years followed by range, and follow-up time is expressed as median days. No categories were significantly different between serogroups by Fisher exact for categoric variables and Mann-Whitney U test for continuous variables. The Fisher exact test was used for disease type, relapse status at transplant, and donor type.
Two patients (one R+/D− and 1 R−/D+) had missing information regarding disease state at transplant.
Factors Associated with CMV Reactivation
Quantitative determination of CMV viral load by PCR over time for each patient who experienced either single or multiple episodes of reactivation, segregated by serostatus, are shown in Figure 1. Incidence of viremia during the first 100 days differed significantly in the R−/D+ group compared with the other 2 groups in which the recipients were seropositive. In the R−/D+ group, 13% (2/15) of patients had detectable viremia. In contrast, viremia was detected in 61% (35/57) and 64% (29/45) of the R+/D+ group and the R+/D− group, respectively, which were both significantly greater than the frequency of viremia seen in the R−/D+ group (P = .001 by Fisher’s exact test; odds ratio (OR) of 10.3 (95% confidence interval [CI] 2.1–50.2) for the R+/D+ group and 11.8 (95% CI 2.4–58.9) for the R+/D− group) (Table 2). Two cases of nonfatal CMV disease, 1 in the R+/D+ group and 1 in the R+/D− group, both with gastrointestinal involvement, occurred in the first 100 days posttransplant.
Figure 1.
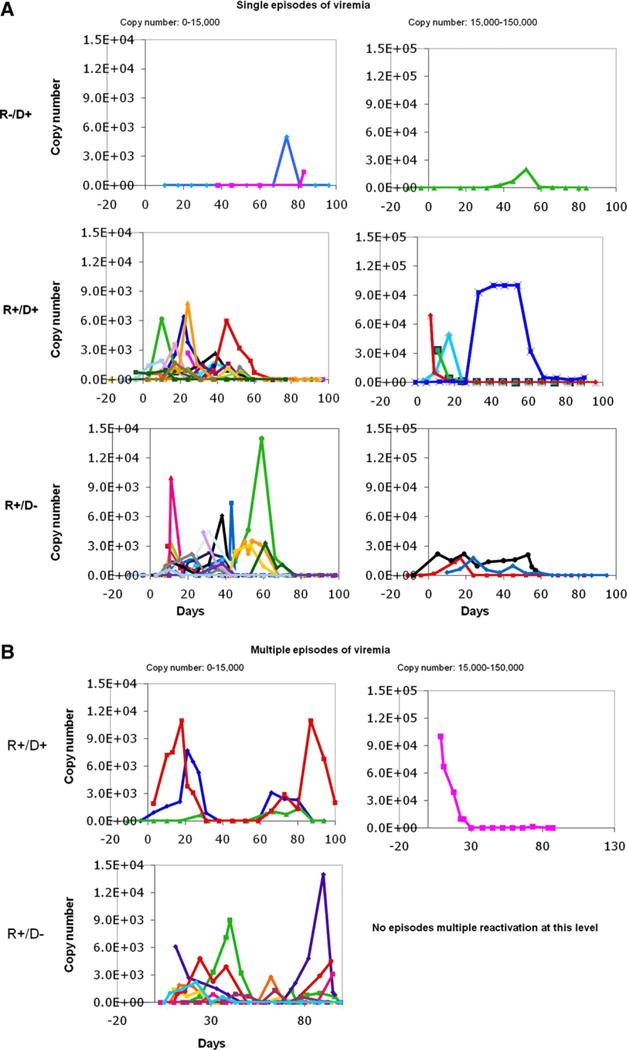
Weekly CMV PCR measurements in the first 100 days posttransplant, separated by serology group and by number of reactivations. CMV PCR results are shown (y-axis), graphed against time after transplantation in days (x-axis). Each patient is represented by a different symbol and color; the left-hand column displays results falling within 0 to 15,000 copy number, and those with 15,000 to 150,000 copy number are displayed in the right-hand column. (A) Patients experiencing single viremia episodes separated by serology group. (B) R+/D+ and R+/D− patients experiencing multiple viremia episodes. No patients in the R−/D+ experienced multiple episodes of viremia, and no patients in the R+/D− group had multiple viremia episodes in the 15,000 to 150,000 range.
Table 2.
Analysis of Factors Affecting CMV Reactivation
| Univariate
|
Multivariate
|
|||
|---|---|---|---|---|
| OR (95% CI) | P-value | OR (95% CI) | P-value | |
| Serogroup | ||||
| R−/D+ | 1.0 (reference group) | N/A | 1.0 (reference group) | N/A |
| R+/D+ | 10.3 (2.1–50.3) | .004* | 23.8 (2.8–202.6) | .004* |
| R+/D− | 11.8 (2.4–58.9) | .003* | 25.7 (3.0–222.2) | .003* |
| Age >55 | 1.5 (.70–3.1) | .319 | ND | |
| Disease type | ||||
| MDS | 1.0 (reference group) | N/A | 1.0 (reference group) | N/A |
| CML/CLL | 1.8 (.50–6.7) | .368 | 3.4 (.77–14.5) | .106 |
| NHL/HD | 2.4 (.73–7.6) | .153 | 4.9 (1.2–119.1) | .023* |
| ALL/AML | 5.5 (1.6–18.1) | .006* | 9.7 (2.5–38.3) | .001* |
| Disease status | ||||
| Remission | 1.0 (reference group) | N/A | ND | |
| Partial remission | .93 (.40–2.1) | .865 | ND | |
| Relapse | .45 (.16–1.3) | .133 | ND | |
| Donor type | ||||
| MRD | 1.0 (reference group) | N/A | ND | N/A |
| Haplo/PMRD | 3.4 (.64–18.2) | .151 | ND | |
| URD | 2.2 (1.0–4.8) | .048* | ND | |
| aGVHD | 4.5 (.93–21.3) | .062 | ND | |
| aGVHD (>1) | 4.1 (.46–36.2) | .204 | ND | |
| Steroid use | 2.2 (.79–6.2) | .132 | ND | |
| Chimerism >95% (d90) | 1.1 (.50–2.2) | .892 | ND |
CI indicates confidence interval; OR, odds ratio; aGVHD, acute graft-versus-host disease; URD, unrelated donor; MRD, matched related donor; PMRD, partially matched related donor; MDS myelodysplastic syndrome; CLL, chronic lymphocytic leukemia; CML, chronic myelogenous leukemia; HD, Hodgkin disease; NHL, non-Hodgkin lymphoma; ALL, acute lymphoblastic leukemia; AML, acute myelogenous leukemia.
In a univariate logistic regression, CMV seropositivity group (R−/D+, R+/D+, or R+/D−), underlying disease, and donor type were all associated with an increased likelihood of CMV reactivation (P < .050). A trend toward increased reactivation was seen with steroid use and aGVHD. Multivariate analysis demonstrated that only CMV seropositivity group and disease type were associated with increased odds of reactivation; adjusted ORs are shown for the variables included in this analysis, ND indicates multivariable analysis was not done.
Statistically significant results compared to relevant reference group (P < .050).
Although various transplant-related risk factors have been associated with CMV viremia, in our analysis only acute lymphoblastic leukemia (ALL) or acute myelogenous leukemia (AML) as an underlying disease, use of an unrelated donor, and recipient seropositivity were statistically associated with increased risk for CMV viremia in univariate analysis. Multivariate analysis was also performed and confirmed the statistically significant increased risk for seropositive patients as well as for disease type. Age over 55 years, disease status at transplantation, donor type, aGVHD, chimerism >95% at day 90, and use of steroids were not associated with an increased risk of viremia during the first 100 days posttransplantation (Table 2).
In the analyses presented before, a clear difference was consistently seen between the R−/D+ group and the 2 R+ groups, as well as a trend toward increased CMV viremia in the R+/D− compared with the R+/D+ group. To quantitatively characterize the number and duration of observed episodes of CMV viremia, the area under the curve (AUC) was calculated for each patient undergoing any CMV reactivation by summing the areas created by multiplying CMV PCR results times the length of time in days between CMV PCR results (Figures 1 and 2). Only PCR results >600 were used for this numeric analysis. The AUC of viremia was greater for the 2 R+ groups when compared to the R−/D+ group (P = .005). Furthermore, the R+/D− group had a greater AUC of viremia than the R+/D+ group (103,000 versus 71,000, P = .049) (Figure 2).
Figure 2.

Area under the curve (AUC) of CMV PCR viremia by serology group. AUC was calculated for each patient by summing the areas generated using the time between CMV PCR results as “length” and the CMV PCR copy number as “height.” Patients in the R−/D+ serology group are indicated by circles, R+/D+ by triangles, and R+/D− by squares. Break in y-axis scale is indicated by a double line. Mann-Whitney U comparison between all 3 groups was statistically significant (P =.005) as was the comparison between the R+/D+ and R−/D+ serotypes (P = .049). Red horizontal bars on scatterplot graph indicate means for each serology group; asterisks (*) indicate statistically significant comparisons.
Timing and Multiplicity of CMV Reactivations
Analysis of the number of reactivation episodes per patient by CMV seropositivity group is shown in Figure 3A. In the R−/D+ group, there were no episodes of multiple reactivations. In the R+/D+ group, 54% experienced single and 7% experienced multiple episodes of reactivation. In contrast, in the R+/D− group 44% of patients had single and 20% had multiple episodes of reactivation. This difference in the number of episodes of viremia was statistically significant when all 3 serogroups were compared (P = .002 by Fisher’s exact test), but statistical significance was not seen between the R+/D− and R+/D+ groups (P = .15 by Fisher’s exact test).
Figure 3.

Percentage of patients experiencing CMV viremia and days to first episode by serology group. (A) Percentage of patients with either 0, 1, or >1 episodes of CMV viremia within each serology group are indicated by white, gray, and black bars, respectively. A maximum of 2 episodes of viremia per patient were seen in the R+/D+ group, and a maximum of 3 episodes of viremia per patient were seen in the R+/D− group. The number of episodes by serology type was statistically significantly different between all 3 groups (P = .002, Fisher’s exact test). Median time in days to reactivation is indicated below the bar graph. There was a trend toward shorter time to reactivation in the R−/D+ group (P = .11) compared with the other 2 serology groups. Range of days to reactivation was 38 to 74 days in the R−/D+ group, −4 to 53 days in the R+/D+ group, and −11 to 46 days in the R+/D− groups. (B) Sixty-six patients who experienced 1 or more episodes of CMV viremia were grouped by single versus multiple episode status (regardless of serology group). Horizontal bars within the scatter plot indicate the median number of days to first detectable CMV PCR, which was significantly different between the 2 groups (P =.008). The median day to first episode of viremia was 17 (range: −4 to 74) in the single episode of viremia group (n = 53), versus 10 (range: −11 to 31) in the multiple episode group (n = 13). Asterisk (*) indicates statistically significant comparisons.
Also observed was a trend toward an earlier median day of reactivation posttransplantation in the R+/D+ and R+/D− groups compared with the R−/D+ group (P= .11), with a median time of first positive CMV PCR at 56 days posttransplant for the R−/D+ serogroup compared with 17 days for the R+/D+ patients and 11 days for the R+/D− patients (Figure 3A).
A strong association was seen between multiple episodes of viremia and an earlier occurrence of viremia posttransplantation: For patients experiencing a single episode of viremia, the median number of days to first positive PCR result was 17, whereas for patients who eventually experienced multiple episodes of viremia, the first detectable episode of viremia occurred at a median of 10 days after transplant (P = .008) (Figure 3B).
Flow Cytometric Analysis of T Cell Subsets
Analysis of absolute lymphocyte counts (ALC) and CD3+, CD4+, CD8+, and CD19+ T cell and natural killer (NK) cell subsets by flow cytometry were performed within 2 weeks of the target time points of 25, 56, and 90 days after transplant, excluding flow cytometry results for patient with a diagnosis of CLL or with relapsed or progressive disease prior to day 56 after transplant, for an average of 61% of the patients in each seropositivity group (Figure 4). There were no differences in CD19+ counts between groups at any time point. By day +90, patients in both R+ groups had significantly higher ALC and subset counts than the patients in the R− groups (Figure 4). In the R−/D− group, absolute NK cells were significantly higher than those from the R+/D− group at days 56 and 90, CMV viremia during the first 100 days posttransplant was associated with a significant trend toward lower cell ALC, CD3+, CD4+, and CD8+ counts; for the NK cell population this trend was observed primarily at day +28 (Figure 5). However, this observation was most striking in certain serogroups; for example, we did note that the median CD4+ counts were significantly lower in the R+/D+ patients who had an episode of viremia when compared to their cohorts in whom CMV viremia had not been detected (P = .014).
Figure 4.

Flow cytometry results by day after transplant and serology group. Means 6 standard error of absolute cell count for each cell type are graphed with absolute lymphocyte (ALC) in black, CD3+ with dots (CD3+), CD4+ in lines (CD4+), and CD8+ in gray (CD8+) at day 28, day 56, and day 90 after transplant. The number of patients with evaluable results within each serology group are shown below. Patient results were not included if the patient had a diagnosis of CLL as their underlying disease or if they experienced documented relapse prior to day 56 posttransplant. Means that were statistically significantly higher than the other groups are indicated by single asterisk (*); means that were statistically significantly than the 2 R− groups are indicated by a double asterisk (**). Statistical analysis performed using the Mann-Whitney U test. Pound sign (#) indicates a mean that was statistically significantly higher than the R+/D− group.
Figure 5.
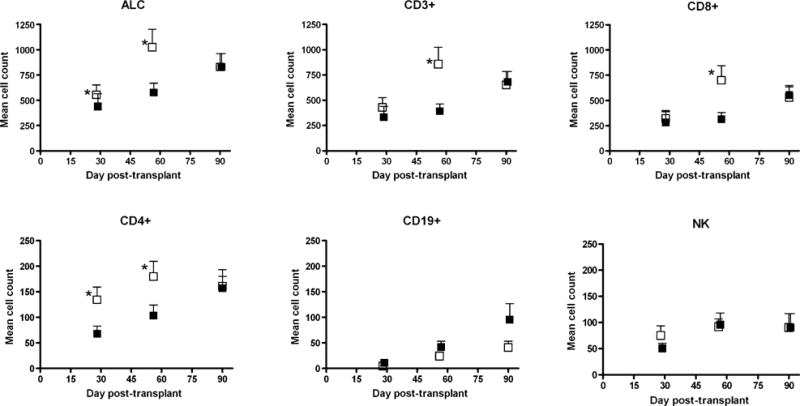
Mean cell count by cell type and experience of CMV viremia. Mean cell counts are indicated by squares with flanking standard error bars for each cell type at days 28, 56, and 90 posttranplantation. Means of cell counts from patients who experienced 1 or more episodes of CMV viremia are shown by black squares; those without CMV viremia in the first 100 days after transplant are shown by open squares. Y-axis scale is smaller for the CD4+ and CD19+ values compared with the other cell types. Asterisks indicated statistically significant difference by Mann-Whitney U test.
DISCUSSION
This analysis of CMV viremia after NMA allogeneic HCT using a preparative regimen of TLI and ATG revealed several important findings that reflect the factors that influence functional immune reconstitution and control of viremia. Following TLI-ATG, the median time to onset of CMV viremia was <18 days, in contrast to 60 days following NMA preparative regimens that included fludarabine and total body irradiation [7,8]. After MA preparative regimens, median time to CMV viremia has frequently been reported to be around 30 days posttransplant [9–11].
Of potentially greater significance is that the earlier reactivation seen following this TLI-ATG preparative regimen is correlated with a significant risk of multiple episodes of viremia. In contrast to NMA preparative regimens that did not include ATG [7], 18% of our patients in the R+/D− group experienced multiple reactivations despite a minimum of 3 weeks of antiviral therapy and confirmation of clearance of viremia.
Other reports of HCT following NMA preparative regimens containing ATG noted the potential for recurrent CMV disease [12,13]. However, both reports summarize a series of patients at a single center who received a preparative regimen including fludarabine, busulfan, and ATG at varying doses on days −4 to −1. In the more recent of the 2 analyses, the 14.2% of 84 patients had multiple viremias following the initial viremia that occurred at a median of 37 days HCT. There were no analyses regarding the effect of time of reactivation on recurrent viremia. As may be predicted, the earlier administration of TLI and ATG (days −11 through −7) resulted in viremia occurring earlier during the initial period of immune reconstitution (median day +17 and day +11 for the R+/D+ and R+/D− patients, respectively). Furthermore, the association between early viremia and the risk of subsequent episodes of viremia suggests that a degree of tolerance may have resulted because of viral antigen exposure to differentiating T cells.
The inclusion of alemtuzumab as part of an NMA preparative regimen has been shown to result in even higher rates of recurrence of CMV viremia (54%–90%) despite the fact that initial viremia occurred significantly later than in our series [14,15]. These observations are consistent with a more profound impact on both numbers and recovery of lymphocytes and CD4+, CD8+, CD19+, and NK cells as a result of alemtuzumab when compared to ATG [16].
To further understand the association between CMV and immune reconstitution, we analyzed the temporal sequence of quantitative lymphocyte recovery. The critical role of CMV-specific CD4+ and CD8+ T cells in functional viral control has been well documented and low levels of total as well as CMV-specific CD4+ and CD8+ cells are associated with reactivation [3,4]. Transfer of either CD4+ or CD8+ CMV-specific cells has been shown to boost immunity and allow control of CMV infection in patients not responding to antiviral therapy alone, and trials are currently underway to test the impact of transferring both CD4+ and CD8+ cells simultaneously [17]. Additionally, after CMV reactivation, increases in CMV-specific CD4+ and CD8+ populations have been shown to be associated with successful control of CMV [4,18,19]. Although our study is limited by the absence of data regarding CMV-specific lymphocyte responses, viremia was associated with lower ALC, CD3+, CD4+, and CD8+ counts at day 56, and a trend toward lower NK cell counts at day 28 (Figure 5). However, because our flow cytometry measurement began at day 28 after transplant it is not possible to distinguish whether depressed lymphocyte levels might be a contributing risk factor for or a consequence of viremia in the early posttransplant period.
It is unclear why patients in the R+/D+ group had significantly higher ALC and lymphocyte subsets when compared with the other groups independent of detectable CMV viremia (Figure 4). As this group of patients demonstrated improved viral control following HCT, it is reasonable to hypothesize that a synergistic interaction between CMV-specific CD4+ and CD8+ cells from both donor and recipient may lead to enhanced lymphocyte expansion after transplantation. Alternatively, other as of yet unidentified factors may result in enhanced lymphocyte reconstitution in these patients.
The association we observed between viremia during the early period of immune reconstitution and the appearance of multiple episodes of reactivation despite appropriate antiviral treatment suggests an impaired immune control of CMV replication, especially in the R+/D− setting. A similar situation may be seen in children congenitally infected with CMV who are exposed to the virus during a period of immune system development and exhibit a selective defect in immune control of CMV compared with other herpesvirues, leading to persistent shedding of virus in saliva and urine [20]. More recent studies have suggested that this lack of effective immune control may be because of decreased interferon-γ response from CD4 T cells in neonates compared with infected adults [21,22]. In prospective analyses, we are also studying potentially important factors influencing reconstitution of antiviral immunity such as differences in ATG pharmacokinetics, chimerism at earlier timepoints, and NK-T cell populations.
Patients with lower CMV-specific lymphocyte responses as quantitated by tetramer and functional assays have been shown to be more likely to experience recurrent viremia [19]. We are now prospectively analyzing the timing of CMV viremia on the kinetics of the reconstitution of quantitative and functional CMV-specific immunity following TLI-ATG.
The contributory role of donor seropositivity remains controversial. Donor seropositivity is not always salutary, as some observed decreased CMV viremia, disease, and patient survival, whereas others reported the opposite effect [1,2]. In our study, the relative contribution of donor seropositivity depended largely on the serostatus of the recipient. In seronegative patients who received grafts from CMV seropositive donors, a single episode of low grade viremia occurred later posttransplant and resolved promptly in association with preemptive therapy. In seropositive patients, however, we found that donor CMV serostatus also had an important influence on control of CMV replication with increased efficiency of immunologic control in the R+/D+ group as reflected by the lower viral load AUC compared with the R+/D− group. This observation suggests that, in addition to the hematopoietic progenitor cells transferred in the graft, mature T cells with CMV-specific activity were also present, leading to expansion and better control of infection after exposure to CMV antigen. The transfer of CMV-specific cells has been demonstrated in MA allogeneic HCT [23].
Other patient factors found in our study to play an important role in control of CMV reactivation after NMA transplant were donor relatedness and underlying hematologic disease. aGVHD also demonstrated a trend toward increased rate of CMV viremia. There may be a direct connection between immune dysregulation caused by GVHD and lack of immune control for CMV, or it may be possible that the presence of GVHD was a marker for high dose steroid use, which has been shown to contribute to CMV reactivation and disease [3]. Increased incidence of CMV reactivation and disease has also been shown previously to be influenced by donor relatedness, with increased burden of infection in patients receiving unrelated donor grafts [1]. In our study, consistent with previous reports, aGVHD is a rare event; therefore, the observations we have made regarding antiviral immune reconstitution are of particular significance as they occurred largely in the absence of this confounding factor. Because there were relatively few patients with haploidentical or partially matched donors, the effect of donor mismatch could not be fully assessed in this study (Table 2).
Regarding underlying disease, however, it is unclear why an increase in odds of reactivation was observed in the patient with ALL and AML, particularly as there was no association between disease status at transplant and viremia, although this may have been a chance observation. If a true association, it may reflect the contribution of recipient immunity to control of CMV replication in the early posttransplant period in the NMA situation where the patient’s native immune system persists for some time as reflected by the measurements of chimerism showing persistent recipient cells at day 90 after transplant. It is possible that residual T cells in leukemic patients are less able to contribute to functional control of virus during the early period of immune reconstitution. Alternatively, the lymphocyte-targeting therapies for ALL that patients receive prior to transplantation may influence viral control; however, we lack sufficient numbers of patients to determine the impact of this potential factor.
In addition to the limitations of this study as a retrospective analysis, the standard practice of administering preemptive antiviral treatment at the first evidence of viremia precludes a study of the natural history of CMV reactivation and immune control. The AUC calculation, although useful in studies of CMV replication [24], is somewhat difficult to interpret in the setting of antiviral administration, although the patients in this cohort were all treated for CMV according to a standardized protocol.
In addition, because the seropositive patients uniformly experienced early CMV viremia, it is difficult to separate the contribution of early viral exposure and recipient CMV serostatus in control of viral replication. Further studies in a mouse model of allogeneic transplant will allow the dissection of factors that contribute to an effective and durable immune response in the setting of NMA preparative regimens. Furthermore, these experiments would permit analysis of the relative contributions of host-versus-donor-derived cells in antiviral responses.
As transplant protocols continue to evolve, so will the understanding of the complex modulation of immune reconstitution. Our findings uniquely demonstrate the dynamic nature of immunity in relation to CMV antigen exposure in the complex environment resulting from NMA conditions where both residual recipient as well as donor immune response may contribute to viral control. Continued study of this common posttransplant viral infection will provide insight into the recapitulation of a functional immune system after allogeneic HCT in the setting of recipient immunomodulation.
Acknowledgments
The authors thank Raymond Balise for helpful input regarding database construction and statistical programming. This work was supported in part by National Institutes of Health funding (Grant 2P01 CA 049605-19).
Footnotes
Financial disclosure: The authors have no conflicts of interest to report.
References
- 1.Boeckh M, Nichols WG, Papanicolaou G, Rubin R, Wingard JR, Zaia J. Cytomegalovirus in hematopoietic stem cell transplant recipients: current status, known challenges, and future strategies. Biol Blood Marrow Transplant. 2003;9:543–558. doi: 10.1016/s1083-8791(03)00287-8. [DOI] [PubMed] [Google Scholar]
- 2.Lilleri D, Fornara C, Chiesa A, Caldera D, Alessandrino EP, Gerna G. Human cytomegalovirus-specific CD4+ and CD8+ T-cell reconstitution in adult allogeneic hematopoietic stem cell transplant recipients and immune control of viral infection. Haematologica. 2008;93:248–256. doi: 10.3324/haematol.11912. [DOI] [PubMed] [Google Scholar]
- 3.Hakki M, Riddell SR, Storek J, et al. Immune reconstitution to cytomegalovirus after allogeneic hematopoietic stem cell transplantation: impact of host factors, drug therapy, and subclinical reactivation. Blood. 2003;102:3060–3067. doi: 10.1182/blood-2002-11-3472. [DOI] [PubMed] [Google Scholar]
- 4.Cwynarski K, Ainsworth J, Cobbold M, et al. Direct visualization of cytomegalovirus-specific T-cell reconstitution after allogeneic stem cell transplantation. Blood. 2001;97:1232–1240. doi: 10.1182/blood.v97.5.1232. [DOI] [PubMed] [Google Scholar]
- 5.Lowsky R, Takahashi T, Liu PL, et al. Protective conditioning for acute graft-versus-host disease. N Engl J Med. 2005;353:1321–1331. doi: 10.1056/NEJMoa050642. [DOI] [PubMed] [Google Scholar]
- 6.Kohrt HE, Turnbull BB, Heydari K, et al. TLI and ATG conditioning with low risk of graft-versus-host disease retains anti-tumor reactions after allogeneic hematopoietic cell transplantation from related and unrelated donors. Blood. 2009;114:1099–1109. doi: 10.1182/blood-2009-03-211441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Junghanss C, Boeckh M, Carter RA, et al. Incidence and outcome of cytomegalovirus infections following nonmyeloablative compared with myeloablative allogeneic stem cell transplantation, a matched control study. Blood. 2002;99:1978–1985. doi: 10.1182/blood.v99.6.1978. [DOI] [PubMed] [Google Scholar]
- 8.Nakamae H, Kirby KA, Sandmaier BM, et al. Effect of conditioning regimen intensity on CMV infection in allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2009;15:694–703. doi: 10.1016/j.bbmt.2009.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boeckh M, Gooley TA, Myerson D, Cunningham T, Schoch G, Bowden RA. Cytomegalovirus pp65 antigenemia-guided early treatment with ganciclovir versus ganciclovir at engraftment after allogeneic marrow transplantation: a randomized double-blind study. Blood. 1996;88:4063–4071. [PubMed] [Google Scholar]
- 10.Yanada M, Yamamoto K, Emi N, et al. Cytomegalovirus antigenemia and outcome of patients treated with pre-emptive ganciclovir: retrospective analysis of 241 consecutive patients undergoing allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2003;32:801–807. doi: 10.1038/sj.bmt.1704232. [DOI] [PubMed] [Google Scholar]
- 11.Schetelig J, Oswald O, Steuer N, et al. Cytomegalovirus infections in allogeneic stem cell recipients after reduced-intensity or myeloablative conditioning assessed by quantitative PCR and pp65-antigenemia. Bone Marrow Transplant. 2003;32:695–701. doi: 10.1038/sj.bmt.1704164. [DOI] [PubMed] [Google Scholar]
- 12.Mohty M, Faucher C, Vey N, et al. High rate of secondary viral and bacterial infections in patients undergoing allogeneic bone marrow mini-transplantation. Bone Marrow Transplant. 2000;26:251–255. doi: 10.1038/sj.bmt.1702509. [DOI] [PubMed] [Google Scholar]
- 13.Mohty M, Jacot W, Faucher C, et al. Infectious complications following allogeneic HLA-identical sibling transplantation with antithymocyte globulin-based reduced intensity preparative regimen. Leukemia. 2003;17:2168–2177. doi: 10.1038/sj.leu.2403105. [DOI] [PubMed] [Google Scholar]
- 14.Chakrabarti S, Mackinnon S, Chopra R, et al. High incidence of cytomegalovirus infection after nonmyeloablative stem cell transplantation: potential role of Campath-1H in delaying immune reconstitution. Blood. 2002;99:4357–4363. doi: 10.1182/blood.v99.12.4357. [DOI] [PubMed] [Google Scholar]
- 15.Park SH, Choi SM, Lee DG, et al. Infectious complications associated with alemtuzumab use for allogeneic hematopoietic stem cell transplantation: comparison with anti-thymocyte globulin. Transplant Infect Dis. 2009;11:413–423. doi: 10.1111/j.1399-3062.2009.00414.x. [DOI] [PubMed] [Google Scholar]
- 16.Juliusson G, Theorin N, Karlsson K, Frodin U, Malm C. Subcutaneous alemtuzumab vs ATG in adjusted conditioning for allogeneic transplantation: influence of Campath dose on lymphoid recovery, mixed chimerism and survival. Bone Marrow Transplant. 2006;37:503–510. doi: 10.1038/sj.bmt.1705263. [DOI] [PubMed] [Google Scholar]
- 17.Einsele H, Kapp M, Grigoleit GU. CMV-specific T cell therapy. Blood Cells Mol Dis. 2008;40:71–75. doi: 10.1016/j.bcmd.2007.07.002. [DOI] [PubMed] [Google Scholar]
- 18.Foster AE, Gottlieb DJ, Sartor M, Hertzberg MS, Bradstock KF. Cytomegalovirus-specific CD4+ and CD8+ T-cells follow a similar reconstitution pattern after allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2002;8:501–511. doi: 10.1053/bbmt.2002.v8.pm12374455. [DOI] [PubMed] [Google Scholar]
- 19.Moins-Teisserenc H, Busson M, Scieux C, et al. Patterns of cytomegalovirus reactivation are associated with distinct evolutive profiles of immune reconstitution after allogeneic hematopoietic stem cell transplantation. J Infect Dis. 2008;198:818–826. doi: 10.1086/591185. [DOI] [PubMed] [Google Scholar]
- 20.Pass RF, Stagno S, Britt WJ, Alford CA. Specific cell-mediated immunity and the natural history of congenital infection with cytomegalovirus. J Infect Dis. 1983;148:953–961. doi: 10.1093/infdis/148.6.953. [DOI] [PubMed] [Google Scholar]
- 21.Tu W, Chen S, Sharp M, et al. Persistent and selective deficiency of CD4+ T cell immunity to cytomegalovirus in immunocompetent young children. J Immunol. 2004;172:3260–3267. doi: 10.4049/jimmunol.172.5.3260. [DOI] [PubMed] [Google Scholar]
- 22.Hassan J, Dolley S, Hall W. Immunological response to cytomegalovirus in congenitally infected neonates. Clin Exp Immunol. 2007;147:465–471. doi: 10.1111/j.1365-2249.2007.03302.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li CR, Greenberg PD, Gilbert MJ, Goodrich JM, Riddell SR. Recovery of HLA-restricted cytomegalovirus (CMV)-specific T-cell responses after allogeneic bone marrow transplant: correlation with CMV disease and effect of ganciclovir prophylaxis. Blood. 1994;83:1971–1979. [PubMed] [Google Scholar]
- 24.Limaye AP, Kirby KA, Rubenfeld GD, et al. Cytomegalovirus reactivation in critically ill immunocompetent patients. JAMA. 2008;300:413–422. doi: 10.1001/jama.300.4.413. [DOI] [PMC free article] [PubMed] [Google Scholar]


