Abstract
This report describes, for the first time in small animal literature, the spontaneous resorption of herniated Hansen type I intervertebral disc material in the cervical spine of a chondrodystrophic dog over a 4-month period, documented by magnetic resonance imaging. Clinical signs (cervical hyperpathia) responded to conservative treatment during the same period.
Résumé
Résorption spontanée d’une hernie discale chez un chien détectée par imagerie par résonance magnétique. Cet article décrit, pour la première fois dans la littérature des petits animaux, la résorption spontanée d’une hernie Hansen de type I du matériel du disque intervertébral dans la colonne cervicale d’un chien chondrodystrophique pendant une période de 4 mois et documentée par imagerie par résonance magnétique (IRM). Les signes cliniques (hyperpathie cervicale) ont répondu à un traitement conservateur durant la même période.
(Traduit par Isabelle Vallières)
Cervical disc herniation is reported in 15% of dogs that suffer from intervertebral disc (IVD) herniation (1–3). A total of 15% to 61% of dogs that suffer from IVD herniation in the cervical spine present with signs of cervical hyperpathia (3,4), guarding of the neck and muscle fasciculations without neurological deficits (1,2). The lower rate of neurological deficits compared to patients with thoracolumbar IVD herniation is thought to be related to the high vertebral canal/spinal cord ratio of the cervical vertebral column (1–4).
Treatments for IVD extrusion in dogs include conservative management (strict avoidance of activity and the administration of anti-inflammatory and/or other analgesic medications) (1,2,4–6) and surgical decompression (1,2,4,5). The choice between conservative and surgical management depends on the severity of neurological dysfunction, the degree of neuronal tissue compression apparent on imaging, and financial considerations (1–7). Most neurologists agree that an initial period of conservative management is appropriate for patients that present with cervical hyperpathia (1,2,4,6).
Spontaneous resorption of degenerate nuclear material within the vertebral canal has not been documented by advanced diagnostic imaging [Computed Tomography (CT)/Magnetic Resonance Imaging (MRI)] in previous reports. The current report describes the MRI-documented spontaneous disappearance of an acute cervical intervertebral disc herniation in 1 patient which was treated conservatively. Relevant cases reported in the human literature are reviewed and the biochemical and biological factors that may be involved in the resorption of herniated cervical intervertebral disc material are discussed.
Case description
A 4-year-old, 14-kg, spayed female, cocker spaniel dog was referred to a clinic with a history of cervical hyperpathia and exercise intolerance of acute onset approximately 4 mo previously. At first examination the local veterinary surgeon had recorded a low head carriage, stiffness affecting all 4 limbs, guarding of the neck, and occasional diffuse muscular tremors affecting the neck and both forelimbs. Management had been conservative, involving cage rest and anti-inflammatory medication [gabapentin (Gabapentin Zentiva; Zentiva, Guildford, Surrey, UK)], 10 mg/kg body weight (BW), PO, q12h for the initial 2 wk.
According to the dog’s owner, the signs of neck pain had responded well during this period. However, following discontinuation of the medication the dog had shown a recurrence of signs of neck pain, again without any evidence of motor or sensory deficits.
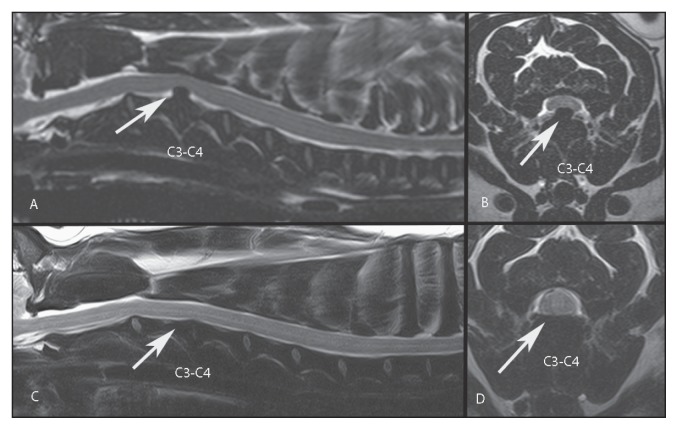
Because of difficulties accessing a referral center a second opinion was delayed and magnetic resonance imaging (MRI) was carried out at the local veterinary practice using a mobile 1.5-Tesla MRI scanner (Philips, Guildford, Surrey, UK). The study included T2-weighted (T2W) and T1-weighted (T1W) sagittal and transverse sequences. The resulting MRI images showed a moderate quantity of well-defined T2W hypointense, T1W hypointense extradural material, located dorsal to the C3–C4 IVD space that extended cranially and caudally over a total length of approximately 7.4 mm. This material was located ventrally and slightly lateral to the right side of the vertebral canal and reduced its cross-sectional diameter by ~25% at the level of its greatest severity. There was an associated loss of both the normal spinal fluid signal of the subarachnoid space and the normal epidural fat signal. The C3–C4 IVD exhibited a marked reduction in T2W signal, indicating degeneration. Subjective narrowing of the C3–C4 IVD was observed (Figures 1A, B). Hansen type I C3–C4-disc extrusion was diagnosed based on the imaging and clinical signs.
Figure 1.
T2-weighted magnetic resonance imaging (MRI). A — Sagittal view. B — Transverse view. Note the T2-weighted hypointense extradural material that is located above the C3–C4 intervertebral disc space and causing mild focal ventral compression of the spinal cord at the level of C3–C4 intervertebral disc space (arrow). Notably, the normal CSF signal of the subarachnoid space and the normal epidural fat signal in the ventral and dorsal aspect of the spinal cord at the compression site have been lost. The C3–C4 intervertebral disc exhibits a marked reduction in T2-weighted signal, indicating degeneration. The narrowing of the C3–C4 intervertebral disc space is observed. C — Sagittal view. D — Transverse view of the cervical vertebral column of the dog depicted in parts A and B, obtained 4 months after the previous MRI. Notably, the intervertebral disc herniation and compression characterized by the presence of spinal fluid and an epidural fat signal (T2W hyperintense) dorsal and ventral to the spinal cord have resolved (arrow). The changes described earlier in the C3–C4 intervertebral disc and the intervertebral disc space remain present.
Conservative treatment consisting of strict cage rest, anti-inflammatory meloxicam (Metacam; Boehringer Ingelheim, Vetmedica GmbH, Ingelheim/Rhein, Germany), 0.1 mg/kg BW, PO, q24h and pain relief medication gabapentin (Gabapentin Zentiva; Zentiva), 10 mg/kg BW, PO, q12h was reintroduced.
Four months later the dog was referred to our practice for investigation. According to the client, the signs of neck pain were well-controlled with the prescribed treatment, which had been administered continuously in the interim, and the dog had been more lively and active, particularly during the previous month. Anti-inflammatory and pain relief medications had been discontinued for 24 h prior to the consultation. A neurological examination was conducted by a board-certified neurologist. Clinical notes from this examination record that the dog was bright and alert. The neurological examination revealed no sensory, motor, or proprioceptive deficits. Gait and posture were within normal limits. There was no evidence of resentment to palpation of the cervical spine.
As 4 mo had passed since the first MRI study at the local veterinary practice, a follow-up MRI investigation was recommended. The MRI was carried out using a 1.5 Tesla MRI scanner (Petvet Hallmarq, Surrey, UK). The following sequences were acquired: sagittal T2W Half-Fourier Acquisition Single-Shot Turbo Spin-Echo (HASTE) (TR 9000, TE 192, slice thickness 2.5 mm), T2W images in the sagittal (TR 2500, TE 110, slice thickness 2.5 mm) and transverse (TR 2522, TE 110, slice thickness 3.5 mm) planes and T1W images in the sagittal (TR 300, TE 17, slice thickness 2.5 mm) and transverse (TR 300, TE 17 slice thickness 3.5 mm) planes. The MRI images revealed complete resolution of the previously described IVD herniation. The changes described in the C3–C4 IVD and this IVD space remained present (Figures 1C, D).
Given the MRI study findings and the dog’s clinical improvement, medical treatment was gradually discontinued over the course of 2 wk. At telephone follow-ups 3 mo and 2 y following the second MRI investigation the clients reported no recurrence of clinical signs of cervical hyperpathia.
Discussion
Limited information is available on the clinical success of conservative treatment for suspected or confirmed IVD herniation in dogs (1,2,6) with recurrence rates reported at 33% to 36%. This report provides the first description of spontaneous resorption of Hansen type I disc extrusion in small animals at the level of the cervical spine that has been documented using MRI.
Only 1 previous veterinary case on small animals reported resolution of a Hansen type I intervertebral disc extrusion at the level of the lumbar spine detected using MRI (8). In contrast, the human literature has several reports and case series of spontaneous regression of herniated lumbar and cervical IVDs (9–19). The difficulty in justifying follow-up advanced imaging in patients which have returned to a healthy clinical status may be a contributing factor to this disparity. The ability to perform advanced imaging in humans without the associated risks of anesthesia, makes it feasible to perform safe standardized follow-up protocols documenting the extent of any disc reabsorption, while in dogs the follow-up is often limited to a clinical examination only.
The previous veterinary case documented a 3-year-old French bulldog that was presented with hyperpathia, neurological deficits, and spastic paraparesis associated with MRI findings consistent with IVD extrusion at the level of the L3–L4 intervertebral disc space (8). According to the report there was a dramatic improvement in the clinical signs (mild residual hind limbs ataxia) over approximately 5 wk and a significant reduction of the extruded intervertebral disc material was documented at a recheck MRI study. Similarities were detected with the case described in our study: both dogs affected were young adults (3- to 4-years-old) and belonging to a chondrodystrophic type breed of dogs.
In the human literature, although the spontaneous regression of herniated IVD material at the level of the lumbar spine documented by imaging has been well-established (9–14), this outcome has less frequently been reported for herniated discs at the level of the cervical spine (15–19).
The mechanism by which herniated IVD material undergoes spontaneous regression remains incompletely understood, although multiple factors related to the resorption process have been identified and described in humans (9–24) and small animals (5,25). The first (inflammatory) phase is triggered by the traumatic rupture of the dorsal annulus fibrosus causing contusion and compression of the spinal cord, local vasoconstriction followed by vasodilation and inflammation. A second phase is characterized by new blood vessel formation extending into the cellular matrix of the herniated intervertebral disc material; and by development of a strong inflammatory process, including invasion neutrophils followed by macrophages. These phagocytic cells are considered essential for removal of cellular debris and extruded IVD material (17–21).
In humans the type of disc herniation can also affect the resorption process, with spontaneous regression observed more frequently for extruded discs (Hansen type I) than for protruded discs (Hansen type II) (14–17,19). It has been suggested that this may relate to the vascularity of the tissues involved. Only the outer layer of the annulus fibrosus and the dorsal longitudinal ligament are believed to be directly supplied by blood vessels, whereas the nucleus pulposus and most of the annulus fibrosus receive nutrition by diffusion from the cartilaginous end plates (1,5,26). Extruded disk material that extends through the ruptured dorsal longitudinal ligament is therefore resorbed more effectively than protruded disc material that is confined between the layers of the annulus fibrosus, where there is a profound lack of vascularization.
These findings are consistent with reports in the small animal literature that have described repeated MRI examinations following the conservative treatment of dogs with Hansen type II IVD — associated cervical spondylomyelopathy that indicated that the protruding disc material does not decrease in size (27–28). In contrast, the Hansen type I disc extrusion previously reported in the lumbar region (8) and the one detailed in this case report spontaneously resorbed.
Studies in humans have suggested a correlation between the size of a cervical disc herniation and the likelihood of spontaneous resorption, with larger volume herniated discs being observed to regress more rapidly than smaller ones. This finding has been consistently reported by various authors and the common conclusion was that larger volumes of extruded disc material and sequestrated-type disc herniations were more likely to regress. This was postulated to be associated with the severity of the inflammatory reactions, and, as such, the strength of the neovascularization and phagocytic response induced at the level of the epidural space (9–15,17–21).
In evaluations of neovascularization, enhancement with the MRI contrast agent gadolinium-diethylenetriamine penta-acetic acid (Gd-DTPA) (Magnevist; Bayer Schering Pharma, Leverkusen, Germany) has been reported to be a good indicator for predicting the probability that a herniated disc will spontaneously regress. The extension and thickness of rim enhancement in MRI is reported to be greater in extruded and sequestrated discs that are spontaneously resorbed over time (20). Unfortunately, in the present report the contrast medium was not administered to the patient during the initial MRI study; therefore, the information that could have been obtained using this medium is missing.
This report demonstrates the spontaneous regression of herniated cervical disc material in a chondrodystrophic dog and the associated resolution of clinical signs of cervical hyperpathia.
Acknowledgment
The authors thank the team at New Era Veterinary Hospital (Jersey) for their collaboration in the management of the reported case and for providing consent for our use of the initial MRI images obtained at their practice. CVJ
Footnotes
Use of this article is limited to a single copy for personal study. Anyone interested in obtaining reprints should contact the CVMA office (hbroughton@cvma-acmv.org) for additional copies or permission to use this material elsewhere.
References
- 1.Brisson BA. Intervertebral disc disease in dogs. Vet Clin North Am Small Anim Pract. 2010;40:829–858. doi: 10.1016/j.cvsm.2010.06.001. [DOI] [PubMed] [Google Scholar]
- 2.Lorenz MD, Coates JR, Kent R. Handbook of Veterinary Neurology. 5th ed. St. Louis, Missouri: Saunders; 2011. pp. 164–188. [Google Scholar]
- 3.Cherrone KL, Dewey CW, Coates JR, Bergman RL. A retrospective comparison of cervical intervertebral disk disease in nonchondrodystrophic large dogs versus small dogs. J Am Anim Hosp Assoc. 2004;40:316–320. doi: 10.5326/0400316. [DOI] [PubMed] [Google Scholar]
- 4.Sharp NJH, Wheeler SJ. Small Animal Spinal Disorders: Diagnosis and Surgery. 2nd ed. Philadelphia, Pennsylvania: Mosby; 2005. Thoracolumbar disc disease; p. 125. [Google Scholar]
- 5.Jeffery ND, Levine JM, Olby NJ, Stein VM. Intervertebral disk degeneration in dogs: Consequences, diagnosis, treatment, and future directions. J Vet Intern Med. 2013;27:1318–1333. doi: 10.1111/jvim.12183. [DOI] [PubMed] [Google Scholar]
- 6.Levine JM, Levine GJ, Johnson SI, Kerwin SC, Hettlich BF, Fosgate GT. Evaluation of success of medical management for presumptive cervical intervertebral disk herniation in dogs. Vet Surg. 2007;36:492–499. doi: 10.1111/j.1532-950X.2007.00296.x. [DOI] [PubMed] [Google Scholar]
- 7.Olby N, Harris T, Burr J, Munana K, Sharp N, Keene B. Recovery of pelvic limb function in dogs following acute intervertebral disc herniations. J Neurotrauma. 2004;21:49–59. doi: 10.1089/089771504772695940. [DOI] [PubMed] [Google Scholar]
- 8.Steffen F, Kircher PR, Dennler M. Spontaneous regression of lumbar Hansen type 1 disc extrusion detected with magnetic resonance imaging in a dog. J Am Vet Med Assoc. 2014;244:715–718. doi: 10.2460/javma.244.6.715. [DOI] [PubMed] [Google Scholar]
- 9.Maigne JY, Deligne I. Computed tomographic follow-up study of 48 cases of nonoperatively treated lumbar intervertebral disc herniation. Spine. 1992;17:1071–1074. doi: 10.1097/00007632-199209000-00010. [DOI] [PubMed] [Google Scholar]
- 10.Saal JA, Saal JS, Herzog RJ. The natural history of lumbar intervertebral disc extrusions treated nonoperatively. Spine. 1990;15:683–686. doi: 10.1097/00007632-199007000-00013. [DOI] [PubMed] [Google Scholar]
- 11.Benson RT, Tavares SP, Robertson SC, Sharp R, Marshall RW. Conservatively treated massive prolapsed discs: A 7-year-follow-up. Ann Royal Coll Surg Engl. 2010;92:147–153. doi: 10.1308/003588410X12518836438840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bozzao A, Gallucci M, Masciocchi C, Aprile I, Barile A, Passariello R. Lumbar disk herniation: MR imaging assessment of natural history in patients treated without surgery. Radiology. 1992;185:135–141. doi: 10.1148/radiology.185.1.1523297. [DOI] [PubMed] [Google Scholar]
- 13.Henmi T, Sairyo K, Nakano S, et al. Natural history of extruded lumbar intervertebral disc herniation. J Med Invest. 2002;49:40–43. [PubMed] [Google Scholar]
- 14.Kim ES, Oladunjoye AO, Li JA, Kim KD. Spontaneous regression of herniated lumbar discs. J Clin Neurosci. 2014;21:909–913. doi: 10.1016/j.jocn.2013.10.008. [DOI] [PubMed] [Google Scholar]
- 15.Maigne JY, Deligne L. Computed tomographic follow-up study of 21 cases of nonoperatively treated cervical intervertebral soft disc herniation. Spine. 1994;19:189–191. doi: 10.1097/00007632-199401001-00013. [DOI] [PubMed] [Google Scholar]
- 16.Vinas FC, Wilner H, Rengachary S. The spontaneous resorption of herniated cervical discs. J Clin Neurosci. 8:542–546. doi: 10.1054/jocn.2000.0894. 200. [DOI] [PubMed] [Google Scholar]
- 17.Han SR, Choi CY. Spontaneous regression of cervical disc herniation: A case report. Korean J Spine. 2014;11:235–237. doi: 10.14245/kjs.2014.11.4.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gurkanlar D, Yucel E, Er U, Keskil S. Sponateous regression of cervical disc herniations. Minim Invasive Neurosurg. 2006;49:179–183. doi: 10.1055/s-2006-932194. [DOI] [PubMed] [Google Scholar]
- 19.Kobayashi N, Asamoto S, Doi H, Ikeda Y, Matusmoto K. Spontaneous regression of herniated cervical disc. Spine J. 2003;3:171–173. doi: 10.1016/s1529-9430(02)00556-9. [DOI] [PubMed] [Google Scholar]
- 20.Autio RA, Karppinen J, Niinima J, et al. Determinants of spontaneous resorption of intervertebral disc herniations. Spine. 2006;31:1247–1252. doi: 10.1097/01.brs.0000217681.83524.4a. [DOI] [PubMed] [Google Scholar]
- 21.Orief T, Orz Y, Attia W. Almusrea: Spontaneous resorption of sequestrated intervertebral disc herniation. World Neurosurg. 2012;77:146–152. doi: 10.1016/j.wneu.2011.04.021. [DOI] [PubMed] [Google Scholar]
- 22.Minamide A, Hashizume H, Yoshida M, Kawakami M, Hayashi N, Tamaki T. Effects of basic fibroblast growth factor on spontaneous resorption of herniated intervertebral discs. An experimental study in the rabbit. Spine. 1999;24:940–945. doi: 10.1097/00007632-199905150-00003. [DOI] [PubMed] [Google Scholar]
- 23.Kato T, Haro H, Komori H, Shinomiya K. Sequential dynamics of inflammatory cytokine, angiogenesis inducing factor and matrix degrading enzymes during spontaneous resorption of the herniated disc. J Orthop Res. 2004;22:895–900. doi: 10.1016/j.orthres.2003.11.008. [DOI] [PubMed] [Google Scholar]
- 24.Haro H, Kato T, Komori H, Osada M, Shinomiya K. Vascular endothelial growth factor (VEGF)-induced angiogenesis in herniated disc resorption. J Orthop Res. 2002;20:409–415. doi: 10.1016/S0736-0266(01)00150-4. [DOI] [PubMed] [Google Scholar]
- 25.Olby NJ. Current concepts in the management of acute spinal cord injury. J Vet Intern Med. 1999;13:399–407. doi: 10.1892/0891-6640(1999)013<0399:ccitmo>2.3.co;2. [DOI] [PubMed] [Google Scholar]
- 26.Bergknut N, Smolders LA, Grinwis GC, et al. Intervertebral disc degeneration in the dog. Part 1: Anatomy and physiology of the intervertebral disc and characteristics of intervertebral disc degeneration. Vet J. 2013;195:282–291. doi: 10.1016/j.tvjl.2012.10.024. [DOI] [PubMed] [Google Scholar]
- 27.Da Costa RC, Parent JM. One-year clinical and magnetic resonance imaging follow-up of Doberman Pinschers with cervical spondylomyelopathy treated medically or surgically. J Am Vet Med Assoc. 2007;231:243–250. doi: 10.2460/javma.231.2.243. [DOI] [PubMed] [Google Scholar]
- 28.De Decker S, Gielen IMVL, Duchateau L, et al. Evolution of clinical signs and predictors of outcome after conservative medical treatment for disk-associated cervical spondylomyelopathy in dogs. J Am Vet Med Assoc. 2012;240:848–857. doi: 10.2460/javma.240.7.848. [DOI] [PubMed] [Google Scholar]