Abstract
Sleep complaints are reported by 40–60 % of menopausal women. Poor sleep is a risk factor for cardiovascular disease, diabetes, and obesity. The effect of menopausal hormone therapy on sleep quality is unclear. A systematic review and meta-analysis were conducted to summarize the efficacy of menopausal hormone therapy on self-reported sleep quality. Electronic databases (PubMed, Scopus, Ovid MEDLINE, EMBASE, EBM Reviews CENTRAL, and Psyclnfo) were searched from 2002 to October 2015. Randomized trials assessing the effect of menopausal hormone therapy with a minimum follow up of 8 weeks were included. Titles, abstracts, and full texts were screened independently and in duplicate. Primary outcome included sleep items within a questionnaire, scale or diary. Standardized mean differences across trials were pooled using random-effects models. The search identified 424 articles, from which 42 trials were included. Seven trials at a moderate to high risk of bias enrolling 15,468 women were pooled in meta-analysis. Menopausal hormone therapy improved sleep quality in women who had vasomotor symptoms at baseline [standardized mean difference −0. 54 (−0. 91 to −0. 18), moderate quality evidence]. No difference was noted when women without such symptoms were analyzed separately or combined. Across 31 sleep quality questionnaires, daytime dysfunction was the most evaluated sleep domain. Menopausal hormone therapy improves sleep in women with concomitant vasomotor symptoms. Heterogeneity of trials regarding study population, formulations, and sleep scales; limit overall certainty in the evidence. Future menopausal hormone therapy trials should include assessment of self-reported sleep quality using standardized scales and adhere to reporting guidelines.
Keywords: Menopause, Sleep disturbances, Vasomotor symptoms, Estrogen replacement
Introduction
From 2000 to 2010 the number of women at the age range expected to transition into menopause (50–54 years of age) increased by 26.6 % [1]. Worldwide, it is estimated that by 2025, the number of postmenopausal women will be 1.1 billion. With increased life expectancies, women live a third of their life after menopause, with some having the decline of menopause symptoms take many years [2]. The burden associated with untreated menopausal symptoms results in more frequent outpatient visits and incremental health care costs [3]. Menopausal hormone therapy (MHT) is one of the most common treatments used to counteract these symptoms.
The applicability of the evidence for the use of MHT is complicated by the heterogeneity of available trials in terms of age at which MHT is initiated, dose and type of estrogen, contraindications, and adjunct therapies. Additionally, concerns from the Women’s Health Initiative (WHI) report in 2002, led guidelines to advise shorter exposure to MHT. Yet, a decade after the WHI, the use of low-dose MHT has remained constant [4], gynecologist continue to favor MHT [5], and guidelines recommend MHT as the most effective treatment for menopause symptoms, including sleep disturbances [6].
Approximately 40–60 % of menopausal women report sleep related symptoms, with the most common complaint nighttime awakenings [7]. The mechanism by which sleep disturbances arise during menopause is still unclear, and studies characterizing how other menopausal symptoms are associated to sleep alterations are conflicting. An inverse relationship between sleep quality and vasomotor symptoms (VMS) has been reported. Sleep difficulties, however, could present independently [8]. Poor sleep is a risk factor for cardiovascular disease, diabetes, obesity, and neuro behavioral dysfunction [9]. Therefore, reducing the burden of emerging sleep symptoms during menopause will result in an improvement in quality of life and overall health.
Sleep symptoms can be measured objectively (e.g., polysomnography) or subjectively (e.g., questionnaire, severity scale or diary). A previous systematic review has shown that patient reported measurements are highly predictive of quality of sleep [10], and a guideline has emphasized that such measures are important for diagnosing and monitoring response to treatment in many sleep disorders, including insomnia [11]. Further, they both empower patients and aid clinicians in recognizing and valuing the patient’s perspective in response to treatment [12, 13]. These previous publications, however, addressed all adults, and did not tailor their conclusions to postmenopausal women. Therefore, understanding the effects of MHT on subjective sleep quality is important in helping patients and their clinicians manage the symptoms of menopause. However, synthesized evidence is scarce in regards to MHT effects on sleep quality leading to clinical uncertainty when choosing the best treatment.
This systematic review and meta-analysis aims to (1) evaluate the effects of MHT on self-reported sleep outcomes when compared to placebo in postmenopausal women and (2) explore the use of a multi-domain assessment of sleep quality across trials.
Methods
This review adheres to the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA statement [14] and was guided by a registered protocol (PROSPERO CRD42015027189). Screening and extraction was performed using online software (https://www.covidence.org/).
Eligibility criteria
Randomized clinical trials (RCTs) that compared the effects of MHT, to each other or placebo, on self-reported outcomes within a sleep questionnaire, symptom scale, or quality of life assessment tool were included. Selection was not restricted by blinding scheme, type or dose of MHT, whether sleep was a primary or secondary outcome, or type of self-reported sleep measurement tool. Minimal intervention length was 8 weeks. This timing was chosen arbitrarily as there is no current agreement on duration for which MHT changes in sleep quality would be anticipated. However, MHT alleviation of other menopausal symptoms have shown benefit as early as 8 weeks [15]. Trials where MHT was combined with compounds other than progesterone derived or selective estrogen receptor modulators were excluded. Women at any stage of natural or surgical menopause above 40 years old were included [16].
Identification and selection of trials
An experienced librarian developed search strategies, using methods recommended by the Institute of Medicine [17], in the following databases: PubMed, Scopus, Ovid MEDLINE, Ovid EMBASE, Ovid EBM Reviews CENTRAL, and Ovid Psyclnfo (for search strategy, eAppendix in electronic supplementary). Search included MESH headings and keywords such as menopause, estrogen, and sleep. Databases were searched from 2002 to October 2015, aiming to gather evidence produced or published during and after the WHI reports. Electronic search was supplemented by hand searching eligible articles. There were no language restrictions with non-English articles translated by fluent bilingual speakers. Full texts of included trials were screened in duplicate and independently (κ = 0.74) [18], and disagreements were resolved by arbitration.
Data collection and study appraisal
Data were extracted using an electronic form designed by the reviewers; which was tested and piloted and contained information on patient characteristics, intervention descriptions, methodological quality indications, and outcomes of interest. Data extraction and risk-of-bias assessment were performed independently by two reviewers. Mean and standard deviation at baseline and longest follow up were extracted for the outcome. RCTs were assessed for methodological quality using the Cochrane Risk of Bias Tool [19]. Available study protocols were searched in trial registries. If the blinding of study participants or personnel was rated to be at a high or unclear risk of bias, the trial was considered to be at high risk of bias overall. If all domains were judged to be at low risk of bias, the trial was considered at a low risk of bias. Otherwise, the trial was considered to be at a moderate risk of bias. The quality of evidence was evaluated using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach [20].
Author contact
When scores for sleep items within questionnaires were not available or when clarification was necessary the corresponding author was contacted by e-mail. If there was no response, a second, final e-mail was sent after 2 weeks. Authors were given 6 weeks to answer and send requested information.
Meta-analysis
Standardized mean differences (SMD) were pooled using random-effects models. This approach was preferred given the construct of sleep quality was evaluated using different scales, thus, the results were standardized and expressed using standard deviation units to allow meta-analysis. SMD results can be interpreted as 0.2 = small effect, 0.5 = moderate effect, and 0.8 = large effect [19]. For all trials, lower scores indicated better sleep quality (the direction was reversed for one trial to be consistent with the rest). In a trial with more than one active MHT arm, the weighted SMD between groups was compared to placebo. To explain possible inconsistencies across trials, a sensitivity analysis was used to assess the effect of the WHI on the pooled estimate effect. Inconsistency of effects across trials was assessed using forest plots and the I2 statistic with values over 50 % indicative of moderate to high heterogeneity [21]. Statistical analyses, including overall and subgroup effect estimates, were done using Review Manager v5.3 [22].
Subgroup analysis
Pre-specified subgroup analyses were performed to explore heterogeneity. Trials with inclusion criteria restricted to women with presence of VMS (hot flashes and night sweats) were compared to trials with no VMS criteria. To address duration of MHT and risk of bias, subgroup analysis by duration of intervention (8 weeks vs. >8 weeks) and by overall risk of bias (moderate vs. high) were performed.
Outcome assessment
Two board-certified sleep specialists (M.L.; R.L.) classified sleep items across multiple measurement tools using the seven sleep domains of the Pittsburgh Sleep Quality Index (PSQI). This allowed standardizing results to seven sleep characteristics routinely assessed in clinical interviews of patients with sleep complaints [23]. The PSQI was found to cover most domains of relevance to researchers when studying sleep disorders [10]. For each questionnaire, with at least one self-reported sleep item, each sleep specialist reviewed questionnaire items and dichotomized each under primary and, if applicable, secondary domains of sleep quality. Conflicts were resolved by consensus.
Results
Search strategy and contact of authors
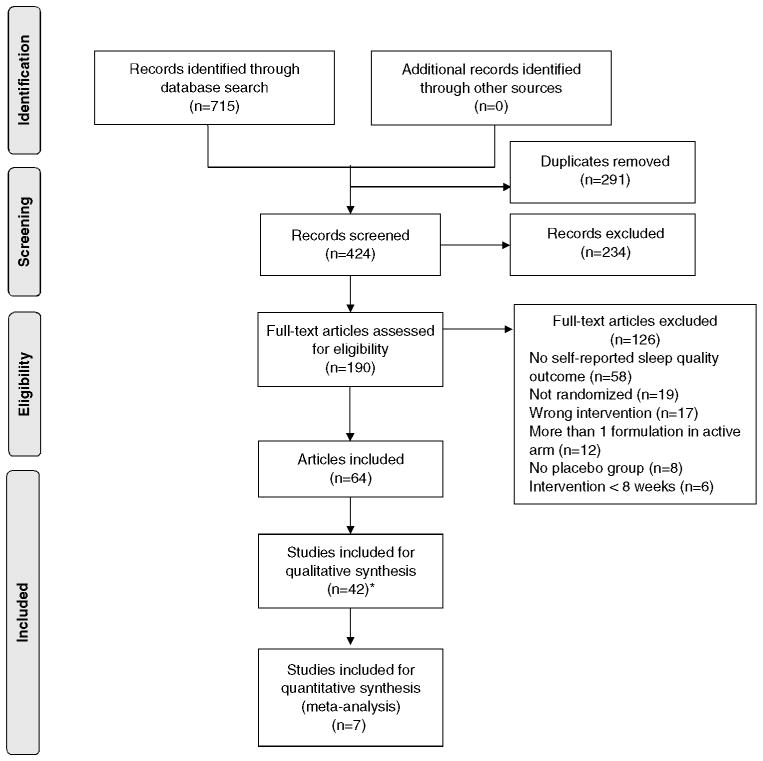
The search identified 424 articles of these, 234 were excluded at title and abstract screening. The full text assessment of the remaining 190 articles resulted in 64 meeting eligibility criteria. After merging multiple publications of a same trial, a total of 42 RCTs were included (Fig. 1). Thirty-five trials were missing data necessary for appraisal. Authors were contacted for 23 trials; the remaining 10 did not provide contact information. Two of 23 contacted authors provided the requested data [24, 25]. Thereafter, a total of nine trials had complete report of sleep quality.
Fig. 1.
PRISMA flow diagram for study selection process
* denotes number of included articles does not match number of included RCTs, as some trials had multiple publications
Description of trials
The data from 42 trials was used for qualitative assessment and is summarized in supplementary eTablel. Across all RCTs, 21 where comparing MHT interventions to each other and nine comparing MHT to placebo. The most commonly administered formulation was oral conjugated equine estrogen (o-CEE) at a dose of 0.625 mg/day (12/42 trials).
The definition of menopause was variable. Most trials used self-report of last menstrual period (LMP) as definition with intervals post-LMP ranging from 6 months to 10 years. Seven of the 42 trials included sleep quality as a primary outcome measure. Across trials significant variability was found on reports of MHT effect on sleeping problems.
One trial was judged to be at low risk of bias, 23 (55 %) at moderate risk and 18 (43 %) at high risk (supplementary eTable 2). Sequence generation and blinding of outcome assessors were the domains least reported. The 18 trials rated at high risk of bias had either not blinded participants or not clearly reported blinding methodology.
Effects of MHT on sleep quality
From the 42 trials, nine trials had mean and standard deviation reports at baseline and longest follow up. Seven of the nine trials included a placebo treatment arm [15, 24–29], while 2 had parallel comparisons of MHT formulations [30, 31]. Therefore, the seven RCTs with placebo arm as comparator had similar interventions and reported sufficient quantitative data to allow for statistical pooling (Table 1). The trials were at moderate to high risk of bias (Table 2).
Table 1.
Characteristics of included eligible randomized placebo controlled trials with available data for meta-analysis
| Reference | Country | Sample size | Menopause definition | Inclusion criteria | Type of MHT (mg/day) | Duration (weeks) | Sleep quality scale |
|---|---|---|---|---|---|---|---|
| Hays et al. [26] | USA | 166,008 | 50–54 y/o + LMP > 12 m >54 y/o + LMP > 6 m |
— | o-CEE (0.625) + MPA (2.5) | 156 | WHIIRS |
| Saletu-Zyhlarz et al. [27] | Austria | 55 | LMP > 24 m + E < 55 pg/mL + FSH > 19 mlU/mL + KI > 15 | Insomnia +VMS | o-EV(2) + dienogest (3) | 8 | PSQI |
| + | o-EV (2) | ||||||
| Sherman et al. [25] | USA | 249 | >55 y/o + LMP > 5 years | Coronary artery disease | o-CEE (0.625) o-CEE (0.625) + MPA (2.5) |
156 | WHIIRS |
| Heinrich et al. [28] | Germany | 51 | Hysterectomy | VMS | o-EV (2) | 24 | ADSK Sleep item + Sleep item from Menopausal Index (combined) |
| o-EV(2) + mP(100) | |||||||
| Kalleinen et al. [29] | Finland | 18 | By age and amenorrhea (no further details) | — | o-EV(2) + MDP (5) | 24 | BNSQ |
| + | |||||||
| LeBlanc et al. [15] | USA | 37 | 6 m > LMP < 36 m + FSH > 920 IU/L | — | o-EV(2) + MDP (5) | 8 | OHSU SL sleep diary |
| Savolainen-Peltonen et al. [24] | Finland | 128 | 6 m > LMP > 36 m + FSH > 30 U/L | ½ women with severe VMS, ½ symptom free | o-E2 | 24 | WHQ sleep item |
| o-E2 + MPA (5) | |||||||
| t-E2 |
LMP last menstrual period, m months, VMS vasomotor symptoms, WHIIRS Women’s Health Initiative Insomnia Rating Scale, ADSK German version of Center for Epidemiological Studies Depression Scale, authors used only sleep item for sleep quality assessment, BNSQ Basic Nordic Sleep Questionnaire, OHSU SL Oregon Health and Science University Sleep Laboratory, PSQI Pittsburgh Sleep Quality Index, WHQ Women’s Health Questionnaire, o- oral, t- transdermal CEE, conjugated equine estrogen, MPA medroxyprogesterone acetate, EV estradiol valerate, mP micronized-progesterone, E2 17 beta estradiol, + primary outcome sleep quality or insomnia
Table 2.
Cochrane risk of bias quality assessment
| Reference | SG | AC | BP | BOA | IOD | SOR | Other | Overall |
|---|---|---|---|---|---|---|---|---|
| Hays et al. [26] | Unclear | Higha | Low | Unclear | Unclear | Unclear | Highb | High |
| Saletu-Zyhlarz et al. [27] | Unclear | Unclear | Low | Unclear | Unclear | Unclear | Highc | Moderate |
| Sherman et al. [25] | Low | Unclear | Low | Low | Unclear | Low | Unclear | Moderate |
| Heinrich et al. [28] | Unclear | Unclear | Unclear | Unclear | Highd | Unclear | Low | High |
| Kalleinen et al. [29] | Low | Low | Unclear | Low | Low | Unclear | Highe | High |
| LeBlanc et al. [15] | Highf | Unclear | Low | Unclear | Low | Unclear | Highg | High |
| Savolainen-Peltonen et al. [24] | Low | Low | Low | Unclear | Low | Unclear | Low | Moderate |
SG sequence generation, AC allocation concealment, BP blinding of participants, BOA blinding of outcome assessors, IOD I incomplete outcome data, SOR selective outcome reporting
Women on estrogen alone group were un-blinded and reassigned to estrogen + progrestin group
Early termination of trial, significant conflict of interest of authors with private sponsor
One of the authors is private sponsor employee and no conflict disclosure was provided
High proportion of patients withdrew from study, unclear to which group participants belonged too
Different time-points across patients for sleep quality outcome assessment, some did not complete 6 months (follow up to 3 months but authors report as 6 months)
Five participants were stratified by a third party after computer generated randomization was completed
Significant baselines sleep outcome imbalances across treatment groups
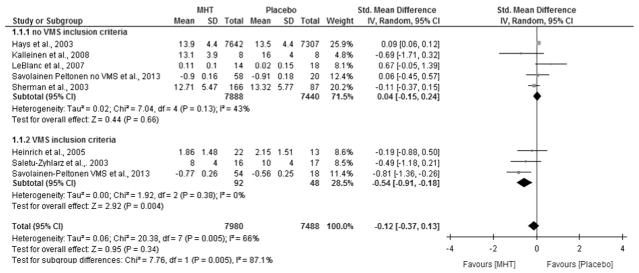
Subgroup analysis showed that MHT improved sleep quality among women who had concomitant VMS [SMD −0.54 (−0.91 to −0.18, I2 = 0%), moderate quality evidence]; test for subgroup difference p = <0.007. No significant difference was noted in trials that included women without VMS criteria [SMD − 0.04 (− 0.15 to 0.24, I2 = 43%)], or when both groups were combined (with and without VMS), [SMD (− 0.12 (− 0.37 to 0.13, I2 =66 %)]. Results are depicted as a forest plot in Fig. 2.
Fig. 2.
SMD for subgroup analysis by VMS, smaller scores indicate better sleep quality. The green square markers indicate standardized mean difference from primary studies, with sizes reflecting the statistical weight of the study using random-effects meta-analysis. The horizontal lines indicate 95 % confidence intervals. The diamond markers represent the subtotal and overall effect estimate and 95 % confidence intervals. SMD interpretation, 0.2 = small effect, 0.5 = moderate effect, >0.8 = large effect
A sensitivity analysis performed to examine whether the WHI affected effect estimate showed no significant difference [SMD (− 0.17 (0.35–0.02, I2 = 53%)]. Subgroup analysis comparing duration of MHT and risk of bias did not show significant differences as shown in supplementary eFig. 1 and eFig. 2.
Outcome assessment
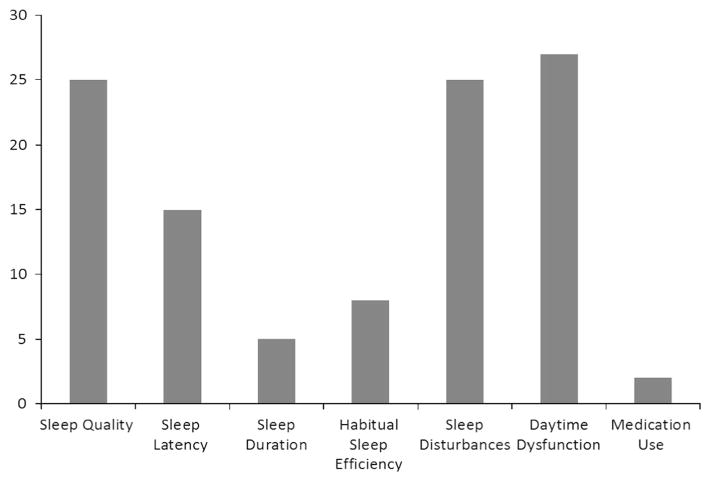
Across 31 self-report sleep tools, the most frequently assessed domains of sleep quality were daytime dysfunction followed by sleep quality and sleep disturbances (Fig. 3). Prior medication use for aid in sleep was only assessed in two scales. Three scales were not accessible for item dichotomizing, two were independently created by authors institutions and were not provided, and one was inaccessible through library resources (supplementary eTable 3)
Fig. 3.
Distribution of the PSQI seven domain of sleep quality across 27 self-reported sleep scales used in included studies
Quality of evidence
The certainty in the estimates following the GRADE approach was moderate confidence in women with VMS and low in women without VMS (Table 3).
Table 3.
Summary of the evidence quality grading using GRADE
| Outcome | Quality assessment | Effect | |||||||
|---|---|---|---|---|---|---|---|---|---|
|
|
|||||||||
| No. of studies | Study design | Risk of bias | Inconsistency | Indirectness | Imprecision | Other onsiderations | SMD (95 %CI) | Quality | |
| Sleep quality overall | 8 | RCT | Seriousa | Not serious | Not serious | Seriousb | Seriousc | −0.12 (−0.36, 0.13) | ⊕⊕○○Low |
| Sleep quality VMS subgroup | 3 | RCT | Seriousa | Not serious | Not serious | Not serious | Seriousc | −0.54 (−0.91, −0.17) | ⊕⊕⊕○Moderate |
The GRADE working group grades of evidence are as follow: 1. High quality, future research is very unlikely to change the group’s confidence in the estimate of effect; 2. Moderate quality, further research is likely to have an important impact on the group's confidence in the estimate of effect and may change the estimate; 3. Low quality, further research is very likely to have an important effect on the group’s confidence in the estimate of effects and is likely to change the estimate; 4. Very low quality, the group is very uncertain about the estimate
The quality was downgraded because there was overall poor reporting of blinding and allocation concealment
Wide confidence intervals in our estimate effect limit conclusions
Large numbers of studies do not contribute to sleep quality outcome, suggesting possible reporting bias
CI confidence interval, SMD standardized mean difference
Discussion
Summary of evidence
In this systematic review and meta-analysis of the effects of MHT on sleep quality, seven RCTs provided similar interventions and sufficient data for meta-analysis. MHT was associated with modest improved sleep quality in women with concomitant VMS at baseline. The effect of MHT is uncertain in women without VMS.
The heterogeneity in trial populations and formulations of MHT limit conclusions. The absorption, distribution, and metabolism of MHT differ among women based on genotype, age, distribution of adipose tissue, comorbidities, and use of other medications [32]. These covariates should help guide the design of future comparative effectiveness trials. Following the WHI, the use of low-dose transdermal estrogen increased more than tenfold [4], yet only three trials [24, 33, 34] had a direct comparison between routes of administration. Additionally, there is still a need for a standard definition of menopause, given that both age and years from menopause have shown to be important indicators of the benefit-risk ratio of MHT [35].
Self-reported sleep quality captures different parameters of sleep than objective measurements [36]. Lack of accessibility to polysomnography resources, and the limited utility of this clinical test within a large population setting, supports the need to develop validated self-reported sleep measurements in menopausal women. It is understood that sleep is best characterized across multiple domains including quality, duration, continuity and effects on daytime function [37]. A thorough assessment of these measurable characteristics of sleep quality, results in a detailed sleep scenario that is understood by both health professionals and patients [38].
In the present analysis, daytime dysfunction, sleep disturbances and overall sleep quality were the most commonly assessed domains. The other domains were infrequently incorporated into questionnaires, including sleep duration and latency (the ease of falling asleep), which have both been associated to negative health outcomes such as higher mortality, coronary heart disease, and diabetes [38]. This finding underscores the importance of further work in the area of standardizing sleep assessment tools.
Limitations and strengths
The systematic review faces a number of limitations. First, the majority of trials lacked a baseline screen for sleep disorders. After menopause, there is an increased risk of sleep disordered breathing due to fluctuating hormones and weight gain. Yet, only three trials [27, 39, 35], had exclusion or testing criteria for sleep related breathing disorders, narcolepsy or periodic limb movements. Second, evidence from this review cannot discern the magnitude of effect on sleep quality through indirect reduction of mood disturbances or frequency and severity of VMS, both known to affect sleep. This is due to heterogeneity in enrollment, as most trials do not follow the recommendations of the Food and Drug Administration for studies assessing treatment of moderate to severe VMS, where participants enrolled should have a minimum of 7 to 8 moderate to severe hot flushes per day, or 50 to 60 per week at baseline [40]. Finally, MHT formulations vary in the inclusion of progesterone or selective estrogen receptor modulator compounds. Progesterone has independent effects on sleep, through anxiolytic and respiratory stimulant action [41]. A number of studies used progestin synthetic derivatives that may not have the same effects as progesterone. Therefore, the independent effects of estrogen vs. progesterone and progestin compounds require further evaluation.
This systematic review also has several strengths. It provides a comprehensive review of the current evidence guided by an a priori registered protocol, with an extensive search for eligible studies in multiple databases including both published and unpublished work. All of the included studies were assessed in duplicate and registered protocols were searched on online trial protocol databases. We incorporated an in depth assessment of the sleep quality outcome scales, to best provide information for clinicians to understand and apply in practice. This is, to our knowledge, the first use of dichotomizing scheme to bring together self-reported sleep quality scales in menopausal women.
Implications for practice and research
Sleep disturbances are a common indication for MHT. Yet, at present there is insufficient evidence to determine how a woman’s self-reported sleep quality during menopause is affected by different routes of administration or formulations of MHT.
Future research focused on determining if MHT is beneficial in improving sleep quality during menopause, will be best addressed by head-to-head RCTs between various formulations and routes of administration. Likewise, sleep quality assessment tools need to be developed with a consensus on a tool with sufficient domains of sleep quality and validated in menopausal women. We suggest use of the seven major sleep domains listed in the PSQI. Yet, important questions specific to menopause must also be addressed in the context of sleep quality. The evidence derived from this systematic review suggests that MHT benefits sleep in women with VMS. This is congruent with current North American Menopause Society recommendations where use of MHT for sleep disturbance is suggested for women with bothersome night time hot flashes [42]. Other guidelines lack specifics regarding MHT use, and may benefit from incorporating these results.
Conclusion
It is imperative to endorse meaningful conversations about sleep quality in the clinical setting with female patients nearing menopausal age and to tailor treatment recommendations towards patient-specific complaints. Validated clinical screening tools addressing various domains of sleep quality are needed in order to better describe the current burden of sleep disturbances in this population.
Supplementary Material
Acknowledgments
D.C., M.R.G., and G.S.B. were supported by TL1 TR000137 from the National Center for Advancing Translational Science, a component of the National Institutes of Health. V.M.M. is supported by National Institutes of Aging P50-AG44170 and the Mayo Foundation.
Footnotes
Compliance with ethical standards
Conflict of interest The authors declare that they have no conflict of interest.
Electronic supplementary material The online version of this article (doi:10.1007/sl2020–016–1072–9) contains supplementary material, which is available to authorized users.
References
- 1.Howden L, Meyer J. Age and sex composition: 2010. 2010 Census Briefs. 2011 [Google Scholar]
- 2.Al-Safi ZA, Santoro N. Menopausal hormone therapy and menopausal symptoms. Fertil Steril. 2014;101:905–915. doi: 10.1016/j.fertnstert.2014.02.032. [DOI] [PubMed] [Google Scholar]
- 3.Sarrel P, Portman D, Lefebvre P, et al. Incremental direct and indirect costs of untreated vasomotor symptoms. Menopause. 2015;22:260–266. doi: 10.1097/GME.0000000000000320. [DOI] [PubMed] [Google Scholar]
- 4.Ettinger B, Wang SM, Leslie RS, et al. Evolution of postmenopausal hormone therapy between 2002 and 2009. Menopause. 2012;19:610–615. doi: 10.1097/gme.0b013e31823a3e5d. [DOI] [PubMed] [Google Scholar]
- 5.Danckers L, Blumel JE, Witis S, et al. Personal and professional use of menopausal hormone therapy among gynecologists: a multinational study (REDLINC VII) Maturitas. 2016;87:67–71. doi: 10.1016/j.maturitas.2016.02.015. [DOI] [PubMed] [Google Scholar]
- 6.Stuenkel CA, Davis SR, Gompel A, et al. Treatment of symptoms of the menopause: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2015;100:3975–4011. doi: 10.1210/jc.2015-2236. [DOI] [PubMed] [Google Scholar]
- 7.Kravitz HM, Ganz PA, Bromberger J, Powell LH, Sutton-Tyrrell K, Meyer PM. Sleep difficulty in women at midlife: a community survey of sleep and the menopausal transition. Menopause. 2003;10:19–28. doi: 10.1097/00042192-200310010-00005. [DOI] [PubMed] [Google Scholar]
- 8.Bruyneel M. Sleep disturbances in menopausal women: aetiology and practical aspects. Maturitas. 2015;81:406–409. doi: 10.1016/j.maturitas.2015.04.017. [DOI] [PubMed] [Google Scholar]
- 9.Luyster FS, Strollo PJ, Jr, Zee PC, Walsh JK. Sleep: a health imperative. Sleep. 2012;35:727–734. doi: 10.5665/sleep.1846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Devine EB, Hakim Z, Green J. A systematic review of patient-reported outcome instruments measuring sleep dysfunction in adults. Pharmacoeconomics. 2005;23:889–912. doi: 10.2165/00019053-200523090-00003. [DOI] [PubMed] [Google Scholar]
- 11.Schutte-Rodin S, Broch L, Buysse D, Dorsey C, Sateia M. Clinical guideline for the evaluation and management of chronic insomnia in adults. J Clin Sleep Med. 2008;4:487–504. [PMC free article] [PubMed] [Google Scholar]
- 12.Deshpande PR, Rajan S, Sudeepthi BL, Abdul Nazir CP. Patient-reported outcomes: a new era in clinical research. Perspect Clin Res. 2011;2:137–144. doi: 10.4103/2229-3485.86879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wiklund I. Assessment of patient-reported outcomes in clinical trials: the example of health-related quality of life. Fundam Clin Pharmacol. 2004;18:351–363. doi: 10.1111/j.1472-8206.2004.00234.x. [DOI] [PubMed] [Google Scholar]
- 14.Moher D, Liberati A, Tetzlaff J, Altaian DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(264–269):W64. doi: 10.7326/0003-4819-151-4-200908180-00135. [DOI] [PubMed] [Google Scholar]
- 15.LeBlanc ES, Neiss MB, Carello PE, Samuels MH, Janowsky JS. Hot flashes and estrogen therapy do not influence cognition in early menopausal women. Menopause. 2007;14:191–202. doi: 10.1097/01.gme.0000230347.28616.1c. [DOI] [PubMed] [Google Scholar]
- 16.Harlow SD, Gass M, Hall JE, et al. Executive summary of the Stages of Reproductive Aging Workshop+10: addressing the unfinished agenda of staging reproductive aging. Climacteric. 2012;15:105–114. doi: 10.3109/13697137.2011.650656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Eden J, Levit L, Berg A, Morton S, editors. Finding What Works in Health Care: Standards for Systematic Reviews. National Academy of Sciences; Washington, DC: 2011. [PubMed] [Google Scholar]
- 18.McGinn T, Wyer PC, Newman TB, Keitz S, Leipzig R. GG For, Tips for learners of evidence-based medicine: 3. Measures of observer variability (kappa statistic) CMAJ. 2004;171:1369–1373. doi: 10.1503/cmaj.1031981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Higgins J, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011] The Cochrane Collaboration; 2011. [Google Scholar]
- 20.Meader N, King K, Llewellyn A, et al. A checklist designed to aid consistency and reproducibility of GRADE assessments: development and pilot validation. Syst Rev. 2014;3:82. doi: 10.1186/2046-4053-3-82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Review Manager (RevMan) [Computer program]. Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration; 2014. [Google Scholar]
- 23.Buysse DJ, Reynolds CF, 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 24.Savolainen-Peltonen H, Hautamaki H, Tuomikoski P, Ylikorkala O, Mikkola TS. Health-related quality of life in women with or without hot flashes: a randomized placebo-controlled trial with hormone therapy. Menopause. 2014;21:732–739. doi: 10.1097/GME.0000000000000120. [DOI] [PubMed] [Google Scholar]
- 25.Sherman AM, Shumaker SA, Sharp P, et al. No effect of HRT on health-related quality of life in postmenopausal women with heart disease. Minerva Ginecol. 2003;55:511–517. [PubMed] [Google Scholar]
- 26.Hays J, Ockene JK, Brunner RL, et al. Effects of estrogen plus progestin on health-related quality of life. N Engl J Med. 2003;348:1839–1854. doi: 10.1056/NEJMoa030311. [DOI] [PubMed] [Google Scholar]
- 27.Saletu-Zyhlarz G, Anderer P, Gruber G, et al. Insomnia related to postmenopausal syndrome and hormone replacement therapy: sleep laboratory studies on baseline differences between patients and controls and double-blind, placebo-controlled investigations on the effects of a novel estrogen-progestogen combination (Cli-modien, Lafamme) versus estrogen alone. J Sleep Res. 2003;12:239–254. doi: 10.1046/j.1365-2869.2003.00356.x. [DOI] [PubMed] [Google Scholar]
- 28.Heinrich AB, Wolf OT. Investigating the effects of estradiol or estradiol/progesterone treatment on mood, depressive symptoms, menopausal symptoms and subjective sleep quality in older healthy hysterectomized women: a questionnaire study. Neuropsychobiology. 2005;52:17–23. doi: 10.1159/000086173. [DOI] [PubMed] [Google Scholar]
- 29.Kalleinen N, Polo O, Himanen SL, Joutsen A, Polo-Kantola P. The effect of estrogen plus progestin treatment on sleep: a randomized, placebo-controlled, double-blind trial in premenopausal and late postmenopausal women. Climacteric. 2008;11:233–243. doi: 10.1080/13697130802112033. [DOI] [PubMed] [Google Scholar]
- 30.Polisseni AF, Andrade AT, Ribeiro LC, et al. Effects of a continuous-combined regimen of low-dose hormone therapy (oestradiol and norethindrone acetate) and tibolone on the quality of life in symptomatic postmenopausal women: a double-blind, randomised study. Maturitas. 2013;74:172–178. doi: 10.1016/j.maturitas.2012.11.001. [DOI] [PubMed] [Google Scholar]
- 31.Elfituri A, Sherif F, Elmahaishi M, Chrystyn H. Two hormone replacement therapy (HRT) regimens for middle-eastern postmenopausal women. Maturitas. 2005;52:52–59. doi: 10.1016/j.maturitas.2004.12.003. [DOI] [PubMed] [Google Scholar]
- 32.Bhavnani BR, Stanczyk FZ. Pharmacology of conjugated equine estrogens: efficacy, safety and mechanism of action. J Steroid Biochem Mol Biol. 2014;142:16–29. doi: 10.1016/j.jsbmb.2013.10.011. [DOI] [PubMed] [Google Scholar]
- 33.Kling JM, Lahr BA, Bailey KR, Harman SM, Miller VM, Mulvagh SL. Endothelial function in women of the Kronos Early Estrogen Prevention Study. Climacteric. 2015;18:187–197. doi: 10.3109/13697137.2014.986719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Serrano D, Mariani L, Mora S, et al. Quality of life assessment in a chemoprevention trial: fenretinide and oral or transdermal HRT. Maturitas. 2006;55:69–75. doi: 10.1016/j.maturitas.2006.01.005. [DOI] [PubMed] [Google Scholar]
- 35.Joffe H, Petrillo LF, Koukopoulos A, et al. Increased estradiol and improved sleep, but not hot flashes, predict enhanced mood during the menopausal transition. J Clin Endocrinol Metab. 2011;96:E1044–E1054. doi: 10.1210/jc.2010-2503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Buysse DJ, Ancoli-Israel S, Edinger JD, Lichstein KL, Morin CM. Recommendations for a standard research assessment of insomnia. Sleep. 2006;29:1155–1173. doi: 10.1093/sleep/29.9.1155. [DOI] [PubMed] [Google Scholar]
- 37.Hall MH, Kline CE, Nowakowski S. Insomnia and sleep apnea in midlife women: prevalence and consequences to health and functioning. FlOOOPrime Rep. 2015;7:63. doi: 10.12703/P7-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Buysse DJ. Sleep health: can we define it? Does it matter? Sleep. 2014;37:9–17. doi: 10.5665/sleep.3298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hachul H, Bittencourt LR, Andersen ML, Haidar MA, Baracat EC, Tufik S. Effects of hormone therapy with estrogen and/or progesterone on sleep pattern in postmenopausal women. Int J Gynaecol Obstet. 2008;103:207–212. doi: 10.1016/j.ijgo.2008.07.009. [DOI] [PubMed] [Google Scholar]
- 40.Guidance for Industry: Estrogen and Estrogen/Progesting Drug Products to Treat Vasomotor Symptoms and Vulvar and Vaginal Atrophy Symptoms-Recommendations for Clinical Evaluation. [accessed 08/05/16]; http://google2.fda.gov/search?q=esrrogen&client=FDAgov&site=EDAgov&h=&proxystylesheet=FDAgov&requiredfields=-archive%3AYes&output=xml_no_dtd&getfields=*
- 41.Eichling PS, Sahni J. Menopause related sleep disorders. J Clin Sleep Med. 2005;1:291–300. [PubMed] [Google Scholar]
- 42.Shifren JL, Gass ML. The North American Menopause Society recommendations for clinical care of midlife women. Menopause. 2014;21:1038–1062. doi: 10.1097/GME.0000000000000319. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.