Abstract
A 2-year-old girl experienced cardiac arrest after cold water drowning. Magnetic resonance imaging (MRI) showed deep gray matter injury on day 4 and cerebral atrophy with gray and white matter loss on day 32. Patient had no speech, gait, or responsiveness to commands on day 48 at hospital discharge. She received normobaric 100% oxygen treatment (2 L/minute for 45 minutes by nasal cannula, twice/day) since day 56 and then hyperbaric oxygen treatment (HBOT) at 1.3 atmosphere absolute (131.7 kPa) air/45 minutes, 5 days/week for 40 sessions since day 79; visually apparent and/or physical examination-documented neurological improvement occurred upon initiating each therapy. After HBOT, the patient had normal speech and cognition, assisted gait, residual fine motor and temperament deficits. MRI at 5 months after injury and 27 days after HBOT showed near-normalization of ventricles and reversal of atrophy. Subacute normobaric oxygen and HBOT were able to restore drowning-induced cortical gray matter and white matter loss, as documented by sequential MRI, and simultaneous neurological function, as documented by video and physical examinations.
Keywords: normobaric oxygen, hyperbaric oxygen, drowning, magnetic resonance imaging, brain volume, gray matter, white matter
INTRODUCTION
Pediatric drowning1,2,3 is treated with normobaric 100% oxygen to maintain adequate systemic oxygenation.1 Serious cases cause angiosomic-based4 deep gray matter and cortical5 injury with resultant atrophy/ventricular dilatation.5,6 To our knowledge and investigation reversal of gray matter (cortical volume loss) and white matter atrophy (ventricular dilatation) are unreported with any therapy. We report subacute treatment of a severe pediatric drowning case with repetitive short-duration normobaric oxygen and then hyperbaric oxygen that resulted in visually apparent and/or physical examination-documented neurological improvement with each therapy and near-complete reversal of cortical and white matter atrophy on magnetic resonance imaging (MRI).
CASE REPORT
A 2-year-old girl was resuscitated at Washington Regional Medical Center, Fayetteville, AR, USA, from a Glasgow Coma Scale (GCS) of 3, fixed dilated pupils, and body temperature of 85.1°F (28.9°C) after a 15-minute submersion in 41°F (5°C) water. After 100 minutes of cardio-pulmonary resuscitation, the arterial pH was 6.53. Following hypothermia, vasopressors, ventilator support (10 days), and critical care at Arkansas Children’s Hospital, Little Rock, AR, USA, the patient was discharged home 35 days post drowning unresponsive to all stimuli, immobile with legs drawn to chest, and with constant squirming and head shaking. MRIs at 3 (Figure 1) and 31 (Figure 2) days post drowning showed thalamic injury then generalized atrophy with evolving gray and white matter injury.
Figure 1.
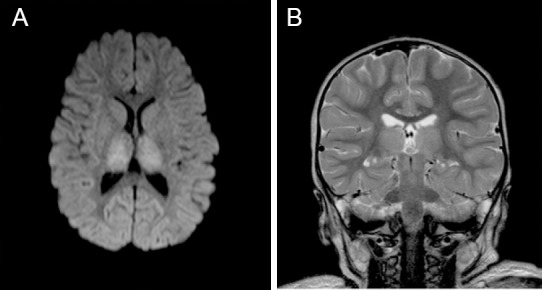
Magnetic resonance imaging at 3 days after injury of the 2-year-old girl who experienced cardiac arrest after cold water drowning.
Note: (A) Axial diffusion weighted image at three days post drowning, showing increased signal from acute ischemic injury to both thalami; (B) Coronal T2 weighted mid thalamic image 3 days post-drowning, showing subtle diffuse thalamic signal changes, normal ventricles, and normal cortical sulcal cerebro-spinal fluid spaces.
Figure 2.
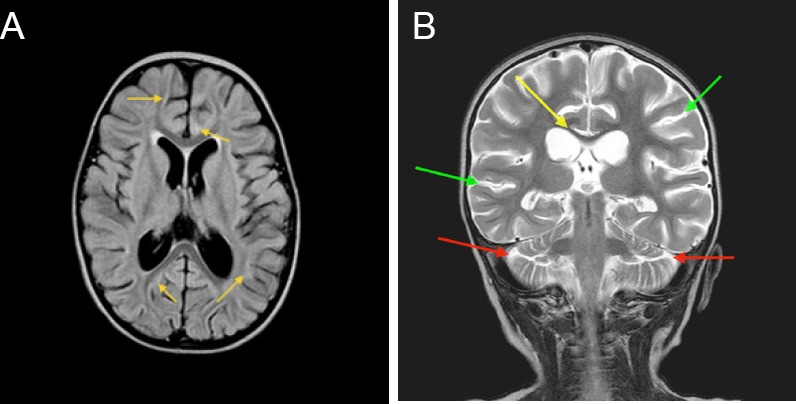
Magnetic resonance imaging at 31 days after injury of the 2-year-old girl who experienced cardiac arrest after cold water drowning.
Note: (A) Axial fluid attenuated inversion recovery (FLAIR) image at the level of the basal ganglia at 31 days post-drowning, showing subtle persistent diffuse signal irregularities in the gray and white matter (yellow arrows), diffuse gray matter atrophy (enlarged sulcal spaces), and white matter atrophy (enlarged lateral and third ventricles); (B) Coronal T2 image at the level of the thalami 31 days post-drowning, showing gray matter atrophy with increased cerebro-spinal fluid spaces at temporal and parietal lobes (green arrows) and cerebellar lobes (red arrows), and white matter atrophy with thinned corpus callosum (yellow arrow) and enlarged ventricles.
Author Paul G. Harch was consulted and commenced 2 L/minute nasal cannula 100% oxygen for 45 minutes twice/day bridging normobaric oxygen therapy at 55 days post-drowning. Within hours the patient was more alert, awake, and stopped squirming (see the movie at https://www.youtube.com/watch?v=Pdy9w_3x2Lw&feature=youtube. Entire movie is pre-, and post-drowning up to pre- and post-normobaric oxygen therapy, shown at 7 minutes 40 seconds mark and then 9 minutes mark). Neurological improvement rate increased over the ensuing 23 days with laughing, increased movement of arms, hands, grasp with the left hand, partial oral feeding, eye tracking, and short sentence speech (pre-drowning speech level, but with diminished vocabulary). Physical (PT), occupational (OT), and speech therapy (ST) were added on the 10th day for 2 weeks.
Seventy-eight days post-drowning, hyperbaric oxygen treatment (HBOT) commenced with compressed air at 1.3 atmosphere absolute (1 ATA = 131.7 kPa) for 45 minutes total treatment time, once daily. Within hours the patient experienced decreased tone, increased gross motor activity, vocabulary, and alertness. After 10 sessions of HBOT, the patient’s mother reported that the patient was “near normal, except for gross motor function;” PT was then reinstituted. After 39 sessions of HBOT, the patient exhibited: assisted gait, speech level greater than pre-drowning, near normal motor function, normal cognition, improvement on nearly all neurological exam abnormalities, discontinuance of all medications (buspar, propranolol, baclofen), and residual emotional, gait, and temperament deficits (Additional Video 1). Gait improvement was documented immediately upon return home at https://www.youtube.com/watch?v=m2SBmdY4RXg&feature=youtu.be. MRI at 27 days after 40-session HBOT and 162 days post-drowning demonstrated mild residual injury and near-complete reversal of cortical and white matter atrophy (Figures 3–6), while further improved gait was documented at: https://www.youtube.com/watch?v=UjtucZa7zmw&feature=youtu.be.
Figure 3.
T2 coronal MRI images and axial fluid attenuated inversion recovery (FLAIR) image of the 2-year-old girl who experienced cold water drowning.
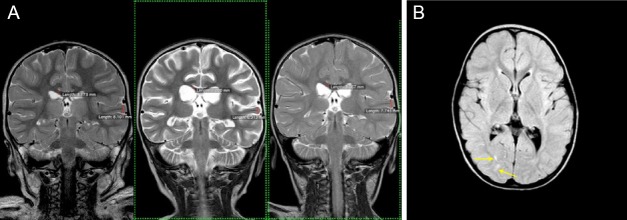
Note: (A) T2 coronal images at the level of the thalami from left to right at 3, 31, and 162 days post-drowning, showing reversal of white matter and cortical atrophy. Corpus callosum white matter and temporal lobe gray matter calculations embedded: 3.17, 2.00, 3.57 mm, and 8.10, 6.31, and 7.75 mm, at 3, 31, and 162 days respectively. (B) Axial FLAIR image at the level of the basal ganglia 162 days post-drowning, showing scattered residual signal change in the white matter (yellow arrows) despite apparent global return to normal tissue volumes. MRI: Magnetic resonance imaging.
Figure 6.
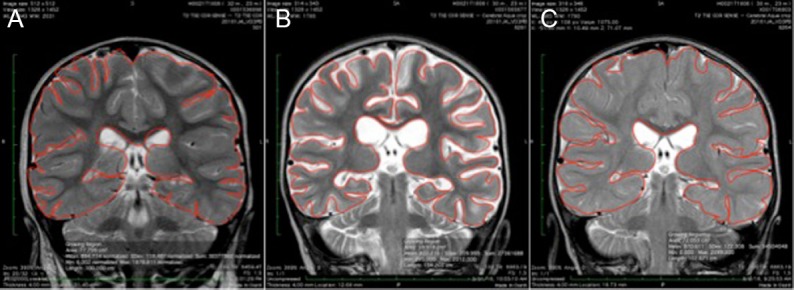
T2 coronal MRI images of the thalami in the 2-year-old girl who experienced cold water drowning.
Note: T2 coronal images at the level of the thalami of MRI 3 (A), 31 (B), and 162 days (C) post-drowning with manually drawn surface area calculations of 77.8, 59.9, and 72.0 cm2, respectively. MRI: Magnetic resonance imaging.
Figure 4.
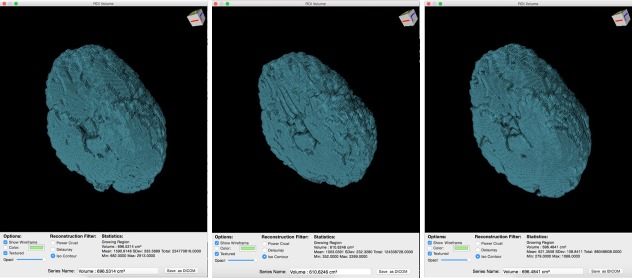
Three-dimensional DWI volumes obtained from the level of the pons to the centrum semi-ovale as viewed from a caudal-oblique projection in the 2-year-old girl who experienced cold water drowning.
Note: Axial images at 3, 31, and 162 days (left to right) post-drowning with iso-contour three-dimensional brain volume calculations of 697, 611, and 696 cm3, respectively. There is smaller volume and worsening of surface texture in the middle image and reversal of both in the third image. DWI: Diffusion weighted imaging.
Figure 5.
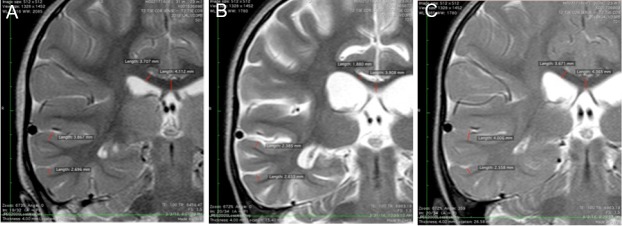
Partial T2 coronal MRI images at the level of the thalami at 3, 31, and 162 days post-drowning of the 2-year-old girl who experienced cold water drowning.
Note: (A) Partial T2 coronal image at the level of the thalami at 3 days post-drowning with right temporal lobe gray matter (2.70–3.87 mm) and corpus callosum white matter (3.71–4.11 mm) thickness measurements. (B) Partial T2 coronal image at the level of the thalami 31 days post-drowning with right temporal lobe gray matter (2.39–2.65 mm) and corpus callosum white matter (1.88–3.91 mm) thickness measurements. (C) Partial T2 coronal image at the level of the thalami 162 days post-drowning with right temporal lobe gray matter (2.56–4.01 mm) and corpus callosum white matter (3.67–4.37 mm) thickness measurements. MRI: Magnetic resonance imaging.
DISCUSSION
Due to concern for oxygen toxicity,7,8 continuous normobaric oxygen in acute cerebral injury is only used for normalization of systemic oxygenation.1 Short duration normobaric oxygen has been applied to acute focal stroke9 and traumatic brain injury,10 yet is unexplored in subacute hypoxic/ischemic encephalopathy (HIE). Short duration hyperoxia at or slightly above the equivalent level of normobaric oxygen in our case, in the form of hyperbaric oxygen or air, has been achieved in chronic toxic brain injury,11 traumatic brain injury,12,13 autism,14 and cerebral palsy15,16 where it is used for deoxyribonucleic acid (DNA) signal transduction17,18 in combination with increased barometric pressure.19,20 Intermittent hyperoxia and increased atmospheric pressure up- or down-regulate 8,101 genes in human endothelial cells.21 Sequential application of normo-baric oxygen and hyperbaric oxygen in our patient caused visually apparent and/or physical examination-documented neurological improvements consistent with gene signaling effects of oxygen and then pressure20 as well as the clinical effects demonstrated in chronic neurological disorders.11,12,13,14,15,16
Eight weeks post-drowning, our patient exhibited severe predictable1,2 neurological deficits and MRI findings4,5,6,22 that were reversed by short-duration normobaric oxygen and hyperbaric oxygen therapy. The decision to apply normobaric 100% oxygen was dictated by author PGH’s inability to obtain HBOT in the patient’s location and PGH’s experience using normobaric 100% oxygen in unpublished cases of chronic multi-infarct dementia, traumatic brain injury and extremity ulcers. Cortical cystic lesion and cortical atrophy regression has been reported in a 2-year-old post-neonatal HIE, but white matter loss was unchanged.23 Spontaneous regression of both cortical and white matter atrophy is contrary to the natural evolution of non-neonatal HIE.24 T2 signal changes on days 4 and 32 indicated permanent brain tissue injury or loss (increased fluid spaces) and limited tissue salvage/future neurological improvement. The diffuse regrowth of tissue was validated by visual inspection and multiple calculations. A minimum 12.3% volume loss (86 cm3, underestimated due to inclusion of enlarged ventricles) was restored to the normal volume of a 24–36-month-old child.
Functional imaging has been used since 1990 in chronic neurological disorders,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43 to document HBOT-induced neurological improvements. These improvements have evaded anatomical imaging due to the paucity of subacute pediatric cases, treatment of severe cases with large tissue loss, and the inability to capture metabolic25,29,30 and microscopic changes,31,35,42,44 including microscopic neurogenesis.42,45 The startling macroscopic regrowth of tissue in this case is explicable by early intervention prior to long-term tissue degeneration (e.g., Wallerian degeneration, apoptosis) in a growing child. The synergy of increased oxygen and increased oxygen with pressure in the hormone-rich childhood cerebral milieu is consistent with the synergy of growth hormones and hyperbaric oxygen17,18 caused by normobaric and hyperbaric oxygen-induced gene signaling trophic,21,46 anti-inflammatory,21,46 and anti-apoptotic effects21,46 on brain tissue.47 Trophism is the basis of oxygen and hyperbaric oxygen based wound-healing in animals and humans,48 including central nervous system injuries48 and is underscored in this patient by the concomitant rapid neurological improvements.
Substantial animal and human literature has demonstrated beneficial effects of hyperacute HBOT for resuscitation and post-resuscitation recovery from global ischemia/anoxia.49 Late application to drowning patients32,36 and other global ischemia patients28,49,50 produces more modest effects. When hyperacute HBOT is precluded by availability or other non-medical factors bridging short-duration repetitive normobaric oxygen therapy may be an option until HBOT is available. Such low-risk medical treatment may have a profound effect on recovery of function in similar patients who are neurologically devastated by drowning; however it is impossible to conclude from this single case if the sequential application of normobaric oxygen then HBOT would be more effective than HBOT alone.
CONCLUSION
Short duration normobaric oxygen and hyperbaric oxygen therapy in the subacute phase of drowning recovery resulted in video-documented near-complete resolution of severe neurological deficits and near-complete reversal of gray and white matter atrophy on MRI. Hyperoxic and hyperbaric gene signaling-induced growth of both gray and white matter is the most likely explanation.
Acknowledgments
We thank Chris and Kristal Carlson for allowing us to report the treatment of their daughter, Henry Arnold, a Certificate of Artistry Media Arts senior student at Lusher Charter School, New Orleans, LA, USA for his expeditious and skilled editing of the final video used in this report, and Juliette Lucarini, R.N., research nurse for the Family Physicians Center and Harch Hyperbarics, Inc., New Orleans, LA, USA, for her counseling/interface with the family during the normobaric oxygen administration and facilitation of their eight week sojourn to New Orleans. We would also like to acknowledge the use of Osirix Open-source workstation software without which the image analysis would not have been possible.
Footnotes
Conflicts of interest
PGH is co-owner of Harch Hyperbarics, Inc., a corporation that performs hyperbaric medicine consulting and expert witness testimony/opinions. He is also on the board of directors of the International Hyperbaric Medical Association (IHMA), a non-profit corporation. He derives no income from the IHMA. EFF is president of the International Hyperbaric Medical Foundation (IHMF), a non-profit corporation that promotes education, research, and teaching in hyperbaric medicine. He derives no income from the IHMF.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient’s parents have given her consent for her images and other clinical information to be reported in the journal. The patient’s parents understand that their names will be published.
Contributor agreement
A statement of “Publishing Agreement” has been signed by an authorized author on behalf of all authors prior to publication.
Plagiarism check
This paper has been checked twice with duplication-checking software iThenticate.
Peer review
A double-blind and stringent peer review process has been performed to ensure the integrity, quality and significance of this paper.
Open peer reviewers
Reviewer 1, Lei Huang, Loma Linda University, USA; Reviewer 2, Wen-wu Liu, Second Military Medical University, China.
Additional file
Additional Video 1: Author PGH video exams at hyper-baric clinic pre- and post-hyperbaric oxygen therapy-Eden Carlson.
REFERENCES
- 1.Szpilman D, Bierens JJ, Handley AJ, Orlowski JP. Drowning. N Engl J Med. 2012;366:2102–2110. doi: 10.1056/NEJMra1013317. [DOI] [PubMed] [Google Scholar]
- 2.Wagner C. Pediatric submersion injuries. Air Med J. 2009;28:116–119. doi: 10.1016/j.amj.2009.02.009. [DOI] [PubMed] [Google Scholar]
- 3.Felton H, Myers J, Liu G, Davis DW. Unintentional, non-fatal drowning of children: US trends and racial/ethnic disparities. BMJ Open. 2015;5:e008444. doi: 10.1136/bmjopen-2015-008444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ishaque M, Manning JH, Woolsey MD, Franklin CG, Tullis EW, Fox PT. Lenticulostriate arterial distribution pathology may underlie pediatric anoxic brain injury in drowning. Neuro-image Clin. 2016;11:167–172. doi: 10.1016/j.nicl.2016.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huang BY, Castillo M. Hypoxic-ischemic brain injury: imaging findings from birth to adulthood. Radiographics. 2008;28:417–439. doi: 10.1148/rg.282075066. quiz 617. [DOI] [PubMed] [Google Scholar]
- 6.Dubowitz DJ, Bluml S, Arcinue E, Dietrich RB. MR of hypoxic encephalopathy in children after near drowning: correlation with quantitative proton MR spectroscopy and clinical outcome. AJNR Am J Neuroradiol. 1998;19:1617–1627. [PMC free article] [PubMed] [Google Scholar]
- 7.Semple-Hess J, Campwala R. Pediatric submersion injuries: emergency care and resuscitation. Pediatr Emerg Med Pract. 2014;11:1. [PubMed] [Google Scholar]
- 8.Gore A, Muralidhar M, Espey MG, Degenhardt K, Mantell LL. Hyperoxia sensing: from molecular mechanisms to significance in disease. J Immunotoxicol. 2010;7:239–254. doi: 10.3109/1547691X.2010.492254. [DOI] [PubMed] [Google Scholar]
- 9.Singhal AB, Benner T, Roccatagliata L, et al. A pilot study of normobaric oxygen therapy in acute ischemic stroke. Stroke. 2005;36:797–802. doi: 10.1161/01.STR.0000158914.66827.2e. [DOI] [PubMed] [Google Scholar]
- 10.Rockswold SB, Rockswold GL, Zaun DA, et al. A prospective, randomized clinical trial to compare the effect of hyperbaric to normobaric hyperoxia on cerebral metabolism, intracranial pressure, and oxygen toxicity in severe traumatic brain injury. J Neurosurg. 2010;112:1080–1094. doi: 10.3171/2009.7.JNS09363. [DOI] [PubMed] [Google Scholar]
- 11.Heuser G, Heuser SA, Rodelander D, Aguilera O, Uszler M. Treatment of Neurologically Impaired Adults and Children with “Mild” Hyperbaric Oxygen (1.3ATA and 24% Oxygen). The Proceedings of the 2nd International Symposium on Hyperbaric Oxygenation for Cerebral Palsy and the Brain-Injured Child; 2002; Flagstaff [Google Scholar]
- 12.Wolf G, Cifu D, Baugh L, Carne W, Profenna L. The effect of hyperbaric oxygen on symptoms after mild traumatic brain injury. J Neurotrauma. 2012;29:2606–2612. doi: 10.1089/neu.2012.2549. [DOI] [PubMed] [Google Scholar]
- 13.Miller RS, Weaver LK, Bahraini N, et al. Effects of hyperbaric oxygen on symptoms and quality of life among service members with persistent postconcussion symptoms: a randomized clinical trial. JAMA Intern Med. 2015;175:43–52. doi: 10.1001/jamainternmed.2014.5479. [DOI] [PubMed] [Google Scholar]
- 14.Rossignol DA, Rossignol LW, Smith S, et al. Hyperbaric treatment for children with autism: a multicenter, randomized, double-blind, controlled trial. BMC Pediatr. 2009;9:21. doi: 10.1186/1471-2431-9-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Collet JP, Vanasse M, Marois P, et al. Hyperbaric oxygen for children with cerebral palsy: a randomised multicentre trial. HBO-CP Research Group. Lancet. 2001;357:582–586. doi: 10.1016/s0140-6736(00)04054-x. [DOI] [PubMed] [Google Scholar]
- 16.Mukherjee A, Raison M, Sahni T, et al. Intensive rehabilitation combined with HBO2 therapy in children with cerebral palsy: a controlled longitudinal study. Undersea Hyperb Med. 2014;41:77–85. [PubMed] [Google Scholar]
- 17.Siddiqui A, Davidson JD, Mustoe TA. Ischemic tissue oxygen capacitance after hyperbaric oxygen therapy: a new physiologic concept. Plast Reconstr Surg. 1997;99:148–155. doi: 10.1097/00006534-199701000-00023. [DOI] [PubMed] [Google Scholar]
- 18.Zhao LL, Davidson JD, Wee SC, Roth SI, Mustoe TA. Effect of hyperbaric oxygen and growth factors on rabbit ear ischemic ulcers. Arch Surg. 1994;129:1043–1049. doi: 10.1001/archsurg.1994.01420340057010. [DOI] [PubMed] [Google Scholar]
- 19.Macdonald AG, Fraser PJ. The transduction of very small hydrostatic pressures. Comp Biochem Physiol A Mol Integr Physiol. 1999;122:13–36. doi: 10.1016/s1095-6433(98)10173-3. [DOI] [PubMed] [Google Scholar]
- 20.Chen Y, Nadi NS, Chavko M, Auker CR, McCarron RM. Microarray analysis of gene expression in rat cortical neurons exposed to hyperbaric air and oxygen. Neurochem Res. 2009;34:1047–1056. doi: 10.1007/s11064-008-9873-8. [DOI] [PubMed] [Google Scholar]
- 21.Godman CA, Chheda KP, Hightower LE, Perdrizet G, Shin DG, Giardina C. Hyperbaric oxygen induces a cytoprotective and angiogenic response in human microvascular endothelial cells. Cell Stress Chaperones. 2010;15:431–442. doi: 10.1007/s12192-009-0159-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Grant PE, Yu D. Acute injury to the immature brain with hypoxia with or without hypoperfusion. Radiol Clin North Am. 2006;44:63–77. doi: 10.1016/j.rcl.2005.08.001. viii. [DOI] [PubMed] [Google Scholar]
- 23.Bekiesinska-Figatowska M, Borszewska-Kornacka MK, Antc zak-Marach D, Szczepanik E. Regression of cystic lesions on brain MRI in a child with hypoxic-ischemic encephalopathy treated with selective head cooling. Ginekol Pol. 2013;84:151–156. doi: 10.17772/gp/1557. [DOI] [PubMed] [Google Scholar]
- 24.Gutierrez LG, Rovira A, Portela LA, Leite Cda C, Lucato LT. CT and MR in non-neonatal hypoxic-ischemic encephalopathy: radiological findings with pathophysiological correlations. Neuroradiology. 2010;52:949–976. doi: 10.1007/s00234-010-0728-z. [DOI] [PubMed] [Google Scholar]
- 25.Neubauer RA, Gottlieb SF, Kagan RL. Enhancing “idling” neurons. Lancet. 1990;335:542. doi: 10.1016/0140-6736(90)90777-3. [DOI] [PubMed] [Google Scholar]
- 26.Neubauer RA, Gottlieb SF, Miale A., Jr Identification of hypo-metabolic areas in the brain using brain imaging and hyperbaric oxygen. Clin Nucl Med. 1992;17:477–481. doi: 10.1097/00003072-199206000-00010. [DOI] [PubMed] [Google Scholar]
- 27.Neubauer RA, James P. Cerebral oxygenation and the recoverable brain. Neurol Res. 1998;20(Suppl 1):S33–36. doi: 10.1080/01616412.1998.11740606. [DOI] [PubMed] [Google Scholar]
- 28.Golden ZL, Neubauer R, Golden CJ, Greene L, Marsh J, Mleko A. Improvement in cerebral metabolism in chronic brain injury after hyperbaric oxygen therapy. Int J Neurosci. 2002;112:119–131. doi: 10.1080/00207450212027. [DOI] [PubMed] [Google Scholar]
- 29.Van Meter KW, Weiss L, Harch PG, et al. Should the pressure be off or on in the use of oxygen in the treatment of carbon monoxide-poisoned patients? Ann Emerg Med. 1994;24:283–288. doi: 10.1016/s0196-0644(05)83748-5. [DOI] [PubMed] [Google Scholar]
- 30.Harch PG, Van Meter KW, Neubauer RA, Gottlieb SF. Use of HMPAO SPECT for assessment of response to HBO in ischemic/hypoxic encephalopathies. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Seattle: Hogrefe & Huber Publishers; 1996. pp. 480–491. [Google Scholar]
- 31.Harch PG. Late treatment of decompression illness and use of SPECT brain imaging. Treatment of Decompression Illness, 45th Workshop of the Undersea and Hyperbaric Medical Society; Undersea and Hyperbaric Medical Society, Kensington, MD, USA. 1996:203–242. [Google Scholar]
- 32.Harch PG, Neubauer RA. Hyperbaric oxygen therapy in global cerebral ischemia/anoxia and coma. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Chapter 18. Seattle: Hogrefe & Huber Publishers; 1999. pp. 318–349. [Google Scholar]
- 33.Harch PG, Neubauer RA, Uszler JM, James PB, Maxfield W. Functional imaging. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Seattle: Hogrefe & Huber Publishers; 1999. pp. 616–625. [Google Scholar]
- 34.Van Meter K, Weiss L, Harch PG. Hyperbaric oxygen in emergency medicine. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Seattle: Hogrefe & Huber Publishers; 1999. pp. 556–589. [Google Scholar]
- 35.Barratt DM, Harch PG, Van Meter K. Decompression illness in divers: a review of the literature. Neurologist. 2002;8:186–202. doi: 10.1097/00127893-200205000-00005. [DOI] [PubMed] [Google Scholar]
- 36.Harch PG. The Dosage of Hyperbaric Oxygen in Chronic Brain Injury. In: James T, Joiner, editors. The Proceedings of the 2nd International Symposium on Hyperbaric Oxygenation for Cerebral palsy and the Brain-Injured Child. Flagstaff, AZ, USA: Best Publishing Co; 2002. pp. 31–56. [Google Scholar]
- 37.Harch PG, Neubauer RA, Uszler JM, James PB. Appendix: diagnostic imaging and hbo therapy. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Chapter 41. Seattle: Hogrefe & Huber Publishers; 2004. pp. 471–485. [Google Scholar]
- 38.Harch PG, Neubauer RA. Hyperbaric oxygen therapy in global cerebral ischemia/anoxia and coma. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Chapter 18. Seattle: Hogrefe & Huber Publishers; 2004. pp. 223–261. [Google Scholar]
- 39.Neubauer V, Neubauer RA, Harch PG. HBO in the management of cerebral palsy. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Chapter 21. Seattle: Hogrefe & Huber Publishers; 2004. pp. 287–296. [Google Scholar]
- 40.Harch PG, McCullough VE. The Oxygen Revolution. New York: Hatherleigh Press; 2007. [Google Scholar]
- 41.Harch PG, Fogarty EF, Staab PK, Van Meter K. Low pressure hyperbaric oxygen therapy and SPECT brain imaging in the treatment of blast-induced chronic traumatic brain injury (post-concussion syndrome) and post traumatic stress disorder: a case report. Cases J. 2009;2:6538. doi: 10.1186/1757-1626-0002-0000006538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Harch PG, Andrews SR, Fogarty EF, et al. A phase I study of low-pressure hyperbaric oxygen therapy for blast-induced post-concussion syndrome and post-traumatic stress disorder. J Neurotrauma. 2012;29:168–185. doi: 10.1089/neu.2011.1895. [DOI] [PubMed] [Google Scholar]
- 43.Boussi-Gross R, Golan H, Fishlev G, et al. Hyperbaric oxygen therapy can improve post concussion syndrome years after mild traumatic brain injury-randomized prospective trial. PLoS One. 2013;8:e79995. doi: 10.1371/journal.pone.0079995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Palmer AC, Calder IM, Yates PO. Cerebral vasculopathy in divers. Neuropathol Appl Neurobiol. 1992;18:113–124. doi: 10.1111/j.1365-2990.1992.tb00773.x. [DOI] [PubMed] [Google Scholar]
- 45.Harch PG, Kriedt C, Van Meter KW, Sutherland RJ. Hyperbaric oxygen therapy improves spatial learning and memory in a rat model of chronic traumatic brain injury. Brain Res. 2007;1174:120–129. doi: 10.1016/j.brainres.2007.06.105. [DOI] [PubMed] [Google Scholar]
- 46.Kendall AC, Whatmore JL, Harries LW, Winyard PG, Eggleton P, Smerdon GR. Different oxygen treatment pressures alter inflammatory gene expression in human endothelial cells. Undersea Hyperb Med. 2013;40:115–123. [PubMed] [Google Scholar]
- 47.Lin KC, Niu KC, Tsai KJ, et al. Attenuating inflammation but stimulating both angiogenesis and neurogenesis using hyperbaric oxygen in rats with traumatic brain injury. J Trauma Acute Care Surg. 2012;72:650–659. doi: 10.1097/TA.0b013e31823c575f. [DOI] [PubMed] [Google Scholar]
- 48.Weaver LK, editor. The Hyperbaric Oxygen Therapy Committee Report. North Palm Beach: Best Publishing Co; 2014. Hyperbaric oxygen therapy indications. [Google Scholar]
- 49.Harch PG. Hyperbaric oxygen therapy in global cerebral ischemia/anoxia and coma. In: Jain KK, editor. Textbook of Hyperbaric Medicine. Chapter 20. Cham: Springer; 2017. pp. 269–319. [Google Scholar]
- 50.Churchill S, Weaver LK, Deru K, et al. A prospective trial of hyperbaric oxygen for chronic sequelae after brain injury (HYBOBI) Undersea Hyperb Med. 2013;40:165–193. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.