Abstract
Background
Anemia is a condition in which the number of red blood cells or their oxygen-carrying capacity is insufficient to meet physiologic needs, which varies by age, sex, altitude, smoking, and pregnancy status. The study aim is to determine the prevalence and factors associated with anemia among pregnant women attending a hospital in southern Ethiopia using a structured interview administered questionnaire.
Methods
Facility-based cross-sectional study was conducted from March 01–April 30 2015 at Butajira General Hospital, Ethiopia. A total of 217 women responded to the questionnaire and provided blood and stool samples for analysis. Data were analyzed using Statistical packages for social sciences version 20 for windows.
Result and conclusions
The overall burden of anemia in this study was 27.6%. Residence, ANC follow up, history of excess menstrual bleeding and interpregnancy interval were statistically associated with anemia among the pregnant women. Therefore, working in the identified gaps could reduce the current burden of anemia among pregnant women in the study area.
Keywords: Anemia, Pregnancy, Antenatal care
Background
Anemia is a condition in which the number of red blood cells or their oxygen-carrying capacity is insufficient to meet physiologic needs, which varies by age, sex, altitude, smoking, and pregnancy status [1]. Anemia is a global public health problem affecting both developing and developed countries with major consequences for human health as well as socio-economic development. It occurs at all stages of the life cycle but is more prevalent in pregnant women and young children [2]. In developing countries, the cause of anemia during pregnancy is multifactorial and includes nutritional deficiencies of iron, folate, and vitamin B12 and parasitic diseases, such as malaria and hookworm. The relative contribution of each of these factors to anemia during pregnancy varies greatly by geographical location, season, and dietary practice [3]. During pregnancy, anemia is a major cause of morbidity and mortality of pregnant women in developing countries and has both maternal and fetal consequences [4–9]. It may also lead to premature births [10], low birth weight [11], fetal cognitive impairment, and death [12, 13]. According to the report of World Health Organization (WHO), the prevalence of anemia among pregnant women is 41.8%. The highest prevalence rate (61.3%) is found among pregnant women in Africa [14]. According to the Ethiopian Demographic Health Survey (EDHS) report, 17% women are estimated to be anemic and 22% of the pregnant women are anemic [15]. Because of different Socio-economic status, behavioral, geographical and methodological difference (design and methods) the national figure of EDHS could not represent the prevalence of anemia among pregnant women found in different part of Ethiopia. Thus, disclosing the burden and associated factor of anemia among pregnant women in the study area will help to develop evidence-based decision and intervention strategies to improve the health status of pregnant women. Furthermore, it will contribute to discount predisposing factors of anemia. Therefore, this study is aimed to determine the burden of anemia and associated risk factors among pregnant women attending Antenatal Care Clinic (ANC) follow up at Butajira General Hospital, Southern Ethiopia.
Methods
Study setting and design
Facility based cross-sectional study was done at Butajira General Hospital from March 01 to April 30, 2015 Southern Ethiopia. Butajira General Hospital is located in the South Nation Nationality and Peoples Region (SNNPR), which is 135 km far from Addis Ababa, the capital city of Ethiopia. On average, 405 pregnant women have been registered in the ANC follow up.
Study population
All pregnant women visiting ANC follow up from March 01 to April 30, 2015 and fulfilled the inclusion criteria included to the study. Current illness and drug treatment for chronic illness were the exclusion criteria. Subject recruitment was conducted on volunteer basis after having a clear understanding of the research.
Sample size determination and sampling procedures
The sample size was determined based on the single population proportion formula using Z2 × p × q/d2 with a 95% confidence interval, 5% margin of error and an assumption that 50% of pregnant women are anemic in the study area. Thus, the total sample size was 384.16. Since the total ANC followers in the study area are 405 which is less than 10,000 we used correction formula to calculate the final sample size. Therefore, considering non-response rate (10%), the final sample size for this study were 217. The sample random sampling (lottery) method was used to select the first study participant and a systematic random sampling method (k = N/n = 405/217 = 1.86 = 2) was used to select the rest of the others. Thus, every second (k = 2) pregnant women who mate our inclusion criteria was selected.
Methods of data collection
Information on socio-demographic characteristics, obstetric and gynecological history, and dietary patterns were collected using interviewer-administered pretested questionnaires. Hematological parameters such as Mean cell volume (MCV), Mean cell hemoglobin (MCH) and Mean cell hemoglobin concentration (MCHC) and the analysis were done using CELL-DYN 1800. Anemia in pregnancy was defined as Hemoglobin (Hb) less than 11 g/dl [16]. Stool specimens were collected from each study participant and an examination of intestinal parasitic infections was conducted using saline wet smear and formol-ether concentration techniques [17]. Bivariate and multivariate logistic regression analysis were done to assess the independent risk factors associated with anemia. The goodness of fit of the final logistic model was tested using Hosmer and Lemeshow test (p < 0.05).
Data quality assurance
To assure the quality of the data Standard operating procedures (SOPs) were followed during specimen collection and other laboratory procedures. All reagents used were checked for their expiry date and prepared according to the manufacturer’s instructions. Training was given for the data collectors to minimize technical and observer biases.
Methods of data analysis
Data were edited, cleaned and checked for its completeness and entered into EpiData 3.1 then exported to Statistical packages for social sciences (SPSS) Version 20 for analysis. Categorical variables were summarized as numbers and percentages. Both bivariate and multivariate logistic regression analysis were done to assess the independent risk associated factors for anemia. All variables with a p value less than 0.05 were considered as statistical significance. The goodness of fit of the final logistic model was tested using Hosmer and Lemeshow test at a p value less than 0.05.
Ethical statement
Ethical clearance was obtained from Jimma University Ethical Review Committee. Letter of permission to conduct the study was obtained from Butajira General Hospital ANC clinic. Written informed consent was obtained from each study participant. The purpose of the study was clearly described to the study participants including the benefits and risks of the study. Any information concerning the participants was kept confidential and the specimen collected from the participants was only analyzed for the intended purposes. The participants were enrolled only after they were sufficiently counseled and their informed consents have been obtained. Positive findings for intestinal helminths and hemoglobin value below accepted value were communicated with the respective health professionals of Butajira General Hospital ANC clinic for possible interventions.
Results
Socio-demographic characteristics
A total of 217 study participants were involved in the study. The mean (±SD) age of the study participants were 26.87 (±5.703). The majority of the study subjects, 213 (98.1%) were married, 94 (43.3%) were primary school completed, 99 (45.6%) where housewife and 168 (77.4%) were urban inhabitants (Table 1).
Table 1.
Bivariate analysis of socio-demographic characteristics and anemia among pregnant women (n = 217) in Butajira General Hospital, Southern Ethiopia, March 1–April 30, 2015
| Variables | Total (n = 217) n (%) | Anaemia | COR (95% CI) | p value | |
|---|---|---|---|---|---|
| Yes (n = 60) n = (%) | No (n = 157) n = (%) | ||||
| Age in years | |||||
| <20 | 17 (7.8) | 7 (3.2) | 10 (4.6) | 1r | 0.755 |
| 21–30 | 94 (43.3) | 29 (48.3) | 65 (41.4) | 2.105 (0.210–1.621) | |
| 31–40 | 70 (32.2) | 18 (30) | 52 (33.1) | 0.606 (0.166–1.540) | |
| >40 | 36 (16.5) | 6 (10) | 30 (19.1) | 1.650 (0.208–4.882) | |
| Educational status | |||||
| Illiterate | 32 (14.7) | 11 (5.1) | 21 (9.6) | 1r | 0.999 |
| Primary | 94 (43.3) | 30 (13.8) | 64 (29.4) | 0.895 (0.383–2.091) | |
| Secondary | 72 (33.2) | 15 (6.9) | 57 (26.2) | 0.502 (0.199-1.267) | |
| Tertiary | 15 (6.9) | 4 (1.8) | 11 (5.1) | 0.694 (0.179–2.697) | |
| Others | 4 (1.8) | 0 (0) | 4 (1.8) | 0.000 (0.000, –) | |
| Occupation status | |||||
| Housewife | 99 (45.6) | 28 (12.9) | 71 (32.7) | 1r | 0.931 |
| Trader | 61 (28.1) | 15 (6.9) | 46 (21.1) | 0.827 (0.399–1.713) | |
| Daily laborer | 11 (5.1) | 4 (1.8) | 7 (3.2) | 1.449 (0.393–5.338) | |
| Farming | 6 (2.8) | 3 (1.3) | 3 (1.3) | 2.536 (0.483–13.323) | |
| Self employed | 10 (4.6) | 2 (0.9) | 8 (3.68) | 0.634 (0.127–3.172) | |
| NGO employee | 6 (2.8) | 1 (0.46) | 5 (2.3) | 0.507 (0.057–4.537) | |
| Governmental employee | 24 (11.1) | 7 (3.2) | 17 (7.8) | 1.044 (0.391–2.790) | |
| Residence | |||||
| Urban | 168 (77.4) | 40 (18.4) | 128 (58.9) | 0.453 (0.232–0.887) | 0.021* |
| Rural | 49 (22.6) | 20 (9.2) | 29 (13.3) | 1r | |
| Marital status | |||||
| Single | 4 (1.8) | 4 (1.8) | 0 (0) | 1r | 0.999 |
| Married | 213 (98.2) | 56 (25.8) | 157 (72.3) | – | |
| Monthly income (ETB) | |||||
| <500 | 80 (36.9) | 24 (11) | 56 (25.8) | 1r | 0.453 |
| 500–1000 | 79 (36.4) | 22 (10.1) | 57 (26.2) | 0.901 (0.453–1.788) | |
| >1000 | 58 (26.7) | 14 (6.4) | 44 (20.2) | 0.742 (0.344–1.601) | |
1r set as reference, NGO non-governmental employee, 1 US $ 18.6 Ethiopian Birr (ETB)
* p < 0.05
Health care related factors
From the total 217 study participants, 54 (24.9%) and 53 (24.4%) study subjects had the history of malarial infection prior to the data collection time and excess menstrual related bleeding respectively. The majority of the study participants, 171 (78.8%) had ANC follow-up and 105 (48.4%) had a habit of eating iron-rich animal foods sources (AFS) such as red meat, milk and milk products, egg, poultry, and fish. Study participants with a birth interval of fewer than 2 years 46 (76.7%) had shown to be more anemic than those with an interval of greater than or equal to 2 years 5 (8.3%). Among the study participants, 28 (13%) were infected with intestinal parasites and 21 (9.6%) of them were anemic. Blood film examination was also done to identify blood hemoparasites, particularly for malaria. However, no hemoparasite was identified during the study period (Table 2).
Table 2.
Bivariate analysis of factors associated with anaemia among pregnant women (n = 217) in Butajira General Hospital, Southern Ethiopia, March 1–April 30, 2015
| Variables | Total (n = 217) n (%) | Anaemia | COR (95% CI) | p value | |
|---|---|---|---|---|---|
| Yes (n = 60) n = (%) | No (n = 157) n = (%) | ||||
| History of Malaria | |||||
| Yes | 54 (24.9) | 24 (40) | 30 (19.1) | 1r | 0.002* |
| No | 163 (75.1) | 36 (60) | 127 (80.9) | 0.354 (0.185–0.680) | |
| History of excess menstrual bleeding | |||||
| Yes | 53 (24.4) | 44 (73.3) | 9 (5.7) | 1r | <0.0001* |
| No | 164 (75.6) | 16 (26.7) | 148 (94.3) | 0.022 (0.009–0.053) | |
| Bare foot walking | |||||
| Yes | 19 (8.8) | 7 (11.7) | 12 (7.5) | 1r | 0.352 |
| No | 198 (91.2) | 53 (88.3) | 145 (92.5) | 0.627 (0.234–1.676) | |
| ANC follow up | |||||
| Yes | 171 (78.8) | 24 (40) | 147 (93.6) | 0.045 (0.020–0.103) | <0.0001 * |
| No | 46 (21.2) | 36 (60) | 10 (6.3) | 1r | |
| ITN utilization | |||||
| Yes | 81 (37.3) | 17 (28.3) | 64 (40.8) | 0.574 (0.301–1.095) | 0.092 |
| No | 136 (62.7) | 43 (71.7) | 93 (59.2) | 1r | |
| Use of deworming | |||||
| Yes | 102 (47.0) | 26 (43.3) | 76 (48.4) | 0.815 (0.4481–0.483) | 0.503 |
| No | 115 (53.0) | 34 (56.7) | 81 (51.6) | 1r | |
| Use of contraceptive | |||||
| Yes | 173 (79.7) | 48 (80) | 125 (79.6) | 1.024 (0.487–2.151) | 0.950 |
| No | 44 (20.3) | 12 (20) | 32 (21.4) | 1r | |
| Iron-foliate supplementations | |||||
| Yes | 156 (71.9) | 42 (70) | 114 (72.6) | 0.880 (0.458–1.693) | 0.702 |
| No | 61 (28.1) | 18 (30) | 43 (27.4) | 1r | |
| Habit of drinking coffee or tea after meal | |||||
| Yes | 175 (80.6) | 49 (81.7) | 126 (80.3) | 0.912 (0.425–1.957) | 0.814 |
| No | 42 (19.4) | 11 (18.3) | 31 (19.7) | 1r | |
| Consumption of ASF | |||||
| Yes | 105 (48.4) | 21 (35) | 84 (53.5) | 0.468 (0.253–0.867) | 0.016* |
| No | 112 (51.6) | 39 (65) | 73 (46.5) | 1r | |
| Consumption of PSF | |||||
| Yes | 35 (16.1) | 14 (23.3) | 21 (13.4) | 1r | 0.078 |
| No | 182 (83.9) | 46 (76.7) | 136 (86.6) | 0.507 (0.239, 1.079) | |
| Pregnancy trimester | |||||
| 1st | 73 (33.6) | 21 (35) | 52 (33.2) | 1r | 0.350 |
| 2nd | 66 (30.4) | 11 (18.3) | 55 (35) | 0.495 (0.218–1.127) | |
| 3rd | 78 (35.9) | 28 (46.7) | 50 (31.8) | 1.387 (0.698–2.754) | |
| Parity | |||||
| 0 | 132 (61.3) | 5 (8.3) | 128 (81.5) | 1r | 0.998 |
| 1–4 | 63 (29) | 33 (55) | 29 (18.5) | 29.131 (10.470–81.054) | |
| ≥5 | 22 (10.1) | 22 (36.7) | 0 (0) | 4.136E10 (0.000) | |
| Inter pregnancy interval (years) | |||||
| <2 | 57 (26.3) | 46 (76.7) | 11 (7) | 1r | <0.0001 * |
| ≥2 | 108 (49.8) | 5 (8.3) | 103 (65.6) | 0.012 (0.004–0.035) | |
| Intestinal parasite | |||||
| Present | 28 (12.9) | 21 (35) | 7 (4.5) | 1r | <0.0001 * |
| Absent | 189 (87.1) | 39 (65) | 150 (95.5) | 0.087 (0.034–0.219) | |
1r set as reference, p p value, PSF plant source food, ASF animal source food
* p < 0.05
Prevalence and severity of anemia
Participant’s hemoglobin level was used to determine the prevalence of anemia. The overall burden of anemia among the study participants was 27.6% (60/217). The mean hemoglobin level (adjusted for altitude) was 11.2 gm/dl. From the current occurrence of anemia, 3 (5%) of them were identified as severely anemic, 27 (45%) of them were moderately anemic and 30 (50%) were mildly anemic (Fig. 1).
Fig. 1.
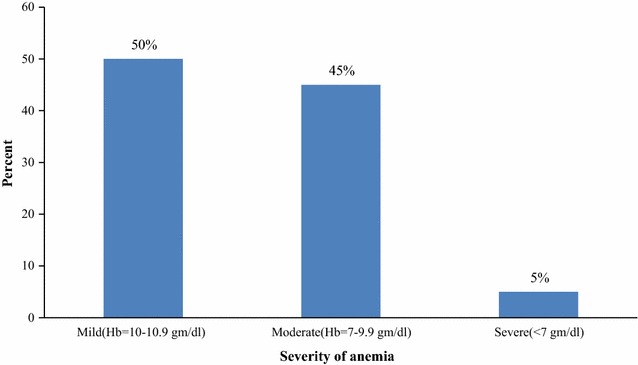
Severity of anemia among pregnant women attending ANC in Butajira General Hospital Southern Ethiopia, March 1–April 30, 2015
Predictors of anemia
Both bivariate and multivariable logistic regression analysis were done to identify the independent predictors of anemia among pregnant women. All the variables were analyzed in bivariate logistic regression analysis and those with a p-value less than 0.05 were considered in multivariable logistic regression analysis. By doing so finally residence, ANC follow up, history of excess menstrual bleeding and interpregnancy interval were identified as predictors of anemia among the study participants (Table 3).
Table 3.
Multivariable logistic regression analysis of factors associated with anemia among pregnant women (n = 217) in Butajira General Hospital, Southern Ethiopia, March 1–April 30, 2015
| Variables | Total (n = 217) n (%) | Anaemia | AOR (95% CI) | p value | |
|---|---|---|---|---|---|
| Yes (n = 60) n = (%) | No (n = 157) n = (%) | ||||
| Residence | |||||
| Urban | 168 (77.4) | 40 (18.4) | 128 (58.9) | 0.167 (0.041–0.682) | 0.013 * |
| Rural | 49 (22.6) | 20 (9.2) | 29 (13.3) | 1r | |
| History of Malaria prior to data collection time | |||||
| Yes | 54 (24.9) | 24 (11) | 30 (13.8) | 1r | 0.525 |
| No | 163 (75.1) | 36 (16.5) | 127 (58.5) | 1.622 (0.365–7.211) | |
| History of excess menstrual bleeding | |||||
| Yes | 53 (24.4) | 16 (7.3) | 9 (4.1) | 1r | <0.001 * |
| No | 164 (75.6) | 44 (20.2) | 148 (68.2) | 0.028 (0.006–0.135) | |
| ANC follow up | |||||
| Yes | 171 (78.8) | 24 (9.2) | 147 (67.7) | 0.082 (0.018–0.370) | <0.001 * |
| No | 46 (21.2) | 36 (16.5) | 10 (4.6) | 1r | |
| Consumption of AFS | |||||
| Yes | 105 (48.4) | 21 (9.6) | 84 (38.7) | 1.264 (0.365–4.380) | 0.712 |
| No | 112 (51.6) | 39 (17.9) | 73 (33.6) | 1r | |
| Inter pregnancy interval (years) | |||||
| <2 | 57 (26.3) | 46 (21.1) | 11 (5) | 1r | 0.016 * |
| ≥2 | 108 (49.8) | 5 (2.3) | 103 (47.4) | 0.133 (0.026–0.685) | |
| Intestinal parasite | |||||
| Present | 28 (12.9) | 21 (9.6) | 7 (3.2) | 1r | 0.121 |
| Absent | 189 (87.1) | 39 (17.9) | 150 (69.1) | 0.245 (0.042–1.450) | |
1r set as reference, AFS animal food sources
* p < 0.05
Discussions
Anemia is a worldwide problem affecting all physiologic group. Pregnant women and children are the most vulnerable group for anemia. In the current study, the overall burden of anemia among pregnant women was 27.6%. The burden of anemia in the current study is in harmony with the study conducted by Ethiopian DHS 2005 (27.4%) and a cross-sectional study carried out in Southeast Ethiopia (27.9%) [18, 19]. However, our finding is slightly higher as compared to the finding from central Ethiopia, Addis Ababa (21.3%) [20], and from Northwest Ethiopia (21.6%) [21]. The burden of anemia in the current study is considerably lower than previous study reports from West Arsi Zone, Eastern part of Ethiopia (36.6%) [22], and Jimma Zone, Southwest part of Ethiopia (38.2-53.9%) [1, 23]. Such magnitude differences are due to geographical, economical, seasonal, dietary and behavioral variations [3, 20]. The presences and absences of malaria and intestinal parasitic infection are also among the contributing factors affecting the magnitude of anemia [24]. From multivariable logistic regression analysis, a significant association was found between anemia and residence, history of excess menstrual bleeding, ANC follow up, and interpregnancy interval. Anaemia among pregnant women who live in an urban setting is 0.167 times as likely as anemia among pregnant women who live rural setting. Association of rural residence with the occurrence of anemia has also been reported earlier [1, 19, 23]. The reason for a higher burden of anemia among pregnant women from rural areas may be related to inaccessibility of health care centers. Thus, they lack information on factors causing anemia and possible strategies to prevent the risk factors of anemia. Pregnant women with a history of heavy or excess menstrual cycle were more anemic than those with normal menstruation cycle [19, 23]. In the present finding, the odds of anemia among pregnant women with the normal menstrual cycle is 0.028 times the odds of anemia among pregnant women who had heavy or excess menstrual cycle. History of excessive bleeding can lead to anemia. The anemia may make her feel weak and tired, and she may also experience shortness of breath, rapid heart rate, and lightheadedness. The odds of anemia among pregnant women who had ANC follow up were 0.918 times less likely than anemia among pregnant women who didn’t have ANC follow up. This result is inconsistent with the study conducted in Addis Ababa [20]. Pregnant women who attend ANC follow up were supported by health professionals to prevent anemia. Antenatal care counseling helps in motivating pregnant women to take iron rich food and iron-folic acid tablets. The present study has shown a statistically significant association between interpregnancy interval and anemia. The odds of anemia among pregnant women who had a birth interval of greater than or equal to 2 years were 0.867 times lower than the odds of anemia among pregnant women who had less than 2 years’ birth interval. Appropriate time after each pregnancy for recovery and replenishment of nutrient stores requires 2–5 years. Pregnancy with a short birth interval leads to iron deficiency anemia as iron requirements are substantially higher than the average [25]. Similar finding has also been documented in the studies conducted at Addis Ababa [20], Jimma Hospital [23], and Nigeria [26]. In this particular study, malaria and intestinal parasitic infection, parity, trimester, age, educational status, occupation and income did not show significant association with anemia among pregnant women but found to be significant by other studies [14, 19–22, 27].
Limitation of the study
Because of the limited fund we had, a number of potential confounding factors such as vitamin B12 and/or folate levels, hemoglobinopathies, HIV/AIDS infection, and food frequency, along with a C-reactive protein test for inflammation, were not measured.
Conclusions
The prevalence of anemia among pregnant women in this study was 27.6%. Living in a rural residence, having a history of excess bleeding during the menstrual cycle, ANC follow up and interpregnancy interval were identified as predictors of anemia among the study participants. Therefore, working in the identified gaps could reduce the current burden of anemia among pregnant women in the study area.
Authors’ contributions
ADW and TB conceived the study. WG, TB and ADW designed the study. WG carried out the data collection, statistical analysis and write up of the first draft. ADW and TB revised and shorten the final version of the manuscript. All authors read and approved the final manuscript.
Acknowledgements
We would like to thank our data collectors for their invaluable effort. Our deep gratitude also goes to our study subjects who were volunteered and took their time to give us all the relevant information for the study.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
The datasets developed and/or analyzed during the current study are available from the first author or from the corresponding author on reasonable request.
Ethics approval and consent to participate
Approval for this study was provided by the Jimma University, collage of health sciences institutional review board. Permission to access patients was obtained from the clinical director of the hospital. The participants provided written informed consent to participate in the study by signing or applying a thumb print.
Funding
The research was funded by ENGEN-USAID nutrition project in Jimma University.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- AFS
animal foods sources
- ANC
Antenatal Care Clinic
- EDHS
Ethiopian Demographic Health Survey
- Hb
hemoglobin
- MCV
mean cell volume
- MCH
mean cell hemoglobin
- MCHC
mean cell hemoglobin concentration
- SNNPR
South Nation Nationality and Peoples Region
- SOPs
standard operating procedures
- SPSS
statistical packages for social sciences
- WHO
World Health Organization
Contributor Information
Weinshet Getahun, Email: weingett@gmail.com.
Tefera Belachew, Email: teferabelachew@gmail.com.
Amare Desalegn Wolide, Email: amaju2002@yahoo.com.
References
- 1.Getachew M, Yewhalaw D, Tafess K, Getachew Y, Zeynudin A. Anaemia and associated risk factors among pregnant women in Gilgel Gibe dam area, southwest Ethiopia. Parasites Vectors. 2012;5(1):296. doi: 10.1186/1756-3305-5-296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.WHO. Worldwide prevalence of anemia 1993–2005. WHO global database on anemia. 2008.
- 3.Addis AK, Mohamed DA. Prevalence of anemia and associated factors among pregnant women in an urban area of eastern Ethiopia. Anemia. 2014 doi: 10.1155/2014/561567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Usanga EA, Chilaka M, Archibong EI. Prevalence of iron deficiency anemia in Nigerian pregnant women. J Med Lab Sci. 1994;4:113. [Google Scholar]
- 5.Shulman CE, Graham WJ, Jilo H, Lowe BS, New L, Obiero J, Snow RW, Marsh K. Malaria is an important cause of anemia in primigravidae: evidence from a district hospital in coastal Kenya. Trans R Soc Trop Med Hyg. 1996;90(5):535–539. doi: 10.1016/S0035-9203(96)90312-0. [DOI] [PubMed] [Google Scholar]
- 6.van den Broek NR, Letsky EA. Etiology of anemia in pregnancy in south Malawi. Am J Clin Nutr. 2000;72(Suppl 1):247–256. doi: 10.1093/ajcn/72.1.247S. [DOI] [PubMed] [Google Scholar]
- 7.Marchant TJ. The health of pregnant women in rural Tanzania with specific emphasis on anemia and the impact of socially marketed insecticide treated bednets (Doctoral dissertation, University of Basel). 2002.
- 8.Crawley J. Reducing the burden of anemia in infants and young children in malaria-endemic countries of Africa: from evidence to action. Am J Trop Med Hyg. 2004;712(Suppl 2):25–34. [PubMed] [Google Scholar]
- 9.Haas JD, Brownlie T. Iron deficiency and reduced work capacity: a critical review of the research to determine a causal relationship. J Nutr. 2001;131(Suppl 2):676–690. doi: 10.1093/jn/131.2.676S. [DOI] [PubMed] [Google Scholar]
- 10.Levy A, Fraser D, Katz M, Mazor M, Sheiner E. Maternal anemia during pregnancy is an independent risk factor for low birthweight and preterm delivery. Eur J Obstetrics Gynecol Reprod Biol. 2005;122(2):182–186. doi: 10.1016/j.ejogrb.2005.02.015. [DOI] [PubMed] [Google Scholar]
- 11.Banhidy F, Acs N, Puho EH, Czeizel AE. Iron deficiency anemia: pregnancy outcomes with or without iron supplementation. Nutrition. 2011;27(1):65–72. doi: 10.1016/j.nut.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 12.Kalaivani K. Prevalence & consequences of anemia in pregnancy. Indian J Med Res. 2009;130(5):627–633. [PubMed] [Google Scholar]
- 13.Walter T. Effect of iron-deficiency anemia on cognitive skills and neuromaturation in infancy and childhood. Food Nutr Bull. 2003;24(4 suppl2):104–110. doi: 10.1177/15648265030244S207. [DOI] [PubMed] [Google Scholar]
- 14.Gebre A, Mulugeta A. Prevalence of anemia and associated factors among pregnant women in North Western zone of Tigray, Northern Ethiopia: a cross-sectional study. J Nutr Metab. 2015 doi: 10.1155/2015/165430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kwast BE, Rochat RW, Kidane-Mariam W. Maternal mortality in Addis Ababa, Ethiopia. Stud Fam Plan. 1986;17(6):288–301. doi: 10.2307/1966906. [DOI] [PubMed] [Google Scholar]
- 16.WHO. Hemoglobin concentrations for the diagnosis of anemia and assessment of severity. Vitamin and Mineral Nutrition Information System. 2011.
- 17.Cheesbrough M. District laboratory practice in tropical countries: examination of faecal specimens. Cambridge: Cambridge university press; 2006. pp 96–110.
- 18.Gebremedhin S, Enquselassie F. Correlates of anemia among women of reproductive age in Ethiopia: evidence from Ethiopian DHS 2005. Ethiop J Health Dev. 2011;25(1):22–30. doi: 10.4314/ejhd.v25i1.69842. [DOI] [Google Scholar]
- 19.Kefiyalew F, Zemene E, Asres Y, Gedefaw L. Anemia among pregnant women in Southeast Ethiopia: prevalence, severity and associated risk factors. BMC Res Notes. 2014;7(1):1. doi: 10.1186/1756-0500-7-771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jufar AH, Zewde T. Prevalence of anemia among pregnant women attending antenatal care at tikur anbessa specialized hospital, Addis Ababa Ethiopia. J Hematol Thromboembolic Dis. 2014;2(1):2–6. [Google Scholar]
- 21.Alem M, Enawgaw B, Gelaw A, Kenaw T, Seid M, Olkeba Y. Prevalence of anemia and associated risk factors among pregnant women attending antenatal care in Azezo Health Center Gondar town, Northwest Ethiopia. J Interdiscip Histopathol. 2013;1(3):137–144. doi: 10.5455/jihp.20130122042052. [DOI] [Google Scholar]
- 22.Obse N, Mossie A, Gobena T. Magnitude of anemia and associated risk factors among pregnant women attending antenatal care in Shalla Woreda, West Arsi Zone, Oromia Region, Ethiopia. Ethiop J Health Sci. 2013;23(2):165–173. [PMC free article] [PubMed] [Google Scholar]
- 23.Belachew T, Legesse Y. Risk factors for anemia among pregnant women attending antenatal clinic at Jimma University Hospital, southwest Ethiopia. Ethiop Med J. 2006;44(3):211–220. [PubMed] [Google Scholar]
- 24.Desalegn A, Mossie A, Gedefaw L. Nutritional iron deficiency anemia: magnitude and its predictors among school age children, southwest Ethiopia: a community based cross-sectional study. PLoS ONE. 2014;9(12):e114059. doi: 10.1371/journal.pone.0114059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Girma W, Genebo T. Determinants of nutritional status of women and children in Ethiopia.
- 26.Nwizu EN, Iliyasu Z, Ibrahim SA, Galadanci HS. Socio-demographic and maternal factors in anaemia in pregnancy at booking in Kano, northern Nigeria. Afr J Reprod Health. 2011;15(4):33–41. [PubMed] [Google Scholar]
- 27.Ejeta E, Alemnew B, Fikadu A, Fikadu M, Tesfaye L, Birhanu T, Nekemte E. Prevalence of anaemia in pregnant womens and associated risk factors in Western Ethiopia. Food Sci Qual Manag. 2014;31.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets developed and/or analyzed during the current study are available from the first author or from the corresponding author on reasonable request.


