Abstract
Purpose
Despite the effectiveness of chemoprevention (Tamoxifen and Raloxifene) in preventing breast cancer among women at high risk for the disease, uptake is low. The objective of this study was to determine the tradeoff preferences for various attributes associated with chemoprevention among women not currently taking the drugs.
Methods
We used rating-based conjoint analysis to evaluate the relative importance of a number of attributes associated with chemoprevention, including risk of side effects, drug effectiveness, time needed to take the drugs, and availability of a blood test to see if the drugs were working in an Internet sample of women. We generated mean importance values and part-worth utilities for all attribute levels associated with taking chemoprevention. We then used multivariable linear regression to examine attribute importance scores controlling for participant age, race, Hispanic ethnicity, educational level, and family history of breast cancer.
Results
Overall interest in taking chemoprevention was low among the 1,094 women included in the analytic sample, even for the scenario in which participants would receive the greatest benefit and fewest risks associated with taking the drugs. Time needed to take the pill for it to work and five-year risk of breast cancer were the most important attributes driving tradeoff preferences between the chemoprevention scenarios.
Conclusions
Interest in taking chemoprevention among this sample of women at average risk was low. Addressing women’s concerns about the time needed to take chemoprevention for it to work may help clinicians improve uptake of the drugs among those likely to benefit.
INTRODUCTION
Breast cancer is the second leading cause of cancer-related death among women in the U.S. with over 230,000 new cases and nearly 40,000 deaths predicted in 2015 [1]. Early detection of breast lesions via screening mammography is currently the most widely-used approach to reduce breast cancer-associated mortality. The use of chemoprevention (i.e., Tamoxifen and Raloxifene) to prevent the development of breast cancer among women at high risk (5-year risk of a cancer diagnosis ≥1.66%) [2], has received comparatively less public attention. Clinical practice guidelines in oncology support the use of these drugs for the prevention of breast cancer among women at high risk [3,4]. While it is estimated that more than 2 million U.S. women could benefit from chemoprevention [2], uptake is extremely low [5–10]. A recent study found that less than 1% of women who would potentially benefit from chemoprevention reported taking these drugs [9].
This low uptake of chemoprevention among women at high risk is concerning, as research indicates that use of chemoprevention can reduce a woman’s risk of breast cancer by up to 50% [11,12]. A secondary benefit is a small but significant decreased risk of bone fractures [13]. However, as with all pharmacologic agents, use of chemoprevention carries the potential for side effects. These include an elevated, albeit low, risk of developing endometrial cancer and/or blood clots, and increased chance of experiencing hormonal symptoms, such as hot flashes or night sweats [11,12]. Despite these possible side effects, among most women at high risk of breast cancer, risks associated with chemoprevention are much lower than the potential for benefit [14]. Perception of personal susceptibility to breast cancer has been found to be related to the use of chemoprevention [15–17], yet women have also reported a number of barriers to use of these drugs. These include concerns about side effects, not wanting to take a pill every day, and worry that the benefits are not worth the risks [8,18,19]. Further, to be most effective, chemoprevention needs to be taken daily for at least five years, which may be burdensome. Additionally, research in other areas has demonstrated that lack of an apparent therapeutic response and thus a perceived lack of drug efficacy by patients is associated with early discontinuation of medications [20,21].
Thus, the decision to take chemoprevention is complex, requiring women to evaluate a number of tradeoffs, primarily between the potential for benefit and perceived barriers to use [22]. Some prior research has explored women’s preferences for chemoprevention after being identified as high risk for breast cancer [16]. However, most women are informed that they are at high risk for breast cancer and offered chemoprevention within the same short clinical encounter [23]. Following widespread recent media coverage of Angelina Jolie’s disclosure of her elevated breast cancer risk, interest in risk reduction strategies among average risk women has increased, including interest in being screened for high risk status [24] Given that individuals often maintain strong preferences for or against certain treatments even in the absence of a diagnosis [25], understanding how women feel about chemoprevention prior to being identified as high risk is critical. Yet little is currently known about how average-risk women value the risks and benefits associated with potentially initiating chemoprevention.
We therefore sought to quantify the relative tradeoff preferences for risks and benefits associated with chemoprevention among women at average risk for breast cancer. Results from this study are intended to inform strategies aimed at improving uptake of chemoprevention among women who are likely to benefit.
METHODS
We used established methods originating in mathematical psychology and later used in marketing research for assessing preferences for products or decisions, specifically conjoint analysis [26]. Several types of conjoint analysis exist; these methods differ in what is presented to the participant and in their underlying modeling [27]. However, all are approaches to preference elicitation that require individuals to make tradeoffs between various attributes of a product or decision [28]. Conjoint analysis is increasingly being used in healthcare to better understand preferences for healthcare interventions as well as health states [29].
Largely, this methodology is based on three concepts: 1) each choice is a bundle of attributes (i.e. characteristics or features of the treatment), 2) each attribute consists of a set of levels (different grades of risk and/or benefit of each attribute), and 3) configurations can be developed via different combinations of attribute levels [28]. We chose to use rating-based conjoint for this study.
Rating-based conjoint analysis
In rating-based conjoint analysis, the configurations of attributes and their corresponding levels are shown to participants in the form of a single hypothetical scenario (i.e. different combinations of levels of each attribute) per page or task. Participants are asked to rate each scenario on its own, based on how appealing the shown combination of attributes and levels is to them. Through a mathematically determined number of these exercises, it is then possible to attach statistical valuations to the contribution of each attribute to participant tradeoffs between preferences for different scenarios. While rating-based conjoint analysis is not as widely used in health as other approaches, like discrete choice exercises, prior health-related studies have used this approach to examine women’s tradeoff preferences for pregnancy prevention [30] and fertility care [31].
Rating-based conjoint has strengths that were particularly suited to our study. Specifically, participants view just one scenario at a time. This has the advantage of making the exercise conceptually easier to follow and allows for testing of a greater number of attributes per page or screen, while minimizing respondent burden. Rating-based conjoint analysis is often recommended with the number of attributes and levels in our study design [32].
Attributes and Levels
Attributes and associated levels were generated by the study team guided by two main factors: 1) randomized controlled trial data reporting on likelihood of possible risk and benefits associated with taking chemoprevention, and 2) prior literature on barriers and facilitators to taking preventive medication, generally and chemoprevention, specifically [8,33–35]. The principal investigator for this study (Dr. Fagerlin) has conducted extensive prior research regarding women’s preferences for chemoprevention [8,33,36]. The conduct of this prior research was informed by a rich patient-centered evidence base, including qualitative interviews, focus groups, and pilot testing of patient surveys among women at average and high risk for breast cancer. Selection of attributes and levels for the current study was based on this formative work.
Consistent with best practices, levels of risk and benefit for each attribute were presented as absolute risks or frequencies, rather than relative risks. Research has demonstrated that absolute risks, either through percentages (out of 100%) or numerical (e.g. 3/100) are the most accurately understood by the broadest range of potential study participants [37–39]. Attributes and associated levels are listed in Table 1 and described briefly below.
Table 1.
Attributes and Levels
| Attributes | Levels |
|---|---|
| Five-year risk of developing breast cancer | 6%; 10%; 20% |
| Reduction in risk of breast cancer | 90%; 50% |
| Time needed to take the pill for it to work | 1 year; 5 years; until age 70 |
| Risk of blood clots | No additional risk; 3/100 will experience blood clots |
| Risk of endometrial cancer | No additional risk; 2/100 will get endometrial cancer |
| Risk of hormone symptoms | No additional risk; 10/100 will experience hormone symptoms |
| Reduction in risk of bone fractures | No change; 1/100 women will avoid a fracture |
| Is there a blood test to tell if pill is working | Yes; no |
The first attribute was designed to assess the impact of one’s estimated five-year risk of developing breast cancer. The levels of possible five-year risk of breast cancer were intended to equate actual risks in the average risk population and in higher risk groups: 1) 6%, 2) 10% and 3) 20% five-year risk, and coded in the analysis as a three-level categorical variable. This attribute was included because research suggests that individuals are more inclined to take preventive medications or engage in preventive behavior when they perceive their own personal risk of the disease to be high [40].
We included an attribute to assess the impact of the secondary benefit of chemoprevention, reduction in risk of bone fractures, with dichotomous levels of 1) no change in risk versus 2) an absolute risk reduction of 1/100.
The remaining attributes consisted of established side effects associated with taking chemoprevention, including risks that were low in prevalence but high in severity, as well as common but not severe risks. These were: 1) risk of developing blood clots (levels: no additional risk vs. risk of 3/100), 2) risk of endometrial cancer (levels: no additional risk vs. risk of 2/100), 3) risk of hormone symptoms such as hot flashes and night sweats, among many others (levels: no additional risk vs. risk of 10/100). Risk numbers were reflective of likelihood of risks as reported in trial data [11,12]. Levels for these attributes were coded dichotomously.
Another attribute of interest was length of time a woman would need to take chemoprevention in order for them to be optimally effective in reducing the risk of breast cancer. Studies have shown individuals are more interested in taking medication when the burden of adherence is lowest [40–42], yet studies of chemoprevention effectiveness indicate the pills need to be taken daily for at least five years to be fully effective. Thus we created another attribute, “time needed to take the pill for it to work,” with levels 1) 1 year, 2) 5 years, and 3) until age 70, coded for analysis as a three-level categorical variable.
Finally, because of established low rates of uptake of chemoprevention, we were interested in evaluating the impact of two potentially novel attributes. The first was designed to evaluate the impact of a higher perceived benefit of breast cancer risk reduction than is currently attainable with existing drugs. We thus generated two levels for the risk reduction attribute: 1) a 50% reduction (similar to what can be obtained from taking chemoprevention in women at high risk), and 2) a 90% risk reduction (a large reduction in risk that is not currently attainable).
The second novel attribute was designed to assess the impact of the availability of a blood test that would inform women whether the drugs were working to reduce their cancer risk (such as is the case with a blood test following initiation of statin therapy). Thus we developed at attribute which we called “Is there a blood test to help tell if the pill is working?” with associated levels of 1) yes, and 2) no. Both this attribute and “reduction in risk of breast cancer” (above) were analytically coded as dichotomous variables.
Conjoint analysis design - construction of tasks
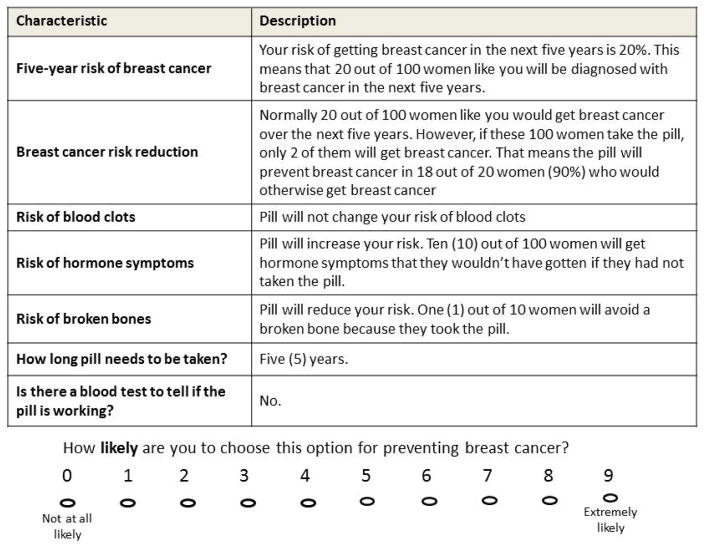
We conducted a traditional full profile rating-based conjoint analysis study [27]. In this computer-based exercise, participants were presented with several hypothetical scenarios including varying levels of each attribute associated with taking chemoprevention. Participants viewed and rated each of the hypothetical scenarios one at a time, while viewing the full set of attributes (and selected levels) [32,43,44]. An example of an individual page from the rating-based conjoint exercise is presented in Figure 1.
Figure 1.
Example of rating-based exercise for preferences for chemoprevention
Additional measures
Certain sociodemograhpic characteristics have been shown to be associated with women’s stated willingness to take chemoprevention [45]. As a result, we included participant age, race, Hispanic ethnicity and educational attainment as control measures. We categorized age as “40 to 54 years” and 55 to 64 years,” race as “white,” “black,” or “other,” and Hispanic ethnicity as “yes’ or “no.” We categorized educational attainment as “high school diploma or less, “some college,” and “four year college graduate or more.” Prior research has shown personal family history of breast cancer to be associated with increased use of some preventive behaviors [46]. As a component of the online survey, participants were asked the number of first degree relatives in their family who had previously been diagnosed with breast cancer. This participant-reported measure was also used to control for participant baseline personal risk of breast cancer (as having a family history increases one’s risk) in the multivariable models. This was categorized as “none,” “one,” or “two or more” and included as a control measure.
Experimental design
We used SAS Market Expo Macro [47] to guide our development of the experimental design. It was determined that 36 scenarios would be the minimal number needed to achieve an efficient, orthogonal (lack of correlation between any two attribute levels) and balanced (equal distribution of each attribute and level in the presented scenarios) design [48]. To reduce respondent burden, a random block design was used to divide the survey into two versions with 18 scenarios each.
An additional 19th “dominant” scenario was included in both versions of the survey. This scenario included all the presumed “best” levels associated with each attribute. These levels included largest breast cancer risk reduction, having a test available to tell if the drug was working, taking the pill for the shortest time, having the highest five-year risk to begin with— as this is the case where taking preventive measures would make most sense — achieving fewer bone fractures, and having no additional risk associated with any of the side effects. We expected participant ratings of the dominant scenario would be higher than ratings for any other scenario.
Sample
Participants were drawn from a panel of female Internet users through Survey Sampling International (SSI). Emails describing the study and providing a link for participation were sent to a stratified random sample of female panel members with the goal of approximating the U.S. Census on race as well as an age range appropriate for taking chemoprevention (40 to 64 years). To ensure at least moderate demographic diversity (but not representativeness) and offset large expected variations in response rates (especially for black and Hispanic participants), we established target response levels roughly matching the prevalence of these racial/ethnic groups in the U.S. population. The number of email invitations in each demographic sub-sample was dynamically adjusted until approximately 12% of completed surveys came from black and Hispanic subgroups.
Participants were deemed ineligible if they reported having previously been personally diagnosed with breast cancer or if they reported ever having taken Tamoxifen or Raloxifene. Eligible individuals were excluded from analysis if their survey completion time was so short (under 200 seconds) that they were unlikely to have seriously considered the survey questions, if they gave the exact same rating on the 0–9 response scale for all 19 scenarios in the survey, or if they did not rate one or more of the 19 scenarios.
Survey administration
In the online survey, participants first viewed an introductory page, which oriented them to the exercise. Participants then completed a number of survey questions related to sociodemographic and health information, including prior use of chemoprevention, prior diagnosis of breast cancer, and family history of breast cancer. Participants were then introduced to the study tasks with language that asked them to imagine they had to consider taking a pill that could prevent breast cancer but had risks associated with it.
Prior to entering the rating exercise, participants were presented with a page including short and plain-language definitions of each of the included attributes and corresponding levels. These definitions were also available to participants on every page of the computer-based conjoint exercise. In the screens that followed, participants were presented with 19 (18 randomized, 1 dominant) different hypothetical scenarios related to taking chemoprevention (one per page), including different combinations of levels of each attribute. While presented with each individual scenario, participants were asked “how likely are you to choose this option for preventing breast cancer?” with a 10-point response scale, ranging from 0 (“not at all likely”) to 9 (“extremely likely.”) Anchors only appeared at the ends of each scale.
Analysis
The statistical approach for this study is based on a main effect fractional factorial design [49]. We first generated descriptive statistics for the analytic sample using Stata Version 13 [50]. We compared individuals in the analytic sample to those excluded (for the reasons outlined above) on sociodemographic information and family history of breast cancer using the chi-squared test. In order to determine participants’ overall willingness to take chemoprevention under the “best case,” we calculated the mean interest in taking chemoprevention for the dominant scenario on the 0 to 9 scale. Using the SAS Market Expo Macro, we then generated unadjusted mean attribute importance values (and standard deviations) for the analytic sample. Importance values reflect the relative contribution of each attribute in driving respondents’ trade-off preferences between scenarios [51].
We then calculated part-worth utilities for each attribute. Part-worth utilities reflect the ranking of levels within each attribute and are calculated to sum to zero. The most positive value represents the most preferred level and the most negative value represents the least preferred level of each attribute. Attributes with the largest range of part-worth utilities are generally considered to be the most important in driving trade-off preferences [51].
In order to examine adjusted mean importance values, we then ran separate multivariable linear regression models for each attribute, controlling for participant age, race, Hispanic ethnicity, educational level, and family history of breast cancer. Participant-reported family history of breast cancer was used to control for individual baseline risk of breast cancer, and participant age was used to control for baseline risk of hormone symptoms. Because we were also interested in understanding whether valuation of the attributes varied by family history of breast cancer, we specifically examined whether coefficients for this variable were statistically significant in adjusted models. This study was not theory-driven, but rather empirical in nature, thus no utility function was specified for the model.
As previously noted, a dominant (best case) scenario was included in the experimental design. As a final step in our study, we conducted a sensitivity analysis comparing adjusted mean attribute importance values between participants who rated the dominant scenario highest versus participants who did not. We did this by including a dummy variable representing whether the participant rated the dominant scenario highest (versus not) in multivariable regression models of attribute importance values, controlling for other covariates. We also explored whether rating the dominant scenario highest was associated with family history of breast cancer using the chi-squared test. All presented tables include the full analytic sample of respondents, not just those who rated the dominant scenario highest.
As anonymous survey research, this study was deemed exempt by the Institutional Review Board of the University of Michigan.
RESULTS
Description of the sample
Of the 1,562 individuals who answered the survey, 70% (1,094) were included in the overall sample. The mean age of sample respondents was 54 years (range: 40–64 years), and 79% were white. A quarter of the sample (26%) reported having completed a four year college degree or higher. The majority of participants (79%) had no first-degree family history of breast cancer; 13% had one first degree relative with breast cancer and 8% had two or more. Relative to those who were excluded, included respondents were younger (p=0.037) had lower levels of education (p<0.001), and a larger proportion were Hispanic (p=0.014). The full description of the sample is presented in Table 2.
Table 2.
Sample Characteristics, comparison of included and excluded participants (x2)
| Included respondents | Excluded respondents | p-value | |
|---|---|---|---|
|
| |||
| N(%) N=1,094 |
N(%) N-468 |
χ2 | |
|
| |||
| Age (years) | |||
| 40–54 | 561(51.3) | 213 (45.5) | 0.037 |
| 55–64 | 533(48.7) | 255 (54.5) | |
|
| |||
| Race | |||
| White | 831(79.1) | 94 (83.9) | 0.487 |
| Black | 161(15.3) | 13(11.6) | |
| Other | 58(5.5) | 5 (4.5) | |
|
| |||
| Hispanic | |||
| No | 917(84.4) | 106(93.0) | 0.014 |
| Yes | 170(15.6) | 8 (7.0) | |
|
| |||
| Education | |||
| High school diploma or less | 316(28.9) | 28(6.0) | <0.001 |
| Some college | 496(45.3) | 61(13.0) | |
| Four year college graduate or more | 282(25.8) | 379(81.0) | |
|
| |||
| Number of first degree relatives with breast cancer | |||
| None | 857(78.8) | 100(79.4) | 0.488 |
| One | 141(13.0) | 19(15.1) | |
| Two or more | 89(8.1) | 7(5.6) | |
Overall interest in taking chemoprevention
Overall interest in taking chemoprevention was low based on responses to the full exercise. The mean unadjusted score for the dominant scenario on the 0–9 scale was 6.0 (Standard Deviation: 2.8).
Part-worth utilities
Part-worth utilities showed that participants preferred to have the lowest five-year risk of breast cancer, only having to take chemoprevention for one year versus longer, achieving a higher versus lower risk reduction of breast cancer, having no additional risk of blood clots, hormone symptoms or endocrine cancer, having a small reduction in the risk of bone fractures versus none, and the availability of a test to determine if the drugs were working. Full part-worth utilities are presented in Table 3.
Table 3.
Dominant scenario
| Mean (SD) | Attribute | Level |
|---|---|---|
| 6.0(2.8) | Five-year risk of breast cancer | 20% five-year risk |
| Time needed to take pill | 1 year | |
| Reduction in risk of breast cancer | 90% | |
| Risk of blood clots | No additional risk | |
| Risk of endometrial cancer | No additional risk | |
| Risk of hormone symptoms | No additional risk | |
| Risk of bone fractures | Reduced risk | |
| Blood test to tell if pill is working | Yes |
Adjusted mean importance values
Results from multivariable linear regression models showed that the amount of time needed to take the pill and five-year risk of breast cancer were the most important attributes driving preferences for different chemoprevention scenarios (mean=16.8; SE=0.66 and mean=16.8; SE=0.61, respectively), after controlling for age, race, Hispanic ethnicity, education, and family history of breast cancer. Reduction in fractures (mean=9.6, SE=0.46) and availability of a blood test to see whether the pills were working (mean=10.3, SE=0.55), in contrast, had the lowest importance values. Table 5 shows adjusted mean attribute importance scores. Family history of breast cancer was only significantly associated with one attribute in the adjusted models: having one family member with breast cancer was associated with small but significantly higher valuation there being a blood test to tell if the chemoprevention was working compared to respondents with no family history.
Table 5.
Adjusted* mean attribute importance scores, overall, and by whether participant rated dominant scenario highest
| Attributes | Attribute importance overall Mean(SE)** |
|---|---|
| Five-year risk of breast cancer | 16.8(0.61) |
| Time needed to take the pill for it to work | 16.8(0.66) |
| Risk of endometrial cancer | 12.4(0.61) |
| Risk of blood clots | 12.1(0.58) |
| Reduction in risk of breast cancer | 11.5(0.54) |
| Risk of hormone symptoms | 10.4(0.46) |
| Is there a blood test to tell if pill is working | 10.3(0.55) |
| Reduction in risk of bone fractures | 9.6(0.46) |
Adjusted for age, race, Hispanic ethnicity, education, and family history of breast cancer
R2 for all models <0.10
Sensitivity analysis
The dominant scenario scored higher than any other scenario, and approximately half (51%) of participants rated the dominant scenario highest of all the scenarios. Compared to respondents who rated the dominant scenario highest, those who did not rate the dominant scenario highest put higher relative valuation on the five-year risk of breast cancer (p<0.001) and reduction in the risk of bone fractures (p=0.014) and lower relative valuation on the risk of blood clots (p<0.001) and the risk of endometrial cancer (p<0.001).
Family history of breast cancer was associated with ratings of the dominant scenario. Respondents with 2 or more family members with a history of breast cancer were significantly less likely to pick the dominant scenario compared to those with no family history (p<0.010) (data not shown).
DISCUSSION
Our results documented low overall interest in taking chemoprevention among study participants, even when women could potentially experience the most benefit and fewest risks, as was the case with the dominant scenario. This finding is consistent with prior research showing that despite understanding the benefits associated with uptake of chemoprevention, interest in taking the drugs is low [6,8].
Prior studies examining women’s interest in taking chemoprevention have approached this question using survey or interview-based methods [6,7,19,45]. Results of these studies have provided valuable information about women’s preferences and feelings regarding the risks and benefits of chemoprevention. Specifically, worry about side effects is generally associated with lower levels of interest in chemoprevention [18,19] and perceived personal susceptibility is associated with greater interest in taking the drugs [15–17].
The use of rating-based conjoint analysis in our study allowed us to examine this topic somewhat differently. Our results reflect women’s tradeoff preferences between the different risks and benefits associated with chemoprevention, which may reflect more “real-world” decision making. Results from our conjoint exercise revealed that the amount of time needed to take chemoprevention and women’s five-year risk of breast cancer were the most important attributes driving women’s tradeoff preferences. Concern about side effects contributed to women’s tradeoff preferences for chemoprevention, but were only moderate drivers of preferences for individual scenarios. The secondary benefit of chemoprevention – the reduction in risk of bone fractures – was the least important attribute driving women’s tradeoff preferences.
Our study is the first to use a conjoint-style methodology to better understand women’s tradeoff preferences related to chemoprevention decision making in the context of primary prevention. In contrast, Wouters and colleagues used adaptive conjoint analysis to understand women’s preferences for chemoprevention as adjuvant therapy to prevent recurrence following a breast cancer diagnosis. They found that efficacy was most important to women when choosing whether to take chemoprevention, followed by side effects and risks of endometrial cancer [52]. Taken together, our results and those of Wouters et al suggest that women may value the relative aspects of chemoprevention differently, depending on whether the drugs are intended to be used for prevention of primary breast cancer, or in reducing the risk of recurrence.
The overall low interest in chemoprevention among women in our study is consistent with findings from research in the domain of cardiovascular prevention. A recent study of U.S. adults found extremely low willingness to take a daily pill for cardiovascular risk prevention, even in cases where the participant would derive the highest level of benefit [25]. As previously noted, prior research has shown that many people find taking daily pill to be burdensome, particularly if the benefits are not readily apparent, as is almost always the case in taking pills for preventive purposes. Further research is needed to determine whether factors other than risk and benefit may play important roles determining individual interest in taking pills to prevent the onset of disease.
Limitations and Strengths
This study should be interpreted in the context of some limitations. In contrast to many other health-related studies employing conjoint-type methodologies, we used a rating-based approach. A major limitation to using rating-based conjoint analysis is that participants may have difficulty placing meaningful numerical ratings on different scenarios. This perhaps explains why a fairly large percentage of participants failed to rate the dominant scenario highest. Other approaches, in particular best-worst scaling [53], would likely prove more informative in understanding women’s preferences for chemoprevention.
An additional limitation of our study was our inclusion of an artificially high (and not currently achievable) level of 90% reduction in risk of breast cancer associated with taking chemoprevention. Our use of an inflated level could have biased participant responses, thereby increasing the importance of the attribute in tradeoff preferences between scenarios. Because of this currently unachievable level, we were unable to test interaction effects between “reduction in risk of breast cancer” and other attributes, as interaction estimates may have been biased. Additionally, because of the large difference between 50% and 90% we were unable to determine if there was an intermediate level of risk reduction at which women’s tradeoff preferences for chemoprevention would change. Our study was further limited by the inclusion of the level “until age 70” for the attribute of “time needed to take pill for it to work.” Unlike the other two levels for this attribute, “until age 70” represented a wide variation in amount of time, dependent on how old the participant was when they completed the conjoint exercise. However, we attempted to account for this through adjusting for participant age in multivariable models.
Our use of a SAS Macro to generate the attribute importance scores further limits our paper, as we are unable to fully describe the computational details of the attribute importance values. Additionally, likely in part because this was not a theory-grounded study, adjusted models did not fit the data well. Because only two levels were used for most quantitative attributes, we were also unable to test for linearity of effects.
Our use of a sample of women at average risk for breast cancer rather than women at high-risk may have resulted in lower participant engagement with the conjoint exercises. However, a number of prior studies in this area have examined women’s preferences for chemoprevention among average-risk women [5,15,17,45]. Because we conducted this study among women not currently eligible for chemoprevention, participants were asked to rate hypothetical scenarios; our ability to infer future behavior from responses to such scenarios is unclear. [54]. Finally, as is a limitation to all conjoint analysis studies, some attributes or levels may have been omitted that could be related to a woman’s decision to take chemoprevention [32].
Despite these limitations, our study provides important patient-centered information about women’s tradeoff preferences for a highly effective yet largely underused pharmacologic agent. To our knowledge, ours is the first study of women’s preferences related to chemoprevention that has used a conjoint-style approach in a population of women at average risk of breast cancer. Importantly, the data from this study allowed us to generate a choice-based conjoint analysis study that is now being fielded with women at high risk for breast cancer who indicated low willingness to take chemoprevention.
Conclusion
Even in the best case scenario, we found that women showed very low interest in taking chemoprevention to prevent breast cancer. Findings from our study suggest women’s preferences for or against chemoprevention are substantially driven by the time they would need to take the pill for it to be most effective. In discussing chemoprevention with patients, clinicians may find it useful to assess patients’ concerns about the time burden of taking chemoprevention, in addition to addressing worries related to side effects. Further exploration of women’s tradeoff preferences for chemoprevention may assist clinicians in improving uptake of the drugs among women who are likely to benefit.
Table 4.
Part worth utilities for attributes and levels related to taking chemoprevention
| Mean | Min–Max | |
|---|---|---|
|
| ||
| Five-year risk of breast cancer | ||
| 20% | −0.13 | −3.5–2.65 |
| 10% | 0 | −3,36–2,78 |
| 6% | 0.13 | −3.1–5.02 |
|
| ||
| Time needed to take pill | ||
| Until 70 years old | −0.25 | −4.25–2.64 |
| 5 Years | 0 | −3–2.54 |
| 1 year | 0.25 | −2.6–6 |
|
| ||
| Reduction in risk by taking pill | ||
| 50% risk reduction | −0.31 | −2.99–1.58 |
| 90% risk reduction | 0.31 | −1.58–2.99 |
|
| ||
| Risk of blood clots | ||
| Small risk (3/100) | −0.41 | −3.37–1.8 |
| No risk | 0.41 | −1.8–3.37 |
|
| ||
| Risk of endometrial cancer | ||
| Small risk (2/100) | −0.32 | −4.5–2.15 |
| No risk | 0.32 | −2.15–4.5 |
|
| ||
| Risk of hormonal side effects | ||
| Some risk (10/100) | −0.23 | −2,78–2.40 |
| No risk | 0.23 | −2.4–2.78 |
|
| ||
| Reduction in bone fractures | ||
| No reduction | −0.18 | −2.46–1.89 |
| Small reduction (1/100) | 0.18 | −1.89–2.46 |
|
| ||
| Test to tell if pill is working | ||
| No | −/0.32 | −2.56–1.67 |
| Yes | 0.32 | −1.67–2.56 |
Key points for decision makers.
Chemoprevention can reduce the risk of breast cancer by up to 50% among women at high risk, yet uptake is extremely low
Women’s tradeoff preferences for chemoprevention are strongly driven by the amount of time they would need to take the pills for them to work as well as their personal susceptibility to breast cancer
The secondary benefit of chemoprevention, a reduction in risk of bone fractures, is less important in driving women’s tradeoff preferences for chemoprevention, compared to risk of side effects
Acknowledgments
This study was funded by the National Institutes for Health (R01 CA87595 and P50 CA101451). Dr. Martinez was supported by a postdoctoral fellowship from the U.S. Department of Veterans Affairs.
Angela Fagerlin, Holly Witteman, Christine Holmberg and Sarah Hawley conceptualized and designed the study, including the online rating-based conjoint exercise. Kathryn Martinez and Holly Witteman conducted data analysis. All authors assisted in interpreting the results. The complete first draft of the paper was written by Kathryn Martinez, with assistance from Angela Fagerlin and Sarah Hawley. All authors commented on the first complete draft as well as subsequent versions. Kathryn Martinez acts as guarantor for the paper.
Footnotes
Compliance with Ethical Standards
Kathryn Martinez, Angela Fagerlin, Holly Witteman, Christine Holmberg, and Sarah Hawley have no conflicts of interest to disclose
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin [Internet] 2015 Jan 5; doi: 10.3322/caac.21254. [cited 2015 Jan 9]; Available from: http://www.ncbi.nlm.nih.gov/pubmed/25559415. [DOI] [PubMed]
- 2.Freedman AN, Graubard BI, Rao SR, McCaskill-Stevens W, Ballard-Barbash R, Gail MH. Estimates of the number of US women who could benefit from tamoxifen for breast cancer chemoprevention. J Natl Cancer Inst [Internet] 2003;95(7):526–32. doi: 10.1093/jnci/95.7.526. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12671020. [DOI] [PubMed] [Google Scholar]
- 3.Bevers TB, Armstrong DK, Arun B, Carlson RW, Cowan KH, Daly MB, et al. Breast cancer risk reduction. J Natl Compr Canc Netw [Internet] 2010 Oct;8(10):1112–46. doi: 10.6004/jnccn.2010.0083. [cited 2013 Nov 19] Available from: http://www.ncbi.nlm.nih.gov/pubmed/20971838. [DOI] [PubMed] [Google Scholar]
- 4.Chlebowski RT, Col N, Winer EP, Collyar DE, Cummings SR, Vogel VG, et al. American Society of Clinical Oncology technology assessment of pharmacologic interventions for breast cancer risk reduction including tamoxifen, raloxifene, and aromatase inhibition. J Clin Oncol [Internet] 2002 Aug 1;20(15):3328–43. doi: 10.1200/JCO.2002.06.029. [cited 2014 Mar 14] Available from: http://www.ncbi.nlm.nih.gov/pubmed/12149307. [DOI] [PubMed] [Google Scholar]
- 5.Kaplan CP, Haas JS, Pérez-Stable EJ, Gregorich SE, Somkin C, Des Jarlais G, et al. Breast cancer risk reduction options: awareness, discussion, and use among women from four ethnic groups. Cancer Epidemiol biomarkers Prev a Publ Am Assoc Cancer Res cosponsored by Am Soc Prev Oncol [Internet] 2006;15(1):162–6. doi: 10.1158/1055-9965.EPI-04-0758. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16434605. [DOI] [PubMed] [Google Scholar]
- 6.Ropka ME, Keim J, Philbrick JT. Patient decisions about breast cancer chemoprevention: a systematic review and meta-analysis. J Clin Oncol [Internet] 2010 Jun 20;28(18):3090–5. doi: 10.1200/JCO.2009.27.8077. [cited 2013 Nov 19] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2903338&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Melnikow J, Paterniti D, Azari R, Kuenneth C, Birch S, Kuppermann M, et al. Preferences of Women Evaluating Risks of Tamoxifen (POWER) study of preferences for tamoxifen for breast cancer risk reduction. Cancer [Internet] 2005 May 15;103(10):1996–2005. doi: 10.1002/cncr.20981. [cited 2013 Nov 19] Available from: http://www.ncbi.nlm.nih.gov/pubmed/15825209. [DOI] [PubMed] [Google Scholar]
- 8.Fagerlin A, Zikmund-Fisher BJ, Nair V, Derry HA, McClure JB, Greene S, et al. Women’s decisions regarding tamoxifen for breast cancer prevention: responses to a tailored decision aid. Breast Cancer Res Treat [Internet] 2010 Feb;119(3):613–20. doi: 10.1007/s10549-009-0618-4. [cited 2013 Nov 19] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3736824&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Waters EA, McNeel TS, Stevens WM, Freedman AN. Use of tamoxifen and raloxifene for breast cancer chemoprevention in 2010. Breast Cancer Res Treat [Internet] 2012 Jul;134(2):875–80. doi: 10.1007/s10549-012-2089-2. [cited 2013 Nov 19] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3771085&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Armstrong K, Quistberg DA, Micco E, Domchek S, Guerra C. Prescription of tamoxifen for breast cancer prevention by primary care physicians. Arch Intern Med [Internet] 2006;166(20):2260–5. doi: 10.1001/archinte.166.20.2260. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17101945. [DOI] [PubMed] [Google Scholar]
- 11.Vogel VG, Costantino JP, Wickerham DL, Cronin WM, Cecchini RS, Atkins JN, et al. Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 Trial: Preventing breast cancer. Cancer Prev Res (Phila) [Internet] 2010 Jun;3(6):696–706. doi: 10.1158/1940-6207.CAPR-10-0076. [cited 2013 Nov 19] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2935331&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fisher B, Costantino JP, Wickerham DL, Redmond CK, Kavanah M, Cronin WM, et al. Tamoxifen for prevention of breast cancer: report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. J Natl Cancer Inst [Internet] 1998 Sep 16;90(18):1371–88. doi: 10.1093/jnci/90.18.1371. [cited 2013 Nov 19] Available from: http://www.ncbi.nlm.nih.gov/pubmed/9747868. [DOI] [PubMed] [Google Scholar]
- 13.Cooke AL, Metge C, Lix L, Prior HJ, Leslie WD. Tamoxifen use and osteoporotic fracture risk: a population-based analysis. J Clin Oncol [Internet] 2008 Nov 10;26(32):5227–32. doi: 10.1200/JCO.2007.15.7123. [cited 2013 Nov 19] Available from: http://www.ncbi.nlm.nih.gov/pubmed/18838712. [DOI] [PubMed] [Google Scholar]
- 14.Nichols HB, DeRoo LA, Scharf DR, Sandler DP. Risk-benefit profiles of women using tamoxifen for chemoprevention. J Natl Cancer Inst [Internet] 2015 Jan;107(1):354. doi: 10.1093/jnci/dju354. [cited 2015 Jan 20] Available from: http://www.ncbi.nlm.nih.gov/pubmed/25475563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bastian LA, Lipkus IM, Kuchibhatla MN, Weng HH, Halabi S, Ryan PD, et al. Women’s interest in chemoprevention for breast cancer. Arch Intern Med. 2001;161(13):1639–44. doi: 10.1001/archinte.161.13.1639. [DOI] [PubMed] [Google Scholar]
- 16.Bober SL, Hoke LA, Duda RB, Regan MM, Tung NM. Decision-making about tamoxifen in women at high risk for breast cancer: clinical and psychological factors. J Clin Oncol. 2004;22(24):4951–7. doi: 10.1200/JCO.2004.05.192. [DOI] [PubMed] [Google Scholar]
- 17.Fasching PA, Von Minckwitz G, Fischer T, Kaufmann M, Schultz-Zehden B, Beck H, et al. The impact of breast cancer awareness and socioeconomic status on willingness to receive breast cancer prevention drugs. Breast Cancer Res Treat [Internet] 2007;101(1):95–104. doi: 10.1007/s10549-006-9272-2. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17033928. [DOI] [PubMed] [Google Scholar]
- 18.Port ER, Montgomery LL, Heerdt AS, Borgen PI. Patient reluctance toward tamoxifen use for breast cancer primary prevention. Ann Surg Oncol [Internet] 2001 Aug;8(7):580–5. doi: 10.1007/s10434-001-0580-9. [cited 2013 Nov 19] Available from: http://www.ncbi.nlm.nih.gov/pubmed/11508619. [DOI] [PubMed] [Google Scholar]
- 19.Heisey R, Pimlott N, Clemons M, Cummings S, Drummond N. Women’s views on chemoprevention of breast cancer: qualitative study. Can Fam Physician [Internet] 2006 May;52:624–5. [cited 2014 Nov 12] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1531726&tool=pmcentrez&rendertype=abstract. [PMC free article] [PubMed] [Google Scholar]
- 20.Wei MY, Ito MK, Cohen JD, Brinton EA, Jacobson TA. Predictors of statin adherence, switching, and discontinuation in the USAGE survey: understanding the use of statins in America and gaps in patient education. J Clin Lipidol [Internet] Jan;7(5):472–83. doi: 10.1016/j.jacl.2013.03.001. [cited 2015 Jan 20] Available from: http://www.ncbi.nlm.nih.gov/pubmed/24079289. [DOI] [PubMed] [Google Scholar]
- 21.Nantz E, Liu-Seifert H, Skljarevski V. Predictors of premature discontinuation of treatment in multiple disease states. Patient Prefer Adherence [Internet] 2009 Jan;3:31–43. doi: 10.2147/ppa.s4633. [cited 2015 Jan 20] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2778422&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mulley AG, Sepucha K. Making good decisions about breast cancer chemoprevention. Ann Intern Med [Internet] 2002 Jul 2;137(1):52–4. doi: 10.7326/0003-4819-137-1-200207020-00014. [cited 2014 Mar 14] Available from: http://www.ncbi.nlm.nih.gov/pubmed/12093247. [DOI] [PubMed] [Google Scholar]
- 23.Owens WL, Gallagher TJ, Kincheloe MJ, Ruetten VL. Implementation in a large health system of a program to identify women at high risk for breast cancer. J Oncol Pract [Internet] 2011 Mar;7(2):85–8. doi: 10.1200/JOP.2010.000107. [cited 2015 Feb 12] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3051867&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Evans DG, Barwell J, Eccles DM, Collins A, Izatt L, Jacobs C, et al. The Angelina Jolie effect: how high celebrity profile can have a major impact on provision of cancer related services. Breast Cancer Res [Internet] 2014 Jan;16(5):442. doi: 10.1186/s13058-014-0442-6. [cited 2015 Apr 21] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4303122&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hutchins R, Viera AJ, Sheridan SL, Pignone MP. Quantifying the Utility of Taking Pills for Cardiovascular Prevention. Circ Cardiovasc Qual Outcomes [Internet] 2015 Feb 3; doi: 10.1161/CIRCOUTCOMES.114.001240. [cited 2015 Feb 9] Available from: http://www.ncbi.nlm.nih.gov/pubmed/25648463. [DOI] [PubMed]
- 26.Luce RD, Tukey JW. Simultaneous conjoint measurement: A new type of fundamental measurement. J Math Psychol. 1964;1:1–27. [Google Scholar]
- 27.Ryan M, Scott D, Reeves C, Bate A, van Teijlingen E, Russell E, et al. Eliciting public preferences for healthcare: a systematic review of techniques. Health Technol Assess (Rockv) 2001;5(5) doi: 10.3310/hta5050. [DOI] [PubMed] [Google Scholar]
- 28.Curry J. Understanding Conjoint Analysis in 15 Minutes. Sequim, WA: 1996. [Google Scholar]
- 29.Bridges JFP, Hauber AB, Marshall D, Lloyd A, Prosser LA, Regier DA, et al. Conjoint analysis applications in health--a checklist: a report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value Health [Internet] 2011 Jun;14(4):403–13. doi: 10.1016/j.jval.2010.11.013. [cited 2014 Jul 10] Available from: http://www.ncbi.nlm.nih.gov/pubmed/21669364. [DOI] [PubMed] [Google Scholar]
- 30.Tanner AE, Katzenstein JM, Zimet GD, Cox DS, Cox AD, Fortenberry JD. Vaginal microbicide preferences among midwestern urban adolescent women. J Adolesc Health [Internet] 2008 Oct;43(4):349–56. doi: 10.1016/j.jadohealth.2008.02.017. [cited 2015 Feb 12] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2593640&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cai QF, Wan F, Dong XY, Liao XH, Zheng J, Wang R, et al. Fertility clinicians and infertile patients in China have different preferences in fertility care. Hum Reprod [Internet] 2014 Apr;29(4):712–9. doi: 10.1093/humrep/deu023. [cited 2015 Feb 12] Available from: http://www.ncbi.nlm.nih.gov/pubmed/24549214. [DOI] [PubMed] [Google Scholar]
- 32.Orme B. Formulating Attributes and Levels in Conjoint Analysis. Sawtooth Software Ltd; 2002. [Google Scholar]
- 33.Fagerlin A, Dillard AJ, Smith DM, Zikmund-Fisher BJ, Pitsch R, McClure JB, et al. Women’s interest in taking tamoxifen and raloxifene for breast cancer prevention: response to a tailored decision aid. Breast Cancer Res Treat [Internet] 2011 Jun;127(3):681–8. doi: 10.1007/s10549-011-1450-1. [cited 2013 Nov 19] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3742062&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wouters H, Van Dijk L, Van Geffen ECG, Gardarsdottir H, Stiggelbout AM, Bouvy ML. Primary-care patients’ trade-off preferences with regard to antidepressants. Psychol Med [Internet] 2014 Jan 7;:1–8. doi: 10.1017/S0033291713003103. [cited 2014 Mar 14] Available from: http://www.ncbi.nlm.nih.gov/pubmed/24398071. [DOI] [PubMed]
- 35.Ryan M. Using conjoint analysis to take account of patient preferences and go beyond health outcomes: an application to in vitro fertilisation. Soc Sci Med [Internet] 1999 Feb;48(4):535–46. doi: 10.1016/s0277-9536(98)00374-8. [cited 2015 Mar 26] Available from: http://www.ncbi.nlm.nih.gov/pubmed/10075178. [DOI] [PubMed] [Google Scholar]
- 36.Dillard AJ, Scherer L, Ubel PA, Smith DM, Zikmund-Fisher BJ, McClure JB, et al. Breast cancer anxiety’s associations with responses to a chemoprevention decision aid. Soc Sci Med [Internet] 2013 Jan;77:13–9. doi: 10.1016/j.socscimed.2012.10.009. [cited 2015 Mar 31] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3760790&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sedrakyan A, Shih C. Improving depiction of benefits and harms: analyses of studies of well-known therapeutics and review of high-impact medical journals. Med Care [Internet] 2007 Oct;45(10 Supl 2):S23–8. doi: 10.1097/MLR.0b013e3180642f69. [cited 2015 Feb 9] Available from: http://www.ncbi.nlm.nih.gov/pubmed/17909378. [DOI] [PubMed] [Google Scholar]
- 38.Griffith JM, Lewis CL, Hawley S, Sheridan SL, Pignone MP. Randomized trial of presenting absolute v. relative risk reduction in the elicitation of patient values for heart disease prevention with conjoint analysis. Med Decis Making [Internet] Jan;29(2):167–74. doi: 10.1177/0272989X08327492. [cited 2014 Sep 8] Available from: http://www.ncbi.nlm.nih.gov/pubmed/19279298. [DOI] [PubMed] [Google Scholar]
- 39.Trevena LJ, Zikmund-Fisher BJ, Edwards A, Gaissmaier W, Galesic M, Han PKJ, et al. Presenting quantitative information about decision outcomes: a risk communication primer for patient decision aid developers. BMC Med Inform Decis Mak [Internet] 2013 Jan;13(Suppl 2):S7. doi: 10.1186/1472-6947-13-S2-S7. [cited 2015 Jan 26] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4045391&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mann DM, Allegrante JP, Natarajan S, Halm EA, Charlson M. Predictors of adherence to statins for primary prevention. Cardiovasc Drugs Ther [Internet] 2007 Aug;21(4):311–6. doi: 10.1007/s10557-007-6040-4. [cited 2014 Dec 2] Available from: http://www.ncbi.nlm.nih.gov/pubmed/17665294. [DOI] [PubMed] [Google Scholar]
- 41.Claxton AJ, Cramer J, Pierce C. A systematic review of the associations between dose regimens and medication compliance. Clin Ther [Internet] 2001 Aug;23(8):1296–310. doi: 10.1016/s0149-2918(01)80109-0. [cited 2014 Mar 14] Available from: http://www.ncbi.nlm.nih.gov/pubmed/11558866. [DOI] [PubMed] [Google Scholar]
- 42.Reginster JY, Rabenda V, Neuprez A. Adherence, patient preference and dosing frequency: understanding the relationship. Bone [Internet] 2006 Apr;38(4 Suppl 1):S2–6. doi: 10.1016/j.bone.2006.01.150. [cited 2014 Mar 5] Available from: http://www.sciencedirect.com/science/article/pii/S8756328206002237. [DOI] [PubMed] [Google Scholar]
- 43.Orme BK, Alpert MICE. Assessing the Validity of Conjoint Analysis - Continued. Sawtooth Software Ltd; 1997. [Google Scholar]
- 44.Louviere J. Stated Choice Methods: Analysis and Application. Cambridge: Cambridge University Press; 2000. [Google Scholar]
- 45.Kaplan CP, Kim SE, Wong ST, Sawaya GF, Walsh JME, Pérez-Stable EJ. Willingness to use tamoxifen to prevent breast cancer among diverse women. Breast Cancer Res Treat [Internet] 2012 May;133(1):357–66. doi: 10.1007/s10549-012-1960-5. [cited 2015 Jan 15] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4039196&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Campitelli MA, Chiarelli AM, Mirea L, Stewart L, Glendon G, Ritvo P, et al. Adherence to breast and ovarian cancer screening recommendations for female relatives from the Ontario site of the Breast Cancer Family Registry. Eur J Cancer Prev [Internet] 2011 Nov;20(6):492–500. doi: 10.1097/CEJ.0b013e3283476217. [cited 2014 Mar 14] Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3179806&tool=pmcentrez&rendertype=abstract. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Market Expo Macros. Cary, North Carolina, USA: [Google Scholar]
- 48.Chrzan K, Orme B. An Overview and Comparison of Design Stratgies for Choice-Based Conjoint Analysis. Sequim, WA: 2000. [Google Scholar]
- 49.Nair V, Strecher V, Fagerlin A, Ubel P, Resnicow K, Murphy S, et al. Screening experiments and the use of fractional factorial designs in behavioral intervention research. Am J Public Heal. 2008 Aug;98(8):1354–9. doi: 10.2105/AJPH.2007.127563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.StataCorp. Stata Statistical Software: Release 13. Vol. 2013 College Station, TX: StataCorp LP; 2013. [Google Scholar]
- 51.Orme B. Software Research Paper Series 2002. Sequin, WA: 2002. Interpreting conjoint analysis data. [Google Scholar]
- 52.Wouters H, Maatman GA, Van Dijk L, Bouvy ML, Vree R, Van Geffen ECG, et al. Trade-off preferences regarding adjuvant endocrine therapy among women with estrogen receptor-positive breast cancer. Ann Oncol [Internet] 2013 Sep;24(9):2324–9. doi: 10.1093/annonc/mdt195. [cited 2013 Nov 19] Available from: http://www.ncbi.nlm.nih.gov/pubmed/23709173. [DOI] [PubMed] [Google Scholar]
- 53.Flynn TN. Valuing citizen and patient preferences in health: recent developments in three types of best-worst scaling. Expert Rev Pharmacoecon Outcomes Res [Internet] 2010 Jun;10(3):259–67. doi: 10.1586/erp.10.29. [cited 2015 Mar 30] Available from: http://www.ncbi.nlm.nih.gov/pubmed/20545591. [DOI] [PubMed] [Google Scholar]
- 54.Persky S, Kaphingst KA, Condit CM, McBride CM. Assessing hypothetical scenario methodology in genetic susceptibility testing analog studies: a quantitative review. Genet Med [Internet] 2007 Nov;9(11):727–38. doi: 10.1097/gim.0b013e318159a344. [cited 2014 Mar 14] Available from: http://www.ncbi.nlm.nih.gov/pubmed/18007141. [DOI] [PMC free article] [PubMed] [Google Scholar]