Abstract
Peripheral myelin protein 22 (PMP22) is associated with a subset of hereditary peripheral neuropathies. Although predominantly recognized as a transmembrane constituent of peripheral nerve myelin, PMP22 is localized to epithelial and endothelial cell-cell junctions, where its function remains unknown. In this report, we investigated the role of PMP22 in epithelial biology. Expression of human PMP22 (hPMP22) slows cell growth and induces a flattened morphology in Madin-Darby canine kidney (MDCK) cells. The transepithelial electrical resistance (TER) and paracellular flux of MDCK monolayers are elevated by hPMP22 expression. After calcium switch, peptides corresponding to the second, but not the first, extracellular loop of PMP22 perturb the recovery of TER and paracellular flux. Finally, subsequent to wounding, epithelial monolayers expressing hPMP22 fail to migrate normally. These results indicate that PMP22 is capable of modulating several aspects of epithelial cell biology, including junctional permeability and wound closure.
INTRODUCTION
The tetraspan glycoprotein peripheral myelin protein 22 (PMP22), also known as growth arrest specific 3 (gas3) gene, has proposed roles in peripheral nerve myelin formation, cell–cell interactions, and cell proliferation (Suter and Snipes, 1995). Although the highest expression levels are found in myelin-forming Schwann cells, PMP22 mRNA can be detected in a multitude of developing and mature non-neural tissues, including epithelia of the intestine (Baechner et al., 1995; Taylor et al., 1995; Wulf and Suter, 1999) and the choroid plexus (Roux et al., 2004). The specific role of PMP22 in Schwann cells remains undefined, although it is known that altered expression is associated with heritable demyelinating peripheral neuropathies (Naef and Suter, 1998). Similarly, the function of the protein at nonneural locations remains undetermined.
In vitro studies have identified a role for PMP22 in the regulation of cell proliferation and morphology. In Schwann cells, elevated expression delays the transition from G0/G1 to the S phase of the cell cycle (Zoidl et al., 1995), and can lead to apoptosis in some instances (Fabbretti et al., 1995; Zoidl et al., 1997). Conversely, reduced PMP22 mRNA levels are associated with enhanced DNA synthesis and entry into the S + G2/M phases (Zoidl et al., 1995). In NIH 3T3 fibroblasts, PMP22 overexpression regulates cell spreading, an effect that is dependent on the Rho–GTPase pathway (Brancolini et al., 1999). Recent studies have detected exogenous PMP22 in ADP-ribosylation factor 6 (Arf-6)-positive plasma membrane-endosomal recycling vacuoles before apoptosis or changes in cell shape (Chies et al., 2003). This pathway is known to be involved in modulating the actin cytoskeleton, cell polarity, adhesion, and migration (Donaldson, 2003). Together, these findings support the notion that PMP22 has a significant role in basic cellular processes, extending beyond an involvement in Schwann cell myelination.
We previously described PMP22 as a constituent of apical intercellular junctions in epithelial and endothelial cells (Notterpek et al., 2001; Roux et al., 2004). Although PMP22 shares significant amino acid homology with members of the claudin superfamily, overexpression of the protein in L-fibroblasts (Notterpek et al., 2001) or C6 glioma cells (Takeda et al., 2001) did not induce tight junction strands. Nonetheless, PMP22 might function in the establishment and maintenance of ion-selective paracellular barriers. Transmembrane proteins of the apical junctional complex such as the claudins, occludin, and the junctional adhesion molecules (González-Mariscal et al., 2003) all participate in the regulation of junctional permeability. In addition, based on the findings of Brancolini and colleagues (Brancolini et al., 1999, 2000; Chies et al., 2003), PMP22 might be involved in the regulation of epithelial proliferation and/or cell migration, dynamic processes that involve changes in cell adhesion and morphology. In support of this possibility, PMP22 contains the carbohydrate L2/HNK1 adhesion/recognition epitope in the first extracellular loop (Snipes et al., 1993; Schachner et al., 1995).
Here, we examined the role of PMP22 in several facets of epithelial cell biology, including proliferation, cell shape, and migration. An elevated level of PMP22 slows the migration of epithelia and reduces the formation of lamellipodial protrusions. The transepithelial electrical resistance (TER) and paracellular flux also are affected in these cultures. Application of a PMP22 peptide perturbs the TER and permeability of Madin-Darby canine kidney (MDCK) monolayers. Together, these results indicate that PMP22 plays a role in modulating growth, morphology, migration, and paracellular permeability in epithelial cells.
MATERIALS AND METHODS
Cell Culture
MDCK type I (high-resistance) and type II (low-resistance) cells were grown in Eagle's minimum essential medium supplemented with 5% fetal calf serum (FCS) or DMEM with 10% FCS, respectively. Cells were cultured on Costar Transwell filters (0.4-μm pores) (Corning, Acton, MA) or on tissue culture dishes and maintained at 37°C and 5% CO2. For TER or paracellular flux studies (see below), MDCK II monolayers were grown on filters (3 × 105 cells/cm2) in low Ca2+ media (see below) for 48 h to ensure confluence before Ca2+ addition. TER measurements were recorded every 24 h after Ca2+ addition until 6 d postplating, at which time TER levels had reached a steady state. After plating MDCK I cells on filters (3 × 105 cells/cm2), the medium was replaced every 24 h until the sixth day when TER levels had reached a steady state. For the Ca2+-switch assay (Gumbiner and Simons, 1986), cells were treated with EDTA (4 mM) containing media for 4 h (Notterpek et al., 2001) or for 18 h in Ca2+- and Mg2+-free media with 5% Chelex (Sigma-Aldrich, St. Louis, MO)-treated FCS.
Establishment of Stable Transgene-expressing Cells
The human PMP22 (kind gift of Dr. Clare Huxley, Imperial College School of Science, Technology, and Medicine, London, United Kingdom) and human occludin (Van Itallie and Anderson, 1997) open reading frames were inserted into the pLNCX-II retroviral vector (BD Biosciences Clontech, Palo Alto, CA), under the control of the cytomegalovirus (CMV) promoter. After transient transfection with LipofectAMINE Plus reagent (Invitrogen, Carlsbad, CA), MDCK cells were treated with 1.1 mg/ml Geneticin (G418 sulfate) (Invitrogen) for 4 wk. Subclones of the stably expressing MDCK II cells were monitored for transgene expression by immunoblotting as described below. Three subclones were analyzed for each construct. Where indicated, 2.5 mM sodium butyrate was added for 20 h to enhance transgene expression under the CMV promoter (Gorman et al., 1983).
Primary Antibodies
Monoclonal mouse antibodies to occludin, ZO-1 (Zymed Laboratories, South San Francisco, CA), β-catenin (BD Transduction Laboratories, Lexington, KY), E-cadherin clone rrl (Developmental Studies Hybridoma Bank, Iowa City, IA), α-tubulin, actin (Sigma-Aldrich), GP-135 (kind gift from Dr. George Ojakian, SUNY Downstate Medical Center, Brooklyn, NY), rat anti-E-cadherin (Zymed Laboratories), and rabbit polyclonal anti-occludin (Zymed Laboratories), anti-PMP22, and affinity-purified anti-PMP22 antibodies (Notterpek et al., 2001; Roux et al., 2004) were used.
Immunofluorescence Microscopy
MDCK cells plated on glass coverslips or 12-mm Transwells were grown to confluence and fixed with either 3% paraformaldehyde (PFA) followed by a 1-min incubation in 100% acetone at -20°C (for PMP22 detection) or 1% PFA followed by permeabilization with 0.2% Triton X-100 (TX-100) (junction staining). For z-plane imaging of confluent monolayers, filters were fixed with 4% PFA followed by 5-min incubation in 100% methanol at -20°C and then frozen in liquid nitrogen-cooled N-methylbutane before cryosectioning filters along the z-plane (Notterpek et al., 2001). After blocking and incubation with primary antibodies, Alexa anti-mouse IgG fluorescein isothiocyanate (FITC)- and anti-rabbit IgG Texas Red-conjugated antibodies were added (Molecular Probes, Eugene, OR). Nuclei were stained with Hoechst dye (10 μg/ml) (Molecular Probes). Actin filaments were visualized with FITC-conjugated phalloidin (Molecular Probes). Coverslips were mounted by using the Pro-Long Antifade kit (Molecular Probes). Samples were imaged with a Spot camera attached to a Nikon Eclipse 800 microscope and formatted for printing by using Adobe Photoshop 5.0. Images were measured using Spot Advanced 3.5.
Cell Cycle Proliferation Studies
The DNA synthesis rate of subconfluent MDCK II cells plated on glass coverslips was analyzed using a bromodeoxyuridine (BrdU) labeling and detection kit (Roche Diagnostics, Indianapolis, IN). The percentage of BrdU-positive cells was determined by counting in four random fields (0.8 mm2) and comparing with the total number of nuclei (Hoechst). More than 500 cells were counted for each condition. The percentage was calculated from the ratio between BrdU-positive and total cells. Cell cycle analysis was performed on exponentially growing MDCK cells by using the CycleTEST PLUS kit (BD Biosciences, San Jose, CA). The data were collected by fluorescence-activated cell sorter (FACScan; BD Biosciences) and analyzed using Modfit version 2.0.
Epithelial Cyst Formation
To generate MDCK II cysts, 5 × 104 cells/ml were suspended in 2 mg/ml rat tail collagen type I (Sigma-Aldrich) on 6.5-mm Transwells (0.4 μm) (Pollack et al., 1998). After the collagen gel solidified at 37°C, medium was added to the apical and basal chambers and was replaced every 48 h. Eight days after plating, the collagen gels were fixed in 4% PFA before freezing in liquid nitrogen-cooled N-methylbutane. Cryosections (7 μm) were processed for immunolabeling as described above.
Cell Surface Biotinylation and Western Blotting
For Western blotting, monolayers of confluent MDCK cells were lysed (Notterpek et al., 2001), and where indicated, treated with N-glycosidase (PNGase F) (Pareek et al., 1997). Samples were separated by SDS-PAGE and transferred to nitrocellulose membrane (Bio-Rad, Hercules, CA) before immunoblotting. Bound horseradish peroxidase-conjugated anti-rabbit or anti-mouse secondary antibodies (Sigma-Aldrich) were detected using ECL reagents (PerkinElmer Life and Analytical Sciences, Boston, MA).
For the detection of PMP22 at the cell surface, confluent monolayers of stably expressing MDCK II cells (6-cm dish) were biotinylated with biocytin hydrazide (Lisanti et al., 1989; Prince et al., 1993). Biotinylated monolayers were lysed for affinity precipitation in NP-40 buffer (25 mM HEPES/NaOH, pH 7.4, 150 mM NaCl, 4 mM EDTA, 25 mM, NaF, 1% NP-40, 1 mM Na3VO4, 1× Complete protease inhibitor [Roche Diagnostics]) as reported previously (Sakakibara et al., 1997). Streptavadin beads (Pierce Chemical, Rockford, IL) suspended in lysis buffer, were added to the cell lysate and gently rocked for 2 h at 4°C. Streptavadin beads were washed four times with NP-40 buffer, boiled in SDS gel sample buffer (Notterpek et al., 1997), and treated with PNGase F. Samples were processed for immunoblotting with anti-PMP22 antibodies.
Measurements of Junctional Permeability
TER was measured in 37°C culture media by using an EVOM epithelial voltohmmeter with an STX-2 electrode (WPI, Sarasota, FL). The TER values were calculated by subtracting the background TER of blank filters and normalized by the area of the monolayer. Steady-state TER measurements (n = 9 wells/construct) were detected 6 d after cell plating under the described culture conditions. To measure nonionic paracellular flux, FITC-dextran of 3 and 40 kDa (Molecular Probes) was dissolved in P-buffer (Balda et al., 1996) at a concentration of 10 mg/ml. Apical and basolateral compartments of cells cultured on Transwell filters (n = 3–4 wells/construct) were rinsed with P-buffer and allowed to equilibrate for 10 min. The FITC-dextran stock solution (25 and 50 μg/100 μl, 3 and 40 kDa, respectively) was added to the apical chamber, and the cells were incubated at 37°C for 30 min. By sampling the basal media, the amount of FITC-dextran diffusion from the apical to the basal chamber was measured in a VersaFluor fluorometer (Bio-Rad). A standard curve was used to convert relative fluorescent units to the concentration of dextran in solution.
Peptide Perturbation
High-performance liquid chromatography-purified peptides, corresponding to a portion of the first (aa 45–63) (NH2-SALGAVQHCYSSSVSEWLQ-COOH) (PMP22-1st) and the entire second loop (aa 117–132) (NH2-YTVRHE-WHVNTDYSY-COOH) (PMP22-2nd) of murine PMP22 were chosen (United Biochemical Research, Seattle, WA). A scrambled peptide using the same amino acids as the second loop of PMP22 (NH2-HDEYVSNTHWYRSYTV-COOH) (scrambled-2nd) served as a control. A 44-aa peptide corresponding to the second extracellular loop of the chicken occludin (Occ-2nd) was used as a positive control (Wong and Gumbiner, 1997). Peptides were dissolved in dimethyl sulfoxide (DMSO) as 10 mM stocks and added to Ca2+-containing media at the indicated concentrations. Monolayers (n = 3–4 wells/condition) were fed every 24 h with fresh peptide-containing medium.
Wound Migration Studies
Wound assays using MDCK cells have been reported previously (Fenteany et al., 2000; Sabo et al., 2001). Highly confluent MDCK II monolayers on either glass coverslips or tissue culture plastic were wounded with a 200-μl pipette tip (Sabo et al., 2001). Long scratches and short wounds were made before rinsing the monolayer in fresh medium to remove detached cells. At the indicated time points after wounding, wound areas were imaged with a Nikon DS camera attached to a Nikon Eclipse TS100 inverted microscope. To ensure that identical areas were imaged between time points, multiple positioning marks were made at the center of the denuded surface with a small needle. Relative wound areas (n = 3/construct) were measured with the NIH Image analysis program. Alternatively, the mean distance migrated along the wound edge (n = 6, measurements in two separate fields) was determined using Adobe Photoshop 5.0. The Rho-kinase inhibitor Y-27632 (10 μM) (Calbiochem, San Diego, CA) was applied to MDCK monolayers 2 h after wounding, and monolayers were fixed for staining after an additional 3 h (Omelchenko et al., 2003). Scatter factor (SF) was obtained by culturing highly confluent NIH 3T3 fibroblasts in DMEM with 1% FCS for 72 h, followed by 0.45-μm filtration. Before use, SF was tested confirming its ability to induce the dispersion of small colonies of neo-MDCK cells (Stoker et al., 1987).
Statistical Analysis
Where indicated, means and SDs were calculated, and statistical significance was determined by unpaired two-tail t test by using GraphPad Prism 4.0 (GraphPad Software, San Diego, CA).
RESULTS
PMP22 Overexpression Alters Epithelial Cell Proliferation and Morphology
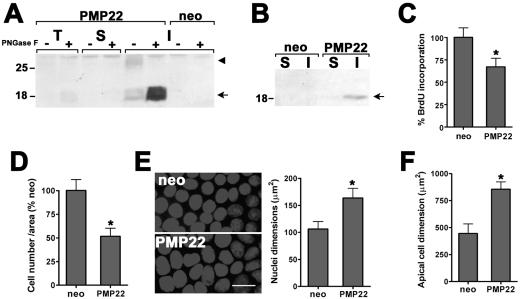
To investigate the role of PMP22 in epithelial biology, human PMP22 (hPMP22) was overexpressed in the pLNCX-2 vector under the control of the CMV promoter in MDCK II cells (hPMP22-MDCK), a frequently used model of polarized epithelia (Stevenson et al., 1988). In total cell lysates (T), by using an antibody optimized to detect the human protein, hPMP22 is faintly observed at ∼26 kDa (Figure 1A, arrowhead). On PNGase F treatment, the protein is detected more prominently at 18 kDa (Figure 1A, arrow). Enhanced immunoreactivity of PMP22 after endoglycosidase treatment has been observed previously (Fabbretti et al., 1995). When the cell lysate is fractionated into detergent-soluble (S) and detergent-insoluble fractions (I), the overexpressed hPMP22 protein is highly enriched in the detergent-insoluble pool. Similar detergent solubility profiles have been observed for both the endogenous canine and exogenous myc-tagged rat PMP22 in MDCK cells (Notterpek et al., 2001). Vector-only cells (neo) are not immunoreactive with the anti-human PMP22 antibody (Figure 1A).
Figure 1.
PMP22 alters epithelial cell proliferation and morphology. (A) Total lysates (T), TX-100–soluble (S) and –insoluble (I) fractions (30 μg/lane) of stable, neo, and human PMP22 (hPMP22)-expressing MDCK II cells were analyzed with (+) and without (-) PNGase F digestion. The glycosylated hPMP22 is detected at ∼26 kDa (arrowhead) and after PNGase F treatment becomes readily visible, migrating at ∼18 kDa (arrow). Compared with total lysates, hPMP22 is enriched in the detergent-insoluble pellet. (B) Plasma membrane targeting of hPMP22 was determined by cell surface biotinylation, followed by PNGase F treatment and immunoblotting with anti-hPMP22 antibodies. The majority of biotinylated hPMP22 is detected in the 1% NP-40–insoluble (I) fraction (arrow). Molecular mass, in kilodaltons. (C) As measured by BrdU incorporation in subconfluent cultures, compared with neo cells, hPMP22 expression reduces DNA synthesis by 32.9 ± 9.5%, (*p < 0.004). (D) In confluent hPMP22-MDCK monolayers, the cell density is 51.7 ± 8.4% of the neo cultures (*p < 0.004). (E) The reduced cell density in confluent PMP22 monolayers is in agreement with an increase in nuclear area. Hoechst staining of representative cultures is shown. Bar, 15 μm. Quantification of nuclei reveals ∼1.5-fold increase in nuclear dimension of hPMP22-MDCK cells compared with neo controls (*p < 0.0001). (F) The apical area of the hPMP22-MDCK cells, outlined by ZO-1 immunostaining, is significantly larger than in neo cells (*p < 0.0004). Error bars in C–F show means ± SD. p values were determined by t test.
We next examined the targeting of hPMP22 to the cell surface, by using biotinylation and subsequent streptavadin precipitation (Ryan et al., 2002) (Figure 1B). Endoglycosidase treatment of the precipitated protein revealed hPMP22 at the plasma membrane. Because the majority of hPMP22 is found in the detergent-insoluble fraction (arrow), the cell surface protein is likely accumulated at apical intercellular junctions and/or, as reported in Schwann cells, possibly in lipid rafts (Erne et al., 2002; Hasse et al., 2002).
Because PMP22 is known to modulate cell cycle progression (Schneider et al., 1988; Manfioletti et al., 1990; Zoidl et al., 1995, 1997; Karlsson et al., 1999), we next examined how hPMP22 might affect epithelial proliferation. In subconfluent cultures, elevated levels of PMP22 lead to a ∼33% reduction in BrdU incorporation, compared with neo cells (Figure 1C) or human occludin (Occ-MDCK)-expressing cells (our unpublished data). Furthermore, cell cycle analysis identified an increase in the percentage of PMP22 cells in G0/G1 (Table 1). At confluence, there were 51.7 ± 8.4% fewer cells in the hPMP22-MDCK cultures, compared with controls (Figure 1D). The lower cell density of confluent hPMP22 monolayers is readily visible by Hoechst imaging of nuclei (Figure 1E). The hPMP22 cell nuclei look larger, and when quantified, they reveal an ∼1.5-fold increase in area (Figure 1E, graph), whereas the Occ-MDCK cells were morphologically similar to neo cells (100.4 ± 2.4%). As predicted from a confluent monolayer with reduced cell density, the apical surface area of the hPMP22 cells, as determined by ZO-1 immunostaining (see below), is ∼1.9-fold larger than that of neo (Figure 1F) or occludin (our unpublished data) controls. These morphological characteristics of hPMP22-MDCK cultures suggest that the monolayer consists of fewer cells with a flattened morphology.
Table 1.
Cell cycle analysis of hPMP22-MDCK cells
| % G0/G1 | % S | % G2/M | |
|---|---|---|---|
| neo-MDCK | 54.08 ± 0.18 | 28.79 ± 0.11 | 17.13 ± 0.20 |
| hPMP22-MDCK | 65.52 ± 0.59 | 22.73 ± 0.79 | 12.75 ± 0.23 |
Asynchronously growing MDCK cells were subjected to flow cytometric analysis to determine cell cycle distribution. hPMP22 expression results in a significant increase in cells in G0/G1 and a decrease in S and G2/M.
PMP22 Does Not Alter Epithelial Polarity or the Localization of Junctional Proteins
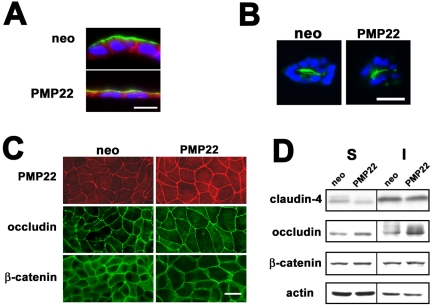
To further evaluate the altered epithelial morphology induced by hPMP22 overexpression, confluent filter-grown monolayers were double immunostained with anti-E-cadherin and anti-GP135 antibodies after z-plane cryosectioning. As expected, in neo-MDCK cultures, GP135 is apical (Ojakian and Schwimmer, 1988) and E-cadherin labels the lateral borders (Figure 2A). Although the hPMP22-MDCK cells are flattened, the protein polarity is similar to that of the neo controls. We also examined protein polarity in a three-dimensional (3-D) model of epithelial cysts in which MDCK cells form a polarized multicellular structure with the apical surface facing the lumen (Ojakian and Schwimmer, 1994). After sectioning and immunostaining for GP135, similar to neo cells, hPMP22 cells formed cysts with normal polarity (Figure 2B).
Figure 2.
Protein polarity and junctional constituents in PMP22-MDCK monolayers. (A) Confluent, filter-grown neo- and hPMP22-MDCK monolayers were immuno-stained and examined by z-plane cryosectioning. In neo- and hPMP22-expressing monolayers, the protein polarity of GP-135 (green) and E-cadherin (red) are apical and basolateral, respectively. The flattened morphology of the PMP22 cells is apparent. Bar, 10 μm. (B) In a 3-D collagen matrix polarization model, both cultures form multicellular cysts with apical GP-135 (green) oriented toward the center. Nuclei are visualized by Hoechst dye (blue). Bar, 15 μm. (C) Compared with neo cultures, an increased level of PMP22 immunoreactivity is detected at intercellular junctions of hPMP22-MDCK cells, when the images are taken at a constant exposure time. In parallel monolayers, the localization of a representative tight and adherens junction constituent, occludin and β-catenin, respectively, seem comparable. Bar, 20 μm. (D) Immunoblotting of 0.5% TX-100–soluble (S) and –insoluble (I) fractions (20 μg/lane) reveals comparable levels and detergent solubilities for claudin-4, occludin, β-catenin, as well as actin, between confluent neo- and hPMP22-MDCK monolayers. Soluble claudin-4 is slightly decreased and insoluble occludin slightly elevated in the hPMP22-MDCK sample.
Because PMP22 is a constituent of apical intercellular junctions (Notterpek et al., 2001; Roux et al., 2004), we next investigated how hPMP22 overexpression affects the localization and detergent solubility properties of tight and adherens junction molecules (Figure 2, C and D). Compared with neo cells, an elevated level of PMP22-like immunoreactivity is associated with the cell-cell contacts of hPMP22-MDCK monolayers (Figure 2C). The expression and localization of a representative tight junction-associated protein, such as occludin, as well as the adherens junction-associated β-catenin, seem similar between neo- and hPMP22-MDCK samples (Figure 2C). Likewise, the distribution of claudin-1, ZO-1, and E-cadherin was unaltered by PMP22 (our unpublished data). As described above (Figure 1F), the larger apical dimensions of the hPMP22 cells are apparent by the junctional immunostaining. To further examine the levels and detergent-solubility characteristics of junction-associated proteins, parallel samples were processed for immunoblotting with the indicated antibodies (Figure 2D). Four examined proteins, claudin-4, occludin, β-catenin, and actin, show similar expression levels and detergent-solubility properties between the neo- and hPMP22-MDCK cells, with a slight decrease in the soluble claudin-4 and an increase in insoluble occludin in the hPMP22 samples. Thus, the overexpression of hPMP22 does not alter epithelial protein polarity nor drastically change the levels or localization of representative tight and adherens junction molecules.
Paracellular Permeability Is Altered by PMP22 Expression
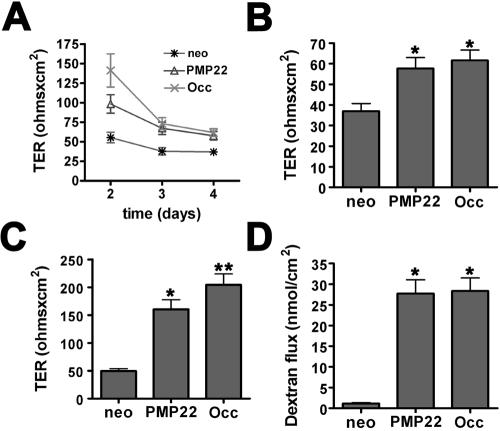
Overexpression of occludin in MDCK cells revealed a role for this protein in regulating paracellular resistance (Balda et al., 1996, 2000; McCarthy et al., 1996, 2000). Because PMP22 is similarly localized to intercellular junctions of MDCK cells (Notterpek et al., 2001), we investigated whether the TER of the hPMP22-expressing epithelial monolayers is altered compared with neo controls. Two days after the addition of Ca2+, hPMP22- and occludin-expressing cells have a high TER level indicative of newly confluent monolayers, after which the TER decreases to steady state by day 4 (Figure 3A). Occ-MDCK monolayers, used as a positive control, have TERs slightly higher than the hPMP22 cells. By 4 d after Ca2+ addition, the monolayers reach a steady-state level of confluence, and the TER of the hPMP22 and occludin cultures is ∼1.6-fold higher than the neo control (Figure 3B). To enhance transgene expression, confluent monolayers were treated with sodium butyrate for 20 h (Gorman et al., 1983), leading to a 5.8- and 1.6-fold increase in hPMP22 and Occ protein levels, respectively. As indicated in Figure 3C, butyrate-treated PMP22- and Occ-MDCK monolayers exhibit a 3.2- and 4.1-fold increase in TER, respectively, compared with butyrate-treated neo cells. A similar phenomenon has been observed previously in epithelial monolayers overexpressing occludin (Balda et al., 1996, 2000; McCarthy et al., 1996, 2000). Because hPMP22 alters MDCK cell morphology, TER values were normalized to the relative linear junctional length for each culture. After correction for junctional length, the differences in TER values between neo control and PMP22- or occludin-overexpressing cells remained highly significant (p < 0.001), with or without butyrate treatment. Expression levels of junctional proteins can alter paracellular flow of nonionic molecules (Balda et al., 1996, 2000; McCarthy et al., 1996, 2000); therefore, we performed a dextran flux assay on confluent hPMP22-MDCK monolayers. Compared with neo controls, the flux of a 3-kDa nonionic FITC-labeled dextran is elevated ∼17-fold in both the hPMP22- and Occ-MDCK monolayers (Figure 3D). These results indicate that elevated expression of hPMP22 results in altered permeability of MDCK monolayers.
Figure 3.
Altered paracellular permeability of epithelial monolayers by hPMP22 expression. (A) TER recordings beginning 2 d after the addition of Ca2+, from newly confluent MDCK monolayers are shown. Three different clones of neo, hPMP22, and Occ cells (n = 3 filters/clone) were used for the quantification. Compared with neo cultures, the TER is elevated in hPMP22 and Occ cells. (B) At steady state, 4 d after plating, hPMP22- and Occ-MDCK monolayers have increased TER, compared with neo cells (*p < 0.0001). (C) Treatment with sodium butyrate (20 h) further enhances the differences in TER values between control and hPMP22, as well as Occ monolayers (*p < 0.0001). (D) The paracellular flux of 3-kDa nonionic FITC-dextran by hPMP22- and Occ-MDCK monolayers is significantly elevated (*p < 0.0001), compared with neo cells. Error bars show means ± SD. p values were determined by t test.
Epithelial Monolayer Permeability Is Perturbed by PMP22 Peptides
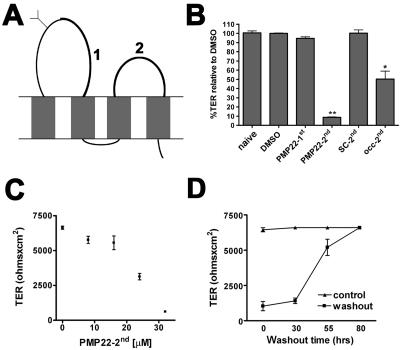
An alternative approach to elucidate the role of junctional proteins is to apply peptides that correspond to extracellular domains of endogenous proteins (Wong, 1997; Wong and Gumbiner, 1997; Lacaz-Vieira et al., 1999; Vietor et al., 2001; Tavelin et al., 2003; Lee et al., 2004). Because the canine PMP22 has not been cloned, we designed peptides representing portions of the first and second extracellular domains of the mouse protein (Figure 4A). Although PMP22 has homology to some members of the claudin family, the proteins are less similar in the extracellular domains (Align software; our unpublished data). Confluent MDCK I monolayers were treated with the indicated peptides, after a Ca2+ switch. Twenty hours after the readdition of Ca2+ with the corresponding peptides (PMP22-1st, -2nd, PMP22-scrambled-2nd, or Occ-2nd), the TER of the monolayers was recorded (Figure 4B). As expected based on the literature (Wong, 1997; Wong and Gumbiner, 1997; Vietor et al., 2001), the Occ-2nd loop peptide inhibited TER recovery by 50.4 ± 8.7%. Similarly, the PMP22-2nd loop peptide diminished TER recovery by 91.3 ± 0.4%. Application of vehicle (DMSO), PMP22-1st, or scrambled-2nd peptides had no significant effect on the TER compared with naïve cells (Figure 4B). The application of PMP22-2nd peptide to intact monolayers had no effect on TER values (our unpublished data). A concentration curve for PMP22-2nd peptide identified an effective range of TER disruption between 8 and 32 μM (Figure 4C). After 60 h of PMP22-2nd treatment, MDCK monolayers remained unable to restore TER (our unpublished data). The washout of PMP22-2nd peptide from MDCK monolayers results in the restoration of the TER to control levels (Figure 4D). Therefore, the disruptive effect of the PMP22-2nd loop peptide on the monolayer TER is reversible.
Figure 4.
PMP22 peptides perturb the TER of epithelial monolayers. (A) Peptides corresponding to a portion of the first (1) and the entire second (2) extracellular loops of murine PMP22, a scrambled second loop, and the second extracellular loop of chicken occludin were applied to confluent MDCK monolayers after a Ca2+ switch. (B) Twenty hours after the addition of the peptides, the TER of PMP22-2nd peptide-treated monolayers (32 μM) remains low compared with naive, DMSO-, PMP22-1st-, or scrambled-2nd–treated cells (**p < 0.0001). TER reformation by monolayers exposed to Occ-2nd peptide (16 μM) is also significantly perturbed (*p < 0.0006). (C) Dosage curve for PMP22-2nd peptide reveals an effective concentration range for TER disruption between 8 and 32 μM. Error bars show means ± SD. p values were determined by t test. (D) After a 20-h PMP22-2nd peptide treatment, and subsequent washout, the monolayers regain a TER similar to untreated samples.
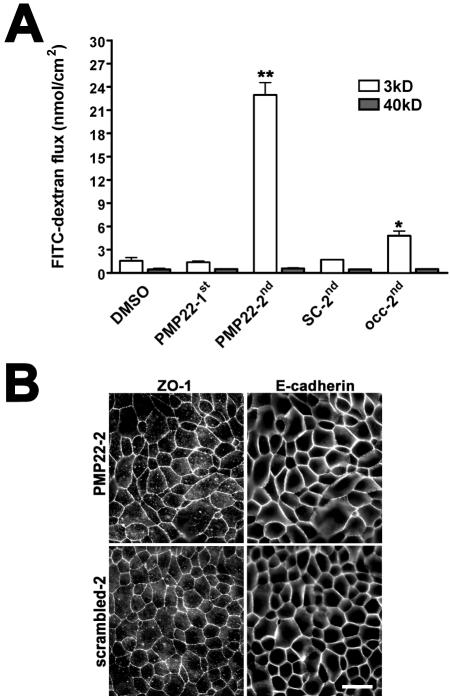
Next, we examined the paracellular flux and the morphology of peptide-treated monolayers (Figure 5). The flux of the 3-kDa, but not the 40-kDa FITC-dextran is significantly elevated in both the PMP22-2nd– and Occ-2nd loop-treated monolayers, indicating a size selective disturbance of paracellular permeability (Figure 5A). The observed increase in the flow of the 3-kDa dextran in Occ-2nd–treated cultures is in agreement with previous reports (Wong and Gumbiner, 1997). To determine whether the PMP22-2nd peptide treatment alters the distribution of junctional constituents, the localization of ZO-1 and E-cadherin was examined in parallel peptide-treated monolayers (Figure 5B). Both ZO-1 and E-cadherin seem unaltered after treatment with PMP22-2nd or scrambled-2nd peptides. These findings indicate that the PMP22-2nd peptide reduces monolayer permeability without radically altering major constituents of apical intercellular junctions.
Figure 5.
PMP22 peptides increase the paracellular flux of epithelial monolayers. (A) MDCK monolayers were treated with the indicated peptides and the paracellular flux of 3-kDa, and 40-kDa FITC-labeled nonionic dextrans were determined. In PMP22-2nd (**p < 0.0001) and Occ-2nd–treated monolayers (*p < 0.002), the paracellular flow of the 3-kDa, but not the 40-kDa, dextran is significantly elevated. Error bars show means ± SD. p values were determined by t test. (B) In PMP22-2nd and scrambled-2nd peptide-treated monolayers (32 μM), the localization of ZO-1 and E-cadherin remain comparable. Bar, 20 μm.
PMP22 Expression Slows the Migration of Epithelial Monolayers
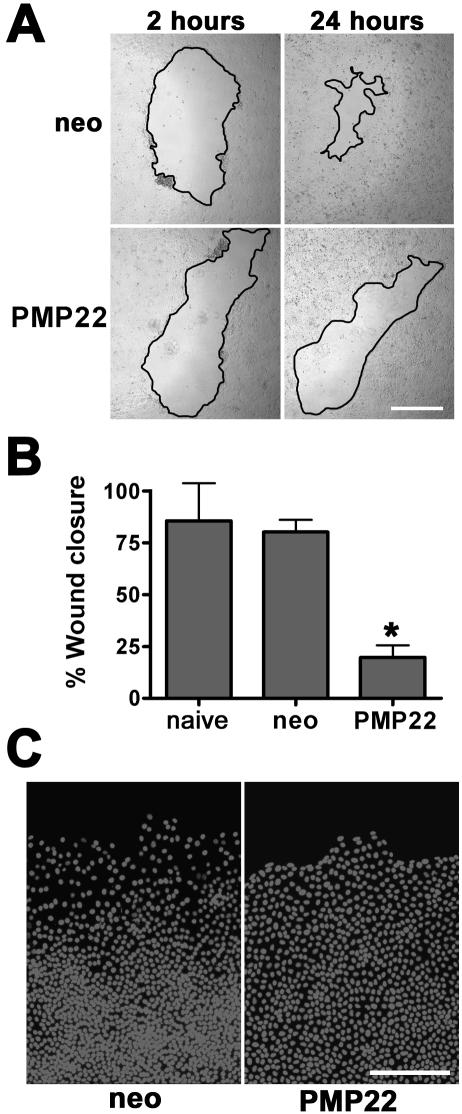
Injured epithelial monolayers display signs of tight junction remodeling before successful wound closure as judged by the down-regulation of ZO-1 and occludin mRNAs (Cao et al., 2002). Because the expression of hPMP22 alters the morphology and permeability of epithelial monolayers, we hypothesized that the protein might modulate the dynamic processes involved in epithelial migration. To investigate this, hPMP22-overexpressing epithelial cells were tested in a two-dimensional wound-migration assay (Figure 6A) (Fenteany et al., 2000). By phase microscopy, 24 h after wounding, the neo cultures nearly close the denuded area (Figure 6A, top). In comparison, the rate of monolayer closure is visibly reduced in hPMP22-MCDK cells (Figure 6A, bottom), which do not cover the denuded area until ∼48 h postwounding. Indeed, within a 24-h period, hPMP22 monolayers exhibit a 60.3 ± 6% reduction in wound closure compared with neo cells (Figure 6B). At higher magnification of Hoechst-stained samples, the neo monolayers display a characteristic wave of migrating cells at the wound edge that look flattened and spread out (Scheffers et al., 2003; Matsubayashi et al., 2004) (Figure 6C). In contrast, the nuclei of hPMP22-expressing cells are more compact at the wound edge and are uniformly spaced throughout the monolayer. Wound closure is unaffected by the overexpression of occludin (our unpublished data).
Figure 6.
PMP22 alters wound healing by epithelial monolayers. (A) Confluent neo- and PMP22-MDCK monolayers were wounded with a pipette tip, and the migration of the cells into the wound area (borders outlined in black) was evaluated at 2 and 24 h. The neo-MDCK monolayers nearly close the wound by 24 h, whereas hPMP22-MDCK cells are unable to similarly reduce the wound area. Bar, 800 μm. (B) Quantification of wounding experiments reveal that compared with naïve and neo cells, hPMP22-MDCK cells are significantly less competent to migrate (*p < 0.0003). Error bars show means ± SD. p values determined by t test. (C) Hoechst staining of parallel samples shows dispersed nuclei along the wound edge in the neo cultures. In comparison, the nuclei of the hPMP22-MDCK cells are densely packed and uniform throughout the image. Bar, 150 μm.
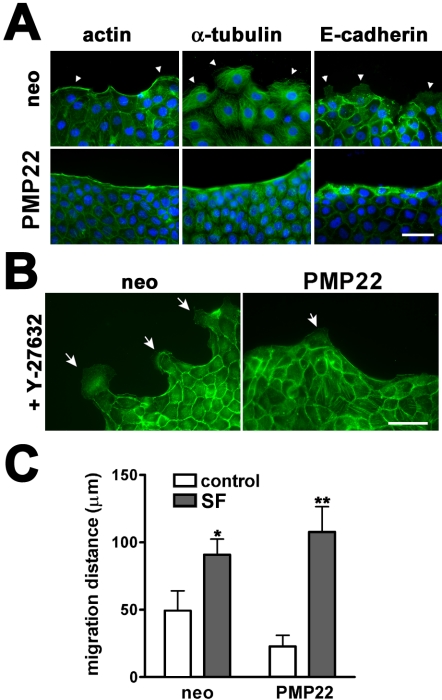
In response to monolayer wounding, migrating MDCK cells maintain cell-cell contacts, form an actin purse-string along the wound edge and pull multiple cell rows forward, by Rac-dependent lamellipodial crawling (Fenteany et al., 2000). Lamellipodial protrusion by leader cells, but not the formation of an actin purse-string, is required by MDCK monolayers to close a wound (Fenteany et al., 2000). Therefore, we examined these two structures in hPMP22-MDCK cells (Figure 7A). As expected, 24 h after monolayer wounding, neo cells (top row) have an actin-belt along the wound edge; with periodic breaks (arrowhead on the right) representative of migrating cells. Leader cell lamellipodia are observed in the neo cultures by tubulin and E-cadherin labeling (arrowheads). An actin purse-string is continuous along the migrating edge of hPMP22 monolayers (Figure 7A, bottom); however, breaks in this actin-belt are largely absent. Additionally, the tubulin and E-cadherin immuno-reactivities seem concentrated along the leading edge in hPMP22 cells of the wound, and the monolayers have fewer cells extending lamellipodia.
Figure 7.
Lamellipodial protrusion in migrating epithelial monolayers is reduced by PMP22. (A) Distribution of actin, α-tubulin, and E-cadherin was examined by fluorescence microscopy along the wound edge at 24 h postwounding. In neo cultures, an actin purse-string and lamellipodial protrusions (arrowheads) are detected (top row). In wounded PMP22 samples, the actin purse-string is uninterrupted and α-tubulin and E-cadherin seem concentrated along the wound edge. Lamellipodial protrusions into the wound space are largely absent in the PMP22-MDCK monolayers. Bar, 40 μm. (B) A 3-h treatment of wounded monolayers with Y-27632, a Rho kinase inhibitor, induces extensive lamellipodial protrusions (arrows) along the wound edge of neo cells, visualized by fluorescent labeling of actin. In PMP22-MDCK monolayers, Y-27632 is unable to bring about a similar response, as cells with lamellipodia are sparse (arrow). Bar, 50 μm. (C) Quantification of epithelial migration after wounding (5 h), in the absence and presence of SF. In normal culture medium, neo cells migrate faster than PMP22-MDCK monolayers. The addition of SF to the medium significantly increases the migration of neo- and PMP22-MDCK cells (*p < 0.0003; **p < 0.0001, respectively). Error bars show means ± SD. p values were determined by t test.
Because lamellipodial protrusion is crucial for MDCK monolayer migration, we examined whether inhibition of Rho-kinase, known to induce lamellipodial expansion in wounded epithelial monolayers (Omelchenko et al., 2003), could overcome the effects of hPMP22 overexpression (Figure 7B). As expected, a 3-h treatment of neo-MDCK cells with a Rho-kinase inhibitor (Y-27632) leads to increased lamellipodial-like cell protrusion, visualized by actin-phalloidin (Figure 7B, arrows). In comparison, hPMP22-MDCK cells seem resistant to the formation of lamellipodia, with few cell protrusions apparent along the wound edge (arrow). Thus, the expression of PMP22 results in the reduced migration of MDCK cells after wounding.
Fibroblast-derived SF has been shown to induce an epithelial to mesenchymal transition (EMT) in MDCK cells (Stoker et al., 1987). Therefore, we investigated whether SF is capable of overriding the inhibitory effect of hPMP22 expression on MDCK monolayer migration. When cultured in SF, neo and hPMP22 cells migrate a similar distance 5 h after a scratch wound (Figure 7C, gray bars). Compared with monolayers in normal medium (Figure 7C, white bars), wound closure in the presence of SF increases by 1.8- and 4.7-fold for the neo and hPMP22-MDCK cells, respectively. These results indicate that although hPMP22-MDCK cells are resistant to the effects of Rho kinase inhibition, they are competent to migrate after SF-induced EMT.
DISCUSSION
The described results indicate that PMP22 plays a role in several aspects of epithelial biology. The overexpression of PMP22 reduces the proliferation and final cell density of epithelial monolayers and induces flattened cell morphology. Monolayers of such cultures have increased TER and paracellular flux of nonionic dextrans. In agreement, a PMP22 peptide disrupts the reformation of paracellular resistance after Ca2+ switch. The migration of epithelial monolayers also is reduced by PMP22 overexpression, possibly due to a deficiency in lamellipodial-forming leader cells. These results suggest that PMP22 takes part in a pathway by which apical cell junctions regulate the proliferation and morphology of epithelial cells and modulate paracellular permeability and cell motility.
Cell junction-associated proteins are known to influence cell proliferation. For example, in addition to regulating paracellular permeability (Balda and Matter, 2000; Reichert et al., 2000), elevated levels of ZO-1 reduce proliferation and cell density in MDCK cells (Balda et al., 2003). This effect is thought to result from sequestration of the transcription factor ZONAB, a ZO-1 binding partner, from the cell nucleus with a concomitant decrease in the level of nuclear CDK4 (Balda et al., 2003). It is yet unknown by what mechanism PMP22 elicits a similar response in the MDCK model, although by quantitative Western analysis the levels of nuclear CDK4 are unchanged between PMP22 and neo control cells (our unpublished data). Thus, the PMP22-mediated effects on cell density and cell cycle do not seem to involve the ZONAB pathway. In Schwann cells and fibroblasts, overexpression of PMP22 by retroviral and transient transfection induces growth arrest, and in some instances apoptosis (Fabbretti et al., 1995; Zoidl et al., 1997, Brancolini et al., 1999). In our stably transduced cell populations, increased apoptosis as judged by Hoechst staining was not observed, possibly due to a lower level of PMP22 expression.
In several cell types the overexpression of PMP22 affects cellular morphology (Brancolini et al., 1999; 2000; Chies et al., 2003). The altered cell shape observed in PMP22-MDCK monolayers is likely the consequence of reduced cell density at confluence, in which a flattened morphology is necessary for maintaining functional cell-cell junctions. An elevated TER can be the result of such a phenomenon, because confluent monolayers with reduced cell density have less linear tight junctional length (Marcial et al., 1984), and paracellular junctions are more permeable than the cell itself (Stefani and Cereijido, 1983). However, after correction for junctional length, the effects of PMP22 overexpression on TER remained highly significant. In accordance, the reduced cell density reported after ZO-1 overexpression did not significantly alter the TER of the MDCK monolayers (Balda and Matter, 2000). In contrast, as shown here and by others (Balda et al., 1996, 2000; McCarthy et al., 1996, 2000), elevated levels of occludin increased the TER, but it was not reported to alter the cell morphology or density of the monolayers.
A role for PMP22 in modulating paracellular flow is supported by the increased ionic and nonionic permeability after exposure to the PMP22-2nd loop peptide. The reduced TER and increased flux of small dextrans may indicate that the second loop peptide disrupts homotypic interactions of PMP22. Indeed, PMP22 is known to form dimers and larger oligomers in vivo and in vitro (Tobler et al., 1999, 2002). As the extracellular domains of PMP22 share no significant homology with the claudins, a potential direct effect on claudins is unlikely. However, the peptides may be perturbing the function of other, as of yet undetermined, epithelial junction-associated proteins that are binding partners for PMP22.
The localization of exogenous and endogenous PMP22 at apical intercellular junctions (Notterpek et al., 2001; Roux et al., 2004), combined with the effects of elevated PMP22 expression or PMP22 peptides on paracellular permeability, supports the notion that PMP22 is a functional constituent of the apical junctional complex. Although it has not been determined ultrastructurally whether PMP22 is at tight or adherens junctions, the protein is capable of altering both the ionic and nonionic permeability of epithelial monolayers. Similar effects have been attained in studies when the expression of the tight junction protein occludin (Balda et al., 1996, 2000; McCarthy et al., 1996, 2000) was modulated. Because PMP22 is detected at intercellular contacts of rat neuroepithelia (Roux et al., 2004), cells devoid of classical tight junctions (Mollgard et al., 1987; Aaku-Saraste et al., 1996), the role of the protein might not be exclusive to tight junctions. In this respect, PMP22 is similar to ZO-1, a junctional protein that in some cell types exists at sites other than the tight junctional complex (Itoh et al., 1993).
Epithelial cells maintain physical contacts during wound closure, whereas they extend Rac-GTPase–dependent lamellipodia (Fenteany et al., 2000). In normal epithelia, lamellipodial protrusion is promoted by inhibiting Rho kinase, likely by disrupting the actin marginal bundles along the wound edge (Omelchenko et al., 2003). PMP22 expression, however, prevents the lamellipodial formation even after Y-27632 treatment. This suggests that an elevated level of the protein interferes with the signaling for lamellipodial protrusion, possibly by acting via the actin cytoskeleton directly or indirectly by modulating the Rac GTPase pathway. In comparison, the same MDCK cells are capable of migration after the application of SF, suggesting that the inhibitory action of PMP22 on wound-induced migration is likely dependent upon epithelial cell-cell contacts, because SF induces a junction-disrupting EMT (Grisendi et al., 1998).
Because PMP22 is predominantly expressed in myelinating Schwann cells, what relevance do these studies in epithelia have to understanding the protein's function in the peripheral nervous system (PNS)? The growth arrest properties of PMP22 seem to occur independently of cell type, but they have yet to be directly linked to PMP22-associated disease pathology. Cell morphology is drastically altered during myelination, an event that involves extensive membrane expansion; however, PMP22 is not required for myelin wrapping (Adlkofer et al., 1995). Cell migration is also crucial to proper nerve development (Lobsiger et al., 2002). Nonetheless, our results show that when epithelia undergo EMT and migrate as individual cells, the overexpression of PMP22 has no inhibitory effect. Typically thought of as a component of compact myelin (Haney et al., 1996), PMP22 has not yet been localized to tight junctions of PNS myelin. Based on the effects of PMP22 on epithelial paracellular permeability, the protein could have a similar role in PNS myelin at claudin-1– and -5–positive autotypic tight junctions (Poliak et al., 2002). Therefore, modulating PMP22 in an epithelial model may provide some clues as to the function of the protein in PNS myelin.
In addition to identifying the participation of PMP22 in epithelial cell biology, we established an in vitro model that is amenable to further experimentation. Using this system, the specific activity of various PMP22 domains can be dissected by examining their effects on epithelial permeability and migration. It will be equally important to identify binding partners of PMP22 at intercellular junctions to fully understand how the protein signals such global changes in epithelial cell biology.
Acknowledgments
We thank Dr. Steven Sugrue and members of the Notterpek laboratory for helpful discussions, Dr. Vivian Wong for technical advice, and Dr. Brian Burke for critical reading of the manuscript. These studies, in part, were supported by the Howard Hughes Medical Institute's Biomedical Research Support Program for Medical Schools (# 76200-550203).
This article was published online ahead of print in MBC in Press (http://www.molbiolcell.org/cgi/doi/10.1091/mbc.E04-07-0551) on January 5, 2005.
Abbreviations used: PMP22, peripheral myelin protein 22; SF, scatter factor; TER, transepithelial electrical resistance; TX-100, Triton X-100; ZO-1, zonula occludens-1.
References
- Aaku-Saraste, E., Hellwig, A., and Huttner, W. B. (1996). Loss of occludin and functional tight junctions, but not ZO-1, during neural tube closure–remodeling of the neuroepithelium prior to neurogenesis. Dev. Biol. 180, 664-679. [DOI] [PubMed] [Google Scholar]
- Adlkofer, K., Martini, R., Aguzzi, A., Zielasek, J., Toyka, K. V., and Suter, U. (1995). Hypermyelination and demyelinating peripheral neuropathy in Pmp22-deficient mice. Nat. Genet. 11, 274-280. [DOI] [PubMed] [Google Scholar]
- Baechner, D., T. Liehr, H. Hameister, H. Altenberger, H. Grehl, U. Suter, and B. Rautenstrauss. (1995). Widespread expression of the peripheral myelin protein-22 gene (PMP22) in neural and non-neural tissues during murine development. J. Neurosci. Res. 42, 733-741. [DOI] [PubMed] [Google Scholar]
- Balda, M. S., Flores-Maldonado, C., Cereijido, M., and Matter, K. (2000). Multiple domains of occludin are involved in the regulation of paracellular permeability. J. Cell. Biochem. 78, 85-96. [PubMed] [Google Scholar]
- Balda, M. S., Garrett, M. D., and Matter, K. (2003). The ZO-1-associated Y-box factor ZONAB regulates epithelial cell proliferation and cell density. J. Cell Biol. 160, 423-432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balda, M. S., and Matter, K. (2000). The tight junction protein ZO-1 and an interacting transcription factor regulate ErbB-2 expression. EMBO J. 19, 2024-2033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balda, M. S., Whitney, J. A., Flores, C., Gonzalez, S., Cereijido, M., and Matter, K. (1996). Functional dissociation of paracellular permeability and transepithelial electrical resistance and disruption of the apical-basolateral intramembrane diffusion barrier by expression of a mutant tight junction membrane protein. J. Cell Biol. 134, 1031-1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brancolini, C., Edomi, P., Marzinotto, S., and Schneider, C. (2000). Exposure at the cell surface is required for gas3/PMP22 to regulate both cell death and cell spreading: implication for the Charcot-Marie-Tooth type 1A and Dejerine-Sottas diseases. Mol. Biol. Cell 11, 2901-2914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brancolini, C., Marzinotto, S., Edomi, P., Agostoni, E., Fiorentini, C., Muller, H. W., and Schneider, C. (1999). Rho-dependent regulation of cell spreading by the tetraspan membrane protein Gas3/PMP22. Mol. Biol. Cell 10, 2441-2459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao, Z., Wu, H. K., Bruce, A., Wollenberg, K., and Panjwani, N. (2002). Detection of differentially expressed genes in healing mouse corneas, using cDNA microarrays. Invest. Ophthalmol. Vis. Sci. 43, 2897-2904. [PubMed] [Google Scholar]
- Chies, R., Nobbio, L., Edomi, P., Schenone, A., Schneider, C., and Brancolini, C. (2003). Alterations in the Arf6-regulated plasma membrane endosomal recycling pathway in cells overexpressing the tetraspan protein Gas3/PMP22. J. Cell Sci. 116, 987-999. [DOI] [PubMed] [Google Scholar]
- Donaldson, J. G. (2003). Multiple roles for Arf 6, sorting, structuring, and signaling at the plasma membrane. J. Biol. Chem. 278, 41573-41576. [DOI] [PubMed] [Google Scholar]
- Erne, B., Sansano, S., Frank, M., and Schaeren-Wiemers, N. (2002). Rafts in adult peripheral nerve myelin contain major structural myelin proteins and myelin and lymphocyte protein (MAL) and CD59 as specific markers. J. Neurochem. 82, 550-562. [DOI] [PubMed] [Google Scholar]
- Fabbretti, E., Edomi, P., Brancolini, C., and Schneider, C. (1995). Apoptotic phenotype induced by overexpression of wild-type gas3/PMP 22, its relation to the demyelinating peripheral neuropathy CMT1A. Genes Dev. 9, 1846-1856. [DOI] [PubMed] [Google Scholar]
- Fenteany, G., Janmey, P. A., and Stossel, T. P. (2000). Signaling pathways and cell mechanics involved in wound closure by epithelial cell sheets. Curr. Biol. 10, 831-838. [DOI] [PubMed] [Google Scholar]
- Gumbiner, B., and Simons, K. A. (1986) A functional assay for proteins involved in establishing an epithelial occluding barrier: identification of a uvomorulin-like polypeptide. J. Cell Biol. 102, 457-468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Mariscal, L., Betanzos, A., Nava, P., and Jaramillo, B. E. (2003). Tight junction proteins. Prog. Biophys. Mol. Biol. 81, 1-44. [DOI] [PubMed] [Google Scholar]
- Gorman, C. M., Howard, B. H., and Reeves, R. (1983). Expression of recombinant plasmids in mammalian cells is enhanced by sodium butyrate. Nucleic Acids Res. 11, 7631-7648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grisendi, S., Arpin, M., and Crepaldi, T. (1998). Effect of hepatocyte growth factor on assembly of zonula occludens-1 protein at the plasma membrane. J. Cell. Physiol. 176, 465-471. [DOI] [PubMed] [Google Scholar]
- Haney, C., Snipes, G. J., Shooter, E. M., Suter, U., Garcia, C., Griffin, J. W., and Trapp, B. D. (1996). Ultrastructural distribution of PMP22 in Charcot-Marie-Tooth disease type 1A. J. Neuropathol. Exp. Neurol. 55, 290-299. [DOI] [PubMed] [Google Scholar]
- Hasse, B., Bosse, F., and Muller, H. W. (2002). Proteins of peripheral myelin are associated with glycosphingolipid/cholesterol-enriched membranes. J. Neurosci. Res. 69, 227-232. [DOI] [PubMed] [Google Scholar]
- Itoh, M., Nagafuchi, A., Yonemura, S., Kitani-Yasuda, T., and Tsukita, S. (1993). The 220-kD protein colocalizing with cadherins in non-epithelial cells is identical to ZO-1, a tight junction-associated protein in epithelial cells: cDNA cloning and immunoelectron microscopy. J. Cell Biol. 121, 491-502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson, C., Afrakhte, M., Westermark, B., and Paulsson, Y. (1999). Elevated level of gas3 gene expression is correlated with G0 growth arrest in human fibroblasts. Cell Biol. Int. 23, 351-358. [DOI] [PubMed] [Google Scholar]
- Lacaz-Vieira, F., Jaeger, M. M., Farshori, P., and Kachar, B. (1999). Small synthetic peptides homologous to segments of the first external loop of occludin impair tight junction resealing. J. Membr. Biol. 168, 289-297. [DOI] [PubMed] [Google Scholar]
- Lee, D. B., Jamgotchian, N., Allen, S. G., Kan, F. W., and Hale, I. L. (2004). Annexin A2 heterotetramer: role in tight junction assembly. Am. J. Physiol. 287, F481-491. [DOI] [PubMed] [Google Scholar]
- Lisanti, M. P., Le Bivic, A., Sargiacomo, M., and Rodriguez-Boulan, E. (1989). Steady-state distribution and biogenesis of endogenous Madin-Darby canine kidney glycoproteins: evidence for intracellular sorting and polarized cell surface delivery. J. Cell Biol. 109, 2117-2127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lobsiger, C. S., Taylor, V., and Suter, U. (2002). The early life of a Schwann cell. Biol. Chem. 383, 245-253. [DOI] [PubMed] [Google Scholar]
- Manfioletti, G., Ruaro, M. E., Del Sal, G., Philipson, L., and Schneider, C. (1990). A growth arrest-specific (gas) gene codes for a membrane protein. Mol. Cell. Biol. 10, 2924-2930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcial, M. A., Carlson, S. L., and Madara, J. L. (1984). Partitioning of paracellular conductance along the ileal crypt-villus axis: a hypothesis based on structural analysis with detailed consideration of tight junction structure-function relationships. J. Membr. Biol. 80, 59-70. [DOI] [PubMed] [Google Scholar]
- Matsubayashi, Y., Ebisuya, M., Honjoh, S., and Nishida, E. (2004). ERK activation propagates in epithelial cell sheets and regulates their migration during wound healing. Curr. Biol. 14, 731-735. [DOI] [PubMed] [Google Scholar]
- McCarthy, K. M., Francis, S. A., McCormack, J. M., Lai, J., Rogers, R. A., Skare, I. B., Lynch, R. D., and Schneeberger, E. E. (2000). Inducible expression of claudin-1-myc but not occludin-VSV-G results in aberrant tight junction strand formation in MDCK cells. J. Cell Sci. 113, 3387-3398. [DOI] [PubMed] [Google Scholar]
- McCarthy, K. M., Skare, I. B., Stankewich, M. C., Furuse, M., Tsukita, S., Rogers, R. A., Lynch, R. D., and Schneeberger, E. E. (1996). Occludin is a functional component of the tight junction. J. Cell Sci. 109, 2287-2298. [DOI] [PubMed] [Google Scholar]
- Mollgard, K., Balslev, Y., Lauritzen, B., and Saunders, N. R. (1987). Cell junctions and membrane specializations in the ventricular zone (germinal matrix) of the developing sheep brain: a CSF-brain barrier. J. Neurocytol. 16, 433-444. [DOI] [PubMed] [Google Scholar]
- Naef, R., and Suter, U. (1998). Many facets of the peripheral myelin protein PMP22 in myelination and disease. Microsc. Res. Tech. 41, 359-371. [DOI] [PubMed] [Google Scholar]
- Notterpek, L., Shooter, E. M., and Snipes, G. J. (1997). Upregulation of the endosomal-lysosomal pathway in the trembler-J neuropathy. J. Neurosci. 17, 4190-4200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Notterpek, L., Roux, K. J., Amici, S. A., Yazdanpour, A., Rahner, C., and Fletcher, B. S. (2001). Peripheral myelin protein 22 is a constituent of intercellular junctions in epithelia. Proc. Natl. Acad. Sci. USA 98, 14404-14409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojakian, G. K., and Schwimmer, R. (1988). The polarized distribution of an apical cell surface glycoprotein is maintained by interactions with the cytoskeleton of Madin-Darby canine kidney cells. J. Cell Biol. 107, 2377-2387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojakian, G. K., and Schwimmer, R. (1994). Regulation of epithelial cell surface polarity reversal by beta 1 integrins. J. Cell Sci. 107, 561-576. [PubMed] [Google Scholar]
- Omelchenko, T., Vasiliev, J. M., Gelfand, I. M., Feder, H. H., and Bonder, E. M. (2003). Rho-dependent formation of epithelial “leader” cells during wound healing. Proc. Natl. Acad. Sci. USA 100, 10788-10793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pareek, S., Notterpek, L., Snipes, G. J., Naef, R., Sossin, W., Laliberte, J., Iacampo, S., Suter, U., Shooter, E. M., and Murphy, R. A. (1997). Neurons promote the translocation of peripheral myelin protein 22 into myelin. J. Neurosci. 17, 7754-7762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poliak, S., Matlis, S., Ullmer, C., Scherer, S. S., and Peles, E. (2002). Distinct claudins and associated PDZ proteins form different autotypic tight junctions in myelinating Schwann cells. J. Cell Biol. 159, 361-372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollack, A. L., Runyan, R. B., and Mostov, K. E. (1998). Morphogenetic mechanisms of epithelial tubulogenesis: MDCK cell polarity is transiently rearranged without loss of cell-cell contact during scatter factor/hepatocyte growth factor-induced tubulogenesis. Dev. Biol. 204, 64-79. [DOI] [PubMed] [Google Scholar]
- Prince, L. S., Tousson, A., and Marchase, R. B. (1993). Cell surface labeling of CFTR in T84 cells. Am. J. Physiol. 264, C491-C498. [DOI] [PubMed] [Google Scholar]
- Reichert, M., Muller, T., and Hunziker, W. (2000). The PDZ domains of zonula occludens-1 induce an epithelial to mesenchymal transition of Madin-Darby canine kidney I cells. Evidence for a role of beta-catenin/Tcf/Lef signaling. J. Biol. Chem. 275, 9492-9500. [DOI] [PubMed] [Google Scholar]
- Roux, K. J., Amici, S. A., and Notterpek, L. (2004). The temporospatial expression of peripheral myelin protein 22 at the developing blood-nerve and blood-brain barriers. J. Comp. Neurol. 474, 578-588. [DOI] [PubMed] [Google Scholar]
- Ryan, M. C., Shooter, E. M., and Notterpek, L. (2002). Aggresome formation in neuropathy models based on peripheral myelin protein 22 mutations. Neurobiol. Dis. 10, 109-118. [DOI] [PubMed] [Google Scholar]
- Sabo, S. L., Ikin, A. F., Buxbaum, J. D., and Greengard, P. (2001). The Alzheimer amyloid precursor protein (APP) and FE65, an APP-binding protein, regulate cell movement. J. Cell Biol. 153, 1403-1414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakakibara, A., Furuse, M., Saitou, M., Ando-Akatsuka, Y., and Tsukita, S. (1997). Possible involvement of phosphorylation of occludin in tight junction formation. J. Cell Biol. 137, 1393-1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schachner, M., Martini, R., Hall, H., and Orberger, G. (1995). Functions of the L2/HNK-1 carbohydrate in the nervous system. Prog. Brain Res. 105, 183-188. [DOI] [PubMed] [Google Scholar]
- Scheffers, M. S., van der Bent, P., van de Wal, A., van Eendenburg, J., Breuning, M. H., de Heer, E., and Peters, D. J. (2004). Altered distribution and co-localization of polycystin-2 with polycystin-1 in MDCK cells after wounding stress. Exp. Cell Res. 292, 219-230. [DOI] [PubMed] [Google Scholar]
- Schneider, C., King, R. M., and Philipson, L. (1988). Genes specifically expressed at growth arrest of mammalian cells. Cell. 54, 787-793. [DOI] [PubMed] [Google Scholar]
- Snipes, G. J., Suter, U., and Shooter, E. M. (1993). Human peripheral myelin protein-22 carries the L2/HNK-1 carbohydrate adhesion epitope. J. Neurochem. 61, 1961-1964. [DOI] [PubMed] [Google Scholar]
- Stefani, E., and Cereijido, M. (1983). Electrical properties of cultured epithelioid cells (MDCK). J. Membr. Biol. 73, 177-184. [DOI] [PubMed] [Google Scholar]
- Stevenson, B. R., Anderson, J. M., Goodenough, D. A., and Mooseker, M. S. (1988). Tight junction structure and ZO-1 content are identical in two strains of Madin-Darby canine kidney cells which differ in transepithelial resistance. J. Cell Biol. 107, 2401-2408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoker, M., Gherardi, E., Perryman, M., and Gray, J. (1987). Scatter factor is a fibroblast-derived modulator of epithelial cell mobility. Nature. 327, 239-242. [DOI] [PubMed] [Google Scholar]
- Suter, U., and Snipes, G. J. (1995). Peripheral myelin protein 22, facts and hypotheses. J. Neurosci. Res. 40, 145-151. [DOI] [PubMed] [Google Scholar]
- Takeda, Y., Notsu, T., Kitamura, K., and Uyemura, K. (2001). Functional analysis for peripheral myelin protein PASII/PMP 22, is it a member of claudin superfamily? Neurochem. Res. 26, 599-607. [DOI] [PubMed] [Google Scholar]
- Tavelin, S., Hashimoto, K., Malkinson, J., Lazorova, L., Toth, I., and Artursson, P. (2003). A new principle for tight junction modulation based on occludin peptides. Mol. Pharmacol. 64, 1530-1540. [DOI] [PubMed] [Google Scholar]
- Taylor, V., Welcher, A. A., Program, A. E., and Suter, U. (1995). Epithelial membrane protein-1, peripheral myelin protein 22, and lens membrane protein 20 define a novel gene family. J. Biol. Chem. 270, 28824-28833. [DOI] [PubMed] [Google Scholar]
- Tobler, A. R., Notterpek, L., Naef, R., Taylor, V., Suter, U., and Shooter, E. M. (1999). Transport of Trembler-J mutant peripheral myelin protein 22 is blocked in the intermediate compartment and affects the transport of the wild-type protein by direct interaction. J. Neurosci. 19, 2027-2036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tobler, A. R., Liu, N., Mueller, L., and Shooter, E. M. (2002). Differential aggregation of the Trembler and Trembler J mutants of peripheral myelin protein 22. Proc. Natl. Acad. Sci. USA 99, 483-488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Itallie, C. M., and Anderson, J. M. (1997). Occludin confers adhesiveness when expressed in fibroblasts. J. Cell Sci. 110, 1113-1121. [DOI] [PubMed] [Google Scholar]
- Vietor, I., Bader, T., Paiha, K., and Huber, L. A. (2001). Perturbation of the tight junction permeability barrier by occludin loop peptides activates beta-catenin/TCF/LEF-mediated transcription. EMBO Rep. 2, 306-312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong, V., and Gumbiner, B. M. (1997). A synthetic peptide corresponding to the extracellular domain of occludin perturbs the tight junction permeability barrier. J. Cell Biol. 136, 399-409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong, V. (1997). Phosphorylation of occludin correlates with occludin localization and function at the tight junction. Am. J. Physiol. 273, C1859-C1867. [DOI] [PubMed] [Google Scholar]
- Wulf, P., and Suter, U. (1999). Embryonic expression of epithelial membrane protein 1 in early neurons. Brain Res. Dev. Brain Res. 116, 169-180. [DOI] [PubMed] [Google Scholar]
- Zoidl, G., Blass-Kampmann, S., D'Urso, D., Schmalenbach, C., and Muller, H. W. (1995). Retroviral-mediated gene transfer of the peripheral myelin protein PMP22 in Schwann cells: modulation of cell growth. EMBO J. 14, 1122-1128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zoidl, G., D'Urso, D., Blass-Kampmann, S., Schmalenbach, C., Kuhn, R., and Muller, H. W. (1997). Influence of elevated expression of rat wild-type PMP22 and its mutant PMP22Trembler on cell growth of NIH3T3 fibroblasts. Cell Tissue Res. 287, 459-470. [DOI] [PubMed] [Google Scholar]