Abstract
Background
Prostate cancer is the second most common type of cancer in Canadian men. Radical prostatectomy is one of the treatment options available, and involves removing the prostate gland and surrounding tissues. In recent years, surgeons have begun to use robot-assisted radical prostatectomy more frequently.
We aimed to determine the clinical benefits and harms of the robotic surgical system for radical prostatectomy (robot-assisted radical prostatectomy) compared with the open and laparoscopic surgical methods. We also assessed the cost-effectiveness of robot-assisted versus open radical prostatectomy in patients with clinically localized prostate cancer in Ontario.
Methods
We performed a literature search and included prospective comparative studies that examined robot-assisted versus open or laparoscopic radical prostatectomy for prostate cancer. The outcomes of interest were perioperative, functional, and oncological. The quality of the body of evidence was examined according to the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) Working Group criteria.
We also conducted a cost–utility analysis with a 1-year time horizon. The potential long-term benefits of robot-assisted radical prostatectomy for functional and oncological outcomes were also evaluated in a 10-year Markov model in scenario analyses. In addition, we conducted a budget impact analysis to estimate the additional costs to the provincial budget if the adoption of robot-assisted radical prostatectomy were to increase in the next 5 years.
A needs assessment determined that the published literature on patient perspectives was relatively well developed, and that direct patient engagement would add relatively little new information.
Results
Compared with the open approach, we found robot-assisted radical prostatectomy reduced length of stay and blood loss (moderate quality evidence) but had no difference or inconclusive results for functional and oncological outcomes (low to moderate quality evidence). Compared with laparoscopic radical prostatectomy, robot-assisted radical prostatectomy had no difference in perioperative, functional, and oncological outcomes (low to moderate quality evidence).
Compared with open radical prostatectomy, our best estimates suggested that robot-assisted prostatectomy was associated with higher costs ($6,234) and a small gain in quality-adjusted life-years (QALYs) (0.0012). The best estimate of the incremental cost-effectiveness ratio (ICER) was $5.2 million per QALY gained. However, if robot-assisted radical prostatectomy were assumed to have substantially better long-term functional and oncological outcomes, the ICER might be as low as $83,921 per QALY gained. We estimated the annual budget impact to be $0.8 million to $3.4 million over the next 5 years.
Conclusions
There is no high-quality evidence that robot-assisted radical prostatectomy improves functional and oncological outcomes compared with open and laparoscopic approaches. However, compared with open radical prostatectomy, the costs of using the robotic system are relatively large while the health benefits are relatively small.
BACKGROUND
Health Condition
Prostate cancer is the second most common cancer (after non-melanoma skin cancers) and the third leading cause of death among Canadian men. It has been estimated that, in Canada, prostate cancer accounted for 21% of all new cancer cases (21,600) and 10% of all cancer deaths (4,000) in men in 2016.1 In Ontario, men have a 15.4% lifetime probability of developing prostate cancer,2 and the incidence among all new cancer cases in 2012 was 21.6%.3
Prostate cancer arises within the prostate gland of the male reproductive system. Most prostate cancers are asymptomatic and slow growing. Symptoms usually appear if the prostate cancer is quite advanced locally, and may include a weak or interrupted flow of urine, frequent or trouble with urination, and painful ejaculation.
Prostate cancer typically affects men over the age of 60 years. Risk factors that have been associated with the development of prostate cancer are family history, African descent, obesity, a high-fat diet, and physical inactivity. The 5-year survival rate is relatively good for prostate cancer, at around 96%.4
Clinical Need and Target Population
Since clinically localized prostate cancer usually causes no symptoms, early detection tests have been developed to identify prostate cancer while it remains confined to the prostate. Most patients are referred to a urologist because they have abnormal results on their digital rectal examination or an elevated prostate-specific antigen (PSA) level. Prostate-specific antigen is a protein produced by prostate cells that can be measured in the blood and is normally present at low levels. Higher PSA levels may indicate prostate cancer or other noncancerous prostate conditions such as benign prostatic hyperplasia or prostatitis (an inflammation of the prostate).
If prostate cancer is suspected in patients, a needle biopsy is performed to confirm the diagnosis. Positive biopsies are scored using the Gleason system, which typically grades biopsies from 6 to 10 (6 being well-differentiated, 7 moderately differentiated, and 8–10 poorly differentiated). Gleason scores ≤ 6 are generally considered low-grade tumours. A less well-differentiated prostate cancer indicates a more aggressive tumour.
Tumour stage is also used for prognosis and refers to the degree in which the tumour has involved the prostate gland or has spread. Tumour stage generally ranges from T1 to T45:
- T1: tumour is too small to be seen on imaging scans or felt during examination, but can be found incidentally during a biopsy or surgery
-
∘T1a: tumour is in ≤ 5% of tissue taken
-
∘T1b: tumour is in > 5% of tissue taken
-
∘T1c: tumour is found by biopsy after an elevated PSA level was detected
-
∘
- T2: tumours are confined within the prostate gland
-
∘T2a: tumour is in ≤ 50% of one lobe
-
∘T2b: tumour is in > 50% of one lobe
-
∘T2c: tumour is in both lobes
-
∘
- T3: tumour has extended through the capsule that surrounds the prostate
-
∘T3a: tumour has gone through the capsule without invading the seminal vesicles
-
∘T3b: tumour has invaded the seminal vesicles
-
∘
T4: tumour has invaded structures or tissues near the prostate other than the seminal vesicles (e.g., bladder neck, rectum, pelvic wall)
The Gleason score and tumour stage are used to predict outcomes for prostate cancer. By using the PSA, Gleason score, and tumour stage, risk stratification schemes have been developed that are significantly associated with biochemical recurrence (elevated PSA levels after radical prostatectomy) and mortality owing to prostate cancer. Different risk stratification systems exist with slight variations in categorical definitions.6 However, patients are generally grouped as low, intermediate, or high risk. Below is a risk stratification from D'Amico et al,7 the first proposed three-group stratification for radical prostatectomy (and radiotherapy):
Low risk: PSA ≤ 10 ng/mL and Gleason score ≤ 6 and T1–T2a
Intermediate risk: PSA 10–20 ng/mL and/or Gleason score 7 and/or T2b and not low-risk
High risk: PSA > 20 ng/mL or Gleason score 8–10 or clinical stage ≥ T2c
Current Treatment Options
Current standard treatment options for prostate cancer are watchful waiting, active surveillance, surgery (radical prostatectomy), radiation therapy, and hormone therapy. The differences between prostate cancer incidence and mortality indicate that many patients may not benefit from immediate treatment (such as surgery) of localized prostate cancer, such as those patients diagnosed with early-stage low-grade prostate cancer. Patients who forgo immediate therapy may opt for watchful waiting or active surveillance. In watchful waiting, the focus is on managing quality of life and other illnesses. In active surveillance, patients are actively monitored (e.g., with repeated diagnostic tests) and offered treatment if the cancer seems as if it is likely to progress.
Radical treatments with a curative aim consist of radical prostatectomy and radiation therapy. In selecting potential candidates for radical prostatectomy, surgeons consider:
Life expectancy
The natural history of the diagnosed prostate cancer
The ability of radical prostatectomy to cure the prostate cancer
The morbidity of radical prostatectomy
Patient choice
Radical prostatectomy is typically offered to patients with localized prostate cancer (stage ≤ T2) and a life expectancy greater than 10 years. Patients with poorer prognostic factors, such as locally advanced prostate cancer (stage T3a), a Gleason score > 8, and a PSA level > 20 ng/mL, may also be offered radical prostatectomy. However, they will likely need further treatments.
Radical prostatectomy involves removing the prostate and the surrounding tissues and seminal vesicles. It is performed via one of four surgical approaches:
-
1.
Open retropubic: the operation is performed through a primary incision through the pubic area
-
2.
Open perineal: the operation is performed through a primary incision in the perineum (a rarely performed approach compared with open retropubic prostatectomy)
-
3.
“Conventional” laparoscopic (referred to in this report as “laparoscopic radical prostatectomy”): this is a minimally invasive approach in which the operation is performed through keyhole incisions in the abdomen; a video camera is inserted to help the surgeon view the prostate
-
4.
Robot-assisted laparoscopic (referred to in this report as “robot-assisted radical prostatectomy”): this is similar to the laparoscopic approach; however, a surgeon manipulates robotic arms of a surgical system that are inserted into the small incision points
Depending on tumour characteristics and a patient's sexual function, either nerve-sparing radical prostatectomy (to preserve erectile function) or non-nerve-sparing radical prostatectomy is commonly performed. Pelvic lymphadenectomy (the removal of lymph nodes in the pelvis) can be performed concurrently and is generally reserved for patients with a higher risk of lymph node involvement.
The laparoscopic approach was introduced in the 1990s and gained acceptance through advances in medical technology. However, laparoscopic radical prostatectomy did not disseminate widely because it is technically difficult to perform. In the early 2000s, with the advent of robotic surgery technology, the option emerged to use a robotic surgical system for radical prostatectomy (i.e., robot-assisted radical prostatectomy).
Technology
The da Vinci Surgical System is the only robotic surgical system currently available for clinical use in Canada. It has four main components:
-
1.
A surgeon's console, where the surgeon sits and views a magnified three-dimensional image of the surgical field
-
2.
A patient side cart, which consists of three instrument arms and one endoscope (a tubular optical instrument for viewing the inside of the body)
-
3.
Detachable instruments, which are used to simulate a person's fine motor movements
-
4.
A three-dimensional vision system
The main procedural advantages of using the robotic system are improved dexterity, its precision, three-dimensional imaging, and its ergonomic design for surgeons. The clinical experts we consulted with noted that the ergonomic ease of the robotic system may allow experienced but ageing surgeons to prolong and maintain their surgical skills. One potential disadvantage of the robotic system is that while it helps the surgeon accurately manipulate tissue, the surgeon experiences only visual feedback and none of the touch experienced in laparoscopic and open radical prostatectomies.
Costs include the initial cost of the robotic surgical system, annual maintenance, training for surgeons and operating room personnel, and disposable instruments.
The overall goals of laparoscopic and robot-assisted radical prostatectomy are to maintain the benefits of minimally invasive surgery, while maximizing:
Cancer control
Urinary continence—the ability to voluntarily control the release of urine
Erectile function—the ability to develop or maintain an erect penis during sexual activity
Compared with the open retropubic approach that is performed through a 6- to 8-cm incision, robot-assisted radical prostatectomy involves multiple incisions ranging from only 5- to 12-mm in diameter.
Regulatory Information
The first generation of the da Vinci Surgical System has been licensed by Health Canada since 2001 as a Class IV medical device. In 2010, Health Canada approved the use of the third generation, the da Vinci Si. In 2010 and 2012, it also approved new components, categorized as Class II medical devices. The da Vinci Surgical Systems are intended to be used by trained physicians in an operating room environment for a wide range of surgeries, including urologic, gynecologic, cardiac, colorectal, oropharyngeal, thoracic, and general surgeries. The latest version of the surgical system, the da Vinci Xi, was approved by Health Canada in July 2016. The da Vinci Surgical Systems are marketed internationally, with about 3,500 systems installed worldwide as of 2015.
Patient Values and Preferences
Patient preference is integral to the decision-making process for prostate cancer treatment. Patients may prefer the minimally invasive nature of robot-assisted radical prostatectomy compared with open radical prostatectomy because there are fewer surgical scars, a faster recovery, and decreased perioperative outcomes (see the Outcomes of Interest section).8,9 Based on data from the United States, patients with a higher socioeconomic status are more likely to undergo minimally invasive radical prostatectomy (laparoscopic or robot-assisted). These patients may be more informed about all surgical options and seek out minimally invasive prostatectomy if it is not offered locally.10
Some experts have also suggested that marketing and promotional language unrelated to presentation of potential benefits and harms may influence patients to prefer robot-assisted over open radical prostatectomy.11 Media coverage and online marketing for robot-assisted radical prostatectomy are also more widespread,12 although there is the potential for inaccurate information from online sources.13,14
Data from the United States have also shown that patients who undergo robot-assisted radical prostatectomy may be more likely to regret their decision, possibly because their expectations were very high.15 Patients undergoing robot-assisted surgery were found to have higher expectations for a significantly shorter hospital stay, earlier return to physical activity, and earlier return of potency than those undergoing open radical prostatectomy.16 Baseline functional outcomes, age, and peri- and postoperative outcomes were independent predictors of patients’ satisfaction and decision regret following robot-assisted radical prostatectomy.17,18
A Canadian study by Davison et al19 examined decision regret using the Decision Regret Scale to measure distress and remorse at 1 year postsurgery. However, in contrast with US findings, the authors found scores were low for the robot-assisted and open groups and that results did not differ significantly between them.
Canadian and International Contexts
Robot-assisted prostatectomy is currently publicly funded through the quality-based procedure pathway for prostate cancer. However, funding rates for robot-assisted and open radical prostatectomy are the same and are determined through the Ontario Ministry of Health and Long-Term Care's case costing methodology (provincial cost per weighted case × institution's case mix index). The choice of open or robot-assisted radical prostatectomy is typically determined by the treating surgeon at institutions where robot-assisted prostatectomy is available.
The da Vinci Surgical Systems that are currently in use in Ontario and their associated maintenance fees have been purchased through charitable donations or have come from within a hospital's budget. Experts have told us that in 2016, there were 10 Ontario hospitals that owned at least one da Vinci Surgical System.
Other than Ontario, as of 2016 only three provinces had hospital centres with at least one da Vinci Surgical System: Alberta (Edmonton and Calgary), Quebec (Montreal and Quebec City), and British Columbia (Vancouver). In provinces where robot-assisted radical prostatectomy is currently available, the provincial fee codes for physician billings do not distinguish between laparoscopic and robot-assisted radical prostatectomy.
Experts also told us that more than 3,500 robot-assisted surgical procedures are performed in Canada each year. Of these procedures, about 64% were urologic surgeries (more than 80% were radical prostatectomies), with gynecologic surgeries being the second most common type of procedure, at 25%.
According to experts, the adoption of robot-assisted surgery has been slower in Canada than in the United States, with about 70% of radical prostatectomies still performed using the open procedure versus less than 15% in the United States. In the United States, funding for robot-assisted radical prostatectomy is covered by the Centers for Medicare and Medicaid Services (CMS) under the same code as laparoscopic radical prostatectomy. This agency reimburses hospitals at the same rate, regardless of surgical approach. Most private insurers also do not pay an additional fee for robot-assisted radical prostatectomy.20 However, a study among privately insured younger patients suggests hospitals may receive more per case for minimally invasive radical prostatectomy than open radical prostatectomy.21
In other comparable jurisdictions such as the United Kingdom and Australia, the adoption of robot-assisted radical prostatectomy has also been higher than in Canada. In the United Kingdom, 2013 data showed an approximately even proportion of robot-assisted versus non-robot-assisted radical prostatectomy procedures.22 According to experts, in Australia in 2015, 58% of radical prostatectomies were performed with robotic assistance. Of note, both the United Kingdom and Australia have a two-tiered hospital system (public and private), with relatively higher usage of the da Vinci Surgical System in each country's private system.
Without robotic assistance, performing laparoscopic radical prostatectomy is technically challenging, with a steep learning curve. The advanced skills required to perform laparoscopic radical prostatectomy have limited its widespread use. Robot-assisted radical prostatectomy has become the primary minimally invasive surgical option for prostate cancer.
It has been proposed that robot-assisted radical prostatectomy can be mastered by most prostate surgeons, although Ontario data are lacking. However, part of the mandate of academic surgeons in Ontario is to train the next generation of surgeons. This includes providing critical analysis of innovative technologies such as robot-assisted surgery, which makes up the majority of performed radical prostatectomies in high-income countries.
Research Questions
Our health technology assessment aimed to answer the following questions:
- What are the benefits and harms of robot-assisted radical prostatectomy for prostate cancer, compared with:
-
∘Open retropubic radical prostatectomy?
-
∘Conventional laparoscopic radical prostatectomy (without robotic assistance)?
-
∘
What is the cost-effectiveness of robot-assisted radical prostatectomy compared with open retropubic radical prostatectomy in men with clinically localized prostate cancer?
What is the potential budget impact in Ontario of publicly funding robot-assisted radical prostatectomy for localized prostate cancer treatment?
What are the needs, priorities, and preferences for those with lived experience in determining the type of radical prostatectomy they receive? How do these factors influence a patient's decision-making process?
CLINICAL EVIDENCE REVIEW
Objective
This study aimed to assess the benefits and harms of robot-assisted radical prostatectomy compared with open retropubic radical prostatectomy (referred to in this report as “open radical prostatectomy”) and laparoscopic radical prostatectomy for prostate cancer.
Methods
Research questions were developed by Health Quality Ontario in consultation with experts in the topic area.
Sources
We performed a literature search on April 21, 2016 to retrieve studies published from January 1, 2006 to the search date. We used the Ovid interface to search the following databases: MEDLINE, Embase, Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews, Health Technology Assessment, National Health Service Economic Evaluation Database (NHSEED), and Database of Abstracts of Reviews of Effects (DARE).
Search strategies were developed by medical librarians using controlled vocabulary (e.g., Medical Subject Headings) and relevant keywords. The final search strategy was peer-reviewed using the PRESS Checklist.23 Database auto-alerts were created in MEDLINE and Embase and monitored for the duration of the HTA review.
See Appendix 1 for Literature Search Strategies, including all search terms.
Literature Screening
A single reviewer reviewed the abstracts and, for those studies meeting the eligibility criteria, we obtained full-text articles. We also examined reference lists for any additional relevant studies not identified through the search.
Inclusion Criteria
English-language full-text publications
Studies published between January 1, 2006, and April 21, 2016
Randomized controlled trials (RCTs), prospective comparative nonrandomized studies, and systematic reviews
Studies comparing robot-assisted radical prostatectomy with laparoscopic or open radical prostatectomy for prostate cancer
Exclusion Criteria
Animal and in vitro studies
Editorials, commentaries, conference abstracts, nonsystematic reviews, noncomparative and retrospective studies (e.g., case reports, case series, registry studies, or studies involving the use of administrative data)
Studies of robot-assisted, laparoscopic, or open radical prostatectomy for salvage treatment
Studies of simple prostatectomy, open perineal radical prostatectomy, or minimally invasive radical prostatectomy (where laparoscopic and robot-assisted radical prostatectomy cannot be distinguished)
Studies that compare different techniques for one type of radical prostatectomy (e.g., nerve-sparing versus non-nerve-sparing robot-assisted radical prostatectomy)
Studies that do not report the outcomes of interest, or where the outcomes of interest cannot be extracted
Outcomes of Interest
Perioperative outcomes of interest were:
Operative time
Length of hospital stay
Estimated blood loss
Transfusion rates
Duration of indwelling catheterization
Rates of hospital readmission
Complication rates
Functional outcomes included:
Urinary function
Erectile function
Health-related quality of life
Pain
Time to mobilization or return to work or activity
Oncological outcomes were:
Positive surgical margin rates
Biochemical (PSA) recurrence-free rates
Data Extraction
We extracted relevant information on study characteristics; the study population, details of the intervention, comparator(s), and outcomes of interest (PICO); and risk-of-bias items. We collected information about the:
Source (e.g., primary author, year, country)
Methods (e.g., study design, inclusion and exclusion criteria, patient assignment, patient population characteristics, details of the intervention and comparators], number of surgeons, surgeon experience, length of follow-up)
Outcomes (e.g., differences in patient characteristics between groups, definition of outcomes of interest, details on outcome assessment and measurement, data time points, loss to follow-up)
Statistical Analysis
We performed an analysis of individual studies using Review Manager v. 5.3 (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Summary measures were expressed as the mean difference for continuous data using the inverse-variance method and risk ratio for dichotomous data using the Mantel-Haenszel method. A random effects model was used according to the Cochrane handbook.24 We also examined graphical displays of the forest plots. We considered a P value of ≤ .05 statistically significant for the overall effect estimate. Where data pooling was considered inappropriate in the case of considerable heterogeneity (I2 > 75%), we summarized the data narratively.
Quality of Evidence
We examined the quality of the body of evidence for each outcome according to the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) Working Group criteria.25 The overall quality was determined to be high, moderate, low, or very low using a step-wise, structured method.
Expert Consultation
Between March 2016 and October 2016, we consulted with several experts on robot-assisted radical prostatectomy. Consulted experts included physicians in the specialty areas of urology, oncology, and surgery and a health economics researcher in prostate cancer. Their role was to refine the clinical review plan, contextualize the evidence, and confirm the volume of robot-assisted radical prostatectomy. However, the statements, conclusions, and views expressed in this report do not necessarily represent the views of the consulted experts.
Results
Literature Search
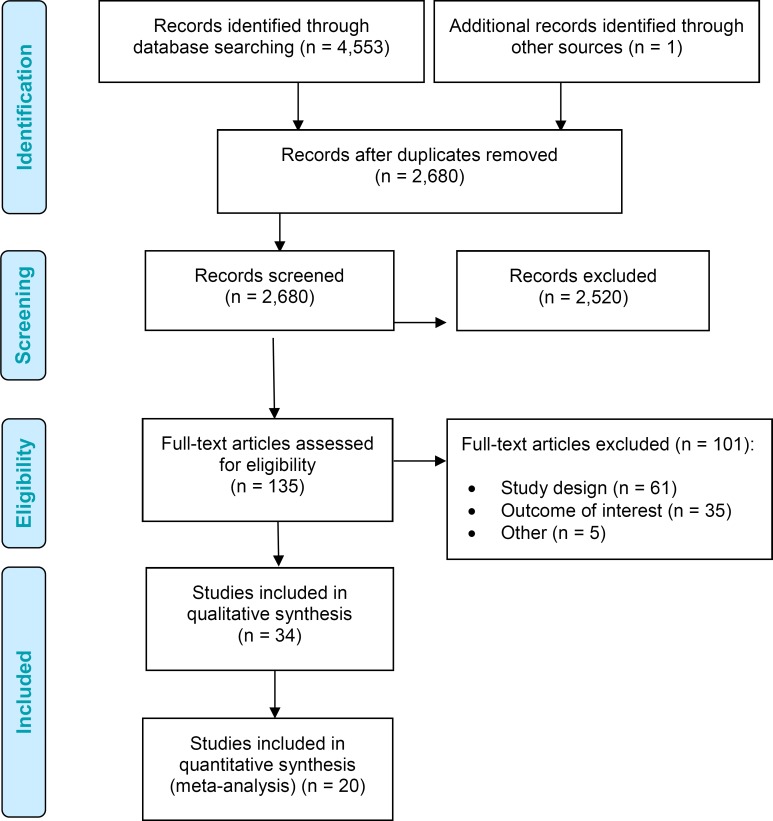
The database search yielded 4,553 citations published between January 1, 2006, and April 21, 2016. After removing duplicates, we reviewed titles and abstracts to identify potentially relevant articles. We obtained the full texts of these articles for further assessment. Thirty-three studies (two RCTs and 31 nonrandomized studies) met the inclusion criteria. We hand-searched the reference lists of the included studies, along with health technology assessment websites and other sources, to identify additional relevant studies. After the search date, we included another RCT26 found through experts.
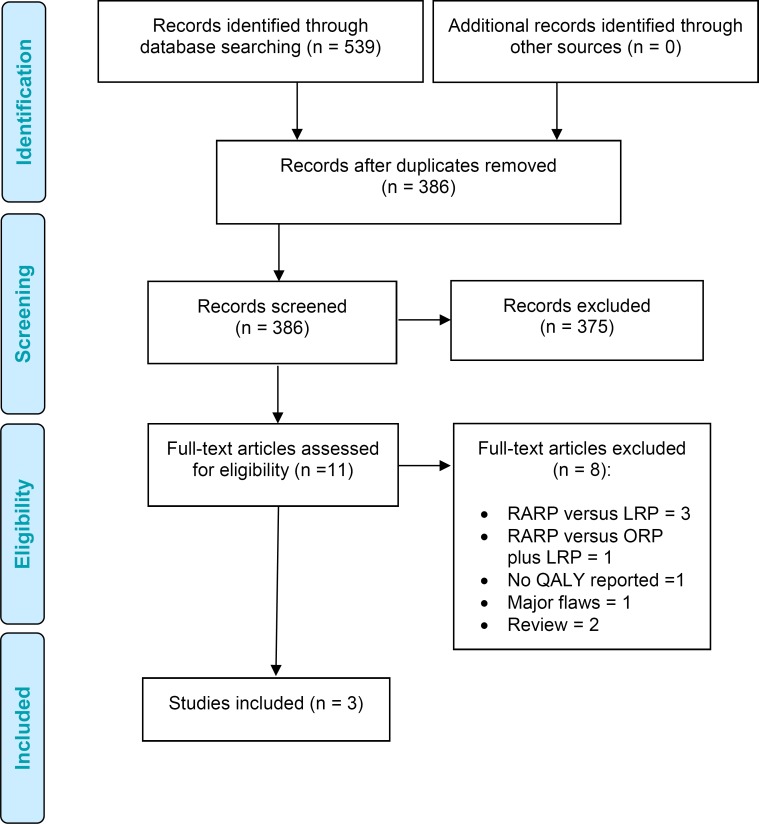
Figure 1 presents the flow diagram for the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA).
Figure 1: PRISMA Flow Diagram for the Clinical Evidence Review.
Source: Adapted from Moher et al.27
Systematic Reviews
Numerous systematic reviews and meta-analyses have been conducted comparing robot-assisted radical prostatectomy with open or laparoscopic approaches (Table A21 in Appendix 5). However, they varied in their study inclusion criteria, included studies, and methods of analyses.
In 2010 the Medical Advisory Secretariat, within the Ontario Ministry of Health and Long-Term Care, conducted a systematic review on robot-assisted surgery for gynecologic and urologic cancers in Ontario.28 It found that compared with open radical prostatectomy, the clinical benefits of robot-assisted radical prostatectomy included reduced lengths of hospital stay, blood loss, transfusion rates, and positive surgical margin rates; and improved erectile function. When compared with the laparoscopic approach, robot-assisted radical prostatectomy reduced blood loss and transfusion rates. However, all included studies were nonrandomized, and surgeon skill was noted to have an impact on functional and oncological outcomes.
None of the health technology assessments or systematic reviews published restricted study design to prospective comparative studies. Typically the authors mixed prospective and retrospective nonrandomized studies in their quantitative analyses. The majority of the published reviews contained literature search end dates from 2011 or earlier, therefore excluding more current literature. However, all of the studies noted limitations within the evidence base for robot-assisted radical prostatectomy. As such, authors usually advised that conclusions be interpreted with caution given the heterogeneity (diversity) within the evidence.
In these reviews, perioperative outcomes such as reduced length of hospital stay, blood loss, transfusion rate, and complication rate were the most consistently reported that favoured the robot-assisted approach compared with open surgery. Increased operative time was also noted for the robot-assisted approach. The results were inconsistent for continence, potency, and positive surgical margin rates. Some results favoured the robot-assisted approach, while others found no difference or inconclusive results. Some reviews also suggested faster recovery of functional outcomes for the robot-assisted approach. When a significant difference for positive surgical margin rates was found, it was typically a reduction favouring robot-assisted radical prostatectomy in patients with pT2 cancer but not pT3 cancer. No reviews found significant differences in biochemical recurrence rates between robot-assisted and open radical prostatectomy.
Differences in outcomes between robot-assisted and laparoscopic radical prostatectomy were generally less apparent.
Since the reviews did not fit our specific inclusion criteria, or had been conducted many years ago, we also evaluated primary studies.
Study Characteristics of Included Primary Studies
We found only two RCTs29,30 for robot-assisted versus laparoscopic radical prostatectomy, and one RCT26 for robot-assisted versus open radical prostatectomy. In the latter, results are so far available for only 3-month outcomes. Tables 1 and 2 summarize the study characteristics of the included RCTs and prospective comparative studies. Two studies included patients from the same study population, and both were included because they reported some different outcomes of interest.31,32
Table 1:
Randomized Controlled Trials on Robot-Assisted Versus Open or Laparoscopic Radical Prostatectomy
| Author, Year | Country | Patient Eligibility | Surgeon | Comparators | Main Outcomes | Follow-Up |
|---|---|---|---|---|---|---|
| Robot-assisted vs. open radical prostatectomy | ||||||
| Yaxley et al, 201626 | Australia | Inclusion: age 35–75 years, newly diagnosed clinically localized prostate cancer; no previous history of head injury, dementia, or psychiatric illness; no concurrent other cancer; estimated life expectancy ≥ 10 years Exclusion: evidence of nonlocalized prostate cancer, PSA > 20 ng/mL, previous laparoscopic hernia repair, previous pelvic radiotherapy or major pelvic surgery, another malignancy within past 5 years |
1 surgeon > 200 RARPs 1 surgeon > 1,500 ORPs |
RARP ORP | Primary: urinary continence, erectile function, oncological outcomes Secondary: pain, physical and mental functioning, fatigue, bowel function, prostate cancer distress, psychological distress, time to return to work |
24 months (results not yet reported) (3-month interim results reported so far) |
| Robot-assisted vs. laparoscopic radical prostatectomy | ||||||
| Asimakopoulos et al, 201130 | Italy | Inclusion: age ≤ 70 years, clinically organ-confined prostate cancer (clinical stage T1–T2), Gleason score ≤ 7, total serum PSA ≤ 10 ng/mL, normal preoperative continence, IIEF-6 ≥ 17, and normal IPSS Exclusion: preoperative incontinence or moderate to severe erectile dysfunction (IIEF-6 < 17); neoadjuvant therapy; any previous prostatic, urethral, or bladder neck surgery; positive MRI results for extracapsular extension; no bilateral nerve sparing |
1 surgeon (> 300 RARPs, > 900 LRPs) | RARP LRP |
Primary: erectile function, continence at 12 months Secondary: perioperative outcomes, complication rate, oncological outcomes |
12 months |
| Porpiglia et al, 201329 | Italy | Inclusion: age 40–75 years, T1-T2N0M0, any prostate size Exclusion: previous radiation therapy, hormonal therapy, transurethral resection of the prostate |
1 surgeon (> 100 RARPs, > 600 LRPs) | RARP LRP |
Primary: continence at 3 months Secondary: continence at different times, perioperative results, rate of PSM, recovery of erectile function |
12 months |
Abbreviations: IIEF, International Index of Erectile Function Questionnaire; IPSS, International Prostate Symptom Score; LRP, laparoscopic radical prostatectomy; MRI, magnetic resonance imaging; PSA, prostate-specific antigen; ORP, open radical prostatectomy; PSM, positive surgical margin; RARP, robot-assisted radical prostatectomy; TNM, tumour staging (tumour, lymph node, metastasis).
Table 2:
Nonrandomized Prospective Comparative Studies for Robot-Assisted Versus Open or Laparoscopic Radical Prostatectomy
| Author, Year | Country | Patient Eligibility | Surgeon | Comparators | Main Outcomes | Follow-Up |
|---|---|---|---|---|---|---|
| Robot-assisted vs. open and laparoscopic radical prostatectomy | ||||||
| Ball et al, 200633 | United States | Newly diagnosed prostate cancer; all patients were candidates for surgical intervention | 7 surgeons (2 performing RARP; 3, ORP; 2, LRP) | 82 RARPs 135 ORPs 125 LRPs |
Health-related quality of life | 36 months |
| Robot-assisted vs. open radical prostatectomy | ||||||
| Bier et al, 201634 | Canada | Patients with clinically localized prostate cancer undergoing radical prostatectomy | 1 surgeon | 128 RARPs 174 ORPs |
Return to work, return to normal daily activity | 12 months |
| Breyer et al, 201035 | United States | Biopsy-confirmed prostate cancer | At least 3 surgeons | 293 RARPs 695 ORPs |
Bladder neck contracture incidence | 12 months |
| Carlsson et al, 201036 | Sweden | Inclusion: clinically localized clinical T1–T2 prostate cancer Exclusion: no previous treatment for prostate cancer, no history of neoadjuvant or adjuvant hormonal therapy |
9 surgeons (6 performing RARP) Varying experience |
1,253 RARPs 485 ORPs |
Adverse events or complications | 24 months |
| Davison et al, 201419 | Canada | Inclusion: clinically localized clinical T1–T2 prostate cancer Exclusion: no previous treatment for prostate cancer, no history of neoadjuvant or adjuvant hormonal therapy |
4 surgeons (2 performing RARP; 2, ORP) | 78 RARPs 73 ORPs |
Health-related quality of life | 12 months |
| Di Pierro et al, 201137 | Switzerland | Patients with prostate cancer undergoing radical prostatectomy and pelvic lymph node dissection | 4 surgeons (1 performing RARP; 3, ORP) | 75 RARPs 75 ORPs |
Oncological outcomes, urinary continence, erectile function, complications, | 12 months |
| Doumerc et al, 201038 | Australia | Inclusion: patients with prostate cancer undergoing radical prostatectomy Exclusion: first 50 cases of RARP, patients with factors increasing surgical difficulty |
1 surgeon (learning curve) | 212 RARPs 502 ORPs |
Operative outcomes | Up to 18 months |
| Farnham et al, 200639 | United States | Clinically localized prostate cancer | 1 surgeon | 176 RARPs 103 ORPs |
Blood loss, perioperative hematocrit, transfusion requirements | 14 months |
| Ficarra et al, 200940 | Italy | Clinically localized prostate cancer | 6 surgeons (2 with > 50 RARPs each, 4 with > 400 ORPs each) | 103 RARPs 105 ORPs |
PSM, surgical time, blood loss, transfusion rate, complications, urinary continence, time to catheter removal, length of hospital stay | 12 months |
| Fode et al, 201441 | Denmark | Patients with biopsy-proven localized prostate cancer, undergoing radical prostatectomy | 9 surgeons (4 performing RARP and ORP; 3, ORP only; 2, RARP only) | 585 RARPs 453 ORPs |
PSM, potency, continence, complications | 12 months |
| Fracalanza et al, 200842 | Italy | Clinically localized prostate cancer | 4 surgeons (1 with > 50 RARPs, 3 with >300 ORPs) | 35 RARPs 26 ORPs |
Acute phase reaction, operative time, length of hospital stay, PSM | Post-RP |
| Geraerts et al, 201331 | Belgium | Inclusion: localized or locally advanced prostate cancer Exclusion: cognitive problems, non-Dutch speaking, simultaneously planned for salvage procedure or other surgery of pelvic region |
3 surgeons (1 with > 150 RARPs, 1 with > 3,000 ORPs, 1 with > 50 RARPs and 700 ORPs) | 64 RARPs 116 ORPs | Primary: time to continence, cumulative continence incidence Secondary: point prevalence of continence |
12 months |
| Haglind et al, 201543,a | Sweden | Patients with prostate cancer undergoing radical prostatectomy, age < 75 years, ability to write or read Swedish, clinical stage T1–T3, no sign of distant metastasis, PSA < 20 ng/mL, surgeon with ≥ 100 procedures | Multiple surgeons | 1,847 RARPs 778 ORPs |
Continence, sexual function, perioperative outcomes | 12 months |
| Ham et al, 200844 | South Korea | Prostate cancer without distant metastasis | 1 surgeon (199 ORPs, 223 RARPs) | 35 early RARPs, 188 late RARPs 89 early ORPs, 110 late ORPs |
Continence, sexual function | 12 months |
| Hong et al, 201045 | South Korea | Patients with American Society of Anesthesiologists physical status I or II | 2 surgeons (1 with > 180 RARPs; 1 with ORP experience) | 26 RARPs 26 ORPs |
Perioperative outcomes, complications (venous gas embolism) | Post-RP |
| Kim et al, 201146 | South Korea | Patients with prostate cancer undergoing radical prostatectomy | 1 surgeon | 528 RARPs 235 ORPs |
Continence, sexual function, perioperative outcomes | 24 months |
| Kordan et al, 201047 | United States | Patients with clinically localized prostate cancer undergoing radical prostatectomy and lymphadenectomy | 4 surgeons (1 performing RARP; 2, ORP; 1, both) | 830 RARPs 414 ORPs |
Primary: transfusion Secondary: PSM, estimated blood loss, change in hematocrit |
Post-RP |
| Loeb et al, 201048 | United States | Patients with prostate cancer undergoing radical prostatectomy | 1 surgeon | 152 RARPs 137 ORPs |
Primary: benign prostate glands at bladder neck margin | Post-RP |
| Lott et al, 201549 | Brazil | Patients with prostate cancer undergoing radical prostatectomy | 8 surgeons (2 performed RARP; they had no previous laparoscopic experience but had > 10 years’ ORP experience. 2 performed ORP and had robotic experience. 4 had an average of 25 years’ experience with RP) | 50 RARPs 34 ORPs |
Primary: urinary continence, erectile function Secondary: histological outcomes |
12 months |
| Ludovico et al, 201350 | Italy | Inclusion criteria: clinically localized prostate cancer (clinical < T2b), PSA < 10 ng/mL, Gleason score < 7, life expectancy > 10 years, preoperative IIEF score > 25, EHS of 4, in a stable relationship Exclusion: other neoplasm, lower urinary tract or major concomitant diseases, previous abdominal surgery, urinary incontinence, or erectile dysfunction treated with phosphodiesterase type 5 inhibitors or intracorporeal injection of prostaglandin E1 |
1 surgeon (> 50 RARPs) | 82 RARPs 48 ORPs |
Primary: potency recovery rate Secondary: continence, PSM, complications |
12 months |
| Miller et al, 200751 | United States | Clinically localized prostate cancer (T1–T2) | Not reported | 42 RARPs 120 ORPs |
Health-related quality of life | 6 weeks |
| Nelson et al, 200752 | United States | Patients with prostate cancer undergoing radical prostatectomy | Not reported | 629 RARPs 374 ORPs |
Length of hospital stay, readmission rates, unscheduled clinical visits, complications | Not specified |
| O'Malley et al, 200653 | Australia | Inclusion criteria: patients with prostate cancer undergoing radical prostatectomy Exclusion criteria: patients treated early in learning curve |
1 surgeon | 102 RARPs 102 ORPs |
PSM, learning curve | 3 months |
| Philippou et al, 201254 | UK | Patients with prostate cancer undergoing radical prostatectomy | 1 surgeon (> 50 ORPs) | 50 RARPs 50 ORPs |
Oncologic outcomes, sexual function and urinary continence, perioperative parameters, complications | 3 months |
| Thompson et al, 201355 | Australia | Patients with prostate cancer undergoing radical prostatectomy | 1 surgeon (> 3,000 ORPs) | 837 RARPs 674 ORPs |
PSM, health-related quality of life | 12 months |
| Wallerstedt et al, 201532,a | Sweden | Patients with prostate cancer undergoing radical prostatectomy, age < 75 years, ability to write or read Swedish, clinical stage T1–T3, no sign of distant metastasis, PSA < 20 ng/mL, surgeon with ≥ 100 procedures | Multiple surgeons | 1,847 RARPs 778 ORPs |
Perioperative, complications, readmission rates | 3 months |
| Wood et al, 200756 | United States | Patients with prostate cancer undergoing radical prostatectomy | 1 surgeon | 117 RARPs 89 ORPs |
Perioperative, oncological, functional, health-related quality of life | 6 months |
| Robot-assisted vs. laparoscopic radical prostatectomy | ||||||
| Asimakopoulos et al, 201357 | Italy | Inclusion: age ≤ 70 years, clinical T1c–cT2, PSA < 10 ng/mL, biopsy Gleason score < 7; fully continent, potent, and candidates for bilateral nerve-sparing radical prostatectomy Exclusion: history of neoadjuvant treatment for prostate cancer, clinical or MRI suspicion for locally advanced prostate cancer |
1 surgeon | 136 RARP 91 LRP |
Primary: pentafecta (potency, urinary continence, no perioperative complications, negative surgical margins, and no BCR) Secondary: preoperative or prognostic factors predicting pentafecta |
Up to 3 years |
| Berge et al, 201358 | Norway | Patients with localized prostate cancer undergoing radical prostatectomy | 4 surgeons | 210 RARPs 210 LRPs |
Health-related quality of life | 36 months |
| Ploussard et al, 200959 | France | Patients with prostate cancer undergoing radical prostatectomy | 3 surgeons (1 performing RARP; 2, LRP) | 83 RARPs 205 LRPs |
Operative time, short-term postoperative complications | Not specified |
| Ploussard et al, 201460 | France | Patients with prostate cancer undergoing radical prostatectomy | At least 5 surgeons (3 senior surgeons performed most of the LRPs; 2 senior surgeons performed RARPs and had experience with > 100 LRPs) | 1,009 RARPs 1,377 LRPs |
Perioperative, functional, oncological | 24 months |
| Willis et al, 201261 | United States | Clinically localized prostate cancer (≤ T2) | 1 surgeon (had performed 250 LRPs; new to RARP) | 121 RARPs 161 LRPs |
Perioperative, oncological, functional, health-related quality of life | 12 months |
Abbreviations: BCR, biochemical recurrence; EHS, Erection Hardness Score; LRP, laparoscopic radical prostatectomy; MRI, magnetic resonance imaging; PSA, prostate-specific antigen; PSM, positive surgical margin; ORP, open radical prostatectomy; RARP, robot-assisted radical prostatectomy; RP, radical prostatectomy; T, tumour stage.
The studies involved one to nine surgeons (or stated “multiple” surgeons) who, prior to the start of the trial, had varying amounts and types of surgical experience (e.g., transition from either open or laparoscopic radical prostatectomy to robot-assisted prostatectomy). Some studies did not report surgeon number or experience. The surgical technique, including nerve-sparing status, also differed between studies.
Follow-up in the studies varied from postsurgery to 3 years, with the majority of the studies having a follow-up duration of 1 year or less. Studies also inconsistently reported pelvic floor training after the radical prostatectomy, rehabilitation for continence recovery, and medication use for erectile dysfunction. Some stated that patients were encouraged to perform training or rehabilitation or given medication to treat erectile dysfunction to use as required. Others did not mention any type of postsurgery care.
These variations resulted in a heterogeneous body of evidence. Because of the clinical and statistical heterogeneity, we primarily summarized the results for outcomes in tabular and narrative formats and used graphics to show the inconsistency of the data. Where possible, we presented the meta-analysis results when the I2 statistic was < 75%. However, the results were unadjusted, and we could not factor in other patient- and surgeon-important factors (e.g., patient risk level, surgeon experience, and surgical technique).
Appendix 2 presents the results of the risk-of-bias assessment for the included RCTs (using Cochrane's Risk of Bias tool) and prospective comparative nonrandomized studies (using the Risk of Bias in Non-randomized Studies—of Interventions tool), along with the results of the methodology checklist. Because of the nature of the nonrandomized studies, many had moderate to serious risks of bias for the pre-intervention phase of the Risk of Bias in Non-randomized Studies—of Interventions tool, under the items of potential confounders and patient selection. For the postintervention domain, we found moderate risks of bias primarily within outcome measurement owing to the use of nonstandardized methods of measurement. For the included RCTs, randomization primarily addressed most of the pre-intervention risks of bias present in the included nonrandomized studies, such as significant differences between baseline patient and tumour characteristics.
Tables 1 and 2 present study characteristics for robot-assisted radical prostatectomy compared with the open approach or with laparoscopic radical prostatectomy.
Robot-Assisted Versus Open Radical Prostatectomy
The following section presents the results for robot-assisted versus open radical prostatectomy.
Operative Time
One RCT and 12 nonrandomized studies reported operative time. Results for operative time between robot-assisted and open radical prostatectomy showed significant heterogeneity among studies when mean operative time was pooled (I2 = 98%) (Table A5). The range for operative time was large, from a median or mean of 125 to 330 minutes for robot-assisted surgery and 103 to 280 minutes for open surgery.
Different definitions for “operative time” were used, from total operative time to skin-to-skin time (i.e., the time from incision to finishing suturing). However, despite differences in measurement, the general trend was a significant decrease in operative time in favour of the open approach in the nonrandomized studies, which may be explained by the extra setup time required for the robot-assisted approach. In contrast, the RCT by Yaxley et al26 showed a significant increase in operative time for the open approach (for both operative time and surgery time). Among the nonrandomized studies, only Hong et al45 showed no difference between groups, and Philippou et al54 found results favouring robot-assisted radical prostatectomy.
A learning curve also exists between operative time and surgeon experience. Doumerc et al38 noted that operative time for robot-assisted radical prostatectomy decreased with experience, and continued to decrease over the study period, with about 110 cases needed to achieve an operative duration of less than 3 hours.
Table 3 presents the GRADE evidence profile for operative time.
Table 3:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Operative Time
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 12 non-RCTs | Serious limitations (−1)b | Serious limitations (−1)c | Serious limitations (−1)d | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
GRADE starts at low for nonrandomized studies. Differences in baseline patient characteristics and surgeon experience between groups may impact operative time.
One study showed results favouring robot-assisted radical prostatectomy,54 while another study showed no difference between groups.
Differences affected by variations in room setup and personnel between hospital centres.
Length of Hospital Stay
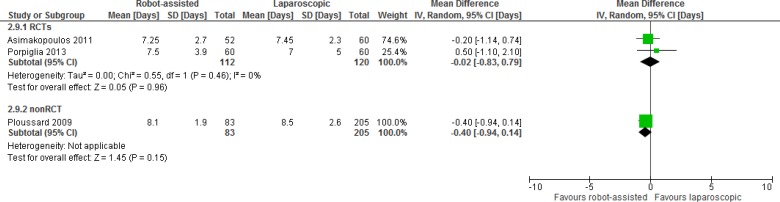
One RCT and 11 nonrandomized studies reported on length of hospital stay, which varied from a mean or median of 1 day to 6 days. This may be explained by differences in postoperative care between hospitals. For example, Nelson et al found that patients undergoing robot-assisted or open radical prostatectomy can be treated on the same clinical pathway and that a targeted hospital discharge date of postoperative day 1 can be achieved in the majority of patients.52 Likely for this reason, of the nonrandomized studies, it is the only one that reports a length of stay of about 1 day for both robot-assisted and open radical prostatectomies. All other studies, including the RCT, found a significant reduction in length of stay with robot-assisted prostatectomy. We did not pool the data because of considerable statistical heterogeneity between studies (I2 = 100%).
Table A6 presents the results, and Table 4 shows the GRADE evidence profile for length of hospital stay.
Table 4:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Length of Hospital Stay
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 11 non-RCTs | Serious limitations (−1)b | No serious limitationsc | Serious limitations (−1)d | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
GRADE starts at low for nonrandomized studies. Differences in baseline clinical factors may impact length of hospital stay.
Differences in hospital discharge pathways likely account for large variability in mean/median lengths of hospital stay between studies; however, almost all studies still show significance for reduced length of stay for the robot-assisted group.
Generalizability concerns from centres where length of stay is not reflective of average Ontario times.
Estimated Blood Loss
One RCT and 16 studies reported estimated blood loss for robot-assisted versus open radical prostatectomy. Estimated blood lost was measured either categorically or, most often, as a continuous variable. All studies showed significantly less estimated blood loss for robot-assisted radical prostatectomy (Table A7). There were large variations in blood loss between groups, ranging from a mean or median of 100 mL to over 400 mL for robot-assisted surgery. The reported mean or median blood loss in the open group was typically higher, at around 500 mL to over 1300 mL.
Fode et al found that, along with a low prostate volume (P < .001), non-nerve-sparing surgery (P < .001), and surgeon (P < .001), robot-assisted radical prostatectomy was a predictor of low perioperative blood loss in multivariable analysis. There was a relative risk of 2.89 (95% confidence interval [CI] 2.52–3.3, P < .001) between the open and robot-assisted groups.41
Table 5 presents the GRADE evidence profile for estimated blood loss.
Table 5:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Estimated Blood Loss
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | Large magnitude of effect (+1) | ⊕⊕⊕ Moderate |
| 16 non-RCTs | Serious limitations (−1)b | No serious limitationsc | No serious limitations | Serious limitations (−1)a | Undetected | Large magnitude of effect (+1) | ⊕⊕ Low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences. Large variability within data in nonrandomized studies. Difficulties in accurately measuring blood loss.
GRADE starts at low for nonrandomized studies. Differences in clinical characteristics between groups may affect outcome.
While results all significantly favour robot-assisted radical prostatectomy for reduced blood loss, large differences in blood loss amount between studies may be impacted by clinical characteristics and surgeon experience between groups.
Transfusion Rates
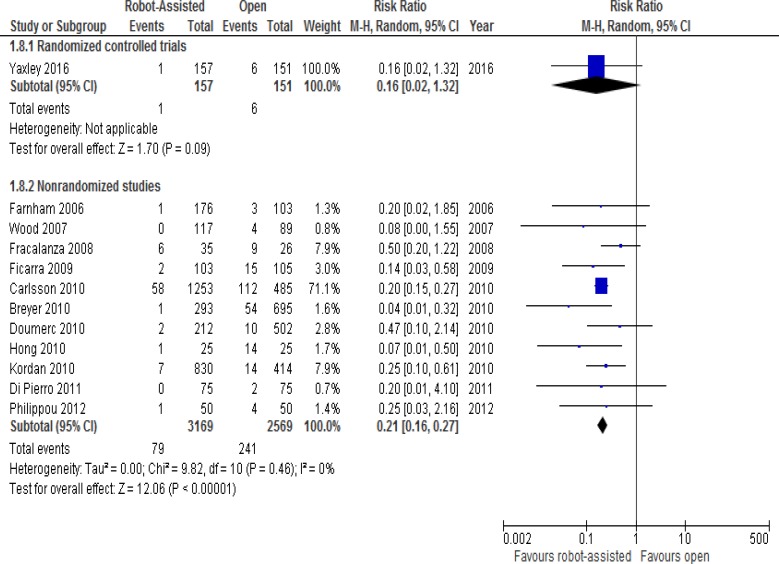
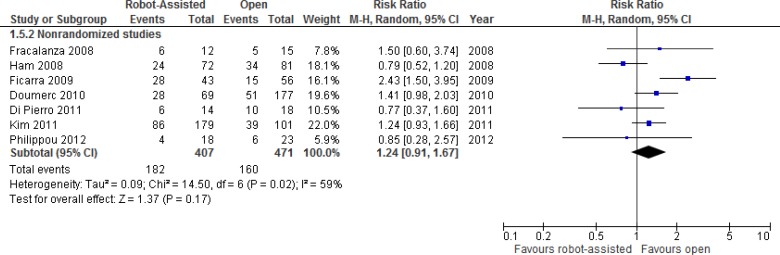
The differences in transfusion rates between robot-assisted and open radical prostatectomy are presented in Figure 2. These rates are from one RCT and 11 nonrandomized studies. While the RCT26 did not find any significant differences between groups, the nonrandomized studies showed decreases in transfusion rates with robot-assisted radical prostatectomy.
Figure 2: Transfusion Rates for Robot-Assisted Versus Open Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel.
Sources: Data from Yaxley et al, 2016,26 Farnham et al, 2006,39 Wood et al, 2007,56 Fracalanza et al, 2008,42 Ficarra et al, 2009,40 Kordan et al, 2010,47 Doumerc et al, 2010,38 Hong et al, 2010,45 Carlsson et al, 2010,36 Breyer et al, 2010,35 Di Pierro et al, 2011,37 and Philippou et al, 2012.54
In the RCT, clinicians used a cell saver device, which recovers a patient's lost blood during the surgery and reinfuses it back into the same patient (known as intraoperative blood salvage, or autologous blood transfusion). Cell savers therefore reduce the requirement for non-autologous (i.e., blood that is not from the same patient) blood transfusions. The RCT found no intraoperative non-autologous blood transfusions in either group, and no difference in postoperative non-autologous blood transfusions between groups (P = .12).
Low hematocrit levels are triggers for transfusion. Farnham et al39 found that differences in the discharge hematocrit (36.8% for robot-assisted versus 32.8% for open, P < .001) and the mean perioperative change in hematocrit (8.0% decrease for robot-assisted versus 10.7% decrease for open, P < .001) were significant between robot-assisted and open groups. Kordan found similar results: a change in hematocrit of 10% (8–12%) in open and 7% (6–9.5%) in robot-assisted surgery (P < .001).47 However, in the study by Philippou et al,54 the decrease in postoperative hematocrit was lower in the open group (4.19 ± 2.21%) than in the robot-assisted group (8.51 ± 3.67%, P < .001).
Kordan et al47 further investigated whether robot-assisted radical prostatectomy was associated with a lower transfusion rate than the open approach. On univariate analysis, the robot-assisted approach, estimated blood loss ≥ 500 mL, and a change in hematocrit ≥ 10% were the only significant predictors of transfusion. In an exploratory multivariate model (limited to only 21 transfusion events), they found that robot-assisted radical prostatectomy was the only significant predictor of reduced need for transfusion (odds ratio [OR] 0.23, 95% CI 0.09–0.58, P = .002). The likelihood of transfusions was not associated with surgeon for the group as a whole, and there was a trend of decreasing likelihood of transfusion with the advancing calendar year because of the increasing volume of robot-assisted radical prostatectomies.
Table 6 presents the GRADE evidence profile for transfusion rates.
Table 6:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Transfusion Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 11 non-RCTs | Serious limitations (−1)b | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences. Studies had differences in postoperative hematocrit thresholds that would trigger the requirement of transfusion.
GRADE starts at low for nonrandomized studies. Differences between groups in patient characteristics and surgeon experience.
Duration of Indwelling Catheterization
Table A8 shows the results for the one RCT and seven nonrandomized studies reporting on the duration of indwelling catheterization for robot-assisted versus open radical prostatectomy. Generally, shorter durations were found for the robot-assisted radical prostatectomy group, with a range of 3 to 12 days for robot-assisted surgery and 6 to 11 days for open surgery. The wide range of results is likely because of different hospital protocols and procedures for removal time.
Di Pierro et al37 measured catheter-free rates at days 10 and 15, and while the rates favoured the robot-assisted group, no P values were reported to show significance. This study was one of only two studies that evaluated prolonged catheterization (> 10 days after surgery), with no significant difference found between robot-assisted and open procedures. Doumerc et al38 also noted prolonged catheterization in the open group; however, they did not report values for significance.
In contrast to the nonrandomized studies reporting significantly shorter durations for indwelling catheterization duration for robot-assisted radical prostatectomy, in the recently published RCT there was no difference between groups.26
Table 7 presents the GRADE evidence profile for indwelling catheterization.
Table 7:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Indwelling Catheterization Duration
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 7 non-RCTs | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
GRADE starts at low for nonrandomized studies. Differences in baseline patient characteristics may impact outcome.
Outcome is impacted by differences in hospital procedures or protocols for actual duration.
Rates of Hospital Readmission
One RCT and two nonrandomized studies examined rates of hospital readmission (Table A9).
Nelson et al52 found no significant differences between robot-assisted and open approaches for readmission or unscheduled clinical or emergency room visits. The authors found that readmission rates or unscheduled hospital visits are necessary in a small percentage of patients treated with an early discharge program.
The most common cause for readmission of patients who had undergone robot-assisted radical prostatectomy was ileus (obstruction of movement in the intestine), at 3.2%.52 Other causes of readmission were port hernia, rectal injury, postoperative hemorrhage, clot retention, and urinary tract infection. For unscheduled visits, the most common causes were clot retention (1.5%), urinary leakage/urinoma, and other unspecified reasons.
For the open surgery group, the most common cause of readmission was also ileus, at 2.5%.52 Deep vein thrombosis, lymphocele, and fever were the other causes. For unscheduled visits, the most common causes were wound infection (1.4%), ileus (1.6%), and unspecified other reasons.
Yaxley et al26 and Wallerstedt et al32 similarly found no significant differences between groups at 3 months. Wallerstedt et al32 investigated predictors of patient-reported readmission to hospital. Factors that significantly increased the risk of readmission included the preoperative PSA level, lymph node dissection, prostate weight, clinical tumour stage, tumour stage of the prostatectomy specimen, the Gleason score of the pathology specimen, and a history of mental disorder. The most common causes of readmission for robot-assisted radical prostatectomy were infection (2%) and surgical reasons (3%). In the open group, the most common causes were infection (1.3%), cardiovascular issues (1.2%), and surgical reasons (1.9%).
Table 8 presents the GRADE evidence profile for rates of hospital readmission.
Table 8:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Hospital Readmission Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)b | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
Nonrandomized studies start at low GRADE. Readmission rates may be impacted by differences in baseline patient characteristics and other comorbid conditions.
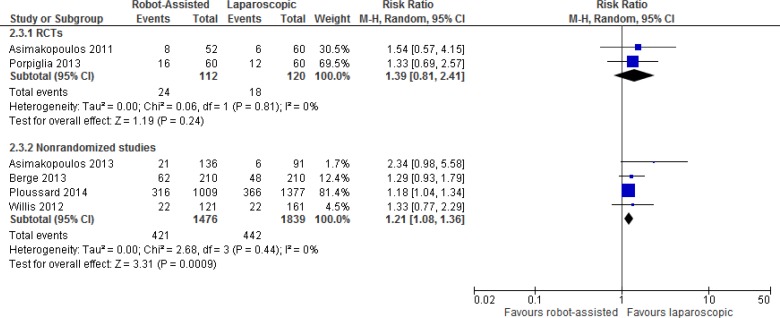
Complication Rates
Table A10 presents the reported complications from one RCT and 14 nonrandomized studies. Common complications were nerve or rectal injury, ileus, bladder neck contracture (narrowing of the connection between the bladder and urethra owing to scarring), and anastomotic stricture (a narrowing of the anastomotic suture line). When grouped using the Clavien or Dindo classification of surgical complications, most complications were minor (grades I and II). Mortality was rare for both procedures.
In general, the nonrandomized studies showed significant reductions in overall rates of complications for robot-assisted radical prostatectomy. However, the RCT found borderline nonsignificant differences for overall complication rates between groups (P = .052).26
Carlsson et al36 specifically examined complications in robot-assisted and open radical prostatectomy. Clavien grade IIIb to grade V complications were more common in the open group (12.9%) than in the robot-assisted group (3.7%). Among late postoperative complications (> 30 days to 15 months), Carlsson et al found that bladder neck contractures were treated more often in the open group (4.5%) than in the robot-assisted group (0.2%) (P < .001). Also, more patients needed surgery because of postoperative incontinence in the open group (2.2% vs. 0.5% robot-assisted, P < .01).
Breyer et al35 examined bladder neck contracture and found that patients were diagnosed at a median of 2.5 months (range 1–13.5 months) in the open group and 6 months (3–24 months) in the robot-assisted group. Life table analysis showed that the rates for being free of bladder neck contracture at 18 months were 97% for open and 99% for robot-assisted surgery (log-rank P = .13). The authors performed a Cox proportional hazards regression analysis of patients who underwent open radical prostatectomy, covarying for year of surgery, age, biopsy Gleason grade, and PSA level at diagnosis. They found that earlier year of surgery (hazard ratio 0.51, 95% CI 0.34–0.79) and higher PSA level at diagnosis (hazard ratio 1.03, 95% CI 1.01–1.06) were significantly associated with bladder neck contracture. In the robot-assisted group, none of the covariates were significantly associated with bladder neck contracture.
Hong et al45 evaluated the incidence of intraoperative venous gas embolism and found a significantly increased rate in the open radical prostatectomy group.
Table 9 presents the GRADE evidence profile for complication rates.
Table 9:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Complication Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 14 non-RCTs | Serious limitations (−1)b | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Few studies powered to detect differences, in particular between individual complications.
Nonrandomized studies start at low GRADE. Differences in baseline patient characteristics between groups may impact types of complication and rates.
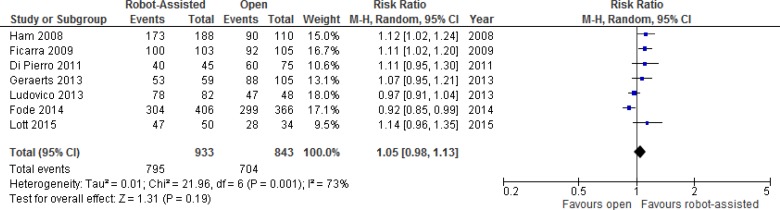
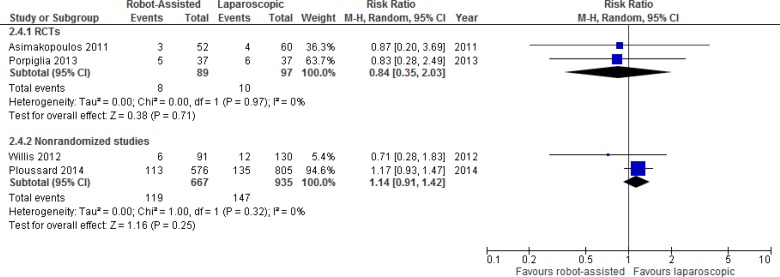
Urinary Function
One RCT and 12 nonrandomized studies compared results for urinary function after robot-assisted versus open radical prostatectomy. We performed a meta-analysis for unadjusted dichotomous continence outcomes (e.g., continent or incontinent) (Figure 3). Pooled data showed large statistical heterogeneity (I2 = 73%). Definitions for dichotomous outcomes were primarily the use of pads or absence of leakage, with definitions varying slightly between studies, from strictly pad free (0 pads) to 0 or 1 pad per day.
Figure 3: Urinary Continence Outcomes for Robot-Assisted Versus Open Radical Prostatectomy at 12 Months.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel.
Sources: Data from Ham et al, 2008,44 Ficarra et al, 2009,40 Di Pierro et al, 2011,37 Ludovico et al, 2013,50 Geraerts et al, 2013,31 Fode et al, 2014,41 and Lott et al, 2015.49
Additional details of urinary outcomes are presented in Table A11. The RCT found no significant differences in urinary continence at 3 months.26 Four studies also reported on time to continence, with three studies showing significantly faster continence recovery with robot-assisted radical prostatectomy.
Four nonrandomized studies adjusted for potential confounders. After correction for covariates, Geraerts et al31 found the difference in time to continence was significant (hazard ratio 1.522, 95% CI 1.027–2.255, P = .036). In addition, younger men, men with positive surgical margins, and men without preoperative incontinence achieved continence sooner. A comparison of time to continence between groups with a sufficient number of patients (intermediate risk and/or bilateral nerve-sparing) still showed a faster return of continence after robot-assisted radical prostatectomy, but the effect was smaller and nonsignificant (hazard ratio > 1.2, P > .05). The robot-assisted group also had significantly fewer voiding symptoms than the open group at 1 month (P = .01) and 3 months (P = .04) after surgery. At 12 months, patients in the open group were more physically limited and took more precautions to avoid a urine leak than did those in the robot-assisted group (P = .01 and P = .01, respectively).
In contrast, in a study by Haglind et al, at 12 months, 366 men (21.3%) who underwent robot-assisted radical prostatectomy were incontinent, as were 144 men (20.2%) in the open group.43 When adjusting for confounders, the results were not significant (OR 1.08, 95% CI 0.87–1.34).
Davison et al19 also found that urinary domain scores in the Expanded Prostate Cancer Index Composite (EPIC) did not differ significantly between the two groups at baseline, 6 months, or 12 months postsurgery after adjustment.
Similarly, Fode et al41 found no difference in the proportion of patients in the robot-assisted or open group who had undergone surgical treatment for incontinence (P = .4). The authors noted, however, that patients who had undergone open radical prostatectomy had an increased chance of subjective continence at 12 months (OR 2.6, P = .01), with the opposite effect seen in the robot-assisted group. On multivariate analysis, the type of surgery remained an independent predictor of regaining continence (subjective assessment) at 12 months (P = .01). Significant predictors of subjective continence were a low preoperative Danish Prostatic Symptom Score (P = .004), younger age at surgery (P = .02), and unilateral or bilateral nerve-sparing surgery compared with non-nerve-sparing surgery (P = .03).
Tables 10 and 11 present the GRADE evidence profiles for urinary function at 3 and 12 months, respectively.
Table 10:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Urinary Function at 3 Months
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | Serious limitation (−1)a | No serious limitations | No serious limitations | No serious limitations | Undetected | None | ⊕⊕⊕ Moderate |
| 12 non-RCTs | Serious limitations (−1)b | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)c | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
No intention-to-treat analysis. Interim 3-month data analysis.
Nonrandomized studies start at low GRADE. Differences in baseline patient, cancer, or surgeon characteristics may impact continence outcomes. Most studies did not adjust for possible confounding.
Use of nonvalidated and possibly subjective outcome measures in some studies for dichotomous urinary outcomes. Nonstandardized reporting and different tools used makes it difficult to directly compare studies.
Table 11:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Urinary Function at 12 Months
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 12 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)c | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Nonrandomized studies start at low GRADE. Differences in baseline patient, cancer, or surgeon characteristics between groups may impact urinary function.
Even when adjusted for differences in baseline characteristics between groups, results were inconsistent.
Use of nonvalidated or nonstandardized assessments for dichotomous outcomes.
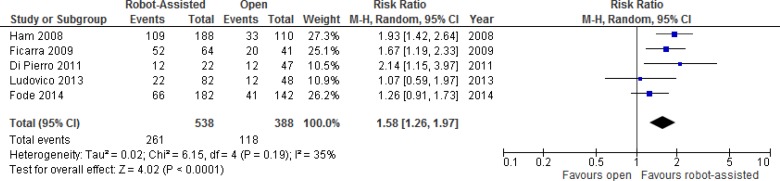
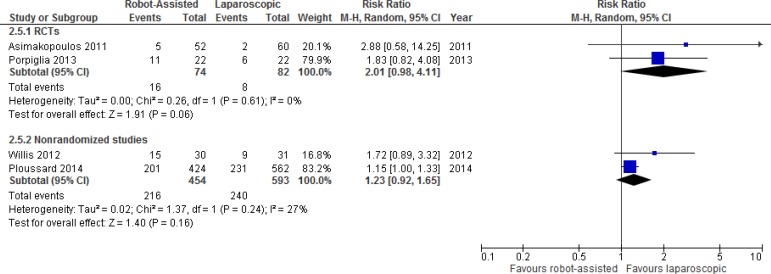
Erectile Function
One RCT and 11 nonrandomized studies reported results comparing erectile function after robot-assisted versus open radical prostatectomy. We performed a meta-analysis for unadjusted dichotomous erectile function outcomes (e.g., potent or impotent) (Figure 4). Outcome results were primarily determined by a single question asking patients if they experienced erections sufficiently firm for sexual intercourse.
Figure 4: Erectile Function Outcomes for Robot-Assisted Versus Open Radical Prostatectomy at 12 Months.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel.
Sources: Data from Ham et al, 2008,44 Ficarra et al, 2009,40 Di Pierro et al, 2011,37 Ludovico et al, 2013,50 and Fode et al, 2014.41
Table A12 presents additional details for erectile function outcomes. The RCT found no significant differences in erectile function at 3 months.26 One study showed that time to potency was significantly less in the robot-assisted group.46
Considering only patients undergoing bilateral nerve-sparing surgery, at ≥ 12 months of follow-up Ficarra et al40 found that 49% in the open group and 81% in the robot-assisted group were potent (P < .001). Similarly, when evaluating only patients aged < 65 years and with a Charlson score of ≤ 2, they found that 58% in the open group and 84% in the robot-assisted group were potent (P < .01).40
Di Pierro et al37 considered patients who were potent without phosphodiesterase type 5 (PDE5) inhibitors before robot-assisted or open radical prostatectomy. They found recovery of erectile function with or without PDE5 inhibitors, respectively, was achieved in 25% and 68% at 3 months postoperatively (P = .009) and in 26% and 55% at 12 months postoperatively (P = .009).
When adjusting for nerve-sparing status and the use of PDE5 inhibitors, Davison et al19 found significant within-subject differences in mean scores across time for both the Expanded Prostate Cancer Index Composite sexual summary and sexual bother domains (P < .001). (“Sexual bother” is the level of interference or annoyance caused by limitations in sexual function.) However, the sexual summary and sexual bother domains were not significantly different between groups at 6 or 12 months. Patients who had either one or two nerves spared reported an overall higher mean sexual quality of life score than those with no (or unknown) nerves spared. Similar results were seen for patients who used a PDE5 inhibitor versus patients with no use of PDE5 inhibitors.
Kim et al46 evaluated factors impacting functional outcomes. In a multivariate analysis, younger age and a longer preoperative membranous urethral length (as seen on magnetic resonance imaging of the prostate) were significant independent factors for the prognosis of continence recovery. Younger age, surgical method (robot-assisted vs. open), and higher preoperative serum testosterone were independent prognostic factors for potency recovery. The extent of nerve sparing (unilateral vs. bilateral) did not make difference to potency outcome. When the preoperative serum testosterone level was ≥ 3.9 ng/mL, potency recovery at 12 months could be expected (OR 2.1, P < .001).
Fode et al41 found on univariate analysis that there was no statistically significant difference between groups in the proportions of potent patients at 3 and 12 months (P = .08 and P = .16, respectively). However, significantly more patients in the robot-assisted group had regained potency at 6 months (P = .02). There was no difference in the use of PDE5 inhibitors or other erection aids in sexually active patients at any point during follow-up. Multivariate analyses showed no difference between groups for potency rates among sexually active patients at 3 months (P = .16), 6 months (P = .11), or 12 months (P = .7). A high preoperative score on the International Index of Erectile Function Questionnaire (IIEF-5; P = .001), younger age at surgery (P = .03), and nerve sparing (P < .001) were all independent predictors of regained potency at 12 months.
In another study, Geraerts et al31 found that after adjustment the odds ratio for any erectile dysfunction was 0.80 (95% CI 0.64–1.00). Classification by the various definitions of “erectile dysfunction” did not substantially affect the odds ratios. When adjustments were made for preoperative clinical tumour characteristics, the odds ratio was 0.74 (95% CI 0.59–0.95); for neurovascular preservation, the odds ratio was 0.75 (95% CI 0.58–0.96); and for lymph node dissection, the odds ratio was 0.78 (95% CI 0.61–1.00).
Bier et al34 found there were no significant differences in robot-assisted or open groups for time between surgery and first postoperative sexual activity (P = .63), or for time between surgery and satisfaction with sexual life (P = .85).
Tables 12 and 13 present the GRADE evidence profiles for erectile function at 3 and 12 months, respectively.
Table 12:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Erectile Function at 3 Months
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | Serious limitations (−1)a | No serious limitations | No serious limitations | No serious limitations | Undetected | None | ⊕⊕⊕ Moderate |
| 11 non-RCTs | Serious limitations (−1)b | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)c | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
No intention-to-treat analysis. Interim 3-month data analysis.
Nonrandomized studies start at low GRADE. Differences in baseline patient, cancer, or surgeon characteristics between groups may impact erectile function. Some studies did not adjust for possible confounding. Even when adjusted, results were inconsistent, showing both significant and nonsignificant results.
Use of nonvalidated or nonstandardized assessments for dichotomous outcomes.
Table 13:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Erectile Function at 12 Months
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 11 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)c | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Nonrandomized studies start at low GRADE. Differences in baseline patient, cancer, or surgeon characteristics between groups may impact erectile function.
Even when adjusted for differences in baseline characteristics between groups, results were inconsistent.
Use of nonvalidated or nonstandardized assessments for dichotomous outcomes.
Health-Related Quality of Life
One RCT and two nonrandomized studies measured generic health-related quality of life (Table A13).
Miller et al51 found that patients in the robot-assisted radical prostatectomy group demonstrated significantly greater physical quality of life compared with those in the open group (P < .001). However, mental quality of life was not significantly related to operative condition (P < .13).
The RCT found no significant differences between groups at 6 or 12 weeks for the Short-Form 36 Health Survey mental function, Expanded Prostate Cancer Index Composite bowel domain, or Revised Impact of Events Scale.26 Minor differences were found between groups for the Short-Form 36 Health Survey at 6 weeks and the Hospital and Depression Scale (HADS) at 12 weeks.
Table 14 presents the GRADE evidence profile for health-related quality of life.
Table 14:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Health-Related Quality of Life
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)b | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
Nonrandomized studies start at low GRADE. Differences in baseline patient characteristics between groups may impact recovery and thus health-related quality of life scores.
Pain
All three studies (one RCT and two nonrandomized studies) examining pain after robot-assisted and open radical prostatectomy found no difference in pain between groups at the latest time point reported (Table A14). However, the RCT noted significantly reduced short-term pain favouring robot-assisted radical prostatectomy at 24 hours and 1 week postsurgery. These results were for pain during activities and when experiencing worst pain, but not while resting.26
Wallerstedt et al32 examined pain at various parts of the body (operation wound, lower abdomen, upper abdomen); however, they found no significant difference between groups at the 3 month follow-up.
Wood et al56 examined pain and discomfort at 2 and 6 weeks postsurgery (overall pain, and also pain in the abdomen, bladder, and flank). They found no significant differences between the robot-assisted and the open groups for overall and site-specific pain.
Table 15 presents the GRADE evidence profile for pain.
Table 15:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Pain
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)b | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences. Individual patient factors and additional medication may affect pain tolerance.
Nonrandomized studies start at low GRADE. Differences in patient characteristics may affect outcome.
Time to Mobilization or Return to Work or Activity
One RCT and three nonrandomized studies evaluated time to mobilization (e.g., movement or limited physical activity), or return to work or (full or normal) activity.
The RCT found no significant differences in return-to-work outcomes at 3 months postsurgery among those who were employed full or part time.26 Similarly, investigators found no significant difference between groups in length of time away from work (robot-assisted group: mean 42.71 days, 95% CI 30.98–54.45; open group: mean 42.71 days, 95% CI 41.09–53.30; P = .49).
Bier et al34 found patients felt affected in their work for a median time of 2 months for both robot-assisted and open groups (P = .67, range < 1 to > 8 months). The median time to pursue their hobbies again without feeling restricted was 3 months after surgery in both groups (P = .71, range < 1 to > 8 months), and to completely pursue work and hobbies again was 4 months (P = .73, range < 1 to > 8 months).
Wood et al56 similarly did not find significant differences for:
Median time to normal activity: robot-assisted group, 9 days (range 1–30 days), versus open group, 7 days (range 7–45 days); P = .57
Median time to 100% activity: robot-assisted group, 21 days (range 6–52), versus open group, 28 days (range 7–45 days); P = .95
Time to driving: robot-assisted group, 13 days (range 4–44), versus open group, 14 days (range 1–31); P = .15
In comparison, Fracalanza et al42 found a significant difference for average time to mobilization and resumption of oral feeding favouring robot-assisted radical prostatectomy (robot-assisted, 1 day, versus open, 1.2 days, P < .001).
Table 16 presents the GRADE evidence profile for time to mobilization or return to work or activity.
Table 16:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Time to Mobilization or Return to Work or Activity
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 3 non-RCTs | Serious limitations (−1)b | Serious limitations (−1)c | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences. No standardized method for measurement.
Nonrandomized studies start at low GRADE. Differences in baseline patient characteristics between robot-assisted and open prostatectomy groups may impact time to mobilization or return to work or activity.
One study found a significant difference favouring robot-assisted radical prostatectomy for time to mobilization.
Rates of Positive Surgical Margins
One RCT and 15 nonrandomized studies reported on rates of positive surgical margins.
In the RCT, no difference was found in the overall rates of positive surgical margins for robot-assisted versus open prostatectomy.26
The results for unadjusted rates, in the RCT and the nonrandomized studies, are presented below for all patients within studies (Figure 5) and, where available, for patients with stage pT2 (Figure 6) or pT3 cancer (Figure 7). When we analyzed results by the overall positive surgical margin rates, we found considerable heterogeneity (I2 = 78%). As a result, we have not shown summary estimates. When we analyzed subgroups pT2 and pT3, the heterogeneity reduced to an I2 of 40% and 59%, respectively, likely owing to controlling for tumour characteristics. There was a general trend in favour of a nonsignificant reduction in positive surgical margins for robot-assisted radical prostatectomy in patients with pT2 cancer. The opposite was seen in patients with pT3 cancer, where a nonsignificant trend favoured open radical prostatectomy.
Figure 5: Overall Positive Surgical Margin Rates for Robot-Assisted Versus Open Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel.
Sources: Data from Yaxley et al, 2016,26 Ham et al, 2008,44 Ficarra et al, 2009,40 Kordan et al, 2010,47 Breyer et al, 2010,35 Doumerc et al, 2010,38 Di Pierro et al, 2011,37 Kim et al, 2011,46 Philippou et al, 2012,54 Ludovico et al, 2013,50 Geraerts et al, 2013,31 Thompson et al, 2013,55 Fode et al, 2014,41 Lott et al, 2015,49 and Haglind et al, 2015.43
Figure 6: Positive Surgical Margin Rates in Cancer Stage pT2 for Robot-Assisted Versus Open Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel.
Sources: Data from Fracalanza et al, 2008,42 Ham et al, 2008,44 Ficarra et al, 2009,40 Doumerc et al, 2010,38 Kim et al, 2011,46 Di Pierro et al, 2011,37 Philippou et al, 2012,54 Ludovico et al, 2013,50 Thompson et al, 2013,55 and Fode et al, 2014.41
Figure 7: Positive Surgical Margin Rates in Cancer Stage pT3 for Robot-Assisted Versus Open Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel.
Sources: Data from Fracalanza et al, 2008,42 Ham et al, 2008,44 Ficarra et al, 2009,40 Doumerc et al, 2010,38 Di Pierro et al, 2011,37 Kim et al, 2011,46 Philippou et al, 2012.54
Doumerc et al38 investigated the effect of learning curve (surgeon experience). They found that the overall rate of positive surgical margins for robot-assisted surgery declined as surgeon experience increased. A learning curve effect was also noted in pT2 cancers. However, rates between pT2 and pT3 remained statistically different at the end of the trial. The authors noted this may have been because of the low number of cases available for analysis, or because the learning curve had not yet been overcome.
Fode et al,41 using univariate analyses, also examined the learning curve. They found that compared with robot-assisted prostatectomy, open radical prostatectomy had a relative risk of positive surgical margins of 1.56 (95% CI 1.23–1.99, P < .001). However, on multivariate analyses, with stratification for tumour characteristics, patient characteristics, and the surgeon, the type of surgery did not affect the surgical margins (P = .96). A large tumour volume (P < .001), pathological tumour stage (P = .005), and a small prostate volume (P = .04) were independent predictors of positive surgical margins.
Thompson et al55 also examined the learning curve, and found that in T2 disease, the odds of a positive surgical margin were 6.19 times higher (95% CI, 1.20–31.80) for robot-assisted than open radical prostatectomy. However, this lowered after 108 robot-assisted radical prostatectomies had been performed, and plateaued at around 400 to 500 procedures. At the end of the study, after 866 cases, the odds of a positive surgical margin were 55% lower for the robot-assisted surgery compared with the open approach (OR 0.45, 95% CI 0.22–0.92).
Table 17 presents the GRADE evidence profile for positive surgical margin rates.
Table 17:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Positive Surgical Margin Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | Serious limitations (−1)a | Serious limitations (−1)b | Undetected | None | ⊕⊕ Low |
| 15 non-RCTs | Serious limitations (−1)c | Serious limitations (−1)d | Serious limitations (−1)a | Serious limitations (−1)b | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Surrogate outcome for patient-important outcome of cancer-free survival or cancer-specific mortality. Impact of potential differences in pathological assessment of positive surgical margins.
Not powered to detect differences.
Nonrandomized studies start at low GRADE. Differences in baseline patient, tumour, or surgeon characteristics between groups may affect outcome.
Inconsistency in results in both direction of effect and significance between studies.
Biochemical Recurrence-Free Rates
Three nonrandomized studies reported biochemical recurrence-free rates (the absence of elevated PSA levels after radical prostatectomy; Table A15). Di Pierro et al37 found no difference between biochemical recurrence-free rates at 3 or 12 months, as did Philippou et al54 at 12 months. Meanwhile, Breyer et al35 found significant differences between groups at 3 years, favouring open radical prostatectomy.
No studies reported information on cancer-free survival rates.
The RCT26 will be evaluating biochemical recurrence rates between groups; however, longer-term results that include biochemical recurrence have yet to be published.
Table 18 presents the GRADE evidence profile for biochemical recurrence-free rates.
Table 18:
GRADE Evidence Profile for Robot-Assisted Versus Open Radical Prostatectomy for Biochemical Recurrence-Free Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 3 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Nonrandomized studies start at low GRADE. Unadjusted differences in baseline patient, tumour, or surgeon characteristics may affect outcome.
One study showed significant results favouring the open group, while other two studies showed no difference.
Surrogate (substitute or proxy) outcome for patient-important outcome of cancer-free survival or cancer-specific mortality. There is an issue with the biological relevancy of the outcome, and the ability to detect true prostate cancer recurrence in patients.
Unpowered to detect differences.
Robot-Assisted Versus Laparoscopic Radical Prostatectomy
We found two RCTs29,30 and five prospective comparative nonrandomized studies57–61 comparing robot-assisted versus laparoscopic radical prostatectomy. An additional study33 included a laparoscopic comparison in addition to robot-assisted versus open radical prostatectomy. No prospective comparative studies were found that examined readmission rate, pain, or return to mobilization or work.
Asimakopoulos et al57 authored the only study that examined the pentafecta of outcomes for (bilateral nerve-sparing) radical prostatectomy: potency, urinary continence, absence of perioperative complications, negative surgical margins, and no biochemical recurrence. Recently proposed by Patel et al,62 the pentafecta builds upon a trifecta of outcomes (potency, continence, and biochemical recurrence-free progression after surgery) and is a composite outcome measure for radical prostatectomy.
Of the 140 patients who did not achieve pentafecta, 90 (64%) missed a single parameter, with a significant difference between laparoscopic (80%) and robot-assisted (53%) groups (P = .007).57 Through regression analysis, Asimakopoulos et al57 found these significant independent factors were associated with pentafecta: lower age (OR 0.94, 95% CI 0.9–1.0, P = .04), lower pathological stage (OR 0.24, 95% CI 0.1–0.7, P = .006), and robot-assisted radical prostatectomy (OR 1.9, 95% CI 1.0–3.5, P = .04).
Operative Time
Mean operative time between robot-assisted and laparoscopic radical prostatectomy was reported in two RCTs and four nonrandomized studies (Figure 8).
Figure 8: Mean Operative Time for Robot-Assisted Versus Laparoscopic Radical Prostatectomy.
Abbreviations: CI, confidence interval; IV, inverse variance; SD, standard deviation.
Sources: Data from Porpiglia et al, 2013,29 Ploussard et al, 2009,59 Willis et al, 2012,61 and Berge et al, 2013.58
In their RCT, Asimakopoulos et al30 found no difference in mean operative time between the two surgeries (actual results and P value not reported). The RCT by Porpiglia et al29 also found no difference.
A nonrandomized study reported reduced mean operative time for robot-assisted surgery, at 128.9 minutes versus 175.5 minutes for laparoscopic surgery (P < .001).60 However, the results provided could not be meta-analyzed.60 Operative time was less in the other nonrandomized studies, favouring robot-assisted radical prostatectomy.58,59,61
A nonrandomized study by Ploussard et al59 examined factors impacting operating room time. The authors found that while total operative time was not significantly different between groups, compared with laparoscopic radical prostatectomy:
The installation step was longer for robot-assisted surgery (33.2 ± 15.8 minutes vs. 24.0 ± 12.1 minutes, P < .01)
Actual skin-to-skin time was reduced for robot-assisted surgery (145.6 ± 34.4 minutes vs. 164.7 ± 49.1 minutes, P < .01)
If lymphadenectomy was performed, this increased the average operative time by 15 minutes for the laparoscopic group (P = .1) and 30 minutes for the robot-assisted group (P = .01).59
Table 19 presents the GRADE evidence profile for operative time.
Table 19:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Operative Time
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 2RCTs | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 4 non-RCTs | Serious limitations (−1)b | Serious limitations (−1)c | Serious limitations (−1)d | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
GRADE starts at low for nonrandomized studies. Differences in baseline patient characteristics and surgeon experience between groups may impact operative time.
Significant and nonsignificant reductions favouring the robot-assisted group.
Generalizability concerns because of differences in hospital discharge pathways.
Length of Hospital Stay
Length of hospital stay was reported in one RCT and two nonrandomized studies. No difference was found in any of the studies (Figure 9). The lengths of hospital stay ranged from 2.2 to 4.6 days for the robot-assisted group, and 2.1 to 4.8 days for the laparoscopic group.30,59,61,63
Figure 9: Length of Hospital Stay for Robot-Assisted Versus Laparoscopic Radical Prostatectomy.
Abbreviations: CI, confidence interval; IV, inverse variance; SD, standard deviation.
Sources: Data from Porpiglia et al, 2013,29 Ploussard et al, 2009,59 and Willis et al, 2012.61
Table 20 presents the GRADE evidence profile for length of hospital stay.
Table 20:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Length of Hospital Stay
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
GRADE starts at low for nonrandomized studies. Differences in baseline patient characteristics and patient factors potentially impact length of hospital stay.
Generalizability concerns for results from centres where length of stay is not reflective of average Ontario times.
Estimated Blood Loss
One RCT and four nonrandomized studies reported estimated blood loss (Table A16). The RCT by Porpiglia et al29 found no significant differences between groups, whereas the nonrandomized studies found a significant reduction favouring the robot-assisted group.58,59,61 However, there was large variability within groups, ranging from an average blood loss of 148 to 469 mL in the robot-assisted group, to 203 to 889 mL in the laparoscopic group.
Table 21 presents the GRADE evidence profile for estimated blood loss.
Table 21:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Estimated Blood Loss
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 4 non-RCTs | Serious limitations (−1)b | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences. Difficulties in accurately measuring blood loss.
GRADE starts at low for nonrandomized studies. Differences in baseline patient characteristics and surgeon experience may impact estimated blood loss.
Transfusion Rates
One RCT and three nonrandomized studies examined transfusion rate. Only one large study found reduced transfusion rates favouring laparoscopic radical prostatectomy60 (Figure 10). In contrast with open radical prostatectomy, both robot-assisted and laparoscopic radical prostatectomy are minimally invasive procedures conducted through keyhole incisions. This likely explains the similarities in outcomes.
Figure 10: Transfusion Rates for Robot-Assisted Versus Laparoscopic Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel.
Sources: Data from Asimakopoulos et al, 2011,30 Ploussard et al, 2009,59 Berge et al, 2013,58 and Ploussard et al, 2014.60
Table 22 presents the GRADE evidence profile for transfusion rate.
Table 22:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Transfusion Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 3 non-RCTs | Serious limitations (−1)b | Serious limitations (−1)c | Serious limitations (−1)d | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
GRADE starts at low for nonrandomized studies.
One large study showed a significant reduction in transfusion rate favouring the robot-assisted group.
Potential differences in postoperative hematocrit levels that trigger transfusion.
Duration of Indwelling Catheterization
The duration of indwelling catheterization was reported in two RCTs and one nonrandomized study. All found nonsignificant differences between robot-assisted and laparoscopic radical prostatectomy (Figure 11).
Figure 11: Indwelling Catheterization Duration for Robot-Assisted Versus Laparoscopic Radical Prostatectomy.
Abbreviations: CI, confidence interval; IV, inverse variance; SD, standard deviation.
Sources: Data from Asimakopoulos et al, 2011,30 Porpiglia et al, 2013,29 and Ploussard et al, 2009.59
Table 23 presents the GRADE evidence profile for indwelling catheterization duration.
Table 23:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Indwelling Catheterization Duration
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 2 RCTs | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 1 non-RCT | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Not powered to detect differences.
GRADE starts at low for nonrandomized studies. Differences in baseline patient characteristics may affect duration.
Generalizability of results may be limited because of differences between health system contexts.
Complication Rates
Complication rates were reported in both RCTs and three nonrandomized studies (Table A17). None of the studies found any differences in total complication rate between the two groups, and they did not report any deaths. The most common complications were “paravesical” hematoma, urinary infection, retention, and anastomotic leakage or stenosis. Complication rates ranged from 0% to 16.6% in the robot-assisted group and 0% to 11.6% in the laparoscopic group.
Table 24 presents the GRADE evidence profile for complication rates.
Table 24:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Complication Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 2 RCTs | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕⊕⊕ Moderate |
| 3 non-RCTs | Serious limitations (−1)b | No serious limitations | No serious limitations | Serious limitations (−1)a | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Studies not powered to detect differences, in particular between individual complications.
Nonrandomized studies start at low GRADE. Differences in baseline patient characteristics between groups may impact type of complication and rates.
Urinary Function
Two RCTs and four nonrandomized studies reported results on urinary function (Table A18).
Of the RCTs, Porpiglia et al29 found results that favoured laparoscopic radical prostatectomy at all time points (3, 6, and 12 months using the Expanded Prostate Cancer Index Composite questionnaire). Step-wise regression analysis also showed that the laparoscopic approach was associated with continence at 3 months after surgery (P = .03).29 However, the other RCT showed no significant difference between groups in continence rates at 3, 6, or 12 months, or time to continence.30
Berge et al58 found no difference for urinary function at 3, 12, or 36 months using the University of California—Los Angeles Prostate Cancer Index questionnaire, even when stratifying for nerve-sparing status (no, unilateral, or bilateral nerve-sparing).
Willis et al61 also found no difference in continence rates based on the Expanded Prostate Cancer Index Composite urinary summary score or pad usage.
Ploussard et al60 found significant findings for robot-assisted radical prostatectomy at 3 months and 24 months (absence of pad use), but results were not significant at 6 months. However, in their univariable analysis, the rate of continence was significantly in favour of the robot-assisted approach at each postoperative visit (P < .001). In their multivariable analysis, the only factor independently associated with a better continence recovery was age (P = .002) at each time point. Surgical experience, nerve-sparing surgery, and surgical approach were not independent predictors for short- or long-term continence recovery. Surgical treatment for persistent incontinence was also more frequent in the laparoscopic group compared with the robot-assisted group (P < .001). Use of the Macroplastique injection, adjustable continence therapy balloon, suburethral sling, or artificial sphincter was reported respectively in 3, 10, 17, and 13 cases in the laparoscopic group versus 0, 2, 5, and 0 cases in the robot-assisted group.
Table 25 presents the GRADE evidence profile for urinary function.
Table 25:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Urinary Function
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 2 RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | No serious limitations | Undetected | None | ⊕⊕ Low |
| 4 non-RCTs | Serious limitations (−1)c | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
No intention-to-treat analysis.
Inconsistent (nonsignificant vs. significant) results.
Nonrandomized studies start at low GRADE. Differences in baseline patient, cancer, or surgeon characteristics may impact continence outcomes without adjustment.
Use of nonvalidated and possibly subjective outcome measures in some studies for dichotomous urinary outcomes. Nonstandardized reporting makes it difficult to directly compare studies.
Erectile Function
Two RCTs and four nonrandomized studies reported on erectile function, with inconsistent results (Table A19).
Asimakopoulos et al30 found significant results favouring robot-assisted radical prostatectomy for scores at 3, 6, and 12 months compared with baseline on the International Index of Erectile Function Questionnaire (IIEF-6):
Mean IIEF-6 change in score per patient
Rates of return to baseline IIEF-6 score
Rates of patients affected by severe dysfunction
The RCT by Porpiglia et al29 found significant improvement at only 12 months, not at the 3- and 6-month time points.
In comparison, Berge et al58 found no significant difference using the University of California—Los Angeles Prostate Cancer Index for sexual function. However, their analysis was limited to patients who received nerve-sparing radical prostatectomy.
Using the Expanded Prostate Cancer Index Composite, Willis et al61 found significant differences for sexual function at only 12 months, not at 3 or 6 months.
In a univariable analysis, Ploussard et al60 found that the rate for potency significantly favoured robot-assisted radical prostatectomy at each postoperative visit. This difference remained significant in a subgroup of patients undergoing bilateral nerve-sparing preservation (P < .001). In their multivariable analysis, age (P = .001), nerve-sparing surgery (P = .033, OR 3.9), and robot-assisted approach (P = .045, OR: 5.9) were significant independent predictors of potency recovery 12 months after surgery. These factors were also associated with potency at each medical visit during follow-up. Surgical experience, the surgeon, and the date of intervention were not associated with potency return. When examining potency using IIEF-5 scores, the scores were significantly better in the robot-assisted group at each time point after surgery.
According to Asimakopoulos et al,57 potency was the most difficult outcome in the pentafecta to achieve. However, the single question used to assess potency was not a validated question, although the authors reported a good correlation between the question's definition and the validated patient-derived IIEF score. The authors reported similar results for the other pentafecta outcomes (continence, absence of perioperative complications, negative surgical margins, and no biochemical recurrence) for pT3 disease.
Table 26 presents the GRADE evidence profile for erectile function.
Table 26:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Erectile Function
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 2 RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | No serious limitations | Undetected | None | ⊕⊕ Low |
| 4 non-RCTs | Serious limitations (−1)c | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
No intention-to-treat analysis.
Inconsistent (nonsignificant vs. significant) results.
Nonrandomized studies start at low GRADE. Differences in baseline patient, cancer, or surgeon characteristics may impact potency outcomes without adjustment.
Use of nonvalidated and possibly subjective outcome measures in some studies for dichotomous potency outcomes. Nonstandardized reporting within studies makes it difficult to directly compare studies.
Health-Related Quality of Life
One nonrandomized study reported on generic health-related quality of life.58 Berge et al58 used the 12-Item Short-Form Health Survey (SF-12) physical and mental component summaries through a mailed questionnaire approach. The authors found no significant difference at any time point (3, 12, and 36 months) between the mean scores for robot-assisted and laparoscopic radical prostatectomy. Similarly, there was no difference in the two groups’ scores in the University of California—Los Angeles Prostate Cancer Index bowel function and bowel bother domains.
At the 36-month follow-up, the authors found:
Better urinary function and lower preoperative comorbidity were associated with better mental health
Surgical method, sexual function score at 36 months, the status of positive surgical margins, tumour stage, preoperative PSA level, body mass index, and age were not associated with better mental health58
Table 27 presents the GRADE evidence profile for health-related quality of life.
Table 27:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Health-Related Quality of Life
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 1 non-RCT | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)b | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Nonrandomized studies start at low GRADE. Differences in baseline patient characteristics between groups may impact recovery and thus health-related quality of life scores.
Not powered to detect differences.
Rates of Positive Surgical Margins
Two RCTs and four nonrandomized studies reported on rates of positive surgical margins. Only one study showed lower positive surgical margin rates for laparoscopic radical prostatectomy60; the others found no difference (Figure 12). When subgrouped by pT2 or pT3 stage, this difference disappeared (Figures 13 and 14).
Figure 12: Overall Positive Surgical Margin Rates for Robot-Assisted Versus Laparoscopic Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Sources: Data from Asimakopoulos et al, 2011,30 Porpiglia et al, 2013,29 Asimakopoulos et al, 2013,57Berge et al, 2013,58 Ploussard et al, 2014,60 and Willis et al, 2012.61
Figure 13: Positive Surgical Margin Rates in pT2 Cancer for Robot-Assisted Versus Laparoscopic Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Sources: Data from Asimakopoulos et al, 2011,30 Porpiglia et al, 2013,29 Willis et al, 2012,61and Ploussard et al, 2014.60
Figure 14: Positive Surgical Margin Rates in pT3 Cancer for Robot-Assisted Versus Laparoscopic Radical Prostatectomy.
Abbreviations: CI, confidence interval; M-H, Mantel-Haenszel; RCT, randomized controlled trial.
Sources: Data from Asimakopoulos et al, 2011,30 Porpiglia et al, 2013,29 Willis et al, 2012,61and Ploussard et al, 2014.60
Asimakopoulos et al30 found no difference in the location of focal positive surgical margins (P = .59).
Another study found the presence of locally advanced disease exposed patients to a statistically higher risk of positive surgical margin (9.7% for pT2 vs. 25% for pT3 cancer, P = .01).57 The authors also found that a positive surgical margin was significantly related to biochemical recurrence both in the entire cohort (P = .003) and in the robot-assisted cases (P = .017).57
Berge et al58 reported no differences in positive surgical margins for pT2 and pT3 groups between nerve- and non-nerve-sparing groups for each technique.
Ploussard et al60 found the following:
Classic prognostic factors—PSA, tumour stage, and Gleason score—were significantly associated with positive surgical margins
Prostate volume was inversely correlated with a risk for positive surgical margins (P = .004)
In multivariable analysis, robot-assisted radical prostatectomy was not associated with an increased positive surgical margin rate
Positive surgical margin rate was significantly reduced for the robot-assisted approach in pT2 cancers, an independent factor associated with better oncologic control of margins in organ-confined disease (P = .030, OR 0.396)
In pT3 cancers, the type of surgical approach did not affect the rate of surgical margins in multivariable analysis (P = .619)
Only the PSA level and surgical experience were independent predictors of positive surgical margins in pT3 cancers (P < .001 and P < .001, respectively)
Table 28 presents the GRADE evidence profile for positive surgical margin rates.
Table 28:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Positive Surgical Margin Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 2 RCTs | No serious limitations | No serious limitations | Serious limitations (−1)a | Serious limitations (−1)b | Undetected | None | ⊕⊕ Low |
| 4 non-RCTs | Serious limitations (−1)c | Serious limitations (−1)d | Serious limitations (−1)a | Serious limitations (−1)b | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Surrogate outcome for patient-important outcome of cancer-free survival or cancer-specific mortality. Impact of potential differences in method of pathological assessment of positive surgical margin.
Not powered to detect differences.
Nonrandomized studies start at low GRADE. Differences in baseline patient, tumour, or surgeon characteristics between groups may affect outcome.
Inconsistency in significance of results.
Biochemical Recurrence-Free Rates
Four studies (two RCTs and two nonrandomized studies) reported biochemical recurrence-free rates. All found nonsignificant differences between the two groups (Table A20).
Table 29 presents the GRADE evidence profile for biochemical recurrence-free rates.
Table 29:
GRADE Evidence Profile for Robot-Assisted Versus Laparoscopic Radical Prostatectomy for Biochemical Recurrence-Free Rates
| # Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| 2 RCTs | No serious limitations | No serious limitations | Serious limitations (−1)a | Serious limitations (−1)b | Undetected | None | ⊕⊕ Low |
| 2 non-RCTs | Serious limitations (−1)c | No serious limitations | Serious limitations (−1)a | Serious limitations (−1)b | Undetected | None | ⊕ Very low |
Abbreviations: GRADE, Grading of Recommendations Assessment, Development, and Evaluation; RCT, randomized controlled trial.
Surrogate outcome for patient-important outcome of cancer-free survival or cancer-specific mortality. There was a potential issue of the biological relevancy of the outcome in some patients, and the ability to detect true prostate cancer recurrence.
Unpowered to detect differences.
Nonrandomized studies start at low GRADE. Unadjusted differences in baseline patient or tumour characteristics may affect the outcome.
Guidelines
We also found four guidelines with recommendations on robot-assisted radical prostatectomy (Appendix 6).64–67 Two were general guidelines for prostate cancer that included recommendations on robot-assisted radical prostatectomy,65,67 while the other two were specific to robot-assisted surgery.64,66 One of the latter guidelines consisted of recommendations from the Pasadena Consensus Panel on robot-assisted radical prostatectomy.66
These guidelines indicated that each approach (open, laparoscopic, and robot-assisted) produces similar results and that surgeon experience is crucial in achieving good outcomes. They concluded that there is limited evidence to suggest the superiority of one approach over another for perioperative, functional, and oncological outcomes.64,67
Discussion
Canada has been slower than the United States to adopt robot-assisted radical prostatectomy (about 70% of radical prostatectomies are still performed using the open procedure versus less than 15% in the United States). However, the current trend is increased yearly adoption. Despite the continued adoption, studies from Canadian centres are limited and either exist in robot-assisted radical prostatectomy case series or have retrospective study designs.68–72
Four Canadian studies were found, but were excluded because of their retrospective design.68,70–72 They generally did not find differences between robot-assisted and open radical prostatectomy outcomes (select perioperative and functional outcomes).
No published studies from Ontario were found through the literature search. A 2013 unpublished report from the Institute for Clinical Evaluative Sciences (excluded based on publication status) examined preliminary provincial data comparing surgical outcomes for robot-assisted versus open or laparoscopic radical prostatectomy. It included four hospital centres with a total of 646 robot-assisted radical prostatectomy cases and 17,065 open and laparoscopic cases. While the study found reduced length of stay and complication rates favouring robot-assisted radical prostatectomy, the data were based on early robot-assisted radical prostatectomy in Ontario, and patient groups were noted to have some baseline differences.
Study Population
Many of the included prospective comparative nonrandomized studies had significant differences in baseline (presurgery) patient characteristics. The authors did not always adequately adjust for these differences when reporting outcome results. In general, when significant baseline differences occurred, they showed that patients who underwent open radical prostatectomy were typically older with more high-risk features (e.g., higher average preoperative PSA levels, higher Gleason score, more advanced clinical stage). If not properly adjusted for, this may result in selection bias since patients in the open radical prostatectomy group may naturally experience worse outcomes because of their older age and higher risk status.
While there are no absolute contraindications for robot-assisted radical prostatectomy, depending on the surgeon's level of expertise and comfort level, anatomic differences such as a large prostate, high body mass index, and previous abdominal surgery may influence the surgeon to perform open rather than robot-assisted radical prostatectomy.
In the RCTs, baseline patient and tumour characteristics were balanced by specific patient eligibility criteria (e.g., age, no other comorbid conditions, life expectancy) and randomization. In contrast, some of the nonrandomized studies did not specify any inclusion or exclusion criteria, other than including patients who were candidates to undergo radical prostatectomy.
Surgical Technique
The evolution of and differences in surgical techniques may influence outcomes.
The nerve-sparing technique is meant to preserve sexual function; however, the experts we consulted with suggested it may lead to higher positive surgical margin rates. Among some of the included studies, there were significant differences in nerve-sparing status between robot-assisted versus open procedures, with the open group using the nerve-sparing approach less often.
Not all details of the surgical technique were available in the included studies, which made exploring surgical technique difficult. When information was available, the techniques varied between studies.
A study73 has also found large heterogeneity in surgical techniques for robot-assisted radical prostatectomy.
Study Design
While we excluded retrospective studies in this review, they comprise a substantial portion of the evidence base for radical prostatectomy, in the form of comparative and noncomparative (case series) studies.
Studies of recent large administrative databases appear to show favourable outcomes for robot-assisted radical prostatectomy compared with open radical prostatectomy. These include reduced lengths of stay, blood loss, risk of positive surgical margins, use of radiation therapy, 30-day mortality, and complications.74–77 Retrospective comparative studies were excluded in this review because of potential methodological limitations and biases in the retrospective study design (such as selection bias for patients included in each surgical group). However, they typically have larger sample sizes because the historical database allows investigators to find relevant patients through various years of follow-up. Administrative databases in particular are not useful in accurately evaluating functional outcomes, but they may provide some insight on health care resource use, including readmission rates and the need for secondary procedures.
The majority of the included studies had a follow-up time of 1 year or less. This does not adequately address longer-term oncological outcomes between different radical prostatectomy approaches. Longer follow-up is also required for functional outcomes, as it has been suggested that continence and erectile function may continue to improve for 2 to 3 years postsurgery.78,79 We also note that currently the highest level of evidence for robot-assisted versus open radical prostatectomy is from an RCT26 that has only short term outcomes (3-month interim results). Therefore, currently there are no reported long-term outcomes (1 year or longer) available from an RCT.
Two RCTs were terminated because of slow recruitment and low enrolment.80,81 One with low recruitment aimed to evaluate robot-assisted versus open and laparoscopic radical prostatectomy. Important reasons for failure to recruit patients included these82:
Surgeons who perform robot-assisted radical prostatectomy did not feel they could present all surgical options with equipoise (i.e., they did not feel all options are equally effective)
There was a market-led bias for patients seeking specific surgeons for particular approaches
One RCT that was only published as a conference abstract was also excluded. The authors found reduced blood loss, improved sexual function, and higher rates of positive surgical margins in the robot-assisted group compared with the open group.83
In contrast, there was more success obtaining earlier RCT evidence comparing robot-assisted versus laparoscopic radical prostatectomy,29,30 perhaps because they are both minimally invasive and therefore easier to recruit and randomize patients.
Surgeon Experience and Learning Curve
It has been suggested that surgeon experience (i.e., where surgeons lie on the learning curve) is the central factor in improving outcomes, rather than the surgical approach itself.84,85 While large international multicentre studies are usually desirable for generalizability of results, surgeon experience is a difficult factor to adjust for in systematic reviews and meta-analyses since the number of cases needed to overcome the learning curve is uncertain. A recent systematic review found the learning curve ranged from 250 to 1,000 cases for open radical prostatectomy, and from 200 to 750 cases for the laparoscopic approach.86 In comparison, the learning curve for robot-assisted radical prostatectomy was reported to be 40 cases at a minimum, with significant reductions in operative time, blood loss, and complication rates after 100 procedures.86
The RCT compared an open radical prostatectomy surgeon with more than 1,500 cases with a robot-assisted radical prostatectomy surgeon with more than 200 cases.26 Some clinical experts we consulted with suggested that the robotic surgical system may have decreased the learning curve for radical prostatectomy, allowing the less experienced surgeon performing robot-assisted radical prostatectomies to achieve outcomes similar to that of the more experienced surgeon performing open radical prostatectomies.
Given this uncertainty in defining surgeon experience and the lack of adequate reporting in some of the included studies, we could not adequately analyze the effect of surgeon experience on the included outcomes of interest.
The experience of a single centre may be difficult to extrapolate to other centres, especially when high-volume centres with very experienced surgeons are involved, such as in the RCT comparing robot-assisted and open radical prostatectomy. The clinical experts we consulted suggested that few surgeons in Ontario perform large caseloads of open radical prostatectomy; thus, the results for the open radical prostatectomy group in the RCT may not be generalizable to the Ontario context.
Also, it has been suggested that low-volume institutions experience inferior outcomes relative to the highest-volume centres, irrespective of radical prostatectomy approach.87 Similarly, surgeons’ days off between robot-assisted radical prostatectomies may result in increased blood loss and operative time.88
Assessment of Outcomes
Some of the results of the perioperative outcomes of interest do not reflect current practice in other countries or health care systems. Outcomes such as length of hospital stay, operative time, and indwelling catheterization duration may depend on context.
For example, in Ontario, patients are typically discharged 2 to 3 days following radical prostatectomy (robot-assisted or open), while lengths of stay of 5 or more days were seen in some of the included studies. Efforts toward early discharge pathways for open radical prostatectomies have also narrowed the potentially larger differences in length of stay between groups.
Definitions of “operative time” differed within the studies, from total operative time (including preparation time and the induction of anaesthesia) to skin-to-skin time (incision to closure).
The duration of indwelling catheterization may also vary by hospital protocols. Prolonged catheterization is likely a better measure of clinically significant differences between groups. However, very few included studies reported prolonged catheterization duration.
Our experts noted that transfusion rates are subject to differences in transfusion triggers and thresholds—hemoglobin or hematocrit below a certain level. Patients who undergo open radical prostatectomy may be discharged with low hemoglobin levels, so postoperative hematocrit levels may be a better measure for transfusion-related differences between groups. However, this information was rarely reported in the included studies.
Some studies reported instead specific steps of the radical prostatectomy procedure. Authors typically did not provide details of the surgical setup and staff personnel, which, in addition to patient and tumour characteristics, may impact operative time. Lasser et al89 suggested that using consistent nonphysician staff may reduce presurgical preparation time and therefore overall operative time for robot-assisted procedures.
Outcomes following radical prostatectomy may also be measured in a composite manner: in the classic trifecta of potency, continence, and no biochemical recurrence,90 or in the more recently proposed pentafecta,62 which adds negative surgical margins and no perioperative complications. These composite measures are important for patients since they represent an ideal result. However, the differences in reporting composite outcomes are highly variable because of how the individual outcomes are defined.91
There is also a lack of standardization in reporting functional outcomes. The reported continence and potency results were obtained through either questionnaires or interviews, which carry a risk of response bias. Most studies also derived single questions from validated questionnaires. Taken separately, these questions were not validated (although authors usually noted there was good correlation between the two). Some dichotomous (yes/no) outcomes for function differed in their definitions and introduced an element of subjectivity.
The assessment of erectile function lacked uniformity and encompassed different factors: partial recovery, adequate rigidity, ability for intercourse, and overall sexual satisfaction. In addition, potency rates need to be correlated with age, preoperative function, oncological outcomes, surgical technique, and the use of medication to achieve erections.
Some studies specified the use of phosphodiesterase type 5 (PDE5) inhibitor regimens in the first few postoperative months, with PDE5 inhibitor use then reduced to an as-needed basis. However, men may continue using PDE5 inhibitors after achieving excellent postoperative erectile function, for various reasons: ease of obtaining an erection, confidence boost after months of erectile dysfunction, or assured erection.
Few studies collected data on partners of patients and frequency of sexual relations.
Reports of continence rates were more consistent, with most studies using safety pad usage for measurement. However, the number of safety pads used may differ based on their absorbency and how frequently patients opted to change them.
Postoperative care for incontinence was inconsistently reported as well. Some studies reported patients receiving instruction on pelvic floor rehabilitation and training, but most studies did not mention this.
There may also be discrepancies between urologists’ and patients’ perceptions of urinary incontinence after robot-assisted radical prostatectomy. Urologists may underestimate how much incontinence affects patients’ health-related quality of life.92,93 It is recommended that common validated prostate-specific or generic questionnaires, as well as objective data collection, be used for functional and health-related quality of life outcomes in particular. This would minimize biases.
Positive surgical margins and biochemical recurrence are reported as indications of cancer control. While they are associated with prostate cancer progression and provide information on prognosis, they are not discrete patient-important outcomes. Instead, they act as surrogates for future cancer-free survival and cancer-specific mortality rates. A systematic review on positive surgical margins after radical prostatectomy found that their long-term impact is highly variable and largely influenced by other risk modifiers.94
Pathologist assessment of surgical margins varies with experience and expertise; interpretation can be difficult. This may result in overdiagnosis of positive surgical margin status.95
Positive surgical margin rates vary with surgeon experience and cancer stage, volume, and grade. They can range widely from less than 10% to greater than 30% for radical prostatectomy. Also, patients with extracapsular extension (pT3—the cancer has spread beyond the prostate capsule) can have an increased risk of positive surgical margins, depending on the extracapsular extension and how much of the neurovascular bundle or tissue around the prostate the surgeon removes.
Study authors highlighted that biochemical recurrence is not necessarily cancer specific. Benign tissue left during apical dissection or to preserve the bladder neck may mimic biochemical recurrence.30 A better indication of cancer control may be the need for secondary cancer treatment; however, this information was limited in the included studies.
Furthermore, there are various definitions of biochemical recurrence in the literature, ranging from a single PSA measurement of a certain threshold (e.g., > 0.2 ng/mL or > 0.4 ng/mL) to combinations of consecutive PSA measurements.96 While the included studies often defined biochemical recurrence as two consecutive PSA levels > 0.2 ng/mL, some studies accepted a single PSA measurement of > 0.2 ng/mL as an indication of biochemical recurrence.
Ultimately, given the relatively short follow-up durations within studies and the natural progression of prostate cancer, the impact of any radical prostatectomy approach on cancer-free survival or cancer-related mortality could not be determined from positive surgical margins and biochemical recurrence.
Ongoing Studies
We searched the World Health Organization's International Clinical Trials Registry Platform for relevant ongoing comparative studies on robot-assisted radical prostatectomy (Appendix 7). Nine ongoing studies were found, ranging from RCTs on robot-assisted versus laparoscopic radical prostatectomy to prospective nonrandomized studies comparing robot-assisted with open or laparoscopic radical prostatectomy. From the search, it seems there is no RCT registered on robot-assisted versus open radical prostatectomy like the one by Yaxley et al.26
According to PROSPERO, an international prospective register of systematic reviews, there are currently two ongoing systematic reviews related to robot-assist radical prostatectomy (Appendix 7). One systematic review is evaluating how the surgical approach (robot-assisted or open radical prostatectomy) impacts positive surgical margins. The other is investigating robotic surgery in urology and includes only RCTs on the topic.
When searching the Cochrane Database of Systematic Reviews, we found a published Cochrane protocol on open and laparoscopic (specifically robot-assisted) radical prostatectomy for localized prostate cancer. However, we did not find a published review in the Cochrane Database.
Summary
To address the uncertainty and limitations in the evidence, more prospective comparative studies are required that adequately adjust for differences in clinical characteristics between groups. These are particularly needed for the Ontario context, given the increased adoption of robot-assisted surgery in the province.
Conclusions
The conclusions below are based on the best-quality evidence available.
When comparing robot-assisted with open radical prostatectomy, we found:
No differences in short-term urinary and erectile functions at 3 months (moderate quality) and inconclusive findings for long-term results (very low quality)
No differences in pain at 6 weeks postsurgery, health-related quality of life, or return to work or activity (low to moderate quality)
No difference in positive surgical margins (low quality)
Inconclusive results for biochemical recurrence (very low quality)
Reduced operative times favouring robot-assisted prostatectomy (moderate quality)
Reduced lengths of hospital stay and estimated blood loss favouring robot-assisted prostatectomy (moderate quality)
No differences in transfusion rates, indwelling catheterization duration, or hospital readmission rates (moderate quality)
No difference in complication rates (in the RCT; moderate quality), and a reduction in complications favouring robot-assisted surgery (in the nonrandomized studies; very low quality)
When comparing robot-assisted with laparoscopic radical prostatectomy, we found:
Inconclusive results for urinary and erectile functions (low quality)
No difference in health-related quality of life (very low quality)
No differences in positive surgical margins and biochemical recurrence (low quality)
No differences in operative times, lengths of hospital stay, estimated blood loss, transfusion rates, indwelling catheterization duration, or complication rates (moderate quality)
ECONOMIC EVIDENCE REVIEW
Objective
This analysis aimed to review the published economic evidence on the cost-effectiveness of robot-assisted radical prostatectomy versus open radical prostatectomy in patients with clinically localized prostate cancer.
Methods
Sources
We performed an economic literature search on April 22, 2016 for studies published from January 1, 2006, to the search date. The search was developed using the clinical search strategy with an economic filter applied. See Clinical Evidence, Literature Search above, for methods used, and Appendix 1 for literature search strategies, including all search terms. The search was updated monthly through the AutoAlert function in Ovid until September 1, 2016. We also reviewed reference lists of the included economic literature for any additional relevant studies not identified through the systematic search.
Literature Screening
A single reviewer reviewed the titles and abstracts. For studies likely to meet the inclusion criteria from the title and abstract screening stage, we obtained the full-text articles and performed further assessment for eligibility.
Inclusion Criteria
Studies comparing robot-assisted radical prostatectomy versus open radical prostatectomy in patients with clinically localized prostate cancer
English-language full-text publications
Studies published between January 1, 2006, and April 22, 2016
Cost—utility analyses with at least 1 year of follow-up
Exclusion Criteria
Reviews
Abstracts, letters, and editorials
Unpublished studies
Outcomes of Interest
Costs
Quality-adjusted life-years (QALYs)
Incremental cost and incremental effectiveness
Cost per QALY gained
Data Extraction
A single reviewer conducted the preliminary data extraction, applying the inclusion criteria. For studies containing several comparators, we extracted only the results for the comparison of robot-assisted radical prostatectomy versus open radical prostatectomy. We mainly extracted the following information:
Source (i.e., first author, country, year of publication)
Population, perspective, and time horizon
Interventions and comparators
Outcomes (e.g., health outcomes, costs, cost-effectiveness)
If we had questions about a publication, we contacted the authors.
Appraisal of Study Applicability
We determined the usefulness of each included cost—utility study for decision-making by applying a modified methodology checklist for economic evaluations developed by the National Institute for Health and Care Excellence (NICE) in the United Kingdom. The original checklist is used to inform the development of clinical guidelines by NICE.97 We modified the wording of the questions to make it Ontario specific. The original NICE checklist was separated into two sections: one for applicability and one for methodological quality. We used only the first section for our review. Using this checklist, we deemed studies directly applicable, partially applicable, or not applicable to our research questions.
Results
Literature Search
The database search yielded 362 citations (after we removed duplicates) published between 2006 and April 22, 2016. After the formal search date, we also obtained 24 unique results from the monthly AutoAlert function in Ovid. We excluded a total of 375 articles based on information in the title and abstract. We obtained 11 full-text articles that were potentially relevant for further assessment. Figure 15 presents the flow diagram for the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA).
Figure 15: PRISMA Flow Diagram for the Economic Evidence Review.
Source: Adapted from Moher et al, 2009.27
Abbreviations: LRP, laparoscopic radical prostatectomy; ORP, open radical prostatectomy; QALY, quality-adjusted life-year; RARP, robot-assisted radical prostatectomy.
We excluded eight studies because they:
Compared robot-assisted radical prostatectomy versus laparoscopic radical prostatectomy98–100
Compared robot-assisted radical prostatectomy versus mixed procedures of open and laparoscopic radical prostatectomy101
Did not report QALYs as an outcome102
Had major flaws in the estimation of QALYs103
Finally, we included three studies from the United States,106 Denmark,107 and Australia.108
Study Applicability of the Included Studies
After reviewing the three cost–utility studies using the quality appraisal checklist, we found the results of these studies were not applicable to the publicly funded health care system in Ontario. These studies were considered not applicable because the estimates of treatment effects were based on earlier clinical evidence,106–108 the studies were not conducted from the perspective of Ontario or the Canadian public payer,106–108 or the cost of purchasing the robotic system was not included.106
The complete results of the quality appraisal checklist applied to all the included full-text articles can be found in Appendix 8.
Review of Included Economic Studies
Table 30 provides a summary of the three included cost–utility analyses, from the United States,106 Denmark,107 and Australia.108 The studies’ results were inconsistent. Cooperberg et al106 concluded that open radical prostatectomy was dominated by robot-assisted radical prostatectomy in three patient subgroups: men with localized prostate cancer at low, intermediate, or high risk. In contrast, Hohwu et al107 found that robot-assisted radical prostatectomy was dominated by open radical prostatectomy. The Australian study108 showed that robot-assisted radical prostatectomy was more likely to be cost-effective if its effect on sexual function was considered ($37,420 per QALY, in 2005 Australian dollars [AUD]). However, it was not cost-effective when only its effect on urinary continence was considered ($450,200 AUD per QALY).
Table 30:
Results of the Economic Literature Review—Summary
| Author, Year, Country | Study Details | Population | Interventions/ Comparators | Results | |||
|---|---|---|---|---|---|---|---|
| Health Outcomes | Costs | Includes Acquisition Cost of Robot? | Cost-Effectiveness ($/QALY) | ||||
| Cooperberg et al, 2013, United States106 |
Type of economic analysis: CUA Study design: Decision-analytic model Perspective: Payer (insurance payments) Time horizon: Lifetime |
Men with clinically localized, low-, intermediate-, or high-risk prostate cancer | 1. RARP 2. ORP |
Primary outcome: QALYs Total QALYsa: 1. 11.3; 10.5; 9.3 2. 11.3; 10.4; 9.2 Discount rate: 3% |
Currency, cost year: USD, 2009 Total costsa: 1. $19,901; $28,017; $35,014 2. $20,245; 428,589; $36,279 Discount rate: 3% |
No | ICER: ORP was dominated by RARP in low-, intermediate-, and high-risk groups |
| Hohwu et al, 2011, Denmark107 |
Type of economic analysis: CUA Study design: economic evaluation alongside cohort-control study Perspective: Societal Time horizon: 1 year |
Men aged 50–69 years with clinically localized prostate cancer | 1. RARP 2. ORP |
Primary outcome: QALYs gained (difference in utility between baseline and 1 year for each group) Total QALYs gainedb: 1. 0.0103 2. 0.0116 |
Currency, cost year: EUR, 2008 Total direct costs: 1. €8,369 2. €3,863 Discount rate: 3% for estimating the equivalent annual cost of robotic system |
Yes | ICER: RARP was dominated by the ORP |
| Medical Services Advisory Committee, 2006, Australia108 |
Type of economic analysis: CUA Study design: Decision-analytic model Perspective: Public payer Time horizon: 10 years |
Men with clinically localized prostate cancer | 1. RARP 2. ORP |
Primary outcome: QALYs Total QALYsc: 1. 6.76; 6.93 2. 6.66; 6.92 Discount rate: 5% |
Currency, cost year: AUD, 2005 Total costsc: 1. $17,562; $17,388 2. $13,820; $12,886 Discount rate: 5% |
Yes | RARP was associated with an ICER of $37,420 AUD per QALY based on sexual function, and of $450,200 AUD per QALY based on urinary continence |
Abbreviations: AUD, Australian dollar; CUA, cost–utility analysis; EUR, Euro; ICER, incremental cost-effectiveness ratio; NA, not applicable; ORP, open radical prostatectomy; QALY, quality-adjusted life-year; RARP, robot-assisted radical prostatectomy.
Results are presented for patients with low-risk, followed by intermediate-risk, and then high-risk prostate cancer.
Authors did not use the common definition of QALY gained, but reported that “the difference in the derived utility weight between baseline and 1 year constitutes the gained QALYs for each group.”107
Results are presented for analysis based on sexual function followed by that based on urinary continence.
Discussion
There are some methodological concerns for all three economic studies included in this review. Cooperberg et al106 conducted a cost–utility analysis from the perspective of a US payer. They compared numerous primary treatment options (including radiation therapy and surgery) for men with localized prostate cancer at low, intermediate, or high risk. The authors found that for all risk groups, open radical prostatectomy was dominated by robot-assisted radical prostatectomy. However, when we evaluated the study, we found that the differences in QALYs between the two treatments were relatively small in each risk level.
In addition, Cooperberg et al106 assumed that the direct medical costs of robot-assisted radical prostatectomy were the same as those for the traditional laparoscopic approach. This study also did not include the capital cost of the robotic surgical system and its disposables since those costs were not paid by insurance companies in the United States. Given the excess cost of the equipment, consumables, and maintenance associated with robotic surgery, the generalizability of the results of this analysis to the Canadian context was limited.
Hohwu et al107 estimated the cost–utility over 1 year for robot-assisted versus open radical prostatectomy in Denmark from a societal perspective. The acquisition cost (amortized over 5 years and assuming an average of 110 procedures per year) and cost of the maintenance contract were included. The authors estimated that patients who underwent robot-assisted radical prostatectomy had a loss of 0.001 QALYs at incremental direct costs of €4,506 (in 2008 Euros). Therefore, robot-assisted surgery was dominated by open radical prostatectomy in the first year postsurgery.
However, when using the other outcome, “successful radical prostatectomy” (PSA < 0.2 ng/mL, urinary continence, and erectile function), the results favoured robot-assisted radical prostatectomy (34% versus 27%), with a corresponding incremental cost of €64,343 per successful operation.107 This suggests an inconsistency between clinical outcomes and QALYs gained by robot-assisted radical prostatectomy.
In 2006, the Medical Services Advisory Committee of Australia assessed the clinical evidence and economic implications of robot-assisted radical prostatectomy.108 The authors compared the cost of robot-assisted versus open radical prostatectomy. In the study's appendix, the authors conducted an “indicative cost–utility analysis” since there was an absence of robust comparative effectiveness data. The cost–utility analysis comparing robot-assisted to open radical prostatectomy over 10 years was conducted from the perspective of the publicly funded health care system.
Under the assumption that the only observable differences in long-term patient outcomes between robot-assisted and open radical prostatectomy were sexual function and urinary continence, the authors used utilities associated with these conditions to adjust patients’ quality of life over their lifetime.108 Because of a lack of combined health-related utility data, the authors ran two decision-analytic models: one based on data for sexual function and one based on data for urinary continence. The results showed that robot-assisted radical prostatectomy was cost-effective when considering its effect on sexual function ($37,420 AUD per QALY). However, it was not cost-effective when considering its effect on only urinary continence ($450,200 AUD per QALY).
These results should be interpreted with caution. The QALY in this cost–utility analysis was derived from a single observational study published in 2003.109 In this study, robot-assisted radical prostatectomy showed large health benefits in continence, erections, and intercourse over open radical prostatectomy. These large benefits were not observed in most other studies. Also, the estimated differences in the blood infusion rate (2% in robot-assisted versus 35% in open radical prostatectomy) and length of stay (2 days in robot-assisted radical prostatectomy versus 7.5 days in open radical prostatectomy) in this study108 were much greater than those in most Canadian hospitals at present.68,110 Overall, the results of this study were not applicable to the Canadian health care system.
Two Canadian cost studies showed that compared with open radical prostatectomy, there was an increased hospitalization cost for robot-assisted radical prostatectomy of $2,893 and $3,860 CAD per patient, respectively.28,111 However, both studies were conducted more than 5 years ago, and several cost components need to be updated to reflect current costs. For example, in the study by the Canadian Agency for Drugs and Technologies in Health (CADTH),111 the costs (in US$) for acquisition of the robotic system, maintenance, and consumables need updating. Also the US to Canadian dollar exchange rate has increased significantly (from $1 USD = $1.02 CAD in 2011 to $1 USD = $1.32 CAD in 2016). This would lead to a substantial increase in costs related to using robotic surgery in Canada. In addition, this report did not include the 5% federal tax for purchasing robotics and disposables from outside Canada. Nor did it include the overhead cost of using a robotic system, which is about 30% of the capital cost, according to experts’ estimates. Finally, the new generation of the robotic system and its disposables may be more expensive.
Conclusions
The systematic review identified three cost–utility analyses on robot-assisted versus open radical prostatectomy for patients with localized prostate cancer. Results ranged from robot-assisted radical prostatectomy being dominant to being dominated, and none of the studies were applicable to the health care system in Ontario.
PRIMARY ECONOMIC EVALUATION
The first RCT comparing robot-assisted versus open radical prostatectomy was published in July 2016.26 The study demonstrated that robot-assisted and open radical prostatectomy have similar outcomes in urinary function, sexual function, positive surgical margin rates, and the Short-Form 36 Health Survey (physical and mental domains) responses after 12 weeks. The clinical evidence review section of this report includes results from this same RCT.
Recognizing that this RCT provides a higher level of evidence than previously published nonrandomized studies for robot-assisted versus open radical prostatectomy, we primarily based our model parameters on the short term data reported in the RCT, where available. We then developed a primary economic evaluation for the Ontario context.
Objective
This study aimed to assess the cost-effectiveness (i.e., the incremental cost per QALY gained) of robot-assisted versus open radical prostatectomy in patients with clinically localized prostate cancer in Ontario.
Methods
The information presented in this report follows the reporting standards set out by the Consolidated Health Economic Evaluation Reporting Standards statement.112
Type of Analysis
We conducted a cost–utility analysis comparing robot-assisted versus open radical prostatectomy in patients with clinically localized prostate cancer in Ontario.
Target Population
The target population in our model was 60-year-old males who are newly diagnosed with clinically localized prostate cancer, with no concurrent cancer or other major comorbidities, and with an estimated remaining life expectancy of over 10 years. We focused on patients with low or intermediate risk of cancer progression since, in Ontario, robot-assisted radical prostatectomy is generally performed in these patients.72
Perspective
We conducted this analysis from the perspective of the Ontario Ministry of Health and Long-Term Care.
Interventions
We compared open radical prostatectomy with robot-assisted radical prostatectomy performed using the da Vinci Surgical System. This is the only robotic surgical system licensed for use in Canada. We excluded laparoscopic radical prostatectomy as a comparator since it is uncommonly performed in Ontario. (According to data from IntelliHealth Ontario, its total volume in 2015 was 98 cases, about 4% of the total radical prostatectomies performed in Ontario.)
Discounting and Time Horizon
We discounted future costs and QALYs (i.e., greater than 1 year) to present values, and we applied an annual discount rate of 5% to both costs and QALYs, following the guidelines for economic evaluations from the Canadian Agency for Drugs and Technologies in Health.113
The results of the clinical review did not find high-quality evidence suggesting differences in long-term outcomes between robot-assisted and open radical prostatectomy. Thus, we used a 1-year time horizon in our base case analysis and a 10-year time horizon for the scenario analyses.
Main Assumptions
To simplify the model, we made the following assumptions:
In the base case, there are some benefits for health-related quality of life for robot-assisted radical prostatectomy within 1 year, but no differences in functional and oncological outcomes between robot-assisted and open radical prostatectomy at 1 year postsurgery
For one of the scenario analyses, we assumed robot-assisted is better than open prostatectomy in functional and oncological outcomes, and we explored the cost-effectiveness of the two treatments in the long-term time horizon
The Surgical Pain Score and Expanded Prostate Cancer Index Composite are adequate to measure patients’ health-related quality of life after a radical prostatectomy. We derived the average utility values for the short-term analysis from these measures
There is no perioperative mortality for either treatment
Except in patients who have progressed to the metastatic state, the mortality risk of patients in other health states is the same as that of the general Canadian age- and sex-specific population. This is because prostate cancer is a low-risk cancer; the 5-year relative survival ratio (observed survival of cancer patients compared with the expected survival of the general population) is high, at 0.98 to 0.99114
Resource use (e.g., operating room time, length of stay) of robot-assisted and open radical prostatectomy from the RCT is applicable to the Ontario context26
Capital costs of the da Vinci Surgical System are fixed and are based on the da Vinci Si HD. In addition, we included the 5% federal tax for purchasing the robotic surgical system, service contract, and disposable items
Service contract costs for the da Vinci Surgical System and costs of disposables remain constant over time, and there are no cost differences between hospitals
The exchange rate between Canadian and US dollars is constant
Model Structure
In the base case analysis, a decision-analytic model was constructed to compare the costs and utilities of patients treated with robot-assisted or open radical prostatectomy within a 1-year time horizon, without considering disease progression. Costs and utilities were estimated separately (see the Model Parameters section, below).
In the scenario analyses, we constructed a Markov decision-analytic model to capture prostate cancer progression and the economic outcomes of patients undergoing open or robot-assisted radical prostatectomy in the long-term (Figure 16). We used a yearly cycle in the model. Patients would receive either robot-assisted or open radical prostatectomy at time zero. The model includes a 1-year transition period after the radical prostatectomy. After the first year (i.e., from the second cycle), patients would remain in the same health state or move in a one-way trajectory from recurrence free to recurrence and metastasis.
Figure 16: Markov Decision-Analytic Model for Scenario Analyses.
Abbreviation: M, Markov model.
The costs and utility values in the first year were estimated based on the results from the base case, plus the cost and the QALY loss owing to urinary or sexual dysfunction. The health utility values and cost in the second year or later were estimated based on patients’ cancer stages and possible urinary and sexual dysfunction.
Model Parameters
We obtained the model parameters from several published studies. When necessary, we contacted the authors to clarify questions or request additional data regarding their publications.115 These inputs—utilities, costs, and clinical outcomes—are provided below.
Utility Parameters
Overview
Utilities represent a person's preference for certain health outcomes, such as being able to walk. These are often measured on a scale of 0 (death) to 1 (full health). Since Yaxley et al26 did not report utility values, we estimated the baseline utility (before the radical prostatectomy) from the literature, and derived utility changes postsurgery from the pain and Expanded Prostate Cancer Index Composite scores reported in the RCT. Figure 17 outlines the process we used to estimate utilities at different follow-up times. Table 31 shows the utility values we used in our analysis.
Figure 17: Process of Estimating Utility Values and QALYs.
Abbreviations: PORPUS-U, Patient-Oriented Prostate Utility Scale—Utility; QALY, quality-adjusted life-year; RCT, randomized controlled trial.
aCoefficient from statistical models in published articles (Krahn et al, 2013,115 and Xie et al, 2016119).
Source: RCT data from Yaxley et al, 2016.26
Table 31:
Utilities Used in the Economic Model
| Health State | Mean (95% CI) | Distribution for Probabilistic Sensitivity Analysis | Reference |
|---|---|---|---|
| Utility in the first year | |||
| Pre-treatment (baseline) | 0.97 | Fixed value | Krahn et al116 |
| Utility change in RARP arm | |||
| 1 day | −0.1406 (−0.1539, −0.1272)a | Pain score: normal (4.60, 0.1786)b | Calculated26,119 |
| 7 days | −0.0767 (−0.0873, −0.0660)a | Pain score: normal (2.51, 0.1607)b | Calculated26,119 |
| 6 weeks | −0.0524 (−0.0607, −0.0423)a | Normal for EPIC urinary, sexual, and bowel domainsc | Calculated26,115 |
| 12 weeks | −0.0326 (−0.0391, −0.0246)a | Normal for EPIC urinary, sexual, and bowel domainsc | Calculated26,115 |
| 1 year | −0.03 (−0.05, −0.01) | Normal (−0.03, 0.01) | Krahn et al116 |
| Utility change in ORP arm | |||
| 1 day | −0.1782 (−0.1938, −0.1624)a | Pain score: normal (5.83, 0.2015)b | Calculated26,119 |
| 7 days | −0.0975 (−0.1078, −0.0872)a | Pain score: normal (3.19, 0.1454)b | Calculated26,119 |
| 6 weeks | −0.0469 (−0.0543, −0.0378)a | Normal for EPIC urinary, sexual, and bowel domainsc | Calculated26,115 |
| 12 weeks | −0.0333 (−0.0401, −0.0248)a | Normal for EPIC urinary, sexual, and bowel domainsc | Calculated26,115 |
| 1 year | −0.03 (−0.05, −0.01) | Normal (−0.03, 0.01) | Krahn et al116 |
| Utility in the second year or later | |||
| Recurrence free | 0.94 | NA | Krahn et al116 |
| Recurrence | 0.83 | NA | Naik et al120 |
| Metastasis | 0.78 | NA | Naik et al120 |
| Utility change because of secondary surgeries for urinary incontinence | −0.0361d | NA | Krahn et al115 |
| Utility change because of sexual dysfunction in the second and third years | –0.0581e | NA | Krahn et al115 |
Abbreviations: CI, confidence interval; EPIC, Expanded Prostate Cancer Index Composite; NA, not applicable; ORP, open radical prostatectomy; RARP, robot-assisted radical prostatectomy.
Since the utility change was calculated from two or more variables, we used a Monte Carlo simulation to estimate 95% CIs.
Distribution of the coefficient of the pain/discomfort domain in EuroQoL—Five Dimensions (EQ-5D-5L): normal (−0.0764, 0.0022).119
Distribution of the coefficient for EPIC domains: urinary—normal (0.000722, 0.000117); sexual—normal (0.001162, 0.000101); and bowel—normal (0.001365, 0.000164).115
We assumed that the secondary surgeries for urinary incontinence would result in a 50-point reduction in the urinary domain of the Prostate Cancer Index, and that the utility reduction would last for 1 year.
We assumed that sexual dysfunction would result in a 50-point reduction in the sexual domain of the Prostate Cancer Index.
Utilities at baseline (0.97) and 1 year post-prostatectomy (0.94) were obtained from Krahn et al,116 who measured quality-of-life changes in a cohort of Toronto men with newly diagnosed, localized prostate cancer. They measured utilities using the Patient-Oriented Prostate Utility Scale (PORPUS).116 This instrument is more sensitive than generic instruments to detecting small changes in quality of life in early prostate cancer.117,118 It should be noted that the baseline utility value would not affect the difference in QALY between two treatments in the present study. The decrease in utility from baseline to 1 year (−0.03)116 showed that patients had not fully recovered at 1 year after radical prostatectomy.
We assumed that patients undergoing robot-assisted or open radical prostatectomy would have the same utility at baseline and 1 year. However, we expected utility to be different during the recovery process since Yaxley et al26 found patients had slightly different pain and functional outcomes (based on actual reported results and not statistical inference). Two studies reported the relationship between utility change and changes in pain and functional scores (i.e., the coefficient).115,119 Thus, the utility values at day 1, day 7, week 6, and week 12 were estimated by subtracting the decrease in utility at given follow-up time points from the baseline value (0.97). In the following sections, we discuss the details for estimating utility change at different time points.
We calculated the QALY in the first year as the area under the utility curve at 1 year following each treatment.
Since we used various sources to derive the utilities, we examined the logical consistency of the calculated utility value at each time point (i.e., face validity). For example, postsurgery utility values would be the lowest on the first day following surgery and would gradually increase over time in both groups. Also, we would expect the utility of the open radical prostatectomy group to be lower than that of the robot-assisted radical prostatectomy group in a short period postsurgery, since open radical prostatectomy is a more invasive and painful treatment and has a higher risk of perioperative complications and needing blood transfusions.26
Days 1 and 7 After Surgery
We estimated the utility change at days 1 and 7 after surgery by using the pain score during normal activities (measured by the Surgical Pain Scale, from 0 to 10) from the RCT26 since there was no Expanded Prostate Cancer Index Composite score measured at days 1 and 7. In a recently published article on the valuation of a five-level version of the EuroQoL—Five Dimensions (EQ-5D-5L) for Canada, the authors reported that the coefficient of the pain/discomfort domain was −0.0764 in the linear term in the main effect model.119 We assumed that the minimum and maximum values in the 10-level Surgical Pain Scale would be equivalent to those in the pain/discomfort domain of EQ-5D-5L (with 0 and 10 in the Surgical Pain Scale corresponding to 1 and 5 in the EQ-5D-5L, respectively). We also assumed the spacing between two consecutive levels would be distributed evenly for each instrument. The change in pain score from the Surgical Pain Scale would be divided by 2.5, which approximates to the change in the pain/discomfort domain in EQ-5D-5L. Assuming “no pain” at baseline (0 in the Surgical Pain Scale), we used the formula below to estimate the utility change at day 1 and day 7,26,119 relative to baseline utility:
Weeks 6 and 12 After Surgery
We also estimated the utility reduction at weeks 6 and 12. We extracted the Expanded Prostate Cancer Index Composite scores for the urinary, sexual, and bowel domains at time 0, week 6, and week 12 from the single RCT26 and calculated the score changes in each domain at 6 weeks and 12 weeks relative to time 0. As obtained through multiple regression analyses by Krahn et al,115 one unit change of Prostate Cancer Index score in the urinary domain, sexual domain, or bowel domain corresponded to a mean utility change of 0.00072, 0.001162, or 0.001365, respectively. We then used the following formula to approximate the utility change at 6 weeks and 12 weeks, compared with baseline:
 |
where EPIC is the Expanded Prostate Cancer Index Composite and PORPUS-U is the Patient-Oriented Prostate Utility Scale—Utility.
It should be noted that Yaxley et al26 reported the results using the Expanded Prostate Cancer Index Composite, whereas Krahn et al115 used the results of the Prostate Cancer Index in their multiple regression. Since the Expanded Prostate Cancer Index Composite and Prostate Cancer Index have the same scale from 0 to 100 and considerable correlations, especially for urinary and sexual function domains,121,122 we made the assumption that the coefficients from the Prostate Cancer Index could also be applied to the Expanded Prostate Cancer Index Composite.
More Than 1 Year After Surgery
We assumed that patients’ long-term utility values were associated with patients’ oncological and functional outcomes (up to 3 years). The health utilities were 0.94, 0.83, and 0.78 for patients in the recurrence-free, recurrence, and metastasis health states, respectively.116,120 (Note: We did not find an appropriate Patient-Oriented Prostate Utility Scale—Utility source for recurrence and metastasis. Thus, the utilities for recurrence and metastasis were based on the EQ-5D-5L method.) We also estimated the utility reduction owing to specific reasons such as sexual dysfunction (Table 31).
Cost Parameters
Costs are reported in 2016 Canadian dollars (CAD), unless otherwise indicated.123
Capital Investment, Service Contract, and Disposables
Experts provided the 2016 purchasing price of the da Vinci Surgical System, as well as the costs for service and its disposables (Table 32).
Table 32:
Costs of the da Vinci Si Surgical System
| Description | USD in 2016 | CAD in 2016 |
|---|---|---|
| Capital cost | ||
| Base of da Vinci Si Surgical System | $2,400,000 | $3,337,310a |
| Options of da Vinci Si Surgical System | $500,000 | $695,273a |
| Start-up reusable equipment (in total) | $200,000 | $278,109a |
| Hospital's overhead cost | NA | $1,000,000 |
| Equivalent annual cost of total capital cost and hospital's overhead cost at different service lifespan | ||
| 5 years | NA | $1,226,636 |
| 7 years | NA | $917,793 |
| 9 years | NA | $747,162 |
| 12 years | NA | $599,181 |
| Service/maintenance cost (per year) | $220,000 | $305,920a |
| Disposable costs for prostatectomy (per procedure) | $2,500 | $3,476a |
Calculated using an exchange rate of $1 USD = $1.32 CAD (the average exchange rate from Jan–Aug 2016), plus 5% federal tax for purchasing medical devices and services.
The capital cost associated with the robotic system included the costs of the base and accessories and a one-time cost for upgrading the operating room. The average total cost of a da Vinci Si Surgical System was estimated to be $3.1 million USD: $2.4 million USD for the base, $500,000 USD for accessories, and $200,000 USD for the start-up reusable equipment (e.g., three-dimensional vision system).
According to the manufacturer, the base of the robotic system costs $1.7 million to $3 million USD. We used an average of $2.4 million USD considering both the quoted price and any potential discount.
The da Vinci Si Surgical System also offers accessories such as a second console, skills simulator, fluorescence imaging starter kit, and vessel sealer starter kit. The total quoted cost of those options (one of each) was slightly over $1,000,000 USD. We used $500,000 USD as the average cost, assuming that a hospital would purchase only a few of these additional options.
According to consulted experts, the indirect (overhead) cost for the hospital of using robotics was estimated to be about 30% of the capital cost. Hospitals often need to upgrade the operating room to conduct robotic-assisted surgeries because there are higher operating room standards for robotic surgeries.111 Thus, we assumed that the total indirect cost for a hospital was $1,000,000 CAD for the whole service life of a robotic system.
In addition, the annual service and maintenance fee of the robotic system was $220,000 USD. The quoted cost of disposables for a typical procedure was $2,825 USD. We used $2,500 USD per procedure in our base case analysis, considering a potential discount.
Since these costs are in US dollars, we converted them into Canadian dollars using the average exchange rate from January to August 2016 ($1 USD = $1.32 CAD).124 There is also a 5% federal tax for purchasing medical devices and services through a hospital in Canada. We calculated the equivalent annual cost of the capital investment of purchasing a robotic surgical system under a few assumptions:
The service lifespan of the da Vinci Si Surgical System is 9 years (base case)
There is no residual value at the end of the surgical system's service life
There is no cost to dispose of the surgical system
We present the capital and annual costs of using robotics in Table 32.
We used the following formula to estimate the attributable cost per procedure of using the robotic system (not including disposable costs):
 |
According to experts, in Ontario in 2015, there were 10 da Vinci Surgical Systems used for 1,616 robotic-assisted surgeries (including 828 prostatectomies, 294 hysterectomies, and 123 lobectomies). On average, one robotic system is used for 162 surgeries per year. We expect that the overall volume of robot-assisted surgeries will continue to increase in the next few years. Thus, we assumed 200 robot-assisted surgeries conducted per robotic system per year in Ontario as the base case (about 24% higher than that in 2015).
We also conducted sensitivity analyses under different assumptions for the service lifespan of the robotic system and the volume of robot-assisted surgeries (Table 33). The attributable cost and the costs of disposables are the total cost related to using the da Vinci Surgical System.
Table 33:
Attributable Cost per Procedure Related to Using the da Vinci Surgical System
| Service Life of the System | Attributable Cost per Procedure (ĆD) by Different Volume per Robotic System per Year | ||
|---|---|---|---|
| n = 162 | n = 200 | n = 300 | |
| 5 years | 9,460 | 7,663 | 5,109 |
| 7 years | 7,554 | 6,119 | 4,079 |
| 9 years | 6,501 | 5,265 | 3,510 |
| 9 years, without the capital cost of purchasing the systema | 2,757 | 2,233 | 1,489 |
| 12 years | 5,587 | 4,526 | 3,017 |
Although there was no capital cost for purchasing the robotic system (e.g., a donated system), there were $1,000,000 in overhead costs for the hospital plus an annual service fee of $305,920 per year.
Hospitalization Costs
We estimated the hospitalization costs of robot-assisted and open radical prostatectomy using the unit price multiplied by the use of health care resources, such as the operating room time, length of stay, recovery room time, blood transfusion rate, and readmission rate. Thus, the hospitalization cost estimates included the hospitalizations for radical prostatectomy treatment and readmission owing to complications.
The resources used for the two treatments were based on the single RCT,26 and the unit prices were obtained from earlier cost comparison studies at McMaster University's Institute of Urology,125 McGill University Health Centre,126 and the Ontario Schedule of Benefits.127 Clinical experts verified our estimations for these unit prices. We assumed that the hospitalization cost was the total cost of two prostatectomy treatments in the first month (Table 34). Costs for the remaining 11 months were estimated to be $2,746 for both groups, based on the Ontario cohort.128
Table 34:
Hospitalization Cost of Robot-Assisted and Open Radical Prostatectomy, Including Readmission
| Health Care Resource | Unit Price, $a | RARPa | ORPa | ||
|---|---|---|---|---|---|
| Resource Use, Mean (95% CI) | Cost, $ | Resource Use, Mean (95% CI) | Cost, $ | ||
| Robotic equipment and service contract | — | — | 5,265 | — | NAb |
| Consumables and disposables | — | — | 3,476c | — | 273 |
| Operating room | 1,278/hour125 | 4.10 (3.96, 4.24)26 | 5,241 | 4.67 (4,57, 4.77)26 | 5,971 |
| Recovery room | 221/hour130 | 1.80 (1.64, 1.96)26 | 397 | 1.79 (1.40, 2.18)26 | 394 |
| Surgical bed | 1,037/day125 | 1.55 (1.41, 1.96)26 | 1,607 | 3.27 (3.03, 3.51)26 | 3,390 |
| Surgeon fees | — | — | 1,508127,c,d | — | 1,081127c,d |
| Anaesthesia | 15/unit127 | 10.00127 | 150 | 10.00127 | 150 |
| Blood transfusion | 757/transfused patient125 | 0.01 (0, 0.03)26 | 8 | 0.04 (0.01, 0.08)26 | 30 |
| Readmissione | 4,334125,127,130 | 0.05 (0.02, 0.1)26 | 217 | 0.08 (0.04, 0.13)26 | 347 |
| Total average costs | — | — | 17,869 | — | 11,636 |
Abbreviations: CI, confidence interval; ORP, open radical prostatectomy; RARP, robot-assisted radical prostatectomy.
In the probabilistic sensitivity analysis, we used the fixed values for the unit price and gamma distributions for the resource uses for both groups. All costs are in 2016 Canadian dollars.
Although there are equipment-related costs for ORP, the surgical equipment is often not solely dedicated for ORP. In the long-term, the attributable cost of equipment for ORP is likely to be small, assuming that the service life of the equipment is long and the volume of (different types of) surgeries is large. Since this cost is relatively small and difficult to quantify, we have excluded it from this analysis.
We used the ± 25% of the point estimate in the deterministic sensitivity analysis, and assumed the standard error is 20% of the point estimate in the probabilistic sensitivity analysis.
The Ontario Schedule of Benefits127 includes the cost for the surgical assistant service (8 units for RARP and 6 units for ORP).
We assumed that one readmission would consume 1 hour of operating room time, 2 hours of recovery room time, 2 days of hospitalization, and a professional fee of 50% of the ORP cost ($4,334 total).
We noticed that the Canadian data for operating room times and lengths of the hospital stay showed considerable differences, compared with those in the RCT26. Thus, we used local data for the operating room time and length of hospital stay in the scenario analyses. Based on the Ontario Case Costing Initiative from Ontario Ministry of Health and Long-Term Care, we estimated that on average the lengths of hospital stay were 2.6 days for robotic surgery and 3.1 days for open surgery, and the direct operating room costs were $2,793 for robotic surgery and $2,589 for open surgery. (Operating room time was not available.) The Ontario hospitalization costs of robot-assisted and open radical prostatectomy were then reduced to $16,511, and $8,079 respectively.
Based on the national statistics from Canadian Institute for Health Information,110 the median hospital stays were 2 days for robotic surgery and 3 days for open surgery, and operating room times were 3.67 hours for robotic surgery and 2.85 hours for open surgery. Using these data, the hospitalization costs of robot-assisted and open radical prostatectomy were $17,785 and $9,028, respectively.
Long-Term Cost in Patients with Prostate Cancer
We also estimated the long-term health care costs of patients following radical prostatectomy. We used only those costs in the scenario analyses for the Markov model. Based on data from Krahn et al,128 we estimated that the yearly costs for the recurrence-free, recurrence, and metastasis health states after prostatectomy were $2,996, $7,854, and $19,110, respectively.
In addition, we estimated the cost of secondary treatments using data from one teaching hospital in Ontario: artificial urinary sphincter insertion ($14,047.50) and urethral sling placement ($4792.5) for incontinence. We estimated the annual cost for erectile dysfunction therapy was $479.129
Parameters of Functional and Oncological Outcomes Favouring Robot-Assisted Radical Prostatectomy in the Scenario Analyses
The recently published RCT concluded that there were no significant differences in oncological and functional outcomes between the two treatments at 12 weeks.26 Our present clinical review (based on prospective comparative studies) found inconclusive findings for these outcomes at 1 year.
However, given that some studies (e.g., those using administrative databases) showed results favouring robot-assisted radical prostatectomy131 and given that some experts believe that robot-assisted radical prostatectomy produces better functional outcomes, we made more favourable assumptions for robot-assisted radical prostatectomy in the scenario analyses. In the Markov model, the probabilities of cancer progression in open radical prostatectomy were obtained from the validated model for prostate cancer.132 Since the clinical review showed inconclusive results for biochemical recurrence, we used an assumed risk ratio of 0.85 for biochemical recurrence for robot-assisted versus open radical prostatectomy. It should be noted that this is a highly favourable and speculative assumption.
The rate of secondary surgeries owing to urinary incontinence in open radical prostatectomy was based on a large cohort in Ontario: about 2.8% and 1.1% of patients underwent artificial urinary sphincter and urethral sling placement, respectively.133 We assumed that the risk ratio of secondary surgeries was 0.5 favouring robot-assisted radical prostatectomy because we did not identify published evidence of these surgeries after the robotic surgery.
In addition, there are few published data comparing sexual function outcomes between these two prostatectomies in Ontario. For simplicity, we extracted data from the time-to-potency curve for both treatments in an observational study with a 30-month follow-up.46 This study showed a large benefit of robot-assisted radical prostatectomy on sexual function.
All the parameters for the long-term model are presented in Table 35.
Table 35:
Long-Term Clinical Outcomes for Scenario Analyses Favouring Robot-Assisted Radical Prostatectomy
| Description | Mean | References |
|---|---|---|
| Oncological outcomes | ||
| Annual transition probability from recurrence-free to local recurrence for patients with low- or intermediate-risk cancer in the ORP groupa | 0.03 | Sanyal et al129,132 |
| Risk ratio of recurrence, RARP versus ORP | 0.85 | Assumptionb |
| Annual transition probability from biochemical recurrence to metastatic castration-resistant prostate cancer for both groups | 0.07 | Sanyal et al132 |
| Mortality in different health states | ||
| Recurrence free or local recurrence | Same as for age- and sex-specific general population in Canada | Assumption |
| Age, years | ||
| 55 | 0.00481 | Life table134 |
| 60 | 0.00791 | Life table134 |
| 61 | 0.00846 | Life table134 |
| 62 | 0.0087 | Life table134 |
| 63 | 0.00994 | Life table134 |
| 64 | 0.01136 | Life table134 |
| 65 | 0.01161 | Life table134 |
| 66 | 0.01306 | Life table134 |
| 67 | 0.01452 | Life table134 |
| 68 | 0.01595 | Life table134 |
| 69 | 0.01713 | Life table134 |
| Metastatic castration-resistant prostate cancer | 0.27 | Sanyal et al132 |
| Functional outcomes: urinary incontinence | ||
| Artificial urinary sphincter insertion in the ORP group per year (up to 3 years) | 0.94% | Nam et al133 |
| Urethral sling placement in the ORP group per year (up to 3 years) | 0.37% | Nam et al133 |
| Risk ratio of surgical interventions owing to urinary incontinence in the RARP group | 0.5 | Assumption |
| Functional outcomes: sexual dysfunction | ||
| Patients with sexual dysfunction in the RARP groupc | ||
| Year 1 | 0.67 | Kim et al46 |
| Year 2 | 0.3 | Kim et al46 |
| Year 3 | 0.08 | Kim et al46 |
| Patients with sexual dysfunction in the ORP groupc | ||
| Year 1 | 0.93 | Kim et al46 |
| Year 2 | 0.55 | Kim et al46 |
| Year 3 | 0.31 | Kim et al46 |
Abbreviations: CI, confidence interval; ORP, open radical prostatectomy; RARP, robot-assisted radical prostatectomy.
The recurrence rates vary greatly in published data for various reasons. We used the value 0.03 annually from the validated model by Sanyal et al.129,132
Two of the prospective comparative studies showed no significant difference for biochemical recurrence between groups.37,54 A third study by Breyer et al35 showed ORP had a statistically significant lower risk of recurrence at 3 years. Thus, we made a hypothetical risk ratio to favour RARP.
We extracted data on the proportion of potent patients at 6, 18, and 30 months in the Kaplan-Meier curve to represent the estimates of the sexual function in years 1, 2, and 3, respectively.
Analysis
Using the parameters above, we estimated the health care costs and QALYs of robot-assisted and open radical prostatectomy. We conducted a base case analysis using the best available point estimates. We applied Ontario and Canadian data regarding lengths of hospital stay and operating room times in the scenario analyses. We also conducted deterministic sensitivity analyses (e.g., changing from the mean to the upper and lower limit 95% CIs of the variables) to assess the impact of key variables on the incremental cost-effective ratio (ICER), and probabilistic sensitivity analyses to assess parameter uncertainty.
Also, we conducted additional analyses to examine the cost-effectiveness of robot-assisted versus open radical prostatectomy in scenarios favouring robot-assisted radical prostatectomy. First, we used the 1-year model, which is the same as the base case but excluded the capital cost of the robotic system. We then applied the upper limits of the 95% CIs of the longitudinal utilities to estimate QALYs in the robot-assisted radical prostatectomy group. Next, we applied functional and oncological outcomes that favoured robot-assisted radical prostatectomy in the Markov model.
We conducted all analyses using SAS version 9.4 (SAS Institute, Cary, North Carolina) for the 1-year model and TreeAge Pro 2016 (TreeAge Software, Williamstown, Massachusetts) for the Markov model.
Generalizability
The findings of this economic analysis cannot be generalized to all patients with localized prostate cancer. They may however be used to guide decision-making about the specific patient populations in Ontario addressed in the studies we evaluated.
Expert Consultation
Throughout the development of this model, we solicited expert consultation. The expert advisors reviewed the model structure and inputs to confirm that the information we used reasonably reflected the clinical context for prostate cancer in Ontario. However, the statements, conclusions, and views expressed in this report do not necessarily represent the views of the consulted experts.
Results
Base Case Analysis
Table 36 presents the base case results for our analysis. Compared with open radical prostatectomy, robot-assisted radical prostatectomy was associated with higher costs ($6,234 per patient) and small QALYs gained (0.0012). The associated ICER was $5.2 million per QALY gained. Based on a willingness to pay of $100,000 per QALY or lower, robot-assisted radical prostatectomy was not cost-effective compared with open radical prostatectomy.
Table 36:
Results of the Base Case Analysis for the Cost–Utility Analysis, Robot-Assisted Versus Open Radical Prostatectomy
| Strategy | Average Total Costs, $a,b | Incremental Cost, $a,b | Average Total QALYsa | Incremental QALYsa | Incremental Cost-Effectiveness Ratio, $a,b/QALY gained |
|---|---|---|---|---|---|
| ORP | 14,369 | — | 0.9284 | — | — |
| RARP | 20,604 | 6,234 | 0.9296 | 0.0012 | 5,200,894 |
Abbreviations: ORP, open radical prostatectomy; QALY, quality-adjusted life-year; RARP, robot-assisted radical prostatectomy.
Numbers may appear inexact because of rounding.
All costs are 2016 Canadian dollars.
Deterministic Sensitivity Analysis
We examined several factors that could affect the ICER values of robot-assisted versus open radical prostatectomy. Figure 18 presents the results of the one-way sensitivity analysis. The blue and orange bars illustrate the ICERs at lower and upper limits of the respective variables. The main variables that influenced the cost-effectiveness included the utility changes after 12 weeks or 1 year of surgery for both groups, the service life of the robotic system, the volume of surgeries per robotic system, and the cost of disposables for robot-assisted radical prostatectomy.
Figure 18: Tornado Diagram of One-Way Sensitivity Analysis, Robot-Assisted Versus Open Radical Prostatectomya.
Abbreviations: ORP, open radical prostatectomy; QALY, quality-adjusted life-year; RARP, robot-assisted radical prostatectomy.
aAll costs are in 2016 Canadian dollars. For each variable, the blue and orange bars show the incremental cost-effectiveness ratios at lower and upper limits, respectively.
Probabilistic Sensitivity Analysis
We conducted a probabilistic sensitivity analysis using the same 1-year model as the base case, and we assigned the parameters with probabilistic distributions instead of point estimates. Figure 19 illustrates the incremental cost and incremental QALYs calculated for each simulation of the probabilistic sensitivity analysis for robot-assisted versus open radical prostatectomy.
Figure 19: Incremental Cost and QALYs of Robot-Assisted Versus Open Radical Prostatectomya.
Abbreviations: ORP, open radical prostatectomy; QALY, quality-adjusted life-year, RARP, robot-assisted radical prostatectomy.
aThe triangle indicates the base case scenario. Each circle surrounding the triangle represents a single result from the simulation, presenting the incremental effects and incremental costs of RARP relative to ORP. The green diagonal line shows the willingness-to-pay threshold of $100,000 per QALY.
The results of the Monte Carlo simulations are consistent with those of the base case. Compared with open radical prostatectomy, the median incremental cost of robot-assisted radical prostatectomy is $6,164 (quartile 1: $5,279; quartile 3: $7,102), and the median QALY gained is 0.0012 (quartile 1: 0.0002; quartile 3: 0.0026). All simulated results are above the threshold line, indicating robot-assisted radical prostatectomy has very little to no chance of being cost-effective at a willingness-to-pay threshold of $100,000 per QALY gained.
Scenario Analyses
Table 37 presents the results of our various scenario analyses. Assuming that the QALYs of both treatments were the same as those in base case, we used the local length of stay data and operating room time data to recalculate the costs in the scenario analyses. When we used the Ontario data, the incremental cost for the robotics treatment increased to $8,432 and the associated ICER was $7.0 million per QALY gained. When we used the Canadian statistics, the incremental cost for the robotics treatment increased to $8,757 with an associated ICER of $7.3 million per QALY gained.
Table 37:
Results of Scenario Analyses for the Cost–Utility Analysis, Robot-Assisted Versus Open Radical Prostatectomy
| Scenario | Incremental Cost, $a (RARP vs. ORP) | Incremental QALYsa (RARP vs. ORP) | Incremental Cost-Effectiveness Ratio, $a/QALY Gained |
|---|---|---|---|
| Using local data for operating room time and length of hospital stay | |||
| Ontario data | 8,432 | 0.0012 | 7,034,376 |
| Canadian national statistics | 8,757 | 0.0012 | 7,305,690 |
| 1-year model (under assumptions favouring RARP) | |||
| Excluding the capital cost of purchasing the robotic systemb | 3,224 | 0.0012 | 2,689,882 |
| Using the upper limits of 95% CI for utility values in days 1, 7, 42, 84, and 365 in the RARP group | 6,234 | 0.0138 | 453,095 |
| Excluding the capital cost of purchasing the robotic systemb and using upper limits of 95% CI for utility values in the RARP group | 3,224 | 0.0138 | 234,339 |
| Excluding the capital cost of using the robotic system and maintenance service feec | 969 | 0.0012 | 808,708 |
| Excluding the capital cost of using the robotic system and maintenance service feec and using upper limits of 95% CI for utility values in the RARP group | 969 | 0.0138 | 70,454 |
| Long-term model (under assumptions favouring RARP) | |||
| RARP with favourable functional outcomes in 3 years | 5,899 | 0.0419 | 140,700 |
| RARP with favourable oncological outcomes in 10 years | 5,542 | 0.0213 | 260,127 |
| RARP with favourable functional outcomes in 3 years and favourable oncological outcomes in 10 years | 5,206 | 0.062 | 83,921 |
Abbreviations: CI, confidence interval; ORP, open radical prostatectomy; QALY, quality-adjusted life-year; RARP, robot-assisted radical prostatectomy.
Note: some numbers may appear inexact because of rounding.
Although there was no capital cost for purchasing the robotic system (e.g., a donated system), there was $1,000,000 CAD in overhead costs for the hospital and an annual service fee of $305,920 CAD per year.
This analysis included only the cost of the disposables for the robotic system; it excluded the cost of the base, options, and start-up equipment of robotic system, the annual service fee, and hospital's overhead cost.
We also conducted various scenario analyses that favoured robot-assisted radical prostatectomy. When we used the upper limits of the 95% CIs of the longitudinal utility data in the robot-assisted radical prostatectomy group, the QALY gain was larger (0.0138). If we also excluded the capital cost for purchasing the robotic system (i.e., assuming the robotic system was donated), the incremental cost was reduced to $3,224 and the associated ICER was $234,339 per QALY gained. If we only excluded the capital cost of adopting the robotic system and the maintenance fee (i.e., to estimate the cost-effectiveness of additional cases using the robotic system), the ICER was $808,708 per QALY gained.
We also created the Markov model to capture the potential health benefits of robot-assisted radical prostatectomy in the long term. If we assumed that it can provide sexual and urinary benefits compared with open radical prostatectomy for 3 years, robot-assisted radical prostatectomy had a much greater QALY gained (0.0419). If we assumed that it can lead to a lower risk of cancer recurrence, robot-assisted radical prostatectomy had a 0.0213 QALY gained over 10 years compared with open radical prostatectomy. Under assumptions that it can provide substantial functional and oncological benefits compared with open radical prostatectomy, the ICER was $83,921 per QALY gained.
Discussion
Likelihood of Robot-Assisted Radical Prostatectomy Being Cost-Effective
Our economic evaluation showed that compared with open radical prostatectomy, robot-assisted surgery had substantially higher costs and slightly greater QALYs, resulting in a very high ICER. Results were robust in the one-way and probabilistic sensitivity analyses. When using Ontario and Canadian data for the lengths of stay and operating room times, we found the ICERs were even higher than those in the base case. In probabilistic sensitivity analysis, almost all simulated results were beyond the commonly used willingness-to-pay threshold in Ontario (Figure 19).
We also explored the likelihood of robot-assisted radical prostatectomy being cost-effective in several scenarios that favoured it. For example, after we excluded the capital cost of purchasing the robotic surgical system, the ICER decreased substantially from $5.2 million per QALY in the base case to $2.7 million per QALY. However, this is still significantly higher than commonly used thresholds in Canada. Increasing the volume of robotic-assisted radical prostatectomies and the service life of the surgical system would also reduce the ICER, but it would still be higher than $2.7 million per QALY. More importantly, the large volume would lead to a greater budget increase.
The results in our long-term model showed that the ICER would be $83,921 per QALY gained if robot-assisted radical prostatectomy had substantially more favourable oncological and functional outcomes, relative to open radical prostatectomy. Based on the available clinical evidence, this should be viewed as a highly speculatively analysis.
We did not conduct a scenario analysis for patients with a high risk of cancer progression since, in Ontario, robot-assisted radical prostatectomy is commonly used for patients with low- or intermediate-risk cancer. However, the results in our 1-year base case model would be applicable for patients with high-risk cancer, under the assumption that there is no difference in oncological outcomes between the two groups.
Since the RCT found that both treatments resulted in the same length of time away from work,26 we did not conduct analyses for productivity loss.
Magnitude of Clinical Benefit of Robot-Assisted Radical Prostatectomy in Canada
Since the high ICER in the present study is largely driven by the small QALY gained from robot-assisted radical prostatectomy, we explored the potential magnitude of its clinical benefit. There are several published studies comparing robot-assisted and open radical prostatectomy in Canada. The comparative studies in British Columbia and Alberta showed that robotic-assisted and open radical prostatectomy have small differences in clinical outcomes.68,135–137
Gagnon et al68 analyzed 200 consecutive robotic-assisted radical prostatectomies by one surgeon (after a learning curve of 70 cases) and 200 consecutive open radical prostatectomies by another surgeon.68 The authors concluded that both treatments had comparable lengths of stay, transfusion rates, positive surgical margin rates, and rates of postoperative urinary incontinence. However, compared with open radical prostatectomy, robot-assisted radical prostatectomy was associated with68:
A higher 90-day postoperative complication rate: 22% versus 11.5%, P = .007
A longer operative time: skin to skin, 234 versus 114 minutes, P < .001
A higher cost: incremental cost of $5,629 per procedure
We identified three conference abstracts involving a cohort of 1,019 consecutive patients (815 undergoing robot-assisted versus 204 open radical prostatectomy) with clinically localized prostate cancer in Alberta.135–137 However, we did not find a full-text publication of this study. In this cohort, patients in both groups had comparable short-term surgical outcomes (complications within 90 days of surgery) and oncologic outcomes (biochemical recurrence-free survival and positive surgical margins). However, robot-assisted radical prostatectomy was associated with an increased chance of achieving pentafecta (defined as no biochemical recurrence, continent, potent, negative surgical margins, and no complications) at 12 months. The authors also found that the surgeon significantly impacted the clinical outcomes.135
Also, a retrospective study by Rush et al72 of Ontario's population showed that after adjusting for potential confounders, robot-assisted and open radical prostatectomy did not result in clinically significant differences in health-related quality of life, as measured by the Patient-Oriented Prostate Utility Scale.
In addition, although there were no statistics available to compare the surgical outcomes of the two treatments in Canada, the risks of unplanned readmission within 30 days postsurgery were similar, at 3.8% for open radical prostatectomy and 3.9% for robot-assisted radical prostatectomy (fiscal years 2009/10–2011/12).110
Since robot-assisted radical prostatectomy is presently used only in select teaching hospitals in Canada, it is appropriate to limit the comparison of robot-assisted to open radical prostatectomy within high-volume teaching hospitals. In recent years, the improvements of surgical techniques in open radical prostatectomy and anaesthesia have substantially reduced the risk of surgical complications and decreased the lengths of hospital stay.68,138,139 Thus, the historical benefits of robot-assisted radical prostatectomy (e.g., the lower surgical complication rates and shorter lengths of stay) have narrowed over time.138 Also, in high-volume hospitals such as Duke University Medical Center, the transfusion rates of both treatments were also comparable.138
Pierorazio et al139 found that, excluding the “off-pathway” patients (e.g., those experiencing ileus, urine leak, anemia, or re-exploration for bleeding), the average length of stay for all radical prostatectomies was reduced from 7.7 days in 1991 to 1.6 days in 2010 at Johns Hopkins University. Both procedures showed comparable complication rates and lengths of stay (1.87 days for open radical vs. 1.96 days in robot-assisted prostatectomy) since 2005.
In summary, the clinical data in Canada suggest that the difference in various outcomes between the two procedures is very small. The historical benefit of robot-assisted radical prostatectomy has narrowed over time because of the improvements in open techniques as well as contemporary patient-management strategies.
Study Strengths
Our study has the following strengths:
We estimated the costs and QALYs based on the most recent RCT comparing these treatments. This is considered higher-quality evidence compared with earlier observational studies
We captured the temporary health-related quality of life benefit immediately after robot-assisted radical prostatectomy
Our 1-year model and long-term Markov model explored the uncertainty of the potential benefits of robot-assisted radical prostatectomy in various scenarios
Study Limitations
The following limitations should be noted when interpreting the findings of this analysis:
Resource use (e.g. operating room time) estimates for both treatments were largely derived from a single RCT from Australia with 12 weeks’ (short term) follow-up
Utilities of these treatments were not available in the RCT; thus, we derived the estimates of longitudinal utility values using indirect methods
No high-quality evidence was available to allow for the comparison of sexual function, urinary function, or oncological outcomes of treatments in long-term follow-up
We did not control for the effect of surgeons’ skills
Conclusion
The cost of using the robotic surgical system is relatively high, while the health benefit is relatively small. Therefore, robot-assisted radical prostatectomy does not appear to be cost-effective in patients with clinically localized prostate cancer.
BUDGET IMPACT ANALYSIS
We conducted a budget impact analysis to estimate the cost burden of continuing public funding of robot-assisted radical prostatectomy at an increased adoption over the next 5 years. All costs are reported in 2016 Canadian dollars.
Objective
This study aimed to assess the budget impact of purchasing robotic surgical systems and continuing public funding of the robot-assisted radical prostatectomy procedure at an increased adoption in men with clinically localized prostate cancer. We conducted this assessment within the context of the Ontario Ministry of Health and Long-Term Care.
Methods
Target Population
The target population was men with newly diagnosed, clinically localized prostate cancer who were eligible for radical prostatectomy.
We estimated the volume of robot-assisted radical prostatectomy in next 5 years based on data from IntelliHealth Ontario. This database showed that the total volume of radical prostatectomy procedures was in decline from 2011 to 2013 but stabilized in 2014 and 2015 (Table 38). However, the volume of robot-assisted radical prostatectomy has constantly been on the rise. In 2015, it reached a total of 816 procedures (34% of the total volume of all radical prostatectomies).
Table 38:
Volumes of the Different Types of Prostatectomy Procedures Performed in Ontario
| Year | RARP | ORP | LRP | Total |
|---|---|---|---|---|
| 2011 | 229a | 2,695 | 335a | 3,259 |
| 2012 | 456a | 2,199 | 242a | 2,897 |
| 2013 | 692 | 1,704 | 157 | 2,553 |
| 2014 | 756 | 1,539 | 110 | 2,405 |
| 2015 | 816 | 1,492 | 98 | 2,406 |
Abbreviations: RARP, robot-assisted radical prostatectomy; ORP, open radical prostatectomy; LRP, laparoscopic radical prostatectomy.
Estimated number. RARP has had a unique procedure code since 2012 in Ontario. Prior to 2012, RARP shared the same code with LRP. Thus, IntelliHealth data do not distinguish between LRP and RARP in 2012 or before 2012. The volume of RARP in 2011 and 2012 was obtained from experts. The volume of LRP in 2011 and 2012 was estimated by subtracting RARP from the total volume of LRP plus RARP. Note: the volumes of RARP from 2013 to 2015 from the two sources (IntelliHealth Ontario and experts) were slightly different.
Based on the trends in the database, we assumed that in the next 5 years, the volume of total procedures and laparoscopic procedures would be fixed at 2,400 and 48 cases (2%), respectively. And we assumed the adoption of robot-assisted radical prostatectomy would gradually increase to 40% in year 1, 45% in year 2, 50% in year 3, 55% in year 4, and 60% in year 5.
In practice, patients’ characteristics are not identical for robot-assisted and open radical prostatectomy. In Ontario, open radical prostatectomy is performed in patients with various risk profiles, while robot-assisted radical prostatectomy is typically used for patients with a low or intermediate risk of cancer progression.72 Open radical prostatectomy is performed in teaching hospitals and community health centres. But robot-assisted radical prostatectomy is generally conducted exclusively in teaching hospitals. We therefore think it is reasonable to assume that robot-assisted surgery can replace open radical prostatectomy in some instances.
In Canada, Alberta has the highest proportion of robot-assisted radical prostatectomy procedures: about 60% in fiscal years 2011/12 and 2012/13.140 Based on these data, we estimated its adoption in Ontario would be as high as 60% in year 5.
Table 39 shows the expected volumes of robot-assisted radical prostatectomy in Ontario in the next 5 years.
Table 39:
Expected Volumes of Robot-Assisted Radical Prostatectomy in the Next 5 Years in Ontario
| Type of Prostatectomy | Expected Number of Patients | ||||
|---|---|---|---|---|---|
| Year 1 (40% RARP) | Year 2 (45% RARP) | Year 3 (50% RARP) | Year 4 (55% RARP) | Year 5 (60% RARP) | |
| Total radical prostatectomies | 2,400 | 2,400 | 2,400 | 2,400 | 2,400 |
| RARP | 960 | 1,080 | 1,200 | 1,320 | 1,440 |
| ORP | 1,392 | 1,272 | 1,152 | 1,032 | 912 |
| LRP | 48 | 48 | 48 | 48 | 48 |
Abbreviations: LRP, laparoscopic radical prostatectomy; ORP, open radical prostatectomy; RARP, robot-assisted radical prostatectomy.
Resources and Canadian Costs
Based on the undiscounted results from our base case economic evaluation model, we estimated the health care cost of two treatments in the first year. (We did not apply discounting in estimating the equivalent annual cost of capital investment of the robotic system.) The costs included the purchase costs of the da Vinci Surgical System and its service contract, the hospital's overhead costs for using the robotic system, costs for disposables, hospitalization costs, physician fees, and the costs of perioperative complications. More details of the cost components included are presented in the primary economic analysis.
Analysis
We explored the budget impact of publicly funding robot-assisted radical prostatectomy, using an increased adoption compared with the 2015 rate of 34%. We assumed that open and robot-assisted radical prostatectomy do not have any difference in cost after 1 year postsurgery.
The estimated costs of both treatments in the first year in the base case and scenario analyses are summarized in Table 40. In scenario one, we used the local resource use data in Ontario. In scenario two, we excluded the cost of the robotic system, assuming it was donated. In scenario three, we excluded all capital costs related to using the robotic surgical system and the cost of maintenance, assuming that those costs are fixed for a hospital in the next few years, regardless of the volume of robotic surgeries, and that no new robotic systems are purchased in hospitals in Ontario.
Table 40:
Estimated 1-Year Per-Patient Cost for Robot-Assisted and Open Radical Prostatectomya
| Scenario | RARP, $b | ORP, $b |
|---|---|---|
| Base case: including the cost of purchasing the robotic system and cost for maintenancea | 19,819 | 14,369 |
| Scenario analysis 1: using the length of stay and operating room data for the Ontario populationa | 18,461 | 10,814 |
| Scenario analysis 2: excluding the cost of purchasing the robotic systemc | 17,424 | 14,369 |
| Scenario analysis 3: excluding the capital cost of using the robotic system and the cost for maintenance | 15,339 | 14,369 |
Abbreviations: ORP, open radical prostatectomy; RARP, robot-assisted radical prostatectomy.
Not applying discounting in estimating the equivalent annual cost of the capital investment of the robotic system.
All costs are in 2016 Canadian dollars. Some numbers may appear inexact because of rounding.
Although excluding the capital cost of the robotic system, it included the hospital's overhead cost and cost for annual maintenance of the robotic system.
The budget impact analysis was conducted using Excel 2013 (Microsoft, Redmond, Washington).
Results
Table 41 presents the projected total costs of robot-assisted and open radical prostatectomy at the current adoption and the increased adoption of robot-assisted radical prostatectomy. It also shows the expected net budget impact in the next 5 years.
Table 41:
Total Costs and Net Budget Impact for an Increasing Adoption for RARP Versus Continued Current Adoption
| Scenario | Adoption of RARP | Results, $a | ||||
|---|---|---|---|---|---|---|
| Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | ||
| Base case | ||||||
| Total cost of RARP and ORP | Current: 34% | 38,243,500 | 38,243,500 | 38,243,500 | 38,243,500 | 38,243,500 |
| Increasing | 39,028,216 | 39,682,146 | 40,336,075 | 40,990,005 | 41,643,934 | |
| Net budget impact | 784,716 | 1,438,645 | 2,092,575 | 2,746,505 | 3,400,434 | |
| Scenario analysis 1: using Ontario data for length of stay and operating room time | ||||||
| Total cost of RARP and ORP | Current: 34% | 31,675,109 | 31,675,109 | 31,675,109 | 31,675,109 | 31,675,109 |
| Increasing | 32,776,312 | 33,693,982 | 34,611,651 | 35,529,320 | 36,446,990 | |
| Net budget impact | 1,101,203 | 2,018,873 | 2,936,542 | 3,854,211 | 4,771,881 | |
| Scenario analysis 2: excluding the capital cost of the robotic surgical system | ||||||
| Total cost of RARP and ORP | Current: 34% | 36,289,180 | 36,289,180 | 36,289,180 | 36,289,180 | 36,289,180 |
| Increasing | 36,729,016 | 37,095,546 | 37,462,075 | 37,828,605 | 38,195,134 | |
| Net budget impact | 439,836 | 806,365 | 1,172,895 | 1,539,425 | 1,905,954 | |
| Scenario analysis 3: excluding the costs related to using the robotic surgical system and maintenance | ||||||
| Total cost of RARP and ORP | Current: 34% | 34,587,820 | 34,587,820 | 34,587,820 | 34,587,820 | 34,587,820 |
| Increasing | 34,727,416 | 34,843,746 | 34,960,075 | 35,076,405 | 35,192,734 | |
| Net budget impact | 139,596 | 255,925 | 372,255 | 488,585 | 604,914 | |
Abbreviations: ORP, open radical prostatectomy; RARP, robot-assisted radical prostatectomy.
Some numbers may appear inexact because of rounding. All costs are in 2016 Canadian dollars.
In the base case analysis, an increasing adoption would lead to a budget increase of about $0.8 million in the first year. With a continued trend of increasing adoption, the net budget increase would reach about $3.4 million by year 5. When we excluded the cost of the robotic surgical system and its maintenance costs, the net budget impact decreased substantially to $0.14 million in year 1 and $0.6 million in year 5.
Discussion
There is some debate whether the capital cost for the robotic surgical system should be included in cost analyses.102 Some published economic analyses have excluded the capital cost of the robotic system because, for example, insurance companies do not directly pay for capital costs.106 The existing robotic surgical systems in use in Ontario have largely been purchased through charitable donations from hospital foundations. Per-procedures costs for disposable items are also sometimes funded through charitable donations.
We considered the following when including the capital cost in the base case analysis:
The donation is often from hospital foundations, not the manufacturer. If not used to purchase a robotic system, hospital foundation donation funds could be used for other health care services
Not every hospital offering radical prostatectomy surgery will receive a donated robotic surgical system. Even if a hospital receives donated robotic equipment, it may not receive another donated system or an upgraded system when the equipment needs to be replaced. In the long-term, the capital costs of robotic systems may need to be covered by public funds
Nevertheless, our sensitivity analyses did address a scenario without the capital cost of purchasing the robotic equipment.
Also, the capital cost of the robotic system (da Vinci Si) has increased substantially, from $2.8 million to $4.3 million, compared with that reported in a 2011 report published by the Canadian Agency for Drugs and Technologies in Health.111 The changes in exchange rate largely contributed to the increased purchasing cost ($1 USD = $1.02 CAD in the 2011 report111 vs. $1 USD = $1.32 CAD in January–August 2016).
A similar increase was identified for the costs of disposables, from $2,542 per procedure in the 2011 report111 to $3,476 in our present analysis. Aside from the uncertainty of the exchange rate, generally the costs for robotic systems and disposables increase a few percent yearly.
Operating rooms used for robotic surgery need to be bigger than regular operating rooms. It was suggested that the operating room should be at least 52 m2 and optimally 65 to 67 m2.26,113 Also, it is preferable to use a dedicated operating room so that the robotic system does not get damaged in transit from one room to another.26,113 However, it is difficult to accurately estimate the cost to upgrade an operating room to use the robotic surgical system, and the opportunity cost of the dedicated operating room for the robotic system. According to consulted experts, the overhead cost for the hospital of using robotics was estimated to be about 30% of the capital cost. Thus, we estimated that in addition to the cost of purchasing the robotic surgical system, it costs about $1 million for the hospital to use it.
Also, a dedicated robotic surgery team would be required to perform the surgeries.111 Thus, the true budget impact of robot-assisted surgeries may be even higher than our current estimate.
Finally, we did not include the assumption of a reduced adoption of robot-assisted radical prostatectomy. However, these results would be straightforward to calculate.
Conclusion
The current adoption of robot-assisted radical prostatectomy in Ontario is 34%. If the adoption continues to increase, this may lead to a considerable budget increase.
PUBLIC AND PATIENT ENGAGEMENT
Background
Public and patient engagement explores the lived experience of a person with a health condition, including how the condition and its treatment affect the patient, the patient's family or other caregivers, and the patient's personal environment. Public and patient engagement is intended to increase awareness and build appreciation for the needs, priorities, and preferences of the person at the centre of a treatment program. Insights gained through public and patient engagement provide an in-depth picture of lived experience, through an intimate look at the values that underpin the experience.
Lived experience is a unique source of evidence about the personal impact of a health condition and how that condition is managed, including what it is like to navigate the health care system with that condition, and how technologies might or might not make a difference in people's lives. Information shared from lived experience can both identify and supplement gaps or limitations in published research (e.g., outcome measures that do not reflect what is important to those with lived experience).141–143 Additionally, lived experience can provide information or perspectives on the ethical and social values implications of technologies and treatments. Because the needs, priorities, preferences, and values of those with lived experience in Ontario are not often adequately explored by published literature, Health Quality Ontario makes an effort to reach out to, and directly speak with, people who live with the health condition, including those who have experience with the intervention in question.
Needs Assessment
For robot-assisted radical prostatectomy, the scope and direction of patient and public engagement were determined through a formal needs assessment by the Public, Patient, and Caregiver Engagement team at Health Quality Ontario. The purpose of this needs assessment was threefold:
To determine if developing an evidence stream of lived experience would add value to the evidence-based analysis phase of the health technology assessment
To define the goals and objectives of engagement, as needed
To scope out the type of engagement activity that might be best for this project, as needed
To complete the needs assessment, we read background information on robot-assisted radical prostatectomy. This included reviewing the clinical review plan and consulting with clinical experts in the field. We also performed a qualitative literature search on patient-centred outcomes related to surgical procedures.
The needs assessment considered whether patient engagement would yield additional and relevant evidence on three dimensions: patient preferences and values in decision-making, patient preferences and values around outcomes, and health equity.
Patient Preferences and Values in Decision-Making
When we are assessing a health technology, engaging patients can often help us learn about the preferences and values that could inform a patient's decision to use the technology.
For robot-assisted prostatectomy, we consulted clinical experts, who were uncertain how much patient preference impacts the type of surgery patients receive. The choice between robot-assisted radical prostatectomy versus open or laparoscopic radical prostatectomy is often not within a patient's control for a number of reasons:
Availability of the robotic technology
Availability of an experienced surgeon
Surgeon preference
Type of surgery preferred at a hospital that is close to where the patient lives
Surgeons often specialize in one kind of surgery or another, not both. A patient rarely has a choice over which surgeon or type of surgery they can access. While direct patient engagement could help us learn patients’ overall impressions and preferences regarding both types of surgery, their preferences and values would likely have little impact on their access to the technology under review.
Patient Preferences and Values Around Outcomes
Patients can also provide insights about the outcomes that are most important to them. For this health technology, the clinical review also included literature on patient values and preferences related to surgical outcomes, such as blood loss, pain, and days lost to work. Because these outcomes were included in the clinical literature, the need for direct patient engagement or a qualitative literature review was low.
Health Equity
Patient engagement can often highlight health equity issues related to the technology being assessed that are not evident in the published literature. In the case of robot-assisted radical prostatectomy, we were able to identify a possible equity issue—geographic access—through the clinical and economic data we examined. However, improved clinical outcomes are not associated with robot-assisted radical prostatectomy, lessening any inequity concerns for those patients who do not live near health centres offering the procedure.
Clinical and economic data include information about how access to the surgery is clustered around certain centres, rather than distributed equally across the province. There is a natural tendency of surgeons and surgery types to be drawn to the same centres, rather than distributed equally across the province. This can affect patient access to these surgeries, owing to their geographic location.
Conclusion
After careful consideration of these factors within the needs assessment, we concluded that direct patient engagement for this health technology assessment would provide limited value and impact to this project. Patient engagement was therefore not prioritized for this health technology assessment.
Acknowledgments
The medical editor was Susan Harrison. Others involved in the development and production of this report were Arshia Ali, Tanveer Singh, Kellee Kaulback, Ana Laing, Claude Soulodre, Andrée Mitchell, Vivian Ng, Anil Thota, Nancy Sikich, and Irfan Dhalla.
We are very grateful to various clinical experts for lending their expertise to this project. The statements, conclusions, and views expressed in this report do not necessarily represent the views of the consulted experts.
ABBREVIATIONS
- CI
Confidence interval
- GRADE
Grading of Recommendations Assessment, Development, and Evaluation
- ICER
Incremental cost-effective ratio
- IIEF
International Index of Erectile Function Questionnaire
- OR
Odds ratio
- PDE5
Phosphodiesterase type 5
- PSA
Prostate-specific antigen
- QALY
Quality-adjusted life-year
- RCT
Randomized controlled trial
GLOSSARY
- Base case
A projected or virtual scenario in which no changes are made to current practice. The base case is used for comparison with an alternative scenario in which the technology under review is used.
- Cohort
A group of individuals who share a common characteristic and who are part of a clinical trial or study.
- Cost-effective
Good value for money. The overall benefit of the technique or intervention justifies the cost.
- Cost–utility analysis
A type of analysis that estimates the value for money of an intervention by weighing the cost of the intervention against the improvements in length of life and quality of life. The result is expressed as a dollar amount per “quality-adjusted life-year” or QALY.
- Decision-analytic model
A chosen method of decision-making to be used when evaluating the trade-offs between competing values, such as when weighing the costs and benefits or harms of a test or intervention.
- Deterministic sensitivity analysis
A type of analysis that changes the variables to determine whether the final answer will change. The analysis is done by first setting values for each factor, and then substituting other possible values for one (in a one-way sensitivity analysis) or more (in a multi-way sensitivity analysis) factors to test how these changes affect the result.
- Discounting
A method that considers that costs and health benefits are worth more today than in the future.
- Dominance
A test or treatment is in a state of dominance over another when it is both more effective and less costly than the other treatment option.
- Incremental cost
The extra cost associated with using one test or treatment instead of another.
- Incremental cost-effectiveness ratio (ICER)
Determines “a unit of benefit” for an intervention by dividing the incremental cost by the effectiveness. The incremental cost is the difference between the cost of the treatment under study and an alternative treatment. The effectiveness is usually measured as additional years of life or as “quality-adjusted life-years.”
- Markov model
A type of modelling that measures the health state of a patient over the course of treatment. A patient may stay in one health state or move from one health state to another, depending on the effect of the treatment and the progression of the disease.
- Monte Carlo simulation
Determines the uncertainty in an economic model by running many trials of the model. In each trial, random numbers are assigned wherever values are uncertain to see how the model result changes.
- Prospective study
Sometimes called a prospective cohort study, a prospective study selects participants before they develop the outcome in question and observes them over time. A prospective study differs from a retrospective study, whose participants already have the outcome in question.
- Quality-adjusted life-year (QALY)
A measurement that takes into account both the number of years gained by a patient from a procedure and the quality of those extra years (ability to function, freedom from pain, etc.). The QALY is commonly used as an outcome measure in cost–utility analyses.
- Randomized controlled trial
A type of study in which subjects are assigned randomly into different groups, with one group receiving the treatment under study and the other group(s) receiving a different treatment or a placebo (no treatment) to determine the effectiveness of one approach compared with the other.
- Retrospective study
Sometimes called a historic cohort study, a retrospective study selects participants after they develop the outcome in question and looks back at their past. A retrospective study differs from a prospective study, whose participants do not already have the outcome in question.
- Sensitivity
The ability of a test to accurately identify persons with the condition tested for (that is, how well it returns positive results in persons who have the condition).
- Sensitivity analysis
Every evaluation contains some degree of uncertainty. Study results can vary depending on the values taken by key parameters. Sensitivity analysis is a method that allows estimates for each parameter to be varied to show the impact on study results. There are various types of sensitivity analyses. Examples include deterministic, probabilistic, and scenario.
- Statistical significance
The outcome of an analysis is statistically significant if the assumption that there is no effect (the “null hypothesis”) is sufficiently unlikely to be true. Typically, the outcome is considered statistically significant if there is less than a 5% chance that the outcome would have occurred if the null hypothesis were true.
- Systematic review
A process to answer a research question by methodically identifying and assessing all available studies that evaluate the specified research question. The systematic review process is designed to be transparent and objective and is aimed at reducing bias in determining the answers to research questions.
- Time horizon
Costs and outcomes are examined within a chosen time frame. In an economic evaluation, this time frame is referred to as the time horizon.
- Utility
The perceived value placed on a person's health status.
APPENDICES
Appendix 1: Literature Search Strategies
Clinical Literature Search Strategy
Databases: EBM Reviews—Cochrane Central Register of Controlled Trials <February 2016>, EBM Reviews—Cochrane Database of Systematic Reviews <2005 to April 20, 2016>, EBM Reviews—Database of Abstracts of Reviews of Effects <1st Quarter 2016>, EBM Reviews— Health Technology Assessment <1st Quarter 2016>, Embase <1980 to 2016 Week 16>, All Ovid MEDLINE(R) <1946 to Present>
Search Strategy:
-
1
exp Prostatic Neoplasms/ (285933)
-
2
Prostate/ (73484)
-
3
exp Neoplasms/ (6390985)
-
4
2 and 3 (37311)
-
5
(prostat* adj3 (cancer* or carcinoma* or neoplas* or tumo?r* or adenoma* or adenocarcinoma* or malignan*)).tw. (270937)
-
6
Prostatectomy/ (68799)
-
7
prostatectom*.tw. (62136)
-
8
or/1,4–7 (366595)
-
9
Robotic Surgical Procedures/ (1551)
-
10
Robotics/ (44514)
-
11
(robot* or RALRP or RALP or RARP or da vinci* or davinci*).tw. (63095)
-
12
or/9–11 (72656)
-
13
8 and 12 (9296)
-
14
Case Reports/ or Comment.pt. or Editorial.pt. or Letter.pt. or Congresses.pt. (4563899)
-
15
13 not 14 (8529)
-
16
exp Animals/ not (exp Animals/ and Humans/) (13432616)
-
17
15 not 16 (5450)
-
18
limit 17 to english language [Limit not valid in CDSR,DARE; records were retained] (5131)
-
19
limit 18 to yr=“2006 -Current” [Limit not valid in DARE; records were retained] (4801)
-
20
19 use pmoz,cctr,coch,dare,clhta,cleed (2111)
-
21
exp prostate tumor/ (178504)
-
22
prostate/ (73484)
-
23
exp neoplasm/ (6385702)
-
24
22 and 23 (37293)
-
25
(prostat* adj3 (cancer* or carcinoma* or neoplas* or tumo?r* or adenoma* or adenocarcinoma* or malignan*)).tw. (270937)
-
26
prostatectomy/ (68799)
-
27
prostatectom*.tw. (62136)
-
28
or/21,24–27 (352528)
-
29
robot assisted surgery/ (1552)
-
30
robotics/ (44514)
-
31
(robot* or RALRP or RALP or RARP or da vinci* or davinci*).tw. (63095)
-
32
or/29–31 (72727)
-
33
28 and 32 (9233)
-
34
(exp animal/ or nonhuman/) not exp human/ (9718093)
-
35
33 not 34 (9194)
-
36
Case Report/ or Comment/ or Editorial/ or Letter/ or conference abstract.pt. (8496106)
-
37
35 not 36 (5204)
-
38
limit 37 to english language [Limit not valid in CDSR,DARE; records were retained] (4798)
-
39
limit 38 to yr=“2006 -Current” [Limit not valid in DARE; records were retained] (4496)
-
40
39 use emez (2442)
-
41
20 or 40 (4553)
-
42
remove duplicates from 41 (2777)
-
43
41 use emez (2442)
-
44
41 use pmoz (1940)
-
45
41 use cctr (122)
-
46
41 use coch (7)
-
47
41 use dare (12)
-
48
41 use clhta (18)
-
49
41 use cleed (12)
Economic Literature Search Strategy
Databases: EBM Reviews—Cochrane Central Register of Controlled Trials <March 2016>, EBM Reviews—Cochrane Database of Systematic Reviews <2005 to April 20, 2016>, EBM Reviews—Database of Abstracts of Reviews of Effects <1st Quarter 2016>, EBM Reviews— Health Technology Assessment <1st Quarter 2016>, EBM Reviews—NHS Economic Evaluation Database <1st Quarter 2016>, Embase <1980 to 2016 Week 16>, All Ovid MEDLINE(R) <1946 to Present>
Search Strategy:
-
1
exp Prostatic Neoplasms/ (285942)
-
2
Prostate/ (73489)
-
3
exp Neoplasms/ (6391232)
-
4
2 and 3 (37312)
-
5
(prostat* adj3 (cancer* or carcinoma* or neoplas* or tumo?r* or adenoma* or adenocarcinoma* or malignan*)).tw. (270988)
-
6
Prostatectomy/ (68800)
-
7
prostatectom*.tw. (62143)
-
8
or/1,4–7 (366647)
-
9
Robotic Surgical Procedures/ (1551)
-
10
Robotics/ (44516)
-
11
(robot* or RALRP or RALP or RARP or da vinci* or davinci*).tw. (63126)
-
12
or/9–11 (72687)
-
13
8 and 12 (9298)
-
14
economics/ (249292)
-
15
economics, medical/ or economics, pharmaceutical/ or exp economics, hospital/ or economics, nursing/ or economics, dental/ (724001)
-
16
economics.fs. (375375)
-
17
(econom* or price or prices or pricing or priced or discount* or expenditure* or budget* or pharmacoeconomic* or pharmaco-economic*).tw. (670652)
-
18
exp “costs and cost analysis”/ (502959)
-
19
cost*.ti. (229501)
-
20
cost effective*.tw. (243277)
-
21
(cost* adj2 (util* or efficacy* or benefit* or minimi* or analy* or saving* or estimate* or allocation or control or sharing or instrument* or technolog*)).ab. (152210)
-
22
models, economic/ (131795)
-
23
markov chains/ or monte carlo method/ (117503)
-
24
(decision adj1 (tree* or analy* or model*)).tw. (32886)
-
25
(markov or markow or monte carlo).tw. (97100)
-
26
quality-adjusted life years/ (25825)
-
27
(QOLY or QOLYs or HRQOL or HRQOLs or QALY or QALYs or QALE or QALEs).tw. (49079)
-
28
((adjusted adj (quality or life)) or (willing* adj2 pay) or sensitivity analys*s).tw. (95126)
-
29
or/14–28 (2235309)
-
30
13 and 29 (929)
-
31
Case Reports/ or Comment.pt. or Editorial.pt. or Letter.pt. or Congresses.pt. (4564377)
-
32
30 not 31 (849)
-
33
32 use pmoz,cctr,coch,dare,clhta (230)
-
34
13 use cleed (13)
-
35
33 or 34 (243)
-
36
limit 35 to english language [Limit not valid in CDSR,DARE; records were retained] (214)
-
37
limit 36 to yr=“2006 -Current” [Limit not valid in DARE; records were retained] (205)
-
38
exp prostate tumor/ (178504)
-
39
prostate/ (73489)
-
40
exp neoplasm/ (6385949)
-
41
39 and 40 (37294)
-
42
(prostat* adj3 (cancer* or carcinoma* or neoplas* or tumo?r* or adenoma* or adenocarcinoma* or malignan*)).tw. (270988)
-
43
prostatectomy/ (68800)
-
44
prostatectom*.tw. (62143)
-
45
or/38,41–44 (352580)
-
46
robot assisted surgery/ (1552)
-
47
robotics/ (44516)
-
48
(robot* or RALRP or RALP or RARP or da vinci* or davinci*).tw. (63126)
-
49
or/46–48 (72758)
-
50
45 and 49 (9235)
-
51
Economics/ (249292)
-
52
Health Economics/ or exp Pharmacoeconomics/ (212687)
-
53
Economic Aspect/ or exp Economic Evaluation/ (388945)
-
54
(econom* or price or prices or pricing or priced or discount* or expenditure* or budget* or pharmacoeconomic* or pharmaco-economic*).tw. (670652)
-
55
exp “Cost”/ (502959)
-
56
cost*.ti. (229501)
-
57
cost effective*.tw. (243277)
-
58
(cost* adj2 (util* or efficacy* or benefit* or minimi* or analy* or saving* or estimate* or allocation or control or sharing or instrument* or technolog*)).ab. (152210)
-
59
Monte Carlo Method/ (49678)
-
60
(decision adj1 (tree* or analy* or model*)).tw. (32886)
-
61
(markov or markow or monte carlo).tw. (97100)
-
62
Quality-Adjusted Life Years/ (25825)
-
63
(QOLY or QOLYs or HRQOL or HRQOLs or QALY or QALYs or QALE or QALEs).tw. (49079)
-
64
((adjusted adj (quality or life)) or (willing* adj2 pay) or sensitivity analys*s).tw. (95126)
-
65
or/51–64 (1834609)
-
66
50 and 65 (781)
-
67
Case Report/ or Comment/ or Editorial/ or Letter/ or conference abstract.pt. (8496580)
-
68
66 not 67 (585)
-
69
68 use emez (372)
-
70
limit 69 to english language [Limit not valid in CDSR,DARE; records were retained] (336)
-
71
limit 70 to yr=“2006 -Current” [Limit not valid in DARE; records were retained] (310)
-
72
37 or 71 (515)
-
73
72 use emez (310)
-
74
72 use pmoz (164)
-
75
72 use cctr (7)
-
76
72 use coch (7)
-
77
72 use dare (3)
-
78
72 use clhta (12)
-
79
72 use cleed (12)
-
80
remove duplicates from 72 (369)
Appendix 2: Evidence Quality Assessment
Table A1:
Risk of Biasa Among Randomized Controlled Trials (Cochrane Risk of Bias Tool)
| Author, Year | Random Sequence Generation | Allocation Concealment | Blinding of Participants and Personnel | Incomplete Outcome Data | Selective Reporting | Other Bias |
|---|---|---|---|---|---|---|
| Robot-assisted vs. open radical prostatectomy | ||||||
| Yaxley et al, 201626 | Low | Low | Lowb | Low | Low | Highc,d |
| Robot-assisted radical vs. laparoscopic radical prostatectomy | ||||||
| Asimakopoulos et al, 201130 | Low | Unclear | Lowb | Low | Low | Highd |
| Porpiglia et al, 201329 | Low | Unclear | Lowb | Low | Low | Highd |
Possible risk of bias levels: low, high, and unclear.
Not possible to blind patients and surgeons because of the nature of the surgical intervention.
Only interim 12-week results, from an anticipated 2-year study. However, interim 3-month results were preplanned. Ended recruitment after nonsignificant differences between groups at 3 months.
No intention-to-treat analysis; per-protocol analysis.
Table A2:
Risk of Biasa Among Nonrandomized Trials (ROBINS-I Tool)
| Author, Year | Pre-intervention | At Intervention | Postintervention | ||||
|---|---|---|---|---|---|---|---|
| Confounding | Study Participant Selection | Classification of Interventions | Deviations From Intended Intervention | Missing Data | Measurement of Outcomes | Selection of Reported Results | |
| Robot-assisted vs. open radical prostatectomy | |||||||
| Ball et al, 200633 | Seriousb,c | Low | Low | Low | Low | Moderateg,h,i | Low |
| Bier et al, 201634 | Seriousb,c | Low | Low | Low | Low | Moderateg,h,i | Low |
| Breyer et al, 201035 | Seriousc | Moderated | Low | Low | Low | Moderateg,h,i | Low |
| Carlsson et al, 201036 | Seriousb,c | Moderated | Low | Low | Low | Low | Low |
| Davison et al, 201419 | Seriousc | Moderated | Low | Low | Low | Low | Low |
| Di Pierro et al, 201137 | Seriousc | Low | Low | Low | Low | Low | Low |
| Doumerc et al, 201038 | Seriousb,c | Moderated | Low | Low | Low | Moderateg,h,i | Low |
| Farnham et al, 200639 | Seriousb,c | Moderated | Low | Low | Low | Serious | Low |
| Ficarra et al, 200940 | Seriousc | Low | Low | Low | Low | Moderateg,h,i | Low |
| Fode et al, 201441 | Seriousc | Low | Low | Low | Moderatee | Low | Moderatej |
| Fracalanza et al, 200842 | Seriousb,c | Moderated | Low | Low | Low | Moderateg,h,i | Low |
| Geraerts et al, 201331 | Seriousc | Moderated | Low | Low | Moderatee | Low | Low |
| Haglind et al, 201543 | Seriousc | Moderated | Low | Low | Moderatee | Moderatef,g,h,i | Low |
| Ham et al, 200844 | Seriousb,c | Moderated | Low | Low | Low | Moderateg,h,i | Low |
| Hong et al, 201045 | Seriousb,c | Moderated | Low | Low | Low | Low | Low |
| Kim et al, 201146 | Seriousc | Moderated | Low | Low | Low | Low | Low |
| Kordan et al, 201047 | Seriousb,c | Moderated | Low | Low | Low | Moderatef,g,h,i | Low |
| Lott et al, 201549 | Seriousb,c | Low | Low | Low | Moderatee | Low | Low |
| Ludovico et al, 201350 | Seriousb,c | Moderated | Low | Low | Moderatee | Moderateg,h,i | Moderatej |
| Miller et al, 200751 | Seriousb,c | Low | Low | Low | Low | Moderateg,h | Low |
| Nelson et al, 200752 | Seriousb,c | Moderated | Low | Low | Low | Serious | Low |
| O'Malley et al, 200653 | Seriousb,c | Moderated | Low | Low | Low | Moderateg,h | Low |
| Philippou et al, 201254 | Seriousb,c | Low | Low | Low | Moderatee | Moderateg,h,i | Low |
| Thompson et al, 201355 | Seriousc | Low | Low | Low | Low | Low | Low |
| Wallerstedt et al, 201532 | Seriousc | Low | Low | Low | Moderatee | Moderateg,h,i | Low |
| Wood et al, 200756 | Seriousb,c | Moderated | Low | Low | Low | Moderateg,h,i | Low |
| Robot-assisted vs. laparoscopic radical prostatectomy | |||||||
| Asimakopoulos et al, 201357 | Seriousb,c | Low | Low | Low | Moderatee | Moderateg,h,i | Low |
| Berge et al, 201358 | Seriousc | Low | Low | Low | Low | Moderateg,h,i | Low |
| Ploussard et al, 200959 | Seriousb,c | Low | Low | Low | Low | Moderateg,h,i | Low |
| Ploussard et al, 201460 | Seriousb,c | Low | Low | Low | Moderatee | Moderateg,h,i | Low |
| Willis et al, 201261 | Seriousc | Low | Low | Low | Moderatee | Moderateg,h,i | Low |
Abbreviation: ROBINS-I, Risk of Bias in Non-randomized Studies—of Interventions.
Possible risk of bias levels: low, moderate, serious, critical, and no information.
Lack of adjustment for differences in baseline patient or tumour characteristics between groups.
No specific patient inclusion and exclusion criteria; unclear patient selection process (e.g., not stated whether patients were consecutive).
Unclear role of physician or patient in deciding treatment group allocation.
Potential differences in dropout rates or characteristics between groups.
Differences in surgeon experiences owing to multiple surgeons performing a type of radical prostatectomy.
Lack of validated measurements for sexual outcomes.
Lack of validated measurements for continence outcomes.
Biochemical recurrence or positive surgical margin as surrogate outcome for more patient-important outcome of cancer survival or mortality.
Some outcomes specified in the analysis were not reported.
Our first consideration was study design; we started with the assumption that randomized controlled trials (RCTs) are high quality whereas observational studies are low quality. We then took into account five additional factors—risk of bias, inconsistency, indirectness, imprecision, and publication bias. Limitations in these areas resulted in downgrading the quality of evidence. Finally, we considered three main factors that may raise the quality of evidence: the large magnitude of effect, the dose-response gradient, and any residual confounding factors.25 For more detailed information, please refer to the latest series of GRADE articles.25
As stated by the GRADE Working Group, the final quality score can be interpreted using the following definitions:
| High | We are very confident that the true prognosis (probability of future events) lies close to that of the estimate |
| Moderate | We are moderately confident that the true prognosis (probability of future events) is likely to be close to the estimate, but there is a possibility that it is substantially different |
| Low | Our confidence in the estimate is limited: the true prognosis (probability of future events) may be substantially different from the estimate |
| Very Low | We have very little confidence in the estimate: the true prognosis (probability of future events) is likely to be substantially different from the estimate |
Table A3:
GRADE Evidence Profile for Comparison of Robot-Assisted Versus Open Radical Prostatectomy
| Number of Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| Operative time | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 12 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Length of hospital stay | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 11 non-RCTs | Serious limitations (−1)a | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Estimated blood loss | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | Large magnitude of effect (+1) | ⊕⊕⊕ Moderate |
| 16 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | Large magnitude of effect (+1) | ⊕ Low |
| Transfusion rates | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 11 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Indwelling catheterization duration | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 7 non-RCTs | Serious limitations (−1)a | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Hospital readmission rates | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Complication rates | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 14 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Urinary function | |||||||
| 1 RCT | Serious limitations (−1)a | No serious limitations | No serious limitations | No serious limitations | Undetected | None | ⊕⊕⊕ Moderate |
| 12 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Erectile function | |||||||
| 1 RCT | Serious limitations (−1)a | No serious limitations | No serious limitations | No serious limitations | Undetected | None | ⊕⊕⊕ Moderate |
| 11 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)e | Undetected | None | ⊕ Very low |
| Health-related quality of life | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Pain | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Time to mobilization or return to work or activity | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 3 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Positive surgical margin rates | |||||||
| 1 RCT | No serious limitations | No serious limitations | Serious limitations (−1)f | Serious limitations (−1)d | Undetected | None | ⊕⊕ Low |
| 15 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | Serious limitations (−1)f | Serious limitations (−1)d | Undetected | None | ⊕ Very Low |
| Biochemical recurrence-free rates | |||||||
| 3 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | Serious limitations (−1)f | Serious limitations (−1)d | Undetected | None | ⊕ Very Low |
Abbreviations: RCT, randomized controlled trial.
Other than 1 RCT (where interim 3-month data are available), the rest of the evidence is limited to prospective comparative nonrandomized studies with methodological limitations. Follow-up period was limited in most studies. Observational studies start at low quality according to GRADE. Differences in baseline patient characteristics may impact results.
Inconsistencies within results.
Outcomes are related to hospital procedures and follow-up care.
Not powered to detect differences between groups.
Use of subjective, nonvalidated, or nonstandardized measurements for some outcomes.
Surrogate outcomes for cancer control. Other factors such as patient characteristics and risk stratification may also impact cancer control.
Table A4:
GRADE Evidence Profile for Comparison of Robot-Assisted Versus Laparoscopic Radical Prostatectomy
| Number of Studies (Design) | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Upgrade Considerations | Quality |
|---|---|---|---|---|---|---|---|
| Operative time | |||||||
| 2 RCTs | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 4 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Length of hospital stay | |||||||
| 1 RCT | No serious limitations | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 2 non-RCTs | Serious limitations (−1)a | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Estimated blood loss | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 4 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Transfusion rates | |||||||
| 1 RCT | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 3 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Indwelling catheterization duration | |||||||
| 2 RCTs | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 1 non-RCT | Serious limitations (−1)a | No serious limitations | Serious limitations (−1)c | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Complication rates | |||||||
| 2 RCTs | No serious limitations | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕⊕⊕ Moderate |
| 3 non-RCTs | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Urinary function | |||||||
| 2 RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | No serious limitations | Undetected | None | ⊕⊕ Low |
| 4 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)e | Undetected | None | ⊕ Very low |
| Erectile function | |||||||
| 2 RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | No serious limitations | Undetected | None | ⊕⊕ Low |
| 4 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | No serious limitations | Serious limitations (−1)e | Undetected | None | ⊕ Very low |
| Health-related quality of life | |||||||
| 1 non-RCT | Serious limitations (−1)a | No serious limitations | No serious limitations | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Positive surgical margin rates | |||||||
| 2 RCTs | No serious limitations | No serious limitations | Serious limitations (−1)f | Serious limitations (−1)d | Undetected | None | ⊕⊕ Low |
| 4 non-RCTs | Serious limitations (−1)a | Serious limitations (−1)b | Serious limitations (−1)f | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
| Biochemical recurrence-free rates | |||||||
| 2 RCTs | No serious limitations | No serious limitations | Serious limitations (−1)f | Serious limitations (−1)d | Undetected | None | ⊕⊕ Low |
| 2 non-RCTs | Serious limitations (−1)a | No serious limitations | Serious limitations (−1)f | Serious limitations (−1)d | Undetected | None | ⊕ Very low |
Abbreviations: RCT, randomized controlled trial.
Prospective comparative nonrandomized (observational) studies with methodological limitations. Observational studies start at low quality according to GRADE. Differences in baseline patient characteristics may impact results. Follow-up period was limited in most studies.
Inconsistencies within results.
Outcomes are related to hospital procedures and follow-up care.
Not powered to detect differences between groups.
Use of subjective, nonvalidated, or nonstandardized measurements for some outcomes.
Surrogate outcomes for cancer control. Other factors such as patient characteristics and risk stratification may also impact cancer control.
Appendix 3: Results of Clinical Evidence Review: Robot-Assisted Versus Open Radical Prostatectomy
Table A5:
Operative Time Outcomes for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Measure | Time, Minutes ± SDa | P Value | |
|---|---|---|---|---|
| Robot-Assisted Surgery | Open Surgery | |||
| Randomized controlled trial | ||||
| Yaxley et al, 201626 | Mean | 246 ± 55 | 280 ± 36 | < .001 |
| Nonrandomized studies | ||||
| Wood et al, 200756 | Mean | 210 ± 41 | 163 ± 29 | < .001 |
| Fracalanza et al, 200842 | Mean | 196 ± 45 | 127 ± 32 | < .001 |
| Doumerc et al, 201038 | Mean | 192 ± 52 | 148 ± 39 | < .001 |
| Hong et al, 201045 | Mean | 167 ± 5 | 169 ± 7 | Not significant |
| Di Pierro et al, 201137 | Mean | 330 ± 54 | 253 ± 41 | .020 |
| Philippou et al, 201254 | Mean | 125 ± 30 | 212 ± 71 | < .001 |
| Ficarra et al, 200940 | Median | 185 | 135 | < .001 |
| Lott et al, 201549 | Median | 272 (IQR 140–570) | 153 (IQR 110–260) | .01 |
| Ludovico et al, 201350 | Median | 221 | 103 | < .001 |
| Haglind et al, 201543 | Median | 236 (IQR 210–270) | 126 (IQR 102–186) | < .001 |
| Bier et al, 201634 | Median | 252 (range 131–428) | 186 (range 104–294) | < .001 |
| Wallerstedt et al, 201532 | Mean | 175 (range 45–575) | 103 (range 40–428) | < .001 |
Abbreviation: IQR, interquartile range; SD, standard deviation.
Where provided.
Table A6:
Length of Hospital Stay Outcomes for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Measure | Length of Stay, Days ± SDa | P Value | |
|---|---|---|---|---|
| Robot-Assisted Surgery | Open Surgery | |||
| Randomized controlled trial | ||||
| Yaxley et al, 201626 | Mean | 1.6 ± 2.6 | 3.3 ± 1.5 | < . 001 |
| Nonrandomized studies | ||||
| Wood et al, 200756 | Mean | 1.2 ± 0.8 | 1.3 ± 1.0 | .048 |
| Ham et al, 200844 | Mean | 3.9 ± 0.7 | 7.6 ± 1.3 | < .001 |
| Doumerc et al, 201038 | Mean | 2.8 ± 2.4 | 5.5 ± 2.7 | < .001 |
| Philippou et al, 201254 | Mean | 1.3 (range 1–3) | 3.8 (range 2–7) | < .001 |
| Wallerstedt et al, 201532 | Mean | 3.3 (range 2–53) | 4.1 (range 1–17) | < .001 |
| Nelson et al, 200752 | Mean | 1.2 | 1.2 | .27 |
| Fracalanza et al, 200842 | Median | 5 (range 5–6) | 8 (range 5–9) | .002 |
| Ficarra et al, 200940 | Median | 6 (range 5–8) | 7 (range 6–9) | .01 |
| Ludovico et al, 201350 | Median | 4 | 8 | < .001 |
| Lott et al, 201549 | Median | 2.6 (range 1–21) | 2.6 (range 1–21) | .01 |
| Haglind et al, 201543 | Median | 3 (2–4) | 4 (3–5) | < .001 |
Abbreviation: SD, standard deviation.
Where provided.
Table A7:
Estimated Blood Loss Outcomes for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Measure | Estimated Blood Loss, mL ± SDa | P Value | |
|---|---|---|---|---|
| Robot-Assisted Surgery | Open Surgery | |||
| Randomized controlled trial | ||||
| Yaxley et al, 201626 | Mean | 444 ± 294 | 1,338 ± 591 | < .001 |
| Nonrandomized studies | ||||
| Farnham et al, 200639 | Mean | 191 ± 149 | 664 ± 418 | < .001 |
| Wood et al, 200756 | Mean | 151 ± 97 | 707 ± 445 | < .001 |
| Ham et al, 200844 | Mean | 382 ± 213 | 897 ± 270 | < .001 |
| Ficarra et al, 200940 | Mean | 300 ± 234 | 500 ± 315 | < .001 |
| Hong et al, 201045 | Mean | 457 ± 281 | 1410 ± 901 | < .05 |
| Philippou et al, 201254 | Mean | 132 ± 151 | 513 ± 343 | < .001 |
| Bier et al, 201634 | Median | 400 (range 100–1,300) | 700 (range 234–1,600) | < .001 |
| Doumerc et al, 201038 | — | Blood loss < 499: 349/502 (69.7%) Blood loss 500–999: 147/502 (29.1%) Blood loss > 1,000: 6/502 (1.2%) |
Blood loss < 499: 208/212 (98.4%) Blood loss 500–999: 4/212 (1.6%) Blood loss > 1,000: 0/212 (0%) |
< .001 < .001 .25 |
| Fode et al, 201441 | Median | 150 (range 5–1,500) | 600 (range 50–4,320) | < .001 |
| Fracalanza et al, 200842 | Median | 300 (range 200–400) | 500 (range 250–650) | < .02 |
| Kordan et al, 201047 | Median | 100 (range 50–200) | 450 (range 300–600) | < .001 |
| Lott et al, 201549 | Median | 212 (range 50–1,200) | 487 (range 150–1,250) | .01 |
| Ludovico et al, 201350 | Not specified | 280 | 565 | < .001 |
| Miller et al, 200751 | — | Blood loss > 500: not reported | Blood loss > 500: not reported | < .001 |
| Haglind et al, 201543 | Mean | 100 (range 50–200) | 550 (range 350–800) | < .001 |
| Wallerstedt et al, 201532 | Mean | 185 (range 0–5,200) | 683 (range 50–8,000) | < .001 |
Abbreviation: SD, standard deviation.
Where provided.
Table A8:
Indwelling Catheterization Duration for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Outcome | Robot-Assisted Surgery | Open Surgery | P Value |
|---|---|---|---|---|
| Randomized controlled trials | ||||
| Yaxley et al, 201626 | Mean duration, days ± SDs | 8.2 ± 3.6 | 8.4 ± 3.3 | .59 |
| Nonrandomized studies | ||||
| Ham et al, 200844 | Mean duration, days ± SDs | 7.3 ± 0.6 | 7.7 ± 0.7 | < .001 |
| Wood et al, 200756 | Median duration, days | 10 | 10 | Not reported |
| Ludovico et al, 201350 | Median duration, days | 3 | 7 | < .001 |
| Doumerc et al, 201038 | Median duration, days | 6.3 (range 6–21) | 7.9 (range 6–20) | < .001 |
| Prolonged catheterization | 4% | 12% | Not reported | |
| Ficarra et al, 200940 | Median duration, days | 5 (range 4–7) | 6 (range 5–12) | < .001 |
| Geraerts et al, 201331 | Median duration, days | 11 | 12 | Not reported |
| Di Pierro et al, 201137 | Catheter-free rate at postoperative day 10 | 71/75 patients (95%) | 66/75 patients (88%) | Not reported |
| Catheter-free rate at postoperative day 15 | 71/75 patients (95%) | 75/75 patients (100%) | Not reported | |
| Prolonged catheterization (> 10 days after surgery) | 4/75 patients (5%) | 9/75 patients (12%) | .056 | |
Abbreviation: SD, standard deviation.
Table A9:
Rates of Hospital Readmission for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Outcome | Robot-Assisted Surgery, n (%) | Open Surgery, n (%) | P Value |
|---|---|---|---|---|
| Randomized controlled trial | ||||
| Yaxley et al, 201626 | Readmission at 3 months | 8 (5) | 12 (8) | .32 |
| Nonrandomized studies | ||||
| Nelson et al, 200752 | Readmission | 45/629 (7) | 18/374 (5) | .12 |
| Unscheduled clinical and emergency room visits | 63/629 (10) | 37/374 (10) | .95 | |
| Wallerstedt et al, 201532 | Readmission at 3 months | 163 (9.3) | 57 (7.7) | Not significant |
| Readmission leading to reoperation | 29 (1.6) | 13 (1.7) | Not significant | |
| Readmission not leading to reoperation | 116 (6.3) | 36 (4.6) | Not significant | |
Table A10:
Complication Rates for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Adverse Event/Complication | Rates of Complications, n (%)a | P Value | |
|---|---|---|---|---|
| Robot-Assisted Surgery | Open Surgery | |||
| Randomized controlled trial | ||||
| Yaxley et al, 201626 | Total complications | 6/157 (3.8) | 14/151 (9.3) | .052 |
| Nonrandomized studies | ||||
| Carlsson et al, 201036 | Bladder neck contracture | 3/1,253 (0.2) | 22/485 (4.5) | < .001 |
| Surgery for urinary incontinence | 7/1,253 (0.5) | 11/485 (2.2) | < .001 | |
| Death | 0 | 1/485 (0.2) | NS | |
| Rectal injury—perioperative | 1/1,253 (0.1) | 7/485 (1.4) | < .05 | |
| Rectal injury—postoperative | 1/1,253 (0.1) | 1/485 (0.2) | NS | |
| Small bowel injury | 1/1,253 (0.1) | 0 | NS | |
| Ureteral injury | 1/1,253 (0.08) | 0 | NS | |
| Femoral nerve injury | 2/1,253 (0.1) | 0 | NS | |
| Obturator nerve injury | 0 | 2/485 (0.4) | NS | |
| Pulmonary embolism | 2/1,253 (0.1) | 5/485 (1.0) | < .001 | |
| Myocardial infarction | 1/1,253 (0.07) | 2/485 (0.4) | NS | |
| Pneumonia | 0 | 4/485 (0.8) | < .001 | |
| Infected lymphocele | 1/1,253 (0.08) | 3/485 (0.6) | NS | |
| Wound infection | 6/1,253 (0.4) | 29/485 (5.9) | < .05 | |
| Anastomotic leakage (grade IIIa) | 8/1,253 (0.6) | 3/485 (0.6) | NS | |
| Anastomotic leakage (grade IIIb) | 5/1,253 (0.3) | 5/485 (1.0) | NS | |
| Surgical reintervention | 24/1,253 (1.9) | 14/485 (2.8) | NS | |
| Total complications | 197/1,253 (15.7) | 159/485 (32.8) | NR | |
| Ficarra et al, 200940 | Colon lesion | 1/103 (1.0) | 0 | NR |
| Rectal lesion | 1/103 (1.0) | 0 | NR | |
| Paralytic ileus | 1/103 (1.0) | 1/105 (1.0) | NR | |
| Cardiovascular complications | 0 | 2/105 (2.0) | NR | |
| Wound dehiscence | 0 | 1/105 (1.0) | NR | |
| Urethrovesical anastomotic stenosis | 3/103 (2.9) | 6/105 (5.7) | .32 | |
| Total complications | 10/103 (9.7) | 11/105 (10.5) | .85 | |
| Di Pierro et al, 201137 | Clavien grade—minor | 26/31 (83.9) | 18/39 (46.1) | .005 |
| Clavien grade—1 | 20/31 (64.5) | 13/39 (33.3) | NR | |
| Clavien grade—2 | 6/31 (19.4) | 5/39 (12.8) | NR | |
| Clavien grade—major | 5/31 (16.1) | 21/39 (53.8) | .02 | |
| Clavien grade—3a | 3/31 (9.7) | 15/39 (38.5) | NR | |
| Clavien grade—3b | 2/31 (6.5) | 6/39 (15.4) | NR | |
| Clavien grade—4a, 4b, 5 | 0 | 0 | NR | |
| Total complications | 28/75 (37.3) | 30/75 (40.0) | ||
| Doumerc et al, 201038 | Dindo—major (IIIa–V) | 4/212 (1.9) | 4/502 (0.8) | .38 |
| Geraerts et al, 201331 | Additional radiotherapy | 8/64 (12.5) | 15/116 (12.9) | NR |
| Conversion to open | 5/64 (7.8) | — | — | |
| Ham et al, 200844 | Conversion to open | 1/188 (0.5) (because of system malfunction) | — | — |
| Major—rectal injury | 1/188 (0.5) | 1/110 (1.0) | NR | |
| Major—infected hematoma | 0 | 0 | NR | |
| Minor—retention | 0 | 0 | NR | |
| Minor—anastomotic leakage | 0 | 2/110 (1.8) | NR | |
| Minor—lymphocele | 3/188 (1.6) | 3/110 (2.7) | NR | |
| Minor—ileus | 0 | 2/110 (1.8) | NR | |
| Total complications | 4/188 (2.1) | 8/110 (7.3) | < .001 | |
| Lott et al, 201549 | Clavien (grades I–IV) | 9/50 (18.0) | 8/34 (23.5) | < .001 |
| Nelson et al, 200752 | Caused unscheduled visit or hospital readmission: | |||
| Post-catheter retention | 2/629 (0.3) | 4/374 (1.1) | NR | |
| Lymphocele | 2/629 (0.3) | 1/374 (0.3) | NR | |
| Wound infection | 2/629 (0.3) | 5/374 (1.4) | NR | |
| DVT/pulmonary embolism | 3/629 (0.5) | 0 | NR | |
| DVT | 0 | 4/374 (1.1) | NR | |
| Urinary tract infection | 5/629 (0.8) | 4/374 (1.1) | NR | |
| Ileus | 6/629 (1.0) | 6/374 (1.6) | NR | |
| Epididymitis | 6/629 (1.0) | 0 | NR | |
| Clot retention | 9/629 (1.5) | 4/374 (1.1) | NR | |
| Urinary leakage/urinoma | 15/629 (2.4) | 4/374 (1.1) | NR | |
| Caused hospital readmission: | ||||
| Port hernia | 1/629 (0.15) | 0 | NR | |
| Pulmonary embolism | 1/629 (0.15) | 0 | NR | |
| Rectal injury | 1/629 (0.15) | 0 | NR | |
| Postoperative hemorrhage | 1/629 (0.15) | 0 | NR | |
| Clot retention | 2/629 (0.3) | 0 | NR | |
| Urinary tract infection | 3/629 (0.5) | 0 | NR | |
| Ileus | 20/629 (3.2) | 0 | NR | |
| DVT | 0 | 9/374 (2.5) | NR | |
| Lymphocele | 0 | 4/374 (1.1) | NR | |
| Fever | 0 | 1/374 (0.3) | NR | |
| Philippou et al, 201254 | Conversion to ORP | 0 | — | — |
| Clavien grades—minor (I + II) | 5/50 (10.0) | 9/50 (18.0) | .25 | |
| Clavien grades—major (III + IV) | 1/50 (2.0) | 2/50 (4.0) | .88 | |
| Total complications | 6/50 (12.0) | 11/50 (22.0) | .18 | |
| Anastomotic strictures | 2/50 (4.0) | 4/50 (8.0) | .68 | |
| Fracalanza et al, 200842 | Fever > 38°C | 2/37 (5.4) | 4/26 (15.4) | .22 |
| Hong et al, 201045 | Total complications | 3/37 (8.1) | 7/26 (26.9) | NR |
| Intraoperative venous gas embolism | 10/26 (38.5) | 20/25 (80.0) | < .05 | |
| Ludovico et al, 201350 | Clavien grades—minor (I + II) | 7/82 (8.5) | 4/48 (8.3) | NR |
| Clavien grades—major (III) | 1/82 (1.2) | 1/48 (2.1) | NR | |
| Total complications | 8/82 (9.8) | 5/48 (10.4) | .01 | |
| Wood et al, 200756 | Total complications | 25/117 (21.4) | 37/89 (41.6) | .002 |
| Breyer et al, 201035 | Bladder neck contracture | 4/293 (1.4) | 18/695 (2.6) | .12 |
Abbreviations: DVT, deep vein thrombosis; NR, not reported; NS, not significant; ORP, open radical prostatectomy.
Unless otherwise indicated.
Table A11:
Urinary Function Outcomes for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Outcome Definition | Time | Robot-Assisted Surgery | Open Surgery | P Value |
|---|---|---|---|---|---|
| Randomized controlled trial | |||||
| Yaxley et al, 201626 | EPIC urinary domain score | 3 months | 82.5 (95% CI 80–85) | 83.8 (95% CI 81.3–86.2) | .48 |
| Nonrandomized studies | |||||
| Davison et al, 201419 | Adjusted EPIC urinary summary | 6 months | 27.2 (17.2) | 20.7 (17.3) | NS |
| score (SD) | 12 months | 32.6 (20.8) | 25.4 (19.9) | NS | |
| Ball et al, 200633 | % baseline urinary function | 3 months | 58% | 62% | NS |
| (UCLA-PCI) | 6 months | 69% | 75% | NS | |
| Ficarra et al, 200940 | Mean time to continence | — | 25 ± 39 days | 75 ± 116 days | < .001 |
| Geraerts et al, 201331 | Mean time to continence | — | 16 days | 46 days | .026 |
| Kim et al, 201146 | Median time to continence | — | 3.7 months | 4.3 months | .161 |
| Ludovico et al, 201350 | Mean time to continence | — | 4.1 ± 0.8 months | 6.9 ± 1.4 months | < .001 |
Abbreviations: CI, confidence interval; EPIC, Expanded Prostate Cancer Index Composite; NS, not significant; UCLA-PCI, University of California–Los Angeles Prostate Cancer Index.
Table A12:
Erectile Function Outcomes for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Outcome Definition | Time | Robot-Assisted Surgery | Open Surgery | P Value |
|---|---|---|---|---|---|
| Randomized controlled trial | |||||
| Yaxley et al, 201626 | EPIC sexual domain score | 3 months | 38.9 (95% CI 34.8–43.0) | 35.0 (95% CI 30.9–39.1) | .18 |
| IIEF total score | 3 months | 30.1 (95% CI 26.5–30.8) | 27.6 (95% CI 24.3–30.8) | .31 | |
| Nonrandomized studies | |||||
| Davison et al, 201419 | Adjusted EPIC sexual summary score (SD) | 6 months | 27.2 (17.2) | 20.7 (17.3) | NS when |
| 12 months | 32.6 (20.8) | 25.4 (19.9) | adjusted | ||
| Ball et al, 200633 | % baseline sexual function (UCLA-PCI) | 3 months | 35% | 24% | < .05 |
| 6 months | 43% | 33% | NS | ||
| Lott et al, 201549 | IIEF-5 | 6 months | Median 13.1 (IQR 5–24) | Median 7.2 (IQR 5–21) | .01 |
| 12 months | Median 15.0 (IQR 5–24) | Median 8.6 (IQR 5–19) | .01 | ||
| Kim et al, 201146 | Erection sufficient for intercourse with or without PDE5 inhibitor | 6 months | 33.0% | 6.7% | Significant |
| 12 months | 57.1% | 28.1% | when | ||
| 24 months | 83.8% | 47.5% | |||
| Median time to potency (months) | — | 9.8 | 24.7 | < .001 | |
| Ludovico et al 201350 | Mean IIEF score | 12 months | 17.0 | 17.2 | .16 |
| Return to baseline IIEF with drugs (%) | 12 months | 19/82 (23.2) | 9/48 (18.7) | NS | |
| Return to baseline IIEF without drugs (%) | 12 months | 3/82 (3.7) | 3/48 (6.3) | NS | |
Abbreviations: CI, confidence interval; IIEF, International Index of Erectile Function Questionnaire; IQR, interquartile range; OR, odds ratio; NS, not significant; PDE5, phosphodiesterase-5; SD, standard deviation; UCLA-PCI, University of California–Los Angeles Prostate Cancer Index.
Table A13:
Outcomes for Health-Related Quality of Life for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Assessment Method | Time | Robot-Assisted Surgery | Open Surgery | P Value |
|---|---|---|---|---|---|
| Randomized controlled trial | |||||
| Yaxley et al, 201626 | SF-36 Physical Function score (95% CI) | Baseline | 59.77 (58.79–60.75) | 59.29 (58.17–60.41) | .52 |
| 12 weeks | 59.57 (58.51–60.63) | 59.39 (58.39–60.39) | .81 | ||
| SF-36 Mental Function score (95% CI) | Baseline | 47.34 (45.61–49.07) | 45.57 (43.71–47.43) | .17 | |
| 12 weeks | 49.52 (47.82–51.21) | 47.45 (45.49–49.41) | .97 | ||
| EPIC bowel domain score (95% CI) | Baseline | 94.10 (92.74–95.41) | 93.70 (92.23–95.21) | .72 | |
| 12 weeks | 94.50 (93.32–95.64) | 93.70 (91.80–95.54) | .46 | ||
| Revised Impact of Events Scale (RIES) score (95% CI) | Baseline | 12.65 (10.52–14.79) | 14.92 (12.52–17.33) | .16 | |
| 12 weeks | 4.30 (2.91–5.69) | 6.47 (4.65–8.29) | .06 | ||
| Hospital Anxiety and Depression Scale (HADS) score (95% CI) | Baseline | 7.82 (6.76–8.87) | 8.35 (7.25–9.46) | .49 | |
| 12 weeks | 5.26 (4.16–6.36) | 7.03 (5.78–8.28) | .04 | ||
| Nonrandomized study | |||||
| Ball et al, 200633 | % baseline UCLA-PCI bowel function domain | 3 months | 98 | 98 | NS |
| 6 months | 98 | 102 | NS | ||
| % baseline UCLA-PCI bowel bother domain | 3 months | 98 | 92 | NS | |
| 6 months | 99 | 99 | NS | ||
| % baseline AUA Symptom Index | 3 months | 106 | 97 | NS | |
| 6 months | 123 | 104 | NS | ||
| Miller et al, 200751 | SF-12 Physical Component | Baseline | 57.6 ± 2.4 | 56.9 ± 6.0 | .67 |
| Week 1 | 34.7 ± 7.6 | 31.7 ± 6.6 | .42 | ||
| Week 3 | 48.6 ± 3.9 | 42.1 ± 7.0 | < .001 | ||
| Week 6 | 56.4 ± 1.7 | 52.8 ± 4.7 | .003 | ||
| SF-12 Mental Component Score ± SDs | Baseline | 49.8 ± 6.2 | 45.7 ± 9.8 | .03 | |
| Week 1 | 52.7 ± 8.5 | 54.6 ± 7.6 | .03 | ||
| Week 3 | 55.5 ± 7.3 | 56.1 ± 5.8 | .25 | ||
| Week 6 | 57.4 ± 4.3 | 58.0 ± 4.7 | .30 | ||
Abbreviations: AUA, American Urological Association; CI, confidence interval; NS, not significant; SD, standard deviation; SF-12, 12-Item Short-Form Health Survey; SF-36, 36-Item Short-Form Health Survey; UCLA-PCI, University of California–Los Angeles Prostate Cancer Index.
Table A14:
Pain Outcomes for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Assessment Method | Pain Outcome | Robot-Assisted Surgery | Open Surgery | P Value |
|---|---|---|---|---|---|
| Randomized controlled trial | |||||
| Yaxley et al, 201626 | Surgical Pain Scale (0–10) score (95% CI) | Pain at rest at 24 hours | 3.01 (2.70–3.32) | 3.02 (2.69–3.35) | .97 |
| Pain during activities at 24 hours | 4.60 (4.25–4.95) | 5.83 (5.44–6.23) | < .001 | ||
| Worst pain at 24 hours | 5.30 (4.92–5.68) | 6.45 (6.08–6.81) | < .001 | ||
| Pain at rest at 1 week | 1.74 (1.47–2.01) | 1.98 (1.71–2.24) | .21 | ||
| Pain during activities at 1 week | 2.51 (2.19–2.82) | 3.19 (2.91–3.48) | .002 | ||
| Worst pain at 1 week | 2.37 (2.01–2.73) | 3.50 (3.12–3.88) | < .001 | ||
| Pain at rest at 6 weeks | 0.82 (0.55–1.10) | 0.78 (0.56–0.99) | .79 | ||
| Pain during activities at 6 weeks | 0.97 (0.73–1.21) | 1.07 (0.84–1.31) | .55 | ||
| Worst pain at 6 weeks | 0.72 (0.49–0.95) | 0.88 (0.62–1.13) | .37 | ||
| Pain at rest at 3 months | 0.39 (0.21–0.58) | 0.48 (0.29–0.66) | .54 | ||
| Pain during activities at 3 months | 0.55 (0.33–0.77) | 0.61 (0.38–0.83) | .70 | ||
| Worst pain at 3 months | 0.49 (0.26–0.72) | 0.48 (0.25–0.72) | .96 | ||
| Nonrandomized controlled trial | |||||
| Wallerstedt et al, 201532 | Patients using analgesics (%) | Pain in the operation wound at 3 months | 42 (2.4) | 49 (6.6) | Not significant |
| Pain in lower abdomen at 3 months | 149 (8.4) | 58 (7.8) | Not significant | ||
| Pain in upper abdomen at 3 months | 57 (3.2) | 20 (2.7) | Not significant | ||
| Wood et al, 200756 | Michigan Urological Survey pain scale (0–10) score ± SDs | Overall mean discomfort at 2 weeks | 2.8 ± 1.9 | 2.7 ± 1.9 | .76 |
| Overall mean discomfort at 6 weeks | 1.8 ± 1.8 | 1.4 ± 1.5 | .15 | ||
Abbreviation: SD, standard deviation.
Table A15:
Biochemical Recurrence-Free Rates for Robot-Assisted Versus Open Radical Prostatectomy
| Author, Year | Definition | Time | Biochemical Recurrence-Free Rates | P Value | |
|---|---|---|---|---|---|
| Robot-Assisted Surgery | Open Surgery | ||||
| Di Pierro et al, 201137 | 2 consecutive | 3 months | 66/75 (88%) | 68/75 (91%) | .71 |
| PSA ≥ 0.2 ng/mL | 12 months | 40/45 (89%) | 65/75 (87%) | .36 | |
| Philippou et al, 201254 | 2 consecutive PSA ≥ 0.2 ng/mL | 12 months | 92% | 88% | .51 |
| Breyer et al, 201035 | 2 consecutive PSA ≥ 0.2 ng/mL | 3 years | 81% | 87% | .02 |
Abbreviations: PSA, prostate-specific antigen.
Appendix 4: Results of Clinical Evidence Review: Robot-Assisted Versus Laparoscopic Radical Prostatectomy
Table A16:
Estimated Blood Loss for Robot-Assisted Versus Laparoscopic Radical Prostatectomy
| Author, Year | Estimated Blood Loss, mL ± SD | P Value | |
|---|---|---|---|
| Robot-Assisted Surgery | Laparoscopic Surgery | ||
| Randomized controlled trial | |||
| Porpiglia et al, 201329 | 202 ± 124 | 234 ± 150 | .20 |
| Nonrandomized studies | |||
| Ploussard et al, 200959 | 469 ± 380 | 889 ± 531 | < .01 |
| Willis et al, 201261 | 148 ± 87 | 173 ± 95 | .01 |
| Berge et al, 201358 | 190 ± 187 | 203 ± 226 | .05 |
| Ploussard et al, 201460 | 515.3 | 800 | < .001 |
Abbreviation: SD, standard deviation.
Table A17:
Complication Rates for Robot-Assisted Versus Laparoscopic Radical Prostatectomy
| Author, Year | Complication | Complication Rates | P Value | |
|---|---|---|---|---|
| Robot-Assisted | Laparoscopic | |||
| Randomized controlled trials | ||||
| Asimakopoulos et al, 201130 | Conversion to open | 0 | 0 | — |
| Deaths | 0 | 0 | — | |
| Cardiac complications | 0 | 0 | — | |
| “Paravesical” hematoma | 5/52 (10%) | 5/60 (8%) | — | |
| Venous thromboembolism | 1/52 (2%) | 0 | — | |
| Bronchitis | 1/52 (2%) | 0 | — | |
| Epididymitis | 1/52 (2%) | 0 | — | |
| Overall complication rate | 8/52 (15%) | 5/60 (8%) | .24 | |
| Porpiglia et al, 201329 | < 30-day Clavien 1–2 minor complications | 9/60 (15%) | 5/60 (8%) | — |
| < 30-day Clavien 3–4 major complications | 0 | 0 | — | |
| 31- to 90-day Clavien 1–2 minor complications | 1/60 (2%) | 2/60 (3%) | — | |
| 31- to 90-day Clavien 1–2 major complications | 0 | 0 | — | |
| Overall complication rate | 10/60 (17%) | 7/60 (12%) | .43 | |
| Nonrandomized studies | ||||
| Asimakopoulos et al, 201357 | Overall complication rate | 15% | 12% | .06 |
| Ploussard et al, 200959 | Urinary infection | 2% | 2% | — |
| Urinary sepsis | 0 | 1% | — | |
| Retention | 0 | 2% | — | |
| Renal insufficiency | 0 | 1% | — | |
| Pelvic hematoma | 1% | 2% | — | |
| Minor complications | 4% | 6% | — | |
| Postoperative bleeding | 0 | 1% | — | |
| Anastomotic leakage | 0 | 2% | — | |
| Rectal injury | 0 | 1% | — | |
| Overall complication rate | 4% | 8% | .16 | |
| Ploussard et al, 201460 | Anastomotic leakage | 2% | 10% | < .001 |
| Anastomotic stenosis | 1% | 2% | .08 | |
| Overall complication rate | 5% | 4% | .80 | |
Table A18:
Urinary Function Outcomes for Robot-Assisted Versus Laparoscopic Radical Prostatectomy
| Author, Year | Assessment Method | Outcome Definition | Time | Robot-Assisted Surgery | Laparoscopic Surgery | P Value |
|---|---|---|---|---|---|---|
| Randomized controlled trials | ||||||
| Asimakopoulos et al, 201130 | Telephone interview | No leakage/need of any protective pad | 3 months | 69% | 63% | .51 |
| 6 months | 88% | 75% | .06 | |||
| 12 months | 94% | 83% | .07 | |||
| — | Time to continence | — | 2.6 ± 4.2 months | 3.0 ± 2.9 months | .27 | |
| Porpiglia et al, 201329 | Telephone interview | 0 or 1 pad/day | 3 months | 48/60 (80%) | 37/60 (62%) | .03 |
| 6 months | 53/60 (88%) | 44/60 (73%) | .04 | |||
| 12 months | 57/60 (95%) | 53/60 (83%) | .04 | |||
| Nonrandomized studies | ||||||
| Berge et al, 201358 | Mailed questionnaire | UCLA-PCI urinary function score | Baseline | 94.1 ± 11.0 | 92.4 ± 13.4 | — |
| 3 months | 62.1 ± 26.2 | 60.2 ± 27.2 | .9 | |||
| 12 months | 77.4 ± 22.4 | 78.5 ± 23.4 | .2 | |||
| 36 months | 77.0 ± 23.2 | 80.4 ± 22.7 | .06 | |||
| Asimakopoulos et al, 201357 | Telephone interview | No leakage/need for any protective pad | 12 months | 123 (90%) | 74 (81%) | .05 |
| Ploussard et al, 201460 | Mailed questionnaire | No pad usage | 3 months | 50% | 39% | .02 |
| 6 months | 72% | 59% | .02 | |||
| 12 months | 75% | 69% | .18 | |||
| 24 months | 84% | 79% | .02 | |||
| Willis et al, 201261 | Questionnaire | 0 or 1 pad/day | Baseline | 99% | 100% | — |
| 3 months | 76% | 72% | .46 | |||
| 6 months | 87% | 89% | .67 | |||
| 12 months | 93% | 93% | .99 | |||
| Questionnaire | EPIC urinary summary | Baseline | 90.3 ± 1.4 | 89.8 ± 11.7 | — | |
| 3 months | 76.6 ± 15.3 | 76.5 ± 14.4 | .96 | |||
| 6 months | 82.7 ± 14.9 | 83.4 ± 13.3 | .70 | |||
| 12 months | 83.5 ± 16.1 | 85.6 ± 13.4 | .25 | |||
Abbreviations: EPIC, Expanded Prostate Cancer Index Composite; UCLA-PCI, University of California–Los Angeles Prostate Cancer Index.
Table A19:
Erectile Function Outcomes for Robot-Assisted Versus Laparoscopic Radical Prostatectomy
| Author, Year | Assessment Method | Outcome Definition | Time | Robot-Assisted Surgery | Laparoscopic Surgery | P Value |
|---|---|---|---|---|---|---|
| Randomized controlled trials | ||||||
| Porpiglia et al, 201329 | Questionnaire | IIEF-5 score > 17 | 3 months | 60% | 40% | .09 |
| 6 months | 66% | 49% | .14 | |||
| 12 months | 80% | 54% | .02 | |||
| Asimakopoulos et al, 201130 | Telephone interview | Erection firm enough for | 3 months | 63% | 13% | < .001 |
| 6 months | 75% | 22% | < .001 | |||
| 12 months | 77% | 32% | < .001 | |||
| — | Time to potency | — | 2.4 ± 2.3 days | 6.3 ± 5.2 days | < .001 | |
| Nonrandomized studies | ||||||
| Asimakopoulos et al, 201357 | Telephone interview | Erection firm enough for intercourse | 12 months | 66% | 40% | < .001 |
| Berge et al, 201358 | Mailed questionnaire | UCLA-PCI sexual function | Baselinea | 76.0 ± 15.7 | 75.3 ± 13.0 | — |
| 3 monthsa | 27.7 ± 16.8 | 24.8 ± 19.9 | .3 | |||
| 12 monthsa | 38.5 ± 25 | 35.2 ± 23.2 | .4 | |||
| 36 monthsa | 46.6 ± 28.6 | 43.2 ± 25.8 | .5 | |||
| Ploussard et al, 201460 | Questionnaire | Erection firm enough for | 3 months | 35% | 16% | .001 |
| 6 months | 42% | 20% | < .001 | |||
| 12 months | 58% | 32% | < .001 | |||
| 24 months | 69% | 55% | < .001 | |||
| Questionnaire | IIEF-5 score | Baseline | 17.7 | 17.6 | .85 | |
| 3 months | 8.8 | 6.1 | < .001 | |||
| 6 months | 10.6 | 7.0 | < .001 | |||
| 12 months | 11.5 | 8.2 | < .001 | |||
| 24 months | 13.5 | 8.1 | < .001 | |||
| Willis et al, 201261 | Questionnaire | Erection firm enough for intercourse | 3 months | 105/121 (87%) | 142/161 (88%) | .72 |
| 6 months | 77/110 (70%) | 122/161 (76%) | .29 | |||
| 12 months | 44/74 (60%) | 120/161 (75%) | .02 | |||
| Questionnaire | EPIC sexual summary score | Baseline | 77.1 ± 14.7 | 76.6 ± 15.5 | — | |
| 3 months | 47.2 ± 23.9 | 31.6 ± 23.8 | < .001 | |||
| 6 months | 54.9 ± 22.1 | 38.6 ± 24.3 | < .001 | |||
| 12 months | 54.5 ± 17.9 | 50.4 ± 23.5 | .33 | |||
Abbreviations: EPIC, Expanded Prostate Cancer Index Composite; IIEF, International Index of Erectile Function Questionnaire; UCLA-PCI, University of California–Los Angeles Prostate Cancer Index.
In patients with preoperative erectile function sufficient for intercourse and who received nerve-sparing surgery.
Table A20:
Biochemical Recurrence-Free Rates for Robot-Assisted Versus Laparoscopic Radical Prostatectomy
| Author, Year | Outcome Definition | Time | Robot-Assisted Surgery (%) | Laparoscopic Surgery (%) | P Value |
|---|---|---|---|---|---|
| Randomized controlled trials | |||||
| Asimakopoulos et al, 201130 | PSA level > 0.2 ng/mL on 2 consecutive measurements | 12 months | 92 | 97 | .3 |
| Porpiglia et al, 201329 | PSA level > 0.2 ng/mL | 12 months | 98 | 93 | .19 |
| Nonrandomized studies | |||||
| Asimakopoulos et al, 201357 | PSA level > 0.2 ng/mL on 2 consecutive measurements | Variable | 96 | 100 | .08 |
| Ploussard et al, 201460,144 | PSA level > 0.2 ng/mL | Variable | 90 | 82 | .82 |
Abbreviation: PSA, prostate-specific antigen.
Appendix 5: Health Technology Assessments and Systematic Reviews on Robot-Assisted Versus Open or Laparoscopic Radical Prostatectomy from 2011 to 2016
Table A21:
Summary of Recent Health Technology Assessments and Systematic Reviews on Robot-Assisted Versus Open or Laparoscopic Radical Prostatectomy
| Author, Year | Search Period and Databases | Included Studies | Comparison | Primary Conclusions |
|---|---|---|---|---|
| Duffey et al, 2011145 | Nov 2009—Nov 2010 (Medline) | 21 non-RCTs | Open | Robot-assisted: lower blood loss, transfusion rate, length of stay; no definitive conclusions for functional and oncological outcomes |
| Ferronha et al, 2011146 | 2000—Oct 2009 (PubMed, Medline) | 37 studies | Open, laparoscopic | Robot-assisted and laparoscopic: lower blood loss, transfusion rate Robot-assisted and open: shorter operative time No difference in PSM rate, continence, and potency between 3 groups |
| Health Information and Quality Authority, 2011147 | 1950—Mar 2011 (Medline, Embase, EBSCO, CINAHL, Cochrane, DARE, HTA database, Journal of Robotic Surgery) | 50 studies (1 RCT, 49 non-RCTs) | Open, laparoscopic | Robot-assisted vs. open: decreased PSM rate for pT2 cancer, improved functional outcomes (urinary continence and sexual function), reduced transfusion rate and length of stay; increased operative time Robot-assisted vs. laparoscopic: no significant differences for operative time, transfusion rate, conversion to open, sexual function, oncologic outcomes; marginal improvements in urinary continence, small reductions in length of stay |
| Ho et al, 2011111 | 1950—Oct 2009 (Medline, BIOSIS Previews, Embase, CINAHL, PubMed, CINAHL, Cochrane) | 51 non-RCTs | Open, laparoscopic | Robot-assisted vs. open: longer operative time; shorter length of stay; reduced PSM rate in pT2 (inconclusive for pT3), complication rate, blood loss, and transfusion rate; improved continence at 3 and 12 months, improved sexual function at 12 months Robot-assisted vs. laparoscopic: reduced operative time, length of stay, blood loss, and transfusion rate; inconclusive for PSM rate (for pT2 and pT3), complication rate, and urinary continence (at 3 months and 12 months) |
| Ficarra et al, 2012148 | Jan 2008—Aug 2011 (Medline, Embase, Web of Science) | 31 studies | Open, laparoscopic | 12- and 24-month potency rates in the robot-assisted group ranged from 54% to 90% and from 63% to 94%, respectively Robot-assisted radical prostatectomy vs. open: better potency rates at 12 months (OR 2.84, 95% CI 1.46–5.43, P = .002) Robot-assisted vs. laparoscopic for potency: nonsignificant improvement (OR 1.89, P = .21) Age, baseline potency status, comorbidities index, and extension of the nerve-sparing procedure were the most relevant preoperative and intraoperative predictors of potency recovery after robot-assisted radical prostatectomy Available data seemed to support the use of cautery-free dissection or pinpointed low-energy cauterization for robot-assisted radical prostatectomy |
| Ficarra et al, 2012149 | Jan 2008—Aug 2011 (Medline, Embase, Web of Science) | 51 studies | Open, laparoscopic | Robot-assisted vs. open: better 12-month urinary continence recovery (OR 1.53, P = .03) Robot-assisted vs. laparoscopic: better 12-month urinary continence recovery (OR 2.39, P = .006) 12-month urinary incontinence rates ranged from 4% to 31%, with a mean value of 16% using a no-pad definition; considering a no-pad or safety-pad definition, incidence ranged from 8% to 11%, with a mean value of 9% Age, body mass index, comorbidity index, lower urinary tract symptoms, and prostate volume were the most relevant preoperative predictors of urinary incontinence after robot-assisted radical prostatectomy Posterior musculofascial reconstruction with or without anterior reconstruction was associated with a small advantage in urinary continence recovery 1 month after robot-assisted radical prostatectomy Only complete reconstruction was associated with a significant advantage in urinary continence 3 months after robot-assisted radical prostatectomy (OR 0.76, P = .04) |
| Gleitsmann et al, 2012150 | Jan 2002—Jan 2012 (Medline, Medicaid Evidence-based Decisions Project) | 55 studies | Open, laparoscopic | Robot-assisted vs. open and laparoscopic: moderate evidence for reduced length of hospital stay, blood loss, and transfusion rates; moderate evidence for no difference in complication rates Robot-assisted vs. open: moderate evidence for increased operative time, reduced PSM rate, increased urinary continence and sexual function; low evidence of no difference in biochemical recurrence-free survival Robot-assisted vs. laparoscopic: moderate evidence for reduced operative time; no difference in PSM rate Moderate evidence that surgeons experienced in robot-assisted had improvements in most clinical outcomes (except blood loss) compared with less experienced surgeons |
| Moran et al, 2013151 | Jan 2000—Mar 2011 (Medline, Embase, CINAHL, Cochrane, Journal of Robotic Surgery) | 51 studies (1 RCT, 50 non-RCT) | Open, laparoscopic | Robot-assisted vs. open: reduced PSM rates for pT2 (RR 0.63, 95% CI 0.49–0.81, P < .001), improved sexual function at 12 months (RR 1.60, 95% CI 1.33–1.93, P < .001), improved urinary function at 12 months (RR 1.06, 95% CI 1.02–1.11, P < .01) Robot-assisted vs. laparoscopic: slightly improved urinary function at 12 months (RR 1.09, 95% CI 1.02—1.17, P = .013) |
| Novara et al, 2012152 | Jan 2008—Aug 2011 (Medline, Embase, Web of Science) | 79 studies | Open, laparoscopic | Mean PSM rate was 15% in all cancers and 9% in pathologically localized cancers, with some tumour characteristics being the most relevant predictors of PSMs Robot-assisted vs. open: similar overall PSM rate (OR 1.21, P = .19) and pT2 PSM rate (OR 1.25, P = .31); similar biochemical recurrence-free survival (HR 0.9, P = .53) Robot-assisted vs. laparoscopic: similar overall PSM rate (OR 1.12, P = .47) and pT2 PSM rate (OR 0.99; P = .97); similar biochemical recurrence-free survival (HR 0.5, P = .14) Several surgeon-related characteristics or procedure-related issues may play a major role in PSM rates 7-year biochemical recurrence-free survival estimates of about 80% |
| Novara et al, 2012153 | Jan 2008—Aug 2011 (Medline, Embase, Web of Science) | 72 studies | Open, laparoscopic | Robot-assisted: overall mean operative time 152 minutes; mean blood loss 166 mL; mean transfusion rate 2%; mean catheterization time 6.3 days; mean length of hospital stay 1.9 days; mean complication rate 9%, with most of the complications being of low grade—lymphocele/lymphorrhea (3.1 %), urine leak (1.8%), and reoperation (1.6%) were the most prevalent surgical complications Robot-assisted vs. open: lower blood loss (weighted mean difference 582.77, P < .001) and transfusion rate (OR 7.55, P < .001); no difference in operative time or complication rate Robot-assisted vs. laparoscopic: reduced transfusion rate (OR 2.56, P = .005); no difference in operative time, blood loss, or complication rate |
| Ramsay et al, 2012154 | Jan 1995—Oct 2010 (Medline, Embase, BIOSIS, Science Citation Index, CENTRAL, DARE, CDSR, conference abstracts) | 58 reports of 54 studies (1 RCT, 57 reports of 53 non-RCTs) | Laparoscopic | Robot-assisted vs. laparoscopic: lower complication rates (anastomotic leak and organ injury only), PSM rate (17.0% vs. 23.6%, OR 0.69, 95% credible interval 0.51–0.96), transfusion rate (OR 0.71, 95% credible interval 0.31–1.62), and operative time (−12.4 minutes, 95% credible interval −16.5 to −8.1 minutes); no difference in urinary continence at 12 months (OR 0.55, 95% credible interval 0.09–2.84) or biochemical recurrence rate (OR 0.89, 95% credible interval 0.24–3.34); insufficient data to assess any differences in quality of life, sexual function, length of hospital stay, or catheterization duration; no difference in surgeon learning rates |
| Tewari et al, 2012155 | Jan 2012—Dec 2010 (PubMed, Scopus) | 400 studies (187 robot-assisted, 96 open, 117 laparoscopic) | Open, laparoscopic | Robot-assisted and laparoscopic vs. open: lower blood loss, transfusion rate, and length of stay Robot-assisted vs. open and laparoscopic: lower complication rates After propensity adjustment, PSM rates for laparoscopic were higher than robot-assisted but were similar to open Complication rates low for all groups, but lowest for robot-assisted Rates for the following showed significant differences between groups, generally favouring robot-assisted: readmission; reoperation; nerve, ureteral, or rectal injury; deep vein thrombosis; pneumonia; hematoma; lymphocele; anastomotic leak; fistula; and wound infection |
| Sandoval Salinas et al, 2013156 | 1948—Oct 2012 (Medline, Embase, LILACS, CENTRAL, DARE) | 2 RCTs | Laparoscopic | Robot-assisted vs. laparoscopic: improved sexual function and urinary continence; no differences in perioperative outcomes |
| De Carlo et al, 2014157 | ?—Dec 2013 (PubMed, Embase) | 25 studies | Open, laparoscopic | Robot-assisted and laparoscopic: operative time, blood loss, transfusion rates, catheterization duration, length of hospital stay, complication rate were most optimal in the laparoscopic approaches Data insufficient to prove superiority of any surgical approach for functional and oncological outcomes |
| Agarwal et al, 2015158 | Search period unspecified (PubMed, Cochrane) | 19 non-RCTs | Open | Robot-assisted: not unequivocally shown to be superior to open for functional and oncological outcomes |
| Pan et al, 2015159 | Jan 2009—Oct 2013 (PubMed, Google Scholar, Embase, Web of Science) | 6 studies | Open | Robot-assisted vs. open: longer operative time (weighted mean difference 64.84, 95% CI 44.12—85.55, P < .001); no difference in transfusion rate, PSM rate, or 3-and 12-month urinary continence; improved potency at 3 months (OR 2.80, 95% CI 1.83—4.27, P < . 001) and 12 months (OR 1.70, 95% CI 1.30—2.23, P < .001) |
| Allan et al, 2016160 | 1950—Dec 2014 (Medline, Scopus, CDSR, Central) | 2 RCTs | Laparoscopic | Robot-assisted: significantly higher rate of return to erectile function (RR 1.51, 95% CI 1.19–1.92) and continence (RR 1.14, 95% CI 1.04–1.24); no significant differences in operative time, blood loss, transfusion rate, or biochemical recurrence (RR 1.01, 95% CI 0.91–1.12) |
| Seo et al, 2016131 | Jan 1980—Aug 2013 (Medline, Embase, Cochrane, KoreaMed, KMbase, RISS4U, KISS, KISTI, NDSL) | 61 non-RCTs (38 from previous systematic reviews) | Open | Robot-assisted: improved perioperative and functional outcomes (urinary continence and erectile function), lower complication rate; no difference for positive surgical margin or biochemical recurrence-free survival |
Abbreviations: CDSR, Cochrane Database of Systematic Reviews; CENTRAL, Cochrane Central Register of Controlled Trials; CI, confidence interval; CINAHL, Cumulative Index of Nursing and Allied Health Literature; DARE, Database of Abstracts and Reviews of Effects; HR, hazard ratio; HTA, health technology assessment; KISS, Korean Studies Information Service System; KISTI, Korean Institute of Science and Technology Information; LILACS, Latin American and Caribbean Literature on the Health Sciences; NDSL, National Digital Science Library; OR, odds ratio; PSM, positive surgical margin; RCT, randomized controlled trial; RISS4U, Research Information Service System for You; RR, relative risk.
Appendix 6: Guideline Recommendations on Robot-Assisted Radical Prostatectomy
Table A22:
Summary of Guideline Recommendations on Robot-Assisted Radical Prostatectomy
| Author, Year Title | Recommendation Excerpts | Level of Evidence |
|---|---|---|
| Montorsi et al, 201266 Best Practices in Robot-Assisted Radical Prostatectomy: Recommendations of the Pasadena Consensus Panel |
The following recommendations are made with regard to patient selection and surgical technique:
The following recommendations are made with regard to cancer control:
The following recommendations are made with regard to functional outcomes and complications of RARP:
|
Not reported |
| National Institute for Health and Care Excellence (NICE), 201465 Prostate Cancer: Diagnosis and Treatment |
Commissioners of urology services should consider providing robotic surgery to treat localised prostate cancer. Commissioners should ensure that robotic systems for the surgical treatment of localised prostate cancer are cost effective by basing them in centres that are expected to perform at least 150 robot-assisted laparoscopic radical prostatectomies per year. |
Not reported |
| Merseburger et al (European Association of Urology), 201464 Guidelines on Robotic- and Single-Site Surgery in Urologya |
Surgical and oncological outcomes:
|
|
Incontinence:
Potency:
|
|
|
| Mottet et al (European Association of Urology), 201567 Guidelines on Prostate Cancer |
In patients who are surgical candidates for radical prostatectomy, all approaches (i.e. open, laparoscopic or robotic) are acceptable as no single approach has shown clear superiority in terms of functional or oncological results. (Grade A recommendation: based on clinical studies of good quality and consistency that addressed the specific recommendations, including at least one randomised controlled trial) |
|
Abbreviations: DVT, deep vein thrombosis; LRP, laparoscopic radical prostatectomy; NICE, National Institute for Health and Care Excellence; PLND, pelvic lymph node dissection; RARP, robot-assisted radical prostatectomy; RP, radical prostatectomy; RRP, retropubic radical prostatectomy.
The expert panel would like to stress that a well-done laparoscopy or open procedure would produce similar results.
The expert panel would like to stress that a well-done laparoscopy or open procedure would produce similar results.
Now a discontinued European Association of Urology (EAU) guideline topic.
Appendix 7: Ongoing Studies Related to Robot-Assisted Radical Prostatectomy
Table A23:
Ongoing Comparative Studies on Robot-Assisted Radical Prostatectomy
| ID | Registry | Country | Design | Scientific Title | Comparator |
|---|---|---|---|---|---|
| ACTRN12611000593932 | Australian New Zealand Clinical Trials Registry | Australia | Prospective Nonrandomized | Patient outcomes after open and minimally invasive surgery for prostate cancer | Open and laparoscopic radical prostatectomy |
| ACTRN12612001219875 | Australian New Zealand Clinical Trials Registry | China | Randomized controlled trial | Effectiveness and safety of robot-assisted laparoscopic radical prostatectomy versus laparoscopic radical prostatectomy in patients with prostate cancer by one experienced laparoscopic surgeon: a prospective controlled trial from China | Laparoscopic radical prostatectomy |
| DRKS00007138 | German Clinical Trials Register | Germany | Randomized controlled trial | Randomized, multicenter study comparing robot-assisted and conventional laparoscopic radical prostatectomy | Laparoscopic radical prostatectomy |
| NCT00578123 | clinicaltrials.gov | United States | Prospective Nonrandomized | Prospective assessment of clinical and quality of life outcomes after open or robotic-assisted laparoscopic radical prostatectomy | Open radical prostatectomy |
| NCT01325506 | clinicaltrials.gov | United States | Prospective Nonrandomized | Effectiveness of open and robotic prostatectomy: the PROSTQA-RP2 study | Open radical prostatectomy |
| NCT01577836 | clinicaltrials.gov | France | Prospective Nonrandomized | Medico-economic comparison of robot-assisted radical prostatectomy using the da Vinci System versus radical prostatectomy via laparotomy | Laparoscopic radical prostatectomy |
| NCT01578356 | clinicaltrials.gov | Belgium | Prospective Nonrandomized | Oncological and functional outcomes after radical prostatectomy for prostate cancer: comparing open with robot-assisted surgery | Open radical prostatectomy |
| NCT02292914 | clinicaltrials.gov | Brazil | Randomized controlled trial | Prospective analysis of robot-assisted surgery (includes robot-assisted radical prostatectomy) | Conventional surgery (includes open radical prostatectomy) |
| NCT02784314 | clinicaltrials.gov | France | Prospective Nonrandomized | Medico-economic evaluation of robotic-assisted radical prostatectomy versus laparoscopic radical prostatectomy | Laparoscopic radical prostatectomy |
Table A24:
Ongoing Systematic Reviews Related to Robot-Assisted Radical Prostatectomy
| ID | Registry | Title | Review Question | Intervention and Comparator |
|---|---|---|---|---|
| CRD42016035336 | PROSPERO | Robotic surgery in urology: a systematic review of randomised controlled trials | To assess the effectiveness and safety of robotic surgery in the treatment of people with urooncological disease | Robot-assisted surgery compared with open or laparoscopic surgery |
| CRD42016043331 | PROSPERO | Positive surgical margins in robot-assisted radical prostatectomy versus open radical prostatectomy: a meta-analysis of comparative studies | How surgical approach (robotic-assisted radical prostatectomy or open radical prostatectomy) impacts positive surgical margin | Open radical prostatectomy |
| CD009625 | Cochrane (Protocol) | Laparoscopic versus open prostatectomy for the treatment of localised prostate cancer | To determine whether laparoscopic radical prostatectomy, specifically robot-assisted radical prostatectomy, is more effective than open radical prostatectomy in reducing prostate cancer-specific mortality and increasing biochemical recurrence-free survival | Open radical prostatectomy |
Appendix 8: Results of Applicability Checklist for Studies Included in the Economic Literature Review
Table A25:
Applicability of Included Studies to the Ontario Context
| Objective: To Assess the Cost-Effectiveness of Robot-Assisted Versus Open Radical Prostatectomy | |||||
|---|---|---|---|---|---|
| Author, Year | Is the Study Population Similar to the Question? | Are the Interventions Similar to the Question? | Is the Health Care System in Which the Study Was Conducted Sufficiently Similar to the Current Ontario Context? | Was the Perspective Clearly Stated and What Was It? | Are Estimates of Relative Treatment Effect From the Best Available Source? |
| Cooperberg et al, 2013106 | Yes | Yes | No (United States) | Yes; payer | Partially |
| Hohwu et al, 2011107 | Yes | Yes | Yes (Denmark) | Yes; societal | No |
| Medical Services Advisory Committee, 2006161 | Yes | Yes | Yes (Australia) | Yes; payer | No |
| Author, Year | Are All Future Costs and Outcomes Discounted? (If Yes, at What Rate?) | Is the Value Of Health Effects Expressed in Terms of Quality-Adjusted Life-Years? | Are Costs and Outcomes From Other Sectors Fully and Appropriately Measured and Valued? | Overall Judgment (Directly Applicable/Partially Applicable/ Not Applicable) | |
| Cooperberg et al, 2013106 | Yes; 3% | Yes | No | Not applicable | |
| Hohwu et al, 2011107 | No (time horizon: 1 year) | Yes | Yes | Not applicable | |
| Medical Services Advisory Committee, 2006161 | Yes; 5% | Yes | No | Not applicable | |
Author contributions
This report was developed by a multidisciplinary team from Health Quality Ontario. The lead clinical epidemiologist was Myra Wang, the lead health economist was Xuanqian Xie, the lead patient engagement analyst was David Wells, and the medical librarian was Caroline Higgins.
KEY MESSAGES
Prostate cancer is the second most common cancer in men in Canada. It forms in the prostate gland of the male reproductive system and often grows very slowly. However, in some patients, prostate cancer grows more quickly and is fatal.
One of the possible treatments for prostate cancer is to surgically remove the prostate gland. This is known as radical prostatectomy. It can be performed in an open, laparoscopic, or robot-assisted approach. The open approach is the traditional surgical approach and involves a large incision. In contrast, laparoscopic and robot-assisted approaches are minimally invasive and performed through small keyhole incisions. The robot-assisted approach is the newest method. It uses a surgical robotic system with arms that the surgeon controls to perform the radical prostatectomy.
We reviewed the evidence to evaluate the effectiveness, safety, and cost-effectiveness of the robotic surgical system for radical prostatectomy compared with the open and laparoscopic approaches.
We did not find high-quality evidence that the robot-assisted approach improves cancer-related outcomes or important functional outcomes (e.g., urinary function and sexual function). The robotic surgical approach does appear to improve some perioperative outcomes, such as the length of the hospital stay and blood loss.
Patients may prefer the robot-assisted method, particularly if their surgeon recommends it as a better treatment.
Our economic analysis showed that compared with open radical prostatectomy, the costs of using the robotic system are relatively large while the health benefits are relatively small. Thus, robot-assisted radical prostatectomy does not appear to be cost-effective in Ontario.
Contributor Information
Health Quality Ontario:
About Health Quality Ontario
Health Quality Ontario is the provincial advisor on the quality of health care. We are motivated by a single-minded purpose: Better health for all Ontarians.
Who We Are
We are a scientifically rigorous group with diverse areas of expertise. We strive for complete objectivity, and look at things from a vantage point that allows us to see the forest and the trees. We work in partnership with health care providers and organizations across the system, and engage with patients themselves, to help initiate substantial and sustainable change to the province's complex health system.
What We Do
We define the meaning of quality as it pertains to health care, and provide strategic advice so all the parts of the system can improve. We also analyze virtually all aspects of Ontario's health care. This includes looking at the overall health of Ontarians, how well different areas of the system are working together, and most importantly, patient experience. We then produce comprehensive, objective reports based on data, facts and the voice of patients, caregivers and those who work each day in the health system. As well, we make recommendations on how to improve care using the best evidence. Finally, we support large scale quality improvements by working with our partners to facilitate ways for health care providers to learn from each other and share innovative approaches.
Why It Matters
We recognize that, as a system, we have much to be proud of, but also that it often falls short of being the best it can be. Plus certain vulnerable segments of the population are not receiving acceptable levels of attention. Our intent at Health Quality Ontario is to continuously improve the quality of health care in this province regardless of who you are or where you live. We are driven by the desire to make the system better, and by the inarguable fact that better has no limit.
REFERENCES
- (1).Canadian Cancer Society. Prostate cancer statistics [Internet]. Toronto (ON): The Society; 2016. [cited 2016 Feb 17]. Available from: http://www.cancer.ca/en/cancer-information/cancer-type/prostate/statistics [Google Scholar]
- (2).Cancer Care Ontario. Ontario cancer statistics 2016 [Internet]. Toronto (ON): Cancer Care Ontario; 2016. [cited 2016 Feb 22]. Available from: https://www.cancercare.on.ca/common/pages/UserFile.aspx?fileId=360956 [Google Scholar]
- (3).Cancer Quality Council of Ontario. Cancer in Ontario [Internet]. Toronto (ON): The Council; 2016. [cited 2016 Feb 22]. Available from: http://www.csqi.on.ca/cancer_in_ontario/ [Google Scholar]
- (4).Canadian Cancer Society. Survival statistics for prostate cancer [Internet]. Toronto (ON): The Society; 2016. [cited 2016 Feb 22]. Available from: http://www.cancer.ca/en/cancer-information/cancer-type/prostate/prognosis-and-survival/survival-statistics [Google Scholar]
- (5).Canadian Cancer Society. Stages of prostate cancer [Internet]. Toronto (ON): The Society; 2016. [cited 2016 Feb 4]. Available from: http://www.cancer.ca/en/cancer-information/cancer-type/prostate/staging [Google Scholar]
- (6).Rodrigues G, Warde P, Pickles T, Crook J, Brundage M, Souhami L, et al. Pre-treatment risk stratification of prostate cancer patients: a critical review. Can Urol Assoc J. 2012; 6(2):121–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (7).D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998; 280(11):969–74. [DOI] [PubMed] [Google Scholar]
- (8).Xu J, Dailey RK, Eggly S, Neale AV, Schwartz KL. Men's perspectives on selecting their prostate cancer treatment. J Natl Med Assoc. 2011; 103(6):468–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (9).Rojo MA, Martinez-Salamanca JI, Maestro MA, Galarza IS, Rodriguez JC. Impact of cosmetic result on selection of surgical treatment in patients with localized prostate cancer. JSLS. 2014 Oct-Dec;18(4):e2014.00024. [DOI] [PMC free article] [PubMed]
- (10).Hofer MD, Meeks JJ, Cashy J, Kundu S, Zhao LC. Impact of increasing prevalence of minimally invasive prostatectomy on open prostatectomy observed in the national inpatient sample and national surgical quality improvement program. J Endourol. 2013; 27(1):102–7. [DOI] [PubMed] [Google Scholar]
- (11).Dixon PR, Grant RC, Urbach DR. The impact of marketing language on patient preference for robot-assisted surgery. Surg Innov. 2015; 22(1):15–9. [DOI] [PubMed] [Google Scholar]
- (12).Alkhateeb S, Lawrentschuk N. Consumerism and its impact on robotic-assisted radical prostatectomy. BJU Int. 2011; 108(11):1874–8. [DOI] [PubMed] [Google Scholar]
- (13).Steinberg PL, Ghavamian R. Searching robotic prostatectomy online: what information is available? Urology. 2011;77 (4):941–5. [DOI] [PubMed] [Google Scholar]
- (14).Mulhall JP, Rojaz-Cruz C, Muller A. An analysis of sexual health information on radical prostatectomy websites. BJU Int. 2010; 105(1):68–72. [DOI] [PubMed] [Google Scholar]
- (15).Schroeck FR, Krupski TL, Sun L, Albala DM, Price MM, Polascik TJ, et al. Satisfaction and regret after open retropubic or robot-assisted laparoscopic radical prostatectomy. Eur Urol. 2008; 54(4):785–93. [DOI] [PubMed] [Google Scholar]
- (16).Schroeck FR, Krupski TL, Stewart SB, Baez LL, Gerber L, Albala DM, et al. Pretreatment expectations of patients undergoing robotic assisted laparoscopic or open retropubic radical prostatectomy. J Urol. 2012; 187(3):894–8. [DOI] [PubMed] [Google Scholar]
- (17).Lavery HJ, Levinson AW, Hobbs AR, Sebrow D, Mohamed NE, Diefenbach MA, et al. Baseline functional status may predict decisional regret following robotic prostatectomy. J Urol. 2012; 188(6):2213–8. [DOI] [PubMed] [Google Scholar]
- (18).Kim JH, Ha YS, Jeong SJ, Kim S, Kim WJ, Jang TL, et al. Factors related to patient-perceived satisfaction after robot-assisted radical prostatectomy based on the expanded prostate cancer index composite survey. Prostate Cancer Prostatic Dis. 2013; 16(4):341–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).Davison BJ, Matthew A, Gardner AM. Prospective comparison of the impact of robotic-assisted laparoscopic radical prostatectomy versus open radical prostatectomy on health-related quality of life and decision regret. Can Urol Assoc J. 2014; 8 (1–2):E68–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (20).Barbash GI, Glied SA. New technology and health care costs–the case of robot-assisted surgery. N Engl J Med. 2010; 363(8):701–4. [DOI] [PubMed] [Google Scholar]
- (21).Kim SP, Gross CP, Smaldone MC, Han LC, Van Houten H, Lotan Y, et al. Perioperative outcomes and hospital reimbursement by type of radical prostatectomy: Results from a privately insured patient population. Prostate Cancer Prostatic Dis. 2015; 18(1):13–7. [DOI] [PubMed] [Google Scholar]
- (22).National Health Service England. Clinical commissioning policy: robotic-assisted surgical procedures for prostate cancer [Internet]. London: National Health Service England; 2015. [cited 2016 Sep 9]. Available from: https://www.england.nhs.uk/commissioning/wp-content/uploads/sites/12/2015/10/b14pa-rbtic-asstd-srgry-prostate-cancer-oct15.pdf [Google Scholar]
- (23).McGowan J, Sampson M, Salzwedel D, Cogo E, Foerster V, Lefebvre C. PRESS peer review of electronic search strategies: 2015 guideline statement. J Clin Epidemiol. 2016; 75: 40–6. [DOI] [PubMed] [Google Scholar]
- (24).The Cochrane Collaboration. Cochrane handbook for systematic reviews of interventions [Internet]. London: The Cochrane Collaboration; 2011. [cited 2016 Feb 10]. Available from: http://handbook.cochrane.org [Google Scholar]
- (25).Guyatt GH, Oxman AD, Schunemann HJ, Tugwell P, Knottnerus A. GRADE guidelines: a new series of articles in the Journal of Clinical Epidemiology. J Clin Epidemiol. 2011; 64(4):380–2. [DOI] [PubMed] [Google Scholar]
- (26).Yaxley JW, Coughlin GD, Chambers SK, Occhipinti S, Samaratunga H, Zajdlewicz L, et al. Robot-assisted laparoscopic prostatectomy versus open radical retropubic prostatectomy: early outcomes from a randomised controlled phase 3 study. Lancet. 2016; 388(10049):1057–66. [DOI] [PubMed] [Google Scholar]
- (27).Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009; 6(7):e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (28).Medical Advisory Secretariat. Robotic-assisted minimally invasive surgery for gynecologic and urologic oncology: an evidence-based analysis. Ont Health Technol Assess Ser. 2010; 10(27):1–118. [PMC free article] [PubMed] [Google Scholar]
- (29).Porpiglia F, Morra I, Lucci Chiarissi M, Manfredi M, Mele F, Grande S, et al. Randomised controlled trial comparing laparoscopic and robot-assisted radical prostatectomy. Eur Urol. 2013; 63(4):606–14. [DOI] [PubMed] [Google Scholar]
- (30).Asimakopoulos AD, Pereira Fraga CT, Annino F, Pasqualetti P, Calado AA, Mugnier C. Randomized comparison between laparoscopic and robot-assisted nerve-sparing radical prostatectomy. J Sex Med. 2011; 8(5):1503–12. [DOI] [PubMed] [Google Scholar]
- (31).Geraerts I, Van Poppel H, Devoogdt N, Van Cleynenbreugel B, Joniau S, Van Kampen M. Prospective evaluation of urinary incontinence, voiding symptoms and quality of life after open and robot-assisted radical prostatectomy. BJU Int. 2013; 112(7):936–43. [DOI] [PubMed] [Google Scholar]
- (32).Wallerstedt A, Tyritzis SI, Thorsteinsdottir T, Carlsson S, Stranne J, Gustafsson O, et al. Short-term results after robot-assisted laparoscopic radical prostatectomy compared to open radical prostatectomy. Eur Urol. 2015; 67(4):660–70. [DOI] [PubMed] [Google Scholar]
- (33).Ball AJ, Gambill B, Fabrizio MD, Davis JW, Given RW, Lynch DF, et al. Prospective longitudinal comparative study of early health-related quality-of-life outcomes in patients undergoing surgical treatment for localized prostate cancer: a short-term evaluation of five approaches from a single institution. J Endourol. 2006; 20(10):723–31. [DOI] [PubMed] [Google Scholar]
- (34).Bier S, Hennenlotter J, Rausch S, Aufderklamm S, Martzog JC, Stenzl A, et al. Return to work and normal daily life activity after open and robot-assisted radical prostatectomy – a single surgeon analysis. Urol Int. 2016; 96(3):280–6. [DOI] [PubMed] [Google Scholar]
- (35).Breyer BN, Davis CB, Cowan JE, Kane CJ, Carroll PR. Incidence of bladder neck contracture after robot-assisted laparoscopic and open radical prostatectomy. BJU Int. 2010; 106(11):1734–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (36).Carlsson S, Nilsson AE, Schumacher MC, Jonsson MN, Volz DS, Steineck G, et al. Surgery-related complications in 1253 robot-assisted and 485 open retropubic radical prostatectomies at the Karolinska University Hospital, Sweden. Urology. 2010; 75(5):1092–7. [DOI] [PubMed] [Google Scholar]
- (37).Di Pierro GB, Baumeister P, Stucki P, Beatrice J, Danuser H, Mattei A. A prospective trial comparing consecutive series of open retropubic and robot-assisted laparoscopic radical prostatectomy in a centre with a limited caseload. Eur Urol. 2011; 59(1):1–6. [DOI] [PubMed] [Google Scholar]
- (38).Doumerc N, Yuen C, Savdie R, Rahman MB, Rasiah KK, Pe Benito R, et al. Should experienced open prostatic surgeons convert to robotic surgery? The real learning curve for one surgeon over 3 years. BJU Int. 2010; 106(3):378–84. [DOI] [PubMed] [Google Scholar]
- (39).Farnham SB, Webster TM, Herrell SD, Smith JA., Jr Intraoperative blood loss and transfusion requirements for robotic-assisted radical prostatectomy versus radical retropubic prostatectomy. Urology. 2006; 67(2):360–3. [DOI] [PubMed] [Google Scholar]
- (40).Ficarra V, Novara G, Fracalanza S, D'Elia C, Secco S, Iafrate M, et al. A prospective, non-randomized trial comparing robot-assisted laparoscopic and retropubic radical prostatectomy in one European institution. BJU Int. 2009; 104(4):534–9. [DOI] [PubMed] [Google Scholar]
- (41).Fode M, Sonksen J, Jakobsen H. Radical prostatectomy: initial experience with robot-assisted laparoscopic procedures at a large university hospital. Scand J Urol. 2014; 48(3):252–8. [DOI] [PubMed] [Google Scholar]
- (42).Fracalanza S, Ficarra V, Cavalleri S, Galfano A, Novara G, Mangano A, et al. Is robotically assisted laparoscopic radical prostatectomy less invasive than retropubic radical prostatectomy? Results from a prospective, unrandomized, comparative study. BJU Int. 2008; 101(9):1145–9. [DOI] [PubMed] [Google Scholar]
- (43).Haglind E, Carlsson S, Stranne J, Wallerstedt A, Wilderang U, Thorsteinsdottir T, et al. Urinary incontinence and erectile dysfunction after robotic versus open radical prostatectomy: a prospective, controlled, nonrandomised trial. Eur Urol. 2015; 68(2):216–25. [DOI] [PubMed] [Google Scholar]
- (44).Ham WS, Park SY, Kim WT, Koo KC, Lee YS, Choi YD. Open versus robotic radical prostatectomy: a prospective analysis based on a single surgeon's experience. J Robot Surg. 2008; 2(4):235–41. [DOI] [PubMed] [Google Scholar]
- (45).Hong JY, Kim JY, Choi YD, Rha KH, Yoon SJ, Kil HK. Incidence of venous gas embolism during robotic-assisted laparoscopic radical prostatectomy is lower than that during radical retropubic prostatectomy. Br J Anaesth. 2010; 105(6):777–81. [DOI] [PubMed] [Google Scholar]
- (46).Kim SC, Song C, Kim W, Kang T, Park J, Jeong IG, et al. Factors determining functional outcomes after radical prostatectomy: robot-assisted versus retropubic. Eur Urol. 2011; 60(3):413–9. [DOI] [PubMed] [Google Scholar]
- (47).Kordan Y, Barocas DA, Altamar HO, Clark PE, Chang SS, Davis R, et al. Comparison of transfusion requirements between open and robotic-assisted laparoscopic radical prostatectomy. BJU Int. 2010; 106(7):1036–40. [DOI] [PubMed] [Google Scholar]
- (48).Loeb S, Epstein JI, Ross AE, Schultz L, Humphreys EB, Jarow JP. Benign prostate glands at the bladder neck margin in robotic vs open radical prostatectomy. BJU Int. 2010; 105(10):1446–9. [DOI] [PubMed] [Google Scholar]
- (49).Lott FM, Favorito LA. Is previous experience in laparoscopic necessary to perform robotic radical prostatectomy? A comparative study with robotic and the classic open procedure in patients with prostate cancer. Acta Cir Bras. 2015; 30(3):229–34. [DOI] [PubMed] [Google Scholar]
- (50).Ludovico GM, Dachille G, Pagliarulo G, D'Elia C, Mondaini N, Gacci M, et al. Bilateral nerve sparing robotic-assisted radical prostatectomy is associated with faster continence recovery but not with erectile function recovery compared with retropubic open prostatectomy: the need for accurate selection of patients. Oncol Rep. 2013; 29(6):2445–50. [DOI] [PubMed] [Google Scholar]
- (51).Miller J, Smith A, Kouba E, Wallen E, Pruthi RS. Prospective evaluation of short-term impact and recovery of health related quality of life in men undergoing robotic assisted laparoscopic radical prostatectomy versus open radical prostatectomy. J Urol. 2007; 178(3):854–9. [DOI] [PubMed] [Google Scholar]
- (52).Nelson B, Kaufman M, Broughton G, Cookson MS, Chang SS, Herrell SD, et al. Comparison of length of hospital stay between radical retropubic prostatectomy and robotic assisted laparoscopic prostatectomy. J Urol. 2007; 177(3):929–31. [DOI] [PubMed] [Google Scholar]
- (53).O'Malley PJ, Van Appledorn S, Bouchier-Hayes DM, Crowe H, Costello AJ. Robotic radical prostatectomy in Australia: initial experience. World J Urol. 2006; 24(2):165–70. [DOI] [PubMed] [Google Scholar]
- (54).Philippou P, Waine E, Rowe E. Robot-assisted laparoscopic prostatectomy versus open: comparison of the learning curve of a single surgeon. J Endourol. 2012; 26(8):1002–8. [DOI] [PubMed] [Google Scholar]
- (55).Thompson JE, Egger S, Bohm M, Haynes AM, Matthews J, Rasiah K, et al. Superior quality of life and improved surgical margins are achievable with robotic radical prostatectomy after a long learning curve: a prospective single-surgeon study of 1552 consecutive cases. Eur Urol. 2014; 65(3):521–31. [DOI] [PubMed] [Google Scholar]
- (56).Wood DP, Schulte R, Dunn RL, Hollenbeck BK, Saur R, Wolf JS, Jr, et al. Short-term health outcome differences between robotic and conventional radical prostatectomy. Urology. 2007; 70(5):945–9. [DOI] [PubMed] [Google Scholar]
- (57).Asimakopoulos AD, Miano R, Di Lorenzo N, Spera E, Vespasiani G, Mugnier C. Laparoscopic versus robot-assisted bilateral nerve-sparing radical prostatectomy: comparison of pentafecta rates for a single surgeon. Surg Endosc. 2013; 27(11):4297–304. [DOI] [PubMed] [Google Scholar]
- (58).Berge V, Berg RE, Hoff JR, Wessel N, Diep LM, Karlsen SJ, et al. A prospective study of transition from laparoscopic to robot-assisted radical prostatectomy: quality of life outcomes after 36-month follow-up. Urology. 2013; 81(4):781–6. [DOI] [PubMed] [Google Scholar]
- (59).Ploussard G, Xylinas E, Paul A, Gillion N, Salomon L, Allory Y, et al. Is robot assistance affecting operating room time compared with pure retroperitoneal laparoscopic radical prostatectomy? J Endourol. 2009;23 (6):939–43. [DOI] [PubMed] [Google Scholar]
- (60).Ploussard G, De La Taille A, Moulin M, Vordos D, Hoznek A, Abbou CC, et al. Comparisons of the perioperative, functional, and oncologic outcomes after robot-assisted versus pure extraperitoneal laparoscopic radical prostatectomy. Eur Urol. 2014; 65(3):610–9. [DOI] [PubMed] [Google Scholar]
- (61).Willis DL, Gonzalgo ML, Brotzman M, Feng Z, Trock B, Su LM. Comparison of outcomes between pure laparoscopic vs robot-assisted laparoscopic radical prostatectomy: a study of comparative effectiveness based upon validated quality of life outcomes. BJU Int. 2012; 109(6):898–905. [DOI] [PubMed] [Google Scholar]
- (62).Patel VR, Sivaraman A, Coelho RF, Chauhan S, Palmer KJ, Orvieto MA, et al. Pentafecta: a new concept for reporting outcomes of robot-assisted laparoscopic radical prostatectomy. Eur Urol. 2011; 59(5):702–7. [DOI] [PubMed] [Google Scholar]
- (63).Porpiglia F, Fiori C. Reply from authors re: Karim A. Touijer. the promise and challenges of randomized controlled trials for surgical interventions. Eur Urol 2013; 63:615–6: Randomised Controlled Clinical Trials in a Surgical Setting: Always the Best? Eur Urol. 2013; 63(4):616–7. [DOI] [PubMed] [Google Scholar]
- (64).Merseburger AS, Nagele U, Herrmann TRW, Traxer O, Kyriazis I, Shariat SF, et al. Guidelines on robotic- and single-site surgery in urology [Internet]. Arnhem (Netherlands): European Association of Urology; 2014. [cited 2016 Feb 22]. Available from: http://uroweb.org/wp-content/uploads/EAU-Guidelines-on-Robotic-and-Single-site-Surgery-in-Urology-V2.pdf [DOI] [PubMed] [Google Scholar]
- (65).National Institute for Health and Care Excellence. Prostate cancer: diagnosis and treatment [Internet]. London (UK): The Institute; 2014. [cited 2016 Mar 3]. Available from: https://www.nice.org.uk/guidance/cg175/evidence/full-guideline-191710765 [Google Scholar]
- (66).Montorsi F, Wilson TG, Rosen RC, Ahlering TE, Artibani W, Carroll PR, et al. Best practices in robot-assisted radical prostatectomy: recommendations of the Pasadena Consensus Panel. Eur Urol. 2012; 62(3):368–81. [DOI] [PubMed] [Google Scholar]
- (67).Mottet N, Bastian PJ, Bellmunt J, van den Bergh RCN, Bolla M, van Casteren NJ, et al. Guidelines on prostate cancer [Internet]. Arnhem (Netherlands): European Association of Urology; 2015. [cited 2016 Feb 22]. Available from: http://uroweb.org/wp-content/uploads/EAU-Guidelines-Prostate-Cancer-2015-v2.pdf [Google Scholar]
- (68).Gagnon LO, Goldenberg SL, Lynch K, Hurtado A, Gleave ME. Comparison of open and robotic-assisted prostatectomy: The University of British Columbia experience. Can Urol Assoc J. 2014; 8 (3–4):92–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (69).Chin JL, Luke PP, Pautler SE. Initial experience with robotic-assisted laparoscopic radical prostatectomy in the Canadian health care system. Can Urol Assoc J. 2007; 1(2):97–101. [PMC free article] [PubMed] [Google Scholar]
- (70).Fuller A, Pautler SE. Complications following robot-assisted radical prostatectomy in a prospective Canadian cohort of 305 consecutive cases. Can Urol Assoc J. 2013; 7 (3–4):116–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (71).Tholomier C, Bienz M, Hueber PA, Trinh QD, Hakim AE, Alhathal N, et al. Oncological and functional outcomes of 722 robot-assisted radical prostatectomy (RARP) cases: the largest Canadian 5-year experience. Can Urol Assoc J. 2014; 8 (5–6):195–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (72).Rush S, Alibhai SM, Xu L, Xu W, Louis AS, Matthew AG, et al. Health-related quality of life in robotic versus open radical prostatectomy. Can Urol Assoc J. 2015; 9 (5–6):179–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (73).Ficarra V, Wiklund PN, Rochat CH, Dasgupta P, Challacombe BJ, Sooriakumaran P, et al. The European Association of Urology Robotic Urology Section (ERUS) survey of robot-assisted radical prostatectomy (RARP). BJU Int. 2013; 111(4):596–603. [DOI] [PubMed] [Google Scholar]
- (74).Pearce SM, Pariser JJ, Karrison T, Patel SG, Eggener SE. Comparison of perioperative and early oncologic outcomes between open and robotic assisted laparoscopic prostatectomy in a contemporary population based cohort. J Urol. 2016; 196(1):76–81. [DOI] [PubMed] [Google Scholar]
- (75).Trinh QD, Sammon J, Sun M, Ravi P, Ghani KR, Bianchi M, et al. Perioperative outcomes of robot-assisted radical prostatectomy compared with open radical prostatectomy: results from the nationwide inpatient sample. Eur Urol. 2012; 61(4):679–85. [DOI] [PubMed] [Google Scholar]
- (76).Hu JC, Gandaglia G, Karakiewicz PI, Nguyen PL, Trinh QD, Shih YCT, et al. Comparative effectiveness of robot-assisted versus open radical prostatectomy cancer control. Eur Urol. 2014; 66(4):666–72. [DOI] [PubMed] [Google Scholar]
- (77).Gandaglia G, Sammon JD, Chang SL, Choueiri TK, Hu JC, Karakiewicz PI, et al. Comparative effectiveness of robot-assisted and open radical prostatectomy in the postdissemination era. J Clin Oncol. 2014; 32(14):1419–26. [DOI] [PubMed] [Google Scholar]
- (78).Isgoren AE, Saitz TR, Serefoglu EC. Erectile function outcomes after robot-assisted radical prostatectomy: is it superior to open retropubic or laparoscopic approach? Sex Med Rev. 2014;2 (1):10–23. [DOI] [PubMed] [Google Scholar]
- (79).Eastham JA, Kattan MW, Rogers E, Goad JR, Ohori M, Boone TB, et al. Risk factors for urinary incontinence after radical prostatectomy. J Urol. 1996; 156(5):1707–13. [PubMed] [Google Scholar]
- (80).Prospective randomized trial comparing robotic versus open radical prostatectomy [Internet]. Rockville Pike (MD): U.S. National Institutes of Health; 2016. [updated 30 May 2014; cited 2016 May 14]. Available from: https://clinicaltrials.gov/show/NCT01365143 [Google Scholar]
- (81).Open prostatectomy versus laparoscopic prostatectomy versus robot-assisted prostatectomy for organ-confined prostate cancer [Internet]. London (UK): BioMed Central Ltd; 2016. [updated 2014 Sep 15; cited 2016 May 15]. Available from: http://www.isrctn.com/ISRCTN59410552 [Google Scholar]
- (82).Jalil R, Mayer E, Sevdalis N, Kerr K, Green JS, Hall E, et al., editors. Why LopeRA failed to recruit? A qualitative study. Proceedings of the Hamlyn Symposium on Medical Robotics; 2014 Jul 12–15; London, UK. London (UK): Hamlyn Symposium on Medical Robotics; 2014. [Google Scholar]
- (83).Passerotti CC, Antunes AA, Okano MTR, da Cruz JAS, Nesrallah AJ, Pontes J, Jr., et al. Early results on a prospective randomized trial: robotic-assisted laparoscopic prostatectomy (RALP) versus retropubic radical prostatectomy (RRP) study. J Urol. 2011; 185(4):e444. [Google Scholar]
- (84).Hu JC, Gold KF, Pashos CL, Mehta SS, Litwin MS. Role of surgeon volume in radical prostatectomy outcomes. J Clin Oncol. 2003; 21(3):401–5. [DOI] [PubMed] [Google Scholar]
- (85).Trinh QD, Bjartell A, Freedland SJ, Hollenbeck BK, Hu JC, Shariat SF, et al. A systematic review of the volume-outcome relationship for radical prostatectomy. Eur Urol. 2013; 64(5):786–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (86).Abboudi H, Khan MS, Guru KA, Froghi S, De Win G, Van Poppel H, et al. Learning curves for urological procedures: a systematic review. BJU Int. 2014; 114(4):617–29. [DOI] [PubMed] [Google Scholar]
- (87).Sammon JD, Karakiewicz PI, Sun M, Sukumar S, Ravi P, Ghani KR, et al. Robot-assisted versus open radical prostatectomy: the differential effect of regionalization, procedure volume and operative approach. J Urol. 2013; 189(4):1289–94. [DOI] [PubMed] [Google Scholar]
- (88).Pearce SM, Pariser JJ, Patel SG, Anderson BB, Eggener SE, Zagaja GP. The impact of days off between cases on perioperative outcomes for robotic-assisted laparoscopic prostatectomy. World J Urol. 2016; 34(2):269–74. [DOI] [PubMed] [Google Scholar]
- (89).Lasser MS, Patel CK, Elsamra SE, Renzulli IJF, Haleblian GE, Pareek G. Dedicated robotics team reduces pre-surgical preparation time. Indian J Urol. 2012; 28(3):263–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (90).Bianco FJ, Jr, Scardino PT, Eastham JA. Radical prostatectomy: long-term cancer control and recovery of sexual and urinary function (“trifecta”). Urology. 2005; 66 (5 Suppl):83–94. [DOI] [PubMed] [Google Scholar]
- (91).Borregales LD, Berg WT, Tal O, Wambi C, Kaufman S, Gaya JM, et al. ‘Trifecta’ after radical prostatectomy: is there a standard definition? BJU Int. 2013;112 (1):60–7. [DOI] [PubMed] [Google Scholar]
- (92).Litwin MS, Lubeck DP, Henning JM, Carroll PR. Differences in urologist and patient assessments of health related quality of life in men with prostate cancer: results of the CaPSURE database. J Urol. 1998; 159(6):1988–92. [DOI] [PubMed] [Google Scholar]
- (93).Lee SR, Kim HW, Lee JW, Jeong WJ, Rha KH, Kim JH. Discrepancies in perception of urinary incontinence between patient and physician after robotic radical prostatectomy. Yonsei Med J. 2010; 51(6):883–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (94).Yossepowitch O, Briganti A, Eastham JA, Epstein J, Graefen M, Montironi R, et al. Positive surgical margins after radical prostatectomy: a systematic review and contemporary update. Eur Urol. 2014; 65(2):303–13. [DOI] [PubMed] [Google Scholar]
- (95).Chuang AY, Epstein JI. Positive surgical margins in areas of capsular incision in otherwise organ-confined disease at radical prostatectomy: histologic features and pitfalls. Am J Surg Pathol. 2008; 32(8):1201–6. [DOI] [PubMed] [Google Scholar]
- (96).Stephenson AJ, Kattan MS, Eastham JA, Dotan ZA, Bianco FJ, Jr, Lilja H, Scardino PT. Defining biochemical recurrence of prostate cancer after radical prostatectomy: a proposal for a standardized definition. J Clin Oncol. 2006; 24(24):3973–8. [DOI] [PubMed] [Google Scholar]
- (97).National Institute for Health and Care Excellence. Process and methods guides. The guidelines manual: appendix G: methodology checklist [Internet]. London (UK): The Institute; 2012. [cited 2015 May 27]. Available from: http://publications.nice.org.uk/the-guidelines-manual-appendices-bi-pmg6b/appendix-g-methodology-checklist-economic-evaluations [Google Scholar]
- (98).Ramsay C, Pickard R, Robertson C, Close A, Vale L, Armstrong N, et al. Systematic review and economic modelling of the relative clinical benefit and cost-effectiveness of laparoscopic surgery and robotic surgery for removal of the prostate in men with localised prostate cancer. Health Technology Assessment (Winchester, England). 2012; 16(41):1–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (99).Close A, Robertson C, Rushton S, Shirley M, Vale L, Ramsay C, et al. Comparative cost-effectiveness of robot-assisted and standard laparoscopic prostatectomy as alternatives to open radical prostatectomy for treatment of men with localised prostate cancer: a health technology assessment from the perspective of the UK national health service. Eur Urol. 2013; 64(3):361–9. [DOI] [PubMed] [Google Scholar]
- (100).Ratchanon S, Apiwattanasawee P, Prasopsanti K. A cost-utility analysis of laparoscopic radical prostatectomy and robotic-assisted laparoscopic radical prostatectomy in men with localized prostate cancer in Thailand. J Med Assoc Thai. 2015; 98: S14–S20. [PubMed] [Google Scholar]
- (101).Teljeur C, O'Neill M, Moran P, Murphy L, Harrington P, Ryan M, et al. Using prediction intervals from random-effects meta-analyses in an economic model. Int J Technol Assess Health Care. 2014; 30(1):44–9. [DOI] [PubMed] [Google Scholar]
- (102).Bijlani A, Hebert AE, Davitian M, May H, Speers M, Leung R, et al. A multidimensional analysis of prostate surgery costs in the United States: robotic-assisted versus retropubic radical prostatectomy. Value Health. 2016; 19(4):391–403. [DOI] [PubMed] [Google Scholar]
- (103).O'Malley SP, Jordan E. Review of a decision by the Medical Services Advisory Committee based on health technology assessment of an emerging technology: the case for remotely assisted radical prostatectomy. Int J Technol Assess Health Care. 2007; 23(2):286–91. [DOI] [PubMed] [Google Scholar]
- (104).Turchetti G, Palla I, Pierotti F, Cuschieri A. Economic evaluation of da Vinci–assisted robotic surgery: a systematic review. Surg Endosc. 2012; 26(3):598–606. [DOI] [PubMed] [Google Scholar]
- (105).Tandogdu Z, Vale L, Fraser C, Ramsay C. A systematic review of economic evaluations of the use of robotic assisted laparoscopy in surgery compared with open or laparoscopic surgery. Appl Health Econ Health Policy. 2015; 13(5):457–67. [DOI] [PubMed] [Google Scholar]
- (106).Cooperberg MR, Ramakrishna NR, Duff SB, Hughes KE, Sadownik S, Smith JA, et al. Primary treatments for clinically localised prostate cancer: a comprehensive lifetime cost-utility analysis. BJU Int. 2013; 111(3):437–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (107).Hohwu L, Borre M, Ehlers L, Venborg Pedersen K. A short-term cost-effectiveness study comparing robot-assisted laparoscopic and open retropubic radical prostatectomy. J Med Econ. 2011; 14(4):403–9. [DOI] [PubMed] [Google Scholar]
- (108).Medical Services Advisory Committee. Laparascopic remotely assisted radical prostatectomy (assessment report) [Internet]. Canberra (Australia): Australian Government; 2006. May [cited 2014 Jun 6]. Available from: http://www.msac.gov.au/internet/msac/publishing.nsf/Content/5A91C9DB46D41F9ACA2575AD0082FCE7/$File/1091-Assessment-Report.pdf [Google Scholar]
- (109).Tewari A, Srivasatava A, Menon M, Members of the VIP Team. A prospective comparison of radical retropubic and robot-assisted prostatectomy: experience in one institution. BJU Int. 2003; 92(3):205–10. [DOI] [PubMed] [Google Scholar]
- (110).Canadian Institute for Health Information. The delivery of radical prostatectomy to treat men with prostate cancer. Ottawa (ON): The Institute; 2014. [Google Scholar]
- (111).Ho C, Tsakonas E, Tran K, Cimon K, Severn M, Mierzwinski-Urban M, et al. Robot-assisted surgery compared with open surgery and laparoscopic surgery: clinical effectiveness and economic analyses [Internet]. Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2011. [cited 2016 Jun 6]. Available from: https://www.cadth.ca/sites/default/files/pdf/H0496_Surgical_robotics_e.pdf [PubMed] [Google Scholar]
- (112).Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated Health Economic Evaluation Reporting Standards (CHEERS) statement. Value Health. 2013; 16(2):e1–5. [DOI] [PubMed] [Google Scholar]
- (113).Canadian Agency for Drugs and Technologies in Health. Guidelines for the economic evaluation of health technologies. 3rd ed. Ottawa (ON): The Agency; 2006. [Google Scholar]
- (114).Lotan Y. Is robotic surgery cost-effective: no. Curr Opin Urol. 2012; 22(1):66–9. [DOI] [PubMed] [Google Scholar]
- (115).Krahn MD, Bremner KE, Alibhai SM, Ni A, Tomlinson G, Laporte A, et al. A reference set of health utilities for long-term survivors of prostate cancer: population-based data from Ontario, Canada. Qual Life Res. 2013; 22(10):2951–62. [DOI] [PubMed] [Google Scholar]
- (116).Krahn MD, Bremner KE, Tomlinson G, Naglie G. Utility and health-related quality of life in prostate cancer patients 12 months after radical prostatectomy or radiation therapy. Prostate Cancer Prostatic Dis. 2009; 12(4):361–8. [DOI] [PubMed] [Google Scholar]
- (117).Krahn M, Bremner KE, Tomlinson G, Ritvo P, Irvine J, Naglie G. Responsiveness of disease-specific and generic utility instruments in prostate cancer patients. Qual Life Res. 2007; 16(3):509–22. [DOI] [PubMed] [Google Scholar]
- (118).Krahn M, Ritvo P, Irvine J, Tomlinson G, Bezjak A, Trachtenberg J, et al. Construction of the Patient-Oriented Prostate Utility Scale (PORPUS): a multiattribute health state classification system for prostate cancer. J Clin Epidemiol. 2000; 53(9):920–30. [DOI] [PubMed] [Google Scholar]
- (119).Xie F, Pullenayegum E, Gaebel K, Bansback N, Bryan S, Ohinmaa A, et al. A time tradeoff-derived value set of the EQ-5D-5L for Canada. Med Care. 2016; 54(1):98–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (120).Naik H, Howell D, Su J, Qiu X, Brown C, Vennettilli A, et al. Stage specific health utility index scores of Canadian cancer patients [abstract 6614]. J Clin Oncol. 2015; 33 Suppl. [Google Scholar]
- (121).Namiki S, Takegami M, Kakehi Y, Suzukamo Y, Fukuhara S, Arai Y. Analysis linking UCLA PCI with Expanded Prostate Cancer Index Composite: an evaluation of health related quality of life in Japanese men with localized prostate cancer. J Urol. 2007; 178(2):473–7; discussion 477. [DOI] [PubMed] [Google Scholar]
- (122).Ham RT, Broering JM, Cooperberg MR, Kornak J, Carroll P, Wilson LS. Relationship between commonly used HRQOL measures: correlations and mapping between the UCLA-PCI and EPIC scales [abstract e16037]. J Clin Oncol. 2014; 32 Suppl. [Google Scholar]
- (123).Nahon I, Waddington G, Dorey G, Adams R. The history of urologic surgery: from reeds to robotics. Urol Nurs. 2011; 31(3):173–80. [PubMed] [Google Scholar]
- (124).Bernie AM, Caire AA, Conley SP, Oommen M, Boylu U, Thomas R, et al. Posterior reconstruction before anastomosis improves the anastomosis time during robot-assisted radical prostatectomy. JSLS. 2011; 14(4):520–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (125).Al-Shaiji TF, Kanaroglou N, Thom A, Prowse C, Comondore V, Orovan W, et al. A cost-analysis comparison of laparoscopic radical prostatectomy versus open radical prostatectomy: the McMaster Institute of Urology experience. Can Urol Assoc J. 2010; 4(4):237–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (126).Xie X, McGregor M. N. D., Radiofrequency ablation for treatment of Barrett's esophagus: a systematic review and cost analysis (report no. 46) [Internet]. Montreal (QC): Technology Assessment Unit, McGill University Health Centre; 2009. [cited 2016 Jun 6]. Available from: https://secureweb.mcgill.ca/tau/sites/mcgill.ca.tau/files/BARRETTs_ESOPHAGUS_REPORT.pdf [Google Scholar]
- (127).Ministry of Health and Long-Term Care. Schedule of benefits for physician services under the Health Insurance Act [Internet]. Toronto (ON): Queen's Printer for Ontario; 2015. [cited 2016 Feb 17]. Available from: http://www.health.gov.on.ca/english/providers/program/ohip/sob/physserv/physserv_mn.html [Google Scholar]
- (128).Krahn MD, Bremner KE, Zagorski B, Alibhai SM, Chen W, Tomlinson G, et al. Health care costs for state transition models in prostate cancer. Med Decis Making. 2014; 34(3):366–78. [DOI] [PubMed] [Google Scholar]
- (129).Sanyal C, Aprikian AG, Cury FL, Chevalier S, Dragomir A. Management of localized and advanced prostate cancer in Canada: a lifetime cost and quality-adjusted life-year analysis. Cancer. 2016; 122(7):1085–96. [DOI] [PubMed] [Google Scholar]
- (130).Ko YH, Coelho RF, Chauhan S, Sivaraman A, Schatloff O, Cheon J, et al. Factors affecting return of continence 3 months after robot-assisted radical prostatectomy: analysis from a large, prospective data by a single surgeon. J Urol. 2012; 187(1):190–4. [DOI] [PubMed] [Google Scholar]
- (131).Seo HJ, Lee NR, Son SK, Kim DK, Rha KH, Lee SH. Comparison of robot-assisted radical prostatectomy and open radical prostatectomy outcomes: a systematic review and meta-analysis. Yonsei Med J. 2016; 57(5):1165–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (132).Sanyal C, Aprikian A, Cury F, Chevalier S, Dragomir A. Clinical management and burden of prostate cancer: a Markov Monte Carlo model. PLoS One. 2014; 9(12):e113432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (133).Nam RK, Herschorn S, Loblaw DA, Liu Y, Klotz LH, Carr LK, et al. Population based study of long-term rates of surgery for urinary incontinence after radical prostatectomy for prostate cancer. J Urol. 2012; 188(2):502–6. [DOI] [PubMed] [Google Scholar]
- (134).The Human Mortality Database Research Teams. Life tables in 2011 in Canada [Internet]. Berkeley (CA): The Human Mortality Database Research Teams; 2013. [cited 2016 May 5]. Available from: http://www.mortality.org [Google Scholar]
- (135).Fairey A, Jacobsen N, Yasui Y, Liu Q, Voaklander D, Estey E. Between-surgeon variation in outcomes of radical prostatectomy for clinically localized prostate cancer: analysis of 1014 consecutive men treated at the University of Alberta [abstract]. Can Urol Assoc J. 2012; 6 (3 Supple 1):S13. [Google Scholar]
- (136).Fairey AS, Jacobsen N, Voaklander D, Estey E. Comprehensive prospective comparison of open and robot-assisted radical prostatectomy for clinically localized prostate cancer: analysis of 1019 consecutive men at a Canadian academic center [abstract]. J Urol. 2012; 187 (4S):e542. [Google Scholar]
- (137).Fairey AS, Jacobsen N, Voaklander D, Estey E. Effect of open versus robot-assisted laparoscopic radical prostatectomy on cancer control in patients with clinically localized prostate cancer: prospective analysis of 1,014 consecutive patients [abstract 80]. J Clin Oncol. 2012; 30 Suppl 5. [Google Scholar]
- (138).Biehn Stewart S, Reed SD, Moul JW. Will the future of health care lead to the end of the robotic golden years? Eur Urol 2014;65 (2):325–7; discussion 327–8. [DOI] [PubMed] [Google Scholar]
- (139).Pierorazio PM, Mullins JK, Ross AE, Hyams ES, Partin AW, Han M, et al. Trends in immediate perioperative morbidity and delay in discharge after open and minimally invasive radical prostatectomy (RP): a 20-year institutional experience BJU Int. 2013; 112(1):45–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (140).Herrell SD, Galloway RL, Su LM. Image-guided robotic surgery: update on research and potential applications in urologic surgery. Curr Opin Urol. 2012; 22(1):47–54. [DOI] [PubMed] [Google Scholar]
- (141).OHTAC Public Engagement Subcommittee. Public engagement for health technology assessment at Health Quality Ontario—final report from the Ontario Health Technology Advisory Committee Public Engagement Subcommittee [Internet]. Toronto (ON): Queen's Printer for Ontario; 2015. [cited 2015 Aug 5]. Available from: http://www.hqontario.ca/Portals/0/documents/evidence/special-reports/report-subcommittee-20150407-en.pdf [Google Scholar]
- (142).Barham L. Public and patient involvement at the UK National Institute for Health and Clinical Excellence. Patient. 2011; 4(1):1–10. [DOI] [PubMed] [Google Scholar]
- (143).Messina J, Grainger DL. A pilot study to identify areas for further improvements in patient and public involvement in health technology assessments for medicines. Patient. 2012; 5(3):199–211. [DOI] [PubMed] [Google Scholar]
- (144).Ploussard G, Briganti A, De La Taille A, Haese A, Heidenreich A, Menon M, et al. Pelvic lymph node dissection during robot-assisted radical prostatectomy: Efficacy, limitations, and complications – A systematic review of the literature. Eur Urol. 2014; 65(1):7–16. [DOI] [PubMed] [Google Scholar]
- (145).Duffey B, Varda B, Konety B. Quality of evidence to compare outcomes of open and robot-assisted laparoscopic prostatectomy. Curr Urol Rep. 2011; 12(3):229–36. [DOI] [PubMed] [Google Scholar]
- (146).Ferronha F, Barros F, Santos VV, Ravery V, Delmas V. Is there any evidence of superiority between retropubic, laparoscopic or robot-assisted radical prostatectomy? International Braz J Urol. 2011;37 (2):146–58. [DOI] [PubMed] [Google Scholar]
- (147).Health Information and Quality Authority. Health technology assessment of robot-assisted surgery in selected surgical procedures [Internet]. Dublin (Ireland): Health Information and Quality Authority; 2011. [cited 2016 May 15]. Available from: https://www.hiqa.ie/publications/health-technology-assessment-robot-assisted-surgery-selected-surgical-procedures [Google Scholar]
- (148).Ficarra V, Novara G, Ahlering TE, Costello A, Eastham JA, Graefen M, et al. Systematic review and meta-analysis of studies reporting potency rates after robot-assisted radical prostatectomy. Eur Urol. 2012; 62(3):418–30. [DOI] [PubMed] [Google Scholar]
- (149).Ficarra V, Novara G, Rosen RC, Artibani W, Carroll PR, Costello A, et al. Systematic review and meta-analysis of studies reporting urinary continence recovery after robot-assisted radical prostatectomy. Eur Urol. 2012; 62(3):405–17. [DOI] [PubMed] [Google Scholar]
- (150).Gleitsmann K, Bunker K, Kriz H, Ryan K, Vandegriff S, Liu R, et al. Robotic assisted surgery [Internet]. Portland (OR): Oregon Health and Science University; 2012. [cited 2016 Jun 23]. Available from: http://www.hca.wa.gov/assets/program/ras_corrected_final_report_050312[1].pdf [Google Scholar]
- (151).Moran PS, O'Neill M, Teljeur C, Flattery M, Murphy LA, Smyth G, et al. Robot-assisted radical prostatectomy compared with open and laparoscopic approaches: a systematic review and meta-analysis. Int J Urol. 2013; 20(3):312–21. [DOI] [PubMed] [Google Scholar]
- (152).Novara G, Ficarra V, Mocellin S, Ahlering TE, Carroll PR, Graefen M, et al. Systematic review and meta-analysis of studies reporting oncologic outcome after robot-assisted radical prostatectomy. Eur Urol. 2012; 62(3):382–404. [DOI] [PubMed] [Google Scholar]
- (153).Novara G, Ficarra V, Rosen RC, Artibani W, Costello A, Eastham JA, et al. Systematic review and meta-analysis of perioperative outcomes and complications after robot-assisted radical prostatectomy. Eur Urol. 2012; 62(3):431–52. [DOI] [PubMed] [Google Scholar]
- (154).Ramsay C, Pickard R, Robertson C, Close A, Vale L, Armstrong N, et al. Systematic review and economic modelling of the relative clinical benefit and cost-effectiveness of laparoscopic surgery and robotic surgery for removal of the prostate in men with localised prostate cancer. Health Technol Assess. 2012; 16(41):1–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (155).Tewari A, Sooriakumaran P, Bloch DA, Seshadri-Kreaden U, Hebert AE, Wiklund P. Positive surgical margin and perioperative complication rates of primary surgical treatments for prostate cancer: a systematic review and meta-analysis comparing retropubic, laparoscopic, and robotic prostatectomy. Eur Urol. 2012; 62(1):1–15. [DOI] [PubMed] [Google Scholar]
- (156).Sandoval Salinas C, Gonzalez Rangel AL, Catano Catano JG, Fuentes Pachon JC, Castillo Londono JS. Efficacy of robotic-assisted prostatectomy in localized prostate cancer: a systematic review of clinical trials. Adv Urol. 2013; 2013: 105651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (157).De Carlo F, Celestino F, Verri C, Masedu F, Liberati E, Di Stasi SM. Retropubic, laparoscopic, and robot-assisted radical prostatectomy: surgical, oncological, and functional outcomes: a systematic review. Urol Int. 2014; 93(4):373–83. [DOI] [PubMed] [Google Scholar]
- (158).Agarwal G, Valderrama O, Luchey AM, Pow-Sang JM. Robotic-assisted laparoscopic radical prostatectomy. Cancer Control. 2015; 22(3):283–90. [DOI] [PubMed] [Google Scholar]
- (159).Pan XW, Cui XM, Teng JF, Zhang DX, Wang ZJ, Qu FJ, et al. Robot-assisted radical prostatectomy vs. open retropubic radical prostatectomy for prostate cancer: a systematic review and meta-analysis. Indian J Surg. 2015; 77 (Suppl 3):1326–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (160).Allan C, Ilic D. Laparoscopic versus robotic-assisted radical prostatectomy for the treatment of localised prostate cancer: a systematic review. Urol Int. 2016; 96(4):373–8. [DOI] [PubMed] [Google Scholar]
- (161).Martinez PF, Billordo Peres N, Cristallo C, Isola M, Villamil W, Giudice CR, et al. Salvage radical prostatectomy after radiotherapy. Arch Esp Urol. 2014; 67(4):313–22. [PubMed] [Google Scholar]