Abstract
Purpose
People with chronic kidney disease (CKD) have an increased prevalence of depression, anxiety, and neuropathic pain. We examined prevalence, incidence, indication for, and choice of antidepressants among patients with and without CKD.
Methods
Using the UK Clinical Practice Research Datalink, we identified patients with CKD (two measurements of estimated glomerular filtration rate < 60 mL/min/1.73m2 for ≥3 months) between April 2004 and March 2014. We compared those with CKD to a general population cohort without CKD (matched on age, sex, general practice, and calendar time [index date]). We identified any antidepressant prescribing in the six months prior to index date (prevalence), the first prescription after index date among non‐prevalent users (incidence), and recorded diagnoses (indication). We compared antidepressant choice between patients with and without CKD among patients with a diagnosis of depression.
Results
There were 242 349 matched patients (median age 76 [interquartile range 70–82], male 39.3%) with and without CKD. Prevalence of antidepressant prescribing was 16.3 and 11.9%, and incidence was 57.2 and 42.4/1000 person‐years, in patients with and without CKD, respectively. After adjusting for confounders, CKD remained associated with higher prevalence and incidence of antidepressant prescription. Regardless of CKD status, selective serotonin reuptake inhibitors were predominantly prescribed for depression or anxiety, while tricyclic antidepressants were prescribed for neuropathic pain or other reasons. Antidepressant choice was similar in depressed patients with and without CKD.
Conclusions
The rate of antidepressant prescribing was nearly one and a half times higher among people with CKD than in the general population. © 2017 The Authors. Pharmacoepidemiology & Drug Safety Published by John Wiley & Sons Ltd
Keywords: antidepressants, chronic kidney disease, prevalence, incidence, depression
Introduction
Antidepressants are among the most commonly prescribed classes of medication in industrialized countries, including the USA1 and UK.2 The recent increase in the prescription of antidepressants is dramatic, with an average 10% increase per year from 1998 to 2010.3 Antidepressants can be prescribed not only for depressive symptoms but also for other conditions such as anxiety and neuropathic pain.4 In addition, off‐label use of antidepressants is common for chronic pain, including non‐neuropathic pain, and conditions where non‐specific sedation is required.5, 6, 7
Chronic kidney disease (CKD), an impairment of kidney structure or function, is now recognized as a major public health problem.8 Chronic kidney disease is associated with a range of comorbidities including obesity, hypertension, diabetes, and cardiovascular disease.9, 10 Level of kidney function, expressed as estimated glomerular filtration rate (eGFR), is closely associated with increased risk of death, cardiovascular events, and hospitalization.11
Chronic kidney disease is also associated with a range of mental health problems including anxiety12 and depression;13 almost one quarter of adults with pre‐dialysis CKD are depressed. These conditions may be due to co‐existing chronic diseases such as diabetes and heart failure, which are also associated with depression and anxiety symptoms,14, 15 or directly related to CKD. In addition, other indications for antidepressants such as chronic pain and insomnia are more common among patients with CKD.16, 17
Patients with CKD are frequently excluded from clinical trials,18, 19 and concerns have been recently raised about the lack of knowledge regarding how kidney function is related to adverse effects of antidepressants.20, 21 Despite this, there has been no systematic research investigating frequency and patterns of antidepressant prescribing among patients with CKD. Understanding how antidepressants are actually prescribed in patients with CKD, compared to those without CKD, is important groundwork for the planning of future studies on the safety of antidepressants in this population. Therefore, we aimed to compare the frequency (prevalence and incidence), indications for, and choice of antidepressant prescription between patients with and without CKD, in the UK general population.
Methods
Data sources
The Clinical Practice Research Datalink (CPRD) is a database of routinely recorded primary care electronic health record data from 7% of the UK population.22 The database includes the following data: patient demographics; diagnoses; prescriptions; laboratory test results; and referrals made by general practitioners (GPs). Diseases can be identified using diagnostic codes (Read codes) recorded in routine data. We used CPRD linked to additional data sources: the inpatient Hospital Episodes Statistics (HES) database to provide data on ethnicity (to improve data completeness);23 Office for National Statistics (ONS) data for mortality; and Index of Multiple Deprivation (IMD) data for deprivation indices. We obtained study approval from the institutional review board of the London School of Hygiene and Tropical Medicine (reference: 9196), as well as the Independent Scientific Advisory Committee, which oversees research involving CPRD data (Protocol 15_219R). Informed consent from individual patients was waived because the data are anonymous.
Study population and matched cohort
All adults (age 18 or older) alive and contributing to HES‐linked CPRD anytime from 1 April 2004 to 31 March 2014 were potentially eligible for inclusion. Patients were eligible for inclusion at the latest of: one year after practice registration,24 the date that the practice reached CPRD quality control standards, or 1 April 2004. Patients were no longer eligible for follow‐up at the first of renal replacement therapy (RRT) initiation, death, change of practice, last data collection from the practice, or 31 March 2014. We excluded patients already receiving RRT (hemodialysis, peritoneal dialysis, and kidney transplantation) prior to cohort entry.
First, we identified patients with CKD based on two consecutive measurements of eGFR <60 mL/min/1.73 m2 more than three months apart.25 Estimated GFR was calculated from serum creatinine values recorded in CPRD, using the Chronic Kidney Disease Epidemiology Collaboration equation.26 Patients, including those who had CKD before April 2004, were included in the cohort on the date when they first satisfied the CKD definition (i.e. second eGFR <60 mL/min/1.73 m2) during eligible follow‐up (index date).
Next, as a control group, we selected at random patients without CKD from the general population. Because (i) CKD status largely depends on age and sex, and (ii) pattern of antidepressant prescription is expected to depend on general practice and calendar time, we matched controls to patients with CKD by age (same year of birth), sex, general practice, and calendar time. Each control entered the cohort on the same index date as their CKD counterpart. Individuals selected as controls (i.e. non‐CKD patients) may be found to have CKD later; in this situation, they were censored as a control at the time of satisfying CKD definition and contributed separately as an incident patient with CKD from that time point forward (with their own matched control).
Prevalence and incidence of antidepressant prescription
We estimated the prevalence of existing users of antidepressants, defined as receiving an antidepressant prescription within six months prior to the index date. Incidence of antidepressant prescription was based on the first antidepressant prescription after index date, after exclusion of existing users.27
Covariates
In order to examine the independent association between CKD status and antidepressant prescription, we considered baseline characteristics of patients: age and sex; ethnicity; socio‐economic status (SES); smoking status; body mass index (BMI); and common chronic physical illnesses that are considered to be associated with mental health conditions (diabetes mellitus, congestive heart failure, myocardial infarction, stroke, chronic obstructive pulmonary disease, rheumatoid arthritis, cancer, Parkinson's disease, and epilepsy).28, 29 Based on previous studies using UK primary care data,30, 31 we classified patients with no record of ethnicity as white. Socio‐economic status was assigned by quintile at an individual level, using 2010 ONS estimates of the IMD (a composite area‐level marker of deprivation).32 For patients with missing individual‐level SES, we used the SES for the patient's general practice. Smoking status and BMI were assigned using the data recorded closest to the index date. We defined each chronic physical illness as present if a relevant diagnosis code of that illness was recorded at least once before a patient's index date.
Indication
We identified morbidity codes for three common diagnoses suggesting indications for antidepressant prescription:4 depression, anxiety, and neuropathic pain (included in Appendix 1). We included symptom codes as well as diagnostic codes because GPs in the UK commonly use symptom codes (e.g. “depressive symptoms”, “anxiousness”) rather than definitive diagnostic codes (e.g. “major depression”, “general anxiety disorder”).33, 34, 35 We included codes recorded by GPs any time prior to the first antidepressant prescription until three months later, to account for possible time lag in recording diagnosis codes in electronic health records.36, 37 We categorized type of antidepressant, according to British National Formulary headings, into the following categories:4 selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants (TCAs), or other antidepressants. Monoamine oxidase inhibitors were grouped into other antidepressants because of a small number of prescriptions. For each type of antidepressant, we identified the proportion of patients with each indication as well as those with none of the three indications.
Choice of antidepressants and initial prescription dose
There are 26 antidepressants currently available in the UK, only a few are indicated for anxiety and neuropathic pain, whilst all 26 are indicated for depressive conditions.4 Therefore, we restricted this analysis to patients with a recorded diagnosis of depression. We compared the pattern of antidepressant choice (the proportion of patients prescribed each antidepressant as their first incident prescription) between depressed patients with and without CKD. We also compared the initial dose prescribed in those with and without CKD to examine whether antidepressants were started at a lower dose in patients with CKD than those without.
Statistical analyses
We compared the baseline characteristics of patients with and without CKD using χ2 tests. We calculated crude prevalence and incidence rates for antidepressant prescribing. We then conducted a conditional logistic regression analysis (to account for matching) to investigate the association between CKD status and prevalence of antidepressant prescription. After excluding existing users of antidepressants (meaning matching was no longer maintained), we conducted an unconditional Poisson regression analysis to investigate the association between CKD status and incidence of new antidepressant prescription, adjusting for age, sex, and financial year, and taking account of clustering by general practice using robust standard errors. We adjusted for financial year (by including financial year as a categorical variable, i.e. from 1 April to 31 March for each year) because the frequency of antidepressant prescribing has been increasing year by year.3 We further adjusted for ethnicity, SES, smoking status, and BMI, and, then, in a fully adjusted model, also included chronic physical illnesses. In models including smoking status and BMI, we included an additional absent category for those with no recorded smoking status or BMI. In a subsequent sensitivity analysis, we dropped all those with missing smoking or BMI status. All the data management and statistical analyses were conducted using STATA version 14 (Stata Corp, Texas).
Renal function subgroup analyses
To examine the association between severity of kidney function and antidepressant prescribing, we classified patients with CKD according to the level of kidney function on the index date into two categories: eGFR 30–59 (CKD stage 3), and <30 mL/min/1.73 m2 (CKD stage 4 and 5).25 In patients without CKD, we differentiated patients with and without serum creatinine results recorded in CPRD prior to index date, because these subgroups are expected to be systematically different due to testing incentives for those at risk of CKD in the UK Quality and Outcomes Framework.38 To compare the prevalence of existing users of antidepressants between subgroups of renal function, we used an unconditional logistic regression analysis, adjusting for age, sex, and financial year, and taking account of clustering by general practice using robust standard errors. We also repeated all other principal analyses (described under ‘Statistical analyses’ subheading) using renal function subgroups.
Additional analyses
Any difference in the duration of follow‐up lengths between patients with and without CKD may affect the likelihood of starting antidepressants. Therefore, as a post hoc analysis, we compared the proportion of patients starting antidepressants within the first six months of follow‐up in those with and without CKD.
We undertook a further analysis to investigate whether patients with CKD were more likely to start antidepressants for the first episode of depression in their life, or for a recurrent episode of depression. In CPRD, GPs routinely record a patient's past medical history shortly after registration with a new practice, and, therefore, a previous episode of depression would be recorded between CPRD registration and index date of the study (as index dates need to be at least one year after CPRD registration by our definition). Therefore, in patients starting antidepressants with a recorded diagnosis of depression, we compared the proportion of those with and without CKD who had: (i) their first depression diagnosis in CPRD recorded between CPRD registration and index date (more likely to suggest a recurrence); and (ii) their first depression diagnosis recorded in CPRD after index date (more likely to suggest the first ever depression diagnosis).
Results
Among 4 070 806 eligible patients (median age 39 [IQR 27–56], male 48.8%), we identified 264 628 patients with CKD (median age 77 [IQR 71–83], male 38.7%) (Figure 1). Of those with CKD, 242 349 (91.6%) (median age 76 [IQR 70–82], male 39.3%) were matched with a control patient without CKD who had the same age, sex, and general practice on the index date of their CKD counterpart. Unmatched patients with CKD (n = 22 279) were older and more likely to be female (median age 88 [IQR 84–92], male 31.5%). Of the 242 349 matched control patients without CKD, 41 151 (17.0%) were subsequently found to have CKD.
Figure 1.
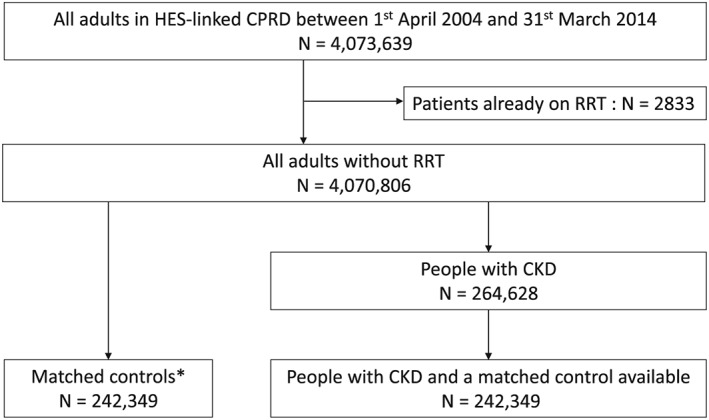
Flow chart for the selection of matched patients with and without chronic kidney disease. CKD = chronic kidney disease, CPRD = clinical practice research datalink, HES = hospital episode statistics, RRT = renal replacement therapy. *Matched cohort: randomly selected individuals without chronic kidney disease matched for age, sex, general practice, and calendar time
Compared to patients without CKD, patients with CKD were more likely to be deprived, ex‐smokers, and overweight (BMI ≥25 kg/m2) (Table 1). Chronic physical illnesses, except for Parkinson's disease and epilepsy, were more common among patients with CKD.
Table 1.
Baseline characteristics of matched patients with and without chronic kidney disease
| Patients without CKD N = 242 349 | Patients with CKD N = 242 349 | p Value | |
|---|---|---|---|
| n (%) | n (%) | ||
| Age (years): | 1.000 | ||
| <55 | 6845 (2.8) | 6845 (2.8) | |
| 55–64 | 23 556 (9.7) | 23 556 (9.7) | |
| 65–74 | 71 112 (29.3) | 71 112 (29.3) | |
| 75–84 | 102 594 (42.3) | 102 594 (42.3) | |
| ≥85 | 38 242 (15.8) | 38 242 (15.8) | |
| Sex (male): | 95 318 (39.3) | 95 318 (39.3) | 1.000 |
| Ethnicity: | |||
| White/not‐recorded* | 238 533 (98.4) | 238 138 (98.3) | <0.001 |
| South Asian | 1796 (0.7) | 2317 (1.0) | |
| Black | 1156 (0.5) | 1060 (0.4) | |
| Other ethnicity | 864 (0.4) | 834 (0.3) | |
| Socio‐economic status**: | |||
| 1 (least deprived) | 56 800 (23.4) | 53 034 (21.9) | <0.001 |
| 2 | 61 647 (25.4) | 60 501 (25.0) | |
| 3 | 50 466 (20.8) | 50 709 (20.9) | |
| 4 | 42 221 (17.4) | 44 692 (18.4) | |
| 5 (most deprived) | 31 215 (12.9) | 33 413 (13.8) | |
| Smoking status: | <0.001 | ||
| Non‐smoker | 92 363 (38.1) | 80 721 (33.3) | |
| Ex‐smoker | 107 737 (44.5) | 131 510 (54.3) | |
| Current‐smoker | 36 338 (15.0) | 29 243 (12.1) | |
| Missing | 5911 (2.4) | 875 (0.4) | |
| Body mass index (kg/m2): | <0.001 | ||
| <18.5 | 6638 (2.7) | 4562 (1.9) | |
| 18.5–25 | 85 473 (35.3) | 70 102 (28.9) | |
| ≥25 | 80 458 (33.2) | 88 083 (36.4) | |
| ≥30 | 40 326 (16.6) | 63 183 (26.1) | |
| Missing | 29 454 (12.2) | 16 419 (6.8) | |
| Chronic physical illnesses: | |||
| Diabetes mellitus | 24 292 (10.0) | 52 802 (21.8) | <0.001 |
| Congestive heart failure | 7581 (3.1) | 23 774 (9.8) | <0.001 |
| Myocardial infarction | 11 459 (4.7) | 25 746 (10.6) | <0.001 |
| Stroke | 12 243 (5.1) | 19 982 (8.3) | <0.001 |
| Chronic obstructive pulmonary disease | 14 996 (6.2) | 18 229 (7.5) | <0.001 |
| Rheumatoid arthritis | 4270 (1.8) | 6031 (2.5) | <0.001 |
| Cancer | 47 431 (19.6) | 54 450 (22.5) | <0.001 |
| Parkinson's disease | 2691 (1.1) | 2293 (1.0) | <0.001 |
| Epilepsy | 3972 (1.6) | 3682 (1.5) | 0.001 |
CKD = chronic kidney disease.
White/not‐recorded: 136 119 (56.2%) and 140 784 (58.1%) patients with and without CKD, respectively, had no recorded ethnicity.
Socio‐economic status: 259 (0.1%) and 272 (0.1%) patients with and without CKD, respectively, did not have individual‐level data; we therefore used the socio‐economic status of their general practice.
Prevalence of existing use of antidepressants at index date was 16.3 and 11.9% in patients with and without CKD, respectively (Table 2). The incidence rate of new antidepressant prescription was 57.2 and 42.4/1000 person‐years in patients with and without CKD, respectively (Table 3). After adjusting for confounding, CKD remained positively associated with increased prevalence and incidence of antidepressant prescribing (Tables 2 and 3). Our results were similar to those in the main analysis after excluding patients with missing smoking status and BMI (data not shown).
Table 2.
Prevalence of antidepressant prescription by chronic kidney disease status
| No. of patients receiving antidepressants in the past 6 months | Prevalence % (95%CI) | Adjusted odds ratio (95%CI) | |||
|---|---|---|---|---|---|
| Model 1* | Model 2** | Model 3*** | |||
| Patients without CKD (N = 242 349) | 28 738 | 11.9 (11.7–12.0) | 1 (Reference) | 1 (Reference) | 1 (Reference) |
| Patients with CKD (N = 242 349) | 39 428 | 16.3 (16.1–16.4) | 1.46 (1.43–1.48) | 1.43 (1.41–1.46) | 1.35 (1.32–1.37) |
CI = confidence interval, CKD = chronic kidney disease.
Model 1: Accounted for the matched nature of the groups (age, sex, general practice, and calendar time) in conditional logistic regression analysis.
Model 2: Model 1 + adjusted by ethnicity, socio‐economic status, smoking status, and body mass index.
Model 3: Model 2 + adjusted by chronic physical illnesses.
Table 3.
Incidence of new antidepressant prescription by chronic kidney disease status
| Total follow‐up length (person‐years) | No. of patients starting antidepressants | Incidence rate (/1000 person‐years) (95%CI) | Adjusted rate ratio (95%CI) | |||
|---|---|---|---|---|---|---|
| Model 1* | Model 2** | Model 3*** | ||||
| Patients without CKD (N = 213 611) | 774 660 | 32 846 | 42.4 (41.9–42.9) | 1 (Reference) | 1 (Reference) | 1 (Reference) |
| Patients with CKD (N = 202 921) | 794 150 | 45 394 | 57.2 (56.6–57.7) | 1.35 (1.33–1.37) | 1.30 (1.28–1.32) | 1.25 (1.23–1.26) |
CI = confidence interval, CKD = chronic kidney disease, IQR = interquartile range.
Model 1: Adjusted by age, sex, and financial year, and taking account of clustering by general practice with robust standard errors using unconditional Poisson regression analysis.
Model 2: Model 1 + adjusted by ethnicity, socio‐economic status, smoking status, and body mass index.
Model 3: Model 2 + adjusted by chronic physical illnesses.
The pattern of recorded diagnoses was broadly similar between patients with and without CKD (Table 4). Regardless of CKD status, the majority of patients prescribed SSRIs had recorded diagnoses of depression or anxiety, while TCAs were prescribed for neuropathic pain or other reasons. Among patients with a recorded diagnosis of depression, the choice of antidepressant was similar between patients with and without CKD (Table 5). Irrespective of CKD status, the most commonly prescribed antidepressant was citalopram, followed by amitriptyline, fluoxetine, sertraline, and mirtazapine. There was no clear evidence that antidepressants were started at a reduced dose in patients with CKD, compared to those without CKD.
Table 4.
Recorded diagnoses for patients prescribed antidepressants stratified by chronic kidney disease status and type of antidepressant
| Patients without CKD (N = 32 846) | Patients with CKD (N = 45 394) | |||||
|---|---|---|---|---|---|---|
| SSRI N = 12 924 | TCA N = 17 672 | Others N = 2250 | SSRI N = 17 992 | TCA N = 24 262 | Others N = 3140 | |
| Depression, n (%)* | 8123 (62.9) | 4430 (25.1) | 1035 (46.0) | 11 363 (63.2) | 6257 (25.8) | 1456 (46.4) |
| Anxiety, n (%)* | 4843 (37.5) | 3902 (22.1) | 708 (31.5) | 6131 (34.1) | 5055 (20.8) | 935 (29.8) |
| Neuropathic pain, n (%)* | 625 (4.8) | 2536 (14.4) | 152 (6.8) | 997 (5.5) | 3491 (14.4) | 209 (6.7) |
| None of the above, n (%) | 3188 (24.7) | 9699 (54.9) | 894 (39.7) | 4683 (26.0) | 13 259 (54.7) | 1256 (40.0) |
CKD = chronic kidney disease, SSRI = selective serotonin reuptake inhibitor, TCA = tricyclic antidepressants.
Percentages are column percentages. Each patient may have one or more recorded diagnosis.
Table 5.
Choice of antidepressants and initial prescription dose for patients with diagnosed depression by chronic kidney disease status
| Patients without CKD N = 13 588 | Patients with CKD N = 19 076 | |||
|---|---|---|---|---|
| n (%)* | Median initial dose (mg/day) [IQR] | n (%)* | Median initial dose (mg/day) [IQR] | |
| Selective serotonin reuptake inhibitors | ||||
| Citalopram | 4934 (36.3) | 10 [10–20] | 7070 (37.1) | 10 [10–20] |
| Escitalopram | 353 (2.6) | 5 [5–10] | 429 (2.3) | 5 [5–10] |
| Fluoxetine | 1651 (12.2) | 20 [20–20] | 2270 (11.9) | 20 [20–20] |
| Fluvoxamine | <5 (<0.1) | n/a | <5 (<0.1) | n/a |
| Paroxetine | 132 (1.0) | 20 [20–20] | 144 (0.8) | 20 [20–20] |
| Sertraline | 1053 (7.8) | 50 [50–50] | 1449 (7.6) | 50 [50–50] |
| Tricyclic and related antidepressants | ||||
| Amitriptyline | 3506 (25.8) | 10 [10–20] | 5024 (26.3) | 10 [10–15] |
| Clomipramine | 26 (0.2) | 25 [10–37.5] | 27 (0.1) | 20 [10–37.5] |
| Dosulepin | 407 (3.0) | 37.5 [25–75] | 512 (2.7) | 37.5 [25–75] |
| Doxepin | 19 (0.1) | 25 [25–37.5] | 24 (0.1) | 25 [20–30] |
| Imipramine | 30 (0.2) | 25 [10–30] | 45 (0.2) | 25 [10–30] |
| Lofepramine | 113 (0.8) | 70 [70–140] | 186 (1.0) | 70 [70–140] |
| Nortriptyline | 94 (0.7) | 15 [10–15] | 158 (0.8) | 10 [10–15] |
| Trimipramine | 15 (0.1) | 25 [10–37.5] | 26 (0.1) | 25 [20–50] |
| Mianserin | 7 (0.1) | 30 [30–30] | 5 (<0.1) | 30 [30–30] |
| Trazodone | 213 (1.6) | 50 [50–100] | 250 (1.3) | 50 [50–75] |
| Monoamine oxidase inhibitors** | <5 (<0.1) | n/a | <5 (<0.1) | n/a |
| Other antidepressants: | ||||
| Agomelatine | <5 (<0.1) | n/a | <5 (<0.1) | n/a |
| Duloxetine | 98 (0.7) | 40 [30–60] | 169 (0.9) | 40 [30–60] |
| Flupentixol | 63 (0.5) | 1 [0.5–1] | 88 (0.5) | 1 [0.5–1] |
| Mirtazapine | 758 (5.6) | 15 [15–15] | 1045 (5.5) | 15 [15–15] |
| Reboxetine | <5 (<0.1) | n/a | <5 (<0.1) | n/a |
| Venlafaxine | 85 (0.6) | 75 [75–75] | 97 (0.5) | 75 [75–75] |
| Two or more different antidepressants | 27 (0.2) | n/a | 53 (0.3) | n/a |
CKD = chronic kidney disease, IQR = interquartile range.
Cell counts less than five have been suppressed to preserve patient privacy.
Phenelzine, isocarboxazid, tranylcypromine, and moclobemide are combined due to small sample sizes.
When we repeated our analyses in subgroups of renal function (Appendix 2 Tables 1–5), as the level of kidney function decreased, patients tended to be older and sicker. Among patients without CKD, those with serum creatinine results recorded in CPRD were sicker than those without. Prevalence and incidence of antidepressant prescribing increased among people with more severe kidney function: prevalence was 16.1 and 18.3%, and incidence was 56.9 and 62.3/1000 person‐years in patients with eGFR 30–59 and <30 mL/min/1.73 m2, respectively. This trend remained after adjusting for confounders. Patterns of indication for and choice of antidepressant, as well as initial prescription dose, were broadly similar for patients with different levels of kidney function.
In additional analyses with follow‐up restricted to the first six months, the percentage of patients starting antidepressants was higher amongst patients with CKD (3.5%; 7155/202 921) than amongst those without it (2.5%; 5233/213 611) (p < 0.001).
The proportion of patients starting antidepressants with their first depression diagnosis recorded between CPRD registration and index date was larger among patients with CKD (5.8%; 11 781/202 921) than those without CKD (4.0%; 8476/213 611) (p < 0.001). Similarly, the proportion of patients starting antidepressants with their first depression diagnosis recorded in CPRD after index date was larger among patients with CKD (3.6%; 7295/202 921) than those without CKD (2.4%; 5112/213 611) (p < 0.001). These results suggest that patients with CKD are more likely than those without CKD to start antidepressants both for recurrent episodes of depression, and for their first ever episode of depression.
Discussion
Main findings
In this large study, we found that patients with CKD were more likely than patients without CKD to be receiving an antidepressant, or among non‐users, to start one during follow‐up. The increase in prevalence and incidence was graded according to severity of kidney function, and the association remained after adjusting for baseline characteristics including chronic physical illnesses. The pattern of indication for and choice of antidepressants, as well as initial prescription dose, were broadly similar between patients with and without CKD.
Strengths and limitations
We used a detailed source of routinely collected data that is representative of UK population demographics.22 In the UK, GPs manage the vast majority of non‐refractory cases of mental health disorders,39, 40 and even when patients see psychiatrists in secondary care, prescriptions are usually administered by primary care.41 Therefore, we expect that most antidepressant prescriptions are captured in CPRD. To better understand the characteristics of patients with CKD, we used a comparison group of patients without CKD matched on age, sex, general practice, and calendar time. Although previous studies suggested that the proportion of patients with CKD receiving antidepressants may be high as an absolute value,42, 43 we are not aware of any study that has directly compared frequency and patterns of antidepressant prescribing between patients with and without CKD. We defined CKD using eGFR calculated from serum creatinine measurement. This method is more accurate than using recorded diagnosis of CKD, which has low sensitivity for detecting people with CKD in UK primary care databases.44
We must acknowledge several limitations of our study. First, serum creatinine testing in primary care is not universal—currently, it is only recommended and incentivized for people who are considered to be at risk for CKD.9, 38 We may have misclassified patients with unmeasured CKD to the matched control cohort, which could dilute the true association between CKD and antidepressant prescription. However, a recent study showed that the prevalence of CKD identified in CPRD is similar to that estimated in a nationally representative survey (Health Survey for England), suggesting that most CKD patients are captured in CPRD.45 Second, although we adjusted for important confounders that may be associated with mental health conditions,28, 29 the observed association between CKD status and the prevalence/incidence of antidepressant prescribing could be influenced by residual confounding due to un‐coded poor health status or access to talking therapies. Third, we examined three common diagnoses associated with antidepressant use (depression, anxiety, and neuropathic pain). However, for patients with two or more different diagnoses (e.g. depression and neuropathic pain), it was not possible to determine the most likely indication for antidepressant prescription because diagnosis and prescription records are separate in CPRD. Also, patients may have received antidepressants for other reasons, such as non‐neuropathic pain and insomnia, but reliable identification of these conditions has not been established in CPRD. Finally, we demonstrated that the initial dose of antidepressant prescribed was similar in depressed patients with and without CKD. However, this does not ensure that the subsequent dose was also similar between those with and without CKD (as doctors may increase or decrease antidepressant dose after initial prescription, according to perceived effectiveness or side effects).
Comparison with other studies
Two studies conducted in the USA have examined antidepressant use in patients with CKD.42, 43 The Chronic Renal Insufficiency Cohort study investigated the proportion of patients with CKD receiving an antidepressant at recruitment.43 Of 3853 participants, 700 (18.2%) were taking antidepressants. This number is close to the prevalence of existing users of antidepressants in patients with CKD (16.3%) found in our study. Another US cohort study showed that around 30% of patients with CKD (with or without diagnosis of depression) were receiving antidepressants at any time during a 2‐year period between 2004 and 2006.42 These antidepressant users appeared to include both existing and new users of antidepressants. Our study demonstrated the incident rate of antidepressant prescription at 57.2/1000 person‐years in patient with CKD. Together with the prevalence of existing users (16.3%), the cumulative effect of this was consistent with over 30% of CKD patients exposed to antidepressants during follow‐up. Neither US study included a comparison group of patients without CKD in order to compare prescribing in CKD patients to that in the general population. Indication and choice of antidepressants were also not examined.
Explanation of findings and implication for future studies
Patients with mild CKD generally do not have related physical symptoms. However, a previous study has suggested that negative perception of CKD is associated with depression and lower quality of life, even in the early stages of CKD.46 Patients with more advanced CKD (eGFR <30 mL/min/1.73m2) tend to have symptoms including fatigue, nausea, sleep disturbances, itching, and peripheral neuropathy, any of which could influence quality of life and mental health. This is in line with our finding that patients with advanced CKD were more likely to be prescribed antidepressants, even without a specifically coded diagnosis of depression and anxiety.
While most SSRIs were associated with a coded diagnosis of depression or anxiety, more than half of patients starting TCAs (mostly amitriptyline) did not have any recorded diagnoses of depression, anxiety, or neuropathic pain. Amitriptyline may have been predominantly prescribed as an off‐label indication for non‐psychiatric conditions such as chronic pain and insomnia.5, 6, 7 When restricted to patients with a coded diagnosis of depression, SSRIs accounted for the majority of antidepressant prescribing, which is in keeping with current guidelines for management of depression.39 Patterns of antidepressant choice did not differ substantially according to CKD status or level of kidney function. This is probably because to date there is no evidence of greater efficacy or safety concerns for specific antidepressants among patients with CKD.20, 21
Increased adverse events as renal function declines are an important concern. For example, amitriptyline clearance is reduced in patients with decreased kidney function.47 As a result, amitriptyline may accumulate, causing serious adverse outcomes through neurotoxicity48 and cardiotoxicity.49 Another example is the potential amplification of bleeding risk both with use of SSRIs and with decreased kidney function itself.50 Finally, the results of our analyses stratified by severity of renal function demonstrate that many patients are prescribed antidepressants at levels of renal function below those where cessation is recommended by manufacturers (e.g. eGFR <30 mL/min/1.73 m2). According to the British National Formulary,4 escitalopram, paroxetine, sertraline, imipramine, lofepramine, trazodone, duloxetine, mirtazapine, and venlafaxine should be used with caution or avoided in those with reduced renal function, but our real‐world data suggest that these drugs are prescribed similarly in patients with moderately or severely decreased kidney function, compared to those with normal kidney function. Better evidence regarding the potential adverse effects of these drugs for patients with decreased kidney function is needed.
Conclusions
This study using a large UK database suggests that patients with CKD are more likely to be prescribed antidepressants than the general population, whilst prescribing patterns did not appear to be influenced by kidney function. These real‐world data emphasize the need for research investigating the potential adverse effects of antidepressant therapy in people with decreased kidney function.
Conflict of Interest
The authors declare no conflict of interest.
Key Points.
This study examined details of antidepressant prescribing in patients with chronic kidney disease using a large, contemporary UK database of routine medical record data. We defined chronic kidney disease using serum creatinine measurements and compared people with and without chronic kidney disease matched for age, sex, general practice, and calendar time.
Patients with chronic kidney disease were exposed to antidepressants more frequently; with higher prevalence and incidence of antidepressant prescribing than the general population. The positive association between chronic kidney disease and increased frequency of antidepressant prescribing remained after adjusting for measured confounders such as diabetes and cardiovascular disease.
Among patients starting antidepressants, indication for antidepressant prescription (recorded diagnoses of depression, anxiety, or neuropathic pain) was similar between patients with and without chronic kidney disease. Antidepressant choice was also similar between depressed patients with and without chronic kidney disease.
Author Contributions
M.I. planned the study, carried out the data extraction, processing and analysis, and drafted the manuscript. D.N. and L.T. contributed substantially to the study design, interpretation of the results, and writing of the manuscript. K.M. and H.M. supported the data processing and writing of the manuscript. L.S. was involved in discussions of the analytical approach to this study and made comments on the results. All authors read and approved the final manuscript.
Supporting information
Appendix S1. List of diagnosis codes indicative of depression, anxiety, and neuropathic pain in Clinical Practice Research Datalink.
Appendix S2. Subgroup analyses according to level of kidney function (among patients with CKD) and creatinine measurement (among patients without CKD).
Acknowledgments
This research received no specific grant from any funding agency in the public, commercial or not‐for‐profit sectors. At an individual level, Dr Masao Iwagami is funded by Honjo International Scholarship Foundation, Dr Laurie Tomlinson is funded by a Wellcome Trust Intermediate Clinical Fellowship [WT101143MA], and Dr Helen McDonald was funded by a Kidney Research UK studentship [ST2/2011]. The funders had no role in the execution of this study or interpretation of results.
Iwagami, M. , Tomlinson, L. A. , Mansfield, K. E. , McDonald, H. I. , Smeeth, L. , and Nitsch, D. (2017) Prevalence, incidence, indication, and choice of antidepressants in patients with and without chronic kidney disease: a matched cohort study in UK Clinical Practice Research Datalink. Pharmacoepidemiol Drug Saf, 26: 792–801. doi: 10.1002/pds.4212.
References
- 1. Hsiao CJ, Cherry DK, Beatty PC, Rechtsteiner EA. National Ambulatory Medical Care Survey: 2007 summary. Natl Health Stat Rep 2010; 27: 1–32. [PubMed] [Google Scholar]
- 2. Health & Social Care Information Centre . Health Survey for England—2013. Available at: http://www.hscic.gov.uk/catalogue/PUB16076 [1 August 2016].
- 3. Ilyas S, Moncrieff J. Trends in prescriptions and costs of drugs for mental disorders in England, 1998–2010. Br J Psychiatry 2012; 200: 393–398 https://doi.org/10.1192/bjp.bp.111.104257. [DOI] [PubMed] [Google Scholar]
- 4. British Medical Association, Royal Pharmaceutical Society of Great Britain . British national formulary. Available at: https://www.medicinescomplete.com/mc/bnf/current/ [1 August 2016].
- 5. Chouinard G. The search for new off‐label indications for antidepressant, antianxiety, antipsychotic and anticonvulsant drugs. J Psychiatry Neurosci 2006; 31: 168–176. [PMC free article] [PubMed] [Google Scholar]
- 6. Mercier A, Auger‐Aubin I, Lebeau JP, et al. Evidence of prescription of antidepressants for non‐psychiatric conditions in primary care: an analysis of guidelines and systematic reviews. BMC Fam Pract 2013; 14: 55 https://doi.org/10.1186/1471‐2296‐14‐55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lai LL, Tan MH, Lai YC. Prevalence and factors associated with off‐label antidepressant prescriptions for insomnia. Drug Healthc Patient Saf 2011; 3: 27–36 https://doi.org/10.2147/DHPS.S21079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Jha V, Garcia‐Garcia G, Iseki K, et al. Chronic kidney disease: global dimension and perspectives. Lancet 2013; 382: 260–272 https://doi.org/10.1016/S0140‐6736(13)60687‐X. [DOI] [PubMed] [Google Scholar]
- 9. National Institute for Health and Care Excellence . Chronic kidney disease in adults: assessment and management. Available at: https://www.nice.org.uk/guidance/cg182 [1 August 2016]. [PubMed]
- 10. MacLaughlin HL, Hall WL, Sanders TA, Macdougall IC. Risk for chronic kidney disease increases with obesity: Health Survey for England 2010. Public Health Nutr 2015; 18: 3349–3354 https://doi.org/10.1017/s1368980015000488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 2004; 351: 1296–1305 https://doi.org/10.1056/NEJMoa041031. [DOI] [PubMed] [Google Scholar]
- 12. Lee YJ, Kim MS, Cho S, Kim SR. Association of depression and anxiety with reduced quality of life in patients with predialysis chronic kidney disease. Int J Clin Pract 2013; 67: 363–368 https://doi.org/10.1111/ijcp.12020. [DOI] [PubMed] [Google Scholar]
- 13. Palmer S, Vecchio M, Craig JC, et al. Prevalence of depression in chronic kidney disease: systematic review and meta‐analysis of observational studies. Kidney Int 2013; 84: 179–191 https://doi.org/10.1038/ki.2013.77. [DOI] [PubMed] [Google Scholar]
- 14. Anderson RJ, Freedland KE, Clouse RE, Lustman PJ. The prevalence of comorbid depression in adults with diabetes: a meta‐analysis. Diabetes Care 2001; 24: 1069–1078. [DOI] [PubMed] [Google Scholar]
- 15. Nair N, Farmer C, Gongora E, Dehmer GJ. Commonality between depression and heart failure. Am J Cardiol 2012; 109: 768–772 https://doi.org/10.1016/j.amjcard.2011.10.039. [DOI] [PubMed] [Google Scholar]
- 16. Cohen SD, Patel SS, Khetpal P, Peterson RA, Kimmel PL. Pain, sleep disturbance, and quality of life in patients with chronic kidney disease. Clin J Am Soc Nephrol 2007; 2: 919–925 https://doi.org/10.2215/cjn.00820207. [DOI] [PubMed] [Google Scholar]
- 17. Wu J, Ginsberg JS, Zhan M, et al. Chronic pain and analgesic use in CKD: implications for patient safety. Clin J Am Soc Nephrol 2015; 10: 435–442 https://doi.org/10.2215/cjn.06520714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Charytan D, Kuntz RE. The exclusion of patients with chronic kidney disease from clinical trials in coronary artery disease. Kidney Int 2006; 70: 2021–2030 https://doi.org/10.1038/sj.ki.5001934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Glassman AH, O'Connor CM, Califf RM, et al. Sertraline treatment of major depression in patients with acute MI or unstable angina. JAMA 2002; 288: 701–709. [DOI] [PubMed] [Google Scholar]
- 20. Nagler EV, Webster AC, Vanholder R, Zoccali C. Antidepressants for depression in stage 3–5 chronic kidney disease: a systematic review of pharmacokinetics, efficacy and safety with recommendations by European Renal Best Practice (ERBP). Nephrol Dial Transplant 2012; 27: 3736–3745 https://doi.org/10.1093/ndt/gfs295. [DOI] [PubMed] [Google Scholar]
- 21. Hedayati SS, Yalamanchili V, Finkelstein FO. A practical approach to the treatment of depression in patients with chronic kidney disease and end‐stage renal disease. Kidney Int 2012; 81: 247–255 https://doi.org/10.1038/ki.2011.358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Herrett E, Gallagher AM, Bhaskaran K, et al. Data resource profile: Clinical Practice Research Datalink (CPRD). Int J Epidemiol 2015; 44: 827–836 https://doi.org/10.1093/ije/dyv098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Hospital Episode Statistics . Available at: http://www.hscic.gov.uk/hes [1 August 2016].
- 24. Lewis JD, Bilker WB, Weinstein RB, Strom BL. The relationship between time since registration and measured incidence rates in the General Practice Research Database. Pharmacoepidemiol Drug Saf 2005; 14: 443–451 https://doi.org/10.1002/pds.1115. [DOI] [PubMed] [Google Scholar]
- 25. National Kidney Foundation . K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis 2002; 39: S1–266. [PubMed] [Google Scholar]
- 26. Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009; 150: 604–612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. van Eijk ME, Bahri P, Dekker G, et al. Use of prevalence and incidence measures to describe age‐related prescribing of antidepressants with and without anticholinergic effects. J Clin Epidemiol 2000; 53: 645–651. [DOI] [PubMed] [Google Scholar]
- 28. Rayner L, Price A, Evans A, Valsraj K, Higginson IJ, Hotopf M. Antidepressants for depression in physically ill people. Cochrane Database Syst Rev 2010; 3: CD007503 https://doi.org/10.1002/14651858.CD007503.pub2. [DOI] [PubMed] [Google Scholar]
- 29. National Institute for Health and Care Excellence . Depression in adults with chronic physical health problem: recognition and management. Available at: https://www.nice.org.uk/guidance/cg91 [1 August 2016].
- 30. Hippisley‐Cox J, Coupland C. Derivation and validation of updated QFracture algorithm to predict risk of osteoporotic fracture in primary care in the United Kingdom: prospective open cohort study. BMJ 2012; 344: e3427 https://doi.org/10.1136/bmj.e3427. [DOI] [PubMed] [Google Scholar]
- 31. Hippisley‐Cox J, Coupland C. Predicting risk of upper gastrointestinal bleed and intracranial bleed with anticoagulants: cohort study to derive and validate the QBleed scores. BMJ 2014; 349: g4606 https://doi.org/10.1136/bmj.g4606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Department for Communities and Local Government . English indices of deprivation 2010. Available at: https://www.gov.uk/government/statistics/english‐indices‐of‐deprivation‐2010 [1 August 2016].
- 33. Rait G, Walters K, Griffin M, Buszewicz M, Petersen I, Nazareth I. Recent trends in the incidence of recorded depression in primary care. Br J Psychiatry 2009; 195: 520–524 https://doi.org/10.1192/bjp.bp.108.058636. [DOI] [PubMed] [Google Scholar]
- 34. Walters K, Rait G, Griffin M, Buszewicz M, Nazareth I. Recent trends in the incidence of anxiety diagnoses and symptoms in primary care. PLoS One 2012; 7: e41670 https://doi.org/10.1371/journal.pone.0041670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Kendrick T, Stuart B, Newell C, Geraghty AW, Moore M. Changes in rates of recorded depression in English primary care 2003–2013: time trend analyses of effects of the economic recession, and the GP contract quality outcomes framework (QOF). J Affect Disord 2015; 180: 68–78 https://doi.org/10.1016/j.jad.2015.03.040. [DOI] [PubMed] [Google Scholar]
- 36. Gardarsdottir H, Heerdink ER, van Dijk L, Egberts AC. Indications for antidepressant drug prescribing in general practice in the Netherlands. J Affect Disord 2007; 98: 109–115 https://doi.org/10.1016/j.jad.2006.07.003. [DOI] [PubMed] [Google Scholar]
- 37. Abbing‐Karahagopian V, Huerta C, Souverein PC, et al. Antidepressant prescribing in five European countries: application of common definitions to assess the prevalence, clinical observations, and methodological implications. Eur J Clin Pharmacol 2014; 70: 849–857 https://doi.org/10.1007/s00228‐014‐1676‐z. [DOI] [PubMed] [Google Scholar]
- 38. Health & Social Care Information Centre . Quality and outcomes framework. Available at: http://www.hscic.gov.uk/qof [1 August 2016].
- 39. National Institute for Health and Care Excellence . Depression in adults: recognition and management. Available at: https://www.nice.org.uk/guidance/cg90 [1 August 2016]. [PubMed]
- 40. National Institute for Health and Care Excellence . Generalised anxiety disorder and panic disorder in adults: management. Available at: https://www.nice.org.uk/guidance/cg113 [1 August 2016]. [PubMed]
- 41. Crump BJ, Panton R, Drummond MF, Marchment M, Hawkes RA. Transferring the costs of expensive treatments from secondary to primary care. BMJ 1995; 310: 509–512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Balogun RA, Abdel‐Rahman EM, Balogun SA, et al. Association of depression and antidepressant use with mortality in a large cohort of patients with nondialysis‐dependent CKD. Clin J Am Soc Nephrol 2012; 7: 1793–1800 https://doi.org/10.2215/cjn.02650312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Fischer MJ, Xie D, Jordan N, et al. Factors associated with depressive symptoms and use of antidepressant medications among participants in the Chronic Renal Insufficiency Cohort (CRIC) and Hispanic‐CRIC Studies. Am J Kidney Dis 2012; 60: 27–38 https://doi.org/10.1053/j.ajkd.2011.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Fraser SD, Parkes J, Culliford D, Santer M, Roderick PJ. Timeliness in chronic kidney disease and albuminuria identification: a retrospective cohort study. BMC Fam Pract 2015; 16: 18 https://doi.org/10.1186/s12875‐015‐0235‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Iwagami M, Tomlinson LA, Mansfield KE, et al. Validity of estimated prevalence of decreased kidney function and renal replacement therapy from primary care electronic health records compared to national survey and registry data in the UK. Nephrol Dial Transplant 2017; 32: ii142–ii150 https://doi.org/10.1093/ndt/gfw318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Shidler NR, Peterson RA, Kimmel PL. Quality of life and psychosocial relationships in patients with chronic renal insufficiency. Am J Kidney Dis 1998; 32: 557–566. [DOI] [PubMed] [Google Scholar]
- 47. Tasset JJ, Singh S, Pesce AJ. Evaluation of amitriptyline pharmacokinetics during peritoneal dialysis. Ther Drug Monit 1985; 7: 255–257. [DOI] [PubMed] [Google Scholar]
- 48. Livingston RL, Zucker DK, Isenberg K, Wetzel RD. Tricyclic antidepressants and delirium. J Clin Psychiatry 1983; 44: 173–176. [PubMed] [Google Scholar]
- 49. Scollins MJ, Robinson DS, Nies A. Cardiotoxicity of amitriptyline. Lancet 1972; 2: 1202. [DOI] [PubMed] [Google Scholar]
- 50. Olesen JB, Lip GY, Kamper AL, et al. Stroke and bleeding in atrial fibrillation with chronic kidney disease. N Engl J Med 2012; 367: 625–635 https://doi.org/10.1056/NEJMoa1105594. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1. List of diagnosis codes indicative of depression, anxiety, and neuropathic pain in Clinical Practice Research Datalink.
Appendix S2. Subgroup analyses according to level of kidney function (among patients with CKD) and creatinine measurement (among patients without CKD).


