Abstract
Although the pathogenesis of non-alcoholic fatty liver disease (NAFLD) is not completely understood, the increased influx of free fatty acids (FFAs) into the liver and the FFA-induced hepatic endoplasmic reticulum (ER) stress are two crucial pathogenic processes in the initiation and development of NAFLD. In this study we investigated the effects of astragaloside IV (AS-IV), a bioactive compound purified from Astragali Radix, on FFA-induced lipid accumulation in hepatocytes and elucidated the underlying mechanisms. Human HepG2 cells and primary murine hepatocytes were exposed to FFAs (1 mmol/L, oleate/palmitate, 2:1 ratio) with or without AS-IV for 24 h. Exposure to FFAs induced marked lipid accumulation in hepatocytes, whereas co-treatment with AS-IV (100 μg/mL) significantly attenuated this phenomenon. Notably, AS-IV (50–200 μg/mL) concentration-dependently enhanced the phosphorylation of AMPK, acetyl-CoA carboxylase (ACC) and SREBP-1c, inhibited the accumulation and nuclear translocation of mature SREBP-1 and subsequently decreased the mRNA levels of lipogenic genes including acc1, fas and scd1. AS-IV treatment also concentration-dependently attenuated FFA-induced hepatic ER stress evidenced by the reduction of the key markers, GRP78, CHOP and p-PERK. Pretreated the cells with the AMPK inhibitor compound C (20 μmol/L) greatly diminished these beneficial effects of AS-IV. Our results demonstrate that AS-IV attenuates FFA-induced ER stress and lipid accumulation in an AMPK-dependent manner in hepatocytes, which supports its use as promising therapeutics for hepatic steatosis.
Keywords: astragaloside IV, non-alcoholic fatty liver disease, hepatocytes, HepG2 cells, lipid accumulation, ER stress, AMPK, ACC, SREBP-1, compound C
Introduction
Non-alcoholic fatty liver disease (NAFLD) affects approximately 15%-30% of the general population1,2, and it has become the most common chronic liver disease worldwide due to the prevalence of obesity3,4. NAFLD is a hepatic manifestation of the metabolic syndrome associated with type 2 diabetes, obesity, and dyslipidemia. The pathological spectrum of NAFLD ranges from simple steatosis to the more severe non-alcoholic steatohepatitis (NASH), which may progress to fibrosis, cirrhosis and hepatocellular carcinoma5. Although the pathogenesis of NAFLD is not completely understood, the increased influx of free fatty acids (FFAs) into the liver and the FFA-induced hepatic endoplasmic reticulum (ER) stress are two crucial pathogenic processes in the initiation and development of NAFLD6,7. No effective and safe therapy is available for NAFLD, except lifestyle intervention-mediated weight loss. Therefore, there is an urgent need to identify new effective therapeutic strategies.
AMP-activated protein kinase (AMPK) is a key cellular energy and nutrient sensor that belongs to the serine/threonine kinase family. AMPK is expressed almost universally in eukaryotes as heterotrimeric complexes composed of a catalytic subunit (α) and two regulatory subunits (β and γ). AMPK is activated by phosphorylation at Thr172 in the α subunit8. Numerous studies also demonstrated that AMPK is a critical regulator of hepatic lipogenesis, which makes this enzyme a potential therapeutic target for the treatment of hepatic steatosis9. AMPK activation phosphorylates and inactivates acetyl-CoA carboxylase (ACC) to switch off fatty acid synthesis acutely10. AMPK activation also decreases fatty acid synthesis chronically via a reduction in the transcriptional activation of sterol regulatory element binding protein-1c (SREBP-1c), which is a pivotal transcription factor in the regulation of lipogenic genes including acc1, fatty acid synthase (fas) and stearoyl-Coenzyme A desaturase 1 (scd1)11. A recent study revealed that AMPK phosphorylates SREBP-1c at Ser372, which suppresses its cleavage and nuclear translocation, and represses its target gene expression in hepatocytes exposed to high glucose, which leads to reduced lipogenesis and lipid accumulation12. Other recent studies demonstrated a negative regulation of AMPK activation on lipid-induced ER stress in hepatocytes13,14,15. ER stress is a disruption of ER homeostasis that is characterized by the increased expression of key markers such as glucose-regulated protein 78 (GRP78), C/EBP homologous protein (CHOP) and phosphorylated protein kinase-like ER kinase (p-PERK), which promote hepatic lipogenesis and lipid accumulation16,17. Taken together, AMPK activation may attenuate hepatic lipid accumulation via inhibition of lipogenesis and ER stress, which supports AMPK as a potential therapeutic target for the treatment of hepatic steatosis.
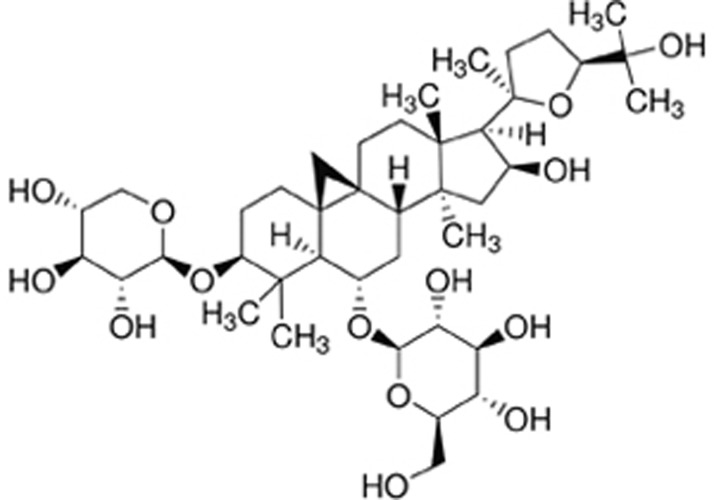
There is a growing interest in the development of AMPK activators as promising treatments for hepatic steatosis. Accumulating evidence indicates that many bioactive compounds purified from traditional Chinese herbs attenuate hepatic lipid accumulation via AMPK activation18,19,20. Astragali Radix is one of the most commonly used traditional Chinese medicines prepared from the roots of Astragalus membranaceus (Fisch) Bunge or Astragalus mongholicus Bunge, and it has been widely used in the treatment of numerous diseases for the past centuries. Astragali Radix is described as a restorative agent that improves immunity and diabetes in Shen Nong's herbal classic and the Compendium of Materia Medica. Astragaloside IV (AS-IV) (Figure 1) is the primary bioactive component purified from Astragali Radix, and it possesses nearly identical therapeutic functions. However, the clinical use of Astragali Radix or AS-IV for fatty liver diseases is not reported. A previous animal study revealed that Astragali Radix ameliorated insulin resistance and fatty liver in type 2 diabetic rats21. Emerging evidence also demonstrates that AS-IV efficiently improves hepatic steatosis in obese mice, likely via an enhancement of leptin sensitivity and modulation of the thermogenic network22. AS-IV also suppresses palmitate-induced ROS generation and inflammation via AMPK activation in endothelial cells23. Taken together, these studies suggest that AS-IV is an AMPK activator in the liver and may serve as potential therapeutics for hepatic steatosis. However, little is known about the direct effect of AS-IV on lipid accumulation in hepatocytes. This study investigated the direct effect of AS-IV on lipid accumulation in hepatocytes and elucidated the underlying mechanisms. We demonstrated, for the first time, that AS-IV attenuated FFA-induced ER stress and lipid accumulation via AMPK activation in hepatocytes, which further supports its use as promising therapeutics for hepatic steatosis.
Figure 1.
Chemical structure of AS-IV. AS-IV, Astragaloside IV.
Materials and methods
Materials and reagents
HBSS buffer, Williams' E Medium, RIPA buffer and Protease & Phosphatase Inhibitors Cocktail were purchased from Thermo Scientific (Rockford, IL, USA). Oleate, palmitate, collagenase IV, Oil red O, compound C and AICAR were purchased from Sigma-Aldrich (St Louis, MO, USA). AS-IV was obtained from Aladdin (Shanghai, China). Primary antibodies, including p-AMPKα (Thr172), p-ACC (Ser79), p-SREBP-1c (Ser372), AMPKα, ACC, PERK and β-actin, were purchased from Cell Signaling Technology (Danvers, MA, USA). SREBP-1, LaminB1, p-PERK and GRP78 were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). CHOP was obtained from Proteintech (Wuhan, China) and Santa Cruz Biotechnology (Santa Cruz, CA, USA). LI-COR IRDye 800CW secondary antibodies were obtained from LI-COR Biosciences (Lincoln, NE, USA). FITC-conjugated goat anti-rabbit IgG was purchased from Proteintech (Wuhan, China). Alexa Fluor® 594-conjugated donkey anti-mouse IgG was obtained from Thermo Scientific (Rockford, IL, USA). SYBR Green I Master was purchased from Roche Applied Science (Mannheim, Germany). Cell Counting Kit-8 (CCK-8) was purchased from Dojindo Laboratories (Tokyo, Japan). The NE-PER Nuclear and Cytoplasmic Extraction Reagent kit was purchased from Thermo Scientific (Rockford, IL, USA). The SuperPrep Cell Lysis & RT Kit for qPCR was purchased from TOYOBO (Tokyo, Japan).
Preparation of AS-IV solution
Purified AS-IV powder was purchased and reconstituted in dimethyl sulfoxide (DMSO) for a stock solution (200 mg/mL) as previously described24. The stock solution was stored at -20 °C until use.
Cell culture and treatment
Human HepG2 hepatoma cells were cultured in MEM medium supplemented with 10% fetal bovine serum, 1 mmol/L sodium pyruvate, 100 U/mL penicillin, and 100 μg/mL streptomycin. Cells at 70% confluence were starved in serum-free medium overnight before treatment.
Primary murine hepatocytes were isolated from up to 12-week-old C57BL/6 mice, using a previously described two-step collagenase perfusion method25. Livers were perfused with HBSS buffer, followed by HBSS containing 100 U/mL collagenase IV for digestion at a flow rate of 9 mL/min. Primary hepatocytes were plated on collagen-coated (8 μg/cm2) plates in Williams' E Medium supplemented with 10% fetal bovine serum, 2 mmol/L L-glutamine, 100 nmol/L dexamethasone and the antibiotics described above for 4 h. Hepatocytes were maintained overnight in serum-free Williams' E Medium and used within 30 h after plating.
All cells were maintained at 37 °C in a humidified incubator containing 5% CO2. Hepatocytes were exposed to 1 mmol/L FFAs (oleate/palmitate, 2:1 ratio) to induce lipid accumulation26, with or without different concentrations (50–200 μg/mL) of AS-IV for 24 h. Cells were pretreated with the AMPK inhibitor compound C (20 μmol/L) for 1 h and treated with AS-IV (100 μg/mL) supplemented with FFAs for 24 h. Cells were treated with the AMPK activator AICAR (1 mmol/L) for 2 h as a positive control.
Cell viability assay
Cell viability was measured using the CCK-8 assay according to the manufacturer's instructions. Briefly, human HepG2 cells or primary murine hepatocytes (4×103 cells/well) were seeded into 96-well plates and treated with FFAs (0.25–2 mmol/L), AS-IV (50–400 μg/mL) or the combination after attachment. The same concentration of vehicle was added to the control group. Cells were treated for 24 h and incubated in a 1:10 diluted CCK-8 solution for 2 h at 37 °C. The absorbance was measured at 450 nm using a microplate reader.
Oil red O staining
Cells were rinsed with ice-cold phosphate buffered saline (PBS) and fixed with 10% formaldehyde at room temperature for 15 min. Cells were washed with PBS and immersed in an Oil red O working solution (60% Oil red O dye and 40% water) at room temperature for 30 min. Cells were washed briefly with 75% ethanol to remove any unbound dye and observed under a Leica DMI4000B inverted microscope after washing with PBS. The dye was extracted using 100% isopropanol immediately via gentle pipetting, and absorbance was measured at 510 nm with a spectrophotometer to quantify the intracellular lipid content.
Western blot analysis
Cells were washed with ice-cold PBS, lysed on ice in RIPA buffer supplemented with a cocktail of protease and phosphatase inhibitors for 15 min and centrifuged (14 000×g, 15 min and 4 °C) to achieve total cellular protein extraction. Nuclear extracts were prepared using the NE-PER Nuclear and Cytoplasmic Extraction Reagents kit according to the manufacturer's instructions. Protein concentrations were determined using a BCA Protein Assay kit. Equal amounts of protein were separated by SDS-PAGE and transferred onto PVDF membranes. The membranes were blocked with 5% bovine serum albumin or non-fat milk for 1 h at room temperature and hybridized overnight at 4 °C with the aforementioned primary antibodies. Membranes were incubated with LI-COR IRDye 800CW secondary antibodies at room temperature for 1 h, and the antibody-antigen complexes were visualized using the Odyssey Infrared Imaging System (LI-COR Biosciences, Lincoln, NE, USA). The results were analyzed quantitatively via densitometry using Image-Pro Plus software.
Immunofluorescence
Cells were fixed with 4% paraformaldehyde in PBS for 15 min and permeabilized with 0.5% Triton X-100 in PBS at room temperature for 15 min. Cells were blocked with 2% bovine serum albumin in PBS for 1 h and incubated with primary anti-GRP78 and anti-CHOP antibodies overnight at 4 °C. Cells were rinsed with PBS and incubated with either FITC-conjugated goat anti-rabbit IgG or Alexa Fluor® 594-conjugated donkey anti-mouse IgG for 1 h at room temperature, followed by incubation in the fluorescent nucleic acid dye Hoechst 33258 for 10 min. Cells were extensively washed in PBS, and images were captured using an Axio Observer Z1 microscope.
Quantitative real-time PCR
Total RNA was isolated from hepatocytes, and reverse transcription was performed using a SuperPrep Cell Lysis & RT Kit for qPCR. Expression of mRNA was quantified using the LightCycler® 480 detection system (Roche Applied Science, Mannheim, Germany) with SYBR Green I Master according to the manufacturer's instructions. The 2−ΔΔCT method was used to analyze the expression of target genes, and β-actin was used as an internal control. PCR primer pair sequences are available upon request.
Statistical analysis
All data are expressed as the mean±SEM of at least three separate experiments in each group. Unpaired two-tailed Student's t test was used for comparisons of differences between groups. A value of P<0.05 was considered statistically significant.
Results
Cytotoxicity of FFAs and AS-IV on hepatocytes
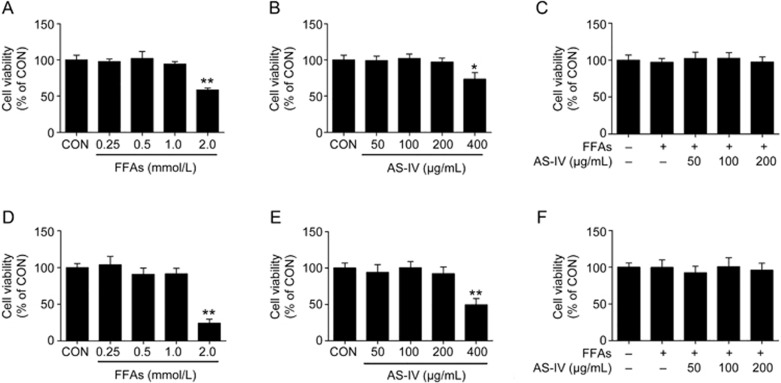
Hepatocytes were exposed to a mixture of FFAs for 24 h to induce intracellular lipid accumulation and treated with or without AS-IV to investigate the effect of AS-IV on hepatic steatosis in vitro. The cytotoxicity of FFAs and AS-IV on hepatocytes was measured using a CCK-8 assay. We found that cell viability was not affected by up to 1 mmol/L FFAs in human HepG2 cells or primary murine hepatocytes, but 2 mmol/L FFA exposure significantly reduced cell viability (Figure 2A, 2D). AS-IV treatment did not exhibit any cytotoxic effects up to 200 μg/mL in hepatocytes, but an obvious decrease in cell viability was observed at 400 μg/mL (Figure 2B, 2E). Therefore, co-treatment of hepatocytes with FFAs (1 mmol/L) and AS-IV (50–200 μg/mL) was used in further studies, and no cytotoxicity was observed (Figure 2C, 2F).
Figure 2.
Cytotoxicity of FFAs and AS-IV on hepatocytes. HepG2 cells or primary murine hepatocytes were treated with various concentrations of FFAs, AS-IV or their combination for 24 h and analyzed using a CCK-8 assay. (A) Cell viability of HepG2 cells exposed to FFAs (0.25–2 mmol/L). (B) Cell viability of HepG2 cells treated with AS-IV (50–400 μg/mL). (C) Cell viability of HepG2 cells co-treated with FFAs (1 mmol/L) and AS-IV (50–200 μg/mL). (D) Cell viability of primary murine hepatocytes exposed to FFAs (0.25–2 mmol/L). (E) Cell viability of primary murine hepatocytes treated with AS-IV (50–400 μg/mL). (F) Cell viability of primary murine hepatocytes co-treated with FFAs (1 mmol/L) and AS-IV (50–200 μg/mL). Data are presented as the mean±SEM of three independent experiments (n=3). *P<0.05, **P<0.01 compared with the control group. FFAs, free fatty acids; AS-IV, astragaloside IV.
AS-IV attenuates FFA-induced lipid accumulation in hepatocytes
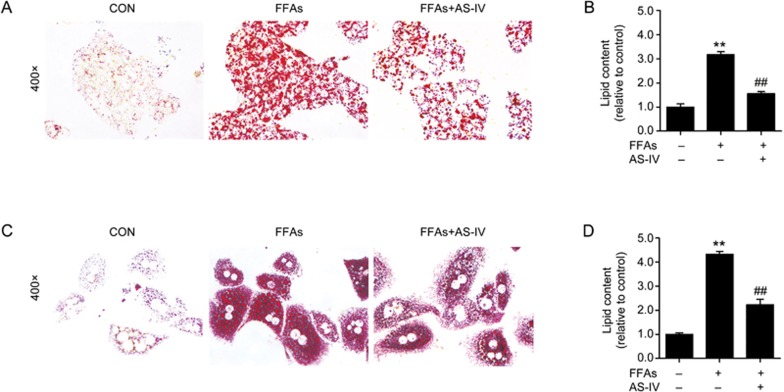
Oil red O staining revealed that exposure to FFAs markedly induced an increase in lipid accumulation compared with the control group in HepG2 cells, which indicated that FFA treatment successfully established a cell model of hepatic steatosis. However, AS-IV under co-treatment with FFAs and AS-IV in HepG2 cells significantly attenuated this accumulation (Figure 3A). The relative quantification of intracellular lipid content confirmed these results (Figure 3B). We also observed that AS-IV treatment significantly alleviated FFA-induced lipid accumulation in primary murine hepatocytes (Figure 3C, 3D). Taken together, these findings suggest that AS-IV attenuates FFA-induced lipid accumulation in hepatocytes.
Figure 3.
AS-IV attenuates FFA-induced lipid accumulation in hepatocytes. HepG2 cells and primary murine hepatocytes exposed to FFAs (1 mmol/L) were treated with or without AS-IV (100 μg/mL) for 24 h. (A) Representative images of Oil red O staining at 400× magnification in HepG2 cells. (B) Quantification of intracellular lipid content by Oil red O staining in HepG2 cells. (C) Representative images of Oil red O staining at 400× magnification in primary murine hepatocytes. (D) Quantification of intracellular lipid content by Oil red O staining in primary murine hepatocytes. Data are presented as the mean±SEM of three independent experiments (n=3). *P<0.05, **P<0.01 compared with the control group; ##P<0.01 compared with the FFA group. FFAs, free fatty acids; AS-IV, astragaloside IV.
AS-IV activates the AMPK pathway and inhibits lipogenesis in FFA-exposed hepatocytes
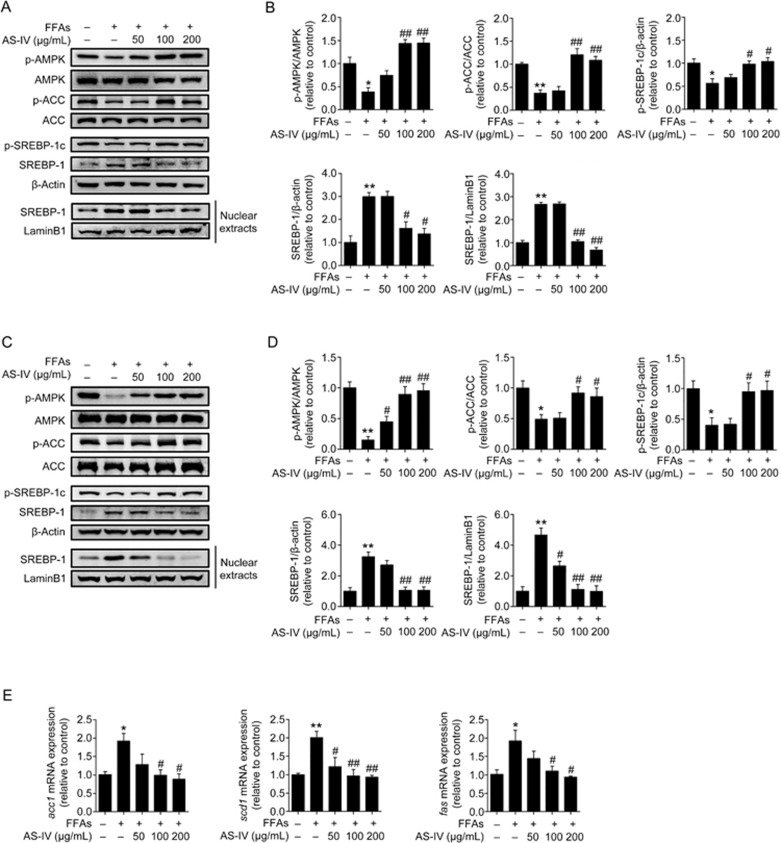
The AMPK signaling pathway was analyzed to elucidate the underlying mechanisms of the beneficial effect of AS-IV on FFA-induced lipid accumulation because AMPK is a critical regulator of hepatic lipogenesis. Figures 4A and 4B show that FFA exposure significantly induced a decrease in the phosphorylation of AMPK in HepG2 cells and AS-IV treatment dose-dependently enhanced this effect. AS-IV treatment also resulted in a dose-dependent increase in ACC phosphorylation, which is a direct target enzyme of AMPK. No obvious changes in the protein levels of AMPK and ACC were observed. AS-IV treatment dose-dependently increased SREBP-1c phosphorylation and decreased the accumulation of mature SREBP-1 in FFA-exposed HepG2 cells. We also found that AS-IV treatment concentration-dependently inhibited the increased nuclear translocation of mature SREBP-1 in the FFA group (Figure 4A, 4B). These results were further confirmed in primary murine hepatocytes (Figure 4C, 4D). The mRNA levels of lipogenic genes that are transcriptionally activated by SREBP-1, such as acc1, fas and scd1, were significantly increased by FFA exposure in HepG2 cells, and AS-IV treatment attenuated this increase in a concentration-dependent manner (Figure 4E). These data demonstrate that AS-IV activates the AMPK pathway and inhibits lipogenesis in FFA-exposed hepatocytes.
Figure 4.
AS-IV activates the AMPK pathway and inhibits lipogenesis in FFA-exposed hepatocytes. HepG2 cells and primary murine hepatocytes exposed to FFAs (1 mmol/L) were treated with or without AS-IV (50–200 μg/mL) for 24 h. (A) Representative immunoblots for Thr172p-AMPKα, AMPKα, Ser79p-ACC, ACC, Ser372p-SREBP-1c and mature SREBP-1 (∼68 kDa) in HepG2 cells. β-Actin and LaminB1 were used as internal controls for total protein extracts and nuclear extracts, respectively. (B) Densitometric analyses of the band intensity ratios for Thr172p-AMPKα/AMPKα, Ser79p-ACC/ACC, Ser372p-SREBP-1c/β-actin, SREBP-1/β-actin and SREBP-1/LaminB1 in HepG2 cells. (C) Representative immunoblots for Thr172p-AMPKα, AMPKα, Ser79p-ACC, ACC, Ser372p-SREBP-1c and mature SREBP-1 in primary murine hepatocytes. β-Actin and LaminB1 were used as internal controls for total protein extracts and nuclear extracts, respectively. (D) Densitometric analyses of the band intensity ratios for Thr172p-AMPKα/AMPKα, Ser79p-ACC/ACC, Ser372p-SREBP-1c/β-actin, SREBP-1/β-actin and SREBP-1/LaminB1 in primary murine hepatocytes. (E) Relative mRNA expression levels of lipogenic genes including acc1, fas and scd1 in HepG2 cells. Data are presented as the mean±SEM from three independent experiments (n=3). *P<0.05, **P<0.01 compared with the control group. #P<0.05, ##P<0.01 compared with the FFA group. FFAs, free fatty acids; AS-IV, astragaloside IV.
AS-IV alleviates FFA-induced ER stress in hepatocytes
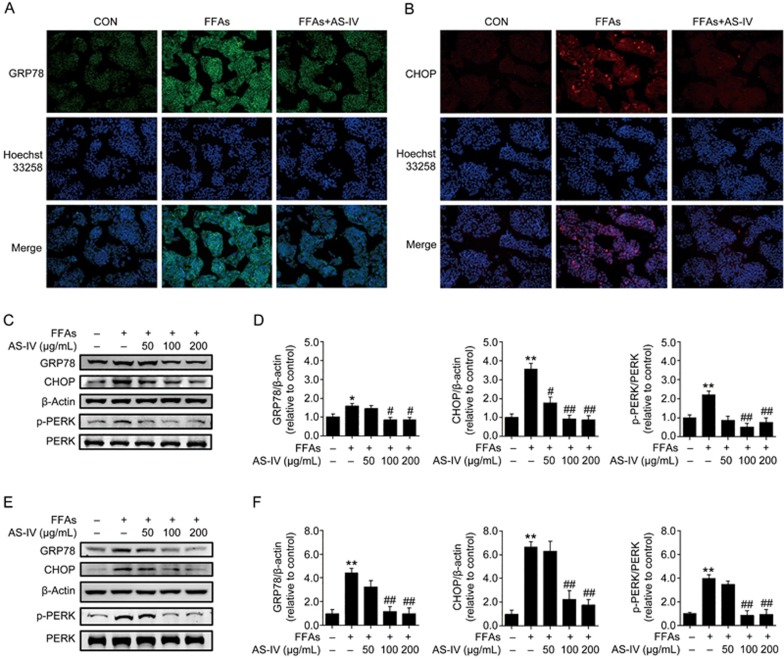
We investigated whether the beneficial effect of AS-IV on FFA-induced lipid accumulation was associated with the alleviation of ER stress by detecting the expression of several key ER stress markers including GRP78, CHOP and p-PERK. Immunofluorescence analyses revealed that GRP78 was primarily located in the cytoplasm of HepG2 cells, and FFA exposure significantly increased its expression (Figure 5A). In contrast, CHOP exhibited very low expression in the cytoplasm and nucleus of HepG2 cells in the control group. FFA exposure obviously increased its expression and nuclear translocation (Figure 5B). AS-IV treatment significantly reversed both of these effects (Figure 5A, 5B). Western blotting analysis demonstrated that FFA exposure substantially increased the protein levels of GRP78, CHOP and p-PERK in HepG2 cells, and AS-IV treatment dose-dependently reduced these increases (Figure 5C, 5D). Similar results were observed in primary murine hepatocytes (Figure 5E, 5F). Collectively, these findings reveal that AS-IV alleviates FFA-induced ER stress in hepatocytes.
Figure 5.
AS-IV alleviates FFA-induced ER stress in hepatocytes. HepG2 cells and primary murine hepatocytes exposed to FFAs (1 mmol/L) were treated with or without AS-IV (50–200 μg/mL) for 24 h. (A) Representative images of immunofluorescence staining for the ER stress marker GRP78 at 200× magnification in HepG2 cells. (B) Representative images of immunofluorescence staining for the ER stress marker CHOP at 200× magnification in HepG2 cells. (C) Representative immunoblots for GRP78, CHOP, Thr981p-PERK and PERK in HepG2 cells. β-Actin was used as an internal control. (D) Densitometric analyses of the band intensity ratios for GRP78/β-actin, CHOP/β-actin and Thr981p-PERK/PERK in HepG2 cells. (E) Representative immunoblots for GRP78, CHOP, Thr981p-PERK and PERK in primary murine hepatocytes. β-Actin was used as an internal control. (F) Densitometric analyses of the band intensity ratios for GRP78/β-actin, CHOP/β-actin and Thr981p-PERK/PERK in primary murine hepatocytes. Data are presented as the mean±SEM of three independent experiments (n=3). *P<0.05, **P<0.01 compared with the control group; #P<0.05, ##P<0.01 compared with the FFA group. FFAs, free fatty acids; AS-IV, astragaloside IV.
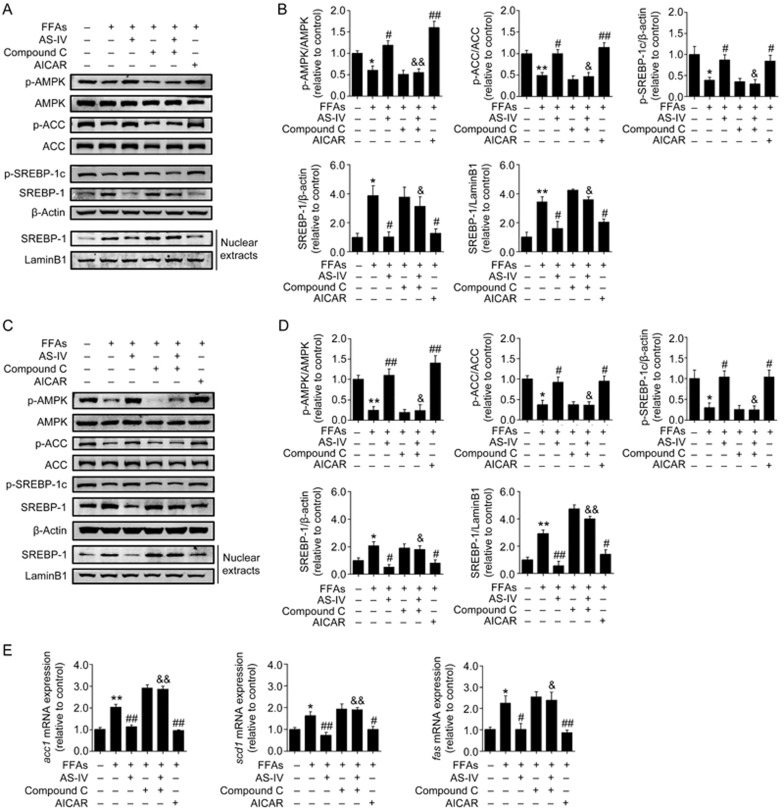
AMPK activation mediates AS-IV-inhibited lipogenesis in FFA-exposed hepatocytes
HepG2 cells and primary murine hepatocytes were pre-treated with the AMPK inhibitor compound C for 1 h prior to FFA and AS-IV treatment to evaluate whether AMPK activation mediates the inhibitory effect of AS-IV on lipogenesis in FFA-exposed hepatocytes. Cells were treated with the AMPK activator AICAR for 2 h as a positive group. AS-IV treatment significantly enhanced the phosphorylation of AMPK, ACC and SREBP-1c in parallel with the decreased accumulation and nuclear translocation of mature SREBP-1 in FFA-exposed hepatocytes. A similar effect was observed in cells treated with AICAR (Figure 6A–6D). Compound C totally or partially blocked these effects of AS-IV treatment (Figure 6A–6D). Similarly, AS-IV and AICAR treatments significantly reduced the mRNA levels of acc1, fas and scd1 in FFA-exposed HepG2 cells, and pretreatment with compound C greatly abolished the inhibitory effect of AS-IV on these changes in gene expression (Figure 6E). These data indicate that AMPK activation mediates AS-IV-inhibited lipogenesis in FFA-exposed hepatocytes.
Figure 6.
AMPK activation mediates AS-IV-inhibited lipogenesis in FFA-exposed hepatocytes. HepG2 cells and primary murine hepatocytes exposed to FFAs (1 mmol/L) were treated with or without AS-IV (100 μg/mL) for 24 h. Compound C (20 μmol/L) was added 1 h prior to the co-treatment with AS-IV and FFAs. Cells were treated with AICAR (1 mmol/L) for 2 h as a positive control. (A) Representative immunoblots for Thr172p-AMPKα, AMPKα, Ser79p-ACC, ACC, Ser372p-SREBP-1c and mature SREBP-1 (∼68 kDa) in HepG2 cells. β-Actin and LaminB1 were used as internal controls for total protein extracts and nuclear extracts, respectively. (B) Densitometric analyses of the band intensity ratios for Thr172p-AMPKα/AMPKα, Ser79p-ACC/ACC, Ser372p-SREBP-1c/β-actin, SREBP-1/β-actin and SREBP-1/LaminB1 in HepG2 cells. (C) Representative immunoblots for Thr172p-AMPKα, AMPKα, Ser79p-ACC, ACC, Ser372p-SREBP-1c and mature SREBP-1 in primary murine hepatocytes. β-Actin and LaminB1 were used as internal controls for total protein extracts and nuclear extracts, respectively. (D) Densitometric analyses of the band intensity ratios for Thr172p-AMPKα/AMPKα, Ser79p-ACC/ACC, Ser372p-SREBP-1c/β-actin, SREBP-1/β-actin and SREBP-1/LaminB1 in primary murine hepatocytes. (E) Relative mRNA expression levels of lipogenic genes including acc1, fas and scd1 in HepG2 cells. Data are presented as the mean±SEM from three independent experiments (n=3). *P<0.05, **P<0.01 compared with the control group. #P<0.05, ##P<0.01 compared with the FFA group. &P<0.05, &&P<0.01 compared with the AS-IV group. FFAs, free fatty acids; AS-IV, astragaloside IV.
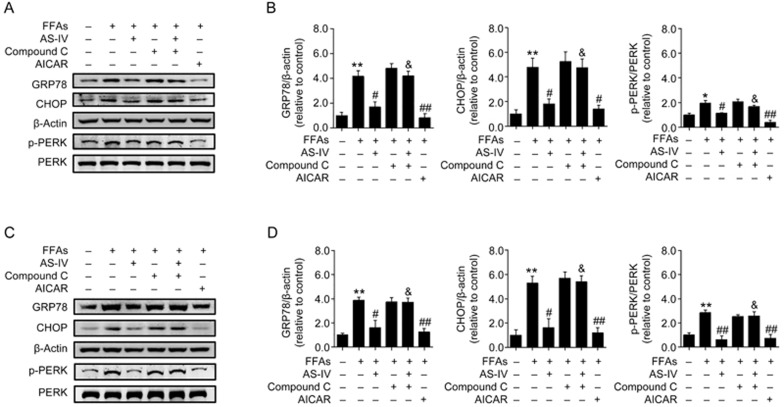
AMPK activation is essential for AS-IV alleviation of FFA-induced ER stress in hepatocytes
The functional relationship between AMPK activation and AS-IV-alleviated ER stress was further investigated in FFA-exposed hepatocytes in our study. AS-IV treatment significantly decreased the protein levels of ER stress markers including GRP78, CHOP and p-PERK in FFA-exposed HepG2 cells, and pretreatment with compound C largely diminished these effects (Figure 7A, 7B). These results were further confirmed in primary murine hepatocytes (Figure 7C, 7D). Altogether, these findings illustrate that AMPK activation is essential for AS-IV alleviation of FFA-induced ER stress in hepatocytes.
Figure 7.
AMPK activation is essential for AS-IV alleviation of FFA-induced ER stress in hepatocytes. HepG2 cells and primary murine hepatocytes exposed to FFAs (1 mmol/L) were treated with or without AS-IV (100 μg/mL) for 24 h. Compound C (20 μmol/L) was added 1 h prior to the co-treatment with AS-IV and FFAs. Cells were treated with AICAR (1 mmol/L) for 2 h as a positive control. (A) Representative immunoblots for GRP78, CHOP, Thr981p-PERK and PERK in HepG2 cells. β-Actin was used as an internal control. (B) Densitometric analyses of the band intensity ratios for GRP78/β-actin, CHOP/β-actin and Thr981p-PERK/PERK in HepG2 cells. (C) Representative immunoblots for GRP78, CHOP, Thr981p-PERK and PERK in primary murine hepatocytes. β-Actin was used as an internal control. (D) Densitometric analyses of the band intensity ratios for GRP78/β-actin, CHOP/β-actin and Thr981p-PERK/PERK in primary murine hepatocytes. Data are presented as the mean±SEM from three independent experiments (n=3). *P<0.05, **P<0.01 compared with the control group. #P<0.05, ##P<0.01 compared with the FFA group. &P<0.05, &&P<0.01 compared with the AS-IV group. FFAs, free fatty acids; AS-IV, astragaloside IV.
Discussion
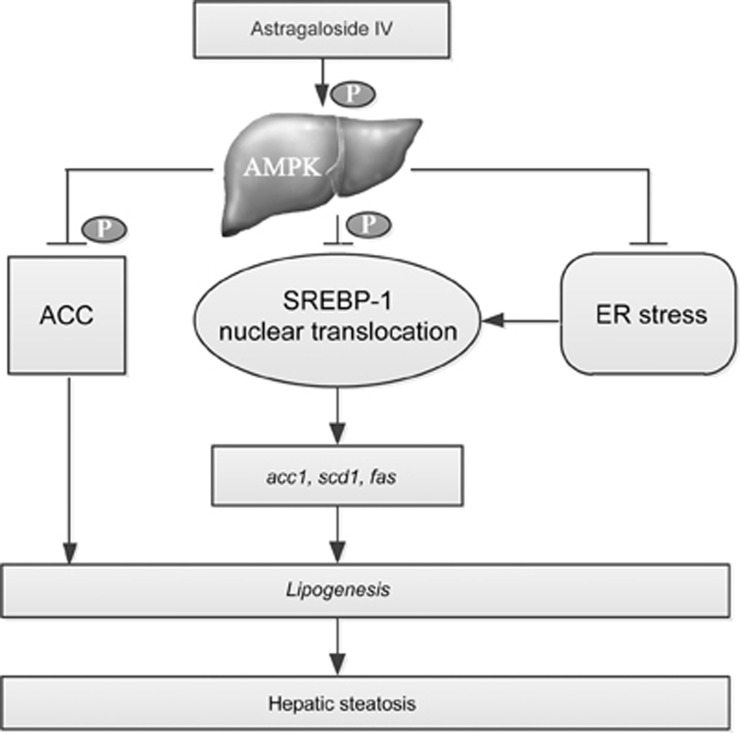
The current study investigated the direct effect of the primary bioactive component of Astragali Radix, AS-IV, on lipid accumulation in hepatocytes and elucidated the underlying mechanisms. We found that AS-IV attenuated FFA-induced lipid accumulation in hepatocytes, possibly through AMPK activation and the subsequent inhibition of ER stress and SREBP-1-mediated lipogenesis (Figure 8). These findings provide molecular evidence to propose AS-IV as promising therapeutics for the treatment of hepatic steatosis.
Figure 8.
Proposed molecular mechanism underlying the lipid-lowering effect of AS-IV on hepatocytes. AS-IV inhibits ER stress and SREBP-1-mediated lipogenesis via AMPK activation to attenuate lipid accumulation in hepatocytes. AS-IV, astragaloside IV.
Hepatic steatosis is the earliest and benign stage of NAFLD. Hepatic steatosis is characterized by the accumulation of triglycerides in the liver. Approximately 60% of the hepatic triglyceride content in NAFLD patients is derived from the FFA influx induced by the lipolysis of adipose tissue, 26% is derived from a sustained increase in de novo lipogenesis in hepatocytes, and 15% is derived from the diet27. In contrast, only <5% of the hepatic triglyceride content is attributed to de novo lipogenesis in healthy persons28. The increased FFA influx and de novo lipogenesis in the liver are two crucial pathogenic processes that contribute to hepatic steatosis. The increased FFA influx delivery into hepatocytes facilitates de novo lipogenesis via an up-regulation of SREBP-1c2. SREBP-1c is the major transcription factor for de novo lipogenesis in the liver, and it translocates into the nucleus to transcriptionally upregulate lipogenic genes including acc1, fas and scd1 upon maturation30. Increased levels of liver SREBP-1c are observed in patients and mice with hepatic steatosis31,32. AMPK activation negatively regulates SREBP-1c expression, cleavage and nuclear translocation via phosphorylation11,12. Therefore, the AMPK/SREBP-1c pathway may be involved in the alleviation of hepatic lipid accumulation. Our study found that AS-IV enhanced the phosphorylation of SREBP-1c, inhibited the accumulation and nuclear translocation of mature SREBP-1 and decreased the mRNA levels of acc1, fas, and scd1 via AMPK activation in FFA-exposed hepatocytes. These results suggest that AMPK activation and the subsequent inhibition of SREBP-1-mediated lipogenesis are a potential mechanism for the beneficial effect of AS-IV on hepatic lipid accumulation. This study is the first report to provide in vitro evidence for the direct lipid-lowing effect of AS-IV on hepatocytes via a mechanism that includes AMPK activation. In contrast, a recent study demonstrated that AS-IV improved hepatic steatosis in obese mice primarily via enhanced fat oxidation rather than decreased lipogenesis22. This discrepancy may be due to the different experimental system used. Nevertheless, these studies indicated that AS-IV ameliorated hepatic steatosis in vivo and in vitro. Many other bioactive compounds purified from Chinese medical herbs also attenuate lipid accumulation via the AMPK/SREBP-1c pathway in hepatocytes20,33,34. Two anti-diabetic drugs, rosiglitazone and metformin, also ameliorate hepatic lipid accumulation via AMPK activation and the subsequent decrease in SREBP-1c activity and expression35,36.
The increased FFA influx delivery into liver, especially saturated FFAs, induces an ER stress response in hepatocytes in the context of NAFLD37,38. Hepatic ER stress may induce SREBP-1c activation and contribute to lipogenesis and hepatic steatosis39. Our results demonstrated that FFA exposure in hepatocytes obviously induced ER stress, which was evidenced by the increased expression of key markers including GRP78, CHOP and p-PERK, in parallel with SREBP-1 activation and hepatic lipid accumulation. AS-IV inhibited SREBP-1 activation in FFA-exposed hepatocytes in our study, and we wondered whether AS-IV treatment also attenuated FFA-induced hepatic ER stress. We observed that AS-IV significantly attenuated ER stress in FFA-exposed hepatocytes, which was indicated by the decreased expression of GRP78, CHOP and p-PERK. Previous studies also demonstrated that AS-IV alleviated ER stress in diabetic nephropathy in vivo and in vitro40,41. Our study revealed that the AS-IV treatment alleviation of FFA-induced ER stress in hepatocytes occurred via an AMPK-dependent manner, which is consistent with previous findings indicating a negative regulation of AMPK on lipid-induced ER stress in hepatocytes13,14,15. Taken together, our results demonstrated that AS-IV-alleviated ER stress via AMPK activation may also contribute to the inhibition of SREBP-1-mediated lipogenesis in FFA-induced hepatocytes. This pathway may be another possible mechanism for the beneficial effect of AS-IV on hepatic lipid accumulation.
However, this study did not clearly define the detailed mechanisms of how AS-IV promoted AMPK activation. AMPK is activated by an increased cellular AMP:ATP ratio and upstream signaling pathways, including LKB1 and CaMKK, via phosphorylation42,43. Our findings are also restricted to in vitro situations and should be confirmed in an animal study. Therefore, further studies should be performed in vivo and in vitro to elucidate the beneficial effect of AS-IV on hepatic steatosis.
In summary, our study demonstrated for the first time that AS-IV attenuated FFA-induced ER stress and lipid accumulation via AMPK activation in hepatocytes, which further supports its use as promising therapeutics for the treatment of hepatic steatosis.
Author contribution
Liao SUN and Jie XU conceived and designed the experiments; Bing ZHOU and Dan-li ZHOU performed the experiments; Bing ZHOU, Xiao-hong WEI, and Rong-yu ZHONG analyzed the data; Bing ZHOU wrote the manuscript.
Acknowledgments
This study was supported by a grant from the Natural Science Foundation of Guangdong Province, China (2016A030313286).
References
- Fan JG, Farrell GC. Epidemiology of non-alcoholic fatty liver disease in China. J Hepatol 2009; 50: 204–10. [DOI] [PubMed] [Google Scholar]
- Cohen JC, Horton JD, Hobbs HH. Human fatty liver disease: old questions and new insights. Science 2011; 332: 1519–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratziu V, Bellentani S, Cortez-Pinto H, Day C, Marchesini G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J Hepatol 2010; 53: 372–84. [DOI] [PubMed] [Google Scholar]
- Williams CD, Stengel J, Asike MI, Torres DM, Shaw J, Contreras M, et al. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: a prospective study. Gastroenterology 2011; 140: 124–31. [DOI] [PubMed] [Google Scholar]
- Malhi H, Gores GJ. Molecular mechanisms of lipotoxicity in nonalcoholic fatty liver disease. Semin Liver Dis 2008; 28: 360–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres DM, Williams CD, Harrison SA. Features, diagnosis, and treatment of nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 2012; 10: 837–58. [DOI] [PubMed] [Google Scholar]
- Pagliassotti MJ. Endoplasmic reticulum stress in nonalcoholic fatty liver disease. Annu Rev Nutr 2012; 32: 17–33. [DOI] [PubMed] [Google Scholar]
- Hardie DG. AMPK — sensing energy while talking to other signaling pathways. Cell Metab 2014; 20: 939–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schimmack G, Defronzo RA, Musi N. AMP-activated protein kinase: Role in metabolism and therapeutic implications. Diabetes Obes Metab 2006; 8: 591–602. [DOI] [PubMed] [Google Scholar]
- Hardie DG. AMPK: a key regulator of energy balance in the single cell and the whole organism. Int J Obes (Lond) 2008; 32: S7–12. [DOI] [PubMed] [Google Scholar]
- Ahmed MH, Byrne CD. Modulation of sterol regulatory element binding proteins (SREBPs) as potential treatments for non-alcoholic fatty liver disease (NAFLD). Drug Discov Today 2007; 12: 740–7. [DOI] [PubMed] [Google Scholar]
- Li Y, Xu S, Mihaylova MM, Zheng B, Hou X, Jiang B, et al. AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab 2011; 13: 376–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J, Hong SW, Park SE, Rhee EJ, Park CY, Oh KW, et al. Exendin-4 inhibits the expression of SEPP1 and Fetuin-A via improvement of palmitic acid-induced endoplasmic reticulum stress by AMPK. Endocrinol Metab (Seoul) 2015; 30: 177–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Min Q, Ou-yang C, Lee J, He C, Zou MH, et al. AMPK activation prevents excess nutrient-induced hepatic lipid accumulation by inhibiting mTORC1 signaling and endoplasmic reticulum stress response. Biochim Biophys Acta 2014; 1842: 1844–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Wu Z, Li D, Wang D, Wang X, Feng X, et al. Involvement of oxygen-regulated protein 150 in AMP-activated protein kinase-mediated alleviation of lipid-induced endoplasmic reticulum stress. J Biol Chem 2011; 286: 11119–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flamment M, Kammoun HL, Hainault I, Ferre P, Foufelle F. Endoplasmic reticulum stress: a new actor in the development of hepatic steatosis. Curr Opin Lipidol 2010; 21: 239–46. [DOI] [PubMed] [Google Scholar]
- Basseri S, Austin RC. ER stress and lipogenesis: a slippery slope toward hepatic steatosis. Dev Cell 2008; 15: 795–6. [DOI] [PubMed] [Google Scholar]
- Lin CL, Huang HC, Lin JK. Theaflavins attenuate hepatic lipid accumulation through activating AMPK in human HepG2 cells. J Lipid Res 2007; 48: 2334–43. [DOI] [PubMed] [Google Scholar]
- Zang M, Xu S, Maitland-Toolan KA, Zuccollo A, Hou X, Jiang B, et al. Polyphenols stimulate AMP-activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic LDL receptor-deficient mice. Diabetes 2006; 55: 2180–91. [DOI] [PubMed] [Google Scholar]
- Seo MS, Kim JH, Kim HJ, Chang KC, Park SW. Honokiol activates the LKB1-AMPK signaling pathway and attenuates the lipid accumulation in hepatocytes. Toxicol Appl Pharmacol 2015; 284: 113–24. [DOI] [PubMed] [Google Scholar]
- Juan YC, Kuo YH, Chang CC, Zhang LJ, Lin YY, Hsu CY, et al. Administration of a decoction of sucrose- and polysaccharide-rich radix astragali (Huang Qi) ameliorated insulin resistance and fatty liver but affected beta-cell function in type 2 diabetic rats. Evid Based Complement Alternat Med 2011; 2011: 349807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H, Gao Y, Shi HL, Qin LY, Huang F, Lan YY, et al. Astragaloside IV improves lipid metabolism in obese mice by alleviation of leptin resistance and regulation of thermogenic network. Sci Rep 2016; 6: 30190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Li Q, Zhao W, Li J, Sun Y, Liu K, et al. Astragaloside IV and cycloastragenol are equally effective in inhibition of endoplasmic reticulum stress-associated TXNIP/NLRP3 inflammasome activation in the endothelium. J Ethnopharmacol 2015; 169: 210–8. [DOI] [PubMed] [Google Scholar]
- Qi H, Wei L, Han Y, Zhang Q, Lau AS, Rong J. Proteomic characterization of the cellular response to chemopreventive triterpenoid astragaloside IV in human hepatocellular carcinoma cell line HepG2. Int J Oncol 2010; 36: 725–35. [DOI] [PubMed] [Google Scholar]
- Li WC, Ralphs KL, Tosh D. Isolation and culture of adult mouse hepatocytes. Methods Mol Biol 2010; 633: 185–96. [DOI] [PubMed] [Google Scholar]
- Gomez-Lechon MJ, Donato MT, Martinez-Romero A, Jimenez N, Castell JV, O'Connor JE. A human hepatocellular in vitro model to investigate steatosis. Chem Biol Interact 2007; 165: 106–16. [DOI] [PubMed] [Google Scholar]
- Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest 2005; 115: 1343–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudgins LC, Hellerstein MK, Seidman CE, Neese RA, Tremaroli JD, Hirsch J. Relationship between carbohydrate-induced hypertriglyceridemia and fatty acid synthesis in lean and obese subjects. J Lipid Res 2000; 41: 595–604. [PubMed] [Google Scholar]
- Mendez-Sanchez N, Arrese M, Zamora-Valdes D, Uribe M. Current concepts in the pathogenesis of nonalcoholic fatty liver disease. Liver Int 2007; 27: 423–33. [DOI] [PubMed] [Google Scholar]
- Shimano H, Yahagi N, Amemiya-Kudo M, Hasty AH, Osuga J, Tamura Y, et al. Sterol regulatory element-binding protein-1 as a key transcription factor for nutritional induction of lipogenic enzyme genes. J Biol Chem 1999; 274: 35832–9. [DOI] [PubMed] [Google Scholar]
- Pettinelli P, Del Pozo T, Araya J, Rodrigo R, Araya AV, Smok G, et al. Enhancement in liver SREBP-1c/PPAR-alpha ratio and steatosis in obese patients: correlations with insulin resistance and n-3 long-chain polyunsaturated fatty acid depletion. Biochim Biophys Acta 2009; 1792: 1080–6. [DOI] [PubMed] [Google Scholar]
- Shimomura I, Bashmakov Y, Horton JD. Increased levels of nuclear SREBP-1c associated with fatty livers in two mouse models of diabetes mellitus. J Biol Chem 1999; 274: 30028–32. [DOI] [PubMed] [Google Scholar]
- Quan HY, Kim do Y, Kim SJ, Jo HK, Kim GW, Chung SH. Betulinic acid alleviates non-alcoholic fatty liver by inhibiting SREBP1 activity via the AMPK-mTOR-SREBP signaling pathway. Biochem Pharmacol 2013; 85: 1330–40. [DOI] [PubMed] [Google Scholar]
- Kamikubo R, Kai K, Tsuji-Naito K, Akagawa M. Beta-Caryophyllene attenuates palmitate-induced lipid accumulation through AMPK signaling by activating CB2 receptor in human HepG2 hepatocytes. Mol Nutr Food Res 2016; 60: 2228–42. [DOI] [PubMed] [Google Scholar]
- Fryer LG, Parbu-Patel A, Carling D. The anti-diabetic drugs rosiglitazone and metformin stimulate AMP-activated protein kinase through distinct signaling pathways. J Biol Chem 2002; 277: 25226–32. [DOI] [PubMed] [Google Scholar]
- Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 2001; 108: 1167–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gentile CL, Frye MA, Pagliassotti MJ. Fatty acids and the endoplasmic reticulum in nonalcoholic fatty liver disease. Biofactors 2011; 37: 8–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leamy AK, Egnatchik RA, Young JD. Molecular mechanisms and the role of saturated fatty acids in the progression of non-alcoholic fatty liver disease. Prog Lipid Res 2013; 52: 165–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kammoun HL, Chabanon H, Hainault I, Luquet S, Magnan C, Koike T, et al. GRP78 expression inhibits insulin and ER stress-induced SREBP-1c activation and reduces hepatic steatosis in mice. J Clin Invest 2009; 119: 1201–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang ZS, Xiong F, Xie XH, Chen D, Pan JH, Cheng L. Astragaloside IV attenuates proteinuria in streptozotocin-induced diabetic nephropathy via the inhibition of endoplasmic reticulum stress. BMC Nephrol 2015; 16: 44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Gui D, Chen J, He D, Luo Y, Wang N. Down-regulation of PERK-ATF4-CHOP pathway by Astragaloside IV is associated with the inhibition of endoplasmic reticulum stress-induced podocyte apoptosis in diabetic rats. Cell Physiol Biochem 2014; 33: 1975–87. [DOI] [PubMed] [Google Scholar]
- Woods A, Johnstone SR, Dickerson K, Leiper FC, Fryer LG, Neumann D, et al. LKB1 is the upstream kinase in the AMP-activated protein kinase cascade. Curr Biol 2003; 13: 2004–8. [DOI] [PubMed] [Google Scholar]
- Birnbaum MJ. Activating AMP-activated protein kinase without AMP. Mol Cell 2005; 19: 289–90. [DOI] [PubMed] [Google Scholar]