Abstract
Self-regulation is a fundamental human process that refers to multiple complex methods by which individuals pursue goals in the face of distractions. Whereas superior self-regulation predicts better academic achievement, relationship quality, financial and career success, and lifespan health, poor self-regulation increases a person’s risk for negative outcomes in each of these domains and can ultimately presage early mortality. Given its centrality to understanding the human condition, a large body of research has examined cognitive, emotional, and behavioral aspects of self-regulation. In contrast, relatively little attention has been paid to specific biologic processes that may underlie self-regulation. We address this latter issue in the present review by examining the growing body of research showing that components of the immune system involved in inflammation can alter neural, cognitive, and motivational processes that lead to impaired self-regulation and poor health. Based on these findings, we propose an integrated, multi-level model that describes how inflammation may cause widespread biobehavioral alterations that promote self-regulatory failure. This immunologic model of self-regulatory failure has implications for understanding how biological and behavioral factors interact to influence self-regulation. The model also suggests new ways of reducing disease risk and enhancing human potential by targeting inflammatory processes that affect self-regulation.
Keywords: cytokines, self-regulation, cognition, motivation, executive function, development, behavior, disease, health
The social and physical worlds that individuals inhabit are filled with temptations and demands that distract people from goals and challenge their best intentions. A fundamental aspect of successfully navigating such an environment thus involves managing impulses that arise as people attempt to engage in short-term behaviors (e.g., skipping dessert) that are consistent with longer-term goals (e.g., losing weight). The method by which humans work to maintain such alignment is generally referred to as self-regulation (Muraven & Baumeister, 2000), and its relevance for shaping peoples’ lives is difficult to overstate. Indeed, whereas closely controlling one’s thoughts and actions can help an individual maintain long-lasting relationships, foster incredible talents, and accomplish remarkable feats, failing to self-regulate can lead to behaviors that have the potential to cause tremendous personal and collective harm.
Self-regulation is an important psychological construct in part because it is relevant for many different outcomes (See Table 1; Baumeister & Vohs, 2005). For example, self-regulation plays a key role in shaping lifelong health by influencing the extent to which people engage in many different actions that can have serious consequences, such as adhering to a healthy diet, scheduling annual visits to the doctor, taking prescribed medications, exercising regularly, and practicing good health behaviors including eating, sleeping, and brushing one’s teeth on a consistent schedule (Bandura, 2005; Schwarzer, 1999). Self-regulatory abilities also greatly affect peoples’ interpersonal and work life by shaping their friendship and romantic relationship quality, academic achievement, and overall career success (Diamond, 2013). Finally, although psychosocial stressors are a part of many peoples’ lives and can increase risk for life-threatening diseases, self-regulatory abilities have been found to buffer individuals against negative health outcomes that are frequently caused by stress (Evans & Fuller-Rowell, 2013).
Table 1.
Multifaceted Benefits of Better Self-Regulation by Life Domain
| Benefits | Example Reference(s) |
|---|---|
| Education | |
| Higher grade point average | Tangney, Baumeister, & Boon, 2004 |
| Decreased likelihood of failing in school | Blair & Diamond, 2008 |
| Career | |
| Better job performance | Porath & Bateman, 2006; Stajkovic & Alexander, 1998 |
| Greater career success (e.g., salary, job satisfaction) | Ng, Eby, Sorensen, & Feldman, 2005 |
| Social Relationships | |
| Better social skills and status | Eisenberg et al., 1993 |
| Better social relationship quality | Tangney, Baumeister, & Boon, 2004 |
| Quality and Length of Life | |
| More positive affect, well-being, and life satisfaction | Hofmann, Luhmann, Fisher, Vohs, & Baumeister, 2014 |
| Reduced occurrence of psychopathology | Tangney, Baumeister, & Boon, 2004 |
| Better overall health | Atherton, Robins, Rentfrow, & Lamb, 2014; Bandura, 2005 |
| Longer lifespan | Friedman et al., 1993 |
In contrast, failures of self-regulation are known to contribute to a variety of personal and societal maladies (Baumeister, Heatherton, & Tice, 1994). For example, failing to self-regulate can result in succumbing to negative social influences (Burkley, Anderson, & Curtis, 2011), debt-procuring impulsive spending (Baumeister, 2002), poor self-presentation (Vohs, Baumeister, & Ciarocco, 2005), relapsing in smoking cigarettes (Muraven, 2010), and committing crimes (Evans, Cullen, Burton, Dunaway, & Benson, 1997; LaGrange & Silverman, 1999). As a result of these effects, poor self-regulation is a very strong, independent predictor of both disease-specific and overall mortality (Friedman et al., 1993; Kröz et al., 2011).
Given the critical importance of self-regulatory abilities for human behavior and health, a large number of studies have been conducted over the past four decades to elucidate social, environmental, and cognitive processes that contribute to self-regulatory failure and the consequences that such failures have for individuals over the life course. Moreover, this body of work has been evaluated in several excellent reviews (e.g., Heatherton & Wagner, 2011; Muraven, Tice, & Baumeister, 1998; Vohs & Heatherton, 2000; Vohs & Baumeister, 2011; Wagner & Heatherton, 2015). At the same time, comparatively little attention has been paid to the roles that specific biologic processes might play in self-regulation, even though elucidating such mechanisms could help refine thinking about self-regulation and possibly lead to the identification of new strategies for enhancing self-regulation and improving the human condition (Klein, Shepperd, Suls, Rothman, & Croyle, 2015; Moffitt et al., 2011; c.f., Heatherton & Wagner, 2011). Discussion of the biological bases of self-regulation has been limited in part because researchers have lacked the types of methodological tools that are necessary to study the full range of biologic processes that might underlie self-regulation. Recent advances in neuroimaging and immunologic techniques have since been developed, though, and one of the most interesting and potentially important discoveries in this area of research has involved the finding that specific neural and immune system processes may interact to directly influence self-regulatory ability.
The purpose of the present review is to examine neural and immune system processes that are relevant for self-regulation, behavior, and health. In doing so, we hope to generate new ideas for future research that use concepts and methods from psychology, neuroscience, and immunology to elucidate the psychological and biological bases of self-regulation. To achieve this goal, we first summarize contemporary thinking and research on self-regulation. Second, we provide an overview of immune system processes that are involved in human behavior and health. Third, we describe pathways by which the immune system can affect neural structure and function. Fourth, we examine how aberrant neural and immune system dynamics can impact processes that are required for effective self-regulation. Fifth, based on this information, we propose an integrative, multi-level model of self-regulation, behavior, and health. Finally, we discuss the possible implications of this work and suggest some possible avenues for future research. Because evidence linking immune system dynamics and self-regulation is more well-developed in some contexts than others, our overarching goal is to provide a comprehensive assessment of what is presently known and a framework for future research on these important issues.
Self-Regulation
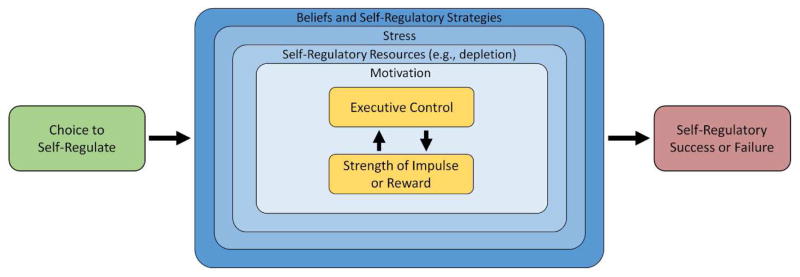
We define self-regulation as the ability through which people create and maintain alignment with distant or abstract goals, especially when immediate motivations and desires compete with those goals. Self-regulation is not a singular process, but rather a set of multiple interrelated processes that enable individuals to control their thoughts, behavior, and future in concrete and abstract ways by managing goals, strategies, plans, intentions, and impulses (Coutlee & Huettel, 2012). Numerous proximal factors have been proposed to influence self-regulation, such as stress, motivation, cognitive resources, and self-regulatory strategies (Hagger, Wood, Stiff, & Chatzisarantis, 2010; Vohs & Baumeister, 2011). Several distal factors have also been proposed, including brain structure, trait-like tendencies to appraise situations as more positive or negative, and individual differences in cognitive abilities (Heatherton & Wagner, 2011). Despite the complex ways in which these many factors can interact to ultimately influence self-regulation, though, a substantial body of research points to the fact that individual acts of self-regulation—resulting in either self-regulatory success or failure—depend to a great extent on the balance between executive control and the strength of an impulse, emotion, or reward. A graphical representation of these dynamics is depicted in Figure 1.
Figure 1.
Processes involved in self-regulation, arranged by their proximity to individual self-regulatory acts. Acts of self-regulation depend most proximally on the strength of an impulse, emotion, or reward, which both influence and are influenced by executive control abilities. This interplay is influenced by a person’s motivation to self-regulate, as motivation to self-regulate is crucial if sustained executive control is needed. Motivation is then in turn influenced by self-regulatory resources, as motivation to self-regulate will be low if one knows self-regulation is ultimately a futile effort. Self-regulatory resources are in turn affected by stress, as stress reduces self-regulatory resources. Finally, and most distally, beliefs and self-regulatory strategies can have an influential impact on self-regulation, due in part to how these factors may alter perceptions of both stress and self-regulatory resources.
Many factors also interact to ultimately influence human behavior and health (Miller, Chen, & Cole, 2009; Toussaint, Shields, Dorn, & Slavich, 2016). In this context, self-regulation has been regarded as an important and sometimes critical factor to the extent that it contributes to cognitive, emotional, and behavioral outcomes that shape individuals’ lives and well-being (Scheier, Carver, & Armstrong, 2012). Self-regulation may influence how individuals in a society live and interact through cognitive means by, for example, enabling them to consider the consequences of being conflictual or verbally combative, or through behavioral means by promoting pro-social actions. Self-regulation can also influence individuals’ lives through emotional means by facilitating effective coping strategies for dealing with negative affect or preventing aggressive or depressive behavior.
Given these far-reaching effects, a large body of research has investigated methods for enhancing self-regulation as a means of improving self regulation-related outcomes. Studies on this topic have examined a variety of different intervention strategies, given self-regulations’ multifaceted nature, and this work has suggested that some strategies may improve self-regulation despite the fact that self-regulatory abilities are largely stable over time (e.g., Berman et al., 2013; Casey et al., 2011; Meichenbaum & Goodman, 1971; Sultan, Joireman, & Sprott, 2011; Verbeken, Braet, Goossens, & van der Oord, 2013). One notable limitation of this work, however, is that it has identified methods for enhancing self-regulation abilities that are broad and nonspecific, and that vary greatly with respect to their efficacy (Berkman, Graham, & Fisher, 2012). Indeed, there is debate as to whether the self-regulation training methods developed to date are effective (e.g., Simons et al., 2016). Understanding the biological mechanisms that underlie self-regulation may address some of these issues and help inform the development of more effective interventions for promoting self-regulation, but research on this topic has been limited, especially compared to the amount of work that has focused on identifying cognitive, emotional, and behavioral aspects of self-regulation.
Biologically Informed Theories of Self-Regulation
With respect to the few biological formulations of self-regulation that have been proposed, two models stand out that provide an initial picture of how specific biological mechanisms may lead to self-regulatory failure. The first model argues that self-regulation depends in part on glucose, and that temporary reductions in self-regulatory ability are mediated by temporary depletions of circulating or focal brain glucose (Gailliot & Baumeister, 2007). Although this model has been challenged (i.e., Hagger & Chatzisarantis, 2013; Job, Walton, Bernecker, & Dweck, 2013; Molden et al., 2012), some of its tenants may hold up to criticism (Baumeister, 2014; Chatzisarantis & Hagger, 2015; though see Baumeister & Vohs, 2016; Dang, 2016; Hagger et al., 2016). The second noteworthy, biologically informed model of self-regulation posits that the glucocorticoid hormone cortisol, which is released during stress, alters brain function in ways that may impair self-regulation (Blair et al., 2011; Raio, Orederu, Palazzolo, Shurick, & Phelps, 2013; c.f. Shields, Bonner, & Moons, 2015). These theories provide important initial accounts of biologic mechanisms that may underlie self-regulatory successes and failures. However, for reasons that we outline below, it is unlikely that cortisol and glucose are the only biological factors that influence self-regulation. Moreover, cortisol and glucose may not represent the most critical factors to focus on in this context.
The Immune System, Inflammation, and their Relevance for Self-Regulation
One system that may be intimately involved in self-regulation, but which has received virtually no attention to date, is the human immune system. As we discuss below, immune system activity—especially components of the immune system involved in inflammation—appear to impair numerous facets of self-regulation. In addition, health conditions that are associated with aberrant immune system activity, such as obesity, are also characterized by poor self-regulation. Moreover, like the nervous system (Knudsen, 2004), the immune system exhibits sensitive periods during which time environmental inputs, such as life stress, can shape the reactivity of the immune system for years to come, thus providing a potential explanation for why early life stress is associated with both poor self-regulation and worse health over the lifespan (Bilbo & Schwarz, 2009; see also John-Henderson, Rheinschmidt, Mendoza-Denton, & Francis, 2014). Consequently, multiple reasons exist for examining the role that the immune system plays in self-regulation.
The primary responsibility of the immune system is to rid the body of foreign pathogens and help the body recuperate during physical injury and infection (Daruna, 2012; Murphy, 2014). One key player in immune system activity is a class of proteins called cytokines, which facilitate communication between immune cells. Cytokines come in several different types and serve many functions (Daruna, 2012). Most relevant for the present discussion, though, is a class of cytokines called proinflammatory cytokines, which upregulate inflammatory activity throughout the body (Slavich & Irwin, 2014).
Proinflammatory cytokines are released from immune cells usually after a microbial invader or tissue injury has been detected (Irwin & Slavich, 2017; Medzhitov, 2008). Under these circumstances, cytokines send chemical messages to attract other cells to target locations and can also cause blood vessels to expand to increase trafficking of immune cells to sites of injury or microbial invasion (Barton, 2008). These dynamics in turn cause redness, heat, pain, and swelling at the affected area(s), and can also cause systemic changes including the induction of fever, fatigue, altered sleep and eating, and social-behavioral withdrawal (Curfs, Meis, & Hoogkamp-Korstanje, 1997; Slavich & Irwin, 2014). Collectively, these effects are referred to as inflammation.
Immune system activity in general, and inflammatory activity in particular, are coordinated by thousands of complex physiochemical interactions that have numerous biobehavioral effects. The immune system, therefore, does not only influence self-regulatory behavior (as described above), nor is it the only system that influences self-regulation. As we have already mentioned, however, components of the immune system involved in inflammation can have very powerful effects on cognition, motivation, and behavior, making this system potentially critical for understanding self-regulatory failure.
As we describe in greater detail below, support for the idea that inflammation can degrade self-regulatory ability comes from numerous studies showing that inflammatory activity can impair both cognitive and emotional self-regulation (e.g., Frydecka et al., 2015; Gianaros et al., 2014; Miller, Capuron, & Raison, 2005; Mooijaart et al., 2013; Prossin et al., 2011; Reichenberg et al., 2001; Trompet et al., 2008). Therefore, although there is very little crosstalk between studies on self-regulation and inflammation, extant evidence suggests that self-regulation influences immune system activity and vice versa. Such effects may be surprising, since inflammation is still typically thought of as the body’s primary response to physical injury or infection (Slavich, 2015). However, biological mediators of the inflammatory response can also have neural, cognitive, and motivational effects, making the immune system highly relevant for self-regulatory behavior (Dantzer & Kelley, 2007; Maier & Watkins, 1998). To describe how the immune system can influence self-regulation, we next provide a brief overview of how the brain and immune system interact in ways that are relevant for self-regulation.
Bi-Directional Pathways Linking the Immune System and Brain
Like other bodily systems, the immune system and brain have the ability to bi-directionally communicate, and the pathways that underlie these links enable the brain to steer the activity of the immune system and vice versa (Irwin & Cole, 2011; Nusslock & Miller, 2015; Slavich & Irwin, 2014; Slavich, Way, Eisenberger, & Taylor, 2010). One pathway by which the brain regulates immune system activity involves the hypothalamic-pituitary-adrenal (HPA) axis, which, through the anti-inflammatory actions of cortisol, functions as a key down-regulator (but also up-regulator) of proinflammatory cytokine activity (Busillo & Cidlowski, 2013). Another pathway by which the brain can regulate immune system activity is the sympathetic-adrenal-medullary (SAM) axis, which, once activated, upregulates proinflammatory cytokine production through the release of epinephrine and norepinephrine (Daruna, 2012). In brief, therefore, the brain can modulate the immune system through at least two main pathways: whereas the HPA axis primarily (but not exclusively) reduces inflammation by inhibiting the release of proinflammatory cytokines, SAM axis activity ultimately increases inflammation by promoting the release of proinflammatory cytokines.
Conversely, the immune system can also influence brain activity via several pathways. In brief, proinflammatory cytokines can directly stimulate neurons via cytokine receptors on neurons (Hopkins & Rothwell, 1995; Louveau et al., 2015; Raison, Capuron, & Miller, 2006; Rothwell & Hopkins, 1995). Such effects can occur through direct interaction between the immune system and brain (Louveau et al., 2015) but also through indirect interactions, such as when neural activity is influenced by stimulation of the vagus nerve or by alterations in the synthesis and degradation of various neurotransmitters (Garcia-Oscos et al., 2015; Maier, Goehler, Fleshner, & Watkins, 1998; Thayer & Sternberg, 2010). Ultimately, via these and other non-mutually exclusive pathways, proinflammatory cytokines can exert strong modulatory effects on the structure and function of brain regions that support self-regulation, such as the prefrontal cortex (PFC; Audet, Jacobson-Pick, Wann, & Anisman, 2011; de Pablos et al., 2006; Garcia-Oscos et al., 2015; Poh, Yeo, Stohler, & Ong, 2012).
Neurocognitive and Behavioral Pathways Linking the Immune System and Self-Regulation
So far, we have reviewed evidence indicating that the brain and immune system interact. Next, we examine psychological processes that support self-regulation and how immune system activation may impact these processes. In particular, we summarize research demonstrating how cytokines and other immune system mediators can influence beliefs about self-regulation, self-regulatory depletion, motivation and reward, appraisals of stress, and executive function. Evidence relating immune system activity to each of these self-regulatory processes varies, and some self-regulatory processes, such as executive function, have a stronger body of evidence linking them to immune system activity than others. Nevertheless, since these processes all represent mechanisms that may underpin successful self-regulation, we describe known connections when appropriate and identify the need for future research when warranted.
Beliefs about Self-Regulation
Personal beliefs exert strong effects on self-regulatory abilities. Beliefs about self-control, self-regulatory capacity, and the efficacy of agency, for example, modulate several self-regulatory abilities, including error monitoring in cognitive tasks (Rigoni, Wilquin, Brass, & Burle, 2013), preconscious action initiating processes (Rigoni, Kühn, Sartori, & Brass, 2011), self-control (Rigoni, Kühn, Gaudino, Sartori, & Brass, 2012), and the effects of glucose supplementation on self-regulation (Job et al., 2013). Moreover, individuals’ belief that their self-regulatory abilities are limitless attenuates the detrimental effects of prior self-regulatory exertion on subsequent self-regulatory behavior (Vohs, Baumeister, & Schmeichel, 2013). Indeed, a recent meta-analytic review examining the effects of beliefs about self-regulation found that beliefs about self-regulatory abilities have a small-to-moderate effect on the efficacy of self-regulation (Burnette, O’Boyle, VanEpps, Pollack, & Finkel, 2013). This is notable given that numerous factors influence self-regulation.
Similarly, the use of regulatory strategies contributes to effective self-regulation (Burnette et al., 2013). Self-regulatory strategies are the preplanned methods by which individuals attempt to attain their goals, such as by deciding to walk out of a room if one sees cupcakes on a table at a party. Although these methods are often dependent on the adequate functioning of more immediate methods of self-control, such as response inhibition, they contribute a unique part of the variance in overall self-regulatory success (Burnette et al., 2013) and may even facilitate more immediate methods of self-control if these strategies are indeed predefined (Crockett et al., 2013). Thus, beliefs about self-regulation and strategies both appear to have a prominent impact on the effectiveness of self-regulatory efforts.
A substantial body of research has examined the effects of different beliefs on cytokines and other immune system mediators. Existing studies support the idea that beliefs related to self-efficacy (Mausbach et al., 2011), spirituality (Ai, Pargament, Kronfol, Tice, & Appel, 2010), and religion (Koenig et al., 1997) may reduce basal proinflammatory cytokine activity, as well as cytokine reactivity to stress. Interestingly, positive beliefs about one’s self-efficacy, as well as religious beliefs, have been shown to promote self-regulation (Burnette et al., 2013; McCullough & Willoughby, 2009). Although correlational, these data indicate that certain beliefs the self or God may create psychological states that lead to decreased levels of proinflammatory cytokine activity, which may have implications for self-regulation.
An additional possibility is that proinflammatory cytokines influence neural systems in ways that shape the development of beliefs that impair self-regulation. Put another way, rather than certain beliefs lowering circulating levels of cytokines and thereby enhancing self-regulation, heightened levels of proinflammatory cytokines may contribute to the development of beliefs that undercut successful self-regulatory strategies. Little direct support is presently available for this formulation (though see Papageorgiou et al., 2006). However, a robust finding pertaining to the cognitive and behavioral effects of inflammation is that proinflammatory cytokines can induce depressed mood (e.g., Eisenberger, Inagaki, Mashal, & Irwin, 2010; for a review, see Maier & Watkins, 1998). To the extent that negative beliefs that degrade self-regulatory abilities increase in salience during depressive mood states (Madigan & Bollenbach, 1986), these links may suggest that proinflammatory cytokine activity may promote self-regulation impairing beliefs in part by inducing depressive mood. One intriguing possibility, then, is that cytokines may help promote the development of beliefs that impair effective self-regulation, especially in individuals who are cognitively vulnerable.
Self-Regulatory Depletion
Another potential aspect of self-regulation that may be modulated by immune system activity is self-regulatory depletion. Research has suggested that exercising self-regulation impairs self-regulatory abilities (Baumeister & Heatherton, 1996; Hagger et al., 2010). This phenomenon has been given various labels, but it is most commonly referred to as depletion. It is important to note that the existence of self-regulatory depletion has been contested by a failed replication attempt (Hagger et al., 2016; for responses, see Baumeister & Vohs, 2016; Dang, 2016) and by meta-analytic techniques that account for publication bias (Carter, Kofler, Forster, & McCullough, 2015; for a response, see Inzlicht, Gervais, & Berkman, 2015). Nevertheless, evidence linking cytokines to alterations in brain regions that are thought to underpin depletion is persuasive, thus providing a justification for examining potential links between cytokine activity, self-regulatory depletion, and self-regulation.
Assuming that self-regulatory depletion exists in some form, there are several mechanisms that may underlie this effect. For example, depletion may be caused by reductions in a neural resource (Berkman & Miller-Ziegler, 2013; Persson, Larsson, & Reuter-Lorenz, 2013), resultant stronger emotions or impulses (Vohs et al., 2012), altered motivation and attention (Inzlicht & Schmeichel, 2012), or some combination of these processes. Depletion, if it exists, may also correspond to alterations in specific neural regions that are employed during self-regulation. For example, depletion may contribute to reduced activity in the ACC (Inzlicht & Gutsell, 2007) and dorsolateral PFC (DLPFC; Friese et al., 2013; Hedgcock, Vohs, & Rao, 2012), increased activity in reward- or emotion-related brain regions, and reduced functional connectivity between reward or emotion-related regions and systems involved in top-down control (Wagner, Altman, Boswell, Kelley, & Heatherton, 2013; Wagner & Heatherton, 2013).
Depletion is hypothesized to impact self-regulation through these dynamics, but premature or more profound depletions of self-regulatory ability may occur as a result of proinflammatory cytokine activity. Cytokines, for example, can alter the function of brain regions that are thought to underpin self-regulatory depletion. More specifically, acute inflammatory challenges, such as typhoid vaccination, can cause an exaggerated DLPFC response when individuals engage in tasks that require self-control (Harrison, Brydon, Walker, Gray, Steptoe, Dolan, et al., 2009). Since the DLPFC plays a central role in self-regulation and self-regulatory fatigue, excessive DLPFC activity may lead to a more pronounced self-regulatory depletion on subsequent tasks involving self-regulation. Consistent with this possibility, prior research has shown that DLPFC activation during a task requiring self-control mediates impairment on subsequent tasks requiring self-control, with greater activation during the first task predicting greater self-regulatory impairment on the second task (Richeson et al., 2003). Thus, cytokine-induced exaggerations in DLFPC activity during a self-control task may lead to quicker and more pronounced self-regulatory depletion. In humans, acute inflammatory challenges have also been shown to reduce functional connectivity between the medial PFC (mPFC) and brain regions involved in mood and emotion (Harrison, Brydon, Walker, Gray, Steptoe, & Critchley, 2009), similar to that seen in self-control depletion (Wagner & Heatherton, 2013). Putting these links together, then, data suggest that proinflammatory cytokine activity may be one mechanism underlying quicker and more pronounced self-regulatory depletion.
Interestingly, an acute inflammatory challenge has been shown to reduce resting glucose metabolism in the ACC in humans (Hannestad et al., 2012), which is a brain region critically important in self-regulation that shows reduced activity during depletion (Inzlicht & Gutsell, 2007). As previously stated, a major theory of self-regulatory depletion is that focal reductions in glucose availability mediate effects of self-regulatory depletion (Baumeister, 2014). Because cytokine expression and production increases following self-regulatory depleting tasks (Brydon et al., 2005), these results suggest that cytokines may not only contribute to quicker and more pronounced depletion but that they may also alter glucose metabolism in ways that impair self-regulation. The data are thus consistent with glucose-dependent models of self-regulatory depletion, but extend this work in an important new direction by identifying cytokines as a potential proximal biological mechanism that plays a key role in altering glucose metabolism in the ACC.
In sum, existing data provide initial evidence suggesting that proinflammatory cytokines could contribute to a quicker or more pronounced self-regulatory depletion by altering neural activity in key brain regions. At the same time, we are not aware of any studies that have directly tested this mechanistic model to date, and alternative directional effects are possible. Rather than cytokine activity contributing to quicker and more pronounced self-regulatory depletion, for example, it is possible that cytokine activity alters processing in brain regions supporting self-regulation, which may in turn impair self-regulation. Additional research is thus needed to examine the nature and directionality of these effects, and to further investigate whether self-regulatory depletion exists as it has been conceptualized to date.
Motivation and Reward
Like self-regulatory beliefs and depletion, immune system activity may alter motivation and reward processing in ways that impair self-regulation. In recent years, substantial attention has also been paid to the ways in which motivation affects self-regulation. Self-regulatory failure can be overcome by increasing motivation, which indicates that inadequate motivation may be one of the critical components of self-regulatory failure (Baumeister & Vohs, 2007; Muraven & Slessareva, 2003). Reward has also been studied in the context of self-regulation. Perhaps counterintuitively, reward expectancy appears to bolster self-control in some situations (Schmidt, Lebreton, Cléry-Melin, Daunizeau, & Pessiglione, 2012), such as when expecting a reward that requires effort to obtain increases one’s motivation to engage in self-regulation. Similarly, tasks that are interesting or have been previously rewarded require less self-regulatory resources, and may even replenish those resources despite task demands (Goto & Kusumi, 2013; Thoman, Smith, & Silvia, 2011). These findings are qualified, though, by other data showing that the reward value of stimuli opposite from long-term goals, such as the reward value of a piece of cake when trying to lose weight, can drive self-regulatory failure (Wagner et al., 2013). Thus, although the relation between reward sensitivity and self-regulation is not as linear as is motivation, reward sensitivity can be beneficial for self-regulation insofar as it influences one’s motivation to engage in self-regulation.
Motivation and reward sensitivity are both heavily influenced by proinflammatory cytokines. Some of the first behavioral effects observed along these lines involved the finding that proinflammatory cytokines induce anhedonia and relatively quick reductions in motivation and socially affiliative behavior, which are now included in the general constellation of behaviors known as sickness behaviors in both human and nonhuman animals (Dantzer & Kelley, 2007; Dantzer, O’Connor, Freund, Johnson, & Kelley, 2008; Maier & Watkins, 1998). In addition, experimentally inducing an inflammatory state strongly reduces motivation in nonhuman animals (e.g., Haba et al., 2012) and reduces neural responses to motivationally salient stimuli in humans (Harrison, Cercignani, Voon, & Critchley, 2015). As such, the consensus is that sickness behaviors reflect a motivational state, wherein motivation is reduced to facilitate recovery from physical injury or infection (Dantzer & Kelley, 2007). This lack of motivation bears directly upon self-regulation, as self-regulatory failure often occurs from a lack of motivation to maintain goals (Altman et al., 2013; Heatherton & Wagner, 2011).
A corollary of decreased motivation is decreased sensitivity to pleasure or reward. And, indeed, inflammatory activity also impairs sensitivity to reward. Inducing an inflammatory state in mice, for example, reduces their pleasure-seeking behavior (Markou & De La Garza, 2005). Similarly in humans, an experimentally-induced inflammatory state has been shown to decrease neural responding to reward in the ventral striatum, which is a brain region that mediates experiences of pleasure (Eisenberger, Berkman, et al., 2010). As mentioned previously, research has shown that decreased sensitivity to reward could either help self-regulation or contribute to its failure. If a goal relies on reward as an incentive, such as losing weight to look good in a new pair of pants, decreasing sensitivity to reward may impair self-regulation. Conversely, if a goal does not rely on reward for motivation, such as losing weight to avoid health problems, then decreasing reward sensitivity will leave those regulatory processes unaffected and may even guard against self-regulatory failure by decreasing the reward value of goal-opposing stimuli. In sum, then, inflammatory activity may impair self-regulation by reducing motivation, and may either help or hinder self-regulation by decreasing sensitivity to reward.
Appraisals of Stress
Immune system activation may also alter appraisals of stress, thereby impairing self-regulation. Perhaps the most commonly discussed cause of self-regulatory failure is exposure to stress. Although stress does not impair all cognitive processes (e.g., Shields, Lam, Trainor, & Yonelinas, 2016), failures of self-regulation are known to occur following several different types of stress, including difficult social interactions (Finkel et al., 2006; Vohs et al., 2005), social exclusion (Baumeister, DeWall, Ciarocco, & Twenge, 2005), academic stress (Oaten & Cheng, 2005), and emotional distress (Tice, Bratslavsky, & Baumeister, 2001). It should be noted, however, that the effects of stress on self-regulatory abilities are more nuanced than is often presented. For example, sex, biological and emotional reactivity to stress, and individual differences in susceptibility and resilience to stress can all moderate or attenuate the impairing effects of stress on self-regulatory abilities (Elzinga & Roelofs, 2005; Shields, Moons, Tewell, & Yonelinas, 2016; Schoofs, Pabst, Brand, & Wolf, 2013; Sprague, Verona, Kalkhoff, & Kilmer, 2011). Still, reduction in self-regulatory abilities that occur following stress can create a cyclical pattern wherein exposure to stress impairs self-regulation, which in turn leads to further occurrences of stress and impairments in self-regulation (Arnsten, 2009; see also Hammen, Kim, Eberhart, & Brennan, 2009). This may potentially explain why stress is such a strong predictor of failures in self-regulation (Muraven & Baumeister, 2000). Conversely, appraising life events as stressful is a key factor shaping the effects of different life events on health (Haley, Levine, Brown, & Bartolucci, 1987; Slavich & Cole, 2013; Slavich, 2016).
Although inflammatory activity has not been shown to contribute directly to the occurrence of stressful life events, research has shown that inflammation can alter experiences of the social world and individuals’ appraisal of stressors. In a sample of chronically stressed women, for example, circulating levels of proinflammatory cytokines were found to predict greater engagement of brain regions that process the affective valence of stimuli—specifically, the subgenual anterior cingulate cortex (sACC) and parts of the orbitofrontal cortex (OFC)—in response to grief; moreover, neural activity in these regions was associated with behavioral indicators of grief (Lane, Wager, O’Connor, Irwin, & Wellisch, 2009). However, it should be noted that mere associations of cytokines with neural activity in regions related to appraisals and perceptions of stress do not necessarily imply that cytokines themselves alter these experiences of stress. More pointedly, experimentally inducing an inflammatory response triggers significant increases in self-reported feelings of social isolation, demonstrating that inflammation is intimately involved in contributing to negative appraisals of events (Eisenberger, Inagaki, et al., 2010; see also Reichenberg et al., 2001). To summarize, then, experiences of stress may impair self-regulatory abilities (Cohen & Lichtenstein, 1990; Crescioni, 2012; Duckworth, Kim, & Tsukayama, 2012; Sprague et al., 2011), and proinflammatory cytokine activity contributes to greater experiences of stress (Eisenberger, Inagaki, et al., 2010; Hannestad, DellaGioia, Ortiz, Pittman, & Bhagwagar, 2011). Proinflammatory cytokine-mediated alterations in appraisals of stress may thus play an important role in impairing self-regulation.
Executive Function
Perhaps the most important psychological resource necessary for effective self-regulation is executive function (Hofmann, Schmeichel, & Baddeley, 2012). There is some debate about whether executive function is distinct from (but supports) self-regulation, or whether executive function and self-regulation are essentially the same construct (Diamond, 2013; Hofmann et al., 2012). For the present discussion, though, these constructs may be thought of as the same. For example, a common task assessing executive function, the Stroop task, is also commonly used to assess self-regulation. Taking a stance on this particular debate is not critical for the present discussion, although it is important to recognize that impairment in executive function indicates impairment in self-regulation.
Executive function refers to higher cognitive processes that enable flexible, stable, goal-directed, and adaptive thought, and it is crucial for appropriate functioning in contemporary society. Factor analysis studies have indicated that there are three primary, interrelated, yet slightly different executive functions—namely, updating, inhibition, and set shifting (Miyake et al., 2000). Updating is the active and intentional monitoring and updating of information, inhibition is the process of overriding pre-potent responses or inhibiting impulses/distractions, and set-shifting refers to the ability to cognitively change one’s mental set or focus on tasks. Executive function abilities arise from the complex interactions of the PFC, other cortical structures, and subcortical structures (Alvarez & Emory, 2006; Heyder, Suchan, & Daum, 2004); still, prefrontal structures do seem to have some primacy (Mansouri, Tanaka, & Buckley, 2009). As the control processes of self-control are dependent upon the PFC, it is not surprising that these three basic executive functions (i.e., working memory, response inhibition, and mental set shifting) enable self-regulation (Hofmann et al., 2012). Working memory permits an active representation of goals and goal-relevant attentional control, inhibition enables the ability to respond to a given stimulus while overriding impulses or habits, and task-switching allows for flexible switching between different ways of meeting the same goal (Hofmann et al., 2012). The interrelated aspect of the three executive functions allows them to be usefully conceptualized as supporting a single construct and thus proves pragmatic for the purposes of this review. Numerous studies have been conducted examining how cytokines influence executive functions, and we now review this work, focusing on four types of studies: cross-sectional and longitudinal association studies, cytokine administration studies, experimental studies involving the induction of acute inflammation, and genetic studies.
Cross-sectional and longitudinal associations of cytokines with executive function
Our understanding of how inflammatory activity influences executive function is gradually becoming clearer. Impairments in executive function in healthy individuals has been associated with several mediators of inflammatory activity, including the cytokines interleukin (IL)-1β (Simpson et al., 2013; Trompet et al., 2008), IL-6 (Marsland et al., 2006; Marsland et al., 2015; Simpson et al., 2013; Trollor et al., 2012), TNF-α (Jefferson et al., 2011), and IL-12 (Trollor et al., 2012), as well as the markers of systemic inflammation haptoglobin (Teunissen et al., 2003) and C-reactive protein (CRP; Marsland et al., 2015; Wersching et al., 2010). In addition, a variety of cytokines and markers of systemic inflammation have been associated with brain alterations that negatively impact executive function in humans. These brain alterations include reduced total brain white matter tract integrity (Marsland, Krajina, & Gianaros, 2012), reduced frontal lobe white matter tract integrity (Wersching et al., 2010), white matter hyperintensities, total brain gray matter atrophy (Satizabal, Zhu, Mazoyer, Dufouil, & Tzourio, 2012), PFC gray matter atrophy, and hippocampal gray matter atrophy (Marsland et al., 2008), thus supporting the abovementioned associations between cytokines and poor executive function. It is impossible to establish a causal effect from these correlational data alone; however, the aforementioned effects have been found to be significant after controlling for multiple potentially confounding factors, including age, body mass index, and smoking status. In addition, many of these studies longitudinally examined associations between these factors and found that higher levels of proinflammatory cytokines at baseline predict lower executive function over time (e.g., Jefferson et al., 2011; Teunissen et al., 2003; Trompet et al., 2008).
One longitudinal study that examined associations between cytokines and executive function over time attempted to partial out other components of the stress response by investigating a perioperative immune response during cardiac surgery, and then examining longitudinal associations between inflammatory activity and post-operative neurocognitive decline (Ramlawi et al., 2006). Although anesthesia can cause cognitive impairment, this study may help to explain why this cognitive impairment occurs. In particular, the study found that increases in CRP, IL-1β, and IL-10 during surgery were all significantly associated with neurocognitive decline, which suggest that longitudinal associations between inflammatory markers and impairments in executive function could be causal in nature. These data thus provide evidence that inflammatory activity is strongly associated with, and also increases prior to, executive function impairments, which impair self-regulation.
Administration of cytokines and executive function
Although the aforementioned correlational and longitudinal studies do not themselves establish that inflammatory activity impairs executive function, they do provide evidence that at least some cytokines may contribute to executive function impairments. Other evidence comes from studies in which cytokines have been carefully administered for medical purposes. For example, multiple studies have found that the use of cytokine therapy for medical treatment, such as exogenous administration of IL-2, leads to deficits in both cognition and executive function in humans (Capuron, Ravaud, & Dantzer, 2001; Meyers & Abbruzzese, 1992; Meyers, Valentine, Wong, & Leeds, 1994; Pavol et al., 1995; cf. Fontana et al., 2007). Research has also shown that cytokine therapy results in functional deficits in the frontal lobes (Juengling et al., 2000; Meyers et al., 1994), although the relation of these neurobiological effects to executive function has only been examined in one study to date (Meyers et al., 1994). These studies provide evidence that excessive peripheral inflammatory activity contributes to cognitive deficits. At the same time, additional research is necessary to determine whether extant alterations in the immune systems of individuals receiving cytokine therapy modulate the neurocognitive effects seen following cytokine administration.
Experimental inductions of acute inflammation and executive function
Despite the potential limited generalizability of studies employing clinical cytokine administration, a small number of studies have investigated executive function in conditions of experimentally-induced acute inflammatory and glucocorticoid activity via endotoxin injection in otherwise healthy individuals. Generally, findings have been mixed. Two small studies (20–24 participants) failed to find support for the idea that endotoxin injection impaired working memory, which is one component of executive function (Grigoleit et al., 2010; Reichenberg et al., 2001). An additional small study (i.e., 10 participants) found that endotoxin administration actually increased working memory (Cohen et al., 2003), while another small study found that endotoxin administration either impaired or enhanced cognition and working memory depending upon the dose of endotoxin given (Grigoleit et al., 2011). Although human studies examining the effects of subclinical cytokine administration on executive function are rare, multiple rodent studies have found that endotoxin injection induces deficits in working memory (e.g., Chen et al., 2008; Sparkman et al., 2006). Notably, one of these rodent studies found that IL-6 was necessary for deficits in working memory (Sparkman et al., 2006).
These mixed findings may be due in part to the fact that endotoxin, which induces acute inflammatory activity, also leads to increased circulating cortisol, which has anti-inflammatory properties. Moreover, several recent human studies have suggested that increases in cortisol may enhance executive function (Henckens et al., 2011; Schwabe, Tegenthoff, Höffken, & Wolf, 2013; Shields et al., 2015) and may be responsible for improving cognitive performance under endotoxin (van den Boogaard et al., 2010). Indeed, exogenous administration of cortisol leads to enhancements in working memory performance during the time at which working memory testing is done following endotoxin administration (Shields et al., 2015). Consistent with these findings, the only study conducted to date that assessed executive function and also employed a vaccine that does not increase cortisol obtained different results than prior studies, although it should be noted that the task employed in this study primarily assessed inhibition, not working memory (see Brydon, Harrison, Walker, Steptoe, & Critchley, 2008). Nevertheless, even with a total sample of 16 participants, this study found that reaction times on the Stroop task varied as a function of IL-6 levels, with higher IL-6 levels predicting slower reaction times. There was also a tendency for participants receiving the typhoid vaccine in this study to commit more errors on incongruent Stroop trials, which indicates poorer executive function, but the study’s small sample size limited the statistical power available to detect significant effects. Given these findings, more research is clearly needed to understand the effects of experimentally-induced inflammatory activity on executive function and self-regulation. These studies could use an improved paradigm of inducing acute inflammatory activity—such as one that does not upregulate cortisol—but they will also need to employ larger sample sizes.
Genetic differences in immune system genes and executive function
Although genetic studies cannot elucidate cause, they provide a unique opportunity for studying associations between inflammatory activity and executive function insofar as they can help isolate effects of proinflammatory cytokines that are not entangled with the effects of stress, cortisol, illness, or behaviors that upregulate inflammation. One cytokine that has been examined in neurogenetic studies of humans is IL-1β. In this context, a genetic variant that results in reduced expression of an enzyme converting IL-1β to its active form in humans—namely, the gene that codes for the interleukin-1β-converting enzyme (ICE)—has been found to predict lower levels of circulating IL-1β and also longitudinally predict better executive function compared to a genetic variant that is associated with higher levels of ICE and IL-1β (Trompet et al., 2008). Similarly, a genetic variant in the promoter region of the human IL-1β gene that results in increased IL-1β has been found to be associated with reduced working memory in healthy elderly males (Tsai et al., 2010) and females (Sasayama et al., 2011). These last two results may be explained by the fact that this genetic variant predicts reduced functional connectivity between neural regions involved in executive function, including the anterior midcingulate cortex, multiple areas in the PFC, and the putamen (Tu et al., 2013). However, additional research is needed to examine whether activity in these brain regions mediate the effects of variation in the human IL-1β gene on executive functions.
In addition to this research, one population-based study investigating the relation between cognitive factors and various cytokine-related genetic polymorphisms including IL-1β in elderly individuals did not find associations between IL-1β polymorphisms and executive function (Marioni, Deary, Murray, Fowkes, & Price, 2010). However, this study only looked at elderly Scottish citizens with asymptomatic atherosclerosis, and it is possible that this null effect is due to subtle effects of atherosclerosis on cognition or undiscovered and possibly culturally-related confounds. In general, studies suggest that IL-1β is associated with poorer executive functioning, independent of other biological dysregulation, which indicates that IL-1β contributes to impairments in self-regulation.
A few studies have examined associations between variation in genes coding for IL-6 and executive function in humans, and these studies also reveal conflicting results. The Scottish elderly population-based study referenced above found no association between genetic polymorphisms related to IL-6 and executive function abilities (Marioni et al., 2010), although that study only examined individuals with atherosclerosis. Additionally, half of the sample was instructed to regularly take aspirin, which can influence cytokine activity. In contrast, a larger population-based study of elderly Americans found that levels of IL-6 and the genetic variant that influences IL-6 production (i.e., the IL-6 -174 CC genotype) predicted poorer executive function and Stroop task performance (Mooijaart et al., 2013). These studies should be interpreted with caution, as the reasons for the conflicting results are not entirely clear. Nevertheless, the studies provide initial support for the notion that genetic variation in the gene that codes for IL-6 is associated with deficits in executive function and may thus impair self-regulation.
Compared to the limited number of genetic studies on genes that code for IL-1β and IL-6, several studies have examined how genetic factors involved in TNF-α production relate to cognitive function in humans. Variations in a single nucleotide polymorphism (SNP) in the human TNF-α gene (AA/AG/GG) leads to differing transcriptional activity, with the -308A allele conferring a stronger transcriptional activity than the -308G allele, likely resulting in higher TNF-α levels for individuals with the AA or AG genotype (Hajeer & Hutchinson, 2001; Wilson, Symons, McDowell, McDevitt, & Duff, 1997). This SNP has also been found to result in changes in cognitive control. In particular, individuals with the AA/AG genotype exhibit both behavioral and neural evidence of enhanced attentional processes, but reduced conflict processing and action selection abilities, relative to their GG counterparts (Beste, Baune, Falkenstein, & Konrad, 2010). Similarly, another study found that individuals with the AA/AG genotype exhibit better response inhibition, but reduced error processing functions, when compared with their GG counterparts (Beste et al., 2011). In sum, these studies have demonstrated that genetic variation that leads to higher circulating levels of TNF-α enhances attention and inhibition, but impairs error processing, conflict monitoring, and action selection abilities. Thus, although TNF-α influences executive functions involved in self-regulation, the association between TNF-α and executive function—and thus self-regulation—is more complex than the association between self-regulation and IL-6 or IL-1β.
In sum, data from several sources, including correlational, longitudinal, experimental, and genetic studies, converge to suggest that heightened proinflammatory cytokine activity contributes to poorer executive function. Each of these lines of research is not completely persuasive by itself. When considered together, though, they begin to provide a stronger case indicating that cytokine-related deficits in executive function are an important factor underlying links between the immune system and impaired self-regulation abilities.
Summary of Links Between Cytokines and Self-Regulation
To summarize, cytokines appear to play a role in influencing several processes that are important for self-regulation. These processes include beliefs about self-regulation, self-regulatory depletion, reward sensitivity and motivation, appraisals of stress, and executive function. In each of these instances, more proinflammatory cytokine activity appears to be related to less self-regulatory ability. Importantly, cytokines may be bi-directionally related to beliefs about self-regulation—that is, they may be both modulated by self-regulatory beliefs and also play a role in shaping such beliefs. Similarly, cytokine activity alters neural processes in ways that appear to exacerbate self-regulatory depletion. In addition, cytokines influence both reward sensitivity and motivation, with cytokine activity reducing motivation and producing anhedonia. Cytokines also alter neurocognitive appraisals of stress, illustrating another pathway through which cytokines may impair self-regulation. Finally, excessive cytokine activity appears to impair executive function. When considered together, then, these findings provide multiple sources of support for the formulation that increased cytokine activity can impair multiple self-regulatory processes.
An Immunologic Model of Self-Regulatory Failure
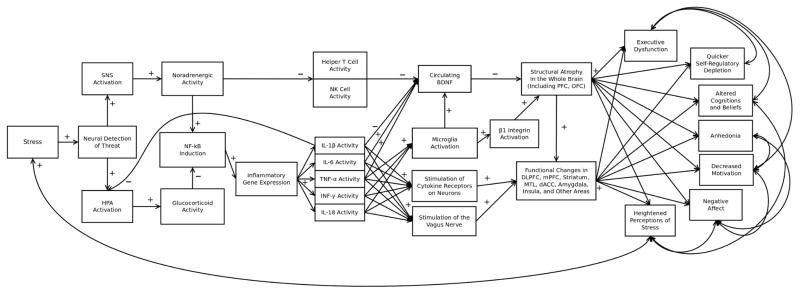
Based on these findings, we propose an integrated, multi-level model of self-regulation, inflammation, and health, which describes how immunologic factors may influence self-regulation, and how self-regulation may in turn shape human health and behavior. According to this immunologic model of self-regulatory failure, elevations in proinflammatory cytokine activity can occur as a result of several factors, including bacterial or viral infection, physical injury, poor diet or sleep, or psychological stress. These cytokines can be released centrally in the brain, but even when elevated in peripheral tissue, there are several mechanistic pathways (e.g., involving active and passive transport, and the SAM and HPA axes) by which peripheral inflammation can communicate with the central nervous system to alter neural activity in brain regions that are critical for self-regulation, such as the DLPFC and ACC. Altered neural activity in these brain systems in turn impairs self-regulatory capacity. These dynamics exert short-term effects on self-regulation. However, long-term changes can also develop. Over repeated exposure to inflammatory triggers, for example, glucocorticoids lose their ability to effectively down-regulate inflammation (i.e., glucocorticoid resistance) and adequately regulate immune system activity. The resulting dysregulation leads to sustained increases in proinflammatory cytokine activity and alterations in immune-related gene expression. As discussed in the section on structural changes in the brain produced by cytokines, several pathways link aberrant immune system activity with functional and structural changes in the brain, including reduced prefrontal cortical volume. These changes can in turn have numerous cognitive and behavioral consequences that predispose a person to experience self-regulatory failure. There are also several neurocognitive and behavioral pathways that give the immune system the ability to influence self-regulation, including personal beliefs about self-regulation, self-regulatory depletion, reward sensitivity and motivation, executive function, and perceptions of stress. These pathways are illustrated in Figure 2.
Figure 2.
Immunologic model of self-regulatory failure. Activation of the immune system is hypothesized to induce biological changes that alter neurocognitive and other biological processes that underpin self-regulation, leading to decrements in self-regulation. In particular, after a social-environmental stressor is neurally detected, the hypothalamic-pituitary-adrenal (HPA) and sympathetic-adrenal-medullary (SAM) axes increase in activity, leading to a decrease in natural killer (NK) cell and helper T cell activity, and an increase in the release of numerous proinflammatory cytokines from immune cells. These immunologic dynamics then directly and indirectly influence neuronal activity, contributing to structural and functional changes in brain regions that support self-regulation. These neural alterations in turn produce an impaired self-regulatory phenotype, which leads to a decreased ability to successfully navigate stressful situations, ultimately increasing the likelihood of experiencing more stress in the future.
Over brief periods of time, increases in immune system activity and decrements in self-regulatory ability typically have only limited effects on health. As increases in inflammation and difficulties self-regulating become more frequent or prolonged, however, immune-related health problems and disease states can eventually arise. A primary driver of these effects involves the fact that inflammatory activity, self-regulatory problems, and the pathways linking these factors can become mutually self-promoting over time. For example, inflammation can lead to failures in self-regulation that cause individuals to choose food options or forego exercise routines in a manner that leads to more adipose tissue, which is proinflammatory (Fantuzzi, 2005). Likewise, increases in proinflammatory cytokine activity may heighten perceptions of stress, leading to further increases in inflammation and neural sensitivity to threat, and the evolution of a positive feedback loop in the cytokine-neural threat network that can have lasting effects on self-regulatory ability and health (Slavich & Irwin, 2014). From this perspective, a person’s biological milieu, psychosocial life history, thoughts, and behaviors all influence one another and interact over time to shape self-regulatory capabilities and lifespan health.
One benefit of this immunologic model of self-regulatory failure is that it may have broad explanatory power. First, it may explain why self-regulatory failure occurs following stress and prior acts of self-regulation, as both stress (Steptoe, Hamer, & Chida, 2007) and self-regulation (Brydon, Edwards, Mohamed-Ali, & Steptoe, 2004; Brydon et al., 2005) increase circulating levels of proinflammatory cytokines. Second, it may explain why individuals with certain medical illnesses involving the immune system or inflammation—such as psoriasis, diabetes, hypertension, and arthritis—have poorer self-regulatory abilities (Brands et al., 2007; Dick, Eccleston, & Crombez, 2002; Fortune et al., 2003; Marek, Placek, & Borkowska, 2011; Saxby, Harrington, McKeith, Wesnes, & Ford, 2003; Schillerstrom, Horton, & Royall, 2005; van den Berg, de Craen, Biessels, Gussekloo, & Westendorp, 2006; Vicario, Martinez, Baretto, Casale, & Nicolosi, 2005). Similarly, the model may help explain why certain TNF-α inhibitors enhance executive function (Tobinick & Gross, 2008), and why several psychiatric disorders, including depression and ADHD, are frequently characterized by both elevated inflammatory activity and difficulties in self-regulation (Oades, Dauvermann, Schimmelmann, Schwarz, & Myint, 2010; Slavich, O’Donovan, Epel, & Kemeny, 2010). Finally, this model may help explain why rates of self-regulatory failure are elevated under conditions of chronic stress, even in the absence of acute stress (Hammen, Kim, Eberhart, & Brennan, 2009), due in part to the fact that elevations in inflammation can become self-promoting over time (Cohen et al., 2012; Slavich & Cole, 2013).
Looking forward, this research may provide clinicians with new ideas for how to treat medical problems that are influenced by both self-regulatory difficulties and inflammation, such as obesity. Obesity is a prototypic example because it is a leading public health problem (Lobstein, Baur, & Uauy, 2004) that is characterized by both chronic inflammation (Cazettes, Cohen, Yau, Talbot, & Convit, 2011) and persistent deficits in self-regulation (Wing, Tate, Gorin, Raynor, & Fava, 2006) that include poorer executive function and worse emotion regulation (Blanco-Gómez et al., 2015; Clyne & Blampied, 2004; Cook et al., 2014; Coppin, Nolan-Poupart, Jones-Gotman, & Small, 2014; Zijlstra et al., 2012). Because obese individuals have more adipose tissue, which is a storehouse for proinflammatory cytokines (Tilg & Moschen, 2006), they typically have greater difficulties with self-regulation, which challenges their ability to make healthy food choices and adhere to a prescribed diet. We describe obesity here only as an example disease condition in which inflammatory activity may both contribute to, and be caused by, the disorder, but others exist including (for example) type 2 diabetes, tobacco-related diseases, and hypertension. Ultimately, to the extent that inflammation-related difficulties in self-regulation help sustain disease states like obesity, inflammation may be an important target for pharmacologic interventions aimed at improving self-regulation and lessening disease symptoms.
Cytokine-Specific Effects on Self-Regulation
The empirical precedent for this model comes from the large body of research on inflammation and sickness behavior, which has demonstrated that increased inflammatory activity can have a variety of neurobehavioral effects on humans (e.g., Dantzer & Kelley, 2007; Dantzer et al., 2008; Maier & Watkins, 1998; Markou & De La Garza, 2005; Raison et al., 2006). Although sickness behaviors may influence some outcomes that are associated with poor self-regulation such as lack of motivation, sickness behaviors do not explain all of the inflammation-related deficits in self-regulation that are described in the model. If sickness behaviors were the primary mechanism linking proinflammatory cytokines and worse executive function, for example, then proinflammatory cytokine activity should be associated with worse overall cognitive performance, but this is not the case (Jefferson et al., 2011; Marsland et al., 2006; Trompet et al., 2008).
In addition, at least five lines of research presently support the notion that proinflammatory cytokines exert several specific effects on self-regulatory processes. First, peripheral administration of the proinflammatory cytokine IL-2 in humans selectively impairs the self-regulatory abilities of executive function and planning without influencing cognitive abilities that are unrelated to self-regulation (Capuron et al., 2001). Second, although circulating levels of the proinflammatory cytokines IL-6, IL-1β, and TNF-α are inversely associated with executive function, they are not associated with processes that are unrelated to self-regulation, such as visual immediate memory (Marsland et al., 2006; Trompet et al., 2008) and language processing (Jefferson et al., 2011; Trollor et al., 2012). Third, acute inflammatory challenges including endotoxin administration and typhoid vaccination do not influence processes that are unrelated to self-regulation, such as procedural memory (Harrison et al., 2014). Fourth, several studies have shown that it is specifically proinflammatory cytokines (and not other types of cytokines) that appear to influence self-regulatory abilities. Administration of antiviral cytokines such as interferon-α, for example, has no effects on executive function, even though antiviral cytokine therapy appears to decrease processing speed (Amodio et al., 2005; Capuron et al., 2001; Fontana et al., 2007). Finally, proinflammatory cytokines have some cognitive-behavioral effects that are more distantly related to self-regulation but still specific in nature. For example, proinflammatory cytokines can impair hippocampus-dependent delayed memory (Harrison et al., 2014), which is important for remembering personal goals and action steps that are necessary for self-regulation.
In sum, we do not suggest that proinflammatory cytokines are the sole biological cause of self-regulatory failure. Indeed, as reviewed above and as depicted in Figure 2, a multitude of other biological factors are also likely implicated (e.g., glucose, cortisol levels, etc.). However, proinflammatory cytokines do appear to play a very important and previously unappreciated role in self-regulatory abilities, and they also appear to exert several effects that are specific to self-regulation. We thus suggest that this immunologic model of self-regulatory failure may help shed new light on the biological bases of self-regulation in humans.
Testable Predictions
Several testable predictions can be derived from this model. For example, if inflammatory cytokines exacerbate self-regulatory depletion, then the effects of self-regulatory depletion should be minimized in individuals who take a drug or supplement that reduces proinflammatory cytokine activity, such as a prescribed TNF-α inhibitor or the over-the-counter supplement stinging nettle (Riehemann, Behnke, & Schulze-Osthoff, 1999; Teucher, Obertreis, Ruttkowski, & Schmitz, 1996). Similarly, if inflammation reduces individuals’ motivation, then taking an anti-inflammatory drug or supplement should increase motivation and enhance self-regulation abilities.
It is important to note that there are alternative interpretations of the findings reviewed here. For example, the physiologic stress response, which helps mobilize the body’s resources to avoid physical harm and preemptively combat infection, could take up available glucose, thereby leaving less glucose available for proximal acts of self-control or self-regulation. Similarly, stress may require behavioral responses that utilize self-regulation—which may or may not be triggered by immune system activation—and in turn deplete a person’s willpower. Importantly, however, these alternative interpretations of the available data are also testable. If immune system activation can impair self-regulation even when glucose is replenished, for example, then the idea that reducing local glucose is the sole mechanism through which inflammatory activity impairs self-regulation would be less parsimonious than the model presented here. Ultimately, these alternative theoretical conceptualizations of available data provide fruitful opportunities for future research.
Developmental Perspective on Inflammation, Self-Regulation, and Health
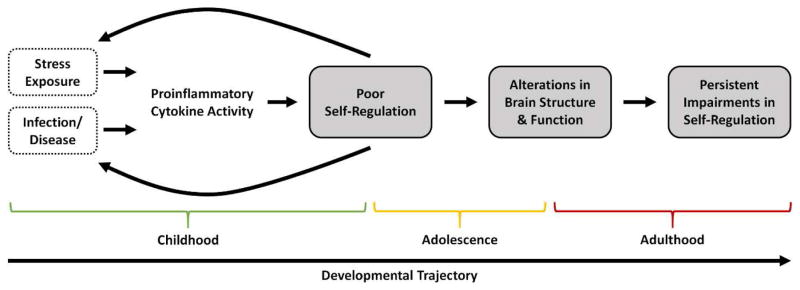
The above description of this immunologic model of self-regulatory failure presents a temporally static view of inflammation, self-regulation, and health. These interactions can also be viewed as developing over time, however, resulting from a complex interplay between stress exposure and ensuing changes in self-regulation and health (see Figure 3). Moreover, these dynamics can have potentially enormous effects on behavior and health over long periods of time. For example, individuals experiencing early adversity, including maltreatment, abuse, or neglect, or who are otherwise chronically or severely stressed, may exhibit structural and functional changes in neural systems that support self-regulation, as well as changes in stress-related biological axes (e.g., the HPA and SAM axis) that lead to persistent immune system dysregulation and related self-regulation difficulties. These difficulties can in turn cause social and behavioral problems, including conflictual relationships and problems at work or school, that engender additional life stressors and reinforce threat-related biases in neural and immune system functioning. Recent population-based studies have shown that self-regulatory abilities are crucial for interpersonal, educational, economic, and career success (Moffitt et al., 2011; see also Diamond, 2013). Consequently, over the long-run, these effects may also become embedded in individuals’ lives in the form of poorer social relationships, educational attainment, economic achievement, or professional development.
Figure 3.
Immunologic model of self-regulatory failure from a developmental perspective. Exposure to stress, infection, and disease in early life increases proinflammatory cytokine activity, which decreases an individual’s self-regulatory ability. Poor self-regulation can in turn feedback to cause stress generation behaviors (e.g., saying a hurtful thing to a friend or loved one during an argument, not arriving on time to work) and poor health behaviors (e.g., not completing a course of antibiotics, not washing one’s hands), which lead to greater exposure to stress and disease. As these dynamics continue over childhood and adolescence, more stable differences in brain structure and function can develop that ultimately produce persistent impairments in self-regulation in adulthood.
In addition to the cyclical nature of the effects described above, the influence of inflammatory activity on self-regulation may be amplified during sensitive or critical periods of development. Early childhood and adolescence are times of prefrontal cortex maturation, but they are also times of greater risk for inflammatory activity because of a nascent and developing immune system and potential stress from a possible chaotic home environment. Because inflammatory activity appears to contribute to structural changes in the prefrontal cortex among other brain regions (Marsland et al., 2015), it is possible that inequalities that predispose certain individuals to develop a more proinflammatory phenotype may have longer-lasting health and behavioral consequences for those individuals.
As these developmental aspects of the model improve over time, its explanatory power may increase. At present, we believe the framework may help explain aspects of how socioeconomic disparities in health arise and widen over the lifespan. For example, the stress of low socioeconomic status during childhood is known to contribute to elevated inflammatory activity that can impair self-regulation, which is crucial for academic achievement (John-Henderson et al., 2014). This discrepancy in self-regulation may in turn affect individuals’ ability to reach their fullest academic potential, which can limit their lifelong earning potential and increase their likelihood of remaining in a lower socioeconomic position. The stress associated with low socioeconomic status may also activate inflammatory processes that impair one’s self-regulatory abilities, which underlie successful coping with stress (Vohs & Baumeister, 2011; see also Shields, Kuchenbecker, Pressman, Sumida, & Slavich, 2016). This reduced ability to cope, coupled with the increased stress burden that accompanies being in a lower socioeconomic position, may in turn promote immune system dysregulation that degrades individuals’ ability to fight off infections and increases their likelihood of developing immune-related disorders. If this working model is substantiated, the framework could help explain how inflammation influences self-regulatory abilities that structure health disparities which are evident across individuals of differing socioeconomic positions.
Pressing Issues and Future Directions
We believe that the model outlined above represents an important new perspective on how cytokines are involved in self-regulatory failure. At the same time, there are several issues that require future clarification. The most immediate issue involves the need to better understand how proinflammatory cytokines interact with other potential biological influences on self-regulation. Currently, no research has explicitly examined the conjoint influences of cytokines, cortisol, and glucose on self-regulatory processes. However, studies of endotoxin administration provide an opportunity to study conjoint influences of cytokines and cortisol on self-regulatory abilities, given that endotoxin administration strongly increases both cytokine and cortisol production (e.g., van den Boogaard et al., 2010). As reviewed above, these paradigms have generally failed to find effects of endotoxin administration on executive functions, indicating that cortisol may help prevent cytokine-induced decrements to executive functions in endotoxin administration studies (Shields et al., 2015; see also Weckesser, Alexander, Kirschbaum, & Menningen, 2016). Ultimately, future studies that experimentally manipulate cytokines, cortisol, and glucose within the same study design are necessary to elucidate the additive and interactive effects that these three mechanisms have on self-regulation.
A second important future direction is to extend this work by employing different inflammatory challenges. As reviewed above, for example, only a few studies have directly manipulated acute inflammatory activity in humans. This is a limitation of the extant literature. More importantly, many of these manipulations, such as endotoxin administration, also affect cortisol levels. It will thus be important for future studies to examine the effects of inflammatory challenges that do not influence cortisol (e.g., typhoid vaccination; Brydon et al., 2008).
A third important issue involves determining the reasons (adaptive or otherwise) for why proinflammatory cytokines affect self-regulation. To address this issue, researchers could draw from the sickness behavior literature. This work has often suggested that the cognitive and behavioral effects of cytokines are an adaptive response to injury or infection (Dantzer & Kelley, 2007; Dantzer et al., 2008; Maier & Watkins, 1998; Markou & De La Garza, 2005; Raison et al., 2006). For example, a lack of motivation and interest in rewarding stimuli may promote more stationary behavior, thus allowing bodily resources to be redirected toward fighting infection and promoting recovery. Impaired self-regulation would help to further limit exploratory behavior by making it difficult to remember goals, execute plans, and maintain a positive mood. Ultimately, these effects, coupled with a lack of motivation and a disinterest in reward, would help ensure that an individual remains as stationary as possible to help facilitate recuperation.
A final important question to consider is whether associations between inflammatory activity and self-regulation are similar in Western, Educated, Industrialized, Rich, and Democratic (WEIRD) populations versus non-WEIRD populations. On the one hand, several of the studies reviewed here suggest that these associations may be generalizable. For example, two studies on elderly Chinese men in Taiwan examined associations between IL-β genotype and executive function (Tsai et al., 2010) and IL-β genotype and brain structure (Tu et al., 2013), respectively, and a third study on elderly Japanese women in Japan examined associations between IL-β genotype and executive function (Sasayama et al., 2011). Notably, each study yielded results that are consistent with those obtained in WEIRD populations.
On the other hand, though, a growing body of research is showing that early physical and microbial environments influence individuals’ inflammatory reactivity to different stimuli, such as social-environmental stressors (see McDade, 2012; McDade, Hoke, Borja, Adair, & Kuwaza, 2013). Therefore, it is possible that associations between inflammatory reactivity and self-regulation may differ in WEIRD versus non-WEIRD populations. Given the paucity of cross-cultural studies conducted on links between inflammation and self-regulation, additional research should examine how early microbial environment moderates the effects of inflammation on self-regulation.
Summary and Conclusion
As described at the beginning of this review, self-regulation refers to the ability to act in goal-directed ways, which is especially important in the face of distractions. Self-regulatory success is the result of a complex interaction of different processes that are all oriented toward goal maintenance in either proximal or distal ways. Self-regulatory failure, in contrast, can result from alterations in several processes that support self-regulation, and the consequences of such failures can be catastrophic for an individual’s life and the collective well-being of a society.
To elucidate mechanisms underlying these effects, we reviewed evidence showing that immune system factors can influence several neural, cognitive, and motivational processes that underpin self-regulation. We also described how dysregulated immune system activity can contribute to self-regulatory failure through multiple pathways, including by influencing beliefs about self-regulation, self-regulatory depletion, reward sensitivity and motivation, appraisals of stress, and executive function. Based on this work, we proposed an immunologic model of self-regulatory failure, which posits that proinflammatory cytokine activity plays a key role in altering neural, cognitive, and behavioral dynamics that can contribute to self-regulatory failure.
Despite evidence supporting this immunologic model, much more research is needed to test this formulation in both its static and developmental form, as well as to evaluate its predictive utility. This research would benefit from identifying additional pathways linking inflammation, self-regulation, and health. Future studies should also aim to elucidate central mechanisms that give cytokines the ability to directly impact self-regulation. Finally, additional research is needed to examine how theoretical advances on these issues could help improve the treatment of disease conditions that cause substantial morbidity and mortality. In sum, the research reviewed here provides an important theoretical account of how the immune system influences self-regulation, but much more work is needed to fully realize the benefits of this research for improving health.
Acknowledgments
Funding: Preparation of this review was supported by a University of California, Davis Provost’s Fellowship to Grant Shields; a Hellman Foundation Fellowship to Wesley Moons; and National Institutes of Health grant K08 MH103443 and a Society in Science—Branco Weiss Fellowship to George Slavich. The authors thank members of the former SNAP Lab at the University of California, Davis, for comments on a prior draft of the manuscript.
Footnotes
Author Contributions: Grant S. Shields and George M. Slavich developed the concept for this article and wrote the manuscript, Wesley G. Moons provided critical revisions to the paper, and all authors read and approved the final version.
Declaration of Conflicting Interests: The authors declare that they had no conflicts of interest with respect to their authorship or the publication of this article.
References
- Ai AL, Pargament K, Kronfol Z, Tice TN, Appel H. Pathways to postoperative hostility in cardiac patients: Mediation of coping, spiritual struggle and interleukin-6. Journal of Health Psychology. 2010;15:186–195. doi: 10.1177/1359105309345556. [DOI] [PubMed] [Google Scholar]
- Amodio P, De Toni EN, Cavalletto L, Mapelli D, Bernardinello E, Del Piccolo F, … Chemello L. Mood, cognition and EEG changes during interferon α (alpha-IFN) treatment for chronic hepatitis C. Journal of Affective Disorders. 2005;84:93–98. doi: 10.1016/j.jad.2004.09.004. [DOI] [PubMed] [Google Scholar]
- Alvarez JA, Emory E. Executive function and the frontal lobes: A meta-analytic review. Neuropsychology Review. 2006;16:17–42. doi: 10.1007/s11065-006-9002-x. [DOI] [PubMed] [Google Scholar]
- Arnsten AF. Stress signalling pathways that impair prefrontal cortex structure and function. Nature Reviews Neuroscience. 2009;10:410–422. doi: 10.1038/nrn2648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atherton OE, Robins RW, Rentfrow PJ, Lamb ME. Personality correlates of risky health outcomes: Findings from a large Internet study. Journal of Research in Personality. 2014;50:56–60. doi: 10.1016/j.jrp.2014.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Audet MC, Jacobson-Pick S, Wann BP, Anisman H. Social defeat promotes specific cytokine variations within the prefrontal cortex upon subsequent aggressive or endotoxin challenges. Brain, Behavior, and Immunity. 2011;25:1197–1205. doi: 10.1016/j.bbi.2011.03.010. [DOI] [PubMed] [Google Scholar]
- Bandura A. The primacy of self-regulation in health promotion. Applied Psychology. 2005;54:245–254. doi: 10.1111/j.1464-0597.2005.00208.x. [DOI] [Google Scholar]
- Barton GM. A calculated response: Control of inflammation by the innate immune system. Journal of Clinical Investigation. 2008;118:413–420. doi: 10.1172/JCI34431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumeister RF. Yielding to temptation: self-control failure, impulsive purchasing, and consumer behavior. Journal of Consumer Research. 2002;28:670–676. doi: 10.1086/338209. [DOI] [Google Scholar]
- Baumeister RF. Self-regulation, ego depletion, and inhibition. Neuropsychologia. 2014;65:313–319. doi: 10.1016/j.neuropsychologia.2014.08.012. [DOI] [PubMed] [Google Scholar]
- Baumeister RF, DeWall CN, Ciarocco NJ, Twenge JM. Social exclusion impairs self-regulation. Journal of Personality and Social Psychology. 2005;88:589–604. doi: 10.1037/0022-3514.88.4.589. [DOI] [PubMed] [Google Scholar]
- Baumeister RF, Heatherton TF. Self-regulation failure: An overview. Psychological Inquiry. 1996;7:1–15. doi: 10.1207/s15327965pli0701_1. [DOI] [Google Scholar]
- Baumeister RF, Heatherton TF, Tice DM. Losing control: How and why people fail at self-regulation. San Diego: Academic Press; 1994. [Google Scholar]
- Baumeister RF, Vohs KD. Self-regulation and the executive function of the self. In: Leary MR, Tangney JP, editors. Handbook of Self and Identity. New York: Guilford Press; 2005. pp. 197–217. [Google Scholar]
- Baumeister RF, Vohs KD. Self-regulation, ego depletion, and motivation. Social and Personality Psychology Compass. 2007;1:115–128. doi: 10.1111/j.1751-9004.2007.00001.x. [DOI] [Google Scholar]
- Baumeister RF, Vohs KD. Misguided effort with elusive implications. Perspectives on Psychological Science. 2016;11:574–575. doi: 10.1177/1745691616652878. [DOI] [PubMed] [Google Scholar]
- Berkman ET, Graham AM, Fisher PA. Training self-control: A domain-general translational neuroscience approach. Child Development Perspectives. 2012;6:374–384. doi: 10.1111/j.1750-8606.2012.00248.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berkman ET, Miller-Ziegler JS. Imaging depletion: fMRI provides new insights into the processes underlying ego depletion. Social Cognitive and Affective Neuroscience. 2013;8:359–361. doi: 10.1093/scan/nss111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berman MG, Yourganov G, Askren MK, Ayduk O, Casey BJ, Gotlib IH, … Jonides J. Dimensionality of brain networks linked to life-long individual differences in self-control. Nature Communications. 2013;4:1373. doi: 10.1038/ncomms2374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beste C, Baune BT, Falkenstein M, Konrad C. Variations in the TNF-α gene (TNF-α-308g→a) affect attention and action selection mechanisms in a dissociated fashion. Journal of Neurophysiology. 2010;104:2523–2531. doi: 10.1152/jn.00561.2010. [DOI] [PubMed] [Google Scholar]
- Beste C, Güntürkün O, Baune BT, Domschke K, Falkenstein M, Konrad C. Double dissociated effects of the functional TNF-α-308g/a polymorphism on processes of cognitive control. Neuropsychologia. 2011;49:196–202. doi: 10.1016/j.neuropsychologia.2010.11.037. [DOI] [PubMed] [Google Scholar]
- Bilbo SD, Schwarz JM. Early-life programming of later-life brain and behavior: A critical role for the immune system. Frontiers in Behavioral Neuroscience. 2009;3:14. doi: 10.3389/neuro.08.014.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blair C, Diamond A. Biological processes in prevention and intervention: The promotion of self-regulation as a means of preventing school failure. Development and Psychopathology. 2008;20:899–911. doi: 10.1017/S0954579408000436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blair C, Granger DA, Willoughby M, Mills-Koonce R, Cox M, Greenberg MT, … Fortunato CK. Salivary cortisol mediates effects of poverty and parenting on executive functions in early childhood. Child Development. 2011;82:1970–1984. doi: 10.1111/j.1467-8624.2011.01643.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanco-Gómez A, Ferré N, Luque V, Cardona M, Gispert-Llauradó M, Escribano J, … Canals-Sans J. Being overweight or obese is associated with inhibition control in children from six to ten years of age. Acta Paediatrica. 2015;104:619–625. doi: 10.1111/apa.12976. [DOI] [PubMed] [Google Scholar]
- Brands AM, Van Den Berg E, Manschot SM, Biessels GJ, Kappelle LJ, De Haan EH, Kessels RP. A detailed profile of cognitive dysfunction and its relation to psychological distress in patients with type 2 diabetes mellitus. Journal of the International Neuropsychological Society. 2007;13:288–297. doi: 10.1017/S1355617707070312. [DOI] [PubMed] [Google Scholar]
- Brydon L, Edwards S, Jia H, Mohamed-Ali V, Zachary I, Martin JF, Steptoe A. Psychological stress activates interleukin-1β gene expression in human mononuclear cells. Brain, Behavior, and Immunity. 2005;19:540–546. doi: 10.1016/j.bbi.2004.12.003. [DOI] [PubMed] [Google Scholar]
- Brydon L, Edwards S, Mohamed-Ali V, Steptoe A. Socioeconomic status and stress-induced increases in interleukin-6. Brain, Behavior, and Immunity. 2004;18:281–290. doi: 10.1016/j.bbi.2003.09.011. [DOI] [PubMed] [Google Scholar]
- Brydon L, Harrison NA, Walker C, Steptoe A, Critchley HD. Peripheral inflammation is associated with altered substantia nigra activity and psychomotor slowing in humans. Biological Psychiatry. 2008;63:1022–1029. doi: 10.1016/j.biopsych.2007.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burkley E, Anderson D, Curtis J. You wore me down: Self-control strength and social influence. Social and Personality Psychology Compass. 2011;5:487–499. doi: 10.1111/j.1751-9004.2011.00367.x. [DOI] [Google Scholar]
- Burnette JL, O’Boyle EH, VanEpps EM, Pollack JM, Finkel EJ. Mind-sets matter: A meta-analytic review of implicit theories and self-regulation. Psychological Bulletin. 2013;139:655–701. doi: 10.1037/a0029531. [DOI] [PubMed] [Google Scholar]
- Busillo JM, Cidlowski JA. The five Rs of glucocorticoid action during inflammation: Ready, reinforce, repress, resolve, and restore. Trends in Endocrinology & Metabolism. 2013;24:109–119. doi: 10.1016/j.tem.2012.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capuron L, Ravaud A, Dantzer R. Timing and specificity of the cognitive changes induced by interleukin-2 and interferon-alpha treatments in cancer patients. Psychosomatic Medicine. 2001;63:376–386. doi: 10.1097/00006842-200105000-00007. [DOI] [PubMed] [Google Scholar]
- Carter EC, Kofler LM, Forster DE, McCullough ME. A series of meta-analytic tests of the depletion effect: Self-control does not seem to rely on a limited resource. Journal of Experimental Psychology: General. 2015;144:796–815. doi: 10.1037/xge0000083. [DOI] [PubMed] [Google Scholar]
- Casey BJ, Somerville LH, Gotlib IH, Ayduk O, Franklin NT, Askren MK, … Shoda Y. Behavioral and neural correlates of delay of gratification 40 years later. Proceedings of the National Academy of Sciences of the United States of America. 2011;108:14998–15003. doi: 10.1073/pnas.1108561108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cazettes F, Cohen JI, Yau PL, Talbot H, Convit A. Obesity-mediated inflammation may damage the brain circuit that regulates food intake. Brain Research. 2011;1373:101–109. doi: 10.1016/j.brainres.2010.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatzisarantis NL, Hagger MS. Unsuccessful attempts to replicate effects of self control operations and glucose on ego-depletion pose an interesting research question that demands explanation. Appetite. 2015;84:328–329. doi: 10.1016/j.appet.2014.10.024. [DOI] [PubMed] [Google Scholar]
- Chen J, Buchanan JB, Sparkman NL, Godbout JP, Freund GG, Johnson RW. Neuroinflammation and disruption in working memory in aged mice after acute stimulation of the peripheral innate immune system. Brain, Behavior, and Immunity. 2008;22:301–311. doi: 10.1016/j.bbi.2007.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clyne C, Blampied NM. Training in emotion regulation as a treatment for binge eating: A preliminary study. Behaviour Change. 2004;21:269–281. doi: 10.1375/bech.21.4.269.66105. [DOI] [Google Scholar]
- Cohen O, Reichenberg A, Perry C, Ginzberg D, Pollmächer T, Soreq H, Yirmiya R. Endotoxin-induced changes in human working and declarative memory associate with cleavage of plasma “readthrough” acetylcholinesterase. Journal of Molecular Neuroscience. 2003;21:199–212. doi: 10.1385/JMN:21:3:199. [DOI] [PubMed] [Google Scholar]
- Cook R, Cheng HL, O’Dwyer N, Raymond J, Rooney K, Steinbeck K, O’Connor H. The relationship between overweight, obesity and cognitive function in adults: A systematic review and meta-analysis. Obesity Research & Clinical Practice. 2014;8:21. doi: 10.1016/j.orcp.2014.10.037. [DOI] [Google Scholar]
- Coppin G, Nolan-Poupart S, Jones-Gotman M, Small DM. Working memory and reward association learning impairments in obesity. Neuropsychologia. 2014;65:146–155. doi: 10.1016/j.neuropsychologia.2014.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen S, Janicki-Deverts D, Doyle WJ, Miller GE, Frank E, Rabin BS, Turner RB. Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proceedings of the National Academy of Sciences of the United States of America. 2012;109:5995–5999. doi: 10.1073/pnas.1118355109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen S, Lichtenstein E. Perceived stress, quitting smoking, and smoking relapse. Health Psychology. 1990;9:466–478. doi: 10.1037/0278-6133.9.4.466. [DOI] [PubMed] [Google Scholar]
- Coutlee CG, Huettel SA. The functional neuroanatomy of decision making: Prefrontal control of thought and action. Brain Research. 2012;1428:3–12. doi: 10.1016/j.brainres.2011.05.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crescioni A. The Frayed Edges Of Willpower: Perceived Stress Impairs Self-Regulatory Performance. Doctoral dissertation. 2012 Retrieved from http://diginole.lib.fsu.edu/
- Crockett MJ, Braams BR, Clark L, Tobler PN, Robbins TW, Kalenscher T. Restricting temptations: Neural mechanisms of precommitment. Neuron. 2013;79:391–401. doi: 10.1016/j.neuron.2013.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curfs JH, Meis JF, Hoogkamp-Korstanje JA. A primer on cytokines: Sources, receptors, effects, and inducers. Clinical Microbiology Reviews. 1997;10:742–780. doi: 10.1128/cmr.10.4.742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang J. Commentary: A multilab preregistered replication of the ego-depletion effect. Frontiers in Psychology. 2016;7:1155. doi: 10.3389/fpsyg.2016.01155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dantzer R, Kelley KW. Twenty years of research on cytokine-induced sickness behavior. Brain, Behavior, and Immunity. 2007;21:153–160. doi: 10.1016/j.bbi.2006.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dantzer R, O’Connor JC, Freund GG, Johnson RW, Kelley KW. From inflammation to sickness and depression: When the immune system subjugates the brain. Nature Reviews Neuroscience. 2008;9:46–56. doi: 10.1038/nrn2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daruna JH. Introduction to psychoneuroimmunology. 2. Academic Press; 2012. [Google Scholar]
- de Pablos RM, Villarán RF, Argüelles S, Herrera AJ, Venero JL, Ayala A, … Machado A. Stress increases vulnerability to inflammation in the rat prefrontal cortex. The Journal of Neuroscience. 2006;26:5709–5719. doi: 10.1523/JNEUROSCI.0802-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diamond A. Executive functions. Annual Review of Psychology. 2013;64:135–168. doi: 10.1146/annurev-psych-113011-143750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dick B, Eccleston C, Crombez G. Attentional functioning in fibromyalgia, rheumatoid arthritis, and musculoskeletal pain patients. Arthritis and Rheumatism. 2002;47:639–644. doi: 10.1002/art.10800. [DOI] [PubMed] [Google Scholar]
- Duckworth AL, Kim B, Tsukayama E. Life stress impairs self-control in early adolescence. Frontiers in Psychology. 2012;3:608. doi: 10.3389/fpsyg.2012.00608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg N, Fabes RA, Bernzweig J, Karbon M, Poulin R, Hanish L. The relations of emotionality and regulation to preschoolers’ social skills and sociometric status. Child Development. 1993;64:1418–1438. doi: 10.1111/j.1467-8624.1993.tb02961.x. [DOI] [PubMed] [Google Scholar]
- Eisenberger NI, Berkman ET, Inagaki TK, Rameson LT, Mashal NM, Irwin MR. Inflammation-induced anhedonia: Endotoxin reduces ventral striatum responses to reward. Biological Psychiatry. 2010;68:748–754. doi: 10.1016/j.biopsych.2010.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberger NI, Inagaki TK, Mashal NM, Irwin MR. Inflammation and social experience: An inflammatory challenge induces feelings of social disconnection in addition to depressed mood. Brain, Behavior, and Immunity. 2010;24:558–563. doi: 10.1016/j.bbi.2009.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elzinga BM, Roelofs K. Cortisol-induced impairments of working memory require acute sympathetic activation. Behavioral Neuroscience. 2005;119:98–103. doi: 10.1037/0735-7044.119.1.98. [DOI] [PubMed] [Google Scholar]
- Evans GW, Fuller-Rowell TE. Childhood poverty, chronic stress, and young adult working memory: The protective role of self-regulatory capacity. Developmental Science. 2013;16:688–696. doi: 10.1111/desc.12082. [DOI] [PubMed] [Google Scholar]
- Evans TD, Cullen FT, Burton VS, Dunaway RG, Benson ML. The social consequences of self-control: Testing the general theory of crime. Criminology. 1997;35:475–504. doi: 10.1111/j.1745-9125.1997.tb01226.x. [DOI] [Google Scholar]
- Fantuzzi G. Adipose tissue, adipokines, and inflammation. Journal of Allergy and Clinical Immunology. 2005;115:911–919. doi: 10.1016/j.jaci.2005.02.023. [DOI] [PubMed] [Google Scholar]
- Fontana RJ, Bieliauskas LA, Lindsay KL, Back-Madruga C, Wright EC, Snow KK, … Padmanabhan L. Cognitive function does not worsen during pegylated interferon and ribavirin retreatment of chronic Hepatitis C. Hepatology. 2007;45:1154–1163. doi: 10.1002/hep.21633. [DOI] [PubMed] [Google Scholar]
- Fortune DG, Richards HL, Corrin A, Taylor RJ, Griffiths CE, Main CJ. Attentional bias for psoriasis-specific and psychosocial threat in patients with psoriasis. Journal of Behavioral Medicine. 2003;26:211–224. doi: 10.1023/A:1023408503807. [DOI] [PubMed] [Google Scholar]
- Friedman HS, Tucker JS, Tomlinson-Keasey C, Schwartz JE, Wingard DL, Criqui MH. Does childhood personality predict longevity? Journal of Personality and Social Psychology. 1993;65:176–185. doi: 10.1037/0022-3514.65.1.176. [DOI] [PubMed] [Google Scholar]
- Friese M, Binder J, Luechinger R, Boesiger P, Rasch B. Suppressing emotions impairs subsequent stroop performance and reduces prefrontal brain activation. PloS One. 2013;8(4):e60385. doi: 10.1371/journal.pone.0060385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frydecka D, Misiak B, Pawlak-Adamska E, Karabon L, Tomkiewicz A, Sedlaczek P, … Beszłej JA. Interleukin-6: The missing element of the neurocognitive deterioration in schizophrenia? The focus on genetic underpinnings, cognitive impairment and clinical manifestation. European Archives of Psychiatry and Clinical Neuroscience. 2015;265:449–459. doi: 10.1007/s00406-014-0533-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gailliot MT, Baumeister RF. The physiology of willpower: Linking blood glucose to self-control. Personality and Social Psychology Review. 2007;11:303–327. doi: 10.1177/1088868307303030. [DOI] [PubMed] [Google Scholar]
- Garcia-Oscos F, Peña D, Housini M, Cheng D, Lopez D, Borland MS, … Atzori M. Vagal nerve stimulation blocks interleukin 6-dependent synaptic hyperexcitability induced by lipopolysaccharide-induced acute stress in the rodent prefrontal cortex. Brain, Behavior, and Immunity. 2015;43:149–158. doi: 10.1016/j.bbi.2014.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gianaros PJ, Marsland AL, Kuan DCH, Schirda BL, Jennings JR, Sheu LK, … Manuck SB. An inflammatory pathway links atherosclerotic cardiovascular disease risk to neural activity evoked by the cognitive regulation of emotion. Biological Psychiatry. 2014;75:738–745. doi: 10.1016/j.biopsych.2013.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goto T, Kusumi T. How can reward contribute to efficient self-control? reinforcement of task-defined responses diminishes ego-depletion. Motivation and Emotion. 2013;37:726–737. doi: 10.1007/s11031-013-9356-3. [DOI] [Google Scholar]
- Grigoleit JS, Kullmann JS, Wolf OT, Hammes F, Wegner A, Jablonowski S, … Schedlowski M. Dose-dependent effects of endotoxin on neurobehavioral functions in humans. PloS One. 2011;6(12):e28330. doi: 10.1371/journal.pone.0028330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grigoleit JS, Oberbeck JR, Lichte P, Kobbe P, Wolf OT, Montag T, … Schedlowski M. Lipopolysaccharide-induced experimental immune activation does not impair memory functions in humans. Neurobiology of Learning and Memory. 2010;94:561–567. doi: 10.1016/j.nlm.2010.09.011. [DOI] [PubMed] [Google Scholar]
- Haba R, Shintani N, Onaka Y, Wang H, Takenaga R, Hayata A, … Hashimoto H. Lipopolysaccharide affects exploratory behaviors toward novel objects by impairing cognition and/or motivation in mice: Possible role of activation of the central amygdala. Behavioural Brain Research. 2012;228:423–431. doi: 10.1016/j.bbr.2011.12.027. [DOI] [PubMed] [Google Scholar]
- Hagger MS, Chatzisarantis NLD. The sweet taste of success: The presence of glucose in the oral cavity moderates the depletion of self-control resources. Personality & Social Psychology Bulletin. 2013;39:28–42. doi: 10.1177/0146167212459912. [DOI] [PubMed] [Google Scholar]
- Hagger MS, Chatzisarantis NL, Alberts H, Anggono CO, Batailler C, Birt A, … Zwienenberg M. A multilab preregistered replication of the ego-depletion effect. Perspectives on Psychological Science. 2015;11:546–573. doi: 10.1177/1745691616652873. [DOI] [PubMed] [Google Scholar]
- Hagger MS, Wood C, Stiff C, Chatzisarantis NLD. Ego depletion and the strength model of self-control: A meta-analysis. Psychological Bulletin. 2010;136:495–525. doi: 10.1037/a0019486. [DOI] [PubMed] [Google Scholar]
- Hajeer AH, Hutchinson IV. Influence of TNFα gene polymorphisms on TNFα production and disease. Human Immunology. 2001;62:1191–1199. doi: 10.1016/S0198-8859(01)00322-6. [DOI] [PubMed] [Google Scholar]
- Haley WE, Levine EG, Brown SL, Bartolucci AA. Stress, appraisal, coping, and social support as predictors of adaptational outcome among dementia caregivers. Psychology and Aging. 1987;2:323–330. doi: 10.1037/0882-7974.2.4.323. [DOI] [PubMed] [Google Scholar]
- Hammen C, Kim EY, Eberhart NK, Brennan PA. Chronic and acute stress and the prediction of major depression in women. Depression and Anxiety. 2009;26:718–723. doi: 10.1002/da.20571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hannestad J, DellaGioia N, Ortiz N, Pittman B, Bhagwagar Z. Citalopram reduces endotoxin-induced fatigue. Brain, Behavior, and Immunity. 2011;25:256–259. doi: 10.1016/j.bbi.2010.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hannestad J, Subramanyam K, DellaGioia N, Planeta-Wilson B, Weinzimmer D, Pittman B, Carson RE. Glucose metabolism in the insula and cingulate is affected by systemic inflammation in humans. Journal of Nuclear Medicine. 2012;53:601–607. doi: 10.2967/jnumed.111.097014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison NA, Brydon L, Walker C, Gray MA, Steptoe A, Critchley HD. Inflammation causes mood changes through alterations in subgenual cingulate activity and mesolimbic connectivity. Biological Psychiatry. 2009;66:407–414. doi: 10.1016/j.biopsych.2009.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison NA, Brydon L, Walker C, Gray MA, Steptoe A, Dolan RJ, Critchley HD. Neural origins of human sickness in interoceptive responses to inflammation. Biological Psychiatry. 2009;66:415–422. doi: 10.1016/j.biopsych.2009.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison NA, Cercignani M, Voon V, Critchley HD. Effects of inflammation on hippocampus and substantia nigra responses to novelty in healthy human participants. Neuropsychopharmacology. 2015;40:831–838. doi: 10.1038/npp.2014.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison NA, Doeller CF, Voon V, Burgess N, Critchley HD. Peripheral inflammation acutely impairs human spatial memory via actions on medial temporal lobe glucose metabolism. Biological Psychiatry. 2014;76:585–593. doi: 10.1016/j.biopsych.2014.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heatherton TF, Wagner DD. Cognitive neuroscience of self-regulation failure. Trends in Cognitive Sciences. 2011;15:132–139. doi: 10.1016/j.tics.2010.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedgcock WM, Vohs KD, Rao AR. Reducing self-control depletion effects through enhanced sensitivity to implementation: Evidence from fMRI and behavioral studies. Journal of Consumer Psychology. 2012;22:486–495. doi: 10.1016/j.jcps.2012.05.008. [DOI] [Google Scholar]
- Henckens MJ, van Wingen GA, Joëls M, Fernández G. Time-dependent corticosteroid modulation of prefrontal working memory processing. Proceedings of the National Academy of Sciences. 2011;108:5801–5806. doi: 10.1073/pnas.1019128108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heyder K, Suchan B, Daum I. Cortico-subcortical contributions to executive control. Acta Psychologica. 2004;115:271–289. doi: 10.1016/j.actpsy.2003.12.010. [DOI] [PubMed] [Google Scholar]
- Hofmann W, Luhmann M, Fisher RR, Vohs KD, Baumeister RF. Yes, but are they happy? Effects of trait self-control on affective well-being and life satisfaction. Journal of Personality. 2014;82:265–277. doi: 10.1111/jopy.12050. [DOI] [PubMed] [Google Scholar]
- Hofmann W, Schmeichel BJ, Baddeley AD. Executive functions and self-regulation. Trends in Cognitive Sciences. 2012;16:174–180. doi: 10.1016/j.tics.2012.01.006. [DOI] [PubMed] [Google Scholar]
- Hopkins SJ, Rothwell NJ. Cytokines and the nervous system I: Expression and recognition. Trends in Neurosciences. 1995;18:83–88. doi: 10.1016/0166-2236(95)80029-2. [DOI] [PubMed] [Google Scholar]
- Inzlicht M, Gervais WM, Berkman ET. Bias-correction techniques alone cannot determine whether ego depletion is different from zero: Commentary on Carter, Kofler, Forster, & McCullough, 2015. 2015 doi: 10.2139/ssrn.2659409. [DOI] [Google Scholar]
- Inzlicht M, Gutsell JN. Running on empty: Neural signals for self-control failure. Psychological Science. 2007;18:933–937. doi: 10.1111/j.1467-9280.2007.02004.x. [DOI] [PubMed] [Google Scholar]
- Inzlicht M, Schmeichel BJ. What is ego depletion? Toward a mechanistic revision of the resource model of self-control. Perspectives on Psychological Science. 2012;7:450–463. doi: 10.1177/1745691612454134. [DOI] [PubMed] [Google Scholar]
- Irwin MR, Cole SW. Reciprocal regulation of the neural and innate immune systems. Nature Reviews Immunology. 2011;11:625–632. doi: 10.1038/nri3042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irwin MR, Slavich GM. Psychoneuroimmunology. In: Cacioppo JT, Tassinary LG, Berntson GG, editors. Handbook of psychophysiology, fourth edition. New York: Cambridge University Press; 2017. [Google Scholar]
- Jefferson AL, Massaro JM, Beiser AS, Seshadri S, Larson MG, Wolf PA, … Benjamin EJ. Inflammatory markers and neuropsychological functioning: The Framingham Heart Study. Neuroepidemiology. 2011;37:1295–1308. doi: 10.1159/000328864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Job V, Walton GM, Bernecker K, Dweck CS. Beliefs about willpower determine the impact of glucose on self-control. Proceedings of the National Academy of Sciences of the United States of America. 2013;110:14837–14842. doi: 10.1073/pnas.1313475110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- John-Henderson NA, Rheinschmidt ML, Mendoza-Denton R, Francis DD. Performance and inflammation outcomes are predicted by different facets of SES under stereotype threat. Social Psychological and Personality Science. 2014;5:301–309. doi: 10.1177/1948550613494226. [DOI] [Google Scholar]
- Juengling FD, Ebert D, Gut O, Engelbrecht MA, Rasenack J, Nitzsche EU, … Lieb K. Prefrontal cortical hypometabolism during low-dose interferon alpha treatment. Psychopharmacology. 2000;152:383–389. doi: 10.1007/s002130000549. [DOI] [PubMed] [Google Scholar]
- Klein WM, Shepperd JA, Suls J, Rothman AJ, Croyle RT. Realizing the promise of social psychology in improving public health. Personality and Social Psychology Review. 2015;19:77–92. doi: 10.1177/1088868314539852. [DOI] [PubMed] [Google Scholar]
- Knudsen EI. Sensitive periods in the development of the brain and behavior. Journal of Cognitive Neuroscience. 2004;16:1412–1425. doi: 10.1162/0898929042304796. [DOI] [PubMed] [Google Scholar]
- Koenig HG, Cohen HJ, George LK, Hays JC, Larson DB, Blazer DG. Attendance at religious services, interleukin-6, and other biological parameters of immune function in older adults. International Journal of Psychiatry in Medicine. 1997;27:233–250. doi: 10.2190/40NF-Q9Y2-0GG7-4WH6. [DOI] [PubMed] [Google Scholar]
- Kröz M, Reif M, Büssing A, Zerm R, Feder G, Bockelbrink A, … Girke M. Does self-regulation and autonomic regulation have an influence on survival in breast and colon carcinoma patients? Results of a prospective outcome study. Health and Quality of Life Outcomes. 2011;9:85. doi: 10.1186/1477-7525-9-85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaGrange TC, Silverman RA. Low self-control and opportunity: Testing the general theory of crime as an explanation for gender differences in delinquency. Criminology. 1999;37:41–72. doi: 10.1111/j.1745-9125.1999.tb00479.x. [DOI] [Google Scholar]
- Lane RD, Wager TD, O’Connor MF, Irwin MR, Wellisch DK. When grief heats up: Pro-inflammatory cytokines predict regional brain activation. NeuroImage. 2009;47:891–896. doi: 10.1016/j.neuroimage.2009.05.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lobstein T, Baur L, Uauy R. Obesity in children and young people: A crisis in public health. Obesity Reviews. 2004;5(s1):4–85. doi: 10.1111/j.1467-789X.2004.00133.x. [DOI] [PubMed] [Google Scholar]
- Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, … Kipnis J. Structural and functional features of central nervous system lymphatic vessels. Nature. 2015;523:337–341. doi: 10.1038/nature14432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madigan RJ, Bollenbach AK. The effects of induced mood on irrational thoughts and views of the world. Cognitive Therapy and Research. 1986;10:547–562. doi: 10.1007/BF01177818. [DOI] [Google Scholar]
- Maier SF, Goehler LE, Fleshner M, Watkins LR. The role of the vagus nerve in cytokine-to-brain communication. Annals of the New York Academy of Sciences. 1998;840:289–300. doi: 10.1111/j.1749-6632.1998.tb09569.x. [DOI] [PubMed] [Google Scholar]
- Maier SF, Watkins LR. Cytokines for psychologists: Implications of bidirectional immune-to-brain communication for understanding behavior, mood, and cognition. Psychological Review. 1998;105:83–107. doi: 10.1037/0033-295X.105.1.83. [DOI] [PubMed] [Google Scholar]
- Mansouri FA, Tanaka K, Buckley MJ. Conflict-induced behavioural adjustment: a clue to the executive functions of the prefrontal cortex. Nature Reviews Neuroscience. 2009;10:141–152. doi: 10.1038/nrn2538. [DOI] [PubMed] [Google Scholar]
- Marek L, Placek W, Borkowska A. Zaburzenia funkcji poznawczych i temperament afektywny u pacjentów z łuszczycą [Cognitive impairment and affective temperament in patients with psoriasis] Przegląd Dermatologiczny. 2011;98:483–490. Retrieved from http://www.termedia.pl/ [Google Scholar]
- Marioni RE, Deary IJ, Murray GD, Fowkes FGR, Price JF. Associations between polymorphisms in five inflammation-related genes and cognitive ability in older persons. Genes, Brain, and Behavior. 2010;9:348–352. doi: 10.1111/j.1601-183X.2010.00565.x. [DOI] [PubMed] [Google Scholar]
- Markou A, De La Garza R. Endotoxin- or pro-inflammatory cytokine-induced sickness behavior as an animal model of depression: Focus on anhedonia. Neuroscience & Biobehavioral Reviews. 2005;29:761–770. doi: 10.1016/j.neubiorev.2005.03.016. [DOI] [PubMed] [Google Scholar]
- Marsland AL, Gianaros PJ, Abramowitch SM, Manuck SB, Hariri AR. Interleukin-6 covaries inversely with hippocampal grey matter volume in middle-aged adults. Biological Psychiatry. 2008;64:484–490. doi: 10.1016/j.biopsych.2008.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsland AL, Gianaros PJ, Kuan DCH, Sheu LK, Krajina K, Manuck SB. Brain morphology links systemic inflammation to cognitive function in midlife adults. Brain, Behavior, and Immunity. 2015;48:195–204. doi: 10.1016/j.bbi.2015.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marsland AL, Krajina K, Gianaros PJ. 64. Systemic inflammation and the integrity of brain white matter tracts. Brain, Behavior, and Immunity. 2012;26(Supplement 1):S18. doi: 10.1016/j.bbi.2012.07.088. [DOI] [Google Scholar]
- Marsland AL, Petersen KL, Sathanoori R, Muldoon MF, Neumann SA, Ryan C, … Manuck SB. Interleukin-6 covaries inversely with cognitive performance among middle-aged community volunteers. Psychosomatic Medicine. 2006;68:895–903. doi: 10.1097/01.psy.0000238451.22174.92. [DOI] [PubMed] [Google Scholar]
- Mausbach BT, von Känel R, Roepke SK, Moore R, Patterson TL, Mills PJ, … Grant I. Self-efficacy buffers the relationship between dementia caregiving stress and circulating concentrations of the proinflammatory cytokine interleukin-6. The American Journal of Geriatric Psychiatry. 2011;19:64–71. doi: 10.1097/JGP.0b013e3181df4498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCullough M, Willoughby BLB. Religion, self-regulation, and self-control: Associations, explanations and implications. Psychological Bulletin. 2009;135:69–93. doi: 10.1037/a0014213. [DOI] [PubMed] [Google Scholar]
- McDade TW. Early environments and the ecology of inflammation. Proceedings of the National Academy of Sciences of the United States of America. 2012;109:17281–17288. doi: 10.1073/pnas.1202244109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDade TW, Hoke M, Borja JB, Adair LS, Kuzawa C. Do environments in infancy moderate the association between stress and inflammation in adulthood? Initial evidence from a birth cohort in the Philippines. Brain, Behavior, and Immunity. 2013;31:23–30. doi: 10.1016/j.bbi.2012.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454:428–435. doi: 10.1038/nature07201. [DOI] [PubMed] [Google Scholar]
- Meichenbaum DH, Goodman J. Training impulsive children to talk to themselves: A means of developing self-control. Journal of Abnormal Psychology. 1971;77(2):115–126. doi: 10.1037/h0030773. [DOI] [PubMed] [Google Scholar]
- Meyers CA, Abbruzzese JL. Cognitive functioning in cancer patients: Effect of previous treatment. Neurology. 1992;42:434–434. doi: 10.1212/WNL.42.2.434. [DOI] [PubMed] [Google Scholar]
- Meyers CA, Valentine AD, Wong FC, Leeds NE. Reversible neurotoxicity of interleukin-2 and tumor necrosis factor: Correlation of SPECT with neuropsychological testing. The Journal of Neuropsychiatry and Clinical Neurosciences. 1994;6:285–288. doi: 10.1176/jnp.6.3.285. Retrieved from http://neuro.psychiatryonline.org/ [DOI] [PubMed] [Google Scholar]
- Miller AH, Capuron L, Raison CL. Immunologic influences on emotion regulation. Clinical Neuroscience Research. 2005;4:325–333. doi: 10.1016/j.cnr.2005.03.010. [DOI] [Google Scholar]
- Miller G, Chen E, Cole SW. Health psychology: Developing biologically plausible models linking the social world and physical health. Annual Review of Psychology. 2009;60:501–524. doi: 10.1146/annurev.psych.60.110707.163551. [DOI] [PubMed] [Google Scholar]
- Miyake A, Friedman NP, Emerson MJ, Witzki AH, Howerter A, Wager TD. The unity and diversity of executive functions and their contributions to complex “frontal lobe” tasks: a latent variable analysis. Cognitive Psychology. 2000;41:49–100. doi: 10.1006/cogp.1999.0734. [DOI] [PubMed] [Google Scholar]
- Moffitt TE, Arseneault L, Belsky D, Dickson N, Hancox RJ, Harrington H, … Caspi A. A gradient of childhood self-control predicts health, wealth, and public safety. Proceedings of the National Academy of Sciences of the United States of America. 2011;108:2693–2698. doi: 10.1073/pnas.1010076108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molden DC, Hui CM, Scholer AA, Meier BP, Noreen EE, D’Agostino PR, Martin V. Motivational versus metabolic effects of carbohydrates on self-control. Psychological Science. 2012;23:1137–1144. doi: 10.1177/0956797612439069. [DOI] [PubMed] [Google Scholar]
- Mooijaart SP, Sattar N, Trompet S, Lucke J, Stott DJ, Ford I, … de Craen AJM. Circulating interleukin-6 concentration and cognitive decline in old age: The PROSPER study. Journal of Internal Medicine. 2013;274:77–85. doi: 10.1111/joim.12052. [DOI] [PubMed] [Google Scholar]
- Muraven M. Practicing self-control lowers the risk of smoking lapse. Psychology of Addictive Behaviors. 2010;24:446–452. doi: 10.1037/a0018545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muraven M, Baumeister RF. Self-regulation and depletion of limited resources: Does self-control resemble a muscle? Psychological Bulletin. 2000;126:247–259. doi: 10.1037/0033-2909.126.2.247. [DOI] [PubMed] [Google Scholar]
- Muraven M, Tice DM, Baumeister RF. Self-control as a limited resource: Regulatory depletion patterns. Journal of Personality and Social Psychology. 1998;74:774–789. doi: 10.1037/0022-3514.74.3.774. [DOI] [PubMed] [Google Scholar]
- Muraven M, Slessareva E. Mechanisms of self-control failure: Motivation and limited resources. Personality & Social Psychology Bulletin. 2003;29:894–906. doi: 10.1177/0146167203029007008. [DOI] [PubMed] [Google Scholar]
- Murphy KM. Janeway’s immunobiology. 8. Garland Science; 2014. [Google Scholar]
- Ng TW, Eby LT, Sorensen KL, Feldman DC. Predictors of objective and subjective career success: A meta-analysis. Personnel Psychology. 2005;58:367–408. doi: 10.1111/j.1744-6570.2005.00515.x. [DOI] [Google Scholar]
- Nusslock R, Miller GE. Early-life adversity and physical and emotional health across the lifespan: A neuroimmune network hypothesis. Biological Psychiatry. 2015 doi: 10.1016/j.biopsych.2015.05.017. Advance online publication. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oades RD, Dauvermann MR, Schimmelmann BG, Schwarz MJ, Myint AM. Attention-deficit hyperactivity disorder (ADHD) and glial integrity: S100B, cytokines and kynurenine metabolism--effects of medication. Behavioral and Brain Functions. 2010;6:29. doi: 10.1186/1744-9081-6-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oaten M, Cheng K. Academic examination stress impairs self–control. Journal of Social and Clinical Psychology. 2005;24:254–279. doi: 10.1521/jscp.24.2.254.62276. [DOI] [Google Scholar]
- Papageorgiou C, Panagiotakos DB, Pitsavos C, Tsetsekou E, Kontoangelos K, Stefanadis C, Soldatos C. Association between plasma inflammatory markers and irrational beliefs; the ATTICA epidemiological study. Progress in Neuro-Psychopharmacology and Biological Psychiatry. 2006;30:1496–1503. doi: 10.1016/j.pnpbp.2006.05.018. [DOI] [PubMed] [Google Scholar]
- Pariante CM. The proinflammatory cytokine, interleukin-1β, reduces glucocorticoid receptor translocation and function. Endocrinology. 1999;140:4359–4366. doi: 10.1210/en.140.9.4359. [DOI] [PubMed] [Google Scholar]
- Pavol MA, Meyers CA, Rexer JL, Valentine AD, Mattis PJ, Talpaz M. Pattern of neurobehavioral deficits associated with interferon alfa therapy for leukemia. Neurology. 1995;45:947–950. doi: 10.1212/WNL.45.5.947. [DOI] [PubMed] [Google Scholar]
- Persson J, Larsson A, Reuter-Lorenz PA. Imaging fatigue of interference control reveals the neural basis of executive resource depletion. Journal of Cognitive Neuroscience. 2013;25:338–351. doi: 10.1162/jocn_a_00321. [DOI] [PubMed] [Google Scholar]
- Poh KW, Yeo JF, Stohler CS, Ong WY. Comprehensive gene expression profiling in the prefrontal cortex links immune activation and neutrophil infiltration to antinociception. The Journal of Neuroscience. 2012;32:35–45. doi: 10.1523/JNEUROSCI.2389-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porath CL, Bateman TS. Self-regulation: From goal orientation to job performance. Journal of Applied Psychology. 2006;91:185–192. doi: 10.1037/0021-9010.91.1.185. [DOI] [PubMed] [Google Scholar]
- Prossin AR, Koch AE, Campbell PL, McInnis MG, Zalcman SS, Zubieta JK. Association of plasma interleukin-18 levels with emotion regulation and μ-opioid neurotransmitter function in major depression and healthy volunteers. Biological Psychiatry. 2011;69:808–812. doi: 10.1016/j.biopsych.2010.10.014. [DOI] [PubMed] [Google Scholar]
- Raio CM, Orederu TA, Palazzolo L, Shurick AA, Phelps EA. Cognitive emotion regulation fails the stress test. Proceedings of the National Academy of Sciences of the United States of America. 2013;110:15139–15144. doi: 10.1073/pnas.1305706110. http://doi.org/10.1073/pnas.1305706110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raison CL, Capuron L, Miller AH. Cytokines sing the blues: Inflammation and the pathogenesis of depression. Trends in Immunology. 2006;27:24–31. doi: 10.1016/j.it.2005.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramlawi B, Rudolph JL, Mieno S, Feng J, Boodhwani M, Khabbaz K, … Sellke FW. C-reactive protein and inflammatory response associated to neurocognitive decline following cardiac surgery. Surgery. 2006;140:221–226. doi: 10.1016/j.surg.2006.03.007. [DOI] [PubMed] [Google Scholar]
- Reichenberg A, Yirmiya R, Schuld A, Kraus T, Haack M, Morag A, Pollmächer T. Cytokine-associated emotional and cognitive disturbances in humans. Archives of General Psychiatry. 2001;58:445–452. doi: 10.1001/archpsyc.58.5.445. [DOI] [PubMed] [Google Scholar]
- Richeson JA, Baird AA, Gordon HL, Heatherton TF, Wyland CL, Trawalter S, Shelton JN. An fMRI investigation of the impact of interracial contact on executive function. Nature Neuroscience. 2003;6:1323–1328. doi: 10.1038/nn1156. [DOI] [PubMed] [Google Scholar]
- Riehemann K, Behnke B, Schulze-Osthoff K. Plant extracts from stinging nettle (Urtica dioica), an antirheumatic remedy, inhibit the proinflammatory transcription factor NF-κB. FEBS Letters. 1999;442:89–94. doi: 10.1016/S0014-5793(98)01622-6. [DOI] [PubMed] [Google Scholar]
- Rigoni D, Kühn S, Gaudino G, Sartori G, Brass M. Reducing self-control by weakening belief in free will. Consciousness and Cognition. 2012;21:1482–1490. doi: 10.1016/j.concog.2012.04.004. [DOI] [PubMed] [Google Scholar]
- Rigoni D, Kühn S, Sartori G, Brass M. Inducing disbelief in free will alters brain correlates of preconscious motor preparation: The brain minds whether we believe in free will or not. Psychological Science. 2011;22:613–618. doi: 10.1177/0956797611405680. [DOI] [PubMed] [Google Scholar]
- Rigoni D, Wilquin H, Brass M, Burle B. When errors do not matter: Weakening belief in intentional control impairs cognitive reaction to errors. Cognition. 2013;127:264–269. doi: 10.1016/j.cognition.2013.01.009. [DOI] [PubMed] [Google Scholar]
- Rothwell NJ, Hopkins SJ. Cytokines and the nervous system II: Actions and mechanisms of action. Trends in Neurosciences. 1995;18:130–136. doi: 10.1016/0166-2236(95)93890-A. [DOI] [PubMed] [Google Scholar]
- Sasayama D, Hori H, Teraishi T, Hattori K, Ota M, Matsuo J, … Kunugi H. Association of interleukin-1β genetic polymorphisms with cognitive performance in elderly females without dementia. Journal of Human Genetics. 2011;56:613–616. doi: 10.1038/jhg.2011.56. [DOI] [PubMed] [Google Scholar]
- Satizabal CL, Zhu YC, Mazoyer B, Dufouil C, Tzourio C. Circulating IL-6 and CRP are associated with MRI findings in the elderly: The 3C-dijon study. Neurology. 2012;78:720–727. doi: 10.1212/WNL.0b013e318248e50f. [DOI] [PubMed] [Google Scholar]
- Saxby BK, Harrington F, McKeith IG, Wesnes K, Ford GA. Effects of hypertension on attention, memory, and executive function in older adults. Health Psychology. 2003;22:587–591. doi: 10.1037/0278-6133.22.6.587. [DOI] [PubMed] [Google Scholar]
- Scheier MF, Carver CS, Armstrong GH. Behavioral self-regulation, health, and illness. In: Baum A, Revenson TA, Singer J, editors. Handbook of Health Psychology. 2. Psychology Press; 2012. pp. 79–112. [Google Scholar]
- Schillerstrom JE, Horton MS, Royall DR. The impact of medical illness on executive function. Psychosomatics. 2005;46:508–516. doi: 10.1176/appi.psy.46.6.508. [DOI] [PubMed] [Google Scholar]
- Schmidt L, Lebreton M, Cléry-Melin ML, Daunizeau J, Pessiglione M. Neural mechanisms underlying motivation of mental versus physical effort. PLoS Biology. 2012;10(2):e1001266. doi: 10.1371/journal.pbio.1001266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoofs D, Pabst S, Brand M, Wolf OT. Working memory is differentially affected by stress in men and women. Behavioural Brain Research. 2013;241:144–153. doi: 10.1016/j.bbr.2012.12.004. [DOI] [PubMed] [Google Scholar]
- Schwabe L, Höffken O, Tegenthoff M, Wolf OT. Stress-induced enhancement of response inhibition depends on mineralocorticoid receptor activation. Psychoneuroendocrinology. 2013;38:2319–2326. doi: 10.1016/j.psyneuen.2013.05.001. [DOI] [PubMed] [Google Scholar]
- Schwarzer R. Self-regulatory processes in the adoption and maintenance of health behaviors. Journal of Health Psychology. 1999;4:115–127. doi: 10.1177/135910539900400208. [DOI] [PubMed] [Google Scholar]
- Shields GS, Bonner JC, Moons WG. Does cortisol influence core executive functions? A meta-analysis of acute cortisol administration effects on working memory, inhibition, and set-shifting. Psychoneuroendocrinology. 2015;58:91–103. doi: 10.1016/j.psyneuen.2015.04.017. [DOI] [PubMed] [Google Scholar]
- Shields GS, Kuchenbecker SY, Pressman SD, Sumida KD, Slavich GM. Better cognitive control of emotional information is associated with reduced pro-inflammatory cytokine reactivity to emotional stress. Stress. 2016;19:63–68. doi: 10.3109/10253890.2015.1121983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shields GS, Lam JC, Trainor BC, Yonelinas AP. Exposure to acute stress enhances decision-making competence: Evidence for the role of DHEA. Psychoneuroendocrinology. 2016;67:51–60. doi: 10.1016/j.psyneuen.2016.01.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shields GS, Moons WG, Tewell CA, Yonelinas AP. The effect of negative affect on cognition: Anxiety, not anger, impairs executive function. Emotion. 2016;6:792–797. doi: 10.1037/emo0000151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simons DJ, Boot WR, Charness N, Gathercole SE, Chabris CF, Hambrick DZ, Stine-Morrow EA. Do “brain-training” programs work? Psychological Science in the Public Interest. 2016;17:103–186. doi: 10.1177/1529100616661983. [DOI] [PubMed] [Google Scholar]
- Simpson EEA, Hodkinson CF, Maylor EA, McCormack JM, Rae G, Strain S, … Wallace JMW. Intracellular cytokine production and cognition in healthy older adults. Psychoneuroendocrinology. 2013;38:2196–2208. doi: 10.1016/j.psyneuen.2013.04.007. [DOI] [PubMed] [Google Scholar]
- Slavich GM. Understanding inflammation, its regulation, and relevance for health: A top scientific and public priority. Brain, Behavior, and Immunity. 2015;45:13–14. doi: 10.1016/j.bbi.2014.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavich GM. Life stress and health: A review of conceptual issues and recent findings. Teaching of Psychology. 2016;43:346–355. doi: 10.1177/0098628316662768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavich GM, Cole SW. The emerging field of human social genomics. Clinical Psychological Science. 2013;1:331–348. doi: 10.1177/2167702613478594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavich GM, Irwin MR. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychological Bulletin. 2014;140:774–815. doi: 10.1037/a0035302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavich GM, O’Donovan A, Epel ES, Kemeny ME. Black sheep get the blues: A psychobiological model of social rejection and depression. Neuroscience and Biobehavioral Reviews. 2010;35:39–45. doi: 10.1016/j.neubiorev.2010.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavich GM, Way BM, Eisenberger NI, Taylor SE. Neural sensitivity to social rejection is associated with inflammatory responses to social stress. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:14817–14822. doi: 10.1073/pnas.1009164107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sparkman NL, Buchanan JB, Heyen JRR, Chen J, Beverly JL, Johnson RW. Interleukin-6 facilitates lipopolysaccharide-induced disruption in working memory and expression of other proinflammatory cytokines in hippocampal neuronal cell layers. The Journal of Neuroscience. 2006;26:10709–10716. doi: 10.1523/JNEUROSCI.3376-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprague J, Verona E, Kalkhoff W, Kilmer A. Moderators and mediators of the stress-aggression relationship: Executive function and state anger. Emotion. 2011;11:61–73. doi: 10.1037/a0021788. [DOI] [PubMed] [Google Scholar]
- Stajkovic AD, Luthans F. Self-efficacy and work-related performance: A meta-analysis. Psychological Bulletin. 1998;124:240–261. doi: 10.1037/0033-2909.124.2.240. [DOI] [Google Scholar]
- Steptoe A, Hamer M, Chida Y. The effects of acute psychological stress on circulating inflammatory factors in humans: A review and meta-analysis. Brain, Behavior, and Immunity. 2007;21:901–912. doi: 10.1016/j.bbi.2007.03.011. [DOI] [PubMed] [Google Scholar]
- Sultan AJ, Joireman J, Sprott DE. Building consumer self-control: The effect of self-control exercises on impulse buying urges. Marketing Letters. 2011;23:61–72. doi: 10.1007/s11002-011-9135-4. [DOI] [Google Scholar]
- Tangney JP, Baumeister RF, Boone AL. High self-control predicts good adjustment, less pathology, better grades, and interpersonal success. Journal of Personality. 2004;72:271–324. doi: 10.1111/j.0022-3506.2004.00263.x. [DOI] [PubMed] [Google Scholar]
- Teunissen C, van Boxtel MP, Bosma H, Bosmans E, Delanghe J, De Bruijn C, … de Vente J. Inflammation markers in relation to cognition in a healthy aging population. Journal of Neuroimmunology. 2003;134:142–150. doi: 10.1016/S0165-5728(02)00398-3. [DOI] [PubMed] [Google Scholar]
- Thayer JF, Sternberg EM. Neural aspects of immunomodulation: Focus on the vagus nerve. Brain, Behavior, and Immunity. 2010;24:1223–1228. doi: 10.1016/j.bbi.2010.07.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thoman DB, Smith JL, Silvia PJ. The resource replenishment function of interest. Social Psychological and Personality Science. 2011;2:592–599. doi: 10.1177/1948550611402521. [DOI] [Google Scholar]
- Tice DM, Bratslavsky E, Baumeister RF. Emotional distress regulation takes precedence over impulse control: If you feel bad, do it! Journal of Personality and Social Psychology. 2001;80:53–67. doi: 10.1037/0022-3514.80.1.53. [DOI] [PubMed] [Google Scholar]
- Tilg H, Moschen AR. Adipocytokines: Mediators linking adipose tissue, inflammation and immunity. Nature Reviews Immunology. 2006;6:772–783. doi: 10.1038/nri1937. [DOI] [PubMed] [Google Scholar]
- Tobinick EL, Gross H. Rapid cognitive improvement in Alzheimer’s disease following perispinal etanercept administration. Journal of Neuroinflammation. 2008;5:2. doi: 10.1186/1742-2094-5-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toussaint L, Shields GS, Dorn G, Slavich GM. Effects of lifetime stress exposure on mental and physical health in young adulthood: How stress degrades and forgiveness protects health. Journal of Health Psychology. 2016;21:1004–1014. doi: 10.1177/1359105314544132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trollor JN, Smith E, Agars E, Kuan SA, Baune BT, Campbell L, … Sachdev P. The association between systemic inflammation and cognitive performance in the elderly: The Sydney Memory and Ageing Study. Age. 2012;34:1295–1308. doi: 10.1007/s11357-011-9301-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trompet S, de Craen AJM, Slagboom P, Shepherd J, Blauw GJ, Murphy MB, … Westendorp RGJ. Genetic variation in the interleukin-1 β-converting enzyme associates with cognitive function. The PROSPER study. Brain. 2008;131:1069–1077. doi: 10.1093/brain/awn023. [DOI] [PubMed] [Google Scholar]
- Tsai SJ, Hong CJ, Liu ME, Hou SJ, Yen FC, Hsieh CH, Liou YJ. Interleukin-1 β (C-511T) genetic polymorphism is associated with cognitive performance in elderly males without dementia. Neurobiology of Aging. 2010;31:1950–1955. doi: 10.1016/j.neurobiolaging.2008.10.002. [DOI] [PubMed] [Google Scholar]
- Tu PC, Su TP, Huang CC, Yang AC, Yeh HL, Hong CJ, … Tsai S-J. Interleukin-1 β C-511T polymorphism modulates functional connectivity of anterior midcingulate cortex in non-demented elderly Han males. Brain Structure and Function. 2013;219:61–69. doi: 10.1007/s00429-012-0484-4. [DOI] [PubMed] [Google Scholar]
- Teucher T, Obertreis B, Ruttkowski T, Schmitz H. Cytokine secretion in whole blood of healthy subjects following oral administration of Urtica dioica L. plant extract. Arzneimittelforschung. 1996;46:906–910. [PubMed] [Google Scholar]
- van den Berg E, De Craen AJM, Biessels GJ, Gussekloo J, Westendorp RGJ. The impact of diabetes mellitus on cognitive decline in the oldest of the old: A prospective population-based study. Diabetologia. 2006;49:2015–2023. doi: 10.1007/s00125-006-0333-1. [DOI] [PubMed] [Google Scholar]
- van den Boogaard M, Ramakers BP, van Alfen N, van der Werf SP, Fick WF, Hoedemaekers CWE, … Pickkers P. Endotoxemia-induced inflammation and the effect on the human brain. Critical Care. 2010;14(3):R81. doi: 10.1186/cc9001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verbeken S, Braet C, Goossens L, van der Oord S. Executive function training with game elements for obese children: A novel treatment to enhance self-regulatory abilities for weight-control. Behaviour Research and Therapy. 2013;51:290–299. doi: 10.1016/j.brat.2013.02.006. [DOI] [PubMed] [Google Scholar]
- Vicario A, Martinez CD, Baretto D, Casale AD, Nicolosi L. Hypertension and cognitive decline: Impact on executive function. The Journal of Clinical Hypertension. 2005;7:598–604. doi: 10.1111/j.1524-6175.2005.04498.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vohs KD, Baumeister RF, editors. Handbook of self-regulation: Research, theory, and applications. Guilford Press; 2011. [Google Scholar]
- Vohs KD, Baumeister RF, Ciarocco NJ. Self-regulation and self-presentation: regulatory resource depletion impairs impression management and effortful self-presentation depletes regulatory resources. Journal of Personality and Social Psychology. 2005;88:632–657. doi: 10.1037/0022-3514.88.4.632. [DOI] [PubMed] [Google Scholar]
- Vohs KD, Baumeister RF, Mead N, Ramanathan S, Hofmann W, Schmeichel BJ. Engaging in self-control heightens urges and feelings. 2012 Unpublished raw data. [Google Scholar]
- Vohs KD, Baumeister RF, Schmeichel BJ. Motivation, personal beliefs, and limited resources all contribute to self-control. Journal of Experimental Social Psychology. 2012;48:943–947. doi: 10.1016/j.jesp.2012.03.002. [DOI] [Google Scholar]
- Vohs KD, Heatherton TF. Self-regulatory failure: A resource-depletion approach. Psychological Science. 2000;11:249–254. doi: 10.1111/1467-9280.00250. [DOI] [PubMed] [Google Scholar]
- Wagner DD, Altman M, Boswell RG, Kelley WM, Heatherton TF. Self-regulatory depletion enhances neural responses to rewards and impairs top-down control. Psychological Science. 2013;24:2262–2271. doi: 10.1177/0956797613492985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner DD, Heatherton TF. Self-regulatory depletion increases emotional reactivity in the amygdala. Social Cognitive and Affective Neuroscience. 2013;8:410–417. doi: 10.1093/scan/nss082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner DD, Heatherton TF. Self-regulation and its failure: The seven deadly threats to self-regulation. In: Mikulincer M, Shaver PR, Borgida E, Bargh JA, editors. APA handbook of personality and social psychology, Volume 1: Attitudes and social cognition. Washington, DC, US: American Psychological Association; 2015. pp. 805–842. [DOI] [Google Scholar]
- Weckesser LJ, Alexander NC, Kirschbaum C, Mennigen E, Miller R. Hydrocortisone Counteracts Adverse Stress Effects on Dual-Task Performance by Improving Visual Sensory Processes. Journal of Cognitive Neuroscience. 2016 doi: 10.1162/jocn_a_01006. Advance online publication. [DOI] [PubMed] [Google Scholar]
- Wersching H, Duning T, Lohmann H, Mohammadi S, Stehling C, Fobker M, … Knecht S. Serum C-reactive protein is linked to cerebral microstructural integrity and cognitive function. Neurology. 2010;74:1022–1029. doi: 10.1212/WNL.0b013e3181d7b45b. [DOI] [PubMed] [Google Scholar]
- Wilson AG, Symons JA, McDowell TL, McDevitt HO, Duff GW. Effects of a polymorphism in the human tumor necrosis factor α promoter on transcriptional activation. Proceedings of the National Academy of Sciences. 1997;94:3195–3199. doi: 10.1073/pnas.94.7.3195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wing RR, Tate DF, Gorin AA, Raynor HA, Fava JL. A self-regulation program for maintenance of weight loss. New England Journal of Medicine. 2006;355:1563–1571. doi: 10.1056/NEJMoa061883. [DOI] [PubMed] [Google Scholar]
- Zijlstra H, van Middendorp H, Devaere L, Larsen JK, van Ramshorst B, Geenen R. Emotion processing and regulation in women with morbid obesity who apply for bariatric surgery. Psychology & Health. 2012;27:1375–1387. doi: 10.1080/08870446.2011.600761. [DOI] [PubMed] [Google Scholar]