Abstract
Background & Aims
Cystic fibrosis liver disease (CFLD), a leading cause of death in cystic fibrosis (CF), is mostly described in pediatric populations. Adult-onset CFLD lacks sufficient characterization and diagnostic tools.
Methods
A cohort of CF patients without CFLD during childhood were followed for up to 38 years with serologic testing, imaging, and noninvasive fibrosis markers. Historical CFLD diagnostic criteria were compared with a newly proposed CFLD criteria.
Results
36 CF patients were followed for a median of 24.5 years(IQR=15.6, 32.9). By the last follow-up, 11(31%) had died. With conventional criteria, 8(22%) patients had CFLD, and by new criterion, 17 (47%) had CFLD at a median age of 36.6 years(IQR=26.5, 43.2). By new criterion, those with CFLD had higher median ALT(42 vs 27, p=0.005), AST(26 vs 21, p=0.01), direct bilirubin(0.13 vs 0.1, p=0.01), PT(14.4 vs 12.4, p=0.002), and APRI(0.31 vs 0.23, p=0.003) over the last two years of follow-up. Subjects with a Fibroscan® >6.8kPa had higher ALT(42 vs 28U/L, p=0.02), AST(35 vs 25U/L, p=0.02), APRI(0.77 vs 0.25, p=0.0004), FIB-4(2.14 vs 0.74, p=0.0003) and lower platelet counts(205 vs 293, p=0.02). One CFLD patient had nodular regenerative hyperplasia. Longitudinally, mean platelet counts significantly declined in the CFLD group(310 to 230U/L, p=0.0005). Deceased CFLD patients had lower platelet counts than those alive with CFLD(143 vs 258 U/L, p=0.004) or those deceased with no CFLD(143 vs 327U/L, p=0.006).
Conclusion
Adult-onset CFLD may be more prevalent than previously described which suggests a later wave of CFLD that impacts morbidity. Routine liver tests, radiologic imaging, noninvasive fibrosis markers and fibroscan® can be utilized algorithmically to identify adult CFLD. Further evaluation in other CF cohorts should be performed for validation.
Introduction
Cystic fibrosis (CF) is the most common autosomal recessive genetic disorder in Caucasians of European decent1, 2 with an annual incidence of 1 in 3,500 live births.3 This disease results from a genetic mutation that codes for the cystic fibrosis transmembrane conductance regulator (CFTR) protein, which plays an important role in chloride transport across epithelial membranes. The ΔF508 gene mutation is the most common cause of CF in North America3, 4, however, more than 1,300 known gene mutations have been associated with this disease. As a result of increased awareness and improved therapies, the median survival in patients with CF has improved to 37 years compared to 16 years in 1970.3
In CF, cystic fibrosis liver disease (CFLD) is the third leading cause of mortality and is estimated to account for 2.5–5% of overall CF mortality.1, 5, 6 While pediatric CFLD is well described with prevalence rates of up to 25% of children3, 6–9, adult onset CFLD has not been adequately characterized, thereby resulting in highly variable prevalence rates, ranging from 2–37%.3, 4, 10 The onset of liver disease in adults is a relatively newer finding in CF and may be a result of improved survival rates along with inconsistent definitions of CFLD.9, 11, 12
Although general agreement exists around the clinical utility of screening for CFLD, no single reliable physical, biochemical or radiological screening tool is available.13 Unlike other diseases with potential liver involvement, the utility of the “gold standard” liver biopsy has been described as “inconsistent” in diagnosing CFLD or has resulted in inadequate staging of CFLD, which may be a result of the various liver diseases that have been associated with CF.6, 14 Thus, the field has generally utilized composite criteria for the diagnosis of CFLD by employing multiple tools including physical examination, biochemical, and imaging modalities.12
More recently, the utility of non-invasive liver fibrosis markers have demonstrated promise in assisting in the diagnosis of CFLD11, 15, 16. In this study, we aimed to expand on the description of adult CFLD utilizing data from a cohort of patients with CF that have been followed at the NIH Clinical Center for up to 38 years, to evaluate the utility of non-invasive markers of liver disease in adult CFLD, and to develop a scoring system that can assist in diagnosing adult CFLD utilizing a combination of routine liver tests, radiologic imaging and noninvasive markers of hepatic fibrosis.
Methods
Patients
A cohort of genetically confirmed CF patients followed prospectively in a natural history protocol at the National Institutes of Health (NIH) Clinical Center was evaluated (NCT00001223 & NCT0001971). Longitudinal demographic, clinical, radiographic and laboratory data were collected. CF patients with suspected liver disease were referred for hepatology evaluation. Reasons for referral included persistently abnormal liver tests, evidence of viral hepatitis and abnormal hepatic imaging. Patients were evaluated by one of three hepatologists (CK, JHH, TH) with history, physical examination, radiologic imaging, laboratory investigations and transient elastography (available beginning in 2007). All patients underwent serologic testing for chronic liver diseases including viral hepatitis, autoimmune hepatitis, Wilson disease, alpha-1-antitrypsin deficiency, primary biliary cholangitis, and primary sclerosing cholangitis. Secondary sclerosing cholangitis was not an exclusion for the diagnosis of CFLD. If clinically indicated, patients were offered liver biopsy. All co-authors had access to the data and reviewed and approved the final manuscript.
Evaluation of Existing and New Diagnostic Criteria for Adult Cystic Fibrosis Liver Disease
The diagnostic criteria for CFLD described by Debray et al. was evaluated in this cohort of CF patients.12 The specifics of this criteria are described in an earlier publication by Debray and colleagues and can also be found in table 1.12 For the purposes of this study, this diagnostic criteria will be designated as the “Debray criteria” hereafter. In brief, this criteria suggests that the diagnosis of CFLD should be considered if significant findings are identified in at least two of the four categories including the physical examination, liver function tests, ultrasonography, or liver biopsy.
Table 1.
Diagnostic criteria for CFLD by the Debray criteria and the new criteria.
| Categories | Debray Criteria^ | New Criteria |
|---|---|---|
| Diagnosis of CFLD should be considered if 1 of the following is present: | ||
| Liver biopsy demonstrating pathology or radiologic evidence demonstrating diffuse liver disease or cirrhosis. | ||
| Diagnosis of CFLD should be considered if ≥2 categories are present: | ||
| Physical Examination | Hepatomegaly: >2 cm below the costal margin on the mid-clavicular line, confirmed by ultrasonography And/or splenomegaly, confirmed by US | Not included |
| Serum Blood Tests | Increase of transaminases and GGT above upper limits of normal at least 3 consecutive determinations over 12 months after excluding other causes of liver disease. | At least 2 persistently abnormal* ALT, AST, GGT or ALP |
| Radiologic Testing | Ultrasonographic evidence of liver involvement or portal hypertension or biliary abnormalities | Evidence of hepatomegaly, splenomegaly, or portal hypertension by imaging |
| Liver Biopsy | May be indicated if there is diagnostic doubt | |
| Vibration Controlled Transient Elastography | Not included | Abnormal fibroscan at any time |
| Noninvasive Fibrosis Biomarker Assessment | Not Included | Persistently abnormal* APRI, FIB-4, or AAR |
Persistently abnormal was defined as having abnormal values on multiple dates over at least 2 consecutive years.
Diagnostic criteria for CFLD as described by Debray et al.12
In addition to evaluating this cohort of CF patients with the Debray criteria for CFLD, a new criterion was developed and evaluated based on expanding evidence of the utility of noninvasive biomarkers in CFLD (table 1). This criterion utilizes routine hepatic biomarkers (alanine aminotransferase (ALT), aspartate aminotransferase (AST), gammaglutamyltransferase (GGT), and alkaline phosphatase (ALP)), radiologic imaging, vibration controlled transient elastography (VCTE) with Fibroscan® (Echosens, Paris), and noninvasive markers of fibrosis (AST-to-platelet-ratio-index (APRI), Fibrosis-4 index (FIB-4), and the AST-to-ALT ratio(AAR)). This criterion considers an individual to have CFLD if there is radiologic or histologic evidence of cirrhosis or diffuse liver disease, or a positive finding in at least two of the four categories including liver function tests, radiologic imaging, vibration controlled transient elastography, or noninvasive fibrosis biomarkers.
Both the Debray criteria and the new criteria were employed at baseline to demonstrate the presence or absence of CFLD at baseline. Subjects were then followed longitudinally with at least one visit annually. Based on the assessments performed during these visits (biochemical and radiological), subjects were identified as developing CFLD when they demonstrated abnormal values that fulfilled the Debray criteria and/or the new criteria with abnormal measurements on multiple dates over a 2-year period.
Normal values for ALT, AST, ALP, and GGT were based on the cut-offs for the assays used by the NIH Clinical Center laboratory. Adult patients with liver and spleen spans greater than 12 cm and 13 cm were considered to have hepatomegaly and splenomegaly respectively.12 In the evaluation of noninvasive fibrosis markers, the APRI, FIB-4, and the AAR were calculated (see supplementary section for formulas) and utilized as they have been extensively evaluated and validated in chronic hepatitis C with additional demonstrations of utility in chronic hepatitis B and nonalcoholic steatohepatitis. Specifically, the use of the APRI and FIB-4 are currently incorporated into practice guidelines for the management of chronic hepatitis C.17 In the APRI, a cut-off value of ≤0.50 was used for APRI for discerning the absence of significant fibrosis based off the seminal publication by Wai et al.17, 18 In the FIB-4 index, a cut-off value of ≥3.25 was utilized for the detection of advanced fibrosis as previously described by Sterling et al. in the seminal study evaluating subjects with HIV/HCV coinfection.17, 19 For the AAR, a cut-off value ≥ 1has been predictive of cirrhosis in chronic viral hepatitis and nonalcoholic steatohepatitis.20–22
Vibration controlled transient elastography, measured via Fibroscan® (Echosens, Paris), was evaluated in patients after 2007 after informed consent.23 Individuals specifically trained in the technique performed the elastography. At least 10 determinations were made and median results were expressed in kilopascals (kPA). A liver stiffness score of >6.8 kPa was used as the cut-off for liver disease based on a study by Kitson et al, which demonstrated a 76% sensitivity and 92% specificity for CFLD.11
Statistical Analysis
Statistical analysis was performed using JMP (Version 10, SAS Inc., Cary, NC, USA, Microsoft Excel (2011) for Mac OS X, and Prism GraphPad version 6 for Mac OS X, GraphPad Software, La Jolla, California, USA. Summary statistics of baseline data was presented as frequencies and median with interquartile range (IQR), unless specified otherwise. Two-sided Fisher’s exact tests were used to compare categorical group characteristics. Comparison of biomarkers between groups at baseline, end of follow-up and with vibration controlled transient elastography was performed via Mann-Whitney test. Paired comparison of biomarkers over the first two years of follow-up and the last two years of follow-up was performed by Wilcoxon signed-rank test. Statistical analysis of mortality and platelet counts were conducted by Mann-Whitney test. A p value of <0.05 was considered to be statistically significant.
Results
Subject Demographics
40 adult patients with CF were evaluated and 4 patients were excluded due to inadequate duration of follow-up (Figure 1). The remaining 36 patients were followed beginning at a median age of 11 years for a median of 24.5 years (Interquartile range(IQR)=15.6, 32.9) with baseline demographics shown in table 2. No subjects had evidence of liver disease during the first two years of follow-up by routine liver tests, radiologic imaging or noninvasive fibrosis markers. Twenty six of 27 patients with known genotypes had class I–III functional mutations of the CFTR allele. Groups with and without CFLD by the new criteria were relatively well matched without differences in the duration of follow-up, median age of diagnosis and age of enrollment. 25 patients were alive at the end of the study period, with 11 deaths occurring at ages 24 to 66 from respiratory failure (3), infection (3), lung transplant (2), and complications of decompensated liver disease with portal hypertension, ascites, and variceal bleeding (2). During the course of the study period, one patient underwent a transjugular liver biopsy with hepatic venous pressure gradient (HVPG) measurements which revealed focal biliary cirrhosis and nodular regenerative hyperplasia with an HVPG of 12mm Hg (Supplemental Figure 1). Seven of 17 subjects with CFLD were placed on ursodeoxycholic acid treatment after diagnosis of CFLD.
Figure 1.
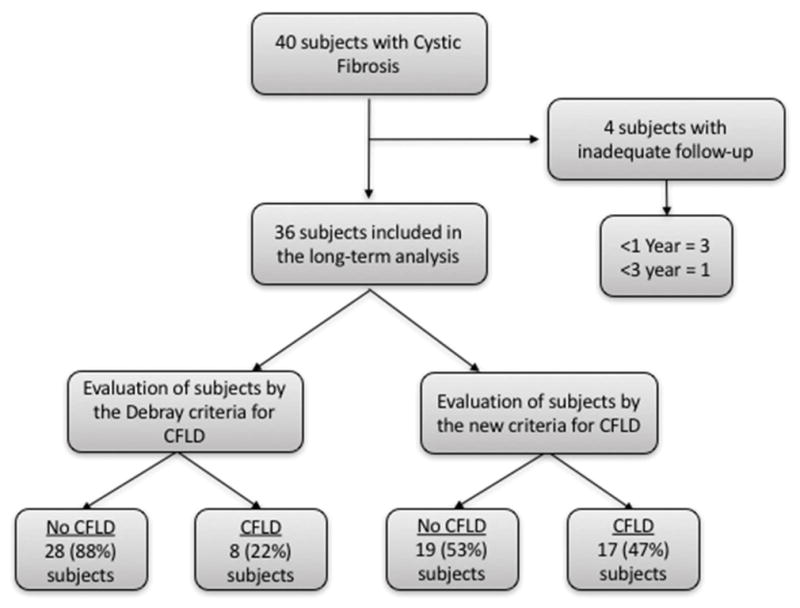
Patient flow throughout the study. Of the 40 patients screened, 36 subjects were enrolled into the study. This figure shows the patients in the study and the number of subjects that were classified as having CFLD or no CFLD by the Debray criteria and the new criteria.
Table 2.
Demographic descriptions of the 36 subjects with genetically confirmed CFLD.
| Feature | All Subjects | Data by New CFLD Criteria | ||
|---|---|---|---|---|
| CFLD | No CFLD | p-value | ||
| Number of Patients | 36 | 17 | 19 | |
| Male/Female | 26/10 | 15/2 | 11/8 | 0.06 |
| Caucasian/Other | 35/1 | 16/1 | 19/0 | |
| Alive/Dead at end of follow-up | 25/11 | 13/4 | 12/7 | |
| Median age of CF diagnosis (IQR) | 11.0 (2.6, 21.7) | 5.2 | 11 | 0.57 |
| Median age at time of enrollment (IQR) | 20.1 (15.3, 29.8) | 16.8 | 26.8 | 0.15 |
| Median duration of follow-up (IQR) | 24.5 (15.6, 32.9) | 28 | 21.5 | 0.08 |
| Genotype | ||||
| ΔF508/ΔF508 | 12 | 8 | 4 | 0.67 |
| ΔF508/Other | 9 | 5 | 4 | 0.29 |
| Other | 6 | 0 | 6 | 0.01 |
| Unknown | 9 | 4 | 5 | 0.43 |
Comparison between those classified as having CFLD and no CFLD by the Debray criteria
Based on the original criteria presented by Debray et al., 8 (22%) patients met criteria for adult CFLD (tables 1 & 3). While there were no differences in sex between groups (88% vs 67% male, p=0.40) more subjects with CFLD were homozygous for ΔF508 (p=0.04) compared to those without CFLD. The median age of CFLD diagnosis was 34.8 years (IQR=29.7, 39.4) and there were 3 deaths in the CFLD group and 8 deaths in the group without CFLD by the end of the study period.
Table 3.
Median laboratory values (with interquartile ranges) for the first two years of follow-up and the last two year of follow-up in subjects classified as CFLD and no CFLD by the Debray criteria
| Biomarker | CFLD n=8 (22%) |
No CFLD n=28 (88%) |
p-value |
|---|---|---|---|
| First Two Years of Follow-up | |||
| AST (U/L) | 28 (26,34) | 23 (20,28) | 0.37 |
| ALT (U/L) | 34 (25,41) | 21 (18,39) | 0.51 |
| Alkaline Phosphatase (U/L) | 182(167, 274) | 127(97, 203) | 0.11 |
| Platelets (K/uL) | 331 (281, 403) | 284 (237, 354) | 0.38 |
| Total Bilirubin | 0.53 (0.39, 0.93) | 0.38 (0.32, 0.47) | 0.18 |
| Direct Bilirubin | 0.10 (0.05,0.10) | 0.10 (0.05,0.10) | 0.59 |
| PT | 12.4 (12.3, 13.4) | 12.5 (11.6, 13.1) | 0.71 |
| Albumin | 3.9 (3.6, 4.2) | 4.1(4.0, 4.5) | 0.05 |
| APRI | 0.18 (0.16,0.35) | 0.21 (0.13,0.25) | 0.81 |
| FIB4 | 0.19 (0.12,0.35) | 0.39 (0.25,0.62) | 0.14 |
| AST/ALT Ratio | 0.78 (0.70,1.02) | 1.01 (0.74,1.28) | 0.09 |
| Last Two Years of Follow-up | |||
| AST (U/L) | 25 (21,32) | 34(25,41) | 0.98 |
| ALT (U/L) | 39 (31, 48) | 30 (23,41) | 0.22 |
| ALP (U/L) | 128 (96,187) | 107 (91, 122) | 0.24 |
| Platelets (K/uL) | 260 (165,283) | 270 (223,302) | 0.23 |
| Total Bilirubin (mg/dL) | 0.60 (0.53,0.88) | 0.38 (0.35,0.44) | 0.08 |
| Direct Bilirubin (mg/dL) | 0.18(0.14, 0.32) | 0.1(0.10, 0.12) | 0.06 |
| PT (sec) | 14.3 (13.7,14.9) | 13.9 (12.2,14.1) | 0.21 |
| Albumin (g/dL) | 3.5 (3.4, 3.7) | 3.7 (3.5, 4.0) | 0.10 |
| APRI | 0.31 (0.22,0.61) | 0.23 (0.19,0.33) | 0.25 |
| FIB-4 | 0.73 (0.48,1.72) | 0.77 (0.57,1.02) | 0.36 |
| AST/ALT Ratio | 0.78 (0.65,1.09) | 1.12 (0.80, 1.35) | 0.21 |
| Transient Elastography (kPa) (n=24) | 3.9 (3.4, 42.2) (n=7) | 4.65 (3.8, 7.2) (n=17) | 0.23 |
During the first two years of follow-up, there were no significant differences between those with and without CFLD comparing routine liver tests, platelet counts, prothrombin time, and noninvasive markers of fibrosis (table 3). However, albumin levels demonstrated a trend towards lower values in the CFLD group compared to the no CFLD group (3.9 vs 4.1, p=0.05). In the last two years of follow-up, total bilirubin (0.6 vs 0.38, p=0.08) and direct bilirubin (0.18 vs 0.1, p=0.06) trended towards higher values CFLD group compared to the no CFLD group, however, all were within normal ranges. All other liver tests, platelets, markers of hepatic synthetic function and noninvasive markers of fibrosis were not different between groups.
In a longitudinal comparison, the mean platelet counts from the first two years were compared with the last two years of follow-up between the CFLD and no CFLD group. The CFLD group demonstrated a significant decline in mean platelet counts from 336 U/L to 217 U/L (p=0.02), whereas the no CFLD group did not demonstrate a signficant decline by the end of follow-up (330 U/L to 271 U/L, p=0.08).
Comparison between those with CFLD vs without CFLD by the new criteria
Based on the aforementioned new CFLD criteria, 17 (47%) patients met criteria for CFLD (tables 1 & 2). There was a trend towards more males having CFLD compared to those without CFLD (88% vs 58%, p=0.06) without significant differences in homozygosity for ΔF508 (p=0.67). The median age of diagnosis of CFLD was 36.6 years (IQR = 26.5, 43.2) and there were 4 deaths in the CFLD group and 7 deaths in the group without CFLD. In comparing the age of diagnosis of the same 8 subjects diagnosed by the Debray criteria and this criteria, although the median age of diagnosis was younger by 5.4 years, this was not statistically significant (p=0.37). By log-rank test, more CF patients were diagnosed with liver disease utilizing the new criteria and at earlier times compared to the Debray criteria (p=0.017) (Figure 2).
Figure 2.

Kaplan-Meier method demonstrating the time to diagnosis of CFLD by the Debray criteria and the new criteria. By log-rank test, more patients were diagnosed with liver disease utilizing the new criteria and at earlier times compared to the Debray criteria (p=0.017).
During the first two years of follow-up, median liver tests (AST, ALT, ALP, total and direct bilirubin) were within normal limits as were platelet counts, PT and albumin, without significant differences between CFLD and non-CFLD groups (table 4). Additionally, noninvasive fibrosis biomarkers (APRI, FIB-4, and AAR) were also within normal limits without differences between groups.
Table 4.
Median laboratory values (with interquartile range) for the first two years of follow-up and last two years of follow-up in subjects classified as CFLD and no CFLD by new criteria.
| Biomarker | CFLD n=17 (47%) |
No CFLD n=19 (53%) |
p-value |
|---|---|---|---|
| First Two Years of Follow-up | |||
| AST (U/L) | 32 (22,46) | 21 (17,33) | 0.3 |
| ALT (U/L) | 28 (22,35) | 23 (19,27) | 0.065 |
| Alkaline Phosphatase (U/L) | 184 (148, 270) | 115 (89, 152) | 0.02 |
| Platelets (K/uL) | 306 (241, 367) | 285 (229, 357) | 0.9 |
| Total Bilirubin | 0.45 (0.26, 0.6) | 0.43 (0.33, 0.49) | 0.4 |
| Direct Bilirubin | 0.1 (0.04, 0.13) | 0.1 (0.05, 0.1) | 0.36 |
| PT | 12.4 (11.8, 13.2) | 12.5 (11.2, 13.2) | 0.96 |
| Albumin | 4.2 (3.8, 4.4) | 4.0 (3.9, 4.3) | 0.98 |
| APRI | 0.20 (0.15,0.34) | 0.19 (0.12,0.24) | 0.33 |
| FIB4 | 0.24 (0.17,0.60) | 0.45 (0.25,0.72) | 0.11 |
| AST/ALT Ratio | 0.59 (0.53,0.92) | 0.88 (0.62,1.07) | 0.15 |
| Last Two Years of Follow-up | |||
| AST (U/L) | 26 (21,33) | 21 (17,26) | 0.01 |
| ALT (U/L) | 42 (29, 53) | 27 (20,34) | 0.005 |
| ALP (U/L) | 110 (94,132) | 107 (83, 129) | 0.17 |
| Platelets (K/uL) | 241 (179,286) | 274 (223,315) | 0.10 |
| Total Bilirubin (mg/dL) | 0.45 (0.35,0.64) | 0.40 (0.32,0.45) | 0.14 |
| Direct Bilirubin (mg/dL) | 0.13 (0.10,0.17) | 0.10 (0.10,0.11) | 0.01 |
| PT (sec) | 14.4 (13.2,15.2) | 12.4 (11.9,13.9) | 0.002 |
| Albumin (g/dL) | 3.6 (3.5,3.9) | 3.6 (3.4,4.1) | 0.36 |
| APRI | 0.34 (0.22,0.54) | 0.20 (0.17,0.23) | 0.003 |
| FIB-4 | 0.78 (0.59,1.33) | 0.70 (0.50,1.00) | 0.08 |
| AST/ALT Ratio | 0.78 (0.65,1.09) | 1.12 (0.80, 1.35) | 0.18 |
| Transient Elastography (kPa) (n=24) | 5.4 (3.3, 10.0) (n=15) | 4 (3.3, 4.8) (n=9) | 0.34 |
During the final two years of follow-up, significant differences were identified in certain biochemical parameters between the two groups (table 4). The adult CFLD group had significantly higher AST (26 vs 21, p=0.01), ALT (42 vs 27 U/L, p=0.005), direct bilirubin (0.13 vs 0.10 mg/dL, p=0.01) and PT (14.4 vs 12.4 sec, p=0.002). However, there were no significant differences in ALP, total bilirubin, platelets, and albumin. In comparing changes in the noninvasive fibrosis biomarkers, APRI was significantly higher in the CFLD group (0.34 vs 0.20, p=0.003) and FIB-4 demonstrated a trend towards higher values in the CFLD group (0.78 vs 0.70, p=0.08). However, there was no difference in the AAR between groups.
Vibration Controlled Transient Elastography in CFLD
Vibration controlled transient elastography was performed in 24 adult subjects with CF and no differences were identified in comparing subjects with and without CFLD (5.4 vs 4 kPa, p=0.34). However, platelet counts were strongly correlated with VCTE (Spearman’s r: −0.60, p=0.002). When comparing subjects with a normal fibroscan (≤6.8 kPa) and subjects with an abnormal fibroscan (>6.8 kPa) with biomarkers of liver disease, significant differences were identified. Interestingly, subjects with a fibroscan >6.8 kPa had significantly higher mean ALT levels (42.2 vs 28.3 IU/L, p=0.02) and AST levels (35.4 vs 24.9 IU/L, p=0.02) and lower mean platelet counts (205.2 vs 293.7 U/L, p=0.02) (figure 3). Additionally, the APRI was significantly higher in subjects with a fibroscan score >6.8 kPa (0.77 vs 0.25, p=0.0004) as was FIB-4 (2.14 vs 0.74, p=0.0003) (figure 3), but not AAR (1.01 vs 0.57, p=0.13). No significant associations were identified with other biomarkers of liver disease including PT or bilirubin.
Figure 3.
Mean comparisons of ALT, AST, Platelets, APRI and FIB-4 in patients with CFLD who have normal and abnormal vibration controlled transient elastography measurements by fibroscan®.
Longitudinal Follow-up of Patients With and Without CFLD by the New Criteria
Since declining platelet counts have been utilized as a marker of progressive liver disease and has been associated with poor outcomes 24, 25, platelet counts were evaluated in a longitudinal fashion in the CF cohort. First, the mean platelet counts during the first two years of follow-up and the last two years of follow-up were compared in subjects with and without CFLD by the new criteria. Subjects with CFLD demonstrated a significant decline in mean platelets over the follow-up period (310 to 230 U/L, p=0.0005) compared to subjects without CFLD (310 to 279 U/L, p=0.06) (figure 4). Next, mean platelet counts over the last two years of follow-up were compared in subjects with and without CFLD by the new criteria between subjects who were alive and deceased by the end of the study period. In this analysis, deceased subjects with CFLD had significantly lower mean platelet counts than those who were alive with CFLD (143 vs 258 U/L, p=0.004) and those that were deceased without CFLD (143 vs 327 U/L, p=0.006) (figure 4). However, there were no significant differences in mean platelet counts between subjects who were dead and alive without CFLD.
Figure 4.
(A) Longitudinal comparison of platelet counts in subjects with and without CFLD. (B) Comparison of mean platelet counts between those alive and deceased at the end of follow-up by subjects classified as having CFLD and those without CFLD by the new criteria.
Discussion
In this prospective, natural history study of patients with genetically confirmed cystic fibrosis followed for up to 38 years, we demonstrate that adult onset cystic fibrosis liver disease can have an impact on morbidity. Additionally, we demonstrate that a composite system utilizing routine serum laboratory measurements, radiologic imaging, vibration controlled transient elastography and noninvasive fibrosis markers may have utility in identifying subjects with adult cystic fibrosis liver disease. In comparison to the widely utilized composite diagnostic algorithm for CFLD12, our new criteria captured an additional 25% (9) patients with CFLD in addition to demonstrating that those that died in the CFLD group had significantly lower platelet counts, which is a surrogate marker of end-stage liver disease with portal hypertension.
One of the major strengths of this study is the long duration of follow-up which allowed for the identification of individuals who developed CFLD in adulthood. While CFLD in childhood is well characterized, adult onset CFLD has not generally been an active area of research concentration. Despite this, several major pieces describing the incidence of CFLD have alluded to the possibility of a second wave of liver disease in adulthood. For example, Colombo et al.9 identified <4 adult subjects with CFLD with one diagnosed at age 28 and another large multinational study excluded 4 subjects from their analysis as they met criteria for CFLD at >30 years of age.26 In our cohort, we describe that adult onset CFLD occurred at a median age of 37 years in the absence of any biochemical or radiological evidence of liver disease during childhood. Additional studies are needed for further understanding and characterization as patients with CF are living longer lives with improved medical management.
Another important finding in this study is the demonstration of the utility of incorporating VCTE (Fibroscan®) and noninvasive fibrosis markers into a diagnostic algorithm for CFLD. Recently, increasing evidence has suggested the potential utility of fibroscan® in the detection of pediatric CFLD27–31, with only a few studies in adults.11, 32 This study adds to the growing body of evidence that VCTE may have utility in clinical practice in adult patients with cystic fibrosis. This is timely given that the fibroscan® is approved by the U.S. Food and Drug Administration for clinical use which has resulted in increased availability in many advanced medical centers that routinely provide care to CF patients. Similarly, studies evaluating noninvasive fibrosis markers (specifically APRI and FIB-4) have solely been performed in children with CFLD.26, 33 In this study, with the long duration of follow-up, we were able to expand on the experience of these biomarkers in pediatrics in addition to demonstrating its utility in identifying adults who develop CFLD. Taken together, by incorporating these biomarkers with routinely available serologic testing and radiologic imaging, we were able to incorporate simple tests to identify 25% more patients with CFLD compared to well-known and utilized historical criteria that has been essential to the CF field.12 However, further experience is needed with this criteria in other CF cohorts prior to widespread application.
In the longitudinal evaluation of CF patients in our cohort, we identified that compared to patients without CFLD, patients with CFLD demonstrated significant declines in platelet counts by the end of the follow-up period. Additionally, subjects with CFLD who died during the follow-up period had lower platelet counts than subjects alive with CFLD and deceased without CFLD. The demonstration of declining platelet counts, while not previously described in CFLD, is an important finding that has been described in various liver diseases ranging from cirrhosis related liver diseases to non-cirrhotic portal hypertension. In chronic hepatitis C, platelets have been described to decline with advancing disease even prior to reaching cirrhosis34 with improvement after sustained viral eradication even at stages prior to cirrhosis.35 In non-cirrhotic portal hypertension, declining platelet counts over time was associated with mortality.25 Thus, the clinical utility of a declining platelet count over time may have the ability to serve as an “early warning” sign suggesting the evolution of liver disease in CF and should be explored further.
Finally, we identified that the pathogenesis of CFLD in adulthood may not be entirely due to cirrhosis related liver diseases as it is often described in pediatric patients. This finding is similar to other systemic diseases where improved survival has revealed the emergence other forms of liver disease, including those from the noncirrhotic portal hypertension spectrum.36–38 In our cohort, one subject with portal hypertension and variceal bleeding was found to have nodular regenerative hyperplasia on liver biopsy. This finding not only expands on the solitary description of CFLD-NRH in the literature, but also explains some of the potential diagnostic difficulties in CFLD where ALP has been reported to be the isolated abnormal liver test in 25% of NRH cases.39–42 Since NRH has been associated with chronic drug induced liver injury from long-standing antibiotic use, as often seen in CF, it is possible that more cases of NRH may come to light with more experience in adult CFLD.43
This study, while strong for the duration of follow-up to demonstrate the evolution of a second wave of liver disease in CF, is limited by the small number of patients followed along with the fact that this study was performed at a single-center. These aspects may limit the generalizability of the findings, particularly with the effects on mortality. Further studies are needed in additional cohorts to expand the experience of adult CFLD and the utility of noninvasive biomarkers for the diagnosis of adult CFLD. Additionally, only two-thirds of the subjects had Fibroscan® measurements performed which were all performed in adulthood. This limitation is due to the fact that Fibroscan® was not performed at the NIH Clinical Center prior to 2007 and not available before 2003.44 As experience expands with VCTE in adults with CF, reporting of long-term fibroscan experience may enhance the ability of early detection of CFLD. Finally, the lack of liver histology in each subject is a relative limitation of this study. However, it should be noted that the CFLD field has acknowledged the significant limitations of liver histology in CF given the often-patchy histologic nature of liver disease in these subjects. Nevertheless, the potential implications from this study remain true and the findings seem worthy of further exploration.
In summary, our study suggests that adult onset CFLD may be more prevalent than previously described and that a second wave of liver disease with impact on mortality exists in CF. Through utilization of routine biomarkers consisting of standard liver tests, radiologic imaging, noninvasive fibrosis markers and vibration controlled transient elastography in an algorithmic fashion, the identification of adult CFLD is possible. Early identification of adult CFLD may help to prevent or delay morbidity and mortality. Further evaluation of other CF cohorts should be performed for continued characterization of adult CFLD and validation of this new diagnostic criteria.
Supplementary Material
Acknowledgments
Financial Support: This research was supported by the Intramural Research Program of the National Institute of Diabetes and Digestive and Kidney Diseases and the National Cancer Institute of the National Institutes of Health.
Abbreviations
- CF
cystic fibrosis
- CFLD
cystic fibrosis liver disease
- ALT
alanine aminotransferase
- AST
aspartate aminotransferase
- IQR
interquartile range
- APRI
ast-to-platelet-ratio-index
- FIB-4
fibrosis index based on four factors
- CFTR
cystic fibrosis transmembrane conductance regulator
- NIH
National Institutes of Health
- GGT
gammaglutamyltransferase
- ALP
alkaline phosphatase
- HVPG
hepatic venous pressure gradient
- VCTE
vibration controlled transient elastography
- AAR
AST-to-ALT ratio
- HIV
human immunodeficiency virus
- HCV
hepatitis C virus
- kPa
kilopascals
- PT
prothrombin time
- U.S
United States
- NRH
nodular regenerative hyperplasia
Footnotes
Conflicts of Interest: None of the authors has financial interests or conflicts of interest related to this research.
Authors’ Contributions:
Study concept and design: Koh, Sakiani, Chernick, Heller
Acquisition of Data: Koh, Herion, Hoofnagle, Chernick, Heller
Analysis and Interpretation of Data: Koh, Sakiani, Surana, Zhao, Eccleston, Kleiner, Herion, Liang, Hoofnagle, Chernick, Heller
Drafting of the manuscript: Koh, Sakiani, Surana, Heller
Critical revision of the manuscript for important intellectual content: Koh, Sakiani, Surana, Zhao, Eccleston, Kleiner, Herion, Liang, Hoofnagle, Chernick, Heller
Statistical Analysis: Surana, Koh, Sakiani, Zhao
Study Supervision: Chernick, Heller
References
- 1.Colombo C. Liver disease in cystic fibrosis. Curr Opin Pulm Med. 2007;13:529–36. doi: 10.1097/MCP.0b013e3282f10a16. [DOI] [PubMed] [Google Scholar]
- 2.Rowe SM, Miller S, Sorscher EJ. Mechanisms of disease: Cystic fibrosis. New England Journal of Medicine. 2005;352:1992–2001. doi: 10.1056/NEJMra043184. [DOI] [PubMed] [Google Scholar]
- 3.Cystic Fibrosis Foundation. Patient Registry Annual Data Report. 2011 [Google Scholar]
- 4.Bhardwaj S, Canlas K, Kahi C, et al. Hepatobiliary Abnormalities and Disease in Cystic Fibrosis Epidemiology and Outcomes Through Adulthood. Journal of Clinical Gastroenterology. 2009;43:858–864. doi: 10.1097/MCG.0b013e31819e8bbd. [DOI] [PubMed] [Google Scholar]
- 5.Sokol RJ, Durie PR. Recommendations for management of liver and biliary tract disease in cystic fibrosis. Cystic Fibrosis Foundation Hepatobiliary Disease Consensus Group. J Pediatr Gastroenterol Nutr. 1999;28(Suppl 1):S1–13. doi: 10.1097/00005176-199900001-00001. [DOI] [PubMed] [Google Scholar]
- 6.Lindblad A, Glaumann H, Strandvik B. Natural history of liver disease in cystic fibrosis. Hepatology. 1999;30:1151–8. doi: 10.1002/hep.510300527. [DOI] [PubMed] [Google Scholar]
- 7.Diwakar V, Pearson L, Beath S. Liver disease in children with cystic fibrosis. Paediatr Respir Rev. 2001;2:340–9. doi: 10.1053/prrv.2001.0170. [DOI] [PubMed] [Google Scholar]
- 8.Colombo C. Anaylsis of risk factors for the development of liver disease associated with cystic fibrosis. The Journal of Pediatrics. 1994;124:393–399. doi: 10.1016/s0022-3476(94)70361-2. [DOI] [PubMed] [Google Scholar]
- 9.Colombo C, Battezzati PM, Crosignani A, et al. Liver disease in cystic fibrosis: A prospective study on incidence, risk factors, and outcome. Hepatology. 2002;36:1374–82. doi: 10.1053/jhep.2002.37136. [DOI] [PubMed] [Google Scholar]
- 10.Nash KL, Allison ME, McKeon D, et al. A single centre experience of liver disease in adults with cystic fibrosis 1995–2006. J Cyst Fibros. 2008;7:252–7. doi: 10.1016/j.jcf.2007.10.004. [DOI] [PubMed] [Google Scholar]
- 11.Kitson MT, Kemp WW, Iser DM, et al. Utility of transient elastography in the non-invasive evaluation of cystic fibrosis liver disease. Liver Int. 2013;33:698–705. doi: 10.1111/liv.12113. [DOI] [PubMed] [Google Scholar]
- 12.Debray D, Kelly D, Houwen R, et al. Best practice guidance for the diagnosis and management of cystic fibrosis-associated liver disease. J Cyst Fibros. 2011;10(Suppl 2):S29–36. doi: 10.1016/S1569-1993(11)60006-4. [DOI] [PubMed] [Google Scholar]
- 13.Flass T, Narkewicz MR. Cirrhosis and other liver disease in cystic fibrosis. J Cyst Fibros. 2013;12:116–24. doi: 10.1016/j.jcf.2012.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lewindon PJ, Shepherd RW, Walsh MJ, et al. Importance of hepatic fibrosis in cystic fibrosis and the predictive value of liver biopsy. Hepatology. 2011;53:193–201. doi: 10.1002/hep.24014. [DOI] [PubMed] [Google Scholar]
- 15.Karlas T, Neuschulz M, Oltmanns A, et al. ARFI and transient elastography for characterization of cystic fibrosis related liver disease: First longitudinal follow-up data in adult patients. J Cyst Fibros. 2013 doi: 10.1016/j.jcf.2013.04.004. [DOI] [PubMed] [Google Scholar]
- 16.Karlas T, Neuschulz M, Oltmanns A, et al. Non-invasive evaluation of cystic fibrosis related liver disease in adults with ARFI, transient elastography and different fibrosis scores. PLoS One. 2012;7:e42139. doi: 10.1371/journal.pone.0042139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Panel AIHG. Hepatitis C guidance: AASLD-IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus. Hepatology. 2015;62:932–54. doi: 10.1002/hep.27950. [DOI] [PubMed] [Google Scholar]
- 18.Wai CT, Greenson JK, Fontana RJ, et al. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology. 2003;38:518–26. doi: 10.1053/jhep.2003.50346. [DOI] [PubMed] [Google Scholar]
- 19.Sterling RK, Lissen E, Clumeck N, et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43:1317–25. doi: 10.1002/hep.21178. [DOI] [PubMed] [Google Scholar]
- 20.Haukeland JW, Schreiner LT, Lorgen I, et al. ASAT/ALAT ratio provides prognostic information independently of Child-Pugh class, gender and age in non-alcoholic cirrhosis. Scand J Gastroenterol. 2008;43:1241–8. doi: 10.1080/00365520802158614. [DOI] [PubMed] [Google Scholar]
- 21.Sheth SG, Flamm SL, Gordon FD, et al. AST/ALT ratio predicts cirrhosis in patients with chronic hepatitis C virus infection. Am J Gastroenterol. 1998;93:44–8. doi: 10.1111/j.1572-0241.1998.044_c.x. [DOI] [PubMed] [Google Scholar]
- 22.Giannini E, Risso D, Botta F, et al. Validity and clinical utility of the aspartate aminotransferase-alanine aminotransferase ratio in assessing disease severity and prognosis in patients with hepatitis C virus-related chronic liver disease. Arch Intern Med. 2003;163:218–24. doi: 10.1001/archinte.163.2.218. [DOI] [PubMed] [Google Scholar]
- 23.Castera L, Forns X, Alberti A. Non-invasive evaluation of liver fibrosis using transient elastography. J Hepatol. 2008;48:835–47. doi: 10.1016/j.jhep.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 24.Poordad F, Theodore D, Sullivan J, et al. Medical resource utilisation and healthcare costs in patients with chronic hepatitis C viral infection and thrombocytopenia. J Med Econ. 2011;14:194–206. doi: 10.3111/13696998.2011.562266. [DOI] [PubMed] [Google Scholar]
- 25.Feld JJ, Hussain N, Wright EC, et al. Hepatic involvement and portal hypertension predict mortality in chronic granulomatous disease. Gastroenterology. 2008;134:1917–26. doi: 10.1053/j.gastro.2008.02.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stonebraker JR, Ooi CY, Pace RG, et al. Features of Severe Liver Disease With Portal Hypertension in Patients With Cystic Fibrosis. Clin Gastroenterol Hepatol. 2016;14:1207–1215 e3. doi: 10.1016/j.cgh.2016.03.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Witters P, De Boeck K, Dupont L, et al. Non-invasive liver elastography (Fibroscan) for detection of cystic fibrosis-associated liver disease. J Cyst Fibros. 2009;8:392–9. doi: 10.1016/j.jcf.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 28.Menten R, Leonard A, Clapuyt P, et al. Transient elastography in patients with cystic fibrosis. Pediatr Radiol. 2010;40:1231–5. doi: 10.1007/s00247-009-1531-z. [DOI] [PubMed] [Google Scholar]
- 29.Van Biervliet S, Verdievel H, Vande Velde S, et al. Longitudinal Transient Elastography Measurements Used in Follow-up for Patients with Cystic Fibrosis. Ultrasound Med Biol. 2016;42:848–54. doi: 10.1016/j.ultrasmedbio.2015.11.011. [DOI] [PubMed] [Google Scholar]
- 30.Breton E, Bridoux-Henno L, Guyader D, et al. Value of transient elastography in noninvasive assessment in children’s hepatic fibrosis. Arch Pediatr. 2009;16:1005–10. doi: 10.1016/j.arcped.2009.03.021. [DOI] [PubMed] [Google Scholar]
- 31.Aqul A, Jonas MM, Harney S, et al. Correlation of Transient Elastography With Severity of Cystic Fibrosis Related Liver Disease. J Pediatr Gastroenterol Nutr. 2016 doi: 10.1097/MPG.0000000000001448. [DOI] [PubMed] [Google Scholar]
- 32.Sadler MD, Crotty P, Fatovich L, et al. Noninvasive methods, including transient elastography, for the detection of liver disease in adults with cystic fibrosis. Can J Gastroenterol Hepatol. 2015;29:139–44. doi: 10.1155/2015/138530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Leung DH, Khan M, Minard CG, et al. Aspartate aminotransferase to platelet ratio and fibrosis-4 as biomarkers in biopsy-validated pediatric cystic fibrosis liver disease. Hepatology. 2015;62:1576–83. doi: 10.1002/hep.28016. [DOI] [PubMed] [Google Scholar]
- 34.Tana MM, Zhao X, Bradshaw A, et al. Factors associated with the platelet count in patients with chronic hepatitis C. Thromb Res. 2015;135:823–8. doi: 10.1016/j.thromres.2015.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Koh C, Heller T, Haynes-Williams V, et al. Long-term outcome of chronic hepatitis C after sustained virological response to interferon-based therapy. Aliment Pharmacol Ther. 2013;37:887–94. doi: 10.1111/apt.12273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fuss IJ, Friend J, Yang Z, et al. Nodular regenerative hyperplasia in common variable immunodeficiency. J Clin Immunol. 2013;33:748–58. doi: 10.1007/s10875-013-9873-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hussain N, Feld JJ, Kleiner DE, et al. Hepatic abnormalities in patients with chronic granulomatous disease. Hepatology. 2007;45:675–83. doi: 10.1002/hep.21524. [DOI] [PubMed] [Google Scholar]
- 38.O’Brien K, Hussain N, Warady BA, et al. Nodular regenerative hyperplasia and severe portal hypertension in cystinosis. Clin Gastroenterol Hepatol. 2006;4:387–94. doi: 10.1016/j.cgh.2005.12.013. [DOI] [PubMed] [Google Scholar]
- 39.Schwarzenberg SJ, Wielinski CL, Shamieh I, et al. Cystic fibrosis-associated colitis and fibrosing colonopathy. J Pediatr. 1995;127:565–70. doi: 10.1016/s0022-3476(95)70113-3. [DOI] [PubMed] [Google Scholar]
- 40.Reshamwala PA, Kleiner DE, Heller T. Nodular regenerative hyperplasia: not all nodules are created equal. Hepatology. 2006;44:7–14. doi: 10.1002/hep.21258. [DOI] [PubMed] [Google Scholar]
- 41.Arvanitaki M, Adler M. Nodular regenerative hyperplasia of the liver. A review of 14 cases. Hepatogastroenterology. 2001;48:1425–9. [PubMed] [Google Scholar]
- 42.Wanless IR. Micronodular transformation (nodular regenerative hyperplasia) of the liver: a report of 64 cases among 2,500 autopsies and a new classification of benign hepatocellular nodules. Hepatology. 1990;11:787–97. doi: 10.1002/hep.1840110512. [DOI] [PubMed] [Google Scholar]
- 43.Stine JG, Chalasani N. Chronic liver injury induced by drugs: a systematic review. Liver Int. 2015;35:2343–53. doi: 10.1111/liv.12958. [DOI] [PubMed] [Google Scholar]
- 44.Sandrin L, Fourquet B, Hasquenoph JM, et al. Transient elastography: a new noninvasive method for assessment of hepatic fibrosis. Ultrasound Med Biol. 2003;29:1705–13. doi: 10.1016/j.ultrasmedbio.2003.07.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.




