Abstract
Venous thrombus was recognized in the upper extremity of a 53-year-old man after blood donation. The patient presented with a 15-day history of swelling in the left upper-extremity that started 6 hours after apheresis. Contrast-enhanced computed tomography revealed clots in the deep veins of the left arm and the peripheral pulmonary artery. Blood donation had proceeded smoothly, and the patient had no thrombotic predisposition, except for a smoking habit. The thrombus resolved following anticoagulant therapy, and the patient’s clinical course was uncomplicated. Despite a thorough investigation, the cause of this thrombus remains unknown.
Keywords: upper-extremity deep vein thrombosis (UEDVT), hypercoagulability, blood donation
Introduction
Blood donation is normally a safe process. A small number of blood donors experience donation-related events (1), and serious complications such as upper-extremity deep vein thrombosis (UEDVT) are extremely rare (2). We herein report the case of a patient with UEDVT following blood donation. This is an extremely rare event, with only 27 similar cases reported worldwide in recent decades.
Case Report
A 53-year-old Japanese man presented to our walk-in clinic with a 15-day history of swelling in the left arm. The patient was a regular blood donor who had just undergone uncomplicated apheresis for the 46th time, with blood collected from the left antecubital fossa. The apheresis procedure involved 3 cycles and lasted 60 minutes. Approximately 6 hours after the donation process was completed, the patient noted swelling that extended from his left elbow to his forearm and fingers. He had no associated numbness, pain, skin discoloration, or congestion. The patient consulted the Blood Center and was referred to a local orthopedic surgery clinic and pain clinic. He was started on NeurotropinⓇ and mecobalamin but had seen no improvement in his condition. Eleven days after donation, there was notable engorgement of the superficial veins on the left precordium, and the patient was subsequently referred to our walk-in center. His vital signs were stable, and his body mass index was 24.3 kg/m2. The patient had no history of surgery or trauma. The patient was currently smoking 15 cigarettes daily and was employed as an information technology professional.
The patient’s entire left arm was swollen, and engorgement of the superficial veins on the left precordium was evident (Fig. 1). Laboratory findings revealed an elevated D-dimer level (2.3 μg/mL, reference range; ≤1.0 μg/mL) but no other abnormal findings. Table 1 shows the results of routine blood tests, and Table 2 shows the results of thrombotic predisposition screening.
Figure 1.
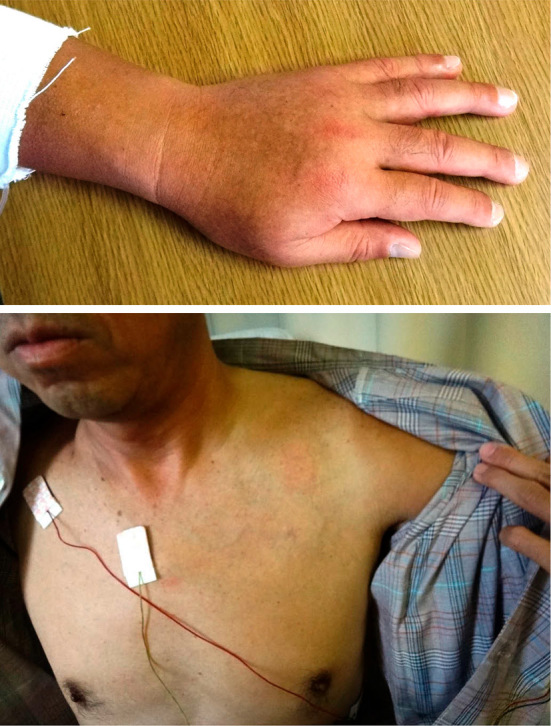
Appearance on the next day of admission. The patient’s entire left arm was swollen, and engorgement of the superficial veins on the left precordium was evident.
Table 1.
Initial Laboratory Findings.
| Laboratory test | Result | Reference range |
|---|---|---|
| WBC (×103/μL) | 6.2 | 3.9-9.7 |
| Hb (g/dL) | 16.6 | 13.4-17.1 |
| Hematocrit (%) | 48.7 | 40.4-51.1 |
| Platelet (×103/μL) | 288 | 153-346 |
| Lactate dehydrogenase (IU/L) | 215 | 119-221 |
| Creatinine kinase (IU/L) | 423 | 57-240 |
| C-reactive protein (mg/dL) | 0.1 | ≤0.2 |
| Prothrombin time-International normalized ratio | 0.99 | 0.90-1.10 |
| Activated partial thromboplastin time (sec) | 33.4 | 35.4 |
| D-dimer (μg/mL) | 2.3 | ≤1.0 |
・Reference range is in our hospital.
Table 2.
Laboratory Findings of Possible Hypercoagulable State.
| Laboratory test | Result | Reference range |
|---|---|---|
| Fibrinogen (mg/dL) | 308.0 | 150-400.0 |
| Antithrombin III activity (%) | 100.0 | 86.6-118.0 |
| Thrombin-antithrombin complex (μg/L) | 4.1 | 1.0-4.0 |
| Protein C activity (%) | 114 | 70-150 |
| Protein S antigen (%) | 84 | 65-135 |
| Lupus anticoagulant (NR) | 1.0 | ≤1.30 |
| Anti-cardiolipin IgG antibody (U/mL) | 8 | <10 |
| Homocysteine (nmol/mL) | 7.2 | 3.7-13.5 |
| Lipoprotein(a) (mg/dL) | 17 | 0-40 |
| Carcinoemboryonic antigen (ng/mL) | 1.3 | ≤5.0 |
| Carbohydrate antigen 19-9 (U/mL) | 8 | ≤37 |
・Reference range is in our hospital.
Venous thrombosis was suspected on the basis of the physical findings and the initial laboratory data. Contrast-enhanced computed tomography (CT) on the day of the first visit revealed a large clot extending from the left subclavian vein to the brachial vein and a small clot in the left pulmonary artery (Fig. 2). The patient was admitted to our hospital, and thrombolytic therapy was started immediately. This consisted of intravenous urokinase 480,000 IU/day on days 1-7 and heparin 50,000 IU/day on days 1-12, with oral warfarin 3 mg started from day 4. The clots regressed with treatment, and contrast-enhanced CT on day 8 showed that only the clot in the left subclavian vein remained. The patient’s subsequent clinical course was uneventful, and he was discharged on day 14 under maintenance therapy with warfarin 3 mg. After discharge, the engorgement of the superficial veins resolved, and the swelling of the arm improved, contracting to the confines of the upper arm. Approximately 3 months after onset, the symptoms had resolved completely, and the D-dimer levels had also decreased (<0.5 μg/mL), indicating complete thrombolysis. There has been no recurrence in the three years since.
Figure 2.
Findings in contrast-enhanced computed tomography at the initial visit. A large clot extending from the left subclavian vein to the brachial vein, and a small clot in the left pulmonary artery.
Blood donation in Japan
A discussion of this patient’s case would be incomplete without a review of the blood donation process in Japan. All blood donations are performed by interrupting the blood flow and puncturing a cutaneous vein in the cubital fossa with a 17-gauge stainless steel needle. During donation, blood flow in the upper limb is temporarily interrupted by applying a manchette tourniquet approximately 10-cm wide to the upper arm. Apheresis usually involves 3-4 cycles of blood collection and reinfusion, each lasting approximately 15 minutes. In the process of apheresis, circulating blood is exposed to a polyvinyl chloride tube during extracorporeal circulation, as well as to acid-citrate-dextrose (ACD), which serves as an anticoagulant in the circuit. The process lasts approximately 45-90 minutes in apheresis donation and 10-15 minutes in whole-blood donation (1).
Discussion
UEDVT is a rare disorder, with an annual incidence of 0.4-1/10,000 (3), and accounts for <4% of all cases of deep vein thrombosis (DVT) (4,5). In UEDVT, unlike lower-extremity deep vein thrombosis (LEDVT), thrombophilic coagulation defects are rare (6). Many of the risk factors for UEDVT resemble those for LEDVT (7,8), in addition to specific risk factors such as surgery or wounds to the arms, catheter insertion, thoracic outlet syndrome, and strenuous muscular efforts with the arms (9-11). The possible causes of thrombus formation in this case are discussed in terms of Virchow's triad.
Endothelial injury. Our patient had a prominent history of smoking 15 cigarettes daily for 33 years. This is relevant in this case, since smoking can lead to endothelial dysfunction (12,13).
Stasis of blood flow. On contrast-enhanced CT, there were no abnormalities in the vascular morphology from the left upper limb to the thorax, but further investigation of the vessels was ceased because the patient developed an allergy to the contrast agent. During donation, blood flow in the upper limb was temporarily interrupted, which may have resulted in a hypercoagulable state. Rasmussen et al. (14) and Hughes et al. (15) described hypercoagulability in healthy individuals induced by a tourniquet applied for 3 and 10 minutes, respectively.
Hypercoagulability. The conventional screening tests for DVT risk factors in this case had negative results (Table 2), and the patient was free from malignancies. However, the process of blood donation per se may have contributed to a hypercoagulable state. Venepuncture results in physical injury to the vessel wall with a subsequent release of tissue factors and activation of the extrinsic coagulation cascade. In addition, blood is exposed to an artificial tube during extracorporeal circulation, which results in initiation of the intrinsic cascade. Indeed, apheresis itself is known to have an effect on coagulability, irrespective of the use of anticoagulants (16-21).
We suspect that a series of processes - interrupted blood flow, venous puncture, placement of a needle, and extracorporeal circulation - in combination with the patient’s smoking history affected the coagulating activity. However, since a thrombus may also develop in whole-blood donation, which is a much shorter process than apheresis that does not involve extracorporeal circulation, it is likely that other factors in addition to the ones mentioned above may have also contributed.
To our knowledge, this is the first report of UEDVT complicating blood donation in Japan. To date, 27 cases have been reported in the English-language literature, including those reported only in abstracts, of which 2 cases involved apheresis (22,23), 4 involved whole-blood donation (24,25), and the others did not mention the method of donation (26,27). Table 3 describes the 8 cases for which the details were recorded. These included patients who were taking oral contraceptives (n=4), had a prothrombin gene mutation (n=1), cases in which there were problems with the blood donation procedure (n=4), as well as cases with unknown causes (n=2). Pain was present in all 7 cases, with swelling and bruising also present in 5, swelling alone in 1, and bruising alone in 1. In the present case, complaints other than swelling were absent. In almost all of the reported cases, including our own, the patient's course was uneventful.
Table 3.
Thrombosis as a Complication of Blood Donation: Comparison with Other Reported Cases.
| Case | Age | Sex | Race | Type of donation | First donation/after multiple donations | Time to onset | Time to diagnosis | Symptoms | Thrombus location (most proximal) | Causative risk factors | Complete resolution? | Date | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 54 | M | Asian | Apheresis | Multiple | 6 hours | 15 days | Swelling of the arm, engorgement of the superficial veins in the precordial area | Subclavian vein, pulmonary artery | Smoker | Yes | 2016 | Present case |
| 2 | 21 | F | Caucasian | Apheresis | Multiple | Not recorded | Not recorded | Pain, swelling of the hand, weakness | Axillary vein | Oral contraceptive, poor retransfusion blood flow | Unknown | 2000 | [22]† |
| 3 | 39 | F | Caucasian | Apheresis | Multiple | 4 days | 14 days | Pain, swelling of the arm, bruise | Axillary vein | Oral contraceptive | Unknown | 2002 | [23] |
| 4 | 33 | F | Caucasian | Whole blood | Multiple | 3 days | 10 days | Pain, swelling of the arm, small bruise* | Proximal brachial vein | Oral contraceptive, prothrombin gene variant, slow phlebotomy, needle adjustment | Yes | 2015 | [24] |
| 5 | 17 | M | Caucasian | Whole blood | First | Several hours | 6 days | Pain, swelling of the arm, large bruise** | Subclavian vein | None identified | No | 2015 | [24] |
| 6 | 17 | M | Asian | Whole blood | First | 1-2 days | 5 days | Pain, swelling of the arm, large bruise**, erythema, sensation of warm | Distal brachial vein | Tight tourniquet | Yes | 2015 | [24] |
| 7 | 20 | F | Caucasian | Whole blood | First | 1-5 days | 5 days | Pain, swelling of the arm, large bruise** | Distal brachial vein | Oral contraceptive, slight access issue | Yes | 2004 | [25] |
| 8 | 44 | F | Not recorded | Not recorded | Multiple | 1 day | 20 days | Pain, bruise | Distal brachial vein | None identified | Yes | 1987 | [26] |
*: small bruise<2×2 in, **: large bruise ≥ 2×2 in, †; Abstract only
We treated a healthy man with no clear predisposition to thrombosis other than smoking who presented with swelling of the upper limb and was diagnosed with UEDVT due to blood donation. As in almost all previously reported cases, this was not a severe situation. Clinicians in outpatient practice should consider thrombosis in their list of differential diagnoses when encountering a patient with swelling of the arm following blood donation. Although the cause was not clarified, we hypothesized that both the existence of a smoking habit and the normal process of blood donation contributed to this UEDVT.
The authors state that they have no Conflict of Interest (COI).
References
- 1. Japanese Red Cross Tokyo Metropolitan Blood Center. [cited 2016 Oct 6] Available from: https://www.tokyo.bc.jrc.or.jp/ (in Japanese) .
- 2. Newman B. Arm complications after manual whole blood donation and their impact. Transfus Med Rev 27: 44-49, 2013. [DOI] [PubMed] [Google Scholar]
- 3. Kucher N. Deep-vein thrombosis of the upper extremities. N Engl J Med 364: 861-869, 2011. [DOI] [PubMed] [Google Scholar]
- 4. Martinelli I, Battaglioli T, Bucciarelli P, Passamonti M, Mannucci PM. Risk factors and recurrence rate of primary deep vein thrombosis of the upper extremities. Circulation 110: 566-570, 2004. [DOI] [PubMed] [Google Scholar]
- 5. Mustafa S, Stein PD, Patel KC, Otten TR, Holmes R, Silbergleit A. Upper extremity deep vein thrombosis. Chest 123: 1953-1956, 2003. [DOI] [PubMed] [Google Scholar]
- 6. Ruggeri M, Castaman G, Tosetto A, Rodeghiero F. Low prevalence of thrombophilic coagulation defects in patients with deep vein thrombosis of the upper limbs. Blood Coagul Fibrynolysis 8: 191-194, 1997. [DOI] [PubMed] [Google Scholar]
- 7. Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Old and new risk factors for upper extremity deep venous thrombosis: result from the RIETE Registry. J Thromb Haemost 3: 2471-2478, 2005. [DOI] [PubMed] [Google Scholar]
- 8. Muñoz FJ, Mismetti P, Poggio R, et al. Clinical outcome of the patients with upper-extremity deep vein thrombosis. Chest 133: 143-148, 2008. [DOI] [PubMed] [Google Scholar]
- 9. Martinelli I, Cattaneo M, Panzeri D, Taioli E, Mannucci PM. Risk factors for deep venous thrombosis of the upper extremities. Ann Intern Med 126: 707-711, 1997. [DOI] [PubMed] [Google Scholar]
- 10. Joffe HV, Kucher N, Tapson VF, Goldhaber SZ. Upper-extremity deep vein thrombosis: a prospective registry of 592 patients. Circulation 110: 1605-1611, 2004. [DOI] [PubMed] [Google Scholar]
- 11. Spiezia L, Simioni P. Upper extremity deep vein thrombosis. Intern Emerg Med 5: 103-109, 2010. [DOI] [PubMed] [Google Scholar]
- 12. Raveendran M, Wang J, Senthil D, et al. Endogenous nitric oxide activation protects against cigarette smoking induced apoptosis in endothelial cells. FEBS Lett 579: 733-740, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Toda N, Toda H. Nitric oxide-mediated blood flow regulation as affected by smoking and nicotine. Eur J Pharmacol 649: 1-13, 2010. [DOI] [PubMed] [Google Scholar]
- 14. Rasmussen K, Moller J, Lyngbak M. Within-person variation of plasma homocysteine and effects of posture and tourniquet application. Clin Chem 45: 1850-1855, 1999. [PubMed] [Google Scholar]
- 15. Hughes SF, Hendricks BD, Edwards DR, Bastawrous SS, Roberts GE, Middleton JF. Mild episode of tourniquet-induced forearm ischaemia-reperfusion injury results in leukocyte activation and changes in inflammatory and coagulation markers. J Inflamm (Lond) 4: 12, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Zimmermann B, Scheel H, Wegener H. Trombophilia in blood-donation for plasma- and cytapheresis caused by antithrombin III depletion. Folia Haematol Int Mag Klin Morphol Blutforsch 115: 314-318, 1988. [PubMed] [Google Scholar]
- 17. Kobayashi I, Hamaoka S, Ozawa H, et al. Hypercoagulable state induced by thrombocytopheresis. J Clin Apher 8: 147-152, 1993. [DOI] [PubMed] [Google Scholar]
- 18. Sagedal S, Hartmann A, Sundstrøm K, Bjørnsen S, Brosstad F. Anticoagulation intensity sufficient for haemodialysis does not prevent activation of coagulation and platelets. Nephrol Dial Transplant 16: 987-993, 2001. [DOI] [PubMed] [Google Scholar]
- 19. Lucchi L, Ligabue G, Marietta M. Activation of coagulation during hemodialysis: effect of blood lines alone and whole extracorporeal circuit. Artif Organs 30: 106-110, 2006. [DOI] [PubMed] [Google Scholar]
- 20. Yilmaz M, Dikmen T, Sonmez M, et al. Change of coagulation parameters after double plateletpheresis. Transfus Apher Sci 37: 161-163, 2007. [DOI] [PubMed] [Google Scholar]
- 21. Siti Nadiah AK, Nor Asiah M, Nur Syimah AT, et al. Effects of platelet apheresis on blood coagulation parameters in healthy donors at National Blood Centre, Kuala Lumpur, Malaysia. Transfus Apher Sci 49: 507-510, 2013. [DOI] [PubMed] [Google Scholar]
- 22. Llop I, Blanquer A, Larrea L, Chaves A. Axilar thrombosis after platlet apheresis procedure. Transfusion 40 (Suppl): 47S, 2000. [Google Scholar]
- 23. Jones DA, Williams E, Riley SA, Makris M. Axillary vein thrombosis in a healthy donor following platelet apheresis. Br J Haematol 116: 390-391, 2002. [DOI] [PubMed] [Google Scholar]
- 24. Newman B, Rajpurkar M, Ozgonenel B, Lal A, Kuriakose P. Upper-extremity deep venous thrombosis after whole blood donation: report of three cases from a single blood center. Transfusion 55: 1290-1293, 2015. [DOI] [PubMed] [Google Scholar]
- 25. Covin RB, Rich NL, Aysola A. Upper-extremity deep venous thrombosis complicating whole-blood donation. Transfusion 44: 586-590, 2004. [DOI] [PubMed] [Google Scholar]
- 26. Featherstone T, Bayliss AP. Deep venous thrombosis of the upper extremity - a case report. Angiology 38: 793-796, 1987. [DOI] [PubMed] [Google Scholar]
- 27. Bell BM, Pink JM, Keller AJ. Upper limb venous thrombosis complicating blood donation. Blood Transfusion 12 (Suppl 2): S455, 2014. [Google Scholar]



