Abstract
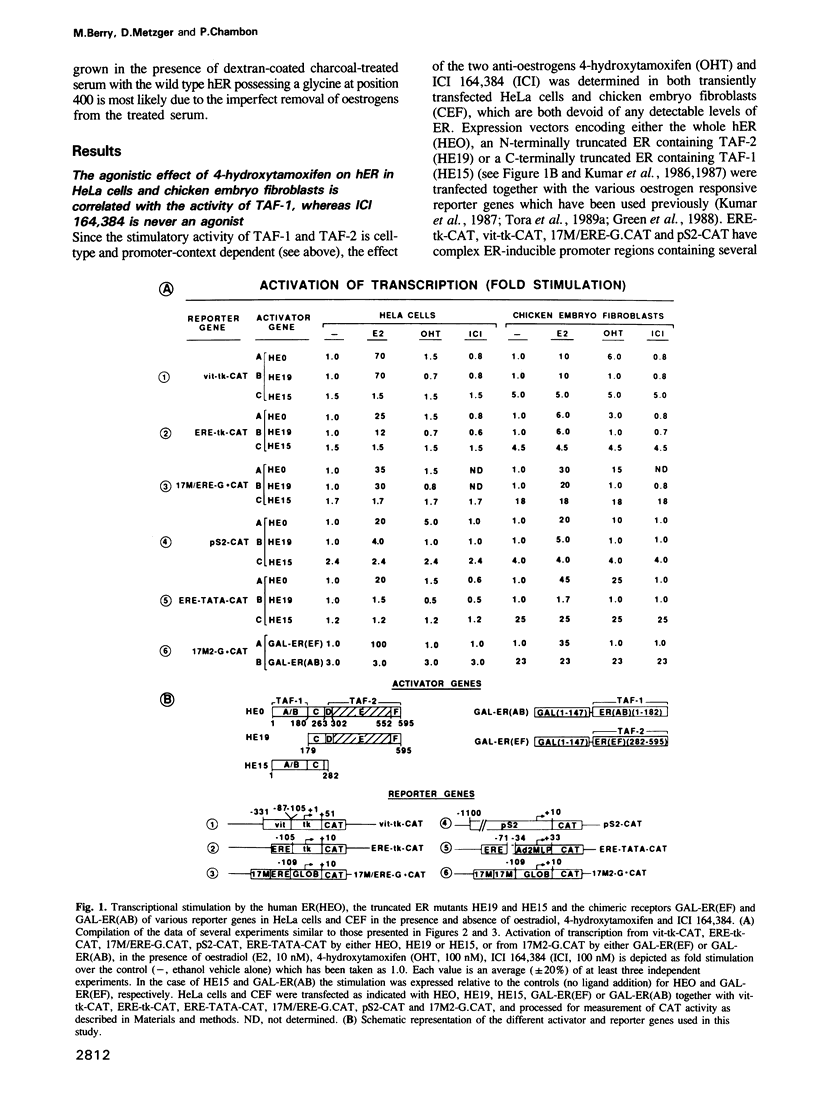
Various oestrogen responsive reporter genes and vectors expressing truncated or chimeric human oestrogen receptors (hER) containing either of the two independent hER transcriptional activation functions (TAF-1 and TAF-2) have been transfected into HeLa cells, chicken embryo fibroblast (CEF) or yeast cells to investigate the agonistic activity of the anti-oestrogen 4-hydroxytamoxifen (OHT). We demonstrate that the agonistic effect of OHT on the whole hER is due to the cell-type and promoter-context dependent activity of TAF-1. In similar experiments, we show that the anti-oestrogen, ICI 164,384, does not exhibit any oestrogenic activity and, therefore, acts always as a pure antagonist, even though it does not inhibit the activity of the isolated TAF-1. We also confirm that the wild type human oestrogen receptor has no ligand independent transcriptional activity. The implications of our results for the variable antagonist/agonist activity of anti-oestrogens in vivo are discussed.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anzai Y., Holinka C. F., Kuramoto H., Gurpide E. Stimulatory effects of 4-hydroxytamoxifen on proliferation of human endometrial adenocarcinoma cells (Ishikawa line). Cancer Res. 1989 May 1;49(9):2362–2365. [PubMed] [Google Scholar]
- Beato M. Gene regulation by steroid hormones. Cell. 1989 Feb 10;56(3):335–344. doi: 10.1016/0092-8674(89)90237-7. [DOI] [PubMed] [Google Scholar]
- Berry M., Nunez A. M., Chambon P. Estrogen-responsive element of the human pS2 gene is an imperfectly palindromic sequence. Proc Natl Acad Sci U S A. 1989 Feb;86(4):1218–1222. doi: 10.1073/pnas.86.4.1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berthois Y., Katzenellenbogen J. A., Katzenellenbogen B. S. Phenol red in tissue culture media is a weak estrogen: implications concerning the study of estrogen-responsive cells in culture. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2496–2500. doi: 10.1073/pnas.83.8.2496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bignon E., Pons M., Gilbert J., Crastes de Paulet A. Analogies and differences in the modulation of progesterone receptor induction and cell proliferation by estrogens and antiestrogens in MCF-7 human breast cancer cells: study with 24 triphenylacrylonitrile derivatives. J Steroid Biochem. 1988 Dec;31(6):877–885. doi: 10.1016/0022-4731(88)90328-7. [DOI] [PubMed] [Google Scholar]
- Boccardo F., Bruzzi P., Rubagotti A., Nicolò G. U., Rosso R. Estrogen-like action of tamoxifen on vaginal epithelium in breast cancer patients. Oncology. 1981;38(5):281–285. doi: 10.1159/000225571. [DOI] [PubMed] [Google Scholar]
- Cavailles V., Augereau P., Garcia M., Rochefort H. Estrogens and growth factors induce the mRNA of the 52K-pro-cathepsin-D secreted by breast cancer cells. Nucleic Acids Res. 1988 Mar 25;16(5):1903–1919. doi: 10.1093/nar/16.5.1903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrazzi E., Cartei G., Mattarazzo R., Fiorentino M. Oestrogen-like effect of tamoxifen on vaginal epithelium. Br Med J. 1977 May 21;1(6072):1351–1352. doi: 10.1136/bmj.1.6072.1351-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fornander T., Rutqvist L. E., Cedermark B., Glas U., Mattsson A., Silfverswärd C., Skoog L., Somell A., Theve T., Wilking N. Adjuvant tamoxifen in early breast cancer: occurrence of new primary cancers. Lancet. 1989 Jan 21;1(8630):117–120. doi: 10.1016/s0140-6736(89)91141-0. [DOI] [PubMed] [Google Scholar]
- Friedl A., Gottardis M. M., Pink J., Buchler D. A., Jordan V. C. Enhanced growth of an estrogen receptor-negative endometrial adenocarcinoma by estradiol in athymic mice. Cancer Res. 1989 Sep 1;49(17):4758–4764. [PubMed] [Google Scholar]
- Furr B. J., Jordan V. C. The pharmacology and clinical uses of tamoxifen. Pharmacol Ther. 1984;25(2):127–205. doi: 10.1016/0163-7258(84)90043-3. [DOI] [PubMed] [Google Scholar]
- Gottardis M. M., Robinson S. P., Satyaswaroop P. G., Jordan V. C. Contrasting actions of tamoxifen on endometrial and breast tumor growth in the athymic mouse. Cancer Res. 1988 Feb 15;48(4):812–815. [PubMed] [Google Scholar]
- Green S., Chambon P. Nuclear receptors enhance our understanding of transcription regulation. Trends Genet. 1988 Nov;4(11):309–314. doi: 10.1016/0168-9525(88)90108-4. [DOI] [PubMed] [Google Scholar]
- Green S., Kumar V., Theulaz I., Wahli W., Chambon P. The N-terminal DNA-binding 'zinc finger' of the oestrogen and glucocorticoid receptors determines target gene specificity. EMBO J. 1988 Oct;7(10):3037–3044. doi: 10.1002/j.1460-2075.1988.tb03168.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green S., Walter P., Kumar V., Krust A., Bornert J. M., Argos P., Chambon P. Human oestrogen receptor cDNA: sequence, expression and homology to v-erb-A. Nature. 1986 Mar 13;320(6058):134–139. doi: 10.1038/320134a0. [DOI] [PubMed] [Google Scholar]
- Ham J., Parker M. G. Regulation of gene expression by nuclear hormone receptors. Curr Opin Cell Biol. 1989 Jun;1(3):503–511. doi: 10.1016/0955-0674(89)90012-4. [DOI] [PubMed] [Google Scholar]
- Horwitz K. B., Koseki Y., McGuire W. L. Estrogen control of progesterone receptor in human breast cancer: role of estradiol and antiestrogen. Endocrinology. 1978 Nov;103(5):1742–1751. doi: 10.1210/endo-103-5-1742. [DOI] [PubMed] [Google Scholar]
- Horwitz K. B., McGuire W. L. Nuclear mechanisms of estrogen action. Effects of estradiol and anti-estrogens on estrogen receptors and nuclear receptor processing. J Biol Chem. 1978 Nov 25;253(22):8185–8191. [PubMed] [Google Scholar]
- Jordan V. C. Biochemical pharmacology of antiestrogen action. Pharmacol Rev. 1984 Dec;36(4):245–276. [PubMed] [Google Scholar]
- Jordan V. C., Phelps E., Lindgren J. U. Effects of anti-estrogens on bone in castrated and intact female rats. Breast Cancer Res Treat. 1987 Oct;10(1):31–35. doi: 10.1007/BF01806132. [DOI] [PubMed] [Google Scholar]
- Katzenellenbogen B. S., Kendra K. L., Norman M. J., Berthois Y. Proliferation, hormonal responsiveness, and estrogen receptor content of MCF-7 human breast cancer cells grown in the short-term and long-term absence of estrogens. Cancer Res. 1987 Aug 15;47(16):4355–4360. [PubMed] [Google Scholar]
- Krust A., Green S., Argos P., Kumar V., Walter P., Bornert J. M., Chambon P. The chicken oestrogen receptor sequence: homology with v-erbA and the human oestrogen and glucocorticoid receptors. EMBO J. 1986 May;5(5):891–897. doi: 10.1002/j.1460-2075.1986.tb04300.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar V., Chambon P. The estrogen receptor binds tightly to its responsive element as a ligand-induced homodimer. Cell. 1988 Oct 7;55(1):145–156. doi: 10.1016/0092-8674(88)90017-7. [DOI] [PubMed] [Google Scholar]
- Kumar V., Green S., Stack G., Berry M., Jin J. R., Chambon P. Functional domains of the human estrogen receptor. Cell. 1987 Dec 24;51(6):941–951. doi: 10.1016/0092-8674(87)90581-2. [DOI] [PubMed] [Google Scholar]
- Kumar V., Green S., Staub A., Chambon P. Localisation of the oestradiol-binding and putative DNA-binding domains of the human oestrogen receptor. EMBO J. 1986 Sep;5(9):2231–2236. doi: 10.1002/j.1460-2075.1986.tb04489.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lees J. A., Fawell S. E., Parker M. G. Identification of two transactivation domains in the mouse oestrogen receptor. Nucleic Acids Res. 1989 Jul 25;17(14):5477–5488. doi: 10.1093/nar/17.14.5477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Love R. R., Mazess R. B., Tormey D. C., Barden H. S., Newcomb P. A., Jordan V. C. Bone mineral density in women with breast cancer treated with adjuvant tamoxifen for at least two years. Breast Cancer Res Treat. 1988 Dec;12(3):297–302. doi: 10.1007/BF01811242. [DOI] [PubMed] [Google Scholar]
- Martinez E., Wahli W. Cooperative binding of estrogen receptor to imperfect estrogen-responsive DNA elements correlates with their synergistic hormone-dependent enhancer activity. EMBO J. 1989 Dec 1;8(12):3781–3791. doi: 10.1002/j.1460-2075.1989.tb08555.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May F. E., Johnson M. D., Wiseman L. R., Wakeling A. E., Kastner P., Westley B. R. Regulation of progesterone receptor mRNA by oestradiol and antioestrogens in breast cancer cell lines. J Steroid Biochem. 1989 Dec;33(6):1035–1041. doi: 10.1016/0022-4731(89)90406-8. [DOI] [PubMed] [Google Scholar]
- May F. E., Westley B. R. Effects of tamoxifen and 4-hydroxytamoxifen on the pNR-1 and pNR-2 estrogen-regulated RNAs in human breast cancer cells. J Biol Chem. 1987 Nov 25;262(33):15894–15899. [PubMed] [Google Scholar]
- Metzger D., White J. H., Chambon P. The human oestrogen receptor functions in yeast. Nature. 1988 Jul 7;334(6177):31–36. doi: 10.1038/334031a0. [DOI] [PubMed] [Google Scholar]
- Pasqualini J. R., Sumida C., Giambiagi N. Pharmacodynamic and biological effects of anti-estrogens in different models. J Steroid Biochem. 1988 Oct;31(4B):613–643. doi: 10.1016/0022-4731(88)90013-1. [DOI] [PubMed] [Google Scholar]
- Satyaswaroop P. G., Zaino R. J., Mortel R. Estrogen-like effects of tamoxifen on human endometrial carcinoma transplanted into nude mice. Cancer Res. 1984 Sep;44(9):4006–4010. [PubMed] [Google Scholar]
- Sonnenschein C., Papendorp J. T., Soto A. M. Estrogenic effect of tamoxifen and its derivatives on the proliferation of MCF7 human breast tumor cells. Life Sci. 1985 Jul 29;37(4):387–394. doi: 10.1016/0024-3205(85)90510-7. [DOI] [PubMed] [Google Scholar]
- Thompson E. W., Katz D., Shima T. B., Wakeling A. E., Lippman M. E., Dickson R. B. ICI 164,384, a pure antagonist of estrogen-stimulated MCF-7 cell proliferation and invasiveness. Cancer Res. 1989 Dec 15;49(24 Pt 1):6929–6934. [PubMed] [Google Scholar]
- Tora L., Mullick A., Metzger D., Ponglikitmongkol M., Park I., Chambon P. The cloned human oestrogen receptor contains a mutation which alters its hormone binding properties. EMBO J. 1989 Jul;8(7):1981–1986. doi: 10.1002/j.1460-2075.1989.tb03604.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tora L., White J., Brou C., Tasset D., Webster N., Scheer E., Chambon P. The human estrogen receptor has two independent nonacidic transcriptional activation functions. Cell. 1989 Nov 3;59(3):477–487. doi: 10.1016/0092-8674(89)90031-7. [DOI] [PubMed] [Google Scholar]
- Wakeling A. E., Bowler J. Biology and mode of action of pure antioestrogens. J Steroid Biochem. 1988;30(1-6):141–147. doi: 10.1016/0022-4731(88)90086-6. [DOI] [PubMed] [Google Scholar]
- Wakeling A. E., Bowler J. Novel antioestrogens without partial agonist activity. J Steroid Biochem. 1988 Oct;31(4B):645–653. doi: 10.1016/0022-4731(88)90014-3. [DOI] [PubMed] [Google Scholar]
- Wakeling A. E. Comparative studies on the effects of steroidal and nonsteroidal oestrogen antagonists on the proliferation of human breast cancer cells. J Steroid Biochem. 1989;34(1-6):183–188. doi: 10.1016/0022-4731(89)90081-2. [DOI] [PubMed] [Google Scholar]
- Walter P., Green S., Greene G., Krust A., Bornert J. M., Jeltsch J. M., Staub A., Jensen E., Scrace G., Waterfield M. Cloning of the human estrogen receptor cDNA. Proc Natl Acad Sci U S A. 1985 Dec;82(23):7889–7893. doi: 10.1073/pnas.82.23.7889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weatherill P. J., Wilson A. P., Nicholson R. I., Davies P., Wakeling A. E. Interaction of the antioestrogen ICI 164,384 with the oestrogen receptor. J Steroid Biochem. 1988;30(1-6):263–266. doi: 10.1016/0022-4731(88)90103-3. [DOI] [PubMed] [Google Scholar]
- Webster N. J., Green S., Jin J. R., Chambon P. The hormone-binding domains of the estrogen and glucocorticoid receptors contain an inducible transcription activation function. Cell. 1988 Jul 15;54(2):199–207. doi: 10.1016/0092-8674(88)90552-1. [DOI] [PubMed] [Google Scholar]
- Webster N. J., Green S., Tasset D., Ponglikitmongkol M., Chambon P. The transcriptional activation function located in the hormone-binding domain of the human oestrogen receptor is not encoded in a single exon. EMBO J. 1989 May;8(5):1441–1446. doi: 10.1002/j.1460-2075.1989.tb03526.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westley B. R., Hölzel F., May F. E. Effects of oestrogen and the antioestrogens, tamoxifen and LY117018, on four oestrogen-regulated RNAs in the EFM-19 breast cancer cell line. J Steroid Biochem. 1989 Mar;32(3):365–372. doi: 10.1016/0022-4731(89)90208-2. [DOI] [PubMed] [Google Scholar]
- White J. H., Metzger D., Chambon P. Expression and function of the human estrogen receptor in yeast. Cold Spring Harb Symp Quant Biol. 1988;53(Pt 2):819–828. doi: 10.1101/sqb.1988.053.01.093. [DOI] [PubMed] [Google Scholar]
- White R., Lees J. A., Needham M., Ham J., Parker M. Structural organization and expression of the mouse estrogen receptor. Mol Endocrinol. 1987 Oct;1(10):735–744. doi: 10.1210/mend-1-10-735. [DOI] [PubMed] [Google Scholar]
- Wilson A. P., Weatherill P. J., Nicholson R. I., Davies P., Wakeling A. E. A comparative study of the interaction of oestradiol and the steroidal pure antioestrogen, ICI 164,384, with the molybdate-stabilized oestrogen receptor. J Steroid Biochem. 1990 Mar;35(3-4):421–428. doi: 10.1016/0022-4731(90)90250-v. [DOI] [PubMed] [Google Scholar]
- Wiseman L. R., Wakeling A. E., May F. E., Westley B. R. Effects of the antioestrogen, ICI 164,384, on oestrogen induced RNAs in MCF-7 cells. J Steroid Biochem. 1989 Jul;33(1):1–6. doi: 10.1016/0022-4731(89)90349-x. [DOI] [PubMed] [Google Scholar]