Abstract
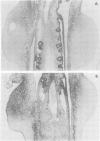
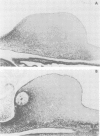
Homeobox gene XlHbox 1 is expressed in a mesodermal gradient in vertebrate forelimbs with maximal expression anteriorly and proximally and may encode positional values. In chick wing buds, anterior cells can be reprogrammed to form posterior structures by grafts of polarizing region tissue and by beads soaked in retinoic acid (RA), which is a good candidate for an endogenous morphogen. Applications of RA anteriorly or at the bud apex, treatments which produce duplicated digits or truncations respectively, substantially increase the extent of mesodermal XlHbox 1 expression. Polarizing region grafts that also produce additional digits lead to a moderate increase. The effects of RA application and the behaviour of transplanted tissue show that only anterior cells are competent to express XlHbox 1 and that expression is cell autonomous. Ectodermal expression in wing buds is enhanced by RA but not by polarizing region grafts and ectoderm/mesoderm recombinations show that the mesoderm is irreversibly affected. The changes in mesodermal expression do not fit the predictions of the simple model that XlHbox 1 encodes anterior positional values but are correlated with a series of novel malformations of the shoulder girdle which, in normal wing buds, is derived from cells expressing XlHbox 1.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brockes J. P. Retinoids, homeobox genes, and limb morphogenesis. Neuron. 1989 Apr;2(4):1285–1294. doi: 10.1016/0896-6273(89)90066-4. [DOI] [PubMed] [Google Scholar]
- Dollé P., Duboule D. Two gene members of the murine HOX-5 complex show regional and cell-type specific expression in developing limbs and gonads. EMBO J. 1989 May;8(5):1507–1515. doi: 10.1002/j.1460-2075.1989.tb03535.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dollé P., Izpisúa-Belmonte J. C., Falkenstein H., Renucci A., Duboule D. Coordinate expression of the murine Hox-5 complex homoeobox-containing genes during limb pattern formation. Nature. 1989 Dec 14;342(6251):767–772. doi: 10.1038/342767a0. [DOI] [PubMed] [Google Scholar]
- Dollé P., Ruberte E., Kastner P., Petkovich M., Stoner C. M., Gudas L. J., Chambon P. Differential expression of genes encoding alpha, beta and gamma retinoic acid receptors and CRABP in the developing limbs of the mouse. Nature. 1989 Dec 7;342(6250):702–705. doi: 10.1038/342702a0. [DOI] [PubMed] [Google Scholar]
- Durston A. J., Timmermans J. P., Hage W. J., Hendriks H. F., de Vries N. J., Heideveld M., Nieuwkoop P. D. Retinoic acid causes an anteroposterior transformation in the developing central nervous system. Nature. 1989 Jul 13;340(6229):140–144. doi: 10.1038/340140a0. [DOI] [PubMed] [Google Scholar]
- Eichele G. Retinoids and vertebrate limb pattern formation. Trends Genet. 1989 Aug;5(8):246–251. doi: 10.1016/0168-9525(89)90096-6. [DOI] [PubMed] [Google Scholar]
- Gehring W. J. Homeo boxes in the study of development. Science. 1987 Jun 5;236(4806):1245–1252. doi: 10.1126/science.2884726. [DOI] [PubMed] [Google Scholar]
- Graham A., Papalopulu N., Krumlauf R. The murine and Drosophila homeobox gene complexes have common features of organization and expression. Cell. 1989 May 5;57(3):367–378. doi: 10.1016/0092-8674(89)90912-4. [DOI] [PubMed] [Google Scholar]
- Maden M. The effect of vitamin A on limb regeneration in Rana temporaria. Dev Biol. 1983 Aug;98(2):409–416. doi: 10.1016/0012-1606(83)90370-6. [DOI] [PubMed] [Google Scholar]
- Molven A., Wright C. V., Bremiller R., De Robertis E. M., Kimmel C. B. Expression of a homeobox gene product in normal and mutant zebrafish embryos: evolution of the tetrapod body plan. Development. 1990 Jun;109(2):279–288. doi: 10.1242/dev.109.2.279. [DOI] [PubMed] [Google Scholar]
- Oliver G., Sidell N., Fiske W., Heinzmann C., Mohandas T., Sparkes R. S., De Robertis E. M. Complementary homeo protein gradients in developing limb buds. Genes Dev. 1989 May;3(5):641–650. doi: 10.1101/gad.3.5.641. [DOI] [PubMed] [Google Scholar]
- Oliver G., Wright C. V., Hardwicke J., De Robertis E. M. A gradient of homeodomain protein in developing forelimbs of Xenopus and mouse embryos. Cell. 1988 Dec 23;55(6):1017–1024. doi: 10.1016/0092-8674(88)90246-2. [DOI] [PubMed] [Google Scholar]
- Oliver G., Wright C. V., Hardwicke J., De Robertis E. M. Differential antero-posterior expression of two proteins encoded by a homeobox gene in Xenopus and mouse embryos. EMBO J. 1988 Oct;7(10):3199–3209. doi: 10.1002/j.1460-2075.1988.tb03187.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SZABO G. A modification of the technique of skin splitting with trypsin. J Pathol Bacteriol. 1955 Oct;70(2):545–545. doi: 10.1002/path.1700700233. [DOI] [PubMed] [Google Scholar]
- Scott M. P., Tamkun J. W., Hartzell G. W., 3rd The structure and function of the homeodomain. Biochim Biophys Acta. 1989 Jul 28;989(1):25–48. doi: 10.1016/0304-419x(89)90033-4. [DOI] [PubMed] [Google Scholar]
- Summerbell D. The zone of polarizing activity: evidence for a role in normal chick limb morphogenesis. J Embryol Exp Morphol. 1979 Apr;50:217–233. [PubMed] [Google Scholar]
- Thaller C., Eichele G. Characterization of retinoid metabolism in the developing chick limb bud. Development. 1988 Jul;103(3):473–483. doi: 10.1242/dev.103.3.473. [DOI] [PubMed] [Google Scholar]
- Thaller C., Eichele G. Identification and spatial distribution of retinoids in the developing chick limb bud. Nature. 1987 Jun 18;327(6123):625–628. doi: 10.1038/327625a0. [DOI] [PubMed] [Google Scholar]
- Tickle C., Alberts B., Wolpert L., Lee J. Local application of retinoic acid to the limb bond mimics the action of the polarizing region. Nature. 1982 Apr 8;296(5857):564–566. doi: 10.1038/296564a0. [DOI] [PubMed] [Google Scholar]
- Tickle C., Crawley A., Farrar J. Retinoic acid application to chick wing buds leads to a dose-dependent reorganization of the apical ectodermal ridge that is mediated by the mesenchyme. Development. 1989 Aug;106(4):691–705. doi: 10.1242/dev.106.4.691. [DOI] [PubMed] [Google Scholar]
- Tickle C., Lee J., Eichele G. A quantitative analysis of the effect of all-trans-retinoic acid on the pattern of chick wing development. Dev Biol. 1985 May;109(1):82–95. doi: 10.1016/0012-1606(85)90348-3. [DOI] [PubMed] [Google Scholar]
- Tickle C., Summerbell D., Wolpert L. Positional signalling and specification of digits in chick limb morphogenesis. Nature. 1975 Mar 20;254(5497):199–202. doi: 10.1038/254199a0. [DOI] [PubMed] [Google Scholar]