B-cell precursor acute lymphoblastic leukemia (BCP-ALL) is the most common type of cancer in children, representing up to 80% of pediatric, and ~20% of adult leukemias.1 BCP-ALL is divided into several genetic subtypes according to acquired chromosomal aberrations with varying prognosis.2 In children and young adults, BCP-ALL patients have an overall survival (OS) at 5 years around 90%. In contrast, OS in adults is much lower (30–40%), in part due to a higher frequency of ALL subtypes with poor prognosis.3, 4 Hence, identifying new biomarkers for patients with poor prognosis is important as it might allow the development of new approaches to treat high-risk BCP-ALL.
CD27 is a member of the tumor necrosis factor receptor superfamily that regulates lymphocyte function.5 Expression of CD27 protein and mRNA has been reported in B-cell lymphomas 6 and adult T-cell leukemia/lymphoma.7 In acute myeloid leukemia CD27 has been shown to be a prognostic biomarker.8 We previously described a pro-B-cell molecular signature enriched in the ETV6-RUNX1 subtype, and found that one of these genes is CD27.9 Consistent with this, previous studies have shown high mRNA and surface levels of CD27 in ETV6-RUNX1 BCP-ALL.10, 11 However, expression of CD27 in BCP-ALL of other subtypes and its potential clinical relevance is still unclear. Here, we determine the protein and mRNA expression pattern of CD27 during early B-cell development and in BCP-ALL, and investigate the prognostic relevance of CD27 mRNA expression in pediatric and adult patients with BCP-ALL.
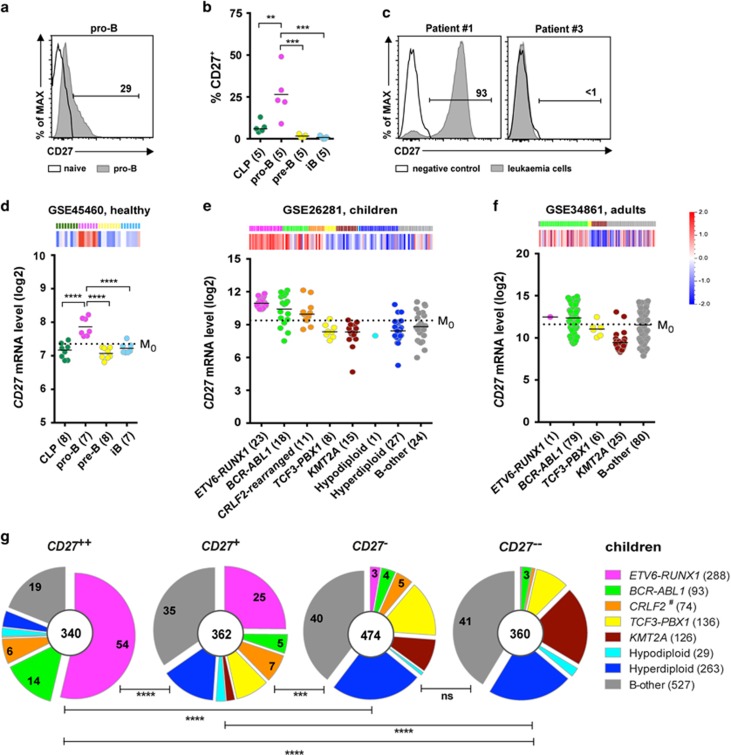
To confirm and extend previous observations,10, 11, 12 we first analyzed CD27 expression during bone marrow (BM) B-cell development using flow cytometry (gating strategy, Supplementary Figure S1a). Pro-B cells (CD19+CD34+IgM−) from all five donors expressed CD27, whereas common lymphoid progenitor (CLP, CD19−CD34+IgM−), pre-B (CD19+CD34−IgM−) and immature B (CD19+CD34−IgM+) cells did not (Figures 1a and b, Supplementary Figure S1b). We also noted that only a fraction, on average 25%, of the pro-B cells were positive (Figure 1b). Thereafter, we analyzed six patient samples, of which two expressed CD27 at the time of diagnosis (Figure 1c and Supplementary Figure S1c). The CD27 levels in these two cases, classified as B-other, were much higher than those in the healthy pro-B cells, with the vast majority of the CD19+ leukemic cells being strongly positive. Consistent with the CD27 protein expression pattern, we found that CD27 mRNA levels were high in pro-B cells as compared to CLP, pre-B and immature B cells (Figure 1d). To determine which BCP-ALL subtypes express CD27 we analyzed its mRNA levels in public data sets (Supplementary Table S1). In pediatric leukemia samples, for example, data set GSE26281, CD27 was as expected highly expressed in over 90% of ETV6-RUNX1, compared to the mean level of the total samples (M0) (Figure 1e). In addition, more than half of BCR-ABL1 and CRLF2-rearranged, and a third of B-other BCP-ALL also expressed high levels of CD27 mRNA. In adult BCP-ALL, for example, data set GSE34861, CD27 was highly expressed in more than 60% of BCR-ABL1 and on average in 45% of B-other BCP-ALL compared to the mean level of the total samples (M0) in each data set (Figure 1f). Thus, CD27 is not only highly expressed in the ETV6-RUNX1, but also in BCR-ABL1, CRLF2-rearranged and B-other BCP-ALL.
Figure 1.
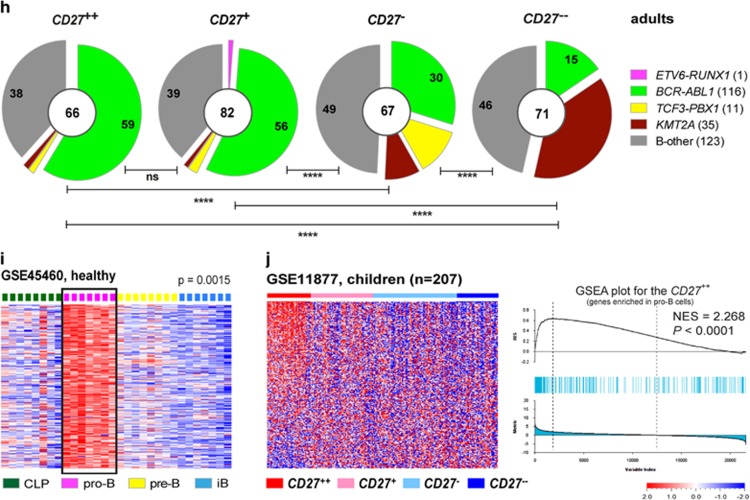
Comparison of CD27 expression and molecular signature between pro-B cells and BCP-ALL. (a) Representative histogram shows surface CD27 expression in pro-B cells. (b) Scatter plot shows percentages of CD27+ cells in indicated subsets of BM samples from five healthy donors. (c) Representative histograms show CD27 surface expression in two BCP-ALL samples (CD27+ and CD27−). (d, e, f) Heat maps and scatter plots show CD27 mRNA expression levels in (d) healthy (GSE45460) BM; (e) pediatric (GSE26281) and (f) adult (GSE34861) BCP-ALL samples. Dashed line in scatter plots represents M0. (g and h) Pie charts show the proportions of BCP-ALL subtypes within each CD27 cluster based on meta-analyzes of (g) eight pediatric data sets (GSE26281, GSE33315, GSE13576, GSE13425, GSE12995, Blood 2003, GSE11877, GSE47051) and (h) two adult data sets (GSE34861 and CCR 2005), after classifying BCP-ALL samples in each data set into four groups according to CD27 expression levels: CD27++ (>M1), CD27+ (<M1 to >M0), CD27− (<M0 to >M−1), CD27−− (<M−1). Numbers in the center of pie charts represent number of patient samples, and those in segments the proportions of the indicated subtype. M0: mean expression level of CD27 in all samples; M1: mean expression of CD27 in samples with levels above M0; M−1: mean expression of CD27 in samples with levels below M0. (i) Heat map shows genes highly expressed in pro-B cells (pro-B signature). Genes are selected according to the criteria: P-value (<0.05), q-value (<0.1) and fold change (>1.5). (j) Heat map (left) and GSEA enrichment plots (right) reveals a pro-B molecular signature in CD27++ high-risk pediatric BCP-ALL data set (GSE11877). CLP, common lymphoid progenitor; iB, immature B. #CRLF2-rearranged (previously defined) and CRLF2-high (defined herein) were pooled. Statistical analysis: (b and d) one-way ANOVA and (g and h) χ2 analysis. **P < 0.01; ***P<0.001; ****P<0.0001.
The CRLF2-rearranged subtype was only recently defined13, 14 and, therefore, these patient samples are found within B-other in most public data sets. To determine the CRLF2-rearranged subtype in data sets where this has not been defined, the CRLF2 expression levels in already defined CRLF2-rearranged samples (Supplementary Table S1, GSE26281 and GSE11877) were analyzed. We found that the expression levels of CRLF2 in all CRLF2-rearranged samples were at least 10-fold higher than the median levels of total samples (Supplementary Figure S2a). Based on this, samples expressing 10-fold higher level of CRLF2 than the median are referred to as CRLF2-high in other data sets (Supplementary Figure S2b) with the assumption that most of these are CRLF2-rearranged. Thereafter, we queried which subtypes were enriched in samples with high CD27 expression levels, by performing meta-analyses based on eight data sets with over 1500 pediatric patient samples in total and two data sets with over 250 adult patient samples (Supplementary Table S1). Samples in each data set were first divided into four clusters (CD27++, CD27+, CD27− and CD27−−) according to CD27 mRNA levels (Supplementary Figure S2c) and thereafter pooled. Subtype distribution analysis in pediatric BCP-ALL showed that ETV6-RUNX1, BCR-ABL1 and CRLF2-rearranged/high were proportionally enriched in the CD27++ and CD27+ clusters, whereas the opposite was observed for, for example, KMT2A-rearranged (Figure 1g). Also in adult samples, the BCR-ABL1 subtype was enriched in the CD27++ and CD27+ clusters, and the opposite was observed for, for example, KMT2A-rearranged (Figure 1h). Thus, pediatric and adult BCP-ALL showed similar expression patterns of CD27.
Because CD27 mRNA is highly expressed in the ETV6-RUNX1 subtype that display a pro-B-cell molecular signature,9 we hypothesized that BCP-ALL samples with high CD27 mRNA levels would also display a pro-B signature (Figure 1i). To test this hypothesis, we performed gene set enrichment analyses (GSEA) in leukemia data sets after excluding the ETV6-RUNX1 subtype. Independent of genetic subtype, pediatric BCP-ALL expressing high CD27 mRNA levels (CD27++) showed a molecular signature similar to pro-B cells (Figure 1j and Supplementary Figure S3). However, we were unable to find a pro-B signature in adult BCP-ALL, which indicates that the molecular signature is different in children and adults.
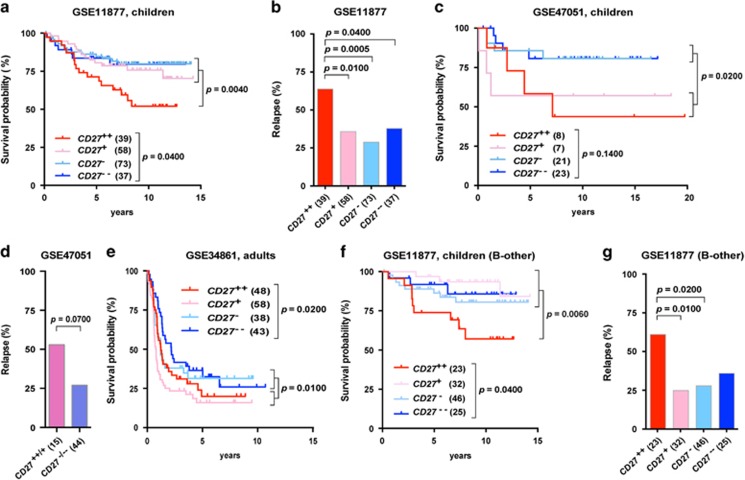
Since CD27 mRNA was highly expressed in BCR-ABL1 and CRLF2-rearranged/high BCP-ALL that are associated with poor prognosis, we asked whether CD27 has prognostic value. To determine this, we first analyzed clinical data available from 207 high-risk pediatric BCP-ALL patients (Supplementary Table S1, GSE11877). Dividing the patient samples into four clusters according to CD27 expression levels, we found that the CD27++ cluster was associated with poor OS compared with the other clusters (Figure 2a). Moreover, approximately 60% of patients in the CD27++ cluster had relapse compared to 25–30% in the remaining clusters (Figure 2b). Thus, CD27 expression levels correlate with clinical outcome in this cohort with high-risk pediatric BCP-ALL patients. To further confirm this observation, we analyzed the clinical data available from an additional pediatric cohort including 75 patients (Supplementary Table S1, GSE47051). Because the ETV6-RUNX1 subtype is associated with good prognosis in pediatric BCP-ALL,2 we excluded this subtype in the analyses. We did not detect a significant difference in OS between the clusters (Figure 2c), whereas a significantly lower OS was observed in the combined CD27++/+ compared to the CD27−/−− clusters (Figure 2c). There was also a trend for a higher proportion of relapse in the former (Figure 2d). We also analyzed the prognostic relevance of CD27 expression levels in adults with BCP-ALL in the cohort with available clinical data (Supplementary Table S1, GSE34861). This showed significantly different OS for patients in the CD27 clusters (Figure 2e). Thus, this suggests that CD27 could be a marker for poor prognosis for non-ETV6-RUNX1 BCP-ALL both in children and adults. Our observation is in contrast to a previous study that did not find any significant differences in OS between CD27 positive and negative BCP-ALL.11 However, this previous study included a low proportion of high-risk patients, and the follow-up time was shorter than 3 years. In our study, all the cohorts included at least a fifth of high-risk BCP-ALL, and the follow-up time was much longer than 3 years.
Figure 2.
Clinical relevance of CD27 mRNA levels in patients with BCP-ALL. (a, c, e, f) Kaplan–Meier Log-rank survival analysis was used to compare survival of patients within the indicated CD27 clusters. (b, d, g) Percentages of patients with relapse within indicated CD27 clusters using Fisher's exact test. (a, b) High-risk pediatric cohort GSE118877, (c, d) pediatric cohort GSE47051 (excluding ETV6-RUNX1); CD27++/+, CD27++ and CD27+; CD27-/—, CD27− and CD27−−, (e) adult cohort GSE34861 and (f, g) B-other patients within high-risk pediatric cohort GSE118877.
In B-other BCP-ALL with unknown or not classifying genetic aberrations there are few prognostic biomarkers. As CD27 showed differential expression pattern in B-other BCP-ALL, we asked whether CD27 could also be used as a marker for these types of leukemia. To investigate this, the clinical data from patients with B-other BCP-ALL in the above cohorts were analyzed. In the high-risk cohort (GSE11877) with 126 B-other samples, CD27++ mRNA levels were associated with poor OS (Figure 2f), and 60% of patients in the CD27++ cluster experienced relapse compared to 25–35% in the remaining clusters (Figure 2g). In the other pediatric cohort (GSE47051) with only 23 B-other samples, the CD27++/+ cluster showed a trend to poor OS (Supplementary Figure S4a). In addition, in adults with B-other BCP-ALL, the CD27++/+ cluster also showed a trend to poor OS (Supplementary Figure S4b). Taken together, these data demonstrate that high CD27 levels represent a poor prognostic marker for high-risk pediatric B-other BCP-ALL.
In conclusion, our findings that CD27 is highly expressed in high-risk pediatric and adult BCP-ALL indicates that this molecule might serve as a therapeutic target, especially for the BCR-ABL1, CRLF2-rearranged/high and B-other subtypes. A human anti-CD27 monoclonal antibody has been developed, and in vitro and in vivo studies have shown that this antibody can exert anti-tumor activity in lymphoma xenograft models.15 Future studies will concentrate on determining whether CD27 can indeed be used as diagnostic/prognostic markers, and whether anti-CD27 antibodies can be used to directly target BCP-ALL expressing surface CD27.
Acknowledgments
This work has been supported by grants from Barncancerfonden (TJ2014-0083, TJ2016-0007, PR2016-0144), Cancerfonden (CAN2014/886, CAN2016/0668), AG Fond (FB 15-57, FB 16-15), Lions Cancerfond (2014:20), ALF, IngaBritt och Arne Lundbergs Forskning Stiftelse, Stiftelsen Wilhelm och Martina Lundgrens Vetenskap, Adlerbertska forskningsstiftelsen, KVVS.
Footnotes
Supplementary Information accompanies this paper on Blood Cancer Journal website (http://www.nature.com/bcj)
The authors declare no conflict of interest.
Supplementary Material
References
- Zuckerman T, Rowe JM. Pathogenesis and prognostication in acute lymphoblastic leukemia. F1000Prime Rep 2014; 6: 59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts KG, Mullighan CG. Genomics in acute lymphoblastic leukaemia: insights and treatment implications. Nat Rev Clin Oncol 2015; 12: 344–357. [DOI] [PubMed] [Google Scholar]
- Boer JM, Koenders JE, van der Holt B, Exalto C, Sanders MA, Cornelissen JJ et al. Expression profiling of adult acute lymphoblastic leukemia identifies a BCR-ABL1-like subgroup characterized by high non-response and relapse rates. Haematologica 2015; 100: e261–e264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toft N, Birgens H, Abrahamsson J, Bernell P, Griskevicius L, Hallbook H et al. Risk group assignment differs for children and adults 1-45yr with acute lymphoblastic leukemia treated by the NOPHO ALL-2008 protocol. Eur J Haematol 2013; 90: 404–412. [DOI] [PubMed] [Google Scholar]
- Gravestein LA, Borst J. Tumor necrosis factor receptor family members in the immune system. Semin Immunol 1998; 10: 423–434. [DOI] [PubMed] [Google Scholar]
- Dong HY, Shahsafaei A, Dorfman DM. CD148 and CD27 are expressed in B cell lymphomas derived from both memory and naive B cells. Leuk Lymphoma 2002; 43: 1855–1858. [DOI] [PubMed] [Google Scholar]
- Shao H, Yuan CM, Xi L, Raffeld M, Morris JC, Janik JE et al. Minimal residual disease detection by flow cytometry in adult T-cell leukemia/lymphoma. Am J Clin Pathol 2010; 133: 592–601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riether C, Schurch CM, Buhrer ED, Hinterbrandner M, Huguenin AL, Hoepner S et al. CD70/CD27 signaling promotes blast stemness and is a viable therapeutic target in acute myeloid leukemia. J Exp Med 2016; 214: 359–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D, Zheng J, Gerasimcik N, Lagerstedt K, Sjogren H, Abrahamsson J et al. The expression pattern of the pre-B cell receptor components correlates with cellular stage and clinical outcome in acute lymphoblastic leukemia. PLoS One 2016; 11: e0162638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaskova M, Mejstrikova E, Kalina T, Martinkova P, Omelka M, Trka J et al. Transfer of genomics information to flow cytometry: expression of CD27 and CD44 discriminates subtypes of acute lymphoblastic leukemia. Leukemia 2005; 19: 876–878. [DOI] [PubMed] [Google Scholar]
- Kamazani FM, Bahoush GR, Aghaeipour M, Vaeli S, Amirghofran Z. CD44 and CD27 expression pattern in B cell precursor acute lymphoblastic leukemia and its clinical significance. Med Oncol 2013; 30: 359. [DOI] [PubMed] [Google Scholar]
- McWilliams L, Su KY, Liang X, Liao D, Floyd S, Amos J et al. The human fetal lymphocyte lineage: identification by CD27 and LIN28B expression in B cell progenitors. J Leukoc Biol 2013; 94: 991–1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullighan CG, Collins-Underwood JR, Phillips LA, Loudin MG, Liu W, Zhang J et al. Rearrangement of CRLF2 in B-progenitor- and Down syndrome-associated acute lymphoblastic leukemia. Nat Genet 2009; 41: 1243–1246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell LJ, Capasso M, Vater I, Akasaka T, Bernard OA, Calasanz MJ et al. Deregulated expression of cytokine receptor gene, CRLF2, is involved in lymphoid transformation in B-cell precursor acute lymphoblastic leukemia. Blood 2009; 114: 2688–2698. [DOI] [PubMed] [Google Scholar]
- Vitale LA, He LZ, Thomas LJ, Widger J, Weidlick J, Crocker A et al. Development of a human monoclonal antibody for potential therapy of CD27-expressing lymphoma and leukemia. Clin Cancer Res 2012; 18: 3812–3821. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.