Abstract
Pain features centrally in numerous illnesses and generates enormous public health costs. Despite its ubiquity, pain’s psychological and neurophysiological nature remains controversial. Here we survey one controversy in particular: the relationship between nociceptive pain, which is somatic in origin, and empathic pain, which arises from observing others in pain. First, we review evidence for neural overlap between nociceptive and empathic pain and what this overlap implies about underlying mental representations. Then, we propose a framework for understanding the nature of the psychological and neurophysiological correspondence across these types of “pain.” This framework suggests new directions for research that can better identify shared and dissociable representations underlying different types of distress, and can inform theories about the nature of pain.
Keywords: Pain, empathy, multivariate pattern analysis, anterior insula & ACC, nociception, fMRI
Nociceptive and Empathic Pain
Imagine accidentally hitting your hand with a hammer. This experience would induce a spectrum of physical and psychological events — tissue damage, visceral discomfort, shifts in attention, arousal, negative affect, and a desire to avoid repeating the experience. These events contribute to the broad phenomenon of “pain,” and more specifically to nociceptive pain, which originates in peripheral nociceptive fibers (see Box 1 for detailed definitions). Though pain plays a crucial role in helping individuals to avoid future harm, it also impairs wellbeing and generates enormous public health burden [1].
Box 1. Definitions of pain.
The International Association for the Study of Pain (IASP) defines pain as “An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage” [90]. This includes effects of noxious physical stimulation, but also other experiences that “hurt”. After witnessing a friend hit herself with a hammer, for instance, you might feel a ‘crushing’ sensation in your own hand, or discomfort in your stomach. Such empathic pain includes bodily sensations described in terms of tissue damage, meeting the IASP criteria for pain.
This IASP’s definition of pain contrasts with its narrower definition for nociceptive pain: “pain that arises from actual or threatened damage to non-neural tissue and is due to the activation of nociceptors.” This definition privileges etiology and excludes empathic pain, which is not triggered by nociceptors in the person experiencing empathy.
Definitions by nature are operational – they serve the study of a phenomenon in a particular context. If scientists investigate a phenomenon like pain at multiple levels (e.g. nociceptors, cortical neurons, patterns of BOLD activity, psychological experience, behavior, and pathology) operational definitions useful at one level may lose their relevance at another, potentially impeding vital efforts to link these levels. At some levels, e.g. the response of certain neurons in the cingulate cortex, nociceptive and empathic pain might trigger identical responses [79]. At the psychological level, both might feel aversive, trigger strong motivations and be described in terms of tissue damage. Yet at the level of peripheral nociceptors, they will seem fundamentally different. Scientists specializing in each of these three levels may then disagree about whether empathic pain is a form of “true” pain. These scientists would disagree not about data, but rather about definitions.
Instead of a universal definition of pain to settle resulting arguments, here we argue for an agnostic approach: investigating particular pain-related dimensions at various levels of analysis, and mapping similarities and dissimilarities between empathic and nociceptive experiences with respect to each dimension. This could allow scientists to more precisely shed light on how nociceptive and empathic experiences relate, as well as how practitioners can effectively intervene to reduce the burden of pain. This approach further allows for a common ground from which each investigator can decide whether they believe empathic experiences constitute ‘pain,’ based on relevant data.
Now imagine observing a friend hit themselves with a hammer. This experience typically generates empathic pain—a pain-related phenomenon that, despite differences in origin, shares features with nociceptive pain. Here we explore the relationship between nociceptive and empathic pain. What does it mean to label both of these experience as “pain?” And how grounded are these labels in shared neurophysiological representation?
The Debate
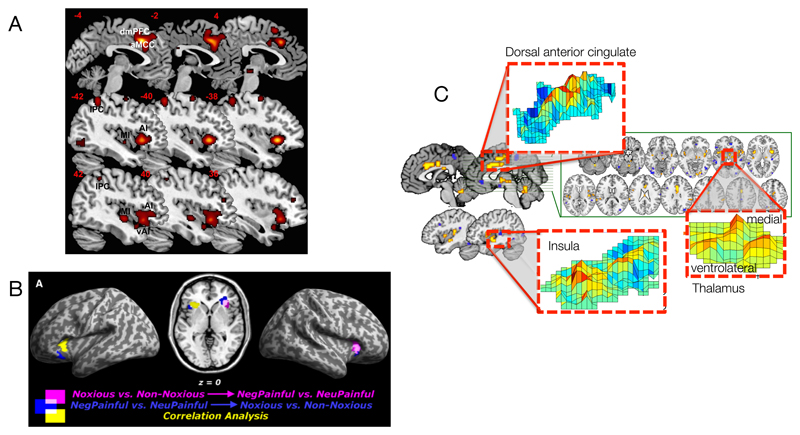
Decades of evidence in humans and animals suggest at least some overlap between nociceptive and empathic pain [2, 3]. Witnessing others in pain can create or intensify behavioral signs of nociceptive pain [4–6], and individuals with congenital insensitivity to nociceptive pain exhibit blunted responses to empathic pain [7]. Neuroscientists have further demonstrated that brain structures such as anterior insula (AI) and parts of the cingulate cortex (CC) commonly respond when people experience nociceptive and empathic pain ([8–15]; see Figure 1a). In some cases, empathic experiences also activate somatosensory cortex [9] and facilitate motor programs associated with nociceptive pain [16]. Brain responses to others’ pain in AI and CC correlate with subjective experiences of pain empathy [2, 17, 18] and willingness to shoulder costs in order to reduce others’ pain [3, 19]. Finally, brain responses to empathic pain diminish after placebo analgesia pain [20, 21].
Figure 1.
- Overlapping activations between nociceptive and empathic pain in a meta-analysis of 32 studies, from Ref [10].
- Overlap between multivariate patterns related to nociceptive pain (noxious versus non-noxious stimuli) and empathic pain (pictures of others in pain vs. neutral pictures), from Ref [8].
- The “Neurologic Pain Signature” (NPS), a multivariate pattern that is sensitive to nociceptive pain but not other aversive emotional experiences, from Ref [50].
These findings signal important relationships between nociceptive and empathic pain, but do not necessarily imply that they rely on the same psychological representations. For instance, AI and CC respond to non-pain states, including arousal and attention [22–28]. Manipulations that affect nociception, such as placebo analgesia, likewise influence not only pain, but also stress and anxiety [29]. Critics suggest that conclusions about the overlap between empathic and nociceptive pain rely heavily on spurious reverse inference (cf. [30]; see Box 2), and that social and nociceptive experiences might not in fact share pain-specific processes [31].
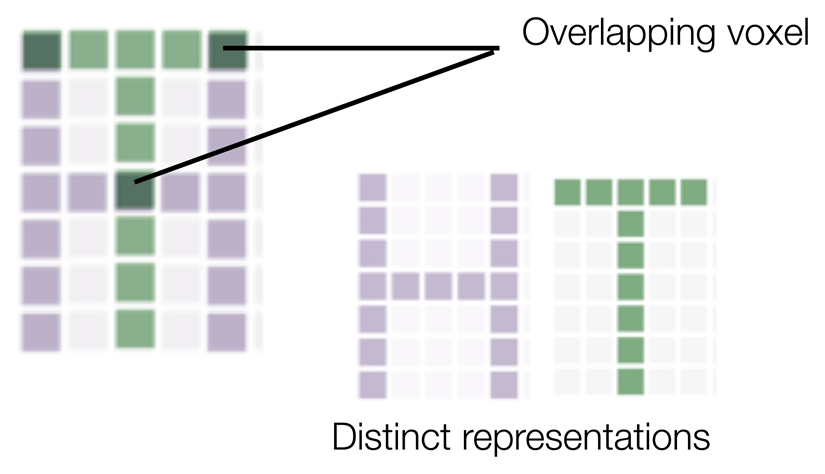
Box 2. Pattern classification and inference.
When scientists use neuroimaging to examining overlap and dissociations between nociceptive and empathic, they must draw inferences about the psychological meaning of brain activity. Such inference is often problematic, especially when brain regions of interest respond to many tasks [91, 92]. In such cases, two phenomena (e.g., empathic and nociceptive pain) could produce overlap in those regions, but nonetheless reflect fundamentally different psychological processes (Figure i).
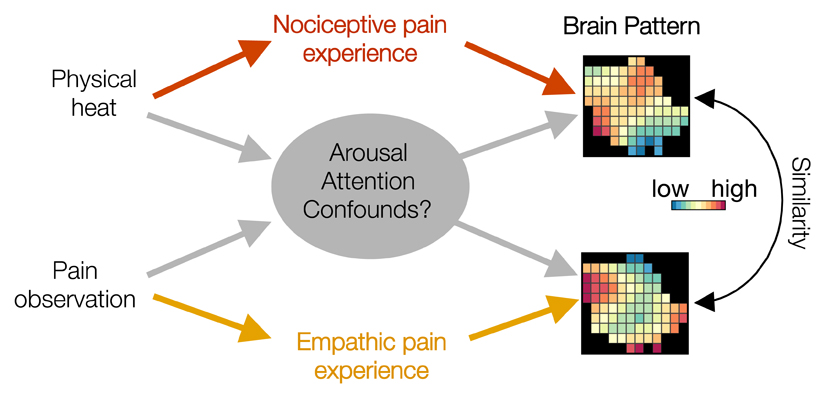
In an effort to overcome this limitation, scientists now commonly examine brain activity across multi-region patterns of voxels, and associate those patterns with variance in stimuli and reported experience [93]. This technique has helped to adjudicate differences and similarities between nociceptive and empathic pain. However, researchers should take two important considerations into account. First, activity patterns need not represent all aspects of a phenomenon in which researchers are interested. For instance, nociceptive and empathic pain might share a multivoxel activity pattern, but this pattern might reflect nonspecific features of arousal and attention, rather than the pain-specific experience (Figure ii). As such, the tuning curve approach we advocate here can help to assess the meaning of activity within a region and multivoxel patterns across regions.
Second, scientists should consider the way in which they extract multivoxel patterns. In assessing pain, one approach is to extract patterns that track the intensity of a localized nociceptive stimuli (e.g. heat to the hand), and test whether these patterns also track manipulations of empathic pain. This approach will identify combinations of voxels that most robustly differentiates levels of pain in the training set (here, nociceptive pain) and as such tests a strong assumption that empathic pain modulates brain activity associated with encoding the intensity of specific pain stimuli. Under our multidimensional framework, failure to document such overlap does not imply that these pain types share no crucial features, but rather that what they do share is not captured by that specific training set. One alternative would be for researchers to develop multivoxel methods that are trained on examples of both forms of pain, and test this pattern on new examples of both pain types. This would address the broader question of whether empathic and nociceptive pain share any key dimensions. Further research could test the responsivity of these shared patterns to pain-specific or non-specific manipulations, thus precisely characterizing the nature of psychological overlap across these states.
Figure i.
Visual representation of how overlapping activity in a cell or voxel across nociceptive and empathic pain could nonetheless reflect dissociable activity patterns.
Figure ii.
Visual representation of potential confounds when using multivoxel patterns to assess similarity across nociceptive and empathic pain.
Often, questions about pain states are posed as a binary: empathic pain either “counts” as pain or does not. We believe that understanding the nature of empathy and pain requires moving away from this simple distinction and instead (i) decomposing pain into its component “ingredients”, (ii) identifying brain markers of these ingredients (see Glossary), and (iii) using those markers to identify exactly which ingredients empathic and nociceptive pain share. This approach transforms the binary question of whether both empathic and nociceptive experiences constitute pain into a graded question: how far from one another these experiences fall in the multi-dimensional space of pain ingredients.
Multi-dimensional Pain
Pain includes a complex suite of processes. Consider the moment in which you hit yourself with a hammer. This event triggers a multi-dimensional experience, including, but not restricted to, processing (i) the location of pain (in your hand, not foot), (ii) its intensity (strong), (iii) qualities (crushing, aching), (iv) generalized discomfort, the negative (v) valence and (vi) high arousal characterizing your emotional response, (vii) redirection of attention to your hand, (viii) motivation to reduce pain, (ix) motor plans to do so (e.g., rubbing the affected area), and (x) learning to avoid future pain by wielding tools more carefully. Decades of research on nociceptive pain demonstrate that some of these dimensions covary more than others, and that it is useful to organize them into three broad groups: sensory-discriminative, affective, and cognitive dimensions [32, 33]. For instance, pharmacological and psychological manipulations—such as hypnosis [34], mood induction [35–37], or opioid drugs [38]— alter the affective qualities of pain more than its sensory qualities, or vice versa.
Most components of pain, when considered individually, are nonspecific, in that they occur both during pain and during non-pain experiences. For instance, arousal and attention play a role in pain, but also pleasure, anger, and stress [39]. Likewise, location coding occurs during processing of both painful and non-painful stimulation. Nociceptive pain, then, represents not a single psychological feature, but rather a set of features coming together in a particular configuration. This reflects a constructionist approach, which posits that phenomena such as emotion or value reflect combinations of more basic psychological ingredients [40–42].
This framework provides a powerful lens for using neuroscience to understand the overlap between pain-types. Consider our example of hitting yourself with a hammer. This experience would produce a complex pattern of activity across many brain areas, which can further be broken down into components, or pieces of this pattern. Does activity in each component constitute a marker of pain? Not necessarily. Crucial here is the pattern or component’s sensitivity and specificity to pain. If a pattern is sensitive and specific to a psychological state, then it qualifies as a marker of a psychological state, because its engagement warrants ‘reverse inference’ about the presence of that state: presence of that marker strongly implies presence of that psychological experience.
Most patterns and components that accompany pain do not meet this criterion. Voxels in sensory cortex, for instance, might respond to hitting your finger, thus exhibiting sensitivity to pain, but might also respond to non-painful tactile experiences, thus not exhibiting specificity. Likewise, activation in the frontal eye fields might respond to hitting yourself, but also to any attentional shift towards unexpected events [43]. On our model, pain constitutes the unique combination of these ingredients, and markers of pain should respond only when those ingredients come together.
Psychological ‘tuning curves’ for pain-related experience
One way of assessing the psychological meaning of brain activity is through examination of tuning curves: plots characterizing the psychological ‘landscape’ characterizing the responsivity of a brain pattern. This approach originates in measures of single neurons [44], but can easily be broadened to assess fMRI activity within and across brain regions [45, 46].
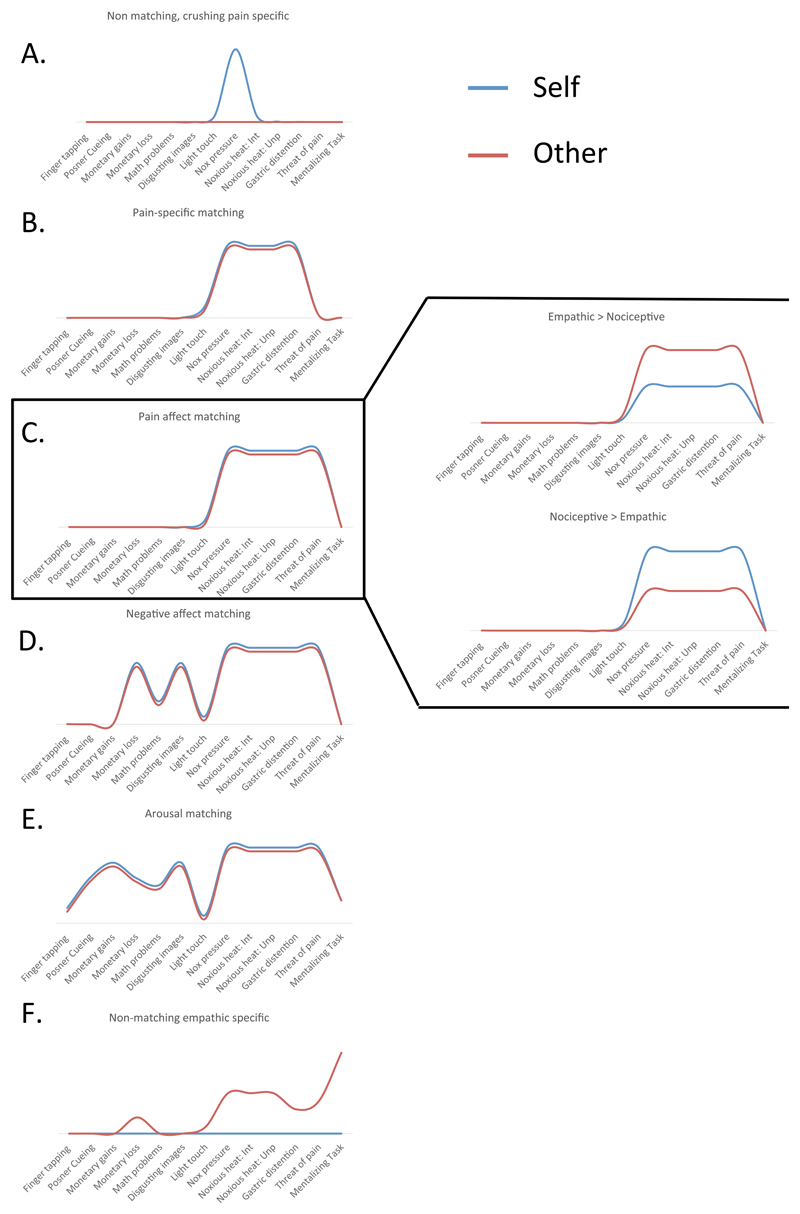
To assess the tuning curves in the domain of pain, scientists should isolate brain patterns that respond to pain, and test the response of these patterns to a systematic set of control conditions that share specific ingredients with pain. Figure 2 suggests some such control tasks, and illustrates tuning curves describing how a hypothetical brain pattern might respond to these tasks.
Figure 2. Inference based on tuning curves.
- This marker responds with high specificity to nociceptive pressure, but not to any empathic experiences.
- This marker responds narrowly to painful pressure and other forms of nociceptive pain (such as heat or gastric distension) in both self and other, suggesting overlap between these states in coding pain intensity and modality.
- This marker responds to multiple types of nociceptive and empathic pain, and also to other unpleasant experiences such as threat of pain in the absence of direct nociception. The amplitude of responses to self and other can vary independently of the markers’ response “landscape,” with some markers showing stronger responses when stimuli are applied to the self (right column, top) or to others (right column, bottom).
- This marker exhibits sensitivity to negative affective experiences, including pain and threat of pain, but also disgusting images and monetary loss.
- This marker exhibits sensitivity to both personal and vicarious experience of many arousing states, and thus captures overlap between these phenomena that is not pain specific.
- This marker responds uniquely to empathic pain states, but not nociceptive pain.
Some tuning curves are pain-specific, in that they only respond when pain ingredients co-occur. For instance, the tuning curve in Figure 2a is narrowly focused on painful pressure, and the curve in Figure 2b additionally responds to other nociceptive pain types, but not to non-pain events. Patterns that exhibit such tuning curves are both sensitive and specific to nociceptive pain, and can thus be considered markers of this state. Of course, any form of reverse inference—including one based on a tuning curve approach—is necessarily probabilistic. However, identifying markers based on their sensitivity and specificity to many tasks and stimuli allows for inferences that are more precise than examining brain responses to only pain, or pain and only one control condition.
Other markers might exhibit broader, less specific tuning curves. Consider a marker of pain affect, or the visceral discomfort brought on by painful stimuli. This marker might respond to noxious pressure and heat, and also exhibit some responsiveness to other “painful” stimuli, such as monetary loss (Figure 2c), but not to other negative affective stimuli such as disgusting images. A broader marker might respond to negatively valenced affective stimuli, including disgusting images and monetary loss (Figure 2d). Finally, an even broader marker for arousal might respond to these states, and also to math problems, winning money, and other states that engage the sympathetic nervous system (Figure 2e), as well as tasks associated with reallocation of attention and motor planning.
This framework provides a substrate for precisely assessing what nociceptive pain shares with empathic pain (e.g., seeing your friend hit with a hammer, marked “other” in Figure 2). If markers specific to nociceptive pain in first person experiences are also engaged by third person pain (Figure 2b), these states likely share a pain-specific configuration of ingredients. If viewing others in pain engages patterns that are activated during first hand experience of other ‘painful’ physical and emotional events (e.g., monetary loss) but not non-painful experiences such as disgust [47], that would support the inference that nociceptive and empathic states share ingredients that characterize “pain affect” (Figure 2c). Alternatively, if markers shared by empathic and nociceptive pain also respond to non-painful stimuli like aversive images or math problems [48], that all have high level or arousal in common, we might conclude that empathic and nociceptive pain share only less-specific representations of negative emotion or arousal (Figure 2d).
Only a few existing studies have examined ‘tuning curves’ associated with empathic pain, and these have produced somewhat discrepant findings, using analysis of multivoxel patterns (see Box 2). For example, AI and CC patterns identified in Ref [8] respond to nociceptive and empathic pain, but also to negative emotional pictures, consistent with a broad response to negative affect (Figure 1b). Newer work focusing on regions of interest in AI and CC has painted a more complex picture, identifying patterns with several types of response profiles [49]. In some regions, such as the right AI, response patterns are specific to both the type of affect (domain) and the target (whether experience is direct or empathetic). This area showed separate patterns for nociceptive pain, empathic pain, disgust, empathic disgust, fairness, and empathic fairness, consistent with cases illustrated in Figures 2a and 2f. Other regions, such as the left AI and CC, appear to respond to all of the types of events listed above roughly equally, consistent with a general representation of negative affect, as illustrated in Figure 2d.
By contrast, other recent work identifies a multivariate “neurologic signature” that tracks the intensity of heat, pressure, and shock pain applied to the arm [50], but does not respond to negative images [51] or social rejection cues [52] (Figure 1c). These findings demonstrate separate modifiability [53] and imply that brain patterns responsive to nociceptive and empathic pain reflect distinct psychological representations. This work also provides a method for assessing to what proportion of representations across these states are shared or unshared.
The discrepancies between across these approaches could reflect several differences in design and analysis. First, markers that show overlap between pain types are drawn from studies that compare high versus low pain in both empathic and nociceptive conditions, whereas patterns found to be specific to nociceptive pain and not social experiences (and vice versa) emerge using markers trained to predict graded ratings of participants’ experience. More importantly, these studies also varied with respect to how they directed participants’ attention. Studies that point towards nociceptive-specific representations typically direct participants’ to make ratings of pain intensity, possibly driving attention towards sensory component of the pain experience. Activity in the neurologic pain signature thus correlates with increases in the intensity of nociceptive pain, but not other aversive experiences [50–52]. By contrast, studies identifying overlap between pain types typically ask participants to report on the affective unpleasantness of pain [8, 49], likely driving their attention towards pain affect.
These data highlight the complexity of drawing inferences about markers of pain based on any one study. Demonstrations of separate modifiability suggest independence between nociceptive and empathic pain, but do not imply that these pain types are entirely or always independent. Instead, separate modifiability here might reflect only some dimensions of pain experience, such as coding of intensity ratings of pain delivered to one’s extremities [50]. Although this intensity-focused marker uniquely tracks nociceptive pain, markers of other pain dimensions such as pain affect might reveal responses shared with empathic pain (though in some cases these rating types tightly correlate with each other; cf. Ref [54]). Future work should directly examine the effects of attentional set on markers for empathic and nociceptive pain, as well as their overlap.
In general, scientists should leverage the ‘tuning curve’ concept to examine responses of nociceptive and empathic neural markers to a broader set of phenomena. Doing so will be a long-term endeavor, requiring many studies across multiple labs to compare neural markers that respond to numerous pain-related states delivered to the self and to others. This dovetails with the increasingly cumulative nature of cognitive neuroscience, under which reverse inference about psychological processes based on brain activity requires synthesis of many studies to estimate neural markers’ specificity and sensitivity [55]. In an affective analogue to the “cognitive ontologies” [56], such a cumulative approach will allow scientists to better decompose pain and understand the relations between self- and other-oriented pain states.
Methodological Suggestions
Thus far, we have suggested a conceptual approach for charting the overlap between empathic and nociceptive pain. We now turn to methodological suggestions through which to apply this approach.
Train classifiers on multidimensional pain experiences
Existing paradigms typically compare empathic pain to nociceptive stimuli delivered to participants’ extremities (such as heat pain to the arm). Patterns trained on intensity judgments for such stimuli likely home in on brain patterns that encode pain in a modality- and location-specific way, with a narrow tuning curve like the one visualized in Figure 2a. If empathic and nociceptive pain instead overlap at an intermediate level, for instance in representations of pain affect (Figure 2c), neural patterns trained on intensity judgments might miss this shared pain experience. Comparing empathic pain to other forms of nociceptive pain that produce more diffuse intensity and location coding—such as gut or rectal distention [57, 58]—or comparing patterns tracking people’s affective discomfort in response to pain, might show more overlap with empathetic pain [59, 60].
Explore factors that modulate overlap
Several factors, including attention, motivation, context and individual differences, powerfully shape the experience of both nociceptive [61–64] and empathic pain [2, 19, 65–71]. For instance, observers who pay close attention to or share group membership with social targets exhibit intensified brain activity in response to empathic pain [19, 72], whereas situational factors such as intergroup barriers [12, 69, 73, 74] and individual factors such as psychopathy [14] and alexithymia [75] diminish or even eliminate these responses. Likewise, empathy training induces functional changes in AI and CC activity during empathic pain, which track increases in self reported empathy [76]. Contextual and individual differences also interact: for instance, individuals with psychopathy exhibit blunted neural responses to empathic pain, but not when explicitly instructed to empathize [68].
Modulatory factors likely alter not only the intensity of empathic pain, but also the dimensions over which it operates. For instance, observers who are highly motivated to process specific characteristics of another person’s pain—such as a parent whose child is injured or, by contrast, an emergency room doctor attempting to objectively treat that injury—could exhibit differential overlap between empathic and nociceptive pain [9, 77]. Future work should merge a tuning curve approach with manipulations of context or individual differences to examine whether these factors indeed modulate the overlap between pain types.
The need for neuronal resolution
The question of whether empathic and nociceptive pain share neural substrates ultimately rests on whether individual neurons co-represent aspects of each pain type. Noninvasive neuroimaging suggests but cannot demonstrate such overlap. For instance, an fMRI voxel or pattern might contain separate neuronal populations that respond to each pain-type, generating false overlap when activity is averaged across those populations. Further, although AI and CC respond to many stimuli and contexts, these regions nonetheless contain pain-specific, as well as non-specific neurons [78, 79], such that averaging across these cell population yields pain non-specific signals. Averaging can also falsely imply independence. For instance, an fMRI pattern sensitive to nociceptive, but not empathic pain, could mean that no nociceptive neurons respond to empathic pain, but could also occur if a minority of neurons in fact do respond to both pain types. Such overlap could be obscured by averaging activity of such shared neurons with others that respond specifically to nociceptive pain.
Single cell recordings can provide crucial converging information in cases like these. Consider the case of “mirror neurons.” Single cell recordings in monkeys and humans showed that many of the voxels that respond to both self and other action indeed contain neurons that co-represent these states [80, 81]. These mirror neurons exhibit specificity and sensitivity for particular actions [80, 81]. However, this is true for only ~10% of neurons, whilst ~90% respond exclusively during self actions [80, 81]. Averaging the 10% of “true” mirror neurons with 90% that respond exclusively to self actions produces activation patterns that translate poorly (although sometimes significantly) from actions of the self to the actions of others [82–84].
Almost no single cell studies have examined empathic pain (but see [79]), but emerging rodent models of empathic pain pave the way for measuring and manipulating cellular activity to shed light on the nature of empathic pain. For instance, deactivating regions involved in pain (e.g., CC) reduces behavioral signs of empathic pain [85]. One key question these techniques will help answer is whether neurons that respond to both empathic and nociceptive pain—even if they comprise a minority of nociceptive neurons overall—suffice to generate pain-relevant experience. Techniques such as optogenetics will allow scientists to address this question by directly triggering or suppress activity in these shared neurons [86].
Concluding Remarks
The relationship between empathic and nociceptive pain has generated great interest and controversy in recent years. Debates about these states’ status as pain or not connect with thorny issues concerning the psychological and biological nature of pain. Here we propose replacing binary questions about whether empathic pain “counts” as pain or not with a multidimensional approach that focuses scientists on finer-grained questions about the particular psychological dimensions empathic and nociceptive pain share. We hope that this nuanced approach, in combination with a growing set of tools and techniques, will deliver increasingly mechanistic accounts of how personal and vicarious pain relate and interact. The coming years will offer new and exciting insight surrounding the connection between pain types, which can inform our basic understanding of what it constitutes (see Outstanding Questions). In the long term, this approach might help assess the nature of pain-related symptoms associated with varying states of illness and dysfunction, and determine the best targets for intervention. Finally, this approach lends itself to many other domains. How for instance does the memory of nociception, rejection [52, 87], shame, embarrassment [88, 89], or guilt relate to nociception? A fine-grained understanding of the many dimensions of pain will allow us to situate these and other experiences as they relate to the broader experience of suffering.
Outstanding Questions.
Given a set of brain markers representing different dimensions of pain—such as location, intensity, discomfort, and arousal—which of these markers (and which levels of pain specificity) generate overlap between nociceptive and empathic pain?
How might contextual factors (e.g., group membership) and individual differences (e.g., in trait empathy or psychopathy) alter the representational dimensions that nociceptive and empathic pain share?
How will patterns of overlap between pain types revealed by neuroimaging map onto similar evidence gleaned from neurophysiological recordings and manipulations in nonhuman animals?
How do the markers associated with different tuning curves map onto clinical disorders, and how does that structure our understanding of the associated experiential symptomatology?
Glossary
- Nociceptive pain
Pain that arises from actual or threatened damage to non-neural tissue and is due to the activation of nociceptors
- Empathic pain
Pain that arises from observing actual or threatened tissue damage in another person.
- Constructionism
An approach to psychology and neuroscience positing that complex states (e.g., emotions) can be best understood not as irreducible entities, but rather as combinations of psychological “ingredients.”
- Sensitivity
The probability of engaging a neural marker given that a particular mental state is present.
- Specificity
The probability of not engaging a neural marker when a particular mental state is not present.
- Pattern
The set of voxels activated (and their accompanying intensity) by a stimulus or task.
- Component
A subset of a brain pattern inferred to track a specific dimension of psychological experience (e.g., attention shifts or location coding).
- Marker
A pattern or component that displays sensitivity and specificity to one psychological state, allowing for reverse inference about that state based on the activation of that pattern.
- Separate modifiability
A state under which activity in two patterns or components are modulated by differing tasks. E.g., activity in pattern A tracks psychological variable X but not variable Y, and activity in pattern B tracks psychological variable Y but not variable X.
References
- 1.Simon LS. Relieving pain in America: A blueprint for transforming prevention, care, education, and research. Journal of Pain and Palliative Care Pharmacotherapy. 2012;26(2):197–198. [Google Scholar]
- 2.Bernhardt BC, Singer T. The neural basis of empathy. Annual Review of Neuroscience. 2012;35:1–23. doi: 10.1146/annurev-neuro-062111-150536. [DOI] [PubMed] [Google Scholar]
- 3.Zaki J, Ochsner K. The neuroscience of empathy: Progress, pitfalls, and promise. Nat Neurosci. 2012;15(5):675–680. doi: 10.1038/nn.3085. [DOI] [PubMed] [Google Scholar]
- 4.Loggia ML, Mogil JS, Bushnell M Catherine. Empathy hurts: compassion for another increases both sensory and affective components of pain perception. Pain. 2008;136(1):168–176. doi: 10.1016/j.pain.2007.07.017. [DOI] [PubMed] [Google Scholar]
- 5.Langford DJ, et al. Social modulation of pain as evidence for empathy in mice. Science. 2006;312(5782):1967–1970. doi: 10.1126/science.1128322. [DOI] [PubMed] [Google Scholar]
- 6.Atsak P, et al. Experience modulates vicarious freezing in rats: a model for empathy. PloS one. 2011;6(7):e21855. doi: 10.1371/journal.pone.0021855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Danziger N, Prkachin KM, Willer J-C. Is pain the price of empathy? The perception of others' pain in patients with congenital insensitivity to pain. Brain. 2006;129(9):2494–2507. doi: 10.1093/brain/awl155. [DOI] [PubMed] [Google Scholar]
- 8.Corradi-Dell'acqua C, Hofstetter C, Vuilleumier P. Felt and seen pain evoke the same local patterns of cortical activity in insular and cingulate cortex. J Neurosci. 2011;31(49):17996–8006. doi: 10.1523/JNEUROSCI.2686-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Keysers C, Kaas JH, Gazzola V. Somatosensation in social perception. Nat Rev Neurosci. 2010;11(6):417–28. doi: 10.1038/nrn2833. [DOI] [PubMed] [Google Scholar]
- 10.Lamm C, Decety J, Singer T. Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. Neuroimage. 2011;54(3):2492–502. doi: 10.1016/j.neuroimage.2010.10.014. [DOI] [PubMed] [Google Scholar]
- 11.Singer T, et al. Empathy for pain involves the affective but not sensory components of pain. Science. 2004;303(5661):1157–62. doi: 10.1126/science.1093535. [DOI] [PubMed] [Google Scholar]
- 12.Singer T, et al. Empathic neural responses are modulated by the perceived fairness of others. Nature. 2006;439(7075):466–9. doi: 10.1038/nature04271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cui F, et al. Responsibility modulates pain-matrix activation elicited by the expressions of others in pain. Neuroimage. 2015 doi: 10.1016/j.neuroimage.2015.03.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Meffert H, et al. Reduced spontaneous but relatively normal deliberate vicarious representations in psychopathy. Brain. 2013;136(Pt 8):2550–62. doi: 10.1093/brain/awt190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zaki J, Ochsner K. You, me, and my brain: Self and other representations in social cognitive neuroscience. In: Todorov A, Fiske ST, Prentice D, editors. Social neuroscience: Toward understanding the underpinnings of the social mind. Oxford University Press; New York: 2011. [Google Scholar]
- 16.Avenanti A, et al. Transcranial magnetic stimulation highlights the sensorimotor side of empathy for pain. Nat Neurosci. 2005;8(7):955–60. doi: 10.1038/nn1481. [DOI] [PubMed] [Google Scholar]
- 17.Saarela MV, et al. The Compassionate Brain: Humans Detect Intensity of Pain from Another's Face. Cereb Cortex. 2007;17:230–237. doi: 10.1093/cercor/bhj141. [DOI] [PubMed] [Google Scholar]
- 18.Kanske P, et al. Dissecting the social brain: Introducing the EmpaToM to reveal distinct neural networks and brain-behavior relations for empathy and Theory of Mind. NeuroImage. 2015 doi: 10.1016/j.neuroimage.2015.07.082. [DOI] [PubMed] [Google Scholar]
- 19.Hein G, et al. Neural responses to ingroup and outgroup members' suffering predict individual differences in costly helping. Neuron. 2010;68(1):149–60. doi: 10.1016/j.neuron.2010.09.003. [DOI] [PubMed] [Google Scholar]
- 20.Rütgen M, et al. Reduction of Empathy for Pain by Placebo Analgesia Suggests Functional Equivalence of Empathy and First-Hand Emotion Experience. The Journal of Neuroscience. 2015;35(23):8938–8947. doi: 10.1523/JNEUROSCI.3936-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rütgen M, et al. Placebo analgesia and its opioidergic regulation suggest that empathy for pain is grounded in self pain. Proceedings of the National Academy of Sciences. 2015;112(41):E5638–E5646. doi: 10.1073/pnas.1511269112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Craig AD. How do you feel--now? The anterior insula and human awareness. Nat Rev Neurosci. 2009;10(1):59–70. doi: 10.1038/nrn2555. [DOI] [PubMed] [Google Scholar]
- 23.Shackman AJ, et al. The integration of negative affect, pain and cognitive control in the cingulate cortex. Nature Reviews Neuroscience. 2011;12(3):154–167. doi: 10.1038/nrn2994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shenhav A, Botvinick MM, Cohen JD. The expected value of control: an integrative theory of anterior cingulate cortex function. Neuron. 2013;79(2):217–240. doi: 10.1016/j.neuron.2013.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Singer T, Critchley HD, Preuschoff K. A common role of insula in feelings, empathy and uncertainty. Trends Cogn Sci. 2009;13(8):334–40. doi: 10.1016/j.tics.2009.05.001. [DOI] [PubMed] [Google Scholar]
- 26.Zaki J, Davis JI, Ochsner KN. Overlapping activity in anterior insula during interoception and emotional experience. Neuroimage. 2012 doi: 10.1016/j.neuroimage.2012.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lyons IM, Beilock SL. When math hurts: math anxiety predicts pain network activation in anticipation of doing math. 2012 doi: 10.1371/journal.pone.0048076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Grinband J, Hirsch J, Ferrera VP. A neural representation of categorization uncertainty in the human brain. Neuron. 2006;49(5):757–763. doi: 10.1016/j.neuron.2006.01.032. [DOI] [PubMed] [Google Scholar]
- 29.Flaten MA, et al. The relation of emotions to placebo responses. Philosophical Transactions of the Royal Society B: Biological Sciences. 2011;366(1572):1818–1827. doi: 10.1098/rstb.2010.0407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Poldrack RA. Can cognitive processes be inferred from neuroimaging data? Trends in cognitive sciences. 2006;10(2):59–63. doi: 10.1016/j.tics.2005.12.004. [DOI] [PubMed] [Google Scholar]
- 31.Iannetti GD, et al. Beyond metaphor: contrasting mechanisms of social and physical pain. Trends in cognitive sciences. 2013;17(8):371–378. doi: 10.1016/j.tics.2013.06.002. [DOI] [PubMed] [Google Scholar]
- 32.Peyron R, Laurent B, Garcia-Larrea L. Functional imaging of brain responses to pain. A review and meta-analysis (2000) Neurophysiol Clin. 2000;30(5):263–88. doi: 10.1016/s0987-7053(00)00227-6. [DOI] [PubMed] [Google Scholar]
- 33.Bushnell MC, Čeko M, Low LA. Cognitive and emotional control of pain and its disruption in chronic pain. Nature Reviews Neuroscience. 2013;14(7):502–511. doi: 10.1038/nrn3516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rainville P, et al. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science. 1997;277(5328):968–71. doi: 10.1126/science.277.5328.968. [DOI] [PubMed] [Google Scholar]
- 35.Wiech K, Tracey I. The influence of negative emotions on pain: behavioral effects and neural mechanisms. Neuroimage. 2009;47(3):987–994. doi: 10.1016/j.neuroimage.2009.05.059. [DOI] [PubMed] [Google Scholar]
- 36.Loggia ML, Mogil JS, Bushnell MC. Experimentally induced mood changes preferentially affect pain unpleasantness. The Journal of Pain. 2008;9(9):784–791. doi: 10.1016/j.jpain.2008.03.014. [DOI] [PubMed] [Google Scholar]
- 37.Villemure C, Bushnell MC. Mood influences supraspinal pain processing separately from attention. The Journal of Neuroscience. 2009;29(3):705–715. doi: 10.1523/JNEUROSCI.3822-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Atlas LY, et al. Dissociable influences of opiates and expectations on pain. The Journal of neuroscience. 2012;32(23):8053–8064. doi: 10.1523/JNEUROSCI.0383-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Beilock SL, et al. More on the fragility of performance: choking under pressure in mathematical problem solving. Journal of Experimental Psychology: General. 2004;133(4):584. doi: 10.1037/0096-3445.133.4.584. [DOI] [PubMed] [Google Scholar]
- 40.Barrett LF. Psychological construction: The Darwinian approach to the science of emotion. Emotion Review. 2013;5(4):379–389. [Google Scholar]
- 41.Roy M, Shohamy D, Wager TD. Ventromedial prefrontal-subcortical systems and the generation of affective meaning. Trends in Cognitive Sciences. 2012 doi: 10.1016/j.tics.2012.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lindquist KA, Barrett LF. Constructing Emotion The Experience of Fear as a Conceptual Act. Psychological science. 2008;19(9):898–903. doi: 10.1111/j.1467-9280.2008.02174.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Corbetta M, Shulman GL. Control of goal-directed and stimulus-driven attention in the brain. Nature reviews neuroscience. 2002;3(3):201–215. doi: 10.1038/nrn755. [DOI] [PubMed] [Google Scholar]
- 44.Britten KH, et al. The analysis of visual motion: a comparison of neuronal and psychophysical performance. The Journal of Neuroscience. 1992;12(12):4745–4765. doi: 10.1523/JNEUROSCI.12-12-04745.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Norman KA, et al. Beyond mind-reading: multi-voxel pattern analysis of fMRI data. Trends in cognitive sciences. 2006;10(9):424–430. doi: 10.1016/j.tics.2006.07.005. [DOI] [PubMed] [Google Scholar]
- 46.Piazza M, et al. Tuning curves for approximate numerosity in the human intraparietal sulcus. Neuron. 2004;44(3):547–555. doi: 10.1016/j.neuron.2004.10.014. [DOI] [PubMed] [Google Scholar]
- 47.Lindquist KA, et al. The brain basis of emotion: a meta-analytic review. Behavioral and Brain Sciences. 2012;35(03):121–143. doi: 10.1017/S0140525X11000446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yeung N, Botvinick MM, Cohen JD. The neural basis of error detection: conflict monitoring and the error-related negativity. Psychological review. 2004;111(4):931. doi: 10.1037/0033-295x.111.4.939. [DOI] [PubMed] [Google Scholar]
- 49.Corradi-Dell'acqua C, et al. Cross-modal representations of first-hand and vicarious pain, disgust and fairness in insular and cingulate cortex. Nat Comm. doi: 10.1038/ncomms10904. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wager TD, et al. An fMRI-based neurologic signature of physical pain. New England Journal of Medicine. 2013;368(15):1388–1397. doi: 10.1056/NEJMoa1204471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chang LJ, et al. A Sensitive and Specific Neural Signature for Picture-Induced Negative Affect. PLoS Biol. 2015;13(6):e1002180. doi: 10.1371/journal.pbio.1002180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Woo C-W, et al. Separate neural representations for physical pain and social rejection. Nature communications. 2014;5 doi: 10.1038/ncomms6380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sternberg S. Separate modifiability, mental modules, and the use of pure and composite measures to reveal them. Acta psychologica. 2001;106(1):147–246. doi: 10.1016/s0001-6918(00)00045-7. [DOI] [PubMed] [Google Scholar]
- 54.Coghill RC, et al. Pain intensity processing within the human brain: a bilateral, distributed mechanism. Journal of neurophysiology. 1999;82(4):1934–1943. doi: 10.1152/jn.1999.82.4.1934. [DOI] [PubMed] [Google Scholar]
- 55.Yarkoni T, et al. Cognitive neuroscience 2.0: building a cumulative science of human brain function. Trends in cognitive sciences. 2010;14(11):489–496. doi: 10.1016/j.tics.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Poldrack RA, Yarkoni T. From Brain Maps to Cognitive Ontologies: Informatics and the Search for Mental Structure. Annual review of psychology. 2015;67(1) doi: 10.1146/annurev-psych-122414-033729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Baciu MV, et al. Central processing of rectal pain: a functional MR imaging study. American Journal of Neuroradiology. 1999;20(10):1920–1924. [PMC free article] [PubMed] [Google Scholar]
- 58.Tillisch K, Mayer EA, Labus JS. Quantitative meta-analysis identifies brain regions activated during rectal distension in irritable bowel syndrome. Gastroenterology. 2011;140(1):91–100. doi: 10.1053/j.gastro.2010.07.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Roy M, et al. Representation of aversive prediction errors in the human periaqueductal gray. Nature neuroscience. 2014;17(11):1607–1612. doi: 10.1038/nn.3832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kucyi A, Salomons TV, Davis KD. Mind wandering away from pain dynamically engages antinociceptive and default mode brain networks. Proceedings of the National Academy of Sciences. 2013;110(46):18692–18697. doi: 10.1073/pnas.1312902110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bantick SJ, et al. Imaging how attention modulates pain in humans using functional MRI. Brain. 2002;125(2):310–319. doi: 10.1093/brain/awf022. [DOI] [PubMed] [Google Scholar]
- 62.Lobanov OV, et al. Frontoparietal mechanisms supporting attention to location and intensity of painful stimuli. Pain. 2013;154(9):1758–1768. doi: 10.1016/j.pain.2013.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Villemure C, Bushnell MC. Cognitive modulation of pain: how do attention and emotion influence pain processing? Pain. 2002;95(3):195–9. doi: 10.1016/S0304-3959(02)00007-6. [DOI] [PubMed] [Google Scholar]
- 64.Atlas LY, et al. Brain mediators of predictive cue effects on perceived pain. The Journal of Neuroscience. 2010;30(39):12964–12977. doi: 10.1523/JNEUROSCI.0057-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hein G, Singer T. I feel how you feel but not always: the empathic brain and its modulation. Curr Opin Neurobiol. 2008;18(2):153–8. doi: 10.1016/j.conb.2008.07.012. [DOI] [PubMed] [Google Scholar]
- 66.Zaki J. Empathy: A motivated account. Psychological Bulletin. 2014;140(6):1608–1647. doi: 10.1037/a0037679. [DOI] [PubMed] [Google Scholar]
- 67.Keysers C, Gazzola V. Dissociating the ability and propensity for empathy. Trends in cognitive sciences. 2014;18(4):163–166. doi: 10.1016/j.tics.2013.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Meffert H, et al. Reduced spontaneous but relatively normal deliberate vicarious representations in psychopathy. Brain. 2013;136(8):2550–2562. doi: 10.1093/brain/awt190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Xu X, et al. Do you feel my pain? Racial group membership modulates empathic neural responses. J Neurosci. 2009;29(26):8525–9. doi: 10.1523/JNEUROSCI.2418-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Decety J, et al. Atypical empathic responses in adolescents with aggressive conduct disorder: a functional MRI investigation. Biological Psychology. 2009;80(2):203. doi: 10.1016/j.biopsycho.2008.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Decety J, Yang C-Y, Cheng Y. Physicians down-regulate their pain empathy response: an event-related brain potential study. Neuroimage. 2010;50(4):1676–1682. doi: 10.1016/j.neuroimage.2010.01.025. [DOI] [PubMed] [Google Scholar]
- 72.Gu X, Han S. Attention and reality constraints on the neural processes of empathy for pain. Neuroimage. 2007;36(1):256–67. doi: 10.1016/j.neuroimage.2007.02.025. [DOI] [PubMed] [Google Scholar]
- 73.Cikara M, Bruneau E, Saxe R. Us and them: Intergroup failures in empathy. Curr Dir Psychol Sci. 2011;20:149–153. [Google Scholar]
- 74.Kaseweter KA, Drwecki BB, Prkachin KM. Racial differences in pain treatment and empathy in a Canadian sample. Pain Research & Management: The Journal of the Canadian Pain Society. 2012;17(6):381. doi: 10.1155/2012/803474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bird G, et al. Empathic brain responses in insula are modulated by levels of alexithymia but not autism. Brain. 2010;133(Pt 5):1515–25. doi: 10.1093/brain/awq060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Klimecki OM, et al. Differential pattern of functional brain plasticity after compassion and empathy training. Social cognitive and affective neuroscience. 2013 doi: 10.1093/scan/nst060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Voisin JI, et al. I am touched by your pain: limb-specific modulation of the cortical response to a tactile stimulation during pain observation. The Journal of Pain. 2011;12(11):1182–1189. doi: 10.1016/j.jpain.2011.06.005. [DOI] [PubMed] [Google Scholar]
- 78.Davis KD, et al. Human anterior cingulate cortex neurons modulated by attention-demanding tasks. Journal of neurophysiology. 2000;83(6):3575–3577. doi: 10.1152/jn.2000.83.6.3575. [DOI] [PubMed] [Google Scholar]
- 79.Hutchison WD, et al. Pain-related neurons in the human cingulate cortex. Nat Neurosci. 1999;2(5):403–5. doi: 10.1038/8065. [DOI] [PubMed] [Google Scholar]
- 80.di Pellegrino G, et al. Understanding motor events: a neurophysiological study. Exp Brain Res. 1992;91(1):176–80. doi: 10.1007/BF00230027. [DOI] [PubMed] [Google Scholar]
- 81.Rizzolatti G, et al. Premotor cortex and the recognition of motor actions. Brain Res Cogn Brain Res. 1996;3(2):131–41. doi: 10.1016/0926-6410(95)00038-0. [DOI] [PubMed] [Google Scholar]
- 82.Etzel JA, Gazzola V, Keysers C. Testing simulation theory with cross-modal multivariate classification of fMRI data. PLoS One. 2008;3(11):e3690. doi: 10.1371/journal.pone.0003690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Oosterhof NN, et al. Surface-based information mapping reveals crossmodal vision-action representations in human parietal and occipitotemporal cortex. J Neurophysiol. 2010;104(2):1077–89. doi: 10.1152/jn.00326.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Dinstein I, et al. Executed and observed movements have different distributed representations in human aIPS. J Neurosci. 2008;28(44):11231–9. doi: 10.1523/JNEUROSCI.3585-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Jeon D, et al. Observational fear learning involves affective pain system and Cav1. 2 Ca2+ channels in ACC. Nature neuroscience. 2010;13(4):482–488. doi: 10.1038/nn.2504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rickgauer JP, Deisseroth K, Tank DW. Simultaneous cellular-resolution optical perturbation and imaging of place cell firing fields. Nature neuroscience. 2014;17(12):1816–1824. doi: 10.1038/nn.3866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Eisenberger NI. The pain of social disconnection: examining the shared neural underpinnings of physical and social pain. Nature Reviews Neuroscience. 2012;13(6):421–434. doi: 10.1038/nrn3231. [DOI] [PubMed] [Google Scholar]
- 88.Paulus FM, et al. Mentalizing and the role of the posterior superior temporal sulcus in sharing others' embarrassment. Cerebral cortex. 2014:bhu011. doi: 10.1093/cercor/bhu011. [DOI] [PubMed] [Google Scholar]
- 89.Müller-Pinzler L, et al. Neural pathways of embarrassment and their modulation by social anxiety. NeuroImage. 2015 doi: 10.1016/j.neuroimage.2015.06.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Loeser JD, Treede R-D. The Kyoto protocol of IASP basic pain terminology. Pain. 2008;137(3):473–477. doi: 10.1016/j.pain.2008.04.025. [DOI] [PubMed] [Google Scholar]
- 91.Yarkoni T, et al. Large-scale automated synthesis of human functional neuroimaging data. Nature methods. 2011;8(8):665–70. doi: 10.1038/nmeth.1635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Poldrack RA. Inferring mental states from neuroimaging data: from reverse inference to large-scale decoding. Neuron. 2011;72(5):692–697. doi: 10.1016/j.neuron.2011.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kriegeskorte N, Mur M, Bandettini P. Representational similarity analysis–connecting the branches of systems neuroscience. Frontiers in Systems Neuroscience. 2008;2(4):1–28. doi: 10.3389/neuro.06.004.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]