Abstract
We investigated the role played by β2-containing neuronal nicotinic receptors [nicotinic acetylcholine receptors (nAChRs)] in mediating nicotine's side effects in the fetus and newborn. Pregnant WT and mutant mice lacking the β2 nAChR subunit were implanted with osmotic minipumps that delivered either water or a controlled dose of nicotine. Subsequently, we compared the development of the sympathoadrenal system and breathing and arousal reflexes of offspring shortly after birth, a period of increased vulnerability to nicotine exposure. Newborn WT pups exposed to nicotine exhibited all of the deficits associated with maternal tobacco and nicotine use, and linked to poor neonatal outcome: growth restriction, unstable breathing, and impaired arousal and catecholamine biosynthesis. Remarkably similar deficits were detected in pups lacking β2-containing nAChRs. Loss-of-function of these nAChRs consequently reproduces with astonishing fidelity many of the abnormalities caused by perinatal nicotine exposure. We propose that the underlying mechanisms of nicotine's detrimental side effects on a range of crucial defensive reflexes involve loss of function of nAChR subtypes, possibly via activity-dependent desensitization.
Keywords: knockout mice, nicotinic acetylcholine receptor, sudden infant death syndrome
Maternal smoking is the major preventable cause of poor pregnancy outcome in the industrialized world. At least 20–25% of women still smoke at some time during pregnancy, with an alarmingly higher prevalence in some (e.g., indigenous) groups (1, 2). These women have a greater risk of abortion, fetal death, and premature delivery, and are more likely to have infants whose physical and mental development is compromised. Infants of smoking mothers are typically smaller at birth, have more behavioral, lung, and respiratory abnormalities, and are more likely to die during sleep from sudden infant death syndrome (3, 4). These infants also appear to be more likely to take up smoking and become nicotine-dependent later in life (5, 6). Although the epidemiological association between passive exposure to tobacco products and poor infant outcome is strong, we still do not fully understand the underlying mechanisms linking the two.
Many of tobacco smoke's side effects on the unborn baby can be attributed to exposure to nicotine. Nicotine absorbed by the mother rapidly passes into the fetal bloodstream, then binds to nicotinic acetylcholine receptors (nAChRs) (7). Parts of the autonomic nervous system involved in regulating breathing, heart rate, blood pressure, and arousal are especially rich in nAChRs. Subtle abnormalities have been identified in these regions in smoke-exposed infants, indicating that one or more critical autonomic reflexes may be transiently or permanently altered (“reprogrammed”) by in utero nicotine exposure (8). Dysfunction in these key reflexes could destabilize breathing, heart rate, and blood pressure, increase the susceptibility to stress, and elevate the risk of sudden death or the prevalence of disease later in life (9).
To determine in detail how perinatal nicotine exposure compromises neonatal development, we have analyzed how nicotine's side effects are altered when genes encoding particular nAChRs are genetically deleted (“knocked out”). We focused on heteromeric nAChRs containing the β2 subunit, a major component of high-affinity nicotine binding sites in the nervous system (10). Our previous study, using adolescent mice, revealed that this nAChR subtype modulated breathing and arousal responses to stress, and mediated acute, nicotine-induced respiratory depression (11). In this new study, we investigated the role played by β2-containing nAChRs in mediating particular side effects of chronic nicotine exposure during fetal and early postnatal life. We implanted pregnant WT and mutant mice with microosmotic minipumps that delivered either water or a controlled dose of nicotine. Subsequently, we compared the development of the sympathoadrenal system and breathing and arousal reflexes of offspring pups shortly after birth, a period of increased vulnerability to nicotine's side effects. Our findings reveal that β2-containing nAChRs are involved in mediating a constellation of important side effects of nicotine, all of which are linked to poor neonatal outcome and sudden infant death syndrome. They also raise new questions about the safety of using nicotine-replacement therapies as alternatives for smoking during pregnancy.
Methods
Animals. We used 6-week-old male and female C57BL/J6 WT control mice, and male and female knockout (KO) mutants lacking β2-subunit-containing nAChRs. Mutants were siblings of parents backcrossed for 12 generations to C57BL/J6 inbred mice. Mice were housed in a quiet room at 22–23°C under a 12-h light-day cycle and provided with water and food pellets ad libitum. Timed matings were performed overnight; the presence of a vaginal “plug” the following morning was taken as a likely sign of successful mating, and this day was designated embryonic day 1.
Minipump Implantation. Alzet (Model 1002) osmotic minipumps of infusion rate 0.25μl·hr-1 for 14 days were implanted in gravid dams on embryonic day 14. Pumps were filled with either water (controls) or nicotine bitartrate calculated to deliver nicotine free base at 6.0 mg·kg-1 maternal weight·day-1, based on the average weight of dams at end-gestation (23 g). Minipumps were inserted s.c. (halothane anesthesia). Pups were studied on postnatal day (P) 5.
Plethysmography. Ventilation was measured by using dual-chamber plethysmograhy (12). A mask molded from soft tubing covered the pup's mouth and nostrils and was affixed to the surrounding skin with dental impression material (Impregum, 3M Co.). Pups breathed from one chamber (the headbox) while pressure changes due to thoracic expansion (inspiration) and contraction (expiration) were measured in a second, calibrated, measurement chamber. Warmed, humidified gas (air or hypoxia) was supplied to the headbox by switching between gas reservoirs. Gas exiting the headbox was forced through a drying column and analyzed for O2 and CO2 concentrations (S3A-II and CD3A gas analyzers, Amatec, Pittsburgh). The internal temperature of the plethysmograph was kept at 36°C (environmental thermoneutrality) by using a water jacket. Analogue signals were digitized at 200 Hz and stored.
Protocol. On admission into the plethysmograph, baseline data (air circulating in the headbox) were collected for 5 min, followed immediately by six cycles of 3 min hypoxia/3 min air. The total test duration was 43 min, because the final recovery period was 5 min. Hypoxia was 10% O2 plus 3% CO2, balance N2. At the conclusion of each study, the mask covering the snout was gently removed, and pups were killed by cervical dislocation after deep (pentobarbital) anesthesia. The medulla-pons (MP), paired carotid bodies (CB), petrosal ganglia (PG), superior cervical ganglia (SCG), and adrenal medullae (AM) were removed under a dissection microscope and immersed in perchloric acid-EDTA. Samples were retained at -80°C until analyzed. We measured dopamine, noradrenaline, and adrenaline content of the BT and (paired) CB, PG, SCG, and AM by using high-performance liquid chromatography coupled to an electrochemical detector (13). Tissues were disrupted ultrasonically, the homogenates were centrifuged, and 10 μl of supernatant was injected directly into a reverse-phase column (Licrospher RP 18 ec, 5 Am, 250 × 3 mm, Merck). The column was eluted with 50 mM citric acid, 50 mM sodium acetate, 1 mM EDTA-Na2, 437 mM acetic acid, 2.13 mM heptane sulfonate, and 7% methanol at 0.7 ml·min-1. Dopamine, noradrenaline, and adrenaline were quantified at +0.67 V with an Ag+/AgCl electrode (ELDEC 102, Chromatofield, Châteauneuf-les-Martigues, France). Detection limits, calculated by doubling the background noise level and expressed in picomoles per injection, were <0.03 pmol for all compounds, and the intraassay coefficient was 0.2%.
Data Analysis. The artifact due to body movement was excised from the recordings to measure ventilation. From the tidal volume (VT), respiratory rate (f), and minute ventilation (VE; = VT × f), we calculated sequential 30-sec mean values, expressed as % initial baseline. The hypoxic ventilatory response was the peak VE during the second to third minute (inclusive) of each hypoxic cycle (P1–P6). Body movements lasting ≥1 sec (“arousals”) were identified by pressure artifact (11). We also calculated the frequency of deep inspiratory efforts, or “sighs”(breaths with a VT more than or equal to twice that of the previous breath) (14). Catecholamine content was the average level measured in paired structures (13). We used a two-factor, repeated-measures ANOVA (statview 5.0, SAS Institute, Cary, NC) to test for significant main effects (genotype and treatment) and interactions between main effects (i.e., whether treatment effects varied with genotype). Where ANOVA was significant, modified t tests with the Bonferroni correction were applied a priori. P ≤ 0.05 was considered significant. Data are presented as group means ± SD in the text and Table 1, which is published as supporting information on the PNAS web site, and (for improved clarity) means ± SEM in figures. All procedures were approved by the Regional Animal Ethics Committee and conformed to European Community regulations.
Results
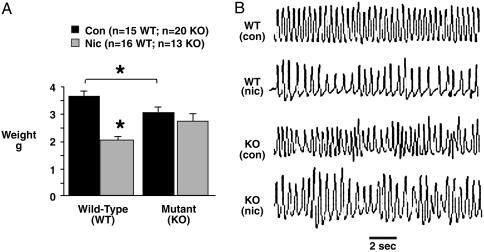
Nicotine Exposure Causes Growth Restriction via nAChRs. Fig. 1 shows growth data derived from pups of seven control dams (three WT and four KO) and six nicotine-exposed dams (three WT and three KO). Perinatal survival (the average number of live-born young per litter) was comparable across genotypes and treatment groups (n = 7 pups each group). Control WT pups were slightly (15%) larger than their mutant counterparts, indicating that the absence of functional β2-containing nAChRs restricts growth. Nicotine had dramatic effects on the growth of WT pups, which were severely growth restricted, weighing 43% less than controls of the same genotype. Nicotine exposure per se did not decrease the size of mutants, which were appropriately, although not normally, grown (Fig. 1 A).
Fig. 1.
Nicotine exposure restricts growth via β2-containing nAChRs. Nicotine-exposed WT pups were severely growth-restricted compared with WT controls (A). *, P < 0.0001. Nicotine-exposed mutants, however, were appropriately grown. Mutant as well as WT pups exposed to nicotine breathed more slowly at rest (B), suggesting that nicotine acts via non-β2-containing nAChRs to alter basal breathing rhythm. Con, control; Nic, nicotine-exposed. Error bars indicate SEM.
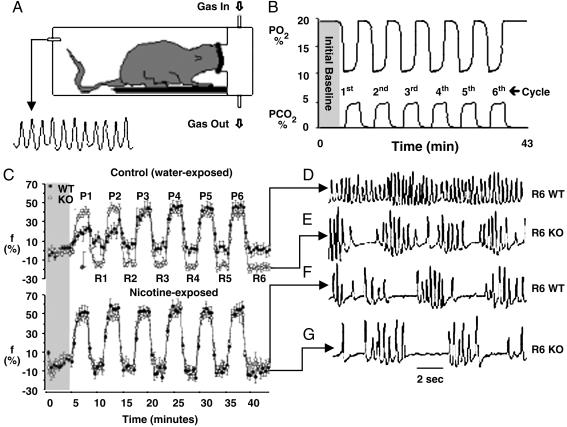
β2-Containing nAChRs Stabilize Breathing. We administered repetitive hypoxia to simulate the sort of stress that presents clinically in syndromes such as sleep apnoea, apnoea of prematurity, and asthma (Fig. 2 A and B) (15). Control pups of both genotypes breathed with similar rate and depth at rest, but major differences between breathing patterns emerged under stress. Mutant pups responded to bouts of hypoxia with a brisk increase in respiratory rate, followed by a precipitous fall during recovery. The mutant's recovery rhythm was always highly irregular because of frequent pauses between breaths (apnoeas) (Fig. 2 C and E), resulting in profound hypoventilation (VE fell 20% below the initial baseline) (data not shown). WT and mutant pups had a comparable net hypoxic ventilatory response (VE increased by ≈80% in both groups) (data not shown). However, WT pups achieved this by initially increasing breath volume (VT) rather than rate, although by the third bout of hypoxia, WT pups responded much like the mutant (compare P1 and P2 in Fig. 2C). The main difference between genotypes was that control WT pups never became apneic between bouts of hypoxia (Fig. 2 D and E).
Fig. 2.
Nicotine exposure destabilizes breathing via β2-containing nAChRs. Pups were studied in a plethysmograph (A). Inspired PO2 and PCO2 varied cyclically (B). Respiratory rate (f; % baseline) during hypoxia (P1–P6) and recovery in air (R1–R6) is shown (C), as is breathing rhythm after 43 min (D–G). Control WT pups had a weak rate response (P1 and P2 in C) and never became apneic during recovery (D). Control mutants were tachypneic during hypoxia (P1 and P2 in C) and became apneic during recovery (E). Nicotine-exposed WT pups behaved like the mutant (C Lower; compare F and G). Error bars indicate SEM.
Nicotine Destabilizes Breathing by Interfering with β2-Containing nAChRs. Nicotine-exposed pups of both genotypes breathed more slowly than did controls of the same genotype (baseline f was reduced by 14% and 25% in mutant and WT pups, respectively; P = 0.0007 nicotine vs. control) (Fig. 1B). Nicotine exposure dramatically altered the way WT pups breathed between bursts of hypoxia: Nicotine-exposed WT pups exhibited the same wild oscillations in respiratory rate (Fig. 2 C and F) and developed the same recovery instability and apnoea as did control mutants. By contrast, nicotine exposure did not significantly alter the respiratory phenotype of the mutant (Fig. 2 C, E, and G). The nicotine-exposed WT pups consequently resembled pups lacking functional β2-containing nAChRs.
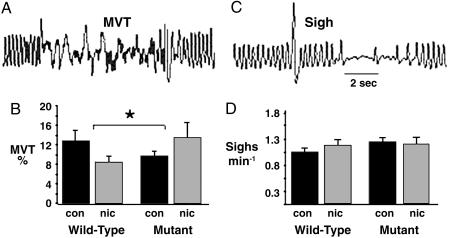
Nicotine Exposure Impairs Behavioral Arousal via Its High-Affinity Receptor. Body movement is an objective measure of the arousal response to distress (11). Nicotine-treated WT pups were less aroused by intermittent hypoxia compared with controls of the same genotype, indicating that nicotine exposure depressed brain (including cortical) arousal mechanisms. Surprisingly, nicotine-exposed mutants were more (not less) aroused by the same stimulus (Fig. 3 A and B). As expected, sighing, a brainstem response to hypoxia (14), occurred more frequently during hypoxia but, unlike movement, was not altered by nAChR mutation or nicotine exposure (Fig. 3 C and D). Nicotine exposure, thus, preferentially depressed cortical rather then brainstem responses to hypoxia, via β2-containing nAChRs.
Fig. 3.
Nicotine depresses hypoxic arousal. Arousal (body movement, MVT) was pressure artifact (A). Nicotine reduced arousal in WT pups but had the reverse effect in mutants (B). *, P = 0.02 for genotype-by-treatment interaction; MVT is expressed as % of total test. The frequency of sighing (a brainstem reflex that stabilizes lung volume) was comparable across genotypes and treatment groups (C and D). Interaction P = 0.3. Nicotine acting at β2-containing nAChRs consequently depresses cortical, not brainstem, reflexes. The brisk arousal response of the nicotine-exposed mutant may reflect greater catecholamine activity (Fig. 4). Error bars indicate SEM.
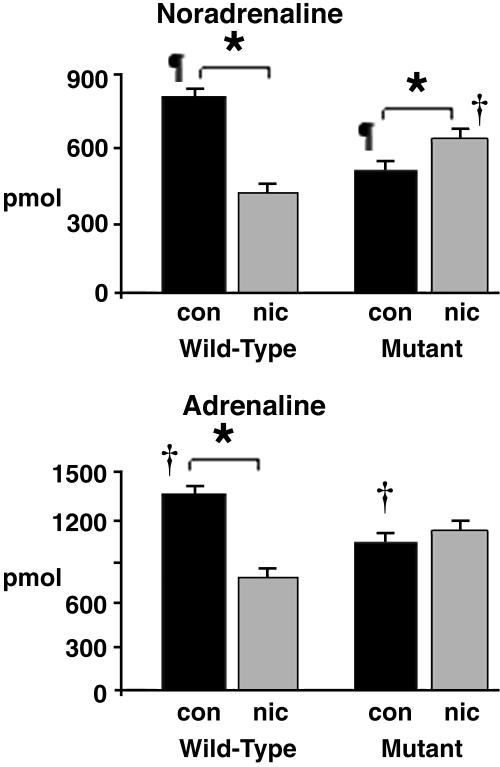
Inhibition of Catecholamine Biosynthesis by Nicotine Is Reversed in Mutants. We measured catecholamine (noradrenaline, adrenaline, and dopamine) levels at key points in the central and peripheral nervous system to determine how catecholamine reserves are affected by nicotine exposure and nAChR mutation. Null mutation resulted in a generalized decrease in catecholamines, especially in the strategically important adrenal medulla (Fig. 4 and Table 1). Nicotine exposure caused catecholamine levels of WT pups to fall to the levels recorded in mutants but had the reverse effect in mutants of increasing (not decreasing) catecholamine levels in most tissues.
Fig. 4.
Nicotine exposure reduces adrenal catecholamine biosynthesis. In control (water-exposed) pups, catecholamine content was lower in the mutant (KO). Nicotine exposure had opposing effects in WT and mutant (KO) pups, reducing levels in the former but elevating them in the latter (Right; *, †, and ¶, P < 0.05). Preservation of this reflex could have important survival benefits (nicotine-treated mutants). Also see Table 1. Error bars indicate SEM.
Discussion
We found that high-affinity, β2-containing nAChRs mediate a constellation of important side effects of nicotine exposure during pre- and postnatal life. Our study focused on the consequences of nicotine exposure during the last third of gestation, commencing around the time nAChRs first appear, and for a short period after birth (16, 17). The dose we used (6.0 mg·kg-1·day-1) is much greater than the average nicotine intake by pregnant women who smoke or use nicotine replacement patches (less than ≈0.5 mg·kg-1·day-1) (18). But because the mouse metabolizes nicotine much more rapidly than humans (plasma half lives are 5–10 min and 120 min, respectively), much higher doses must be given to the former to “mimic” consumption by the latter (19).
Smoking during pregnancy doubles the risk of giving birth to a small (“growth-restricted”) newborn (20). Slow fetal growth in this setting could partly reflect placental insufficiency, a complication of maternal smoking (7). Here, we show that chronic, low-level exposure to the nicotine in tobacco slows growth but not in the absence of high-affinity, β2-subunit-containing nAChRs. Nicotine, thus, acts via this nAChR subtype to “reprogram” growth and development, most likely directly within fetal tissues (21), and possibly by multiple mechanisms (7, 22, 23). Growth deficits associated with dysfunction in particular nAChRs early in life could predispose to disease later in life (24). We also provide intriguing evidence that nicotine exposure increases the risk of neonatal respiratory failure by disrupting nAChR-dependent mechanisms that help to stabilize breathing. Episodes of fast, deep breathing are normally followed by a slow, smooth return to the resting rhythm, due to compensatory mechanisms (neuroplasticity) that transiently prolong respiratory drive. We have previously shown that hypoxic neuroplasticity is partly regulated by the β2-containing nAChRs (11). The most likely explanation for the poststress apnoea seen in mutant and nicotine-exposed pups is that these mechanisms were not appropriately activated. As a result, respiratory drive fell too quickly during recovery from hypoxia, causing prolonged dysrhythmia (25). Instability of this sort could involve dysfunction in brainstem respiratory pacemakers. The pacemakers express multiple (including β2-containing) nAChRs and require cholinergic signals to develop and function normally (26). Chronic nicotine exposure could interfere with the central modulation of pacemaker excitability by these receptors, impairing the way breathing rhythm is fine-tuned to keep pace with sudden changes in demand (27). Peripheral complications associated with growth restriction (e.g., lung hypoplasia) could contribute to this instability (28). If the mechanisms underlying hypoxic respiratory neuroplasticity are weak in nicotine-exposed infants, breathing could become spontaneously, perhaps dangerously, unstable (29).
A failure to arouse during life-threatening situations is almost certainly part of the sequence of events leading to sudden infant death syndrome (30). We reveal here that chronic nicotine exposure depresses the arousability of the newborn via its high-affinity nAChR subtype, probably by impairing sensorimotor transmission within arousal pathways (11, 31, 32). Remarkably, exposing mutant pups to nicotine reversed this arousal deficit, indicating that nicotine has compensatory actions at non-β2-containing (e.g., α7 or β4) nAChRs that can restore arousal thresholds. This finding suggests that using drugs that discriminate between nAChR subtypes could be therapeutically useful in treating arousal disorders. The compensation shown by the mutant could be due to the partial “restoration” of catecholamine biosynthesis caused by nicotine exposure. Activation of catecholaminergic pathways (especially the adrenal medulla) by hypoxia stimulates arousal, as well as breathing, heart rate, and blood pressure. One of the recognized side effects of fetal nicotine exposure is a blunted adrenal catecholaminergic response to stress, which elevates perinatal mortality and worsens outcome (33, 34). It seems likely that this occurs partly in response to a loss of function of β2-containing nAChRs, because we found that catecholamine biosynthesis in WT pups was down-regulated by nicotine exposure. A critical role for these nAChRs in catecholamine biosynthesis, especially in the strategically important adrenal medulla, is somewhat surprising given that β2-containing nAChRs are not the predominant subtype in the peripheral nervous system (35).
The remarkable, multiple similarities we observed between mutant and nicotine-treated WT pups suggest that many of nicotine's side effects result from loss of function, plausibly a desensitization of β2-containing nAChRs. Prolonged nAChR activation by chronic nicotine indeed modifies slow conformational changes of the receptor protein and drives the receptors into long-lasting, inactive (desensitized) states. The result is a loss of synaptic response (36). The β2-containing nAChRs are preferentially desensitized by low-level nicotine (37), which may be the effect of perinatal nicotine exposure (38). Chronic nicotine exposure also triggers another well described phenomenon: a compensatory up-regulation in nAChR number and increase in nicotine binding sites (39). Whether the net effect of parallel changes in receptor sensitivity and density is to amplify or attenuate receptor function is uncertain. Our data suggest that β2-containing nAChR function is essentially lost during the period of nicotine exposure. The loss of one nAChR subtype could alter the way crucial defensive reflexes develop because it may unmask other (e.g., α7 or β4) nAChRs, enabling them to better express their potential roles. A shift in the balance of receptors may facilitate or attenuate the release of secondary neurotransmitters (37, 38), perhaps illustrated by our finding that nicotine exposure “restored” catecholamine levels and “reversed” the arousal deficit in mutants. Nicotinic receptor-mediated alterations in secondary neurotransmitter systems are strongly implicated in the pathophysiology of nicotine (40, 41).
In practical terms, because variability in nAChR sequence and gene expression affect the response to nicotine, an infant's nAChR genetic profile could partially affect his/her sensitivity to nicotine exposure during particular stages of development (10, 42). More importantly, because low-level nicotine exposure occurs under a variety of guises in pregnant women [use of “snuff” or nicotine replacement gum or patches (43, 44)], we warn that these may not be safe alternatives to smoking during pregnancy.
Supplementary Material
Acknowledgments
We thank Vivelca Karlsson and Eva Lundberg for administrative support, Ann-Christine Eklöf for implanting the minipumps, Erik Herlenius for data acquisition, Håkan Eriksson for constructing the plethysmograph, Yuri Shvarev and Miriam Katz-Salamon for useful discussions, and Hélen Braxenholm and Carina Tennfors for breeding the mice. This work was supported by Société de Tabacologie (France), the European Communities, the Collège de France, l'Association pour la Recherche sur le Cancer, the Swedish Heart and Lung foundation, the Swedish Research Council 5234, and I. L. Pesce (private bequest).
Author contributions: G.C., H.L., and J.-P.C. designed research; G.C., J.-C.R., and H.L. performed research; and R.G., G.M., and J.-P.C. contributed new reagents/analytic tools.
Abbreviations: KO, knockout; nAChR, nicotinic acetylcholine receptor; Pn, postnatal day n.
References
- 1.Walsh, R. S., Lowe, J. B. & Hopkins, P. J. (2001) Med. J. Aust. 175, 320-323. [DOI] [PubMed] [Google Scholar]
- 2.NSW Mothers and Babies Report 1997 (1997) Part 3: Aboriginal and Torres Strait Islander Mothers and Babies, www.health.nsw.gov.au/public-health/mdc97/3_7.htm.
- 3.Kleinman, J. C. (1988) Am. J. Epidemiol. 127, 274-282. [DOI] [PubMed] [Google Scholar]
- 4.Mitchell, E. A. & Milerad, J. (1999) in WHO International Consultation on Environmental Tobacco Smoke and Child Health (W.H.O., Geneva), 115-129.
- 5.Buka, S. L., Shenassa, E. D. & Niaura, R. (2003) Am. J. Psychiatry 160, 1978-1984. [DOI] [PubMed] [Google Scholar]
- 6.Abreu-Villaca, Y., Seidler, F. J. & Slotkin, T. A. (2004) Neuropsychopharmacology 29, 1440-1450. [DOI] [PubMed] [Google Scholar]
- 7.Slotkin, T. (1998) J. Pharmacol. Exp. Ther. 285, 931-945. [PubMed] [Google Scholar]
- 8.Nachmanoff, D. B., Panigrahy, A., Filiano, J. J., Mandell, F., Sleeper, L. A., Valdes-Dapena, M., Krous, H. F., White, W. F. & Kinney, H. C. (1998) J. Neuropathol. Exp. Neurol. 57, 1018-1025. [DOI] [PubMed] [Google Scholar]
- 9.Harper, R. M., Kinney, H. C., Fleming, P. J. & Thach, B. T. (2000) Respir. Physiol. 119, 123-132. [DOI] [PubMed] [Google Scholar]
- 10.Le Novere, N., Corringer, P. J. & Changeux, J.-P. (2002) J. Neurobiol. 53, 447-456. [DOI] [PubMed] [Google Scholar]
- 11.Cohen, G., Han, Z., Grailhe, R., Gallego, J., Gaultier, C., Changeux, J.-P. & Lagercrantz, H. (2002) Proc. Natl. Acad. Sci. USA 99, 13273-13277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Frappell, P. B. & Mortola, J. P. (2000) Respir. Physiol. 120, 35-45. [DOI] [PubMed] [Google Scholar]
- 13.Roux, J.-C., Mamet, J., Perrin, D., Peyronnet, J., Royer, C., Cottet-Emard, J. M., Pequignot, J. M. & Dalmaz, Y. (2003) J. Neural Transm. 110, 51-65. [DOI] [PubMed] [Google Scholar]
- 14.Cohen, G. & Henderson-Smart, D. J. (1996) J. Physiol. 490, 551-557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Neubauer, J. A. (2001) J. Appl. Physiol. 90, 1593-1599. [DOI] [PubMed] [Google Scholar]
- 16.Atluri, P., Fleck, M. W., Shen, Q., Mah, S. J., Stadfelt, D., Barnes, W., Goderie, S. K., Temple, S. & Schneider, A. S. (2001) Dev. Biol. 240, 143-156. [DOI] [PubMed] [Google Scholar]
- 17.Narayanan, U., Birru, S., Vaglenova, J. & Breese, C. R. (2002) NeuroReport 13, 961-963. [DOI] [PubMed] [Google Scholar]
- 18.Dempsey, D., Jacob, P. & Benowitz, N. L. (2002) J. Pharmacol. Exp. Ther. 301, 594-598. [DOI] [PubMed] [Google Scholar]
- 19.Van de Kamp, J. L. & Collins, A. C. (1994) Pharmacol. Biochem. Behav. 47, 889-900. [DOI] [PubMed] [Google Scholar]
- 20.Haug, K., Irgens, L. M., Skjaerven, R., Markestad, T., Baste, V. & Schreuder, P. (2000) Acta Obstet. Gynecol. Scand. 79, 485-489. [PubMed] [Google Scholar]
- 21.Sharma, G. & Vijayaraghavan, S. (2002) J. Neurobiol. 53, 524-534. [DOI] [PubMed] [Google Scholar]
- 22.Grove, K. L., Sekhon, H. S., Brogan, R. S., Keller, J. A., Smith, M. S. & Spindel, E. R. (2001) Clin. Endocrinol. Metab. 86, 5420-5426. [DOI] [PubMed] [Google Scholar]
- 23.Sekhon, H. S., Keller, J. A., Benowitz, N. L. & Spindel, E. R. (2001) Am. J. Respir. Crit. Care Med. 164, 989-994. [DOI] [PubMed] [Google Scholar]
- 24.Robinson, S. M. & Barker, D. J. (2002) Proc. Nutr. Soc. 61, 537-542. [DOI] [PubMed] [Google Scholar]
- 25.Khoo, M. C. (2000) Respir. Physiol. 122, 167-182. [DOI] [PubMed] [Google Scholar]
- 26.Hilaire, G. & Duron, B. (1999) Physiol. Rev. 79, 325-360. [DOI] [PubMed] [Google Scholar]
- 27.Shao, X. M. & Feldman, J. L. (2002) J. Neurophysiol. 88, 1851-1858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lechner, A. J. & Tull, D. S. (1986) Pediatr. Res. 20, 920-924. [DOI] [PubMed] [Google Scholar]
- 29.Fleming, P. J., Goncalves, A. L., Levine, M. R. & Woollard, S. (1984) J. Physiol. 347, 1-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sawnani, H., Jackson, T., Murphy, T., Beckerman, R. & Simakajornboon, N. (2004) Am. J. Respir. Crit. Care Med. 169, 733-738. [DOI] [PubMed] [Google Scholar]
- 31.Oliff, H. S. & Gallardo, K. A. (1991) Front. Biosci. 4, D883-D897. [DOI] [PubMed] [Google Scholar]
- 32.Saint-Mleux, B., Eggermann, E., Bisetti, A., Bayer, L., Machard, D., Jones, B. E., Muhlethaler, M. & Serafin, M. (2004) J. Neurosci. 24, 63-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Slotkin, T. A., Lappi, S. E., McCook, E. C., Lorber, B. A. & Seidler, F. J. (1995) Brain Res. Bull. 38, 69-75. [DOI] [PubMed] [Google Scholar]
- 34.Oncken, A., Henry, K. M., Campbell, W. A., Kuhn, C. M., Slotkin, T. A. & Kranzler, H. R. (2003) Pediatr. Res. 53, 119-124. [DOI] [PubMed] [Google Scholar]
- 35.Di Angelantonio, S., Matteoni, C., Fabbretti, E. & Nistri, A. (2003) J. Neurosci. 17, 2313-2322. [DOI] [PubMed] [Google Scholar]
- 36.Changeux, J.-P. & Edelstein, S. J. (1998) Neuron 21, 959-980. [DOI] [PubMed] [Google Scholar]
- 37.Mansvelder, H. D., Keath, J. R. & McGehee, D. S. (2002) Neuron 33, 905-919. [DOI] [PubMed] [Google Scholar]
- 38.Cormier, A., Paas, Y., Zini, R., Tillement, J.-P., Lagrue, G., Changeux, J.-P. & Grailhe, R. (2004) Mol. Pharmacol. 66, 1712-1718. [DOI] [PubMed] [Google Scholar]
- 39.Sparks, J. A. & Pauly, J. R. (1999) Psychopharmacology 141, 145-153. [DOI] [PubMed] [Google Scholar]
- 40.Cordero-Erausquin, M. & Changeux, J.-P. (2001) Proc. Natl. Acad. Sci. USA 98, 2803-2807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kinney, H. C., Filiano, J. J. & White, W. F. (2001) J. Neuropathol. Exp. Neurol. 60, 228-247. [DOI] [PubMed] [Google Scholar]
- 42.Weese-Mayer, D. E., Berry-Kravis, E. M., Zhou, L., Maher, B. S., Curran, M. E., Silvestri, J. M. & Marazita, M. L. (2004) Pediatr. Res. 56, 391-395. [DOI] [PubMed] [Google Scholar]
- 43.England, L. J., Levine, R. J., Mills, J. L., Klebanoff, M. A., Yu, K. F. & Cnattingius, S. (2003) Am. J. Obstet. Gynecol. 189, 939-943. [DOI] [PubMed] [Google Scholar]
- 44.Coleman, T., Britton, J. & Thornton, J. (2004) Brit. Med. J. 328, 965-966. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.