Abstract
BACKGROUND
It is unknown why human metapneumovirus (HMPV) and respiratory syncytial virus (RSV) cause severe respiratory infection in children, particularly in premature infants. Our aim was to investigate if there are defective airway antiviral responses to these viruses in young children with history of prematurity.
METHODS
Nasal airway secretions were collected from 140 children ≤3 y old without detectable virus (n = 80) or with PCR-confirmed HMPV or RSV infection (n = 60). Nasal protein levels of IFNγ, CCL5/RANTES, IL-10, IL-4, and IL-17 were determined using a multiplex magnetic bead immunoassay.
RESULTS
Full-term children with HMPV and RSV infection had increased levels of nasal airway IFNγ, CCL5, and IL-10 along with an elevation in Th1 (IFNγ)/Th2 (IL-4) ratios, which is expected during antiviral responses. In contrast, HMPV-infected premature children (< 32 wk gestation) did not exhibit increased Th1/Th2 ratios or elevated nasal airway secretion of IFNγ, CCL5, and IL-10 relative to uninfected controls.
CONCLUSION
Our study is the first to demonstrate that premature infants have defective IFNγ, CCL5/RANTES, and IL-10 airway responses during HMPV infection and provides novel insights about the potential reason why HMPV causes severe respiratory disease in children with history of prematurity.
Viruses are the primary cause of respiratory illness in humans. During infancy and early childhood, two RNA viruses from the Paramyxoviridae family, respiratory syncytial virus (RSV), and human metapneumovirus (HMPV), are responsible for a large number of cases of lower respiratory tract infections (1–5). These RNA viruses are not only phylogenetically related (1), but also share similar clinical respiratory signs and symptoms including cough, wheezing, rales, hypoxemia, and respiratory distress in high-risk groups (6–10). Interestingly, both viruses tend to cause more severe disease in young individuals with history of prematurity (gestational age <32 wk) (2–5). Indeed, RSV is associated with severe respiratory infections in premature babies, and prophylaxis with a humanized monoclonal antibody against the fusion (F) protein of RSV (Palimizumab) is recommended for them during their first RSV seasons (11). There are also reports describing severe HMPV infections in young children born extremely premature (5). Importantly, the nature of the human antiviral airway immune responses to RSV and HMPV infections in premature children has been remarkably understudied.
Previous studies have established that RSV and HMPV elicit antiviral immune responses consisting of virally-induced production of IFNγ, CCL5//RANTES, and IL-10 (12). Interestingly, despite the clinical and genetic similarities between these respiratory viruses, HMPV infections are characterized by attenuated IFN-γ, IL-10, and CCL5/RANTES responses relative to RSV (12). Although the mechanism by which HMPV alters IFN-mediated responses is not completely understood, there is in vitro evidence demonstrating that HMPV alters IFN-mediated activation of interferon-stimulated response elements, interferon-stimulated genes and STAT 1 downstream signaling (13,14). It is however unclear if this HMPV-induced defective antiviral immunity is present during naturally occurring infections in humans, particularly in young children with history of prematurity in which HMPV is known to cause more severe disease. Accordingly, the overall goal of this study was to investigate if infants and young children (<3 y of age) born premature (<32 wk gestation) exhibit impaired airway secretion of IFNγ, CCL5//RANTES, and IL-10 during naturally occurring HMPV infections.
RESULTS
Baseline Characteristics
One hundred and fourty children were included in this study. The total study population was subdivided by viral-specific PCR analysis into control without detectable virus (n = 80) and cases of RSV (n = 30) or HMPV infection (n = 30). Comparison of baseline demographic characteristics among groups with regards to gender, age, or ethnicity revealed no significant differences. The vast majority of children included in the study were inpatient in all the study groups (Table 1).
Table 1.
Baseline characteristics for subjects
| Group | Control | Respiratory syncytial virus | Human metapneumovirus | P value |
|---|---|---|---|---|
| N | 80 | 30 | 30 | |
| Premature (<32 wk), n (%) | 40 (50) | 7 (23) | 9 (30) | NS |
| Male, n (%) | 49 (62) | 19 (63) | 15 (50) | NS |
| Age (y), mean (SD) | 1.3 (0.8) | 1.5 (1.1) | 1.9 (1.2) | NS |
| Black, n (%) | 48 (40) | 13 (43) | 14 (45) | NS |
| Hospitalized, n (%) | 76 (95) | 29 (97) | 29 (97) | NS |
Airway Antiviral IFNγ Responses in Infants During RSV and HMPV Infection
Analysis of nasal IFNγ protein levels showed that young children born full term with RSV or HMPV infection had higher mean nasal IFNγ responses compared to controls without an identifiable virus (RSV: 0.80 ± 0.15 pg/ml vs. control: 0.29 ± 0.09 pg/ml; P < 0.05; HMPV: 0.88 ± 0.13 pg/ml vs. control: 0.29 ± 0.09 pg/ml; P < 0.05; Figure 1). Interestingly, subjects with history of prematurity (GA < 32 wk) and RSV infection had higher mean nasal levels of IFNγ than premature children (RSV: 0.75 ± 0.12 pg/ml vs. control: 0.32 ± 0.07 pg/ml; P < 0.05; Figure 1a), however, children born premature with HMPV infection did not exhibit significant differences in the IFNγ nasal levels compared to controls (HMPV: 0.11 ± 0.29 pg/ml vs. control: 0.32 ± 0.07 pg/ml; P = 0.29; Figure 1b). Collectively, these results suggest that there is a defective antiviral IFNγ airway response in premature children with HMPV.
Figure 1.
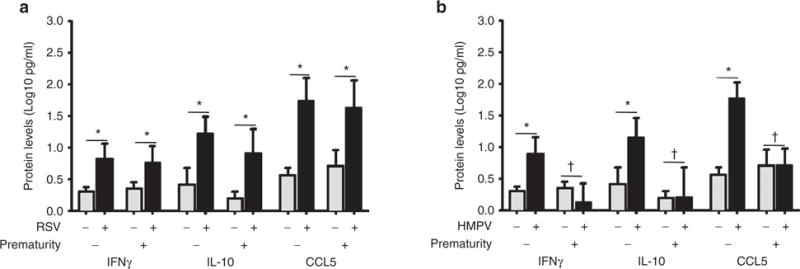
Nasal airway IFNγ, IL-10, and CCL5/RANTES protein levels in full-term and premature children with respiratory syncytial virus (RSV) or human metapneumovirus (HMPV). (a) IFNγ, IL-10, and CCL5/RANTES during RSV infection (black bars) in full-term (n = 23) and premature children (n = 7) vs. uninfected controls (gray bars) born full-term (n = 40) or premature (n = 40). (b) IFNγ, IL-10, and CCL5/RANTES during HMPV infection (black bars) in full-term (n = 21) and premature children (n = 9) vs. uninfected controls (gray bars) born full-term (n = 40) or premature (n = 40). Data are presented as mean and 95% confidence interval; P values presented as *< 0.05; †not significant.
Premature Infants Have Deficient CCL5/RANTES and IL-10 Airway Responses During HMPV Infection
Airway secretion of CCL5/RANTES and IL-10 has been identified in vitro and in vivo during infection with paramixoviruses (8,12,15,16). Accordingly, to further investigate the antiviral responses to RSV and HMPV, we measured nasal protein levels of CCL5/RANTES and IL-10 in full-term and premature children. As shown in Figure 1, relative to controls, full-term subjects with RSV had higher nasal protein levels of CCL5/RANTES (RSV: 1.72 ± 0.18 pg/ml vs. control: 0.54 ± 0.12 pg/ml; P < 0.05; Figure 1a) and IL-10 (RSV: 1.2 ± 0.14 pg/ml vs. control: 0.4 ± 0.13 pg/ml; P < 0.05; Figure 1a). Full-term children with HMPV infection also exhibited increased nasal protein levels of CCL5/RANTES (HMPV: 1.76 ± 0.12 vs. 0.54 ± 0.06 pg/ml; P < 0.05; Figure 1b) and IL-10 (HMPV: 1.13 ± 0.16 vs. 0.4 ± 0.13 pg/ml; P < 0.05; Figure 3b) compared to controls.
Figure 3.
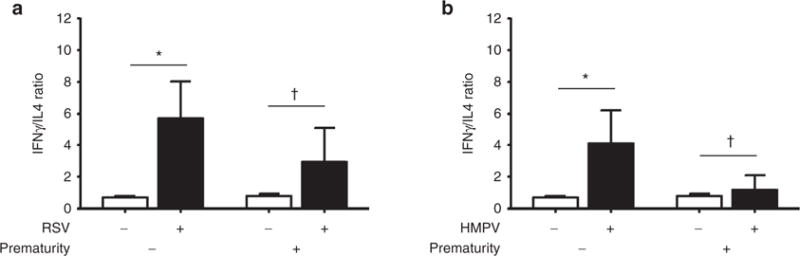
Nasal airway IFN γ/Th2 cytokine ratio in full-term and premature children with respiratory syncytial virus (RSV) or human metapneumovirus (HMPV). (a) IFN γ and Th2 (IL-4) cytokine ratios in children during RSV infection (black bars) in full-term (n = 23) and premature children (n = 7) vs. uninfected controls (white bars) born full-term (n = 40) or premature (n = 40). (b) IFN γ and Th2 (IL-4) cytokine ratios during HMPV infection (black bars) in full-term (n = 21) and premature children (n = 9) vs. uninfected controls (white bars) born full-term (n = 40) or premature (n = 40). Data are presented as mean and 95% confidence interval; P values presented as *< 0.05; † not significant.
Interestingly, although premature children with RSV infection also had higher protein levels of CCL5/RANTES (RSV: 1.61 ± 0.19 vs. 0.7 ± 1.2 pg/ml; P < 0.05; Figure 1a) and IL-10 (RSV: 0.89 ± 0.19 vs. 0.18 ± 0.05 pg/ml; P < 0.05), premature subjects with HPMV infection did not have significant differences in the nasal airway protein levels of CCL5/RANTES and IL-10 compared to controls (CCL5/RANTES: HMPV 0.68 ± 0.13 pg/ml vs. control 0.7 ± 0.12 pg/ml; P = 0.9; IL-10: HMPV 0.19 ± 0.24 pg/ml vs. control 0.18 ± 0.05 pg/ml; P = 0.9; Figure 1b).
Th2/Th17 Cytokine Profile in Infants With Naturally Occurring HMPV Infection
We next investigated the natural Th2 and Th17 immune response to RSV and HMPV infections in full-term and premature individuals by measuring nasal protein levels of IL-4 and IL-17 in the same study groups. As shown in Figure 2, there were no significant differences in the protein levels of IL-4 and IL-17 among the full-term and premature groups with nondetectable virus (control), RSV infection (Figure 2a) or HMPV infection (Figure 2b). When we examined relative antiviral Th1 (IFNγ) vs. allergic Th2 (IL-4) responses (Figure 3), we identified that full-term subjects elicited an expected predominance of antiviral Th1 response during RSV or HMPV infections according to significant increased IFNγ/IL-4 ratios (RSV: 5.7, 95% CI 2.87–8.5 vs. control: 0.71 95% CI 0.63–0.78; P < 0.05; HMPV: 4.11, 95% CI 1.5–6.7 vs. control: 0.71 95% CI 0.63–0.78; P < 0.05).
Figure 2.
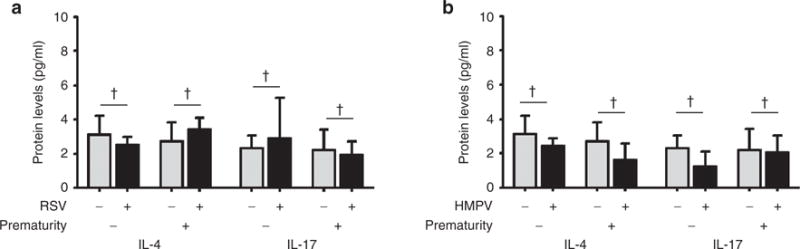
Nasal airway Th2/Th17 cytokines in full-term and premature children with respiratory syncytial virus (RSV) or human metapneumovirus (HMPV). (a) Th2 (IL-4) and Th17 (IL-17) cytokine levels in children during RSV infection (black bars) in full-term (n = 23) and premature children (n = 7) vs. uninfected controls (gray bars) born full-term (n = 40) or premature (n = 40). (b) IL-4 and IL-17 during HMPV infection (black bars) in full-term (n = 21) and premature children (n = 9) vs. uninfected controls (gray bars) born full-term (n = 40) or premature (n = 40). Data are presented as mean and 95% confidence interval; P values presented as † not significant.
Premature children overall exhibited a less prominent increase in IFNγ/IL4 ratios during RSV or HMPV infection. Specifically, premature children with RSV infection had increase in IFNγ/IL4 ratio but it did not reach statistical significance (RSV: 2.95, 95% CI 0.37–5.54 vs. control: 0.81 95% CI 0.65–0.97; P = 0.06; Figure 3a). Moreover, the IFNγ/IL4 ratio in premature children infected with HMPV was essentially unchanged relative to controls (HMPV: 1.18, 95% CI 0.05–2.3 vs. control: 0.81 95% CI 0.65–0.97; P = 0.23; Figure 3b). Collectively, these data suggest that premature children have less preponderance of antiviral Th1 response during naturally occurring RSV or HMPV infections compared to full-term children.
DISCUSSION
We studied a prospective cohort of young children aged less than or equal to 3 y to determine the association between prematurity and nasal antiviral airway cytokine responses to HMPV or RSV. To the best of our knowledge, our study is the first to demonstrate that premature infants have defective IFNγ, CCL5/RANTES, and IL-10 airway responses during HMPV infection and provides novel insights about the potential reason why HMPV causes severe respiratory disease in premature children.
Prematurity has been linked to an increased risk of severe viral respiratory infections during the first years of life (2–5). Diminished lung function may predispose prematurely born infants to severe viral respiratory illnesses (17). Moreover, antibody-mediated immunity, which depends maternal transfer of specific IgG across the placenta, seems to be immature in premature babies (18). In addition, T helper (Th) cytokine responses in early life are largely driven toward a Th2 phenotype leading to lower production of Th1 antiviral cytokines such as IFNγ (19–21). This deficient cytokine production in neonates may explain the susceptibility of this age group to certain pathogens. For instance, preterm infants have deficient cytokine responses to group B streptococcus, an important cause of sepsis in newborns (22). Complementing these findings, our study indicates that prematurity is also associated with abnormal airway cytokine responses during paramixovirus infections (RSV and HMV) according to IFNγ/IL-4 ratios (indicative of Th1 antiviral vs. Th2 atopic responses), which were lower in premature children infected with RSV and essentially unchanged during HMPV infection.
HMPV is a virus phylogenetically related to RSV that causes severe respiratory disease in premature infants (5). Interestingly, HMPV induces an airway immune response distinct from that of RSV (12,23–25). In fact, the ability of HMPV—but not RSV—to ameliorate the nasal airway secretion of CCL5/RANTES was originally reported in 2002 by Jartti et al. (8), in one of the first articles describing HMPV infections as a cause of wheezing in children. This initial finding suggested the presence of IFN-mediated defects during HMPV infection since CCL5/RANTES expression is regulated via IFN regulatory factor 1 (IRF-1) (26). Recent reports have confirmed that HMPV elicits weak IFN-mediated antiviral responses and consequent lower production of CCL5/RANTES and IL-10 in human peripheral blood mononuclear cells relative to RSV (12). The latter results coincide with the reduced HMPV-induction of CCL5/RANTES and IL-10 seen in murine models (22,23). In agreement with these studies, we have now demonstrated that premature children infected with HMPV exhibit significantly lower levels of nasal airway IFNγ, CCL5/RANTES, and IL-10 compared to RSV-infected premature children (Figure 1a) or full-term individuals with HMPV infection (Figure 1b). These new data indicate that HMPV is capable of attenuating IFNγ, CCL5/RANTES, and IL-10 responses in vivo and, to the best of our knowledge, is the first study to link prematurity to the previously reported defective antiviral immunity observed during HMPV infection (12–14).
The exact reason why HMPV elicits markedly weaker IFNγ (and IL-10, CCL5) responses than RSV is still an area of active research, but there are several postulated mechanisms of defective IFN-driven antiviral immunity during HMPV infection. For instance, the structural encoding proteins of HMPV—glycoprotein G and M2-2—seem to play a critical role in modulating antiviral IFN production during HMPV infections (26–28) as demonstrated by the observation that a mutated HMPV virus lacking glycoprotein G and M2 protein induces significantly higher amounts of IFN and antiviral cytokines/chemokines than wild-type HMPV (27). HMPV also appears to interfere with the activation of the IFN signaling cascade at different points, including Jak1, Tyk2, and IFNAR1 membrane expression (29), all of which may lead to downstream inhibition of STAT1 and STAT2 (13,29). Although our study supports the concept that HMPV infection is associated with abnormal IFNγ secretion, future translational studies are needed to investigate whether the postulated HMPV-induced mechanisms of defective antiviral IFN-driven airway responses are relevant to natural HMPV infection in humans, particularly in the pediatric population.
Only few studies have investigated antiviral airway immune responses during naturally occurring HMPV and RSV infections in children. The most important limitation of pediatric studies has been the access to lung samples in this age group. To overcome this limitation, we used nasal samples, an approach used as surrogate in the evaluation of airway cytokine responses in children during naturally occurring viral respiratory infections (16,25,30). Indeed, a pioneering study conducted by the INFANT foundation used this strategy to delineate the airway cytokine profile during natural viral infections in a prospective cohort of infants (25). Similar to our current results, this study identified that subjects with HMPV had lower IFN-γ/IL-4 ratios than did those infected with RSV or influenza virus (25), however, this study did not specifically investigate these responses in premature children. Using the same nasal washing approach, we recently identified that rhinovirus (RV) infection in early life is associated with increased nasal airway levels of the classical Th2 cytokine IL-4 (31). In our current study, we did not observe significant differences in the nasal airway secretion of IL-4 or the Th17 cytokine IL-17 in children with naturally occurring RSV or HMPV infection (Figure 2). These data highlight the prevailing notion that airway cytokine profiles are influenced by host- and viral-specific factors (32). For instance, pediatric hosts with wheeze and/or atopy have a differential antiviral cytokine profile influenced by the infecting virus (32). Our study suggests that in the case of the premature host infected with HMPV, the predominant immune profile is an attenuated airway secretion of IFNγ, CCL5/RANTES, and IL-10, which might underlie the severity of HMPV in this group of children.
Limitations of the present study include the lack of information on clinical data (i.e., severity scores, outcomes) and viral load, which we recognize would be important to establish the clinical relevance of the different cytokine profiles among premature and full-term groups. In addition, given the cross-sectional design of our study we do not have comparisons of baseline vs. viral induces cytokines, thus the results from this study could also be interpreted as premature infant responses being consistent with previous reports in the literature and the full-term infant responses as being abnormally exuberant. Accordingly, longitudinal studies that correlate baseline and viral-induced cytokines levels with disease relevant markers (hospitalization rates, severity scores) are still required to confirm our findings and to establish the clinical significance of the results presented. Future studies should also investigate if children born late preterm (32 to <37 wk gestation) exhibit impaired airway antiviral IFNγ responses to RSV and or HMPV. This is an important issue because of changing RSV prophylaxis recommendations for the older preterm infants (11).
Conclusion
In summary, in this cross sectional study, premature infants exhibited defective IFNγ, CCL5/RANTES, and IL-10 airway responses during HMPV infection. These new data support the previously described HMPV-induced defective IFN antiviral immunity in human-based cellular and animal models (12–14), as well as in the nasal airway secretions of infants with naturally occurring viral infections (27), and provide new insights about the potential underlying reason why HMPV causes severe respiratory disease in young individuals with history of prematurity.
METHODS
Study Population
This is a prospective cohort (n = 140) in which we determined the association between prematurity and nasal airway cytokine responses to HMPV or RSV. Our study population were children aged less than or equal to 3 y who underwent nasal lavage for diagnostic purposes (respiratory virus detection by PCR) either in the outpatient/inpatient setting or during emergency department visits in our pediatric medical center (February 2013 to March 2014). For the purpose of the study “prematurity” was defined a priori by a gestational age of less than 32 wk to include extremely preterm and very preterm subjects based on WHO definition of prematurity (33,34). We did not include children born late preterm (32 to <37 wk gestation) as we wanted to focus initially in the group of very premature children that is at the highest risk of developing severe respiratory illnesses during RSV and HMPV infections (2–5). “Full term” was defined by a gestational age of more than or equal to 37 wk. HMPV and RSV infections were confirmed by PCR analysis used for clinical purposes in our institution. Subjects with other viruses or mixed viral infections were excluded. “Controls” were defined as age-matched full-term and premature subjects with a negative viral PCR. Clinical and demographic variables included gestational age in weeks, age, gender, and ethnicity and were obtained by reviewing electronic medical records in our institution. We excluded subjects with insufficient/inadequate nasal sample for cytokine profiling and those with incomplete medical records. This study was approved by the Institutional Review Board of Children’s National Medical Center, Washington D.C. Waiver of consent was obtained by the Institutional Review Board, as the study was carried out with samples already collected for clinical purposes (respiratory viral diagnosis).
Nasal Washing Collection, Viral PCR Analysis, and Cytokine Measurements
Nasal airway secretions were collected at the onset of acute respiratory illnesses by a standard nasal lavage technique consisting of gently washing the nasal cavity with 3–4 ml sterile normal saline. Secretions were aliquoted and stored at −80 °C until further analysis. Nasal samples were analyzed by a viral multiplex PCR panel for 10 targets (rhinovirus, RSV, HMPV, parainfluenza 1–3, influenza A and B, H1N1, H1N3, Adenovirus) used for clinical purposes (Luminex, Austin, TX) according to the microbiology laboratory protocol of our institution. Nasal washings were analyzed for protein levels of IFNγ, CCLX5/RANTES, IL-10, IL-4, and IL-17 using a commercially available multiplex magnetic bead immunoassay (Millipore, Billerica, MA) according to the manufacturers’ instructions using provided standards and quality controls.
Statistical Analysis
Data were analyzed using Minitab 16 software package for Windows (Minitab, State College, PA). All data are reported as mean ± SE, 95% confidence intervals or as fold changes relative to control values. Log10 transformation of cytokines values was done to normalize the distribution of IFN-γ, IL-10, and CCL-5/RANTES. Data within each group (prematurity and control) and between prematurity and control groups were analyzed with two sample t-test or nonparametric Mann–Whitney U-test when applicable. A probability of <0.05 was considered statistically significant.
Acknowledgments
STATEMENT OF FINANCIAL SUPPORT: This work was supported by National Institutes of Health, Bethesda, MD, grants K12-HL090020, K12-HD001399 and UL1TR000075 KL2TR000076.
Footnotes
Disclosure: Authors do not have any financial ties to products in the study or potential/perceived conflicts of interest
References
- 1.van den Hoogen BG, de Jong JC, Groen J, et al. A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat Med. 2001;7:719–24. doi: 10.1038/89098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rodríguez DA, Rodríguez-Martínez CE, Cárdenas AC, et al. Predictors of severity and mortality in children hospitalized with respiratory syncytial virus infection in a tropical region. Pediatr Pulmonol. 2014;49:269–76. doi: 10.1002/ppul.22781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Edwards KM, Zhu Y, Griffin MR, et al. New Vaccine Surveillance Network Burden of human metapneumovirus infection in young children. N Engl J Med. 2013;368:633–43. doi: 10.1056/NEJMoa1204630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Williams JV, Harris PA, Tollefson SJ, et al. Human metapneumovirus and lower respiratory tract disease in otherwise healthy infants and children. N Engl J Med. 2004;350:443–50. doi: 10.1056/NEJMoa025472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Papenburg J, Hamelin MÈ, Ouhoummane N, et al. Comparison of risk factors for human metapneumovirus and respiratory syncytial virus disease severity in young children. J Infect Dis. 2012;206:178–89. doi: 10.1093/infdis/jis333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Widmer K, Zhu Y, Williams JV, Griffin MR, Edwards KM, Talbot HK. Rates of hospitalizations for respiratory syncytial virus, human metapneumovirus, and influenza virus in older adults. J Infect Dis. 2012;206:56–62. doi: 10.1093/infdis/jis309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mullins JA, Erdman DD, Weinberg GA, et al. Human metapneumovirus infection among children hospitalized with acute respiratory illness. Emerg Infect Dis. 2004;10:700–5. doi: 10.3201/eid1004.030555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jartti T, van den Hoogen B, Garofalo RP, Osterhaus AD, Ruuskanen O. Metapneumovirus and acute wheezing in children. Lancet. 2002;360:1393–4. doi: 10.1016/S0140-6736(02)11391-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Madhi SA, Ludewick H, Kuwanda L, et al. Pneumococcal coinfection with human metapneumovirus. J Infect Dis. 2006;193:1236–43. doi: 10.1086/503053. [DOI] [PubMed] [Google Scholar]
- 10.Williams JV, Tollefson SJ, Heymann PW, Carper HT, Patrie J, Crowe JE. Human metapneumovirus infection in children hospitalized for wheezing. J Allergy Clin Immunol. 2005;115:1311–2. doi: 10.1016/j.jaci.2005.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Committee on Infectious Diseases and Bronchiolitis Guidelines Committee. Updated guidance for palivizumab prophylaxis among infants and young children at increased risk of hospitalization for respiratory syncytial virus infection. Pediatrics. 2014;134:415–20. doi: 10.1542/peds.2014-1665. [DOI] [PubMed] [Google Scholar]
- 12.Douville RN, Bastien N, Li Y, Pochard P, Simons FE, HayGlass KT. Human metapneumovirus elicits weak IFN-gamma memory responses compared with respiratory syncytial virus. J Immunol. 2006;176:5848–55. doi: 10.4049/jimmunol.176.10.5848. [DOI] [PubMed] [Google Scholar]
- 13.Kolli D, Bao X, Casola A. Human metapneumovirus antagonism of innate immune responses. Viruses. 2012;4:3551–71. doi: 10.3390/v4123551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dinwiddie DL, Harrod KS. Human metapneumovirus inhibits IFN-alpha signaling through inhibition of STAT1 phosphorylation. Am J Respir Cell Mol Biol. 2008;38:661–70. doi: 10.1165/rcmb.2007-0285OC. [DOI] [PubMed] [Google Scholar]
- 15.Bacharier LB, Cohen R, Schweiger T, et al. Determinants of asthma after severe respiratory syncytial virus bronchiolitis. J Allergy Clin Immunol. 2012;130:91–100.e3. doi: 10.1016/j.jaci.2012.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lewis TC, Henderson TA, Carpenter AR, et al. Nasal cytokine responses to natural colds in asthmatic children. Clin Exp Allergy. 2012;42:1734–44. doi: 10.1111/cea.12005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Drysdale SB, Wilson T, Alcazar M, et al. Lung function prior to viral lower respiratory tract infections in prematurely born infants. Thorax. 2011;66:468–73. doi: 10.1136/thx.2010.148023. [DOI] [PubMed] [Google Scholar]
- 18.Mussi-Pinhata M, Gonçalves AL. Serum immunoglobulin levels and incidence of infection during the first year of life in full-term and preterm infants. J Trop Pediatr. 1989;35:147–53. doi: 10.1093/tropej/35.4.147. [DOI] [PubMed] [Google Scholar]
- 19.Härtel C, Adam N, Strunk T, Temming P, Müller-Steinhardt M, Schultz C. Cytokine responses correlate differentially with age in infancy and early childhood. Clin Exp Immunol. 2005;142:446–53. doi: 10.1111/j.1365-2249.2005.02928.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Maródi L. Innate cellular immune responses in newborns. Clin Immunol. 2006;118:137–44. doi: 10.1016/j.clim.2005.10.012. [DOI] [PubMed] [Google Scholar]
- 21.Melville JM, Moss TJ. The immune consequences of preterm birth. Front Neurosci. 2013;7:79. doi: 10.3389/fnins.2013.00079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Currie AJ, Curtis S, Strunk T, et al. Preterm infants have deficient monocyte and lymphocyte cytokine responses to group B streptococcus. Infect Immun. 2011;79:1588–96. doi: 10.1128/IAI.00535-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guerrero-Plata A, Casola A, Garofalo RP. Human metapneumovirus induces a profile of lung cytokines distinct from that of respiratory syncytial virus. J Virol. 2005;79:14992–7. doi: 10.1128/JVI.79.23.14992-14997.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guerrero-Plata A, Casola A, Suarez G, et al. Differential response of dendritic cells to human metapneumovirus and respiratory syncytial virus. Am J Respir Cell Mol Biol. 2006;34:320–9. doi: 10.1165/rcmb.2005-0287OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Melendi GA, Laham FR, Monsalvo AC, et al. Cytokine profiles in the respiratory tract during primary infection with human metapneumovirus, respiratory syncytial virus, or influenza virus in infants. Pediatrics. 2007;120:e410–5. doi: 10.1542/peds.2006-3283. [DOI] [PubMed] [Google Scholar]
- 26.Liu J, Guan X, Ma X. Interferon regulatory factor 1 is an essential and direct transcriptional activator for interferon {gamma}-induced RANTES/CCl5 expression in macrophages. J Biol Chem. 2005;280:24347–55. doi: 10.1074/jbc.M500973200. [DOI] [PubMed] [Google Scholar]
- 27.Bao X, Kolli D, Ren J, Liu T, Garofalo RP, Casola A. Human metapneumovirus glycoprotein G disrupts mitochondrial signaling in airway epithelial cells. PLoS One. 2013;8:e62568. doi: 10.1371/journal.pone.0062568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ren J, Liu G, Go J, Kolli D, Zhang G, Bao X. Human metapneumovirus M2-2 protein inhibits innate immune response in monocyte-derived dendritic cells. PLoS One. 2014;9:e91865. doi: 10.1371/journal.pone.0091865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ren J, Kolli D, Liu T, et al. Human metapneumovirus inhibits IFN-β signaling by downregulating Jak1 and Tyk2 cellular levels. PLoS One. 2011;6:e24496. doi: 10.1371/journal.pone.0024496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mukherjee S, Lindell DM, Berlin AA, et al. IL-17-induced pulmonary pathogenesis during respiratory viral infection and exacerbation of allergic disease. Am J Pathol. 2011;179:248–58. doi: 10.1016/j.ajpath.2011.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Perez GF, Pancham K, Huseni S, et al. Rhinovirus infection in young children is associated with elevated airway TSLP levels. Eur Respir J. 2014;44:1075–8. doi: 10.1183/09031936.00049214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Spann KM, Baturcam E, Schagen J, et al. Viral and host factors determine innate immune responses in airway epithelial cells from children with wheeze and atopy. Thorax. 2014;69:918–25. doi: 10.1136/thoraxjnl-2013-204908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.World Health Organization. 2013 http://www.who.int/mediacentre/fact-sheets/fs363/en/
- 34.Blencowe H, Cousens S, Oestergaard MZ, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet. 2012;379:2162–72. doi: 10.1016/S0140-6736(12)60820-4. [DOI] [PubMed] [Google Scholar]


